Hybrid Alkaline Cements: Bentonite-Opc Binders
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
3. Results and Discussion
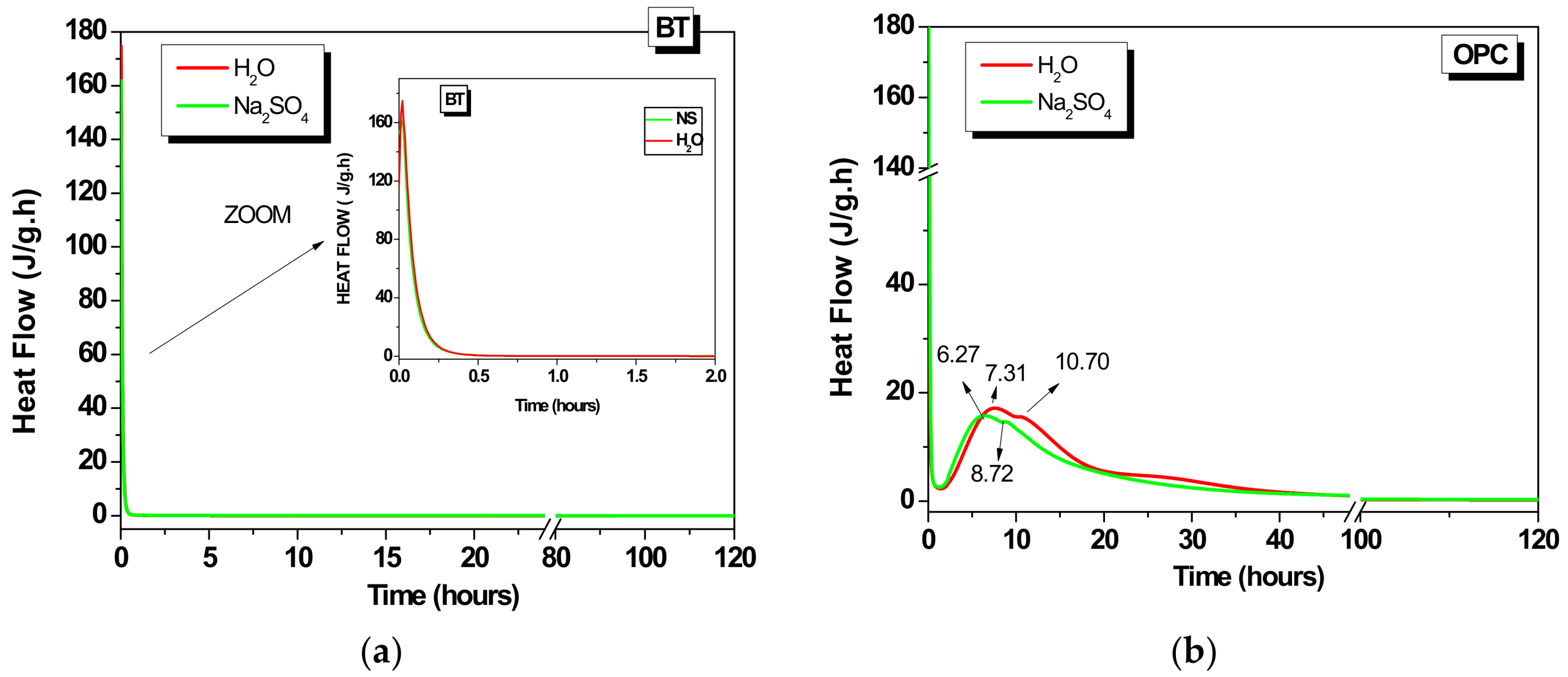
3.1. Calorimetric Study
3.1.1. 100% OPC (OPC)
3.1.2. 100% BT (BT)
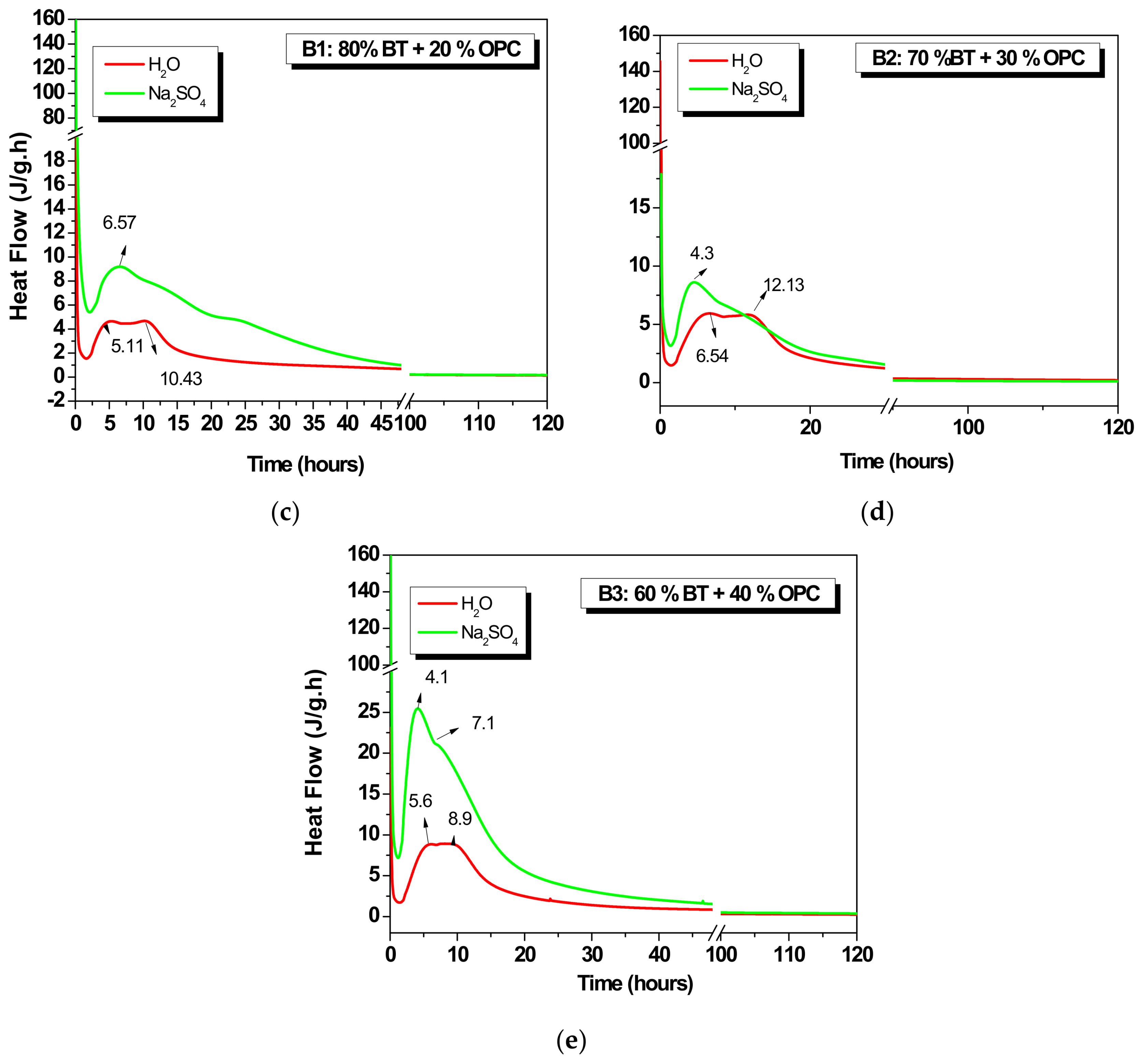
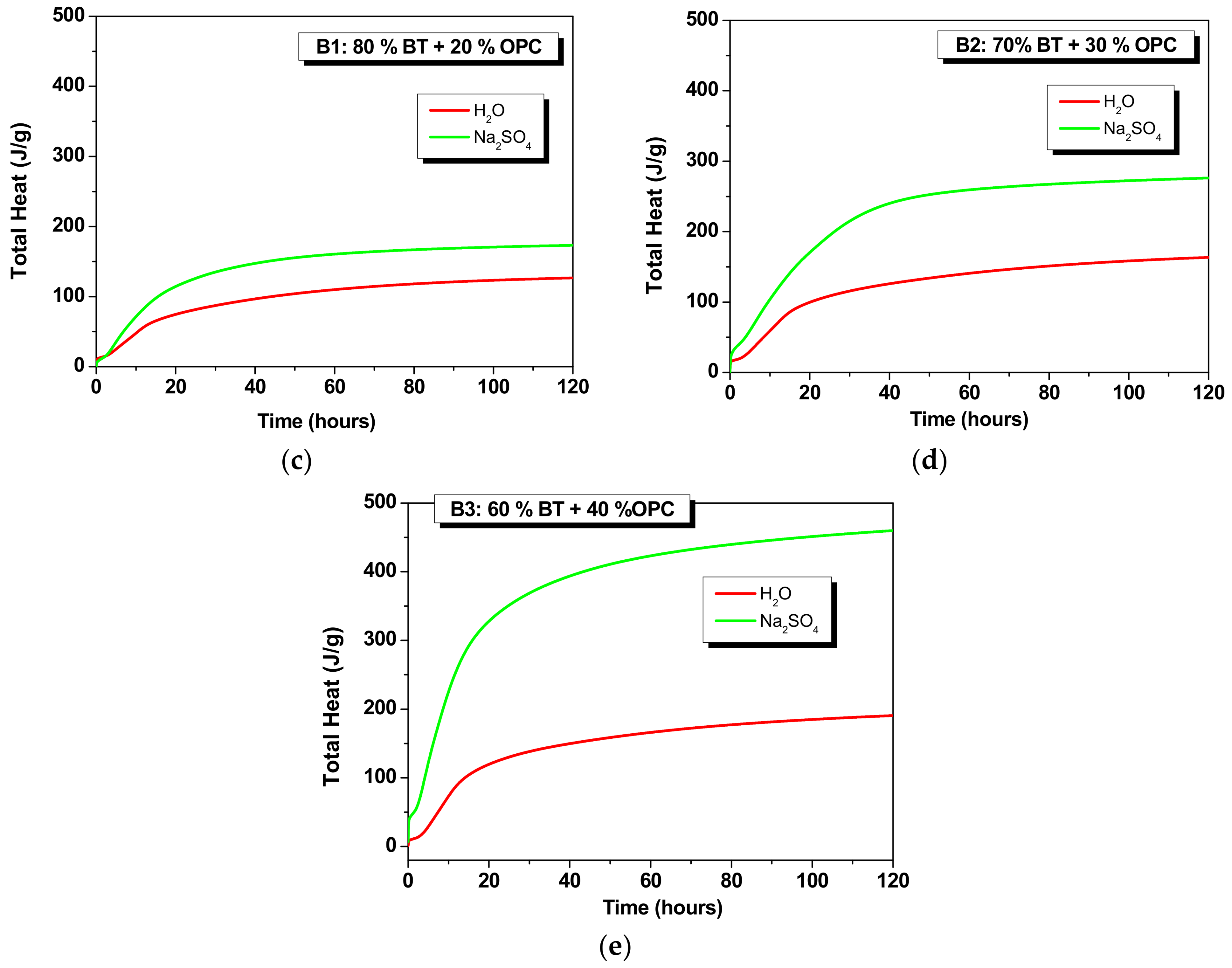
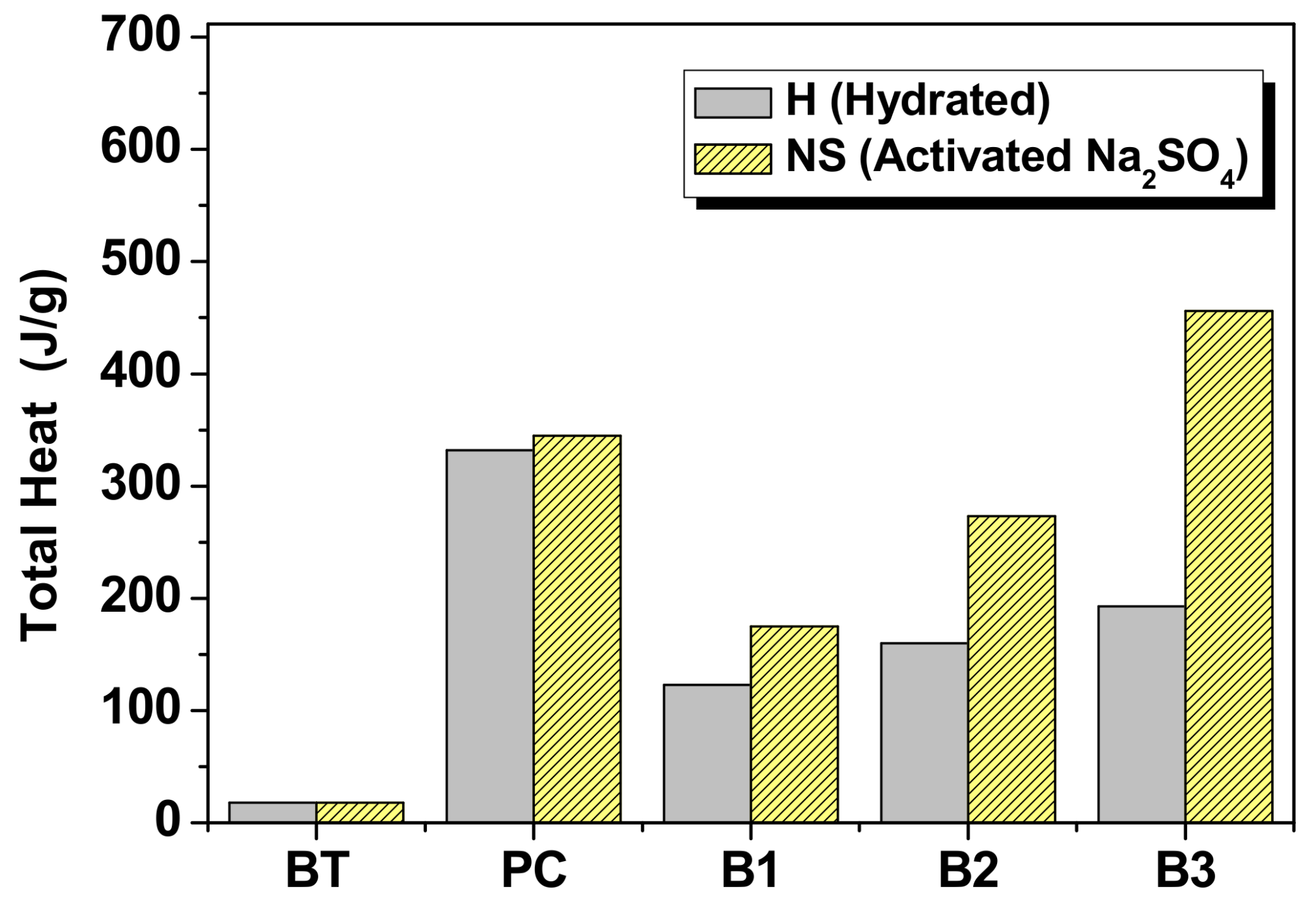
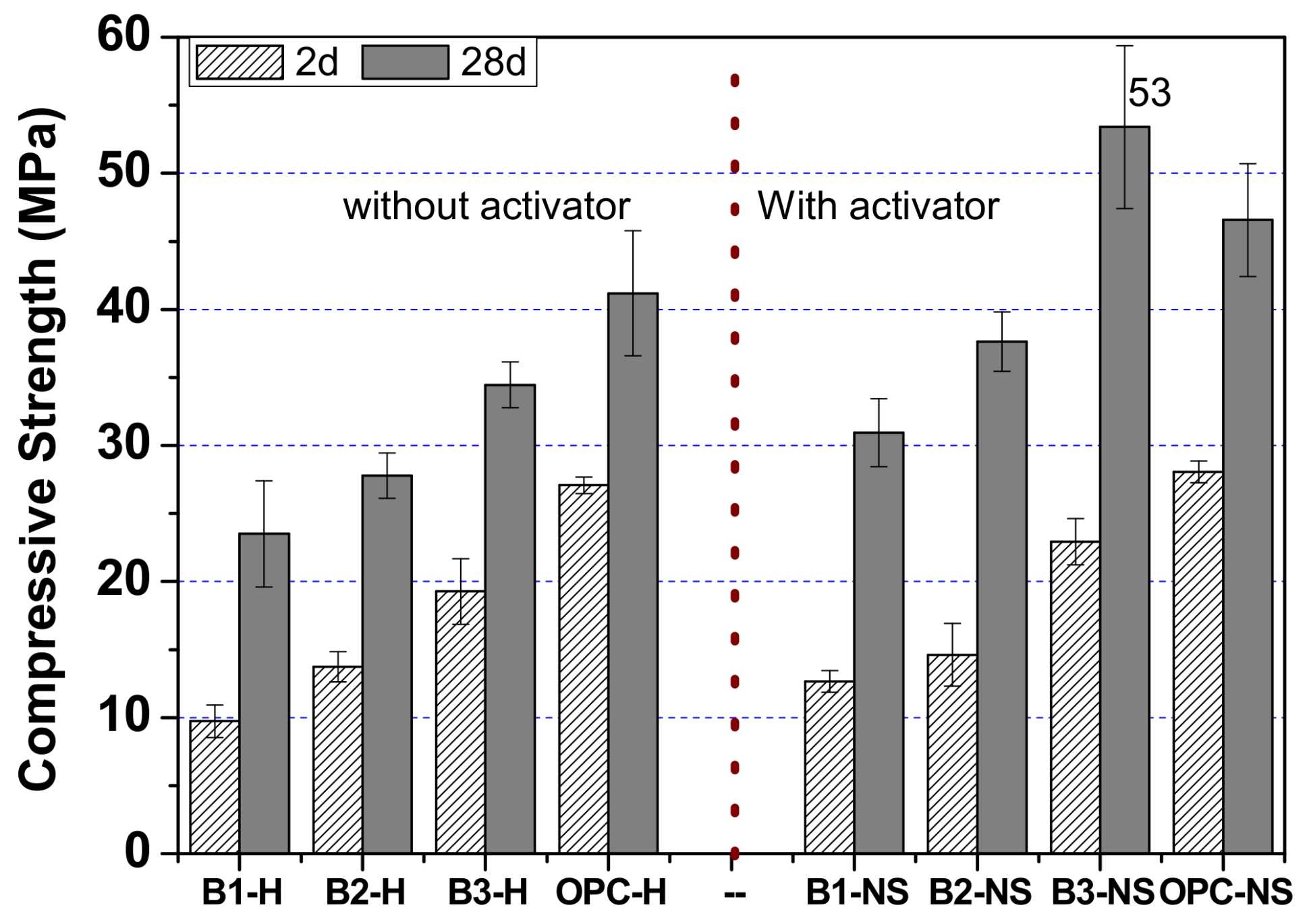
3.1.3. Hybrids (B1, B2 and B3)
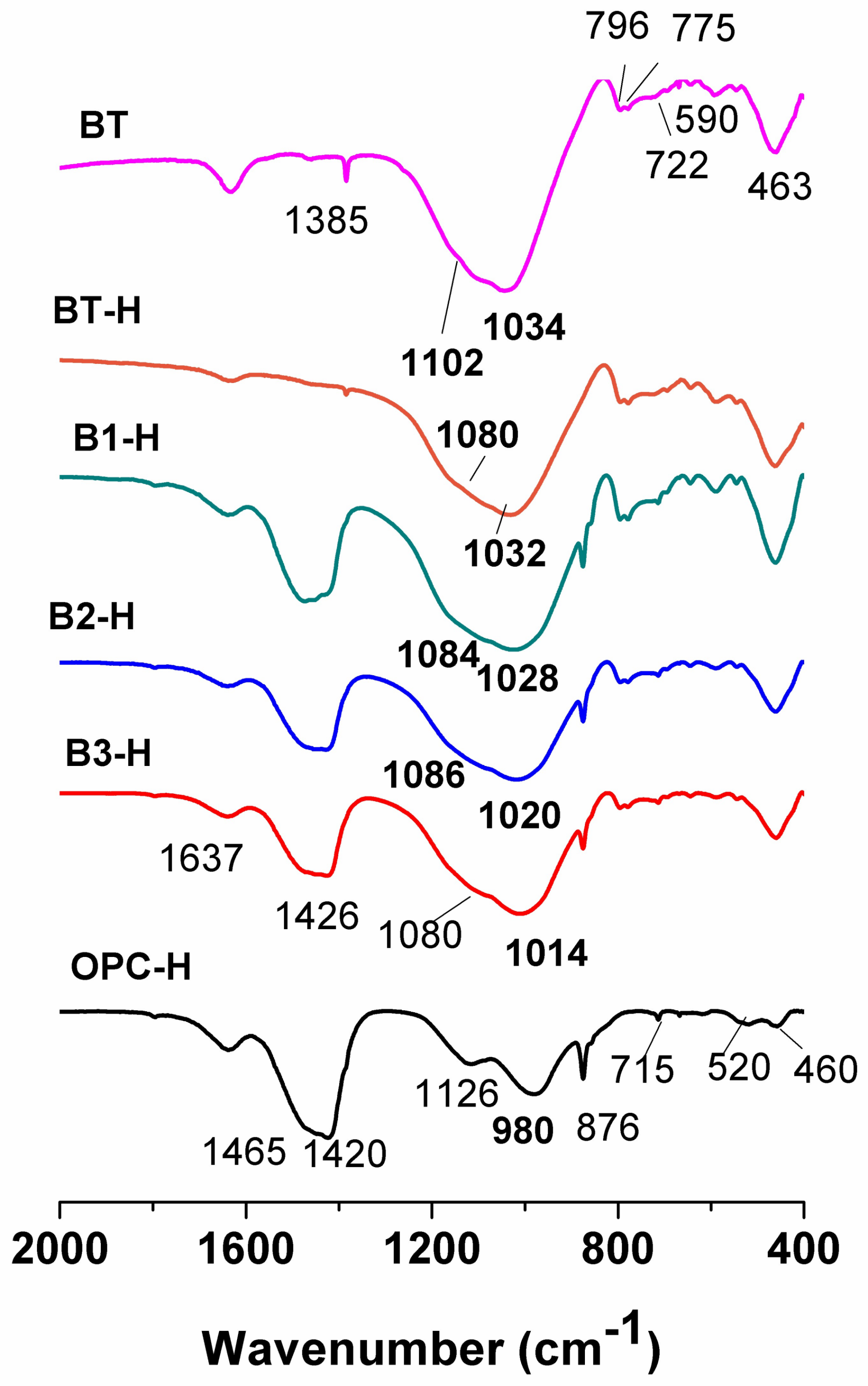
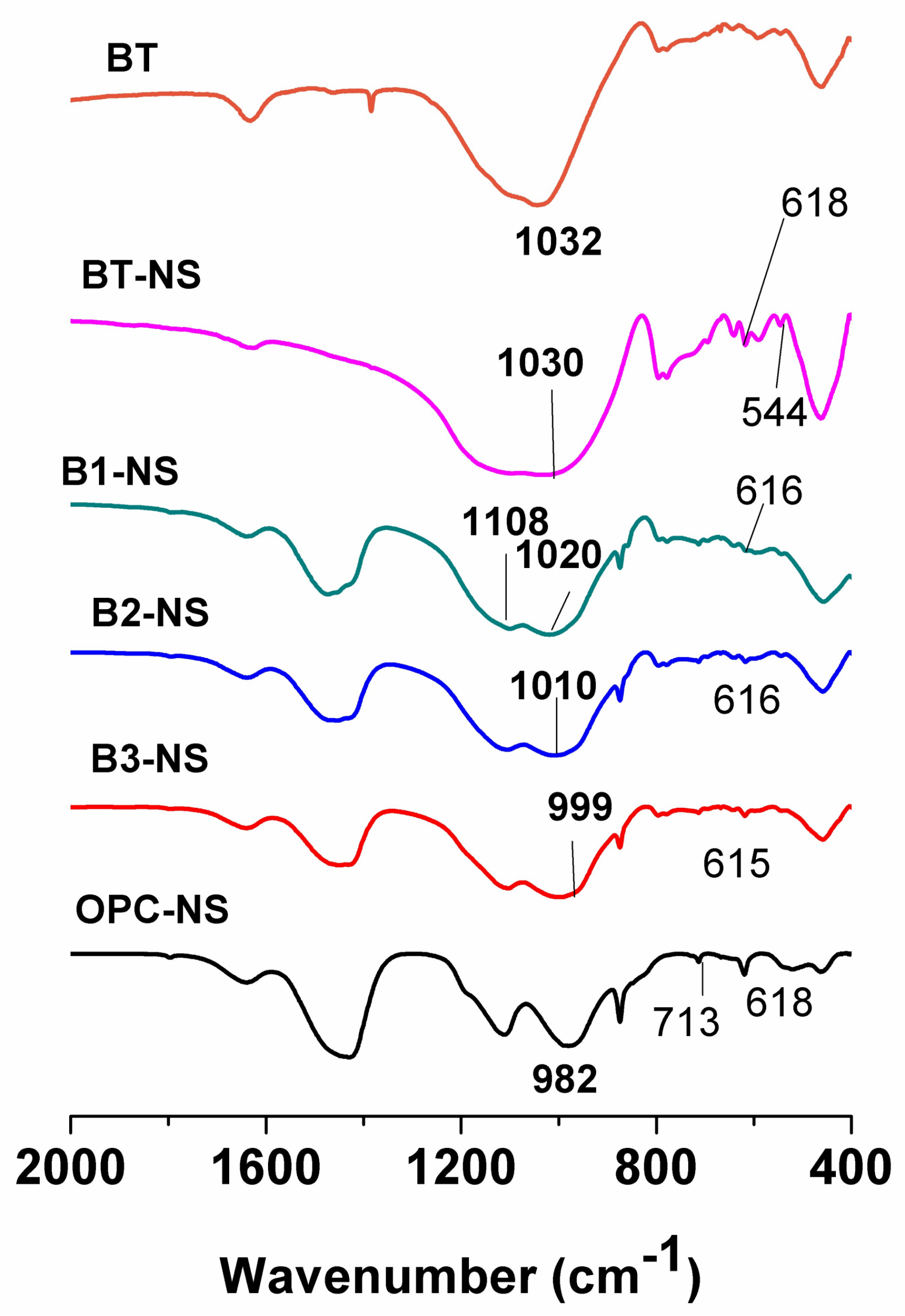
3.2. Characterisation of the Reaction Products
- (i)
- At higher portland cement contents, the main band associated with T–O bond vibrations (B1-NS, 1020 cm−1; B2-NS, 1010 cm−1 and B3-NS, 999 cm−1) shifted toward lower frequencies, narrowed and sharpened. As noted above, this main band overlapped with several others. In this case and given its position, it may have been the result of the overlap between a C-S-H-like gel (cement hydration product) and a N-A-S-H-like gel (product of bentonite alkali-activation).
- (ii)
- The band associated with S–O bond asymmetric stretching vibrations (at around 1110 cm−1), more visible and intense in the Na2SO4-activated systems, was associated with the formation of more ettringite in these systems, as previously detected by XRD.
