On the Color and Genesis of Prase (Green Quartz) and Amethyst from the Island of Serifos, Cyclades, Greece
Abstract
1. Introduction
2. Materials and Methods
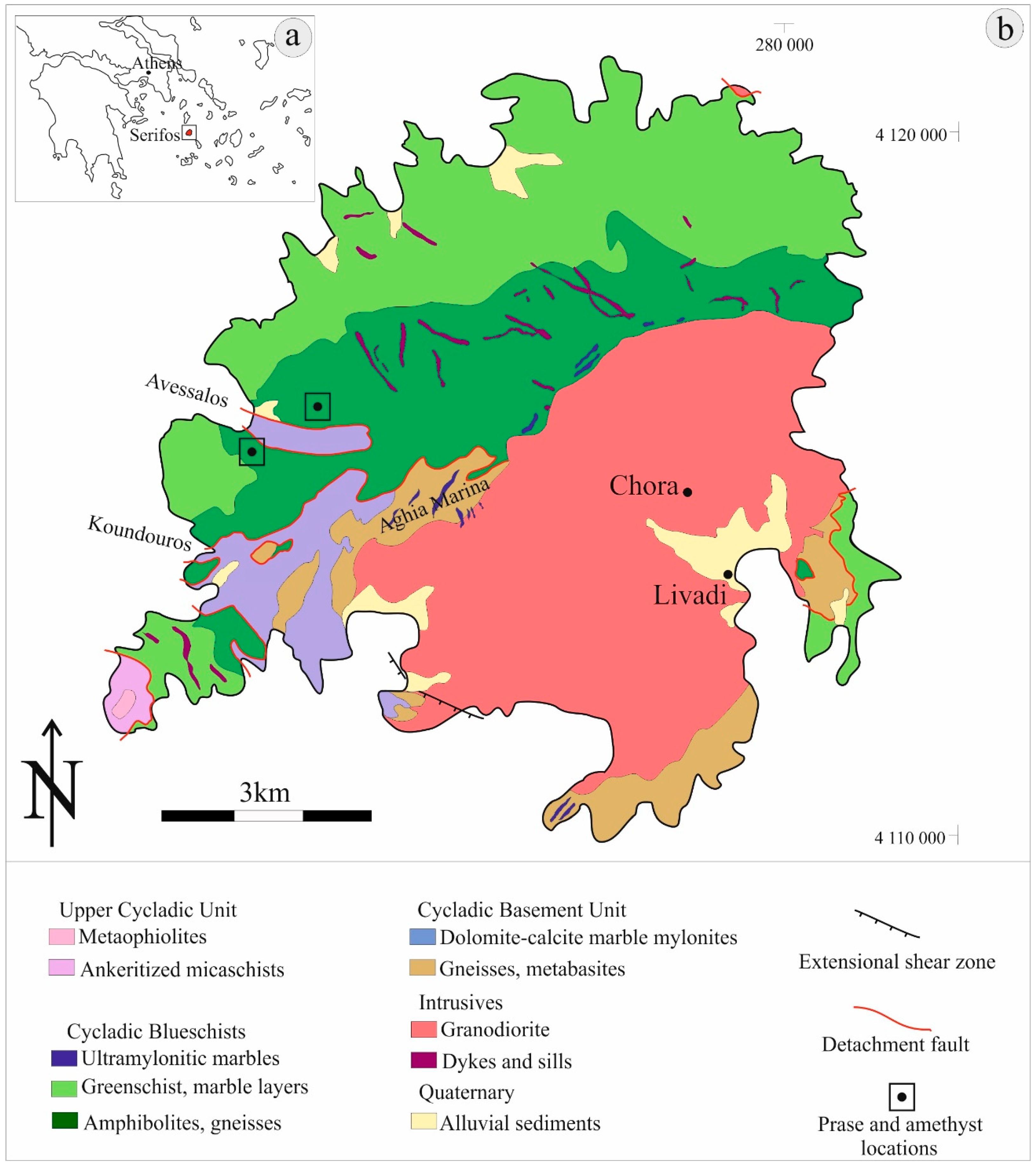
3. Geological Setting
4. Results
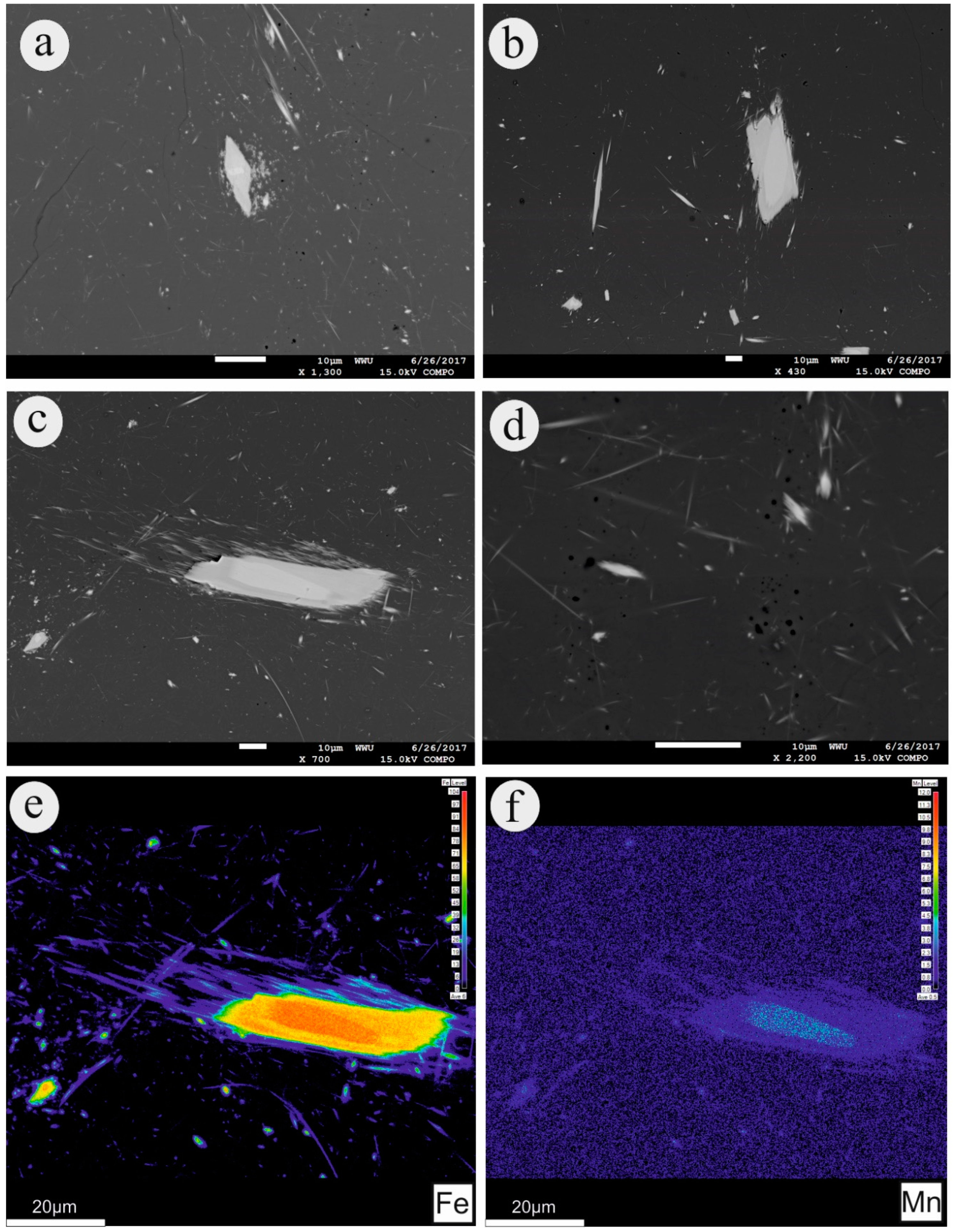
4.1. Mineral Chemistry
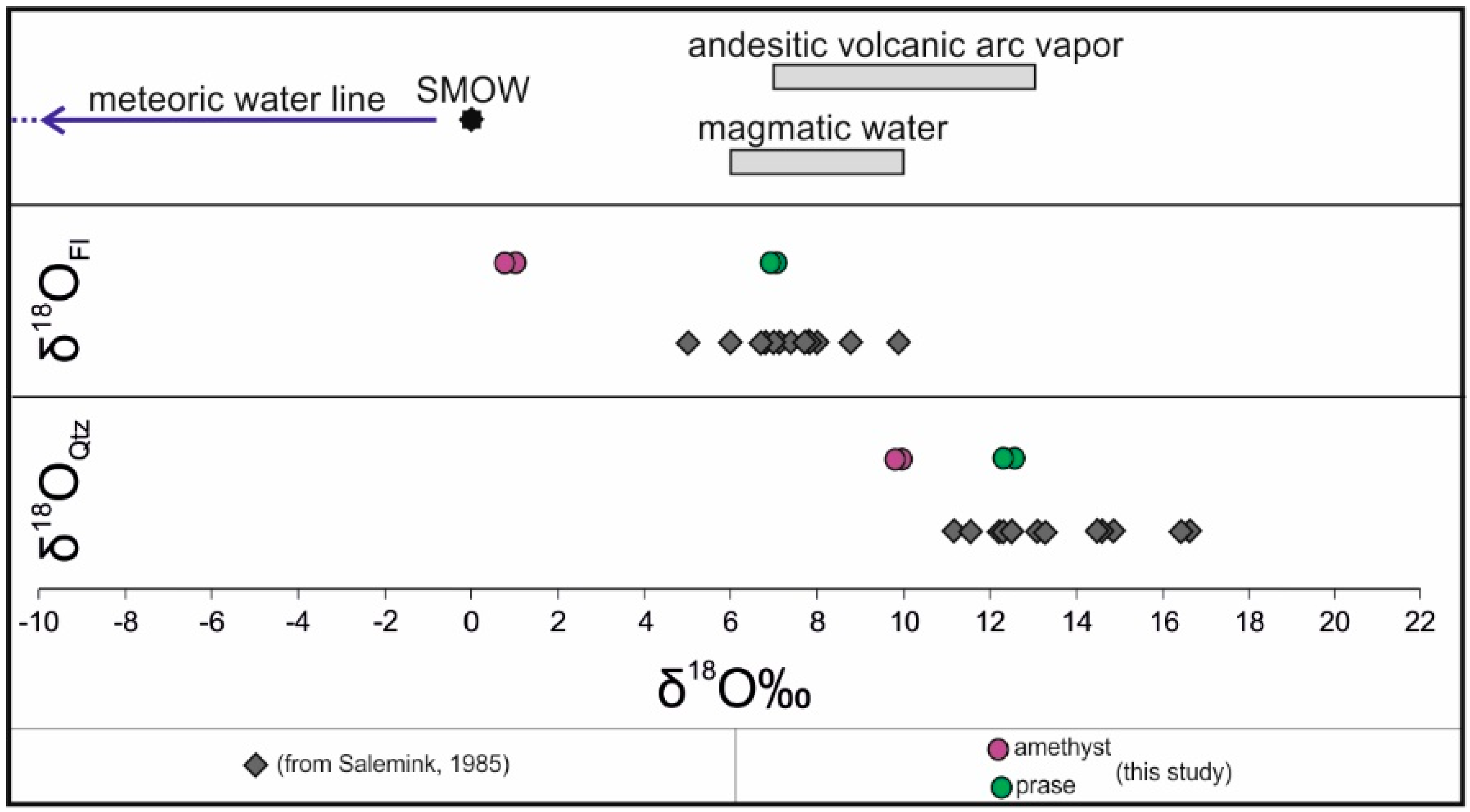
4.2. Quartz Oxygen Isotopes
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hutton, D. Defects in the coloured varieties of quartz. J. Gemmol. 1974, 14, 156–166. [Google Scholar] [CrossRef]
- Lehmann, G.; Bambauer, H.U. Quarzkristalle und ihre Farben. Angew. Chem. 1973, 85, 281–289. [Google Scholar] [CrossRef]
- Rossman, G.R. Coloured Varieties of the Silica Minerals. Rev. Mineral. 1994, 29, 433–467. [Google Scholar]
- Applin, K.R.; Hicks, B.D. Fibers of Dumortierite in quartz. Am. Mineral. 1987, 72, 170–172. [Google Scholar]
- Chauhan, M. Prasiolite with inclusion influenced by Brazil-law twinning. Gems Gemol. 2014, 50, 159–160. [Google Scholar]
- Czaja, M.; Kądziołka-Gaweł, M.; Konefał, A.; Sitko, R.; Teper, E.; Mazurak, Z.; Sachanbińskiet, M. The Mossbauer spectra of prasiolite and amethyst crystals from Poland. Phys. Chem. Miner. 2017, 44, 365–375. [Google Scholar] [CrossRef]
- Neumann, E.; Schmetzer, K. Farbe, Farbursache und Mechanismen der Farbumwandlung von Amethyst. Zeitschrift der Deutschen Gemmologischen Gesellschaft 1984, 33, 35–42. [Google Scholar]
- Neumann, E.; Schmetzer, K. Mechanism of thermal-conversion of colour and colour-centers by heat treatment of amethyst. N. Jahr. Miner. Monatshefte 1984, 6, 272–282. [Google Scholar]
- Henn, U.; Schultz-Güttler, R. Review of some current coloured quartz varieties. J. Gemmol. 2012, 33, 29–43. [Google Scholar] [CrossRef]
- Paradise, J.R. The natural formation and occurrence of green quartz. Gems Gemol. 1982, 18, 39–42. [Google Scholar] [CrossRef]
