Galvanic Leaching of Seafloor Massive Sulphides Using MnO2 in H2SO4-NaCl Media
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results and Discussion
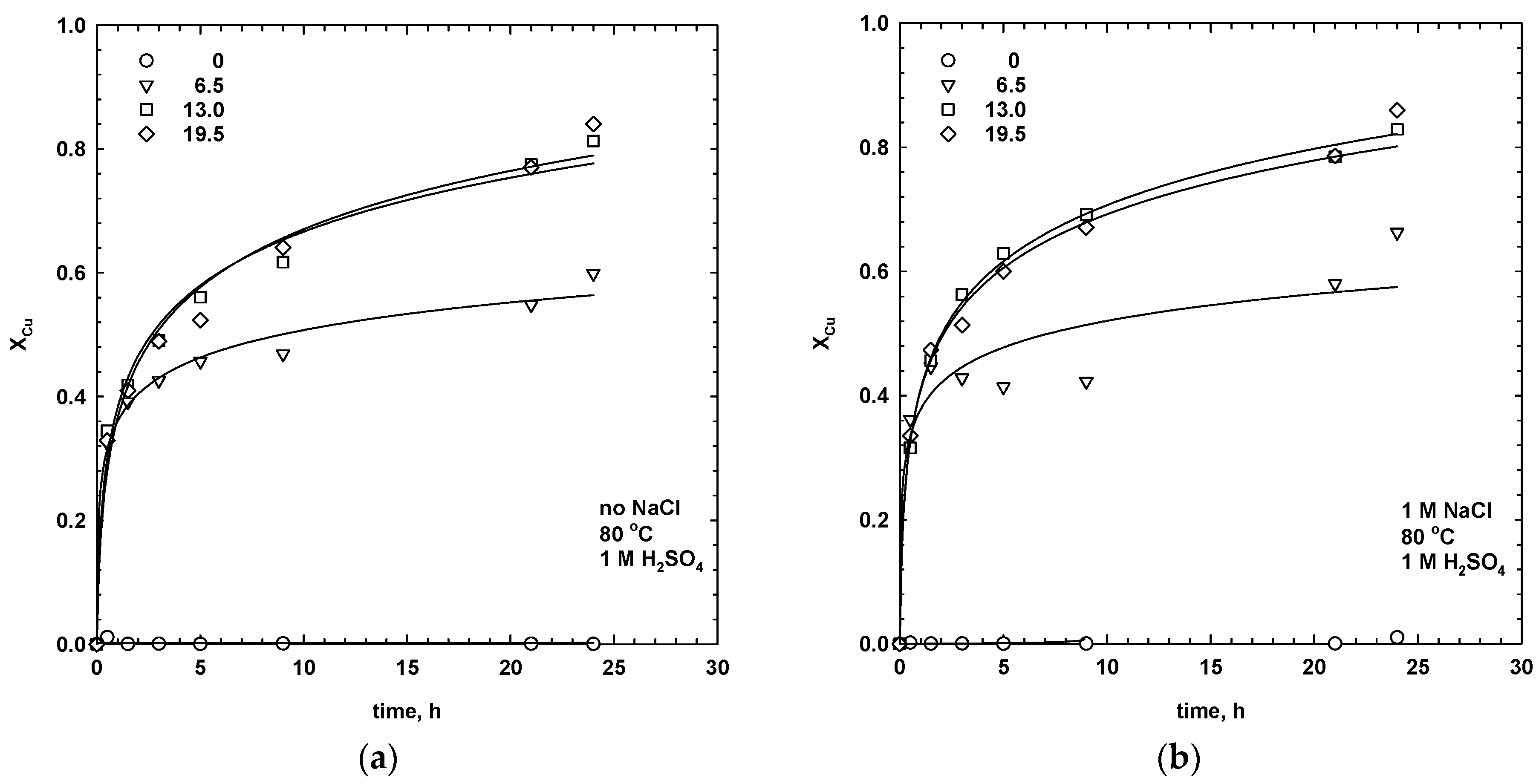
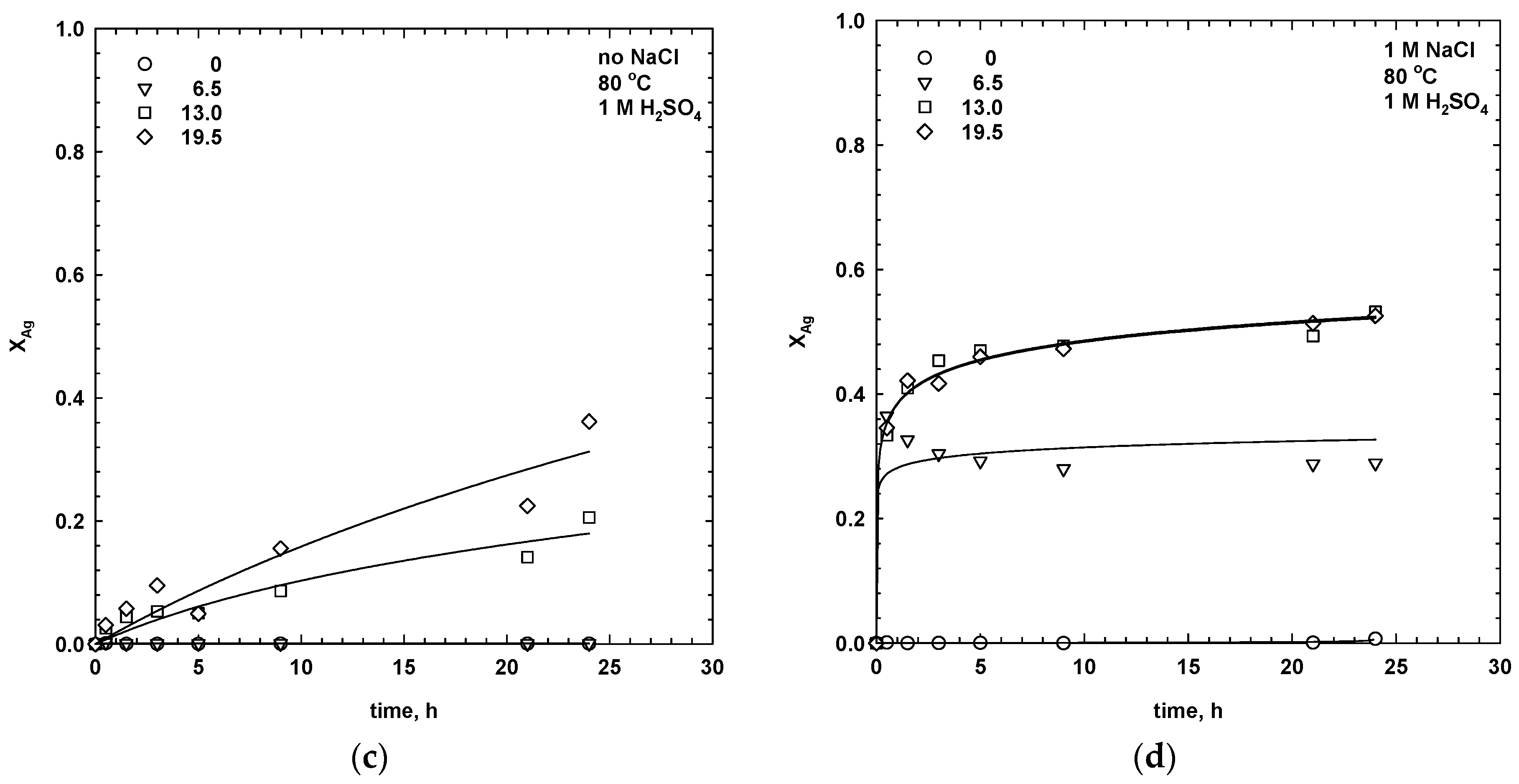
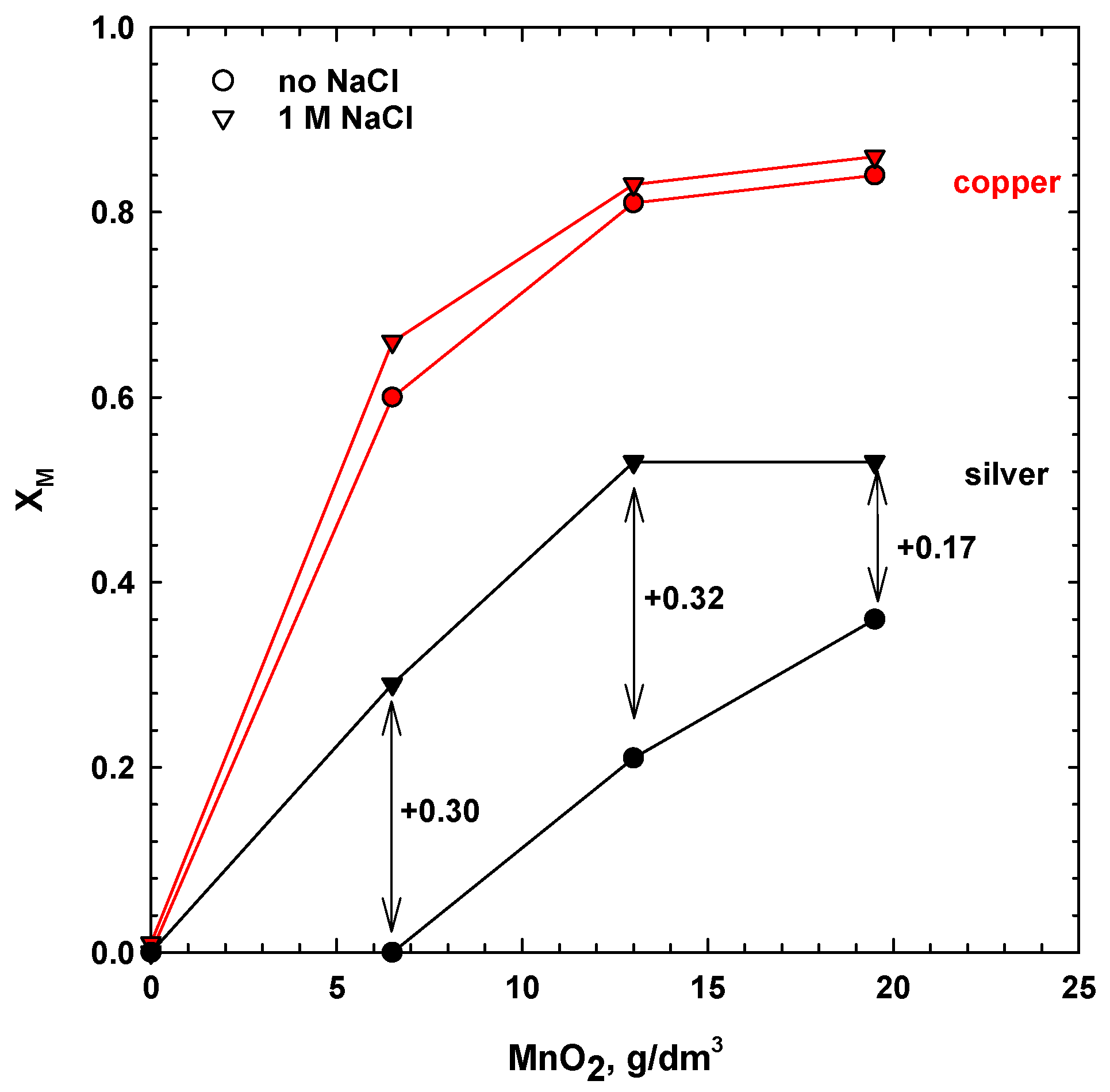
3.1. Effect of Manganese Dioxide and Sodium Chloride Addition
- (i)
- Primary galvanic interactions between the chalcopyrite, isocubanite, and manganese oxide, or
- (ii)
- Synergistic effects of both the ferric chloride and cupric chloride leaching.
