Critical Metal Particles in Copper Sulfides from the Supergiant Río Blanco Porphyry Cu–Mo Deposit, Chile
Abstract
:1. Introduction
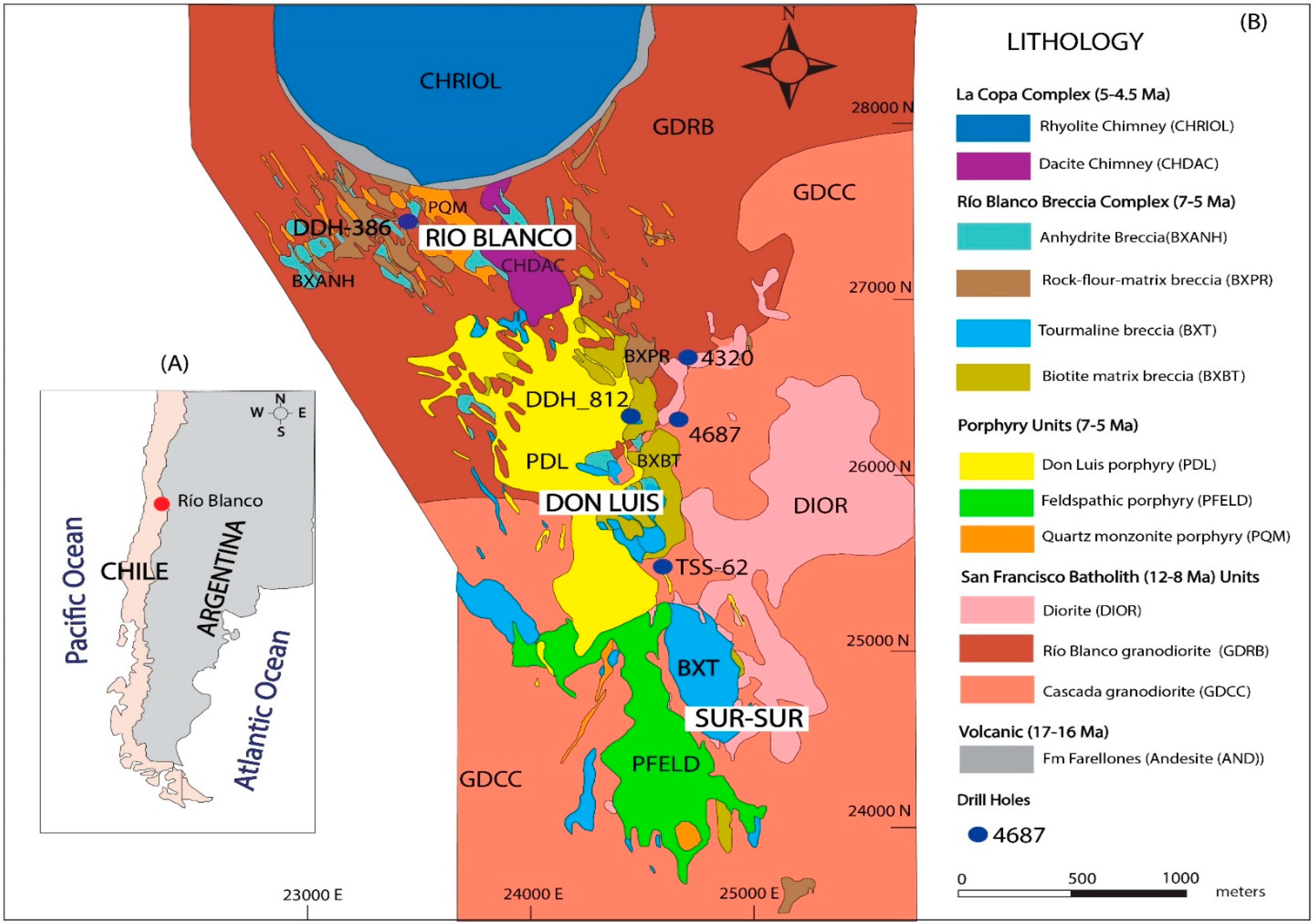
2. Geology of the Río Blanco Deposit
3. Samples and Methods