- Hebert, L.B.; Rossman, G. Greenish quartz from the Thunder Bay Amethyst Mine Panorama, Thunder Bay, Ontario, Canada. Can. Min. 2008, 46, 61–74. [Google Scholar] [CrossRef]
- McArthur, J.R.; Jennings, E.A.; Kissin, S.A.; Sherlock, R.L. Stable-isotope, fluid-inclusion, and mineralogical studies relating to the genesis of amethyst, Thunder Bay Amethyst Mine, Ontario. Can. J. Earth Sci. 1993, 30, 1955–1969. [Google Scholar] [CrossRef]
- Brooks, J. Marlborough Creek chrysoprase deposits, Rockhampton district, central Queensland. Gems Gemol. 1965, 11, 323–330. [Google Scholar]
- Visser, J. Nephrite and chrysoprase of Silesia. Gemmologist 1947, 16, 229–230. [Google Scholar]
- Sachanbinski, M.; Janeczek, J.; Platonov, A.; Rietmeijer, F.J.M. The origin of colour of chrysoprase from Szklary (Poland) and Sarykul Boldy (Kazakhstan). N. Jahrb. Miner. 2001, 177, 61–76. [Google Scholar] [CrossRef]
- Yang, L.; Mashkovtsev, R.I.; Botis, S.M.; Pan, Y. Multi-spectroscopic study of green quartzite (Guizhou Jade) from the Qinglong antimony deposit, Guizhou Province, China. J. China Univ. Geosci. 2007, 18, 327–329. [Google Scholar]
- Lieber, W. Amethyst: Geschichte, Eigenschaften, Fundorte; Christian Weise Verlag: München, Germany, 1994; 188p. [Google Scholar]
- Dedushenko, S.K.; Makhina, I.B.; Mar’in, A.A.; Mukhanov, V.A.; Perfiliev, Y.U.D. What oxidation state of iron determines the amethyst colour? Hyperfine Interact. 2004, 156, 417–422. [Google Scholar] [CrossRef]
- Di Benedetto, F.; Innocenti, M.; Tesi, S.; Romanelli, M.; D’Acapito, F.; Fornaciai, G.; Montegrossi, G.; Pardi, L.A. A Fe K-edge XAS study of amethyst. Phys. Chem. Miner. 2010, 37, 283–289. [Google Scholar] [CrossRef]
- SivaRamaiah, G.; Lin, J.; Pan, Y. Electron paramagnetic resonance spectroscopy of Fe3+ ions in amethyst: thermodynamic potentials and magnetic susceptibility. Phys. Chem. Miner. 2011, 38, 159–167. [Google Scholar] [CrossRef]