- (i)
- For sulphate media leaching without NaCl
- (ii)
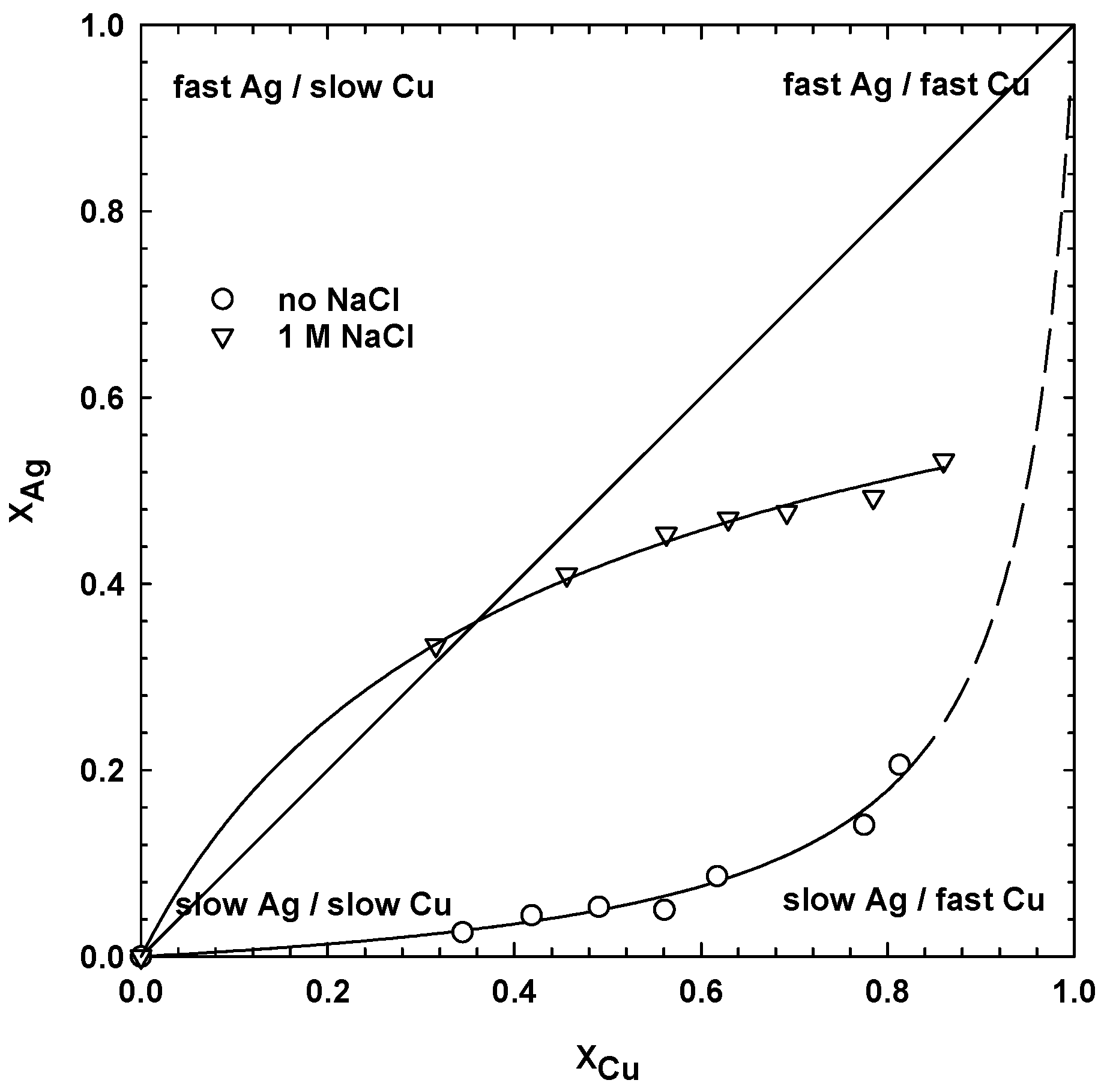
- For sulphate media leaching with 1 M NaClwhere a1 and a2 are the empirical constants for leaching in the absence and presence of chloride ions, respectively. The constants, a1 and a2, were calculated using the Sigma Plot software package. The constants assume values at different ranges, but both start from 0 for the dissolution of copper only, and increases when the dissolution of silver also increases. Thus, the higher the value of a1 and a2, the faster the dissolution of silver, and the slower the extraction rate of the copper. For simultaneous leaching of the copper and silver phases, the constant, a1, is equal to 1. The calculated values of the empirical constants, a1 and a2, for leaching tests, with different dosages of MnO2, are collected in Table 1. For leaching in the absence of NaCl, the constant, a1, was equal to zero in water and 6.5 mg/dm3 of MnO2, indicating that only copper was extracted. The values of a1 and a2 increased with the manganese dioxide dosage, which resulted in a higher extraction of silver.
