Mineralogical and Geochemical Constraints on Magma Evolution and Late-Stage Crystallization History of the Breivikbotn Silicocarbonatite, Seiland Igneous Province in Northern Norway: Prerequisites for Zeolite Deposits in Carbonatite Complexes
Abstract
1. Introduction
2. Geological Setting
2.1. Seiland Igneous Province
2.2. Breivikbotn Carbonatite
3. Analytical Methods
3.1. Mineral Analyses
3.2. Whole-Rock Analyses
4. Petrography and Mineral Compositions: Results and Interpretation
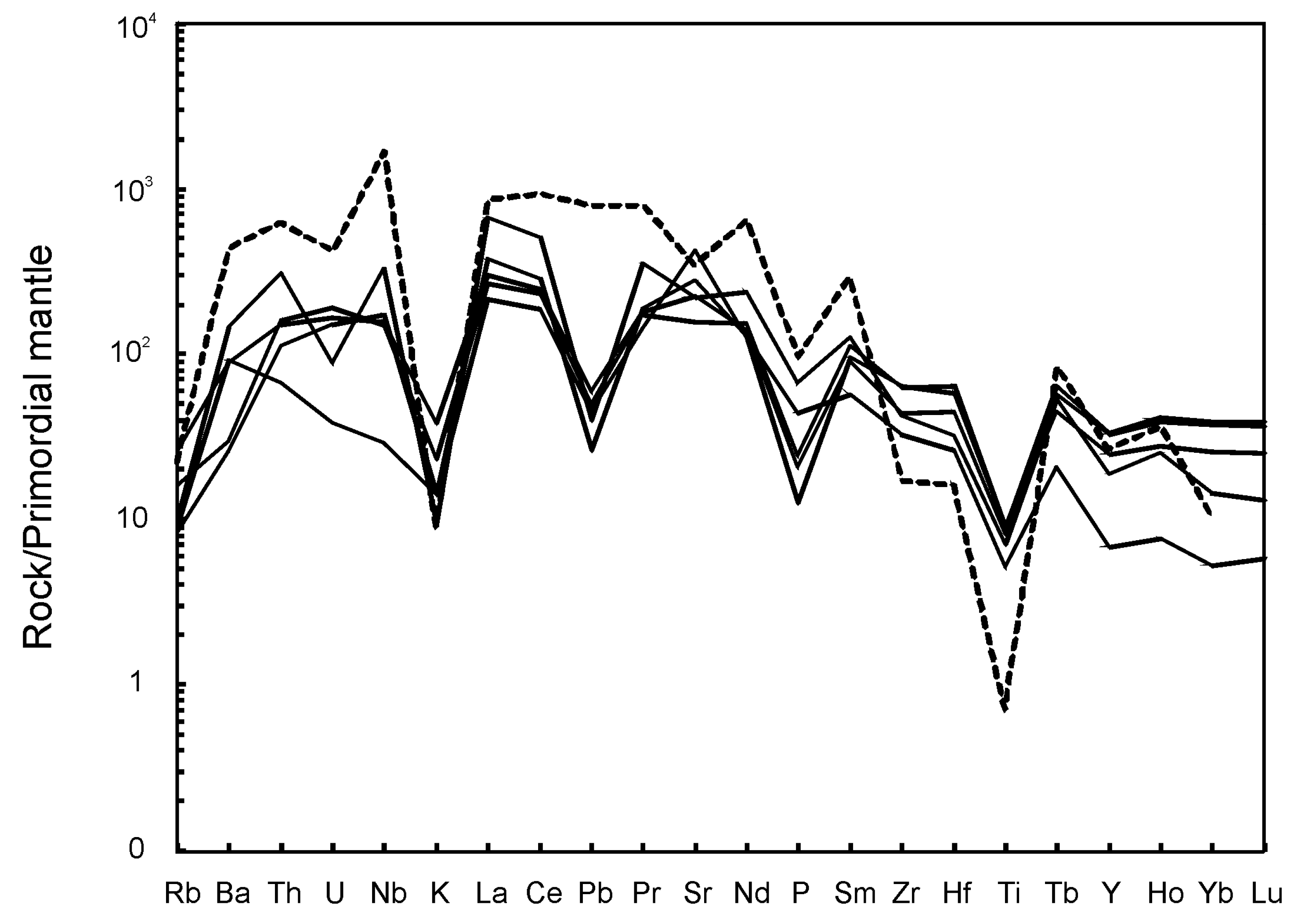
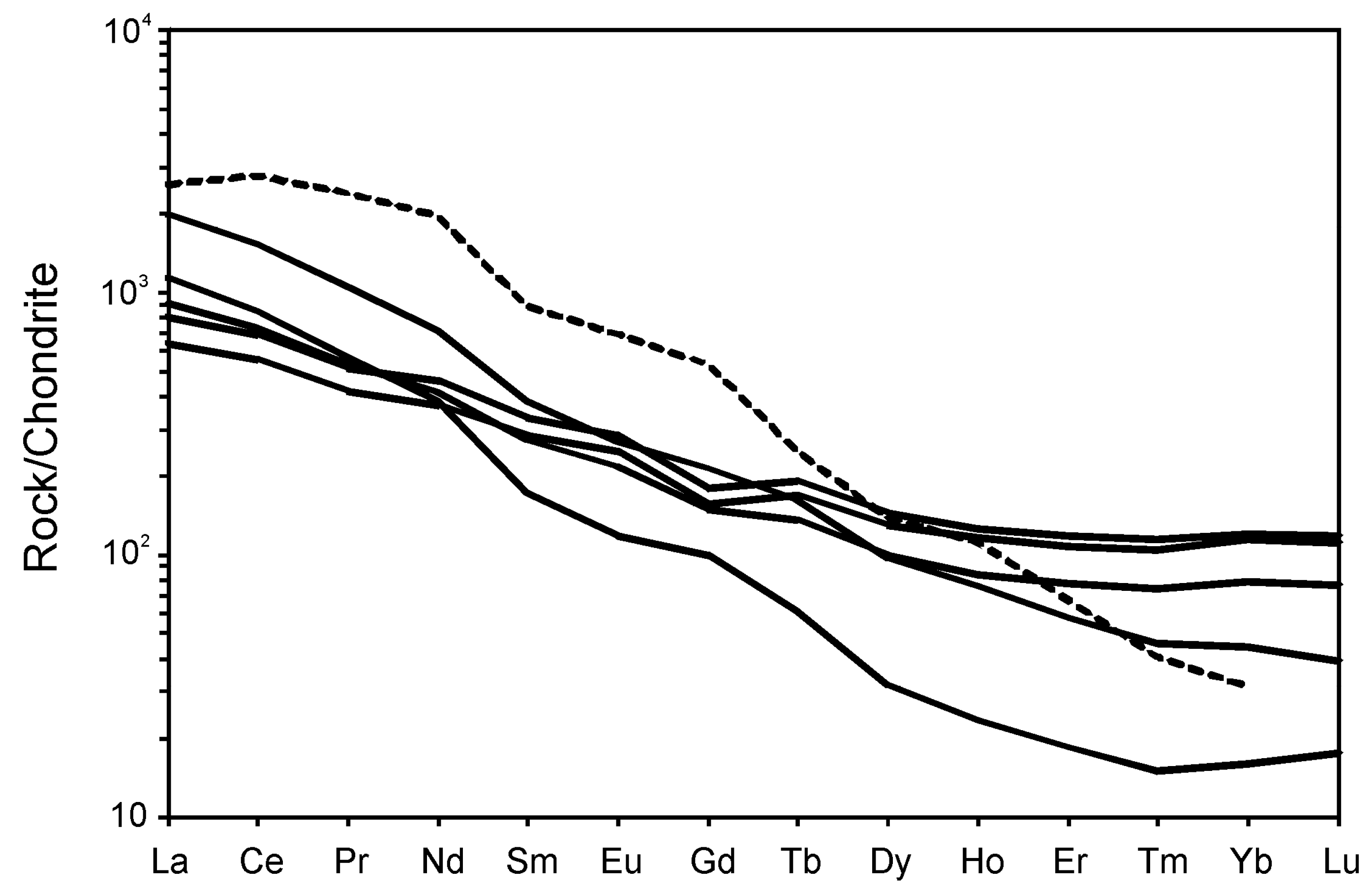
5. Whole-Rock Compositions
5.1. Major Elements
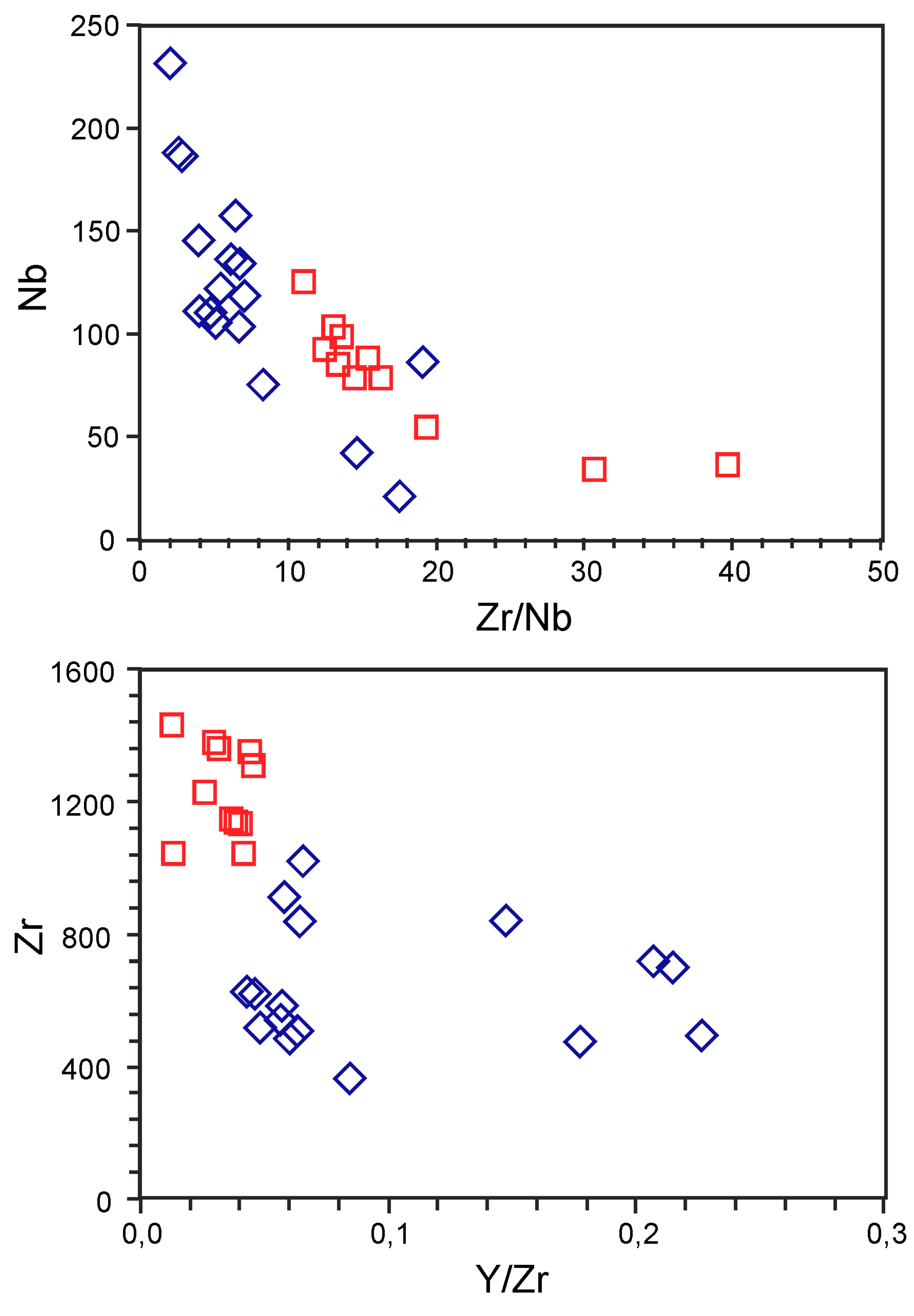
5.2. Trace Elements
6. Discussion
6.1. Petrographical Classification of the Breivkbotn Carbonatite and Alkaline Rocks
6.2. Geochemical Constraints on Primary Melts, Magma Evolution and Geodynamic Setting of Breivikbotn Complex
6.3. Late- to Post-Magmatic Alteration and Possible Metamorphic Overprints
(Na,Ca)2(Si,Al)5O10·3H2O (Gonn)
NaCa2(Al5Si5)O20·6H2O (Thoms)
2(Na,Ca)2(Si,Al)5O10·3H2O (Gonn)
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Woolley, A.R.; Kjarsgaard, B.A. Paragenetic types of carbonatite as indicated by the diversity and relative abundances of associated silicate rocks: Evidence from a global database. Can. Mineral. 2008, 46, 741–752. [Google Scholar] [CrossRef]
- Hou, Z.Q.; Tian, S.H.; Yuan, Z.X.; Xie, Y.L.; Yin, S.P.; Yi, L.S.; Fei, H.C.; Yang, Z.M. The Himalayan collision zone carbonatites in western Sichuan, SW china: Petrogenesis, mantle source and tectonic implication. Earth Planet. Sci. Lett. 2006, 244, 234–250. [Google Scholar] [CrossRef]
- D’Orazio, M.; Innocenti, F.; Tonarini, S.; Doglioni, C. Carbonatites in a subduction system: The Pleistocene alvikites from Mt. Vulture (southern Italy). Lithos 2007, 98, 313–334. [Google Scholar] [CrossRef]
- Xu, C.; Taylor, R.N.; Kynicky, J.; Chakhmouradian, A.R.; Song, W.L.; Wang, L.J. The origin of enriched mantle beneath north China block: Evidence from young carbonatites. Lithos 2011, 127, 1–9. [Google Scholar] [CrossRef]
- Hagen-Peter, G.; Cottle, J.M. Synchronous alkaline and subalkaline magmatism during the late Neoproterozoic-Early Paleozoic Ross orogeny, Antarctica: Insights into magmatic sources and processes within a continental arc. Lithos 2016, 262, 677–698. [Google Scholar] [CrossRef]
- Ravna, E.K.; Zozulya, D.; Kullerud, K.; Corfu, F.; Nabelek, P.I.; Janak, M.; Slagstad, T.; Davidsen, B.; Selbekk, R.S.; Schertl, H.P. Deep-seated carbonatite intrusion and metasomatism in the UHP Tromso Nappe, northern Scandinavian Caledonides—A natural example of generation of carbonatite from carbonated eclogite. J. Petrol. 2017, 58, 2403–2428. [Google Scholar] [CrossRef]
- Elliott, H.A.L.; Wall, F.; Chakhmouradian, A.R.; Siegfried, P.R.; Dahlgren, S.; Weatherly, S.; Finch, A.A.; Marks, M.A.W.; Dowman, E.; Deady, E. Fenites associated with carbonatite complexes: A review. Ore Geol. Rev. 2018, 93, 38–59. [Google Scholar] [CrossRef]
