The Role of Clay Swelling and Mineral Neoformation in the Stabilization of High Plasticity Soils Treated with the Fly Ash- and Metakaolin-Based Geopolymers
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil
2.2. Geopolymer Synthesis
2.3. Soil Preparation
2.4. Cation Exchange Capacity (CEC)
2.5. X-ray Powder Diffraction (XRD)
3. Results and Discussion
3.1. CEC Geochemistry
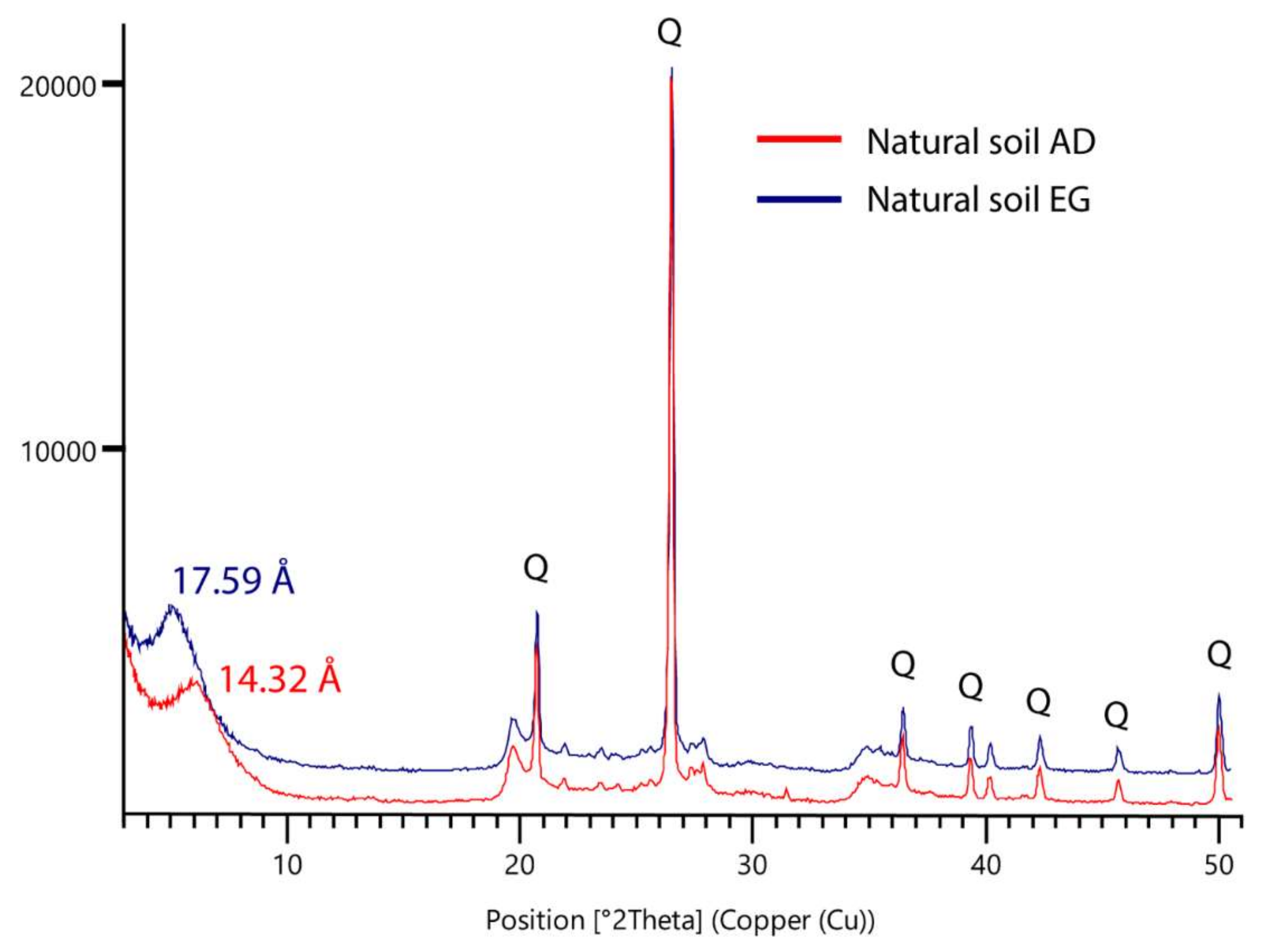
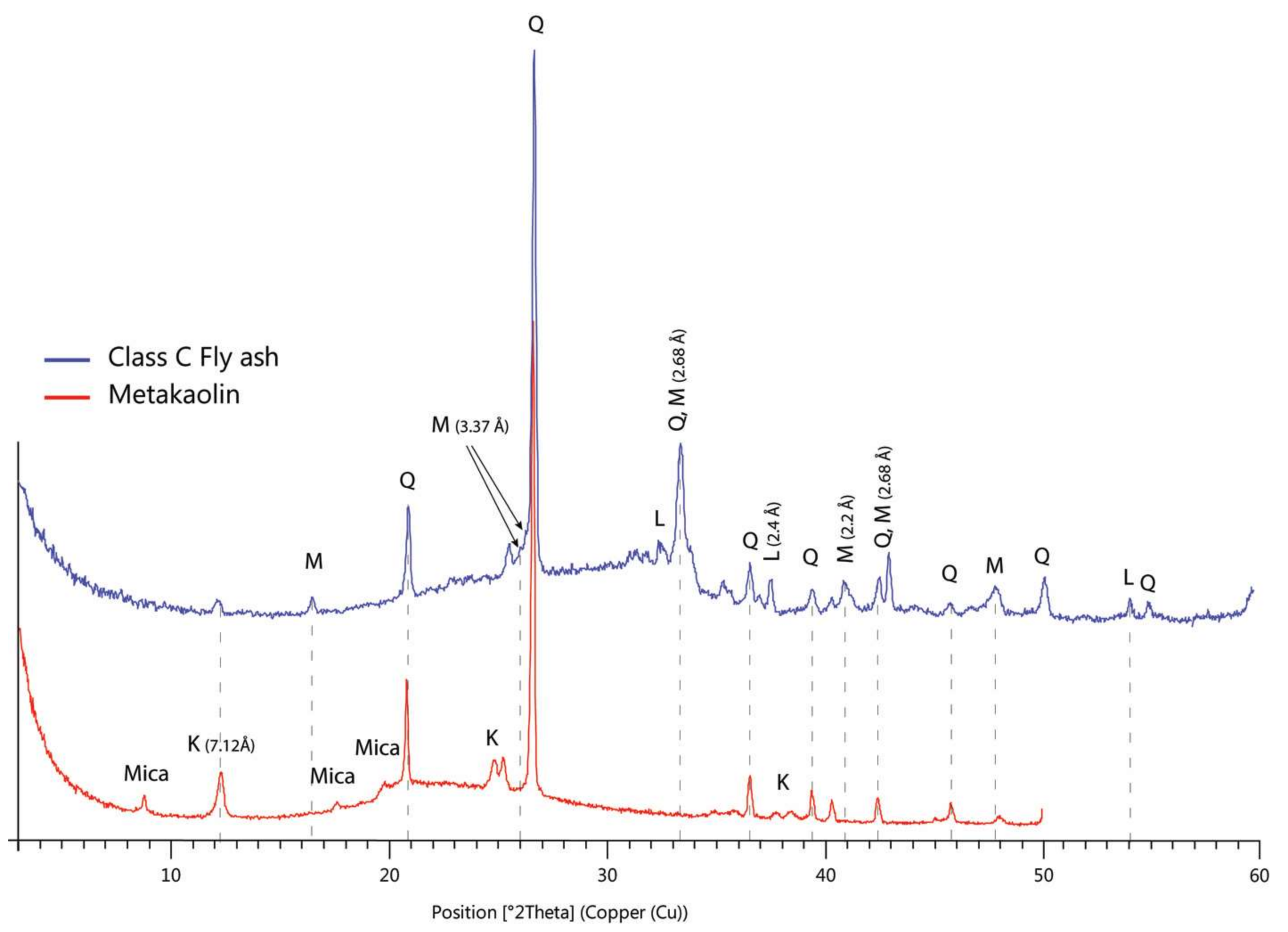
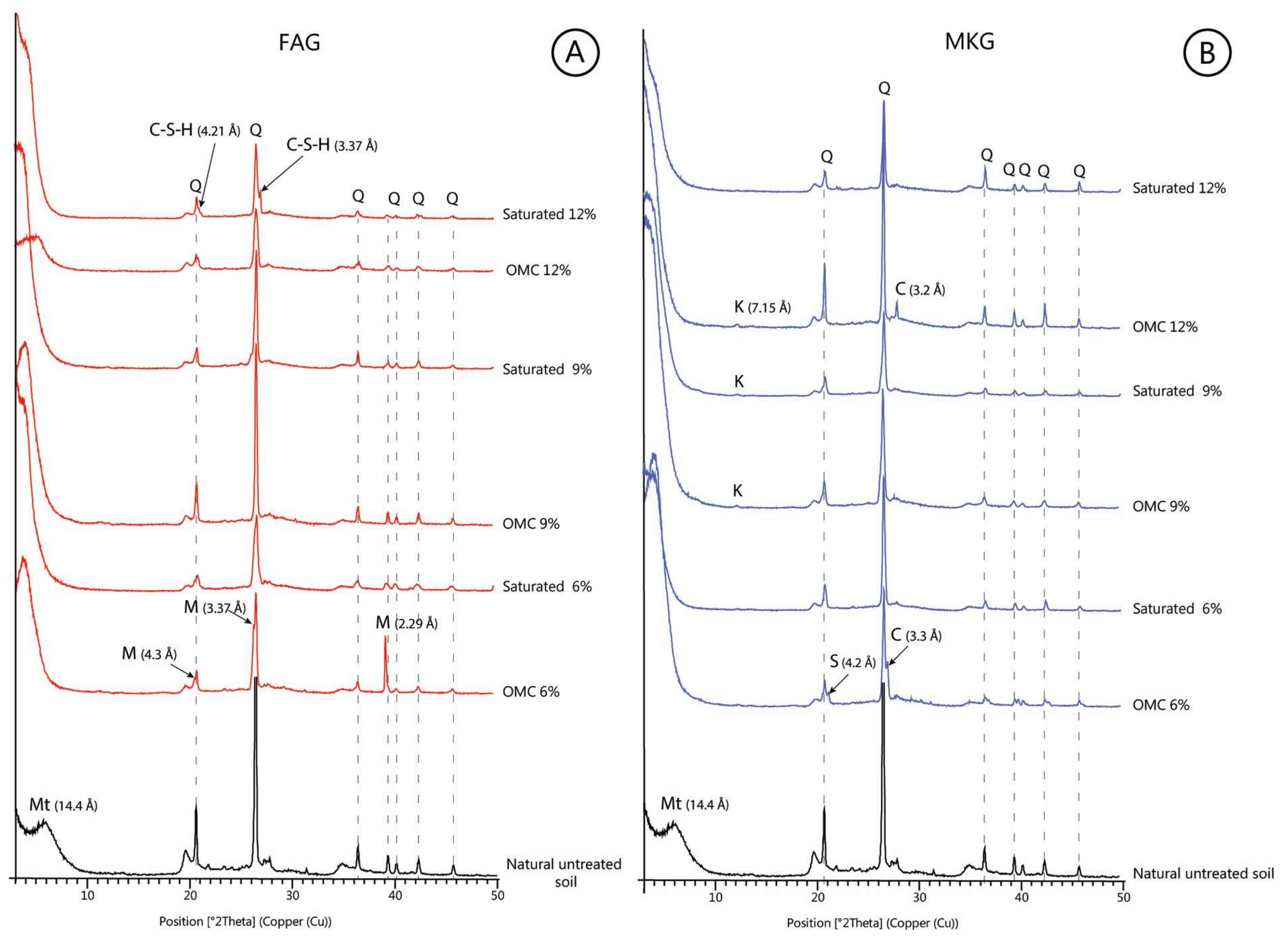
3.2. Mineralogy of Natural and Treated Soils
4. Conclusions
- 1-
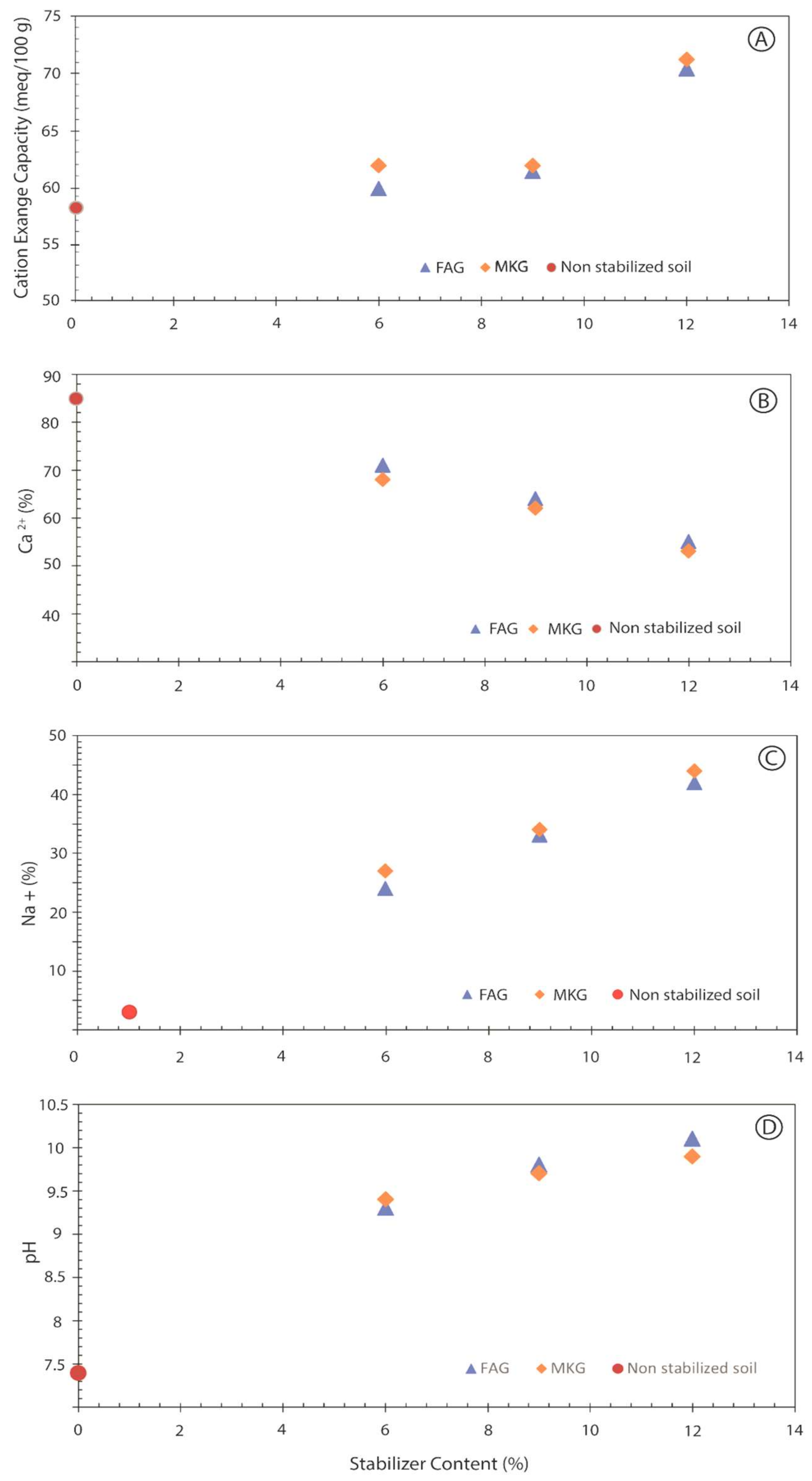
- The pH values increased for all stabilized soils upon increasing the stabilizer content, which is attributed to the alkali content of the additives.
- 2-
- Geopolymer-stabilized soils exhibited a significant reduction in Ca2+ content in the base saturation, which is correlated to increased Na+ availability as a result of stabilizer addition. Increased Na+ concentrations might have facilitated clay particle dispersion and swelling of stabilized soil.