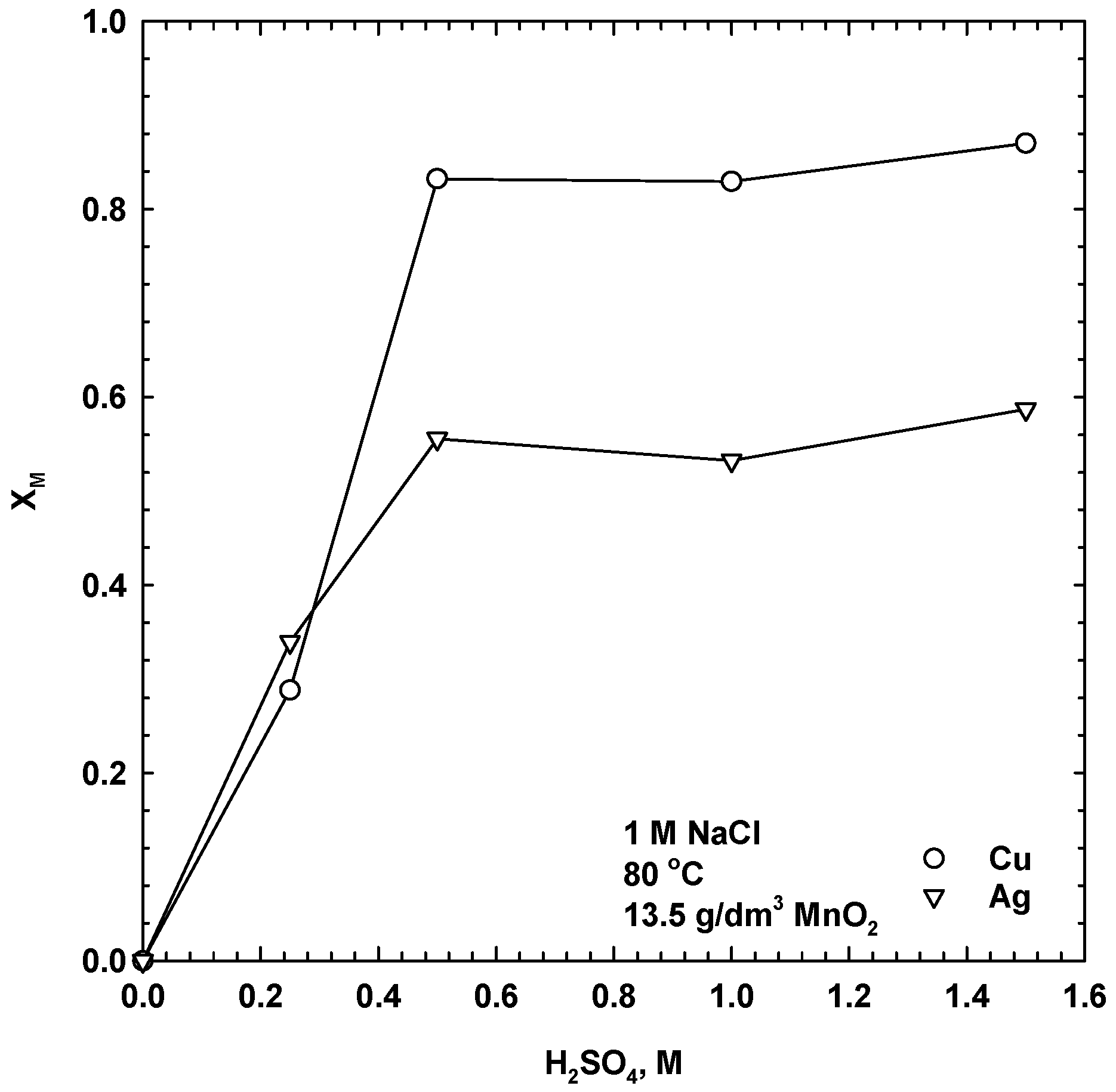
3.2. Effect of Acid Concentration
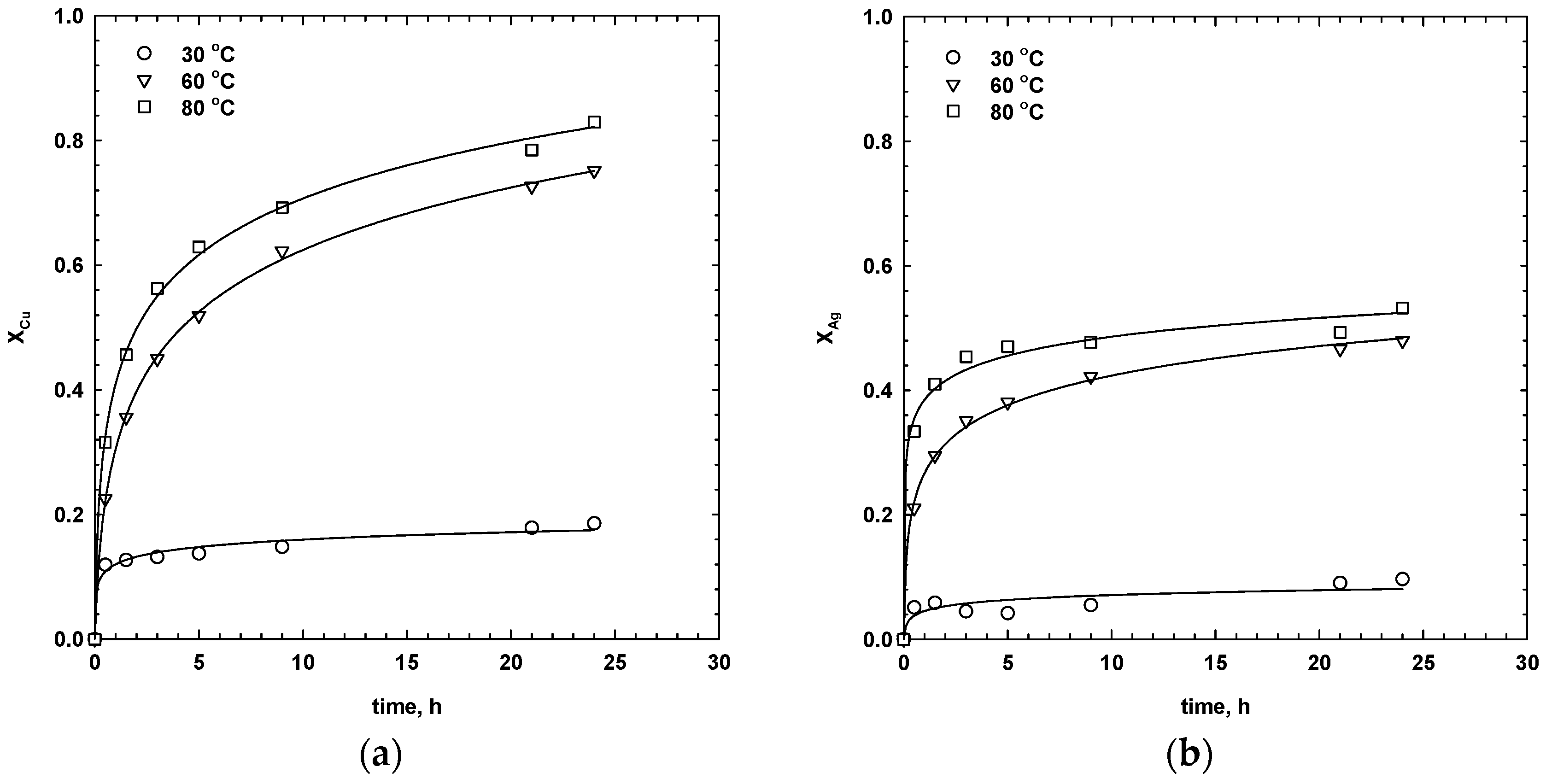
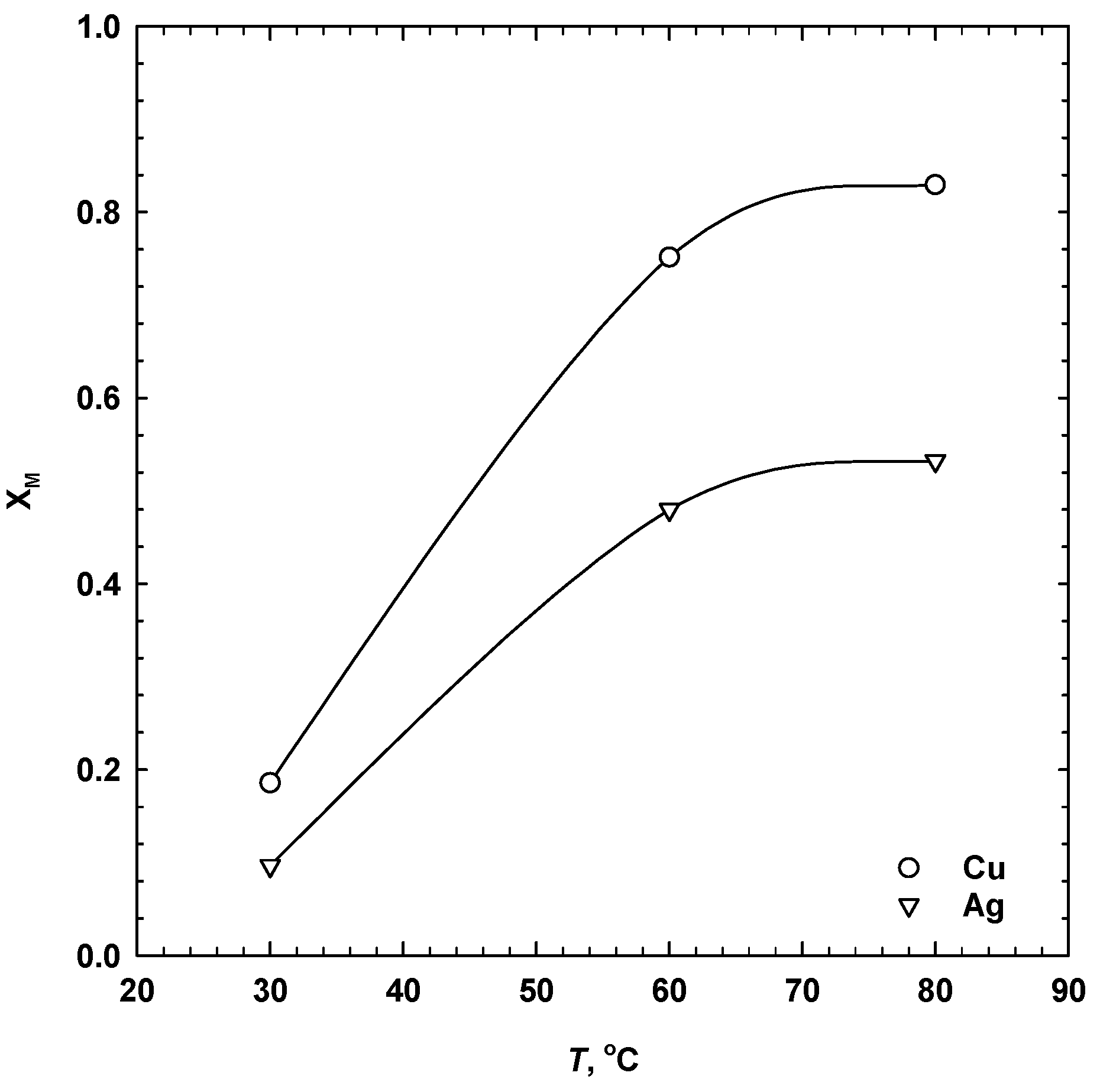
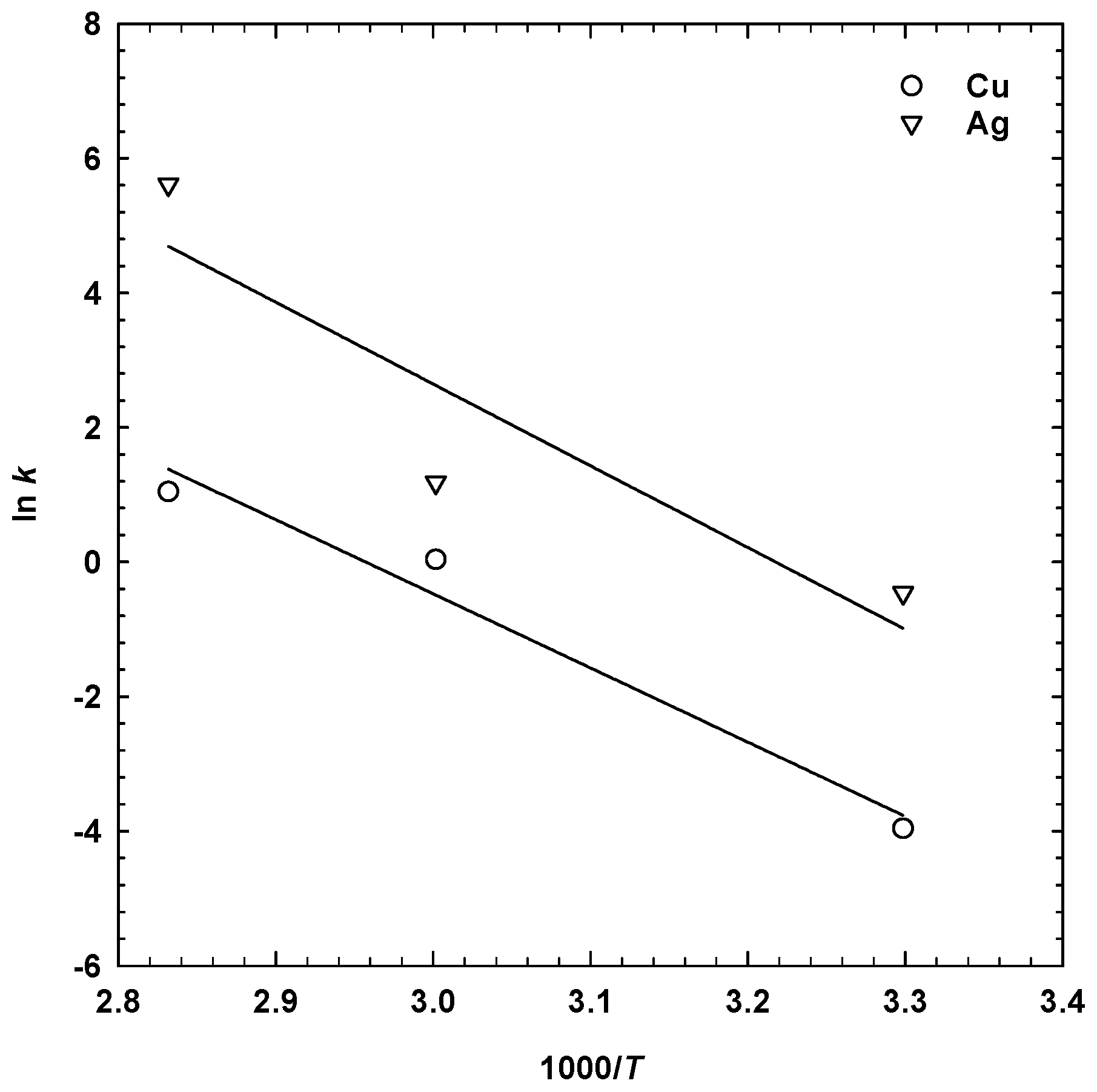
3.3. Effect of Temperature
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kowalczuk, P.B.; Snook, B.; Kleiv, R.A.; Aasly, K. Efficient extraction of copper and zinc from seafloor massive sulphide rock samples from the Loki’s Castle area at the Arctic Mid-Ocean Ridge. Miner. Eng. 2018, 115, 106–116. [Google Scholar] [CrossRef]
- Devi, N.B.; Madhuchhanda, M.; Rao, K.S.; Rath, P.C.; Paramguru, R.K. Oxidation of chalcopyrite in the presence of manganese dioxide in hydrochloric acid medium. Hydrometallurgy 2000, 57, 57–76. [Google Scholar] [CrossRef]
- Gantayat, B.P.; Rath, P.C.; Paramguru, R.K.; Rao, S.B. Galvanic interaction between chalcopyrite and manganese dioxide in sulphuric acid medium. Metall. Mater. Trans. B 2000, 31B, 55–61. [Google Scholar] [CrossRef]
- Tian, Q.; Jiao, C.; Guo, X. Extraction of valuable metals from manganese-silver ore. Hydrometallurgy 2012, 119–120, 8–15. [Google Scholar] [CrossRef]
- Dutrizac, J.E.; MacDonald, R.J.C. The effect of sodium chloride on the dissolution of chalcopyrite under simulated dump leaching conditions. Metall. Trans. 1971, 2, 2310–2312. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Jeffrey, M.I.; Lawson, F. The effect of chloride ions on the dissolution of chalcopyrite in acidic solutions. Hydrometallurgy 2000, 56, 189–202. [Google Scholar] [CrossRef]
- Hait, J.; Jana, R.K.; Kumar, V.; Sanyal, S.K. Some studies on sulphuric acid leaching of anode slime with additives. Ind. Eng. Chem. Res. 2002, 41, 6593–6599. [Google Scholar] [CrossRef]
- Carneiro, M.F.C.; Leao, V.A. The role of sodium chloride on surface properties of chalcopyrite leached with ferric sulfsulphate. Hydrometallurgy 2007, 87, 73–82. [Google Scholar] [CrossRef]