- Ramsay, D.M.; Sturt, B.A.; Zwaan, K.B.; Roberts, D. Caledonides of Northern Norway. In The Caledonian Orogen: Scandinavia and Related Areas; Gee, D.G., Sturt, B.A., Eds.; Wiley: New York, NY, USA, 1985; pp. 163–184. [Google Scholar]
- Corfu, F.; Torsvik, T.H.; Andersen, T.B.; Ashwal, L.D.; Ramsay, D.M.; Roberts, R.J. Early Silurian mafic-ultramafic and granitic plutonism in contemporaneous flysch, Mageroy, northern Norway: U-Pb ages and regional significance. J. Geol. Soc. 2006, 163, 291–301. [Google Scholar] [CrossRef]
- Sturt, B.A.; Pringle, I.R.; Ramsay, D.M. The Finnmarkian phase of the Caledonian Orogeny. J. Geol. Soc. Lond. 1978, 135, 597–610. [Google Scholar] [CrossRef]
- Ramsay, D.M.; Sturt, B.A. The contribution of Finnmarkian Orogeny to the framework of the Scandinavian Caledonides. In Synthesis of the Caledonian Rocks of Britain; Fettes, D.J., Harris, A.L., Eds.; D. Reidel Publishing Company: Dordrecht, The Netherlands, 1986. [Google Scholar]
- Sturt, B.A.; Ramsay, D.M. The alkaline complex of the Breivikbotn area, Sørøy, Northern Norway. Nor. Geol. Unders. Bull. 1965, 231, 1–142. [Google Scholar]
- Krill, A.G.; Zwaan, B. Reinterpretation of Finnmarkian deformation on western Soroy, northern Norway. Nor. Geol. Tidsskr. 1987, 67, 15–24. [Google Scholar]
- Roberts, R.J.; Corfu, F.; Torsvik, T.H.; Hetherington, C.J.; Ashwal, L.D. Age of alkaline rocks in the Seiland igneous province, northern Norway. J. Geol. Soc. 2010, 167, 71–81. [Google Scholar] [CrossRef]
- Jonassen, A. Geologiske og petrologiske undersøkelser av alkaline bergarter og metasedimenter tilknyttet Breivikbotnkomplekset pa Sørøy, Vest Finnmark. Cand. Scient. Master’s Thesis, University of Tromsø, Tromsø, Norway, 1996. (In Norwegian). [Google Scholar]
- Cazes, J. Ewing’s Analytical Instrumentation Handbook, 3rd ed.; Marcel Dekker: New York, NY, USA, 2005; p. 1037. [Google Scholar]
- Govindaraju, K. 1994 compilation of working values and sample description for 383 geostandards (vol 18, pg 53, 1994). Geostand. Newsl. 1994, 18, 331. [Google Scholar] [CrossRef]
- Chakhmouradian, A.R.; McCammon, C.A. Schorlomite: A discussion of the crystal chemistry, formula, and inter-species boundaries. Phys. Chem. Miner. 2005, 32, 277–289. [Google Scholar] [CrossRef]
- Huggins, F.E.; Virgo, D.; Huckenholz, H.G. Titanium-containing silicate garnets. 1. Distribution of Al, Fe3+, and Ti4+ between octahedral and tetrahedral sites. Am. Mineral. 1977, 62, 475–490. [Google Scholar]
- Lupini, L.; Williams, C.T.; Woolley, A.R. Zr-rich garnet and Zr-rich and Th-rich perovskite from the Polino carbonatite, Italy. Mineral. Mag. 1992, 56, 581–586. [Google Scholar] [CrossRef]
- Russell, J.K.; Dipple, G.M.; Lang, J.R.; Lueck, B. Major-element discrimination of titanian andradite from magmatic and hydrothermal environments: An example from the Canadian Cordillera. Eur. J. Mineral. 1999, 11, 919–935. [Google Scholar] [CrossRef]