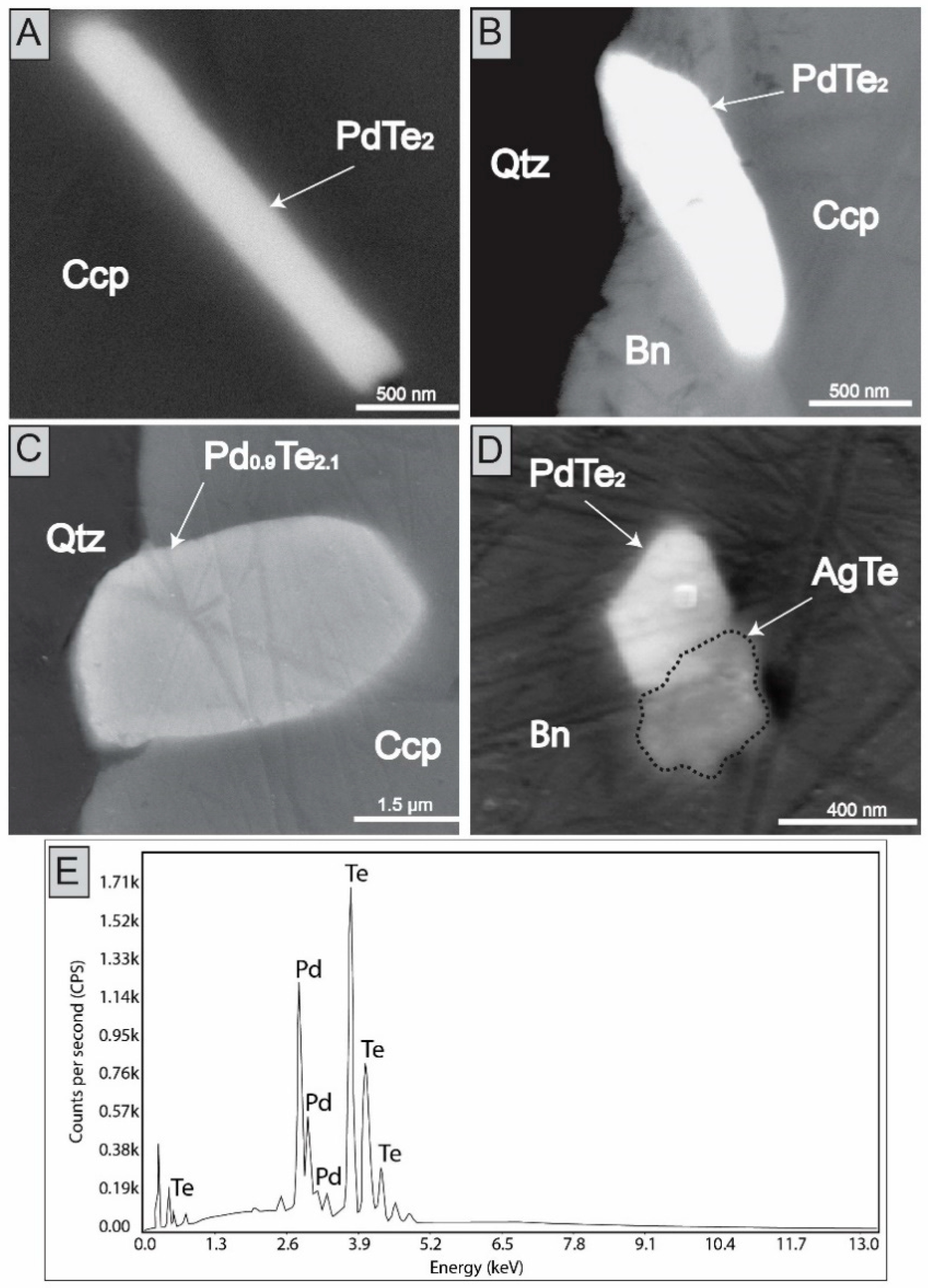
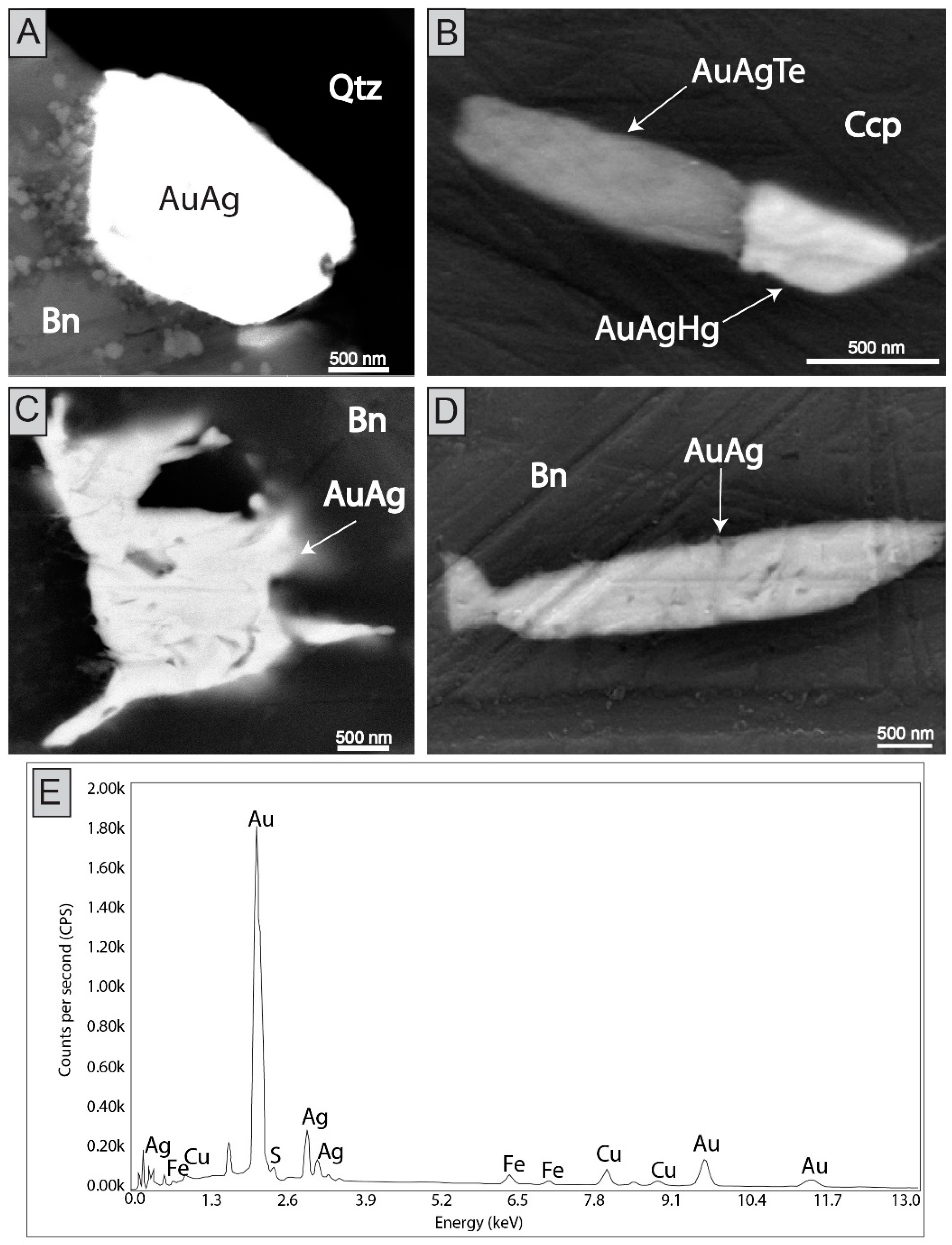
4. Results
5. Discussion
6. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnson, K.M.; Hammarstrom, J.M.; Zientek, M.L.; Dicken, C.L. Estimate of Undiscovered Copper Resources of the World, 2013. USGS Fact Sheet 2014, 3004, 3. [Google Scholar] [CrossRef]
- US Geological Survey 2018. Mineral Commodity Summaries; U.S. Geological Survey: Reston, VA, USA, 2018; 200p.