- Rona, P.A. Resources of the Sea Floor. Science 2003, 299, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Das, R.P.; Anand, S.; Das, S.C.; Jena, P.K. Leaching of manganese nodules in ammoniacal medium using glucose as reductant. Hydrometallurgy 1986, 16, 335–344. [Google Scholar] [CrossRef]
- Kanungo, S.B.; Jena, P.K. Reduction leaching of manganese nodules of Indian Ocean origin in dilute hydrochloric acid. Hydrometallurgy 1988, 21, 41–58. [Google Scholar] [CrossRef]
- Senanayake, G. Acid leaching of metals from deep-sea manganese nodules—A critical review of fundamentals and applications. Miner. Eng. 2011, 24, 1379–1396. [Google Scholar] [CrossRef]
- Pedersen, R.B.; Rapp, H.T.; Thorseth, I.H.; Lilley, M.D.; Barriga, F.J.A.S.; Baumberger, T.; Flesland, K.; Fonseca, R.; Fruh-Green, G.L.; Jorgensen, S.L. Discovery of a black smoker vent field and vent fauna at the Arctic Mid-Ocean Ridge. Nat. Commun. 2010, 1, 126. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsen, M.; Aasly, K.; Ellefemo, S.; Hilario, A.; Ramirez-Llodra, E.; Søreide, F.; Falcon-Suarez, I.; Juliani, C.; Kieswetter, A.; Lim, A.; et al. NTNU Cruise Reports 2016 No 1 MarMine Arctic Mid Ocean Ridge 15.08.2016–05.09.2016; NTNU: Trondheim, Norway, 2016; ISSN 2535-2520. [Google Scholar]
- Barton, P.B., Jr.; Bethke, P.M. Chalcopyrite disease in sphalerite: Pathology and epidemiology. Am. Miner. 1987, 72, 451–467. [Google Scholar]
- Nakazawa, H.; Hareyama, W. Galvanic leaching of chalcopyrite using manganese oxides in spent zinc-carbon batteries. Resour. Process. 2016, 63, 3–11. [Google Scholar] [CrossRef]
- Natarajan, K.A.; Iwasaki, I. Role of galvanic interactions in the bioleaching of Duluth gabbro copper-nickel sulphides. Sep. Sci. Technol. 1983, 18, 1095–1111. [Google Scholar] [CrossRef]
- Dutrizac, J.E.; MacDonald, R.J.C. Ferric ion as a leaching medium. Miner. Sci. Eng. 1974, 6, 59–100. [Google Scholar]