- Saha, A.; Ray, J.; Ganguly, S.; Chatterjee, N. Occurrence of melanite garnet in syenite and ijolite-melteigite rocks of samchampi-samteran alkaline complex, mikir hills, northeastern india. Curr. Sci. 2011, 101, 95–100. [Google Scholar]
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Blancher, S.B.; D’Arco, P.; Fonteilles, M.; Pascal, M.L. Evolution of nepheline from mafic to highly differentiated members of the alkaline series: The Messum complex, Namibia. Mineral. Mag. 2010, 74, 415–432. [Google Scholar] [CrossRef]
- Henderson, C.M.B.; Gibb, F.G.F. Plagioclase-Ca-rich-nepheline intergrowths in a syenite from the Marangudzi complex, Rhodesia. Mineral. Mag. 1972, 38, 670–677. [Google Scholar] [CrossRef]
- Lumpkin, G.R. Nepheline and sodalite in a barred olivine chondrule from the Allende meteorite. Meteoritics 1980, 15, 139–147. [Google Scholar] [CrossRef]
- Ross, D.K.; Simon, J.I.; Simon, S.B.; Grossman, L. Two generations of sodic metasomatism in an Allende Type B CAI. In Proceedings of the 46th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 16–20 March 2015. [Google Scholar]
- Carswell, D.A.; Wilson, R.N.; Zhai, M. Ultra-high pressure aluminous titanites in carbonate-bearing eclogites at Shuanghe in Dabieshan, central China. Mineral. Mag. 1996, 60, 461–471. [Google Scholar] [CrossRef]
- Mitchell, R.H. Undersaturated Alkaline Rocks: Mineralogy, Petrogenesis, and Economic Potential; Mineralogical Association of Canada, Short Courses: Quebec City, QC, Canada, 1996; Volume 24, p. 312. [Google Scholar]
- Sun, S.S.; McDonough, W.F. Chemical and isotopic systematics of oceanic basalts: Implications for mantle composition and processes. Geol. Soc. Lond. Spec. Publ. 1989, 42, 313–345. [Google Scholar] [CrossRef]
- Streckeisen, A. Classification and nomenclature of volcanic-rocks, lamprophyres, carbonatites, and melilitic rocks—Recommendations and suggestions of the IUGS sub-commission on the systematics of igneous rocks. Geology 1979, 7, 331–335. [Google Scholar]
- Woolley, A.R.; Kempe, D.R.C. Carbonatites: Nomenclature, average compositions, and element distribution. In Carbonatites: Genesis and Evolution; Bell, K., Ed.; Unwin Hyman: London, UK, 1989; pp. 1–14. [Google Scholar]
- LeMaitre, R.W. Igneous Rocks: A classification and Glossary of Terms; Cambridge University Press: Cambridge, UK, 2002; p. 236. [Google Scholar]
- Heinrich, E.W. The Geology of Carbonatites; Rand McNally: Chicago, IL, USA, 1966; p. 555. [Google Scholar]
- Andersen, T. Evolution of peralkaline calcite carbonatite magma in the Fen complex, southeast Norway. Lithos 1988, 22, 99–112. [Google Scholar] [CrossRef]
- Robins, B. The Seiland Igneous Province, N. Norway: General Geology and Magmatic Evolution. Field Trip Guidebook, Part II, IGCP Project 336. Norges Geologiske Undersøkelse Report. 1996, p. 34. Available online: http://www.ngu.no/upload/Publikasjoner/Rapporter/1996/96_127.pdf (accessed on 22 September 2018).