- 3-
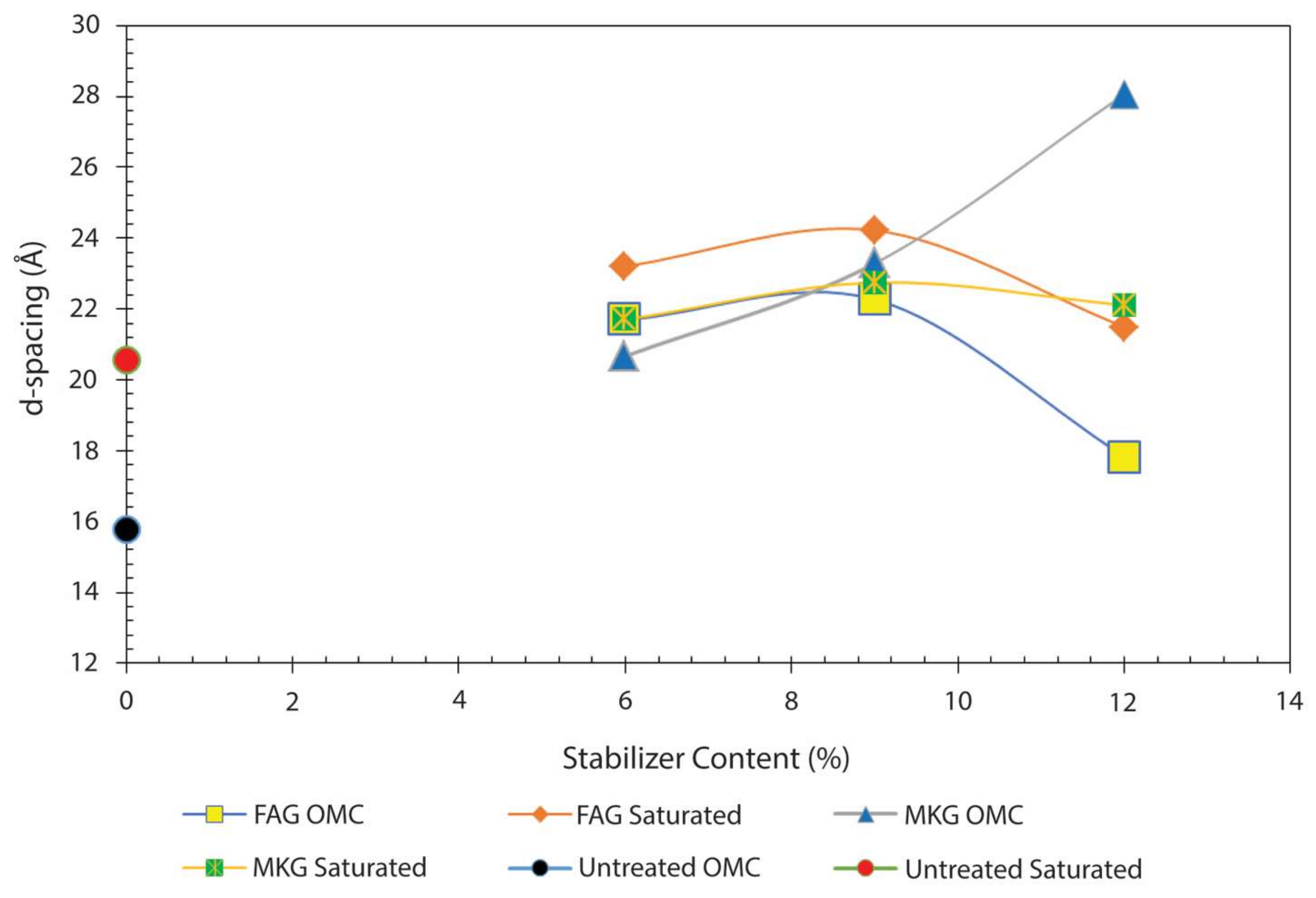
- Soils stabilized with MKG showed slightly higher values of CEC, pH, Na+, and lower Ca2+ compared to the samples stabilized with FAG, thus contributing to higher swelling of the soil treated with MKG.