- Einaudi, M.T.; Burt, D.M. A Special Issue Devoted to Skarn Deposits. Introduction-Terminology, Classification, and Composition of Skarn Deposits. Econ. Geol. 1982, 77, 745–754. [Google Scholar] [CrossRef]
- Tarkian, M.; Stribrny, B. Platinum-group elements in porphyry copper deposits: A reconnaissance study. Miner. Petrol. 1999, 65, 161–183. [Google Scholar] [CrossRef]
- Economou-Eliopoulos, M.; Eliopoulos, D.G. Palladium, platinum and gold concentration in porphyry copper systems of Greece and their genetic significance. Ore Geol. Rev. 1999, 16, 59–70. [Google Scholar] [CrossRef]
- Simon, G.; Kesler, S.E.; Essene, E.J. Gold in Porphyry Copper Deposits: Experimental Determination of the Distribution of Gold in the Cu–Fe–S System at 400 to 700 °C. Econ. Geol. 2000, 95, 259–270. [Google Scholar] [CrossRef]
- Kesler, S.E.; Chryssoulis, S.L.; Simon, G. Gold in porphyry copper deposits: Its abundance and fate. Ore Geol. Rev. 2002, 21, 103–124. [Google Scholar] [CrossRef]
- Economou-Eliopoulos, M. Platinum-group element potential of porphyry deposits. Min. Assoc. Can. Short Course 2005, 35, 203–245. [Google Scholar]
- Pašava, J.; Vymazalova, A.; Kosler, J.; Koneev, R.; Jukov, A.V.; Khalmatov, R.A. Platinum-group elements in ores from the Kalmakyr porphyry Cu–Au–Mo deposit, Uzbekistan: Bulk geochemical and laser ablation ICP–MS data. Miner. Depos. 2010, 45, 411–418. [Google Scholar] [CrossRef]
- McFall, K.A.; Roberts, S.; Teagle, D.; Naden, J.; Lusty, P.; Boyce, A. The origin and distribution of critical metals (Pd, Pt, Te and Se) within the Skouries Cu–Au porphyry deposit, Greece. Appl. Earth Sci. 2016, 125, 100–101. [Google Scholar] [CrossRef]
- McFall, K.A.; Naden, J.; Roberts, S.; Baker, T.; Spratt, J.; McDonald, I. Platinum-group minerals in the Skouries Cu–Au (Pd, Pt, Te) porphyry deposit. Ore Geol. Rev. 2018, 99, 344–364. [Google Scholar] [CrossRef]
- U.S. Departament of Energy. Critical Materials Strategy, December 2011. Available online: https://www.energy.gov/sites/prod/files/DOE_CMS2011_FINAL_Full.pdf (accessed on 10 June 2018).
- Geological Society of America. GSA Position Statement 2013 Critical Mineral and Materials. Available online: http://www.geosociety.org/gsa/positions/position23.aspx (accessed on 20 June 2018).
- Arndt, N.T.; Fontboté, L.; Hedenquist, J.W.; Kesler, S.E.; Thompson, J.F.H.; Wood, D.G. Future global mineral resources. Geochem. Perspect. 2017, 6, 1–171. [Google Scholar] [CrossRef]
- Huston, D.L.; Sie, S.H.; Suter, G.F.; Cooke, D.R.; Both, R.A. Trace Elements in sulfide minerals from eastern australian volcanic-hosted massive sulfide deposits: Part I. proton microprobe analyses of pyrite, chalcopyrite, and sphalerite, and Part II. selenium levels in pyrite: Comparison with (δ34S values and implications for the source of sulfer in volcanogenic hydrothermal system. Econ. Geol. 1995, 90, 1167–1196. [Google Scholar] [CrossRef]
- Kesler, S.E.; Russell, N.; Mccurdy, K. Trace-metal content of the Pueblo Viejo precious-metal deposits and their relation to other high-sulfidation epithermal systems. Miner. Depos. 2003, 38, 668–682. [Google Scholar] [CrossRef]