- Garson, M.S. Carbonatites in Malawi. In Carbonatites; Tuttle, O.F., Gittins, J., Eds.; Interscience: New York, NY, USA, 1966; pp. 33–71. [Google Scholar]
- King, B.C.; Sutherland, D.S. The carbonatite complexes of Eastern Uganda. In Carbonatites; Tuttle, O.F., Gittins, J., Eds.; Interscience: New York, NY, USA, 1966; pp. 73–126. [Google Scholar]
- Le Bas, M.J. Carbonatite–Nephelinite Volcanism: An African Case History; John Wiley: London, UK, 1977. [Google Scholar]
- Nelson, D.R.; Chivas, A.R.; Chappell, B.W.; McCulloch, M.T. Geochemical and isotopic systematics in carbonatites and implications for the evolution of ocean-island sources. Geochim. Cosmochim. Acta 1988, 52, 1–17. [Google Scholar] [CrossRef]
- Cooper, A.F.; Reid, D.L. Nepheline sovites as parental magmas in carbonatite complexes: Evidence from Dicker Willem, southwest Namibia. J. Petrol. 1998, 39, 2123–2136. [Google Scholar] [CrossRef]
- Nedosekova, I.L.; Belousova, E.A.; Sharygin, V.V.; Belyatsky, B.V.; Bayanova, T.B. Origin and evolution of the Ilmeny-Vishnevogorsky carbonatites (Urals, Russia): Insights from trace-element compositions, and Rb-Sr, Sm-Nd, U-Pb, Lu-Hf isotope data. Mineral. Petrol. 2013, 107, 101–123. [Google Scholar] [CrossRef]
- Kapustin, Y.L. The origin of early calcitic carbonatites. Int. Geol. Rev. 1986, 28, 1031–1044. [Google Scholar] [CrossRef]
- Treiman, A.H.; Essene, E.J. The Oka carbonatite complex, Quebec—Geology and evidence for silicate-carbonate liquid immiscibility. Am. Mineral. 1985, 70, 1101–1113. [Google Scholar]
- Mitchell, R.H. Carbonatites and carbonatites and carbonatites. Can. Mineral. 2005, 43, 2049–2068. [Google Scholar] [CrossRef]
- Wallace, M.E.; Green, D.H. An experimental-determination of primary carbonatite magma composition. Nature 1988, 335, 343–346. [Google Scholar] [CrossRef]
- Eggler, D.H. Carbonatites, primary melts, and mantle dynamics. In Carbonatites: Genesis and Evolution; Bell, K., Ed.; Unwin Hyman: London, UK, 1989; pp. 561–579. [Google Scholar]
- Wyllie, P.J.; Lee, W.J. Model system controls on conditions for formation of magnesiocarbonatite and calciocarbonatite magmas from the mantle. J. Petrol. 1998, 39, 1885–1893. [Google Scholar] [CrossRef]
- Dalton, J.A.; Wood, B.J. The compositions of primary carbonate melts and their evolution through wallrock reaction in the mantle. Earth Planet. Sci. Lett. 1993, 119, 511–525. [Google Scholar] [CrossRef]
- Le Bas, M.J. Nephelinites and carbonatites. In Alkaline Igneous Rocks; Fitton, J.G., Upton, B.G.J., Eds.; Geological Society Special Publications; Blackwell Scientific Publications: Oxford, UK, 1987; pp. 85–94. [Google Scholar]
- Gittins, J. The origin and evolution of carbonatite magmas. In Carbonatites: Genesis and Evolution; Bell, K., Ed.; Unwin Hyman: London, UK, 1989; pp. 580–600. [Google Scholar]
- Gittins, J.; Jago, B.C. Differentiation of natrocarbonatite magma at Oldoinyo Lengai volcano, Tanzania. Mineral. Mag. 1998, 62, 759–768. [Google Scholar] [CrossRef]
- Veksler, I.V.; Petibon, C.; Jenner, G.A.; Dorfman, A.M.; Dingwell, D.B. Trace element partitioning in immiscible silicate-carbonate liquid systems: An initial experimental study using a centrifuge autoclave. J. Petrol. 1998, 39, 2095–2104. [Google Scholar] [CrossRef]
- Chakhmouradian, A.R. High-field-strength elements in carbonatitic rocks: Geochemistry, crystal chemistry and significance for constraining the sources of carbonatites. Chem. Geol. 2006, 235, 138–160. [Google Scholar] [CrossRef]
- Chakhmouradian, A.R.; Mumin, A.H.; Demeny, A.; Elliott, B. Postorogenic carbonatites at Eden Lake, Trans-Hudson orogen (northern Manitoba, Canada): Geological setting, mineralogy and geochemistry. Lithos 2008, 103, 503–526. [Google Scholar] [CrossRef]
- Kogarko, L.N. Geochemistry of radioactive elements in the rocks of the Guli massif, Polar Siberia. Geochem. Int. 2012, 50, 719–725. [Google Scholar] [CrossRef]
- Ying, J.; Zhou, X.; Zhang, H. Geochemical and isotopic investigation of the Laiwu-Zibo carbonatites from western Shandong Province, China, and implications for their petrogenesis and enriched mantle source. Lithos 2004, 75, 413–426. [Google Scholar] [CrossRef]