- 4-
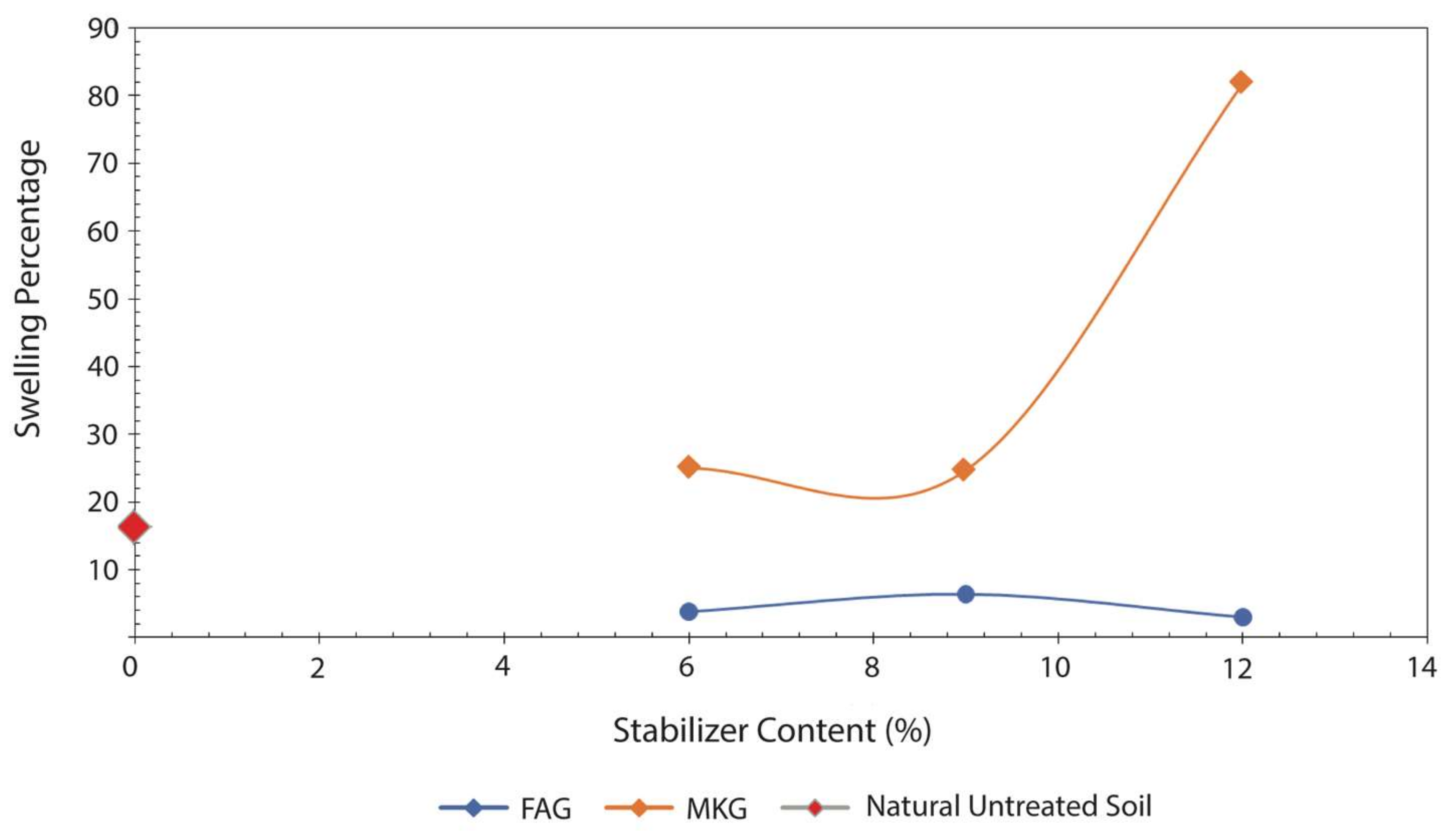
- Soils stabilized with the 12% FAG exhibited the lowest degree of swelling due to the reduced expansion of the soil smectite and the formation of cementitious calcium silicate hydrate (C–S–H). The former is explained by an increase in the availability of Ca2+ accommodated at the exchangeable sites of montmorillonite. The newly formed C–S–H gel strengthened the soil structure by occupying the soil pore spaces and mechanically bound the clay particles, thus precluding any further swelling of the soil smectite.
- 5-
- An abnormal swelling of soil samples stabilized with the 6% and 12% MKG is attributed to an instantaneous swelling of montmorillonite and subsequent formation of feldspathoids (sodalite and cancrinite) as suggested by the interpretation of the XRD patterns.
- 6-
- The study proved that the CEC test in combination with XRD mineralogy could be used as a cost-effective and quick tool to explain the swelling behavior of soils stabilized with chemical additives such as MKG and FAG.
- 7-
- Further analyses are needed, especially electron microbeam investigation, to corroborate the conclusions based on the XRD and basic geochemical inquiry.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hamzah, H.N.; Al Bakri Abdullah, M.M.; Cheng Yong, H.; Arif Zainol, M.R.R.; Hussin, K. Review of Soil Stabilization Techniques: Geopolymerization Method one of the New Technique. Key Eng. Mater. 2015, 660, 298–304. [Google Scholar] [CrossRef]
- Giannopoulou, I.; Dimas, D.; Maragos, I.; Panias, D. Utilization of solid by-products for the development of inorganic polymeric construction materials. Glob. Nest J. 2009, 11, 127–136. [Google Scholar]
- Zhang, Y.; Zhang, W.; Sun, W.; Li, Z.; Liu, Z. Preparation of metakaolin based geopolymer and its three-dimensional pore structural characterization. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2015, 30, 550–555. [Google Scholar] [CrossRef]
- Abdullah, M.M.A.B.; Hussin, K.; Bnhussain, M.; Ismail, K.N.; Yahya, Z.; Abdul Razak, R. Fly Ash-based Geopolymer Lightweight Concrete Using Foaming Agent. Int. J. Mol. Sci. 2012, 13, 7186–7198. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, F.U.A.; Vimonsatit, V. Compressive strength of fly-ash-based geopolymer concrete at elevated temperatures. Fire Mater. 2015, 39, 174–188. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Ngo, T.; Mendis, P.; Nguyen, T.; Kashani, A.; van Deventer, J.S.J. Pore characteristics in one-part mix geopolymers foamed by H2O2: The impact of mix design. Mater. Des. 2017, 130, 381–391. [Google Scholar] [CrossRef]
- Duxson, P.; Provis, J.L.; Lukey, G.C.; van Deventer, J.S.J. The role of inorganic polymer technology in the development of ‘green concrete’. Cem. Concr. Res. 2007, 37, 1590–1597. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; van Deventer, J.S.J. Solid Reactant-Based Geopolymers from Rice Hull Ash and Sodium Aluminate. Waste Biomass Valor 2017, 8, 2131–2140. [Google Scholar] [CrossRef]
- Zhang, M.; Guo, H.; El-Korchi, T.; Zhang, G.; Tao, M. Experimental feasibility study of geopolymer as the next-generation soil stabilizer. Constr. Build. Mater. 2013, 47, 1468–1478. [Google Scholar] [CrossRef]
- Sindhunata; van Deventer, J.S.J.; Lukey, G.C.; Xu, H. Effect of Curing Temperature and Silicate Concentration on Fly-Ash-Based Geopolymerization. Ind. Eng. Chem. Res. 2006, 45, 3559–3568. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymers: Inorganic polymeric new materials. J. Therm. Anal. 1991, 37, 1633–1656. [Google Scholar] [CrossRef]
- Rangan, B.V. Geopolymer concrete for environmental protection. Indian Concr. J. 2014, 88, 41–59. [Google Scholar]
- Barbosa, V.; Mackenzie, K.; Thaumaturgo, C. Synthesis and Characterisation of Materials Based on Inorganic Polymers of Alumina and Silica: Sodium Polysialate Polymers. Int. J. Inorg. Mater. 2000, 2, 309–317. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymer Chemistry and Properties. In Proceedings of the 1st International Conference on Geopolymer ’88, Compiegne, France, 1–3 June 1988; Volume 1, pp. 25–48. [Google Scholar]