- Kievlenko, E.Y. Geology of Gems; Ocean Pictures Ltd.: Romsey, UK, 2003; 468p. [Google Scholar]
- Tatatrinov, A.V. Silic Minerals and Amethyst formation Conditions in Skarn-Magnetite Fields of the South of Siberian Platform. In Mineralogy and Genesis of East Siberia Gems; Nauka: Novosibirsk, Russia, 1983; pp. 34–41. (In Russian) [Google Scholar]
- Flink, G. Bidrag till Sveriges mineralogy. Arkiv Kemi. Mineral. Geol. 1910, 3, 163–166. [Google Scholar]
- Geijer, P.; Magnusson, N.H. De mellansvenska järnmalmernas geologi. Sver. Geol. Und. 1944, 35, 558. [Google Scholar]
- Vogt, M. Serifos-die Mineralien-Insel in der griechischen Ägäis. Lapis 1991, 16, 26–37. (In German) [Google Scholar]
- Gauthier, G.; Albandakis, N. Minerals from the Serifos skarn, Greece. Mineral. Rec. 1991, 22, 303–308. [Google Scholar]
- Dibble, H.L. Quartz: An Introduction to Crystalline Quartz; Dibble Trust Fund Publ.: New York, NY, USA, 2002; 100p. [Google Scholar]
- Hyrsl, J.; Niedermayr, G. Magic World: Inclusions in Quartz/Geheimnisvolle Welt: Einschlüsse im Quarz; Bode Verlag GmbH: Haltern, Germany, 2003. [Google Scholar]
- Maneta, V.; Voudouris, P. Quartz megacrysts in Greece: Mineralogy and environment of formation. Bull. Geol. Soc. Greece 2010, 43, 685–696. [Google Scholar] [CrossRef]
- Voudouris, P.; Katerinopoulos, A. New occurences of mineral megacrysts in tertiary magmatic-hydrothermal and epithermal environments in Greece. Doc. Nat. 2004, 151, 1–21. [Google Scholar]
- Ducoux, M.; Branquet, Y.; Jolivet, L.; Arbaret, L.; Grasemann, A.; Rabillard, C.; Gumiaux, C.; Drufin, S. Synkinematic skarns and fluid drainage along detachments: The West Cycladic Detachment System on Serifos Island (Cyclades, Greece) and its related mineralization. Tectonophysics 2017, 695, 1–26. [Google Scholar] [CrossRef]