- Stoppa, F.; Woolley, A.R. The Italian carbonatites: Field occurrence, petrology and regional significance. Mineral. Petrol. 1997, 59, 43–67. [Google Scholar] [CrossRef]
- Gottardi, G. The genesis of zeolites. Eur. J. Mineral. 1989, 1, 479–487. [Google Scholar] [CrossRef]
- Senderov, E.E.; Khitarov, N.I. Synthesis of thermodynamically stable zeolites in the Na2O-Al2O3-SiO2-H2O system. Am. Chem. Soc. Adv. Chem. Ser. 1971, 101, 149–154. [Google Scholar]
- Kristmannsdóttir, H.; Tómasson, J. Zeolite zones in geothermal areas in Iceland. In Natural Zeolites, Occurrence, Properties, and Use; Sand, L.B., Mumpton, F.A., Eds.; Pergamon: New York, NY, USA, 1978; pp. 277–284. [Google Scholar]
- Carpenter, A.B. Graphical analysis of zeolite mineral assemblages from the Bay of Fundy area, Nova Scotia. Am. Chem. Soc. Adv. Chem. Ser. 1971, 101, 328–333. [Google Scholar]
- Dingwell, D.B.; Brearley, M. Mineral chemistry of igneous melanite garnets from analcite-bearing volcanic-rocks, Alberta, Canada. Contrib. Mineral. Petrol. 1985, 90, 29–35. [Google Scholar] [CrossRef]
- Gwalani, L.G.; Rock, N.M.S.; Ramasamy, R.; Griffin, B.J.; Mulai, B.P. Complexly zoned Ti-rich melanite-schorlomite garnets from Ambadungar carbonatite-alkalic complex, Deccan igneous province, Gujarat state, western India. J. Asian Earth Sci. 2000, 18, 163–176. [Google Scholar] [CrossRef]




| Sample No. | 13-4-1-5-1 | 13-4-1-5-2 | 13-2-1a |
|---|---|---|---|
| wt % | |||
| FeO | 0.16 | 0.00 | 0.16 |
| MnO | 0.25 | 0.22 | 0.27 |
| MgO | 0.07 | 0.00 | - |
| CaO | 53.65 | 55.29 | 51.32 |
| SrO | 2.07 | 0.11 | 2.15 |
| Formulae on the basis of 1 cation | |||
| Fe | 0.002 | - | 0.002 |
| Mn | 0.004 | 0.003 | 0.004 |
| Mg | 0.002 | - | - |
| Ca | 0.957 | 0.986 | 0.972 |
| Sr | 0.020 | 0.001 | 0.022 |
| Mineral | Pyroxene | Amphibole | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sample | 13-4 | 13-2 | 13-3 | 13-3a | 13-3b | 13-4a | 13-4b | 13-2 | 13-3a | 13-3b |
| wt % | ||||||||||
| SiO2 | 49.74 | 50.99 | 48.47 | 51.16 | 51.91 | 37.90 | 37.46 | 39.33 | 39.14 | 39.28 |
| Al2O3 | 3.18 | 3.44 | 4.97 | 3.70 | 3.26 | 12.32 | 12.95 | 11.72 | 12.98 | 12.76 |
| TiO2 | 0.49 | 0.54 | 1.02 | 0.45 | 0.34 | 1.37 | 1.31 | 1.16 | 1.12 | 1.25 |
| FeO | 17.75 | 14.16 | 14.16 | 13.45 | 12.62 | 25.61 | 24.30 | 21.18 | 19.89 | 19.52 |
| MnO | 0.85 | 0.69 | 0.92 | 0.60 | 0.60 | 1.02 | 0.99 | 0.89 | 1.01 | 0.92 |
| MgO | 6.47 | 7.59 | 7.06 | 7.65 | 8.15 | 5.26 | 4.98 | 7.62 | 7.64 | 7.72 |
| CaO | 17.33 | 18.59 | 20.59 | 17.86 | 18.91 | 9.04 | 9.86 | 9.85 | 10.47 | 10.36 |
| Na2O | 4.17 | 3.59 | 2.66 | 4.33 | 3.73 | 3.75 | 3.50 | 2.85 | 3.17 | 3.32 |
| K2O | - | - | - | - | - | 2.21 | 2.24 | 1.96 | 2.28 | 2.06 |
| ZnO | - | - | - | - | - | 0.09 | 0.10 | 0.07 | 0.09 | 0.06 |
| Total | 99.98 | 99.59 | 99.85 | 99.20 | 99.51 | 98.57 | 97.66 | 96.62 | 97.80 | 97.25 |
| apfu (4 cations) | apfu (23 oxygen atoms) | |||||||||
| Si | 1.882 | 1.923 | 1.838 | 1.921 | 1.946 | 6.031 | 5.993 | 6.215 | 6.100 | 6.135 |
| Al(iv) | 0.118 | 0.077 | 0.162 | 0.079 | 0.054 | 1.969 | 2.007 | 1.785 | 1.900 | 1.865 |
| Al(vi) | 0.024 | 0.076 | 0.060 | 0.084 | 0.090 | 0.342 | 0.434 | 0.398 | 0.485 | 0.484 |
| Al(tot) | 0.142 | 0.153 | 0.222 | 0.164 | 0.144 | 2.311 | 2.442 | 2.183 | 2.385 | 2.349 |
| Ti | 0.014 | 0.015 | 0.029 | 0.013 | 0.010 | 0.164 | 0.158 | 0.138 | 0.132 | 0.146 |
| Fe3+ | 0.373 | 0.233 | 0.238 | 0.285 | 0.216 | - | - | - | - | - |
| Fe2+ | 0.189 | 0.213 | 0.211 | 0.137 | 0.180 | 3.408 | 3.251 | 2.800 | 2.592 | 2.550 |
| Mn | 0.027 | 0.022 | 0.030 | 0.019 | 0.019 | 0.138 | 0.134 | 0.119 | 0.133 | 0.122 |
| Mg | 0.365 | 0.427 | 0.399 | 0.428 | 0.455 | 1.247 | 1.187 | 1.794 | 1.774 | 1.797 |
| Ca | 0.703 | 0.751 | 0.837 | 0.718 | 0.759 | 1.541 | 1.689 | 1.667 | 1.749 | 1.733 |
| Na | 0.306 | 0.262 | 0.196 | 0.315 | 0.271 | 1.158 | 1.084 | 0.874 | 0.959 | 1.005 |
| K | - | - | - | - | - | 0.448 | 0.456 | 0.394 | 0.453 | 0.411 |
| Mineral | Garnet | Titanite | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | 13-4 C | 13-4 R | 13-4 | 13-4 | 13-4 C | 13-4 R | 13-4 C | 13-4 R | 13-1 | 13-4 | 13-4 | 13-1 |