- Dutrizac, J.E. The dissolution of chalcopyrite in ferric sulfate and ferric chloride media. Metall. Mater. Trans. B 1981, 12, 371–378. [Google Scholar] [CrossRef]
- Rath, P.C.; Paramguru, R.K.; Jena, P.K. Kinetics of dissolution of sulphide minerals in ferric chloride solution. I. Dissolution of galena, sphalerite and chalcopyrite. Trans. Inst. Min. Metall. 1988, 97, C150–C158. [Google Scholar]
- Winand, R. Chloride hydrometallurgy. Hydrometallurgy 1991, 27, 285–316. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93, 81–87. [Google Scholar] [CrossRef]
- Harmer, S.L.; Thomas, J.E.; Fornasiero, D.; Gerson, A.R. The evolution of surface layers formed during chalcopyrite leaching. Geochim. Cosmochim. Acta 2006, 70, 4392–4402. [Google Scholar] [CrossRef]
- Xian, Y.J.; Wen, S.M.; Deng, J.S.; Liu, J.; Nie, Q. Leaching chalcopyrite with sodium chlorate in hydrochloric acid solution. Can. Metall. Q. 2012, 51, 133–140. [Google Scholar] [CrossRef]
- Ghahremaninezhad, A.; Dixon, D.G.; Asselin, E. Electrochemical and XPS analysis of chalcopyrite (CuFeS2) dissolution in sulfsulphuric acid solution. Electrochim. Acta 2013, 87, 97–112. [Google Scholar] [CrossRef]
- Yang, Y.; Harmer, S.; Chen, M. Synchrotron-based XPS and NEXAFS study of surface chemical species during electrochemical oxidation of chalcopyrite. Hydrometallurgy 2015, 156, 89–98. [Google Scholar] [CrossRef]
- Jiang, T.; Yang, Y.; Huang, Z.; Qiu, G. Simultanous leaching of manganese and silver from manganese-silver ores at room temperature. Hydrometallurgy 2003, 69, 177–186. [Google Scholar] [CrossRef]
- Bahram, B.; Javad, M. Chloride leaching of lead and silver from refractory zinc plant residue. Res. J. Chem. Environ. 2011, 15, 473–480. [Google Scholar]
- Chmielewski, T.; Gibas, K.; Borowski, K.; Adamski, Z.; Wozniak, B.; Muszer, A. Chloride leaching of silver and lead from a solid residue after atmospheric leaching of flotation copper concentrates. Physicochem. Probl. Miner. Process. 2017, 53, 893–907. [Google Scholar] [CrossRef]