- Large, R.R.; Danyushevsky, L.; Hollit, C.; Maslennikov, V.; Meffre, S.; Gilbert, S.; Bull, S.; Scott, R.; Emsbo, P.; Thomas, H.; et al. Gold and trace element zonation in pyrite using a laser imaging technique: Implications for the timing of gold in orogenic and carlin-style sediment-hosted deposits. Econ. Geol. 2009, 104, 635–668. [Google Scholar] [CrossRef]
- Deditius, A.P.; Utsunomiya, S.; Reich, M.; Kesler, S.E.; Ewing, R.C.; Hough, R.; Walshe, J. Trace metal nanoparticles in pyrite. Ore Geol. Rev. 2011, 42, 32–46. [Google Scholar] [CrossRef]
- Reich, M.; Deditius, A.; Chryssoulis, S.; Li, J.-W.; Ma, C.-Q.; Parada, M.A.; Barra, F.; Mittermayr, F. Pyrite as a record of hydrothermal fluid evolution in a porphyry copper system: A SIMS/EMPA trace element study. Geochim. Cosmochim. Acta 2013, 104, 42–62. [Google Scholar] [CrossRef]
- Cioacǎ, M.E.; Munteanu, M.; Qi, L.; Costin, G. Trace element concentrations in porphyry copper deposits from Metaliferi Mountains, Romania: A reconnaissance study. Ore Geol. Rev. 2014, 63, 22–39. [Google Scholar] [CrossRef]
- Franchini, M.; Mcfarlane, C.; Maydagán, L.; Reich, M.; Lentz, D.R.; Meinert, L.; Bouhier, V. Trace metals in pyrite and marcasite from the Agua Rica porphyry-high sulfidation epithermal deposit, Catamarca, Argentina: Textural features and metal zoning at the porphyry to epithermal transition. Ore Geol. Rev. 2015, 66, 366–387. [Google Scholar] [CrossRef]
- Barra, F.; Deditius, A.; Reich, M.; Kilburn, M.; Guagliardo, P.; Roberts, M.P. Dissecting the Re–Os molybdenite geochronometer. Sci. Rep. 2017, 7, 16054. [Google Scholar] [CrossRef] [PubMed]
- Zarasvandi, A.; Rezaei, M.; Raith, J.G.; Pourkaseb, H.; Asadi, S.; Saed, M.; Lentz, D.R. Metal endowment reflected in chemical composition of silicates and sulfides of mineralized porphyry copper systems, Urumieh-Dokhtar magmatic arc, Iran. Geochim. Cosmochim. Acta 2018, 223, 36–59. [Google Scholar] [CrossRef]
- Singer, D.A.; Berger, V.I.; Moring, B.C. Porphyry Copper Deposits of the World: Database and Grade and Tonnage Models, 2008; U.S. Geological Survey: Reston, VA, USA, 2008. Available online: https://pubs.usgs.gov/of/2008/1155/of2008-1155.pdf (accessed on 10 June 2018).
- Sinclair, W.D. Porphyry deposits, in Goodfellow W, ed., Mineral Deposits of Canada: A synthesis of major deposit-types, district metallogeny, the evolution of geological provinces, and exploration methods. Geol. Assoc. Can. Miner. Depos. Div. 2007, 5, 223–243. [Google Scholar]
- Reich, M.; Chryssoulis, S.L.; Deditius, A.; Palacios, C.; Zuniga, A.; Weldt, M.; Alvear, M. “Invisible” silver and gold in supergene digenite (Cu1.8S). Geochim. Cosmochim. Acta 2010, 74, 6157–6173. [Google Scholar] [CrossRef]
- Reich, M.; Palacios, C.; Barra, F.; Chryssoulis, S. “Invisible” silver in chalcopyrite and bornite from the Mantos Blancos Cu deposit, northern Chile. Eur. J. Mineral. 2013, 25, 453–460. [Google Scholar] [CrossRef]
- Hanley, J.J.; MacKenzie, M. Incorporation of platinum-group elements and cobalt into subsidiary pyrite in alkalic Cu–Au porphyry deposits: Significant implications for precious metal distribution in felsic magmatic-hydrothermal systems. In AGU Spring Meeting Abstracts; American Geophysical Union: Washington, DC, USA, 2009. [Google Scholar]
- Ferraz, G.; Cruz, J. Inventario de Recursos Geológicos 2011, División Andina Superintendencia de Geología Gerencia de Recursos Mineros y Desarrollo; Informe Interno; CODELCO: Santiago, Chile, 2011. [Google Scholar]