| wt % | ||||||||||||
| SiO2 | 34.43 | 34.68 | 33.98 | 35.06 | 34.87 | 34.63 | 35.68 | 34.83 | 36.44 | 28.86 | 29.02 | 30.17 |
| Al2O3 | 4.65 | 3.39 | 3.65 | 3.56 | 4.43 | 3.52 | 5.62 | 3.51 | 3.19 | 1.11 | 1.19 | 1.11 |
| TiO2 | 3.19 | 2.88 | 2.58 | 1.53 | 2.95 | 2.06 | 1.25 | 2.63 | 2.03 | 37.23 | 38.28 | 37.19 |
| FeO | 19.95 | 21.79 | 22.25 | 22.52 | 20.25 | 22.25 | 20.65 | 21.60 | 21.36 | 1.88 | 1.58 | 1.68 |
| MnO | 1.28 | 1.41 | 1.48 | 1.20 | 1.10 | 1.25 | 1.70 | 1.35 | 1.25 | 0.08 | 0.06 | 0.05 |
| MgO | 0.34 | 0.21 | 0.17 | 0.15 | 0.27 | 0.18 | 0.14 | 0.29 | 0.15 | 0.00 | 0.00 | 0.00 |
| CaO | 32.67 | 32.87 | 31.64 | 32.76 | 32.70 | 32.19 | 32.14 | 31.63 | 32.81 | 26.79 | 27.82 | 27.92 |
| Na2O | 0.16 | - | 0.22 | - | - | 0.11 | - | - | - | 0.00 | 0.00 | 0.00 |
| ZnO | - | - | - | - | - | - | - | - | - | 0.07 | 0.00 | - |
| Y2O3 | - | 0.17 | 0.17 | - | - | - | 0.18 | 0.15 | - | 0.91 | 0.00 | - |
| ZrO2 | 0.64 | 0.29 | 0.31 | 0.35 | 0.78 | 0.42 | 0.14 | 0.76 | 0.21 | 0.69 | 0.65 | 0.53 |
| Yb2O3 | - | 0.04 | - | - | - | - | - | - | - | - | - | - |
| V2O5 | 0.06 | 0.06 | 0.06 | 0.06 | - | 0.06 | 0.04 | 0.04 | 0.10 | 0.00 | 0.00 | 0.17 |
| Nb2O5 | - | - | - | - | - | - | - | - | - | 0.37 | 0.10 | 0.63 |
| La2O3 | - | - | - | - | - | - | - | - | - | 0.12 | 0.06 | 0.26 |
| Ce2O3 | - | - | - | - | - | - | - | - | - | 0.30 | 0.26 | 0.71 |
| Nd2O3 | - | - | - | - | - | - | - | - | - | 0.45 | 0.33 | - |
| Sm2O3 | - | - | - | - | - | - | - | - | - | 0.24 | 0.12 | - |
| Gd2O3 | - | - | - | - | - | - | - | - | - | 0.36 | 0.00 | - |
| Total | 99.59 | 100.23 | 98.97 | 99.70 | 99.61 | 99.16 | 99.83 | 99.19 | 99.92 | 99.45 | 99.45 | 100.40 |
| apfu (8 cations) | apfu (3 cations) | |||||||||||
| Si | 2.862 | 2.888 | 2.863 | 2.928 | 2.906 | 2.909 | 2.949 | 2.935 | 3.031 | 0.961 | 0.956 | 0.986 |
| Al | 0.456 | 0.333 | 0.362 | 0.350 | 0.435 | 0.349 | 0.547 | 0.349 | 0.312 | 0.043 | 0.046 | 0.043 |
| Ti | 0.199 | 0.181 | 0.163 | 0.096 | 0.185 | 0.130 | 0.078 | 0.167 | 0.127 | 0.933 | 0.949 | 0.914 |
| Fe2+ | 1.387 | 1.517 | 1.568 | 1.573 | 1.412 | 1.564 | 1.428 | 1.522 | 1.486 | 0.052 | 0.043 | 0.046 |
| Mn | 0.090 | 0.100 | 0.106 | 0.085 | 0.077 | 0.089 | 0.119 | 0.096 | 0.088 | 0.002 | 0.002 | 0.001 |
| Mg | 0.042 | 0.026 | 0.021 | 0.019 | 0.034 | 0.023 | 0.018 | 0.036 | 0.019 | - | - | - |
| Ca | 2.909 | 2.933 | 2.857 | 2.931 | 2.920 | 2.898 | 2.846 | 2.855 | 2.923 | 0.956 | 0.982 | 0.978 |
| Na | 0.026 | - | 0.036 | - | - | 0.018 | - | - | - | - | - | - |
| Zn | - | - | - | - | - | - | - | - | - | 0.002 | - | - |
| Y | - | 0.008 | 0.007 | - | - | - | 0.008 | 0.007 | - | 0.016 | - | - |
| Zr | 0.026 | 0.012 | 0.013 | 0.014 | 0.032 | 0.017 | 0.006 | 0.031 | 0.008 | 0.011 | 0.010 | 0.008 |
| Nb | - | - | - | - | - | - | - | - | - | 0.006 | 0.002 | 0.009 |
| V | 0.003 | 0.003 | 0.003 | 0.003 | 0.003 | 0.002 | 0.002 | 0.006 | - | - | 0.004 | |
| La | - | - | - | - | - | - | - | - | - | 0.001 | 0.001 | 0.003 |
| Ce | - | - | - | - | - | - | - | - | - | 0.004 | 0.003 | 0.008 |
| Nd | - | - | - | - | - | - | - | - | - | 0.005 | 0.004 | - |
| Sm | - | - | - | - | - | - | - | - | - | 0.003 | 0.001 | - |
| Gd | - | - | - | - | - | - | - | - | - | 0.004 | - | - |
| Yb | - | 0.001 | - | - | - | - | - | - | - | - | - | - |
| Mineral | Natrolite | Gonnardite | Thomsonite-(Ca) | Altered Nepheline | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | 13-1 | 13-3 * | 13-3 * | 13-3 * | 13-3 * | 13-3 * | 13-3 * | 13-3 * | 13-3 * | 13-4a | 13-4 | 13-4 | 14-3 * | 14-3 * | 14-3 * | 14-3 * | 14-3 * | 13-1 | 13-1 | 13-1 |
| wt % | ||||||||||||||||||||
| SiO2 | 45.22 | 46.05 | 46.35 | 45.04 | 41.28 | 39.47 | 38.85 | 40.44 | 38.40 | 36.34 | 35.58 | 34.93 | 37.28 | 37.07 | 36.84 | 37.59 | 37.54 | 49.35 | 42.87 | 41.36 |
| Al2O3 | 20.38 | 26.46 | 27.33 | 28.36 | 30.20 | 30.50 | 30.56 | 28.89 | 30.16 | 32.06 | 31.56 | 30.36 | 29.57 | 30.23 | 30.04 | 29.54 | 29.47 | 36.67 | 42.32 | 41.09 |
| CaO | 0.08 | 0.12 | 0.31 | 1.48 | 4.17 | 5.12 | 6.43 | 4.34 | 6.25 | 13.16 | 12.19 | 7.08 | 11.36 | 12.61 | 12.08 | 12.06 | 11.14 | 0.24 | 6.05 | 5.97 |