- Liu, W.; Yang, T.; Xia, X. Behaviour of silver and lead in selective chlorination leaching process of gold-antimony alloy. Trans. Nonferr. Met. Soc. China 2010, 20, 322–329. [Google Scholar] [CrossRef]
- De Vreese, P.; Brooks, N.R.; Hecke, K.V.; Meervelt, L.V.; Matthijs, E.; Binnemans, K.; van Deun, R. Speciation of copper(II) complexes in an ionic liquid based on choline chloride and in choline chloride/water mixtures. Inorg. Chem. 2012, 51, 4972–4981. [Google Scholar] [CrossRef] [PubMed]
- Parker, G.K.; Woods, R.; Hope, G.A. Raman investigation of chalcopyrite oxidation. Coll. Surf. A Physicochem. Eng. Asp. 2008, 318, 160–168. [Google Scholar] [CrossRef]
- O’Malley, M.L.; Liddell, L.C. Leaching of CuFeS2 by aqueous FeCl3, HCl, and NaCl: effect of solution composition and limited oxidant. Metall. Trans. B-Process Metall. 1987, 18, 505–510. [Google Scholar] [CrossRef]
- Habashi, F. Chalcopyrite Its Chemistry and Metallurgy; McGraw-Hill: New York, NY, USA, 1978. [Google Scholar]
- Lundstrom, M.; Aromaa, J.; Forsen, O.; Hyvarinen, O.; Barker, M. Leaching of chalcopyrite in cupric chloride. Hydrometallurgy 2005, 77, 89–95. [Google Scholar] [CrossRef]
- Berger, J.M.; Winand, R. Solubilities, densities, and electrical conductivities of aqueous copper (I) and copper (II) chlorides in solutions containing other chlorides such as zinc, sodium, and hydrogen chlorides. Hydrometallurgy 1984, 12, 61–81. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.; Kingman, S.; Al-Harahsheh, A. Ferric chloride leaching of chalcopyrite: synergistic effect of CuCl2. Hydrometallurgy 2008, 91, 89–97. [Google Scholar] [CrossRef]
- Drzymala, J. Mineral Processing. Foundation of Theory and Practice of Minerallurgy; Oficyna Wydawnicza Politechniki Wroclawskiej: Wroclaw, Poland, 2007; ISBN 978-83-7493-362-9. [Google Scholar]
- McLintock, I.S. The Elovich equation in chemisorption kinetics. Nature 1967, 216, 1204–1205. [Google Scholar] [CrossRef]