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Massaza, F. Pozzolanic cements. Cem. Concr. Compos. 1993, 15, 185–214. [Google Scholar] [CrossRef]
- Massaza, F. Pozzolana and Pozzolanic Cements. In Lea’s Chemistry of Cement and Concrete, 4th ed.; P.C. Hewlett: Oxford, UK, 2006. [Google Scholar]
- Calleja, J. Adiciones y Cementos con Adiciones. Materiales de Construccion 1983, 190, 25–52. [Google Scholar] [CrossRef]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary cementitious materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Fernandez-Jimenez, A.; Palomo, A. Cements with low Clinker content: Versatile use of raw materials. J. Sustain. Cem. Based Mater. 2015, 4, 140–151. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Donatello, S.; Fernandez-Jimenez, A.; Palomo, A. Hydration of Hybrid Alkaline Cement containing a very large proportion of fly ash: Descriptive model. Materials 2016, 9, 605. [Google Scholar] [CrossRef] [PubMed]
- Angulo Ramirez, D.E.; Mejia de Gutierrez, R.; Puertas, F. Alkali activated Portland Blast-Furnace Slag Cement: Mechanical properties and Hydration. Constr. Build. Mater. 2017, 140, 119–128. [Google Scholar] [CrossRef]
- Abdollahnejad, Z.; Hlavacek, P.; Miraldo, S.; Pacheco-Torgal, F.; De Aguiar, J.L.B. Compressive strength, microstructure and hydration products of hybrid alkaline cements. Mater. Res. 2014, 17, 829–837. [Google Scholar] [CrossRef]
- Cárdenas Pulido, J.; Lizarazo-Marriaga, J.; Aperador Chaparro, W. Mechanical performance of cementitious binary systems (Portland Cement-fly ash- Blast Furnace Slag). Rev. Latinoam. Metal. Mater. 2016, 36, 78–98. [Google Scholar]
- Buchwald, A.; Hohmann, M.; Posern, K.; Brendler, E. The suitability of thermally activated illite/smectite clay as raw material for geopolymer binders. Appl. Clay Sci. 2009, 46, 300–304. [Google Scholar] [CrossRef]
- Sagoe-Crentsil, K.; Brown, T. Some key materials and process parameters governing geopolymer binder performance. In Proceedings of the International Conference on Pozzolan, Concrete and Geopolymer, Khon Kaen, Thailand, 24–25 May 2006. [Google Scholar]
- Ferone, C.; Colangelo, F.; Cioffi, R.; Montagnaro, F.; Santoro, L. Use of reservoir clay sediments as raw materials for geopolymer binders. Adv. Appl. Ceram. 2013, 112, 184–189. [Google Scholar] [CrossRef]
- Ruiz-Santaquiteria, C.; Fernandez-Jimenez, A.; Skibsted, J.; Palomo, A. Clay reactivity: Production of alkali activated cements. Appl. Clay Sci. 2013, 73, 11–16. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Cherfa, N.; Zibouche, F.; Fernandez-Jimenez, A.; Palomo, A. The role of aluminium in alkali activated bentonites. Mater. Struct. 2014, 48, 585–597. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Cherfa, N.; Fernandez-Jimenez, A.; Palomo, A. Use of Clays in Alklaine Hybrid Cement Preparation. The role of Bentonites. Mater. Lett. 2018. (under review). [Google Scholar]
- Luukkonena, T.; Abdollahnejada, Z.; Yliniemia, J.; Kinnunena, P.B.; Illikainen, M. One-part alkali-activated materials: A review. Cem. Concr. Res. 2018, 103, 21–34. [Google Scholar] [CrossRef]
- Wadsö, U. Applications of an eight-channel isothermal conduction calorimeter for cement hydration studies. Cem. Int. 2005, 3, 94–101. [Google Scholar]
- Ylmén, R.; Wadsö, L.; Panas, I. Insights into early hydration of Portland limestone cement from infrared spectroscopy and isothermal calorimetry. Cem. Concr. Res. 2010, 40, 1541–1546. [Google Scholar] [CrossRef]
- Older, I. Hydration, Setting and Hardening of Portland Cement. In Leas’s Chemistry of Cement and Concrete, 4th ed.; Peter, C., Ed.; Hewlett: Oxford, UK, 2006; p. 351. [Google Scholar]
- Garcia-Lodeiro, I.; Palomo, A.; Fernández-Jiménez, A.; Macphee, D.E. Compatibility studies between N-A-S-H and C-A-S-H gels. Study in the ternary diagram Na2O–CaO–Al2O3–SiO2–H2O. Cem. Concr. Res. 2011, 41, 923–931. [Google Scholar] [CrossRef]