| Na2O | 16.61 | 15.67 | 15.13 | 14.41 | 12.67 | 12.96 | 11.41 | 11.80 | 11.62 | 3.82 | 3.75 | 3.68 | 4.09 | 4.38 | 4.35 | 5.07 | 4.57 | 14.57 | 10.86 | 10.80 |
| K2O | - | 0.02 | 0.03 | 0.02 | 0.03 | 0.04 | 0.02 | 0.00 | 0.02 | - | - | - | - | - | - | - | - | - | - | - |
| SrO | - | - | - | - | - | - | - | - | - | 0.73 | 2.69 | 11.76 | 3.21 | 1.75 | 2.14 | 1.80 | 1.71 | - | - | - |
| Total | 82.29 | 88.32 | 89.15 | 89.31 | 88.36 | 88.09 | 87.28 | 85.47 | 86.44 | 86.12 | 85.78 | 87.81 | 85.51 | 86.05 | 85.45 | 86.07 | 84.43 | 100.83 | 102.09 | 99.22 |
| apfu (7 cations) | apfu (7 cations) | apfu (13 cations) | apfu (3 cations) | |||||||||||||||||
| Si | 3.118 | 2.991 | 2.997 | 2.919 | 2.727 | 2.605 | 2.617 | 2.775 | 2.605 | 4.932 | 4.940 | 5.176 | 5.220 | 5.070 | 5.092 | 5.116 | 5.225 | 1.223 | 1.069 | 1.059 |
| Al | 1.656 | 2.026 | 2.083 | 2.166 | 2.352 | 2.372 | 2.426 | 2.336 | 2.411 | 5.129 | 5.164 | 5.302 | 4.878 | 4.874 | 4.894 | 4.739 | 4.835 | 1.071 | 1.244 | 1.240 |
| Ca | 0.006 | 0.008 | 0.021 | 0.103 | 0.295 | 0.362 | 0.464 | 0.319 | 0.454 | 1.914 | 1.813 | 1.123 | 1.705 | 1.848 | 1.790 | 1.759 | 1.662 | 0.006 | 0.162 | 0.164 |
| Na | 2.220 | 1.974 | 1.897 | 1.810 | 1.622 | 1.658 | 1.490 | 1.570 | 1.528 | 1.005 | 1.009 | 1.058 | 1.109 | 1.161 | 1.166 | 1.338 | 1.232 | 0.700 | 0.525 | 0.537 |
| K | - | 0.002 | 0.002 | 0.002 | 0.003 | 0.003 | 0.002 | 0.000 | 0.002 | - | - | - | - | - | - | - | - | - | - | - |
| Sr | - | - | - | - | - | - | - | - | - | 0.019 | 0.073 | 0.341 | 0.088 | 0.047 | 0.058 | 0.048 | 0.047 | - | - | - |
| Sample | 13_1 | 13_2 | 13_3 | 13_4 | 13_5 | B9.5 | B19.4 | B21.4 | B11.1 | H6.2 | H40.3 | H10.7 | H41.3 | H11.6 | H15.5 | H31.3 | H48.2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rock | Carbonatite | “Malignite” | |||||||||||||||
| SiO2 | 31.74 | 26.4 | 32.1 | 28.97 | 26.51 | 31.54 | 28.67 | 28.96 | 29.76 | 20.47 | 34.95 | 37.1 | 37.46 | 37.53 | 37.71 | 39.18 | 39.43 |
| TiO2 | 1.73 | 1.1 | 1.86 | 1.89 | 1.51 | 0.87 | 0.99 | 0.98 | 1.62 | 1.21 | 2.01 | 1.94 | 1.59 | 1.61 | 1.39 | 1.49 | 0.83 |
| Al2O3 | 11.7 | 3.35 | 11.27 | 14.9 | 2.46 | 13.11 | 13.82 | 13.63 | 11.71 | 6.87 | 9.72 | 11.89 | 12.91 | 12.66 | 13.74 | 13.05 | 17.35 |
| Fe2O3 | 6.12 | 8.92 | 6.93 | 9.69 | 5.25 | 5.46 | 4.41 | 4.37 | 5.19 | 9.62 | 13.59 | 12.39 | 10.85 | 10.39 | 10.23 | 10.27 | 6.73 |
| FeO | 7 | 6.19 | 5.74 | 5.48 | 3.63 | 4.62 | 3.46 | 3.43 | 3.64 | 6.39 | 2.79 | 2.55 | 2.96 | 3.5 | 2.86 | 2.89 | 1.16 |
| MnO | 0.52 | 0.57 | 0.56 | 0.45 | 0.36 | 0.5 | 0.43 | 0.44 | 0.47 | 0.69 | 1.19 | 0.99 | 0.91 | 0.91 | 0.84 | 0.87 | 0.55 |
| MgO | 2.22 | 3.98 | 2.17 | 1.1 | 4.22 | 2.33 | 1.99 | 2.13 | 2.08 | 1.49 | 0.77 | 0.49 | 1.02 | 1.08 | 0.82 | 0.92 | 0.24 |
| CaO | 20.46 | 30.22 | 21.49 | 19.98 | 33.6 | 21.32 | 22.54 | 22.5 | 24.16 | 30.41 | 26.42 | 23.08 | 21.28 | 21.41 | 20.41 | 20.16 | 17.12 |
| Na2O | 4.08 | 1.8 | 4.52 | 3.1 | 1.78 | 4.77 | 5.82 | 5.75 | 5.13 | 2.44 | 2.04 | 2.94 | 3.82 | 3.53 | 3.97 | 3.74 | 5.97 |
| K2O | 1.1 | 0.41 | 0.67 | 0.41 | 0.27 | 0.56 | 0.33 | 0.33 | 0.29 | 0.27 | 0.45 | 0.77 | 0.66 | 0.77 | 0.77 | 1.16 | 1.15 |
| H2O | 0.89 | 0.46 | 0.91 | 0.64 | 0.4 | - | - | - | - | - | - | - | - | - | - | - | - |
| LOI | 2.94 | 0.26 | 3.83 | 5.35 | 2.06 | - | - | - | - | - | - | - | - | - | - | - | - |
| P2O5 | 0.45 | 0.95 | 0.52 | 0.27 | 1.44 | 0.76 | 0.78 | 0.79 | 0.63 | 0.68 | 0.23 | 0.20 | 0.26 | 0.26 | 0.33 | 0.25 | 0.17 |
| F | 0.068 | 0.086 | 0.067 | 0.042 | 0.1 | - | - | - | - | - | - | - | - | - | - | - | - |
| Cl | 0.011 | 0.011 | 0.029 | 0.014 | 0.02 | - | - | - | - | - | - | - | - | - | - | - | - |
| CO2 | 7.91 | 13.17 | 6.57 | 6.07 | 15.35 | 9.65 | 10.72 | 10.57 | 10.41 | 15.74 | 1.39 | 0.84 | 1.49 | 1.59 | 1.6 | 1.42 | 2.96 |
| Total | 98.94 | 97.88 | 99.24 | 98.36 | 98.96 | 95.49 | 93.96 | 93.88 | 95.09 | 96.28 | 95.55 | 95.18 | 95.21 | 95.24 | 94.67 | 95.40 | 93.66 |
| Sample | 13_1 | 13_2 | 13_3 | 13_4 | 13_5 | B19.4 | B21.4 | B11.1 | H6.2 | H40.3 | H10.7 | H41.3 | H11.6 | H31.3 | H48.2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rock | Carbonatite | “Malignite” | |||||||||||||