- Kronz, A.; van den Kerkhof, A.M.; Müller, A. Analysis of low element concentrations in Quartz by Electron Microprobe. In Quartz: Deposits, Mineralogy, and Analytics; Götze, J., Möckel, R., Eds.; Springer: New York, NY, USA, 2012; pp. 191–217. [Google Scholar]
- Mattey, D.P. LaserPrep: An Automatic Laser-Fluorination System for Micromass “Optima” or “Prism” Mass Spectrometers. Micromass Appl. Note 1997, 207, 8. [Google Scholar]
- Jolivet, L.; Brun, J.P. Cenozoic geodynamic evolution of the Aegean region. Int. J. Earth Sci. 2010, 99, 109–138. [Google Scholar] [CrossRef]
- Ring, U.; Glodny, J.; Will, T.; Thomson, S. The Hellenic subduction system: high-pressure metamorphism, exhumation, normal faulting, and large-scale extension. Annu. Rev. Earth Planet. Sci. 2010, 38, 45–76. [Google Scholar] [CrossRef]
- Jolivet, L.; Faccenna, C.; Huet, B.; Labrousse, L.; Le Pourhiet, L.; Lacombe, O.; Lecomte, E.; Burov, E.; Denèle, Y.; Brun, J.-P.; et al. Aegean tectonics: Strain localisation, slab tearing and trench retreat. Tectonophysics 2013, 597–598, 1–33. [Google Scholar] [CrossRef]
- Maluski, H.; Bonneau, M.; Kienast, J.R. Dating the metamorphic events in the Cycladic area; 39Ar/40Ar data from metamorphic rocks of the Island of Syros (Greece). Bull. Soc. Géol. Fr. 1987, 3, 833–842. [Google Scholar]
- Grasemann, B.; Petrakakis, K. Evolution of the Serifos metamorphic core complex. J. Virtual Explor. 2007, 27, 1–18. [Google Scholar] [CrossRef]
- Rabillard, A.; Arbaret, L.; Jolivet, L.; Le Breton, N.; Gumiaux, C.; Augier, R.; Grasemann, B. Interactions between plutonism and detachments during metamorphic core complex formation, Serifos Island (Cyclades, Greece). Tectonics 2015, 34, 1080–1106. [Google Scholar] [CrossRef]
- Schneider, D.A.; Senkowski, C.; Vogel, H.; Grasemann, B.; Iglseder, Ch.; Schmitt, A.K. Eocene tectonometamorphism on Serifos (western Cyclades) deduced from zircon depth-profiling geochronology and mica thermochronology. Lithos 2011, 125, 151–172. [Google Scholar] [CrossRef]