- Rios, S.; Ramos, C.; da Fonseca, A.V.; Cruz, N.; Rodrigues, C. Colombian Soil Stabilized with Geopolymers for Low Cost Roads. Procedia Eng. 2016, 143, 1392–1400. [Google Scholar] [CrossRef]
- Locat, J.; Bérubé, M.-A.; Choquette, M. Laboratory investigations on the lime stabilization of sensitive clays: Shear strength development. Can. Geotech. J. 1990, 27, 294–304. [Google Scholar] [CrossRef]
- Yeskis, D.; van Groos, A.F.K.; Guggenheim, S. The dehydroxylation of kaolinite. Am. Mineral. 1985, 70, 159–164. [Google Scholar]
- Alehyen, S.; Achouri, M.E.L.; Taibi, M. Characterization, microstructure and properties of fly ash-based geopolymer. J. Mater. Environ. Sci. 2017, 8, 1783–1796. Available online: http://www.jmaterenvironsci.com/Document/vol8/vol8_N5/190-JMES-2930-Alehyen.pdf (accessed on 20 February 2018).
- Millot, G. Geology of Clays: Weathering Sedimentology Geochemistry; Springer Science & Business Media: Berlin, Germany, 2013; ISBN 978-3-662-41609-9. [Google Scholar]
- Khadka, S.D.; Jayawickrama, P.W.; Senadheera, S. Shrink/Swell Behavior of High Plastic Clay Treated with Geopolymer. Transp. Res. Rec. J. Transp. Res. Board 2018. accepted for publication. [Google Scholar]
- American Society for Testing and Material (ASTM International). Standard Test Methods for Liquid Limit, Plastic Limit, and Plasticity Index of Soils; D4318-00; ASTM International: West Conshohocken, PA, USA, 2010. [Google Scholar]
- American Society for Testing and Material (ASTM International). Classification of Soils for Engineering Purposes: Annual Book of ASTM Standards; D 2487-83, 04.08; ASTM International: West Conshohocken, PA, USA, 1985; pp. 395–408. [Google Scholar]
- Haby, V.A.; Russelle, M.P.; Skoley, E.O. Testing Soils for Potassium, Calcium, and Magnesium. In Soil Testing and Plant Analysis, 3rd ed.; Westerman, R.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1990; pp. 181–227. [Google Scholar]
- Warncke, D.; Brown, J.R. Potassium and Other Basic Cations. In Recommended Chemical Soil Test Procedures for the North Central Region; North Central Regional Publication No. 221 (revised); Brown, J.R., Ed.; University of Missouri Agricultural Experiment Station: Columbia, MO, USA, 1998; pp. 31–33. [Google Scholar]
- Mc Lean, E.O. Soil pH and Lime Requirement. In Methods of Soil Analysis, Chemical and Microbiological Properties-Agronomy Monograph, 2nd ed.; Page, A.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1982; pp. 199–223, no. 9; Part 2. [Google Scholar]
- Rhoades, J.D. Soluble Salts. In Methods of Soil Analysis, Chemical and Microbiological Properties-Agronomy Monograph, 2nd ed.; Page, A.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1982; pp. 167–179, no. 9; Part 2. [Google Scholar]
- Whitney, D.A. Soil Salinity. In Recommended Chemical Soil Test Procedures for the North Central Region; North Central Regional Publication No. 221 (revised); Brown, J.R., Ed.; University of Missouri Agricultural Experiment Station: Columbia, MO, USA, 1998; pp. 59–60. [Google Scholar]
- Watson, M.E.; Brown, J.R. pH and Lime Requirement. In Recommended Chemical Soil Test Procedures for the North Central Region; North Central Regional Publication No. 221 (revised); Brown, J.R., Ed.; University of Missouri Agricultural Experiment Station: Columbia, MO, USA, 1998; pp. 13–16. [Google Scholar]
- Joint Committee on Powder Diffraction Standards (JCPDS). Mineral Powder Diffraction File; International Center for Diffraction Data: Newtown Square, PA, USA, 1996. [Google Scholar]
- Zanoni, G.; Šegvić, B.; Moscariello, A. Clay mineral diagenesis in Cretaceous clastic reservoirs from West African passive margins (the South Gabon Basin) and its impact on regional geology and basin evolution history. Appl. Clay Sci. 2016, 134, 186–209. [Google Scholar] [CrossRef]