- Shi, C.; Day, R.L. Pozzolanic reaction in the presence of chemical activators Part II: Reaction products and mechanism. Cem. Concr. Res. 2000, 30, 607–613. [Google Scholar] [CrossRef]
- Shi, C.; Day, R.L. Comparison of different methods for enhancing reactivity of pozzolans. Cem. Concr. Res. 2001, 31, 813–818. [Google Scholar] [CrossRef]
- Wu, Z.; Naik, T. Chemically activated Blended Cements. ACI Mater. 2003, 100, 434–440. [Google Scholar]
- Farmer, V.C. The Layers Silicates: The Infrared Spectra of Minerals; Mineralogical Society: London, UK, 1974. [Google Scholar]
- Gil Alabarse, F.; Vieira Conceição, R.; Balzaretti, N.M.; Schenato, F.; Xavier, A.M. In-situ FTIR analyses of bentonite under high-pressure. Appl. Clay Sci. 2011, 51, 202–208. [Google Scholar] [CrossRef]
- Gadsden, J.A. Infrared Spectra of Minerals and Related Inorganic Compounds; Butterworths: London, UK, 1975. [Google Scholar]
- Yu, P.; Kirkpatrick, R.J.; Poe, B.; McMillan, P.F.; Cong, X. Structure of Calcium Silicate Hydrate (C-S-H): Near-, Mid- and Far-Infrared Spectroscopy. J. Am. Ceram. Soc. 1999, 82, 742–748. [Google Scholar] [CrossRef]