- Salemink, J. On the Geology and Petrology of Serifos Island (Cyclades, Greece). Annu. Geol. Pays Hell. 1980, 30, 342–365. [Google Scholar]
- Grasemann, B.; Schneider, D.A.; Stockli, D.F.; Iglseder, C. Miocene bivergent crustal extension in the Aegean: Evidence from the western Cyclades (Greece). Lithosphere 2012, 4, 23–39. [Google Scholar] [CrossRef]
- Salemink, J. Skarn and Ore Formation at Serifos, Greece. Ph.D. Thesis, University of Utrecht, Utrecht, The Netherlands, 1985. [Google Scholar]
- Stouraiti, C.; Baziotis, I.; Asimow, P.D.; Downes, H. Geochemistry of the Serifos calc-alkaline granodiorite pluton, Greece: Constraining the crust and mantle contributions to I-type granitoids. Int. J. Earth Sci. (Geol. Rundsch) 2018, 107, 1657. [Google Scholar] [CrossRef]
- Sharp, Z.D.; Gibbons, J.A.; Maltsev, O.; Atudorei, V.; Pack, A.; Sengupta, S.; Shock, E.L.; Knauth, L.P. A calibration of the triple oxygen isotope fractionation in the SiO2–H2O system and applications to natural samples. Geochim. Cosmochim. Acta 2016, 186, 105–119. [Google Scholar] [CrossRef]
- Taylor, H.P., Jr. Oxygen and hydrogen isotope relationships in hydrothermal mineral deposits. In Geochemistry of Hydrothermal Ore Deposits, 2nd ed.; Barnes, H.L., Ed.; Wiley: New York, NY, USA, 1979; pp. 236–318. [Google Scholar]
- Giggenbach, W.F. The origin and evolution of fluids in magmatic-hydrothermal systems. In Geochemistry of Hydrothermal Ore Deposits, 3rd ed.; Barnes, H.L., Ed.; Wiley: New York, NY, USA, 1997; pp. 737–796. [Google Scholar]
- Simmons, S.F.; Browne, P.R.L. Hydrothermal minerals and precious metals in the Broadlands-Ohaaki geothermal system: Implications for understanding low-sulfidation epithermal environments. Econ. Geol. 2001, 95, 971–999. [Google Scholar] [CrossRef]