- Al-Rawas, A.A.; Taha, R.; Nelson, J.D.; Al-Shab, B.T.; Al-Siyabi, H. A comparative evaluation of various additives used in the stabilization of expansive soils. Geotech. Test. J. 2002, 25, 199–209. [Google Scholar]
- Oh, J.E.; Monteiro, P.J.M.; Jun, S.S.; Choi, S.; Clark, S.M. The evolution of strength and crystalline phases for alkali-activated ground blast furnace slag and fly ash-based geopolymers. Cem. Concr. Res. 2010, 40, 189–196. [Google Scholar] [CrossRef]
- Moore, D.M.; Reynolds, R.C. X-Ray Diffraction and the Identification and Analysis of Clay Minerals, 2nd ed.; Oxford University Press: Oxford, UK, 1997; ISBN 0195087135. [Google Scholar]
- Viani, A.; Gualtieri, A.F.; Artioli, G. The nature of disorder in montmorillonite by simulation of X-ray powder patterns. Am. Mineral. 2002, 87, 966–975. [Google Scholar] [CrossRef]
- Seppälä, A.; Puhakka, E.; Olin, M. Effect of layer charge on the crystalline swelling of Na+, K+ and Ca2+ montmorillonites: DFT and molecular dynamics studies. Clay Miner. 2016, 51, 197–211. [Google Scholar] [CrossRef]
- Knauss, K.G.; Wolery, T.J. The dissolution kinetics of quartz as a function of pH and time at 70 °C. Geochim. Cosmochim. Acta 1988, 52, 43–53. [Google Scholar] [CrossRef]
- Maraghechi, H.; Rajabipour, F.; Pantano, C.G.; Burgos, W.D. Effect of calcium on dissolution and precipitation reactions of amorphous silica at high alkalinity. Cem. Concr. Res. 2016, 87, 1–13. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Huang, X.; Hein, A.M. Synthesizing mullite from beneficiated fly ash. JOM 1994, 46, 36–39. [Google Scholar] [CrossRef]
- Cristelo, N.; Glendinning, S.; Fernandes, L.; Pinto, A.T. Effect of calcium content on soil stabilisation with alkaline activation. Constr. Build. Mater. 2012, 29, 167–174. [Google Scholar] [CrossRef]
- Deer, W.A.; Howie, R.A.; Zussman, J. Rock-Forming Minerals, Volume 4A: Framework Silicates: Feldspars, 2nd ed.; The Geological Society: London, UK, 2001. [Google Scholar]
- Chavali, R.V.P.; Vindula, S.K.; Ponnapureddy, H.P.R.; Babu, A.; Pillai, R.J. Swelling behavior of kaolinitic clays contaminated with alkali solutions: A micro-level study. Appl. Clay Sci. 2017, 135, 575–582. [Google Scholar] [CrossRef]
- Bender Koch, C. Non-Crystalline Hydrous Feldspathoids in Late Permian Carbonate Rock. Clay Minerals 1991, 26, 527–534. [Google Scholar] [CrossRef]
- Weitkamp, J.; Puppe, L. Catalysis and Zeolites: Fundamentals and Applications; Springer: Berlin/Heidelberg, Germany, 2010; ISBN 978-3-642-08347-1. [Google Scholar]
- Gougazeh, M.; Buhl, J.-C. Synthesis and characterization of zeolite A by hydrothermal transformation of natural Jordanian kaolin. J. Assoc. Arab. Univ. Basic Appl. Sci. 2014, 15, 35–42. [Google Scholar] [CrossRef]
- Zhao, H.; Deng, Y.; Harsh, J.; Flury, M.; Boyle, J. Alteration of Kaolinite to Cancrinite and Sodalite by Simulated Hanford Tank Waste and Its Impact on Cesium Retention. Clays Clay Miner. 2004, 52. [Google Scholar] [CrossRef]
- Lapides, I.; Heller-Kallai, L. Reactions of metakaolinite with NaOH and colloidal silica—Comparison of different samples (part 2). Appl. Clay Sci. 2007, 35, 94–98. [Google Scholar] [CrossRef]
- Madani, A.; Aznar, A.; Sanz, J.; Serratosa, J.M. 29Si and 27Al NMR study of zeolite formation from alkali-leached kaolinites. Influence of thermal preactivation. J. Phys. Chem. 1990, 94, 760–765. [Google Scholar] [CrossRef]
- Gualtieri, A.; Norby, P.; Artioli, G.; Hanson, J. Kinetics of formation of zeolite Na-A [LTA] from natural kaolinites. Phys. Chem. Min. 1997, 24, 191–199. [Google Scholar] [CrossRef]
- Demortier, A.; Gobeltz, N.; Lelieur, J.P.; Duhayon, C. Infrared evidence for the formation of an intermediate compound during the synthesis of zeolite Na–A from metakaolin. Int. J. Inorg. Mater. 1999, 1, 129–134. [Google Scholar] [CrossRef]