- Ding, Z.; Yin, Z.; Hu, H.; Chen, Q. Dissolution kinetics of zinc silicate (hemimorphite) in ammoniacal solution. Hydrometallurgy 2010, 104, 201–206. [Google Scholar] [CrossRef]
- Zhao, D.; Yang, S.; Chen, Y.; Tang, C.; He, J.; Li, H. Leaching kinetics of hemimorphite in ammonium chloride solution. Metals 2017, 7, 237. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Montes, K.S.; Padilla, R. Chalcopyrite leaching in sulfate–chloride media at ambient pressure. Hydrometallurgy 2011, 109, 37–42. [Google Scholar] [CrossRef]


| MnO2, g/dm3 | No NaCl | 1 M NaCl | ||||||
|---|---|---|---|---|---|---|---|---|
| a1 | R2 | XCu | XAg | a2 | R2 | XCu | XAg | |
| 0 | 0 | - | 0.00 | 0.00 | 1.33 | 0.990 | 0.01 | 0.00 |
| 6.5 | 0 | - | 0.60 | 0.00 | 2.28 | 0.750 | 0.66 | 0.29 |
| 13.0 | 0.054 | 0.975 | 0.81 | 0.21 | 4.01 | 0.997 | 0.82 | 0.53 |
| 19.5 | 0.096 | 0.965 | 0.84 | 0.36 | 3.97 | 0.999 | 0.86 | 0.53 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kowalczuk, P.B.; Manaig, D.O.; Drivenes, K.; Snook, B.; Aasly, K.; Kleiv, R.A. Galvanic Leaching of Seafloor Massive Sulphides Using MnO2 in H2SO4-NaCl Media. Minerals 2018, 8, 235. https://doi.org/10.3390/min8060235
Kowalczuk PB, Manaig DO, Drivenes K, Snook B, Aasly K, Kleiv RA. Galvanic Leaching of Seafloor Massive Sulphides Using MnO2 in H2SO4-NaCl Media. Minerals. 2018; 8(6):235. https://doi.org/10.3390/min8060235
Chicago/Turabian StyleKowalczuk, Przemyslaw B., Dan Oliric Manaig, Kristian Drivenes, Ben Snook, Kurt Aasly, and Rolf Arne Kleiv. 2018. "Galvanic Leaching of Seafloor Massive Sulphides Using MnO2 in H2SO4-NaCl Media" Minerals 8, no. 6: 235. https://doi.org/10.3390/min8060235
APA StyleKowalczuk, P. B., Manaig, D. O., Drivenes, K., Snook, B., Aasly, K., & Kleiv, R. A. (2018). Galvanic Leaching of Seafloor Massive Sulphides Using MnO2 in H2SO4-NaCl Media. Minerals, 8(6), 235. https://doi.org/10.3390/min8060235