- Dana, J.D.; Dana, E.S.; Frondel, C. The System of Mineralogy, Vol. 3: Silica Minerals; John Wiley & Sons: New York, NY, USA, 1962. [Google Scholar]
- Fournier, R.O. The behaviour of silica in hydrothermal solutions. Geology and geochemistry of epithermal systems. Rev. Econ. Geol. 1985, 2, 45–61. [Google Scholar]
- Voudouris, P.; Melfos, V.; Mavrogonatos, C.; Tarantola, A.; Götze, J.; Alfieris, D.; Maneta, V.; Psimis, I. Amethyst occurrences in Tertiary volcanic rocks of Greece: Mineralogical, fluid inclusion and oxygen isotope constraints on their genesis. Minerals 2018, 8, 324. [Google Scholar] [CrossRef]

| Amphibole | Prase | Amethyst | |||||||
|---|---|---|---|---|---|---|---|---|---|
| n.o.a. | 3 (core) | 6 (rim) | 11 | 14 | |||||
| wt % | 2σ | wt % | 2σ | wt % | 2σ | wt % | 2σ | ||
| SiO2 | 51.1 | 0.04 | 50.6 | 0.57 | 99.3 | 0.56 | 99.9 | 0.44 | |
| TiO2 | 0.05 | 0.00 | 0.01 | 0.02 | - | - | 0.005 | 0.01 | |
| Al2O3 | 0.38 | 0.01 | 1.54 | 0.57 | 0.26 | 0.12 | 0.123 | 0.11 | |
| Cr2O3 | n.a. | - | n.a. | - | n.a. | - | 0.006 | 0.01 | |
| FeO | 29.4 | 0.40 | 28.3 | 1.15 | Fe2O3 | 0.027 | 0.019 | 0.021 | 0.03 |
| MnO | 0.56 | 0.02 | 0.25 | 0.12 | n.a. | - | 0.006 | 0.01 | |
| MgO | 3.81 | 0.07 | 4.98 | 0.72 | n.a. | - | 0.01 | 0.01 | |
| CaO | 11.6 | 0.04 | 11.3 | 0.09 | n.a. | - | 0.008 | 0.01 | |
| Na2O | 0.03 | 0.02 | 0.26 | 0.11 | n.a. | - | 0.01 | 0.01 | |
| K2O | 0.09 | 0.02 | 0.19 | 0.05 | 0.008 | 0.007 | 0.006 | 0.01 | |
| P2O5 | n.a. | - | n.a. | - | n.a. | - | 0.012 | 0.02 | |
| Total | 96.9 | 97.5 | 99.6 | 100.2 | |||||
| Structural formulae | |||||||||
| Si | 8.08 | 7.85 | Si | 0.998 | 0.998 | ||||
| AlVI | 0.00 | 0.15 | Ti | 0.000 | 0.000 | ||||
| M1-3 site | Al | 0.003 | 0.000 | ||||||
| AlVI | 0.07 | 0.13 | Fe3+ | 0.000 | 0.0002 | ||||
| Ti | 0.01 | 0.00 | Mn | - | - | ||||
| Fe3+ | 0.00 | 0.13 | Mg | - | 0.000 | ||||
| Mg | 0.89 | 1.15 | Ca | - | - | ||||
| Fe2+ | 3.87 | 3.55 | Na | - | - | ||||