- Sivapullaiah, P. Induced swelling of kaolinitic soil in alkali solution. Soils Found. Tokyo 2007, 47, 59–66. [Google Scholar] [CrossRef][Green Version]
- Aksu, I.; Bazilevskaya, E.; Karpyn, Z.T. Swelling of clay minerals in unconsolidated porous media and its impact on permeability. Geo. Res. J. 2015, 7, 1–13. [Google Scholar] [CrossRef]
| Material Type | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | SO4 |
|---|---|---|---|---|---|---|---|---|
| Metakaolin | 52.00 | 43.00 | <2.20 | <0.20 | <0.10 | <0.05 | <0.40 | <0.05 |
| Class C fly ash | 41.96 | 20.04 | 5.76 | 21.09 | 4.30 | 1.44 | 0.73 | 0.99 |
| Atlanta clay | 64.28 | 16.39 | 9.03 | 3.92 | 1.78 | - | 1.72 | - |
| Geopolymer Type | g/s (%) | Aluminosilicate Source | Alkaline Activators | Curing Temp (°C) | |||
|---|---|---|---|---|---|---|---|
| MK | FAC | NaOH | Na2SiO3 | H2O | |||
| FAG | 6 | 0 | 6.6 | 2.51 | 8.24 | 0.66 | Ambient conditions |
| FAG | 9 | 0 | 9.89 | 3.76 | 12.36 | 0.99 | |
| FAG | 12 | 0 | 13.19 | 5.01 | 16.48 | 1.32 | |
| MKG | 6 | 6.6 | 0 | 2.51 | 8.24 | 0.66 | |
| MKG | 9 | 9.89 | 0 | 3.76 | 12.36 | 0.99 | |
| MKG | 12 | 13.19 | 0 | 5.01 | 16.48 | 1.32 | |
| Stabilization | pH | Soluble Salts 1:1 dS/m | Sum of Cations meq/100 g | % Base Saturation | |||||
|---|---|---|---|---|---|---|---|---|---|
| H | K | Ca | Mg | Na | |||||
| Natural soil (no stabilization) | 7.4 | 1.64 | 58.1 | 0 | 2 | 85 | 10 | 3 | |
| FAG | 6% | 9.3 | 2.92 | 65.9 | 0 | 1 | 71 | 4 | 24 |
| 9% | 9.8 | 3.56 | 66.8 | 0 | 1 | 64 | 2 | 33 | |
| 12% | 10.1 | 4.42 | 72.2 | 0 | 1 | 55 | 2 | 42 | |
| MKG | 6% | 9.4 | 2.5 | 67.1 | 0 | 1 | 68 | 4 | 27 |
| 9% | 9.7 | 2.84 | 67.1 | 0 | 1 | 62 | 3 | 34 | |
| 12% | 9.9 | 4.64 | 72.7 | 0 | 1 | 53 | 2 | 44 | |
| d-Spacing in Å | Identified Phases | |||||
|---|---|---|---|---|---|---|
| FAC | MK | 6% FAG | 12% FAG | 6% MKG | 12% MKG | |
| 2.2 | - | - | - | - | - | mullite (Al6Si2O13) |
| - | - | 2.29 | - | - | - | mullite (Al6Si2O13) |
| 2.4 | - | - | - | - | - | lime (CaO) |
| 2.68 | - | - | - | - | - | mullite (Al6Si2O13) |
| - | - | - | - | - | 3.2 | cancrinite (Na6Ca2Al6Si6O24(CO3)2) |
| - | - | - | - | 3.3 | - | cancrinite (Na6Ca2Al6Si6O24(CO3)2) |
| 3.37 | - | 3.37 | - | - | - | mullite (Al6Si2O13) |
| - | - | - | 3.37 | - | - | C–S–H (Ca6Si3O12·xH2O) |
| - | - | - | - | 4.2 | - | sodalite (Na8Al6Si6O24Cl2) |
| - | - | - | 4.21 | - | - | C–S–H (Ca6Si3O12·xH2O) |
| - | - | 4.3 | - | - | - | mullite (Al6Si2O13) |
| - | 7.12 | - | - | - | - | kaolinite (Al2Si2O5(OH)4) |
| - | - | - | - | - | 7.15 | kaolinite (Al2Si2O5(OH)4) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahrous, M.A.; Šegvić, B.; Zanoni, G.; Khadka, S.D.; Senadheera, S.; Jayawickrama, P.W. The Role of Clay Swelling and Mineral Neoformation in the Stabilization of High Plasticity Soils Treated with the Fly Ash- and Metakaolin-Based Geopolymers. Minerals 2018, 8, 146. https://doi.org/10.3390/min8040146
Mahrous MA, Šegvić B, Zanoni G, Khadka SD, Senadheera S, Jayawickrama PW. The Role of Clay Swelling and Mineral Neoformation in the Stabilization of High Plasticity Soils Treated with the Fly Ash- and Metakaolin-Based Geopolymers. Minerals. 2018; 8(4):146. https://doi.org/10.3390/min8040146
Chicago/Turabian StyleMahrous, Mahmoud A., Branimir Šegvić, Giovanni Zanoni, Suraj D. Khadka, Sanjaya Senadheera, and Priyantha W. Jayawickrama. 2018. "The Role of Clay Swelling and Mineral Neoformation in the Stabilization of High Plasticity Soils Treated with the Fly Ash- and Metakaolin-Based Geopolymers" Minerals 8, no. 4: 146. https://doi.org/10.3390/min8040146
APA StyleMahrous, M. A., Šegvić, B., Zanoni, G., Khadka, S. D., Senadheera, S., & Jayawickrama, P. W. (2018). The Role of Clay Swelling and Mineral Neoformation in the Stabilization of High Plasticity Soils Treated with the Fly Ash- and Metakaolin-Based Geopolymers. Minerals, 8(4), 146. https://doi.org/10.3390/min8040146