- Vazquez-Moreno, T.; Blanco Varela, M.T. Tabla de frecuencias y espectros de absorcion Infraroja de Compuestos relacionados con la Quimica del Cemento. Materiales de Construccion 1981, 182, 31–48. [Google Scholar] [CrossRef]
- Periasamy, A.; Muruganand, S.; Palaniswamy, M. Vibrational Studies of Na2SO4, K2SO4, NaHSO4 and KHSO4 Crystals, Rasayan. J. Chem. 2009, 2, 981–989. [Google Scholar]
- Liu, Y.; Wang, A.; Freeman, J.J. Raman, MIR and NIR spectroscopic study of calcium sulfates:gypsum, basanite and anhydrite. In Proceedings of the 40th Lunar and Planetary Science Conferece, The Woodlands, TX, USA, 23–27 March 2009. [Google Scholar]
- Learch, W. The influence of gypsum on the hydration and properties of Portland cement pastes. Portland Cem. Assoc. Bull. 1946, 12, 41. [Google Scholar]
- Jawed, I.; Skalny, J. Alkalis in cement: A review II. Effects of alkalis on hydration and performance of Portland Cement. Cem. Concr. Res. 1978, 8, 37–52. [Google Scholar]
- Odler, I.; Wonnemann, R. Effec of alkalies on Portland Cement hydration. II. Alkalis present in form of sulphates. Cem. Concr. Res. 1982, 13, 771–777. [Google Scholar] [CrossRef]
- He, C.; Osback, D.; Makolicky, E. Pozzolanic Reactions of six principal clay minerals. Activation reactivity assessments and technological effects. Cem. Concr. Res. 1995, 737, 1691–1702. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Fernandez-Jimenez, A.; Palomo, A. Hydration Kinetics in hybrid binders. Cem. Concr. Compos. 2013, 39, 82–92. [Google Scholar] [CrossRef]