| La | 214.4 | 265.7 | 190.5 | 150.4 | 470.2 | - | - | - | - | - | - | - | - | - | - |
| Ce | 449.7 | 516.8 | 421.8 | 341.1 | 930.9 | - | - | - | - | - | - | - | - | - | - |
| Pr | 49.2 | 51.8 | 47.6 | 39 | 96.4 | - | - | - | - | - | - | - | - | - | - |
| Nd | 189.6 | 174.4 | 209.4 | 169.7 | 324.2 | - | - | - | - | - | - | - | - | - | - |
| Sm | 40 | 25.3 | 49.4 | 42.4 | 56.2 | - | - | - | - | - | - | - | - | - | - |
| Eu | 12.1 | 6.66 | 16 | 13.9 | 15.1 | - | - | - | - | - | - | - | - | - | - |
| Gd | 29.7 | 19.8 | 35.7 | 31.1 | 42.5 | - | - | - | - | - | - | - | - | - | - |
| Tb | 4.86 | 2.2 | 6.87 | 6.06 | 5.77 | - | - | - | - | - | - | - | - | - | - |
| Dy | 24.5 | 7.83 | 35.4 | 31.9 | 23.7 | - | - | - | - | - | - | - | - | - | - |
| Ho | 4.57 | 1.26 | 6.79 | 6.32 | 4.12 | - | - | - | - | - | - | - | - | - | - |
| Er | 12.5 | 2.95 | 18.9 | 17.2 | 9.17 | - | - | - | - | - | - | - | - | - | - |
| Tm | 1.84 | 0.37 | 2.8 | 2.58 | 1.14 | - | - | - | - | - | - | - | - | - | - |
| Yb | 12.7 | 2.59 | 19.2 | 18.3 | 7.14 | - | - | - | - | - | - | - | - | - | - |
| Lu | 1.87 | 0.43 | 2.88 | 2.72 | 0.97 | - | - | - | - | - | - | - | - | - | - |
| Y | 111.7 | 30.8 | 149.8 | 148.3 | 84 | 30 | 32 | 124 | 28 | 43 | 60 | 46 | 44 | 42 | 14 |
| Ta | 3.23 | 0.8 | 3.27 | 4.19 | 18 | - | - | - | - | - | - | - | - | - | - |
| Nb | 111.2 | 20.7 | 106.3 | 124 | 231.4 | 188 | 187 | 136 | 42 | 88 | 103 | 78 | 85 | 92 | 34 |
| Hf | 13.7 | 8.05 | 19.9 | 17.9 | 9.84 | - | - | - | - | - | - | - | - | - | |
| Zr | 492.5 | 363.2 | 696.7 | 716 | 473.1 | 493 | 505 | 840 | 616 | 1359 | 1350 | 1133 | 1137 | 1145 | 1043 |
| Sr | 4735 | 5803 | 3290 | 8856 | 4656 | 6169 | 6173 | 3780 | 8985 | 645 | 958 | 1211 | 1235 | 1505 | 3993 |
| Rb | 16.4 | 5.82 | 10 | 5.23 | 6.41 | bd | bd | bd | bd | 8 | 10 | 6 | 8 | 12 | 9 |
| Ba | 618.5 | 638.4 | 204.7 | 178.2 | 1003 | - | - | - | - | - | - | - | - | - | - |
| U | 3.46 | 0.8 | 3.99 | 3.13 | 1.86 | - | - | - | - | - | - | - | - | - | - |
| Th | 12.7 | 5.63 | 13.4 | 9.43 | 25.8 | - | - | - | - | - | - | - | - | - | - |
| Pb | 4.17 | 1.86 | 3.53 | 3.2 | 2.81 | - | - | - | - | - | - | - | - | - | - |
| Mo | 5.02 | 1.1 | 8.9 | 13.8 | 0.62 | - | - | - | - | - | - | - | - | - | - |
| Cs | 0.16 | 0.016 | 0.11 | 0.02 | 0.091 | - | - | - | - | - | - | - | - | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zozulya, D.R.; Kullerud, K.; Ravna, E.K.; Savchenko, Y.E.; Selivanova, E.A.; Timofeeva, M.G. Mineralogical and Geochemical Constraints on Magma Evolution and Late-Stage Crystallization History of the Breivikbotn Silicocarbonatite, Seiland Igneous Province in Northern Norway: Prerequisites for Zeolite Deposits in Carbonatite Complexes. Minerals 2018, 8, 537. https://doi.org/10.3390/min8110537
Zozulya DR, Kullerud K, Ravna EK, Savchenko YE, Selivanova EA, Timofeeva MG. Mineralogical and Geochemical Constraints on Magma Evolution and Late-Stage Crystallization History of the Breivikbotn Silicocarbonatite, Seiland Igneous Province in Northern Norway: Prerequisites for Zeolite Deposits in Carbonatite Complexes. Minerals. 2018; 8(11):537. https://doi.org/10.3390/min8110537
Chicago/Turabian StyleZozulya, Dmitry R., Kåre Kullerud, Erling K. Ravna, Yevgeny E. Savchenko, Ekaterina A. Selivanova, and Marina G. Timofeeva. 2018. "Mineralogical and Geochemical Constraints on Magma Evolution and Late-Stage Crystallization History of the Breivikbotn Silicocarbonatite, Seiland Igneous Province in Northern Norway: Prerequisites for Zeolite Deposits in Carbonatite Complexes" Minerals 8, no. 11: 537. https://doi.org/10.3390/min8110537
APA StyleZozulya, D. R., Kullerud, K., Ravna, E. K., Savchenko, Y. E., Selivanova, E. A., & Timofeeva, M. G. (2018). Mineralogical and Geochemical Constraints on Magma Evolution and Late-Stage Crystallization History of the Breivikbotn Silicocarbonatite, Seiland Igneous Province in Northern Norway: Prerequisites for Zeolite Deposits in Carbonatite Complexes. Minerals, 8(11), 537. https://doi.org/10.3390/min8110537