- Rivano, S.; Godoy, E.; Vergara, M.; Villaroel, R. Redefinición de la formacion farellones en la cordillera de los andes de chile central (32–34° S). Rev. Geol. Chile 1990, 17, 205–214. [Google Scholar]
- Deckart, K.; Clark, A.H.; Aguilar, A.C.; Vargas, R.R.; Bertens, A.N.; Mortensen, J.K.; Fanning, M. Magmatic and hydrothermal chronology of the giant Río Blanco porphyry copper deposit, central Chile: Implications of an integrated U–Pb and 40Ar/39Ar database. Econ. Geol. 2005, 100, 905–934. [Google Scholar] [CrossRef]
- Thiele, R.; Cubillos, E. Hoja Santiago: Región Metropolitana: Carta Geológica de Chile Escala 1:250.000; Instituto de Investigaciones Geológicas: Santiago, Chile, 1980. [Google Scholar]
- Stambuck, V.; Blondel, J.; Serrano, L. Geología del Yacimiento Río Blanco. Congr. Geol. Chileno Actas 1982, 2, 419–442. [Google Scholar]
- Toro, J.C.; Ortúzar, J.; Zamorano, J.; Cuadra, P.; Juan, H.; Spröhnle, C. Protracted Magmatic-Hydrothermal History of the Río Blanco-Los Bronces District, Central Chile: Development of World’s Greatest Known Concentration of Copper. Soc. Econ. Geol. Spec. Publ. 2012, 16, 105–126. [Google Scholar] [CrossRef]
- Piquer, J.; Skarmeta, J.; Cooke, D.R. Structural Evolution of the Río Blanco-Los Bronces District, Andes of Central Chile: Controls on Stratigraphy, Magmatism, and Mineralization. Econ. Geol. 2012, 110, 1995–2023. [Google Scholar] [CrossRef]
- Salinero, J. Eventos de Alteración y Mineralización en el Sector Río Blanco, Yacimiento Río Blanco, V Región, Chile. Bachelor’s Thesis, Universidad de Chile, Santiago, Chile, 2004. [Google Scholar]
- Xiong, Y.; Wood, S.A. Experimental quantification of hydrothermal solubility of platinum-group elements with special reference to porphyry copper environments. Miner. Petrol. 2000, 68, 1–28. [Google Scholar] [CrossRef]
- Sullivan, N.A.; Zajacz, Z.; Brenan, J.M. The solubility of Pd and Au in hydrous intermediate silicate melts: The effect of oxygen fugacity and the addition of Cl and S. Geochim. Cosmochim. Acta 2018, 231, 15–29. [Google Scholar] [CrossRef]
- Hanley, J.J. The aqueous geochemistry of the platinum-group elements (PGE) in surficial, low-T Hydrothermal and High-T magmatic-hydrothermal environments. In Exploration for Platinum-Group Elements Deposits; Mungall, J.E., Ed.; Mineralogical Association of Canada: Québec, QC, Canada, 2005; Volume 35, pp. 35–56. [Google Scholar]
- Gammons, C.H. Experimental investigations of the hydrothermal geochemistry of platinum and palladium: IV. The stoichiometry of Pt(IV) and Pd(II) chloride complexes at 100 to 300 °C. Geochim. Cosmochim. Acta 1995, 59, 1655–1667. [Google Scholar] [CrossRef]
- Gammons, C.H. Experimental investigations of the hydrothermal geochemistry of platinum and palladium: V. Equilibria between Pt metal, Pt(II) and Pt(IV) chloride complexes at 25 to 300 °C. Geochim. Cosmochim. Acta 1996, 60, 1683–1694. [Google Scholar] [CrossRef]
- Sillitoe, R.H. Porphyry copper systems. Econ. Geol. 2010, 105, 3–41. [Google Scholar] [CrossRef]
- Kouzmanov, K.; Pokrovski, G.S. Hydrotermal controls on metal distribution in Porphyry Cu(–Mo–Au) Systems. Soc. Econ. Geol. Spec. Publ. 2012, 16, 573–618. [Google Scholar]