| Raw Material | Chemical Composition (XRF, wt %) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LoI (2) | SiO2 | Al2O3 | CaO | Na2O | K2O | Fe2O3 | MgO | SO3 | Other | |
| BT (1) | 0.56 | 62.6 | 17.93 | 1.60 | 6.56 | 2.37 | 3.12 | 3.89 | 0.87 | 0.83 |
| OPC | 2.56 | 20.26 | 6.33 | 62.70 | 0.59 | 0.79 | 2.30 | 0.18 | 2.82 | 1.47 |
| Mineralogical Composition (XRD) | ||||||||||
| BT | Albite (alb: NaAlSi3O8): Quartz (q: SiO2) (amorphous phase (3) 66.58%) | |||||||||
| PC | Alite (C3S); Belite (C2S); Ferritic phase (C4AF); Tricalcium aluminate (C3A); gypsum (g: CaSO4·2H2O) | |||||||||
| System | Crystalline Phases Detected in the Precursors * | Reaction Products * | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H (Water Hydrated) | NS (Activated) | H (Water Hydrated) | NS (Activated) | ||||||||||||||
| A | B | q | alb | A | B | q | alb | e | p | c | g | e | p | c | g | ||
| 2 days | OPC | √ | √ | x | x | √ | √ | x | x | √ | √ | x | x | √ | √ | x | x |
| BT | x | x | √ | √ | x | x | √ | √ | x | x | x | x | x | x | x | √ | |
| B1 | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | x | √ | √ | √ | x | |