- McFall, K.; Roberts, S.; Naden, J.; Wilkinson, C.; Wilkinson, J.; Boyce, A. Hydrothermal transport of PGEs in porphyry systems—A fluid history of the Skouries Cu–Au (PGE) porphyry deposit. Appl. Earth Sci. 2017, 126, 79–80. [Google Scholar] [CrossRef]
| Element | Cu | Fe | S | Au | Ag | Bi | Hg | Te | Se | Zn | As | Pb | Sb | Co | Ni | Total | |
| mdl | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.03 | 0.03 | 0.01 | 0.01 | 0.01 | 0.02 | 0.02 | 0.01 | 0.02 | 0.01 | ||
| Sample DDH386-564 | |||||||||||||||||
| C1-Cpy2 | 35.32 | 29.10 | 34.11 | b.d | b.d | b.d | 0.10 | 0.03 | b.d | b.d | 0.02 | 0.05 | b.d | b.d | b.d | 98.78 | |
| C1-Cpy3 | 35.17 | 29.42 | 34.09 | b.d | b.d | b.d | b.d | 0.03 | b.d | b.d | b.d | 0.03 | b.d | 0.04 | b.d | 98.78 | |
| C4-Cpy10 | 35.24 | 29.43 | 34.33 | b.d | b.d | 0.06 | b.d | b.d | b.d | b.d | b.d | b.d | b.d | b.d | b.d | 99.07 | |
| C4-Cpy11 | 35.31 | 29.32 | 34.26 | 0.08 | b.d | b.d | b.d | b.d | 0.02 | b.d | b.d | b.d | b.d | 0.04 | 0.01 | 99.05 | |
| C4-Cpy12 | 35.38 | 29.42 | 34.46 | b.d | b.d | b.d | 0.12 | b.d | 0.02 | b.d | b.d | 0.07 | b.d | b.d | b.d | 99.49 | |
| C3-Cpy13 | 35.55 | 29.88 | 34.26 | b.d | 0.04 | 0.07 | 0.03 | 0.02 | 0.03 | b.d | 0.03 | 0.03 | b.d | 0.06 | b.d | 100.00 | |
| C3-Cpy14 | 35.69 | 29.84 | 34.38 | b.d | b.d | 0.05 | 0.09 | b.d | 0.02 | b.d | 0.06 | 0.04 | b.d | 0.02 | b.d | 100.19 | |
| C4-Bn8 | 63.85 | 10.99 | 25.55 | b.d | 0.14 | b.d | 0.10 | b.d | b.d | b.d | b.d | b.d | b.d | 0.02 | b.d | 100.65 | |
| C4-Bn9 | 63.90 | 11.09 | 25.48 | b.d | 0.13 | b.d | 0.05 | 0.03 | b.d | b.d | 0.04 | b.d | b.d | b.d | b.d | 100.74 | |
| C3-Bn15 | 62.67 | 11.62 | 25.76 | b.d | 0.04 | 0.06 | b.d | b.d | b.d | b.d | 0.08 | 0.07 | b.d | b.d | b.d | 100.30 | |
| C3-Bn16 | 62.88 | 11.27 | 25.93 | b.d | 0.06 | b.d | 0.09 | b.d | 0.02 | b.d | 0.13 | b.d | b.d | 0.02 | 0.01 | 100.42 | |
| C3-Bn17 | 62.47 | 11.28 | 26.02 | 0.05 | 0.06 | 0.07 | 0.10 | b.d | b.d | b.d | 0.12 | b.d | b.d | 0.02 | b.d | 100.22 | |
| Element | Cu | Fe | S | Au | Ag | Bi | Te | Pd | Pt | Re | As | Rh | Ru | Ir | Os | Ni | Total |
| mdl | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.06 | 0.03 | 0.03 | 0.04 | 0.03 | 0.02 | 0.02 | 0.02 | 0.03 | 0.03 | 0.02 | |
| Sample DDH386-564 | |||||||||||||||||
| C4-PdTe8 | 2.75 | 1.83 | 1.56 | 0.13 | b.d | 0.35 | 65.34 | 23.69 | 1.32 | b.d | b.d | b.d | b.d | b.d | 0.12 | b.d | 97.11 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crespo, J.; Reich, M.; Barra, F.; Verdugo, J.J.; Martínez, C. Critical Metal Particles in Copper Sulfides from the Supergiant Río Blanco Porphyry Cu–Mo Deposit, Chile. Minerals 2018, 8, 519. https://doi.org/10.3390/min8110519
Crespo J, Reich M, Barra F, Verdugo JJ, Martínez C. Critical Metal Particles in Copper Sulfides from the Supergiant Río Blanco Porphyry Cu–Mo Deposit, Chile. Minerals. 2018; 8(11):519. https://doi.org/10.3390/min8110519
Chicago/Turabian StyleCrespo, Jorge, Martin Reich, Fernando Barra, Juan José Verdugo, and Claudio Martínez. 2018. "Critical Metal Particles in Copper Sulfides from the Supergiant Río Blanco Porphyry Cu–Mo Deposit, Chile" Minerals 8, no. 11: 519. https://doi.org/10.3390/min8110519
APA StyleCrespo, J., Reich, M., Barra, F., Verdugo, J. J., & Martínez, C. (2018). Critical Metal Particles in Copper Sulfides from the Supergiant Río Blanco Porphyry Cu–Mo Deposit, Chile. Minerals, 8(11), 519. https://doi.org/10.3390/min8110519