| B2 | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | x | x | √ | √ | x | x | |
| B3 | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | x | x | √ | √ | x | x | |
| 28 days | OPC | √ | √ | x | x | √ | √ | x | x | √ | √ | √ | x | √ | √ | √ | x |
| BT | x | x | √ | √ | x | x | √ | √ | x | x | √ | x | x | x | x | √ | |
| B1 | x | x | √ | √ | x | x | √ | √ | √ | x | √ | x | √ | x | √ | x | |
| B2 | x | x | √ | √ | x | x | √ | √ | x | √ | √ | x | √ | x | √ | x | |
| B3 | x | √ | √ | √ | x | √ | √ | √ | √ | √ | √ | x | √ | x | √ | x | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Lodeiro, I.; Fernandez-Jimenez, A.; Palomo, A. Hybrid Alkaline Cements: Bentonite-Opc Binders. Minerals 2018, 8, 137. https://doi.org/10.3390/min8040137
Garcia-Lodeiro I, Fernandez-Jimenez A, Palomo A. Hybrid Alkaline Cements: Bentonite-Opc Binders. Minerals. 2018; 8(4):137. https://doi.org/10.3390/min8040137
Chicago/Turabian StyleGarcia-Lodeiro, Ines, Ana Fernandez-Jimenez, and Angel Palomo. 2018. "Hybrid Alkaline Cements: Bentonite-Opc Binders" Minerals 8, no. 4: 137. https://doi.org/10.3390/min8040137
APA StyleGarcia-Lodeiro, I., Fernandez-Jimenez, A., & Palomo, A. (2018). Hybrid Alkaline Cements: Bentonite-Opc Binders. Minerals, 8(4), 137. https://doi.org/10.3390/min8040137