| Mn | 0.08 | 0.03 | K | 0.000 | - | ||||
| Σ M1-3 | 4.92 | 5.00 | P | - | 0.000 | ||||
| M4 site | Σ All | 1.001 | 1.009 | ||||||
| Mg | 0.00 | 0.00 | |||||||
| Fe2+ | 0.00 | 0.00 | |||||||
| Mn | 0.00 | 0.00 | |||||||
| Ca | 1.95 | 1.88 | |||||||
| Na | 0.01 | 0.08 | |||||||
| Σ M4 | 1.96 | 1.96 | |||||||
| A site | |||||||||
| Na | 0.00 | 0.00 | |||||||
| K | 0.02 | 0.04 | |||||||
| Σ A | 0.02 | 0.04 | |||||||
| Σ All | 15.0 | 15.0 | |||||||
| Sample | Variety | Associated Minerals | δ18OQz | T (°C) | δ18OFl |
|---|---|---|---|---|---|
| SR2a | prase | ac | 12.72 | 350 | 7.32 |
| SR2b | prase | ac | 12.54 | 350 | 7.14 |
| SR1a | amethyst | hem | 9.94 | 250 | 1.04 |
| SR1b | amethyst | hem | 9.85 | 250 | 0.95 |
| 20–11 * | quartz | ep | 11.4 | 400 | 6.8 |
| 27–30 * | quartz | ep | 12.6 | 400 | 8.0 |
| 26–49 * | quartz | Mt + hem | 11.6 | 410 | 7.2 |
| 26–48 * | quartz | Mt + hem | 12.3 | 400 | 7.7 |
| 26–87 * | quartz | Ep + ac | 13.3 | 405 | 13.3 |
| 137 * | quartz | Ac + cc | 14.7 | 390 | 14.7 |
| 55 * | quartz | iv | 12.6 | 350 | 6.8 |
| 55-(1) * | quartz | iv | 12.5 | 325 | 6.0 |
| 56 * | quartz | iv | 13.2 | 325 | 6.7 |
| 26–35 * | quartz | mt | 14.4 | 325 | 7.8 |
| ML-2.2 * | quartz | Ac + mt | 16.6 | 265 | 7.8 |
| ML-1 * | quartz | Ac + mt | 16.5 | 260 | 7.5 |
| 27-7B * | quartz | Py + hem + cc | 14.8 | 250 | 5.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klemme, S.; Berndt, J.; Mavrogonatos, C.; Flemetakis, S.; Baziotis, I.; Voudouris, P.; Xydous, S. On the Color and Genesis of Prase (Green Quartz) and Amethyst from the Island of Serifos, Cyclades, Greece. Minerals 2018, 8, 487. https://doi.org/10.3390/min8110487
Klemme S, Berndt J, Mavrogonatos C, Flemetakis S, Baziotis I, Voudouris P, Xydous S. On the Color and Genesis of Prase (Green Quartz) and Amethyst from the Island of Serifos, Cyclades, Greece. Minerals. 2018; 8(11):487. https://doi.org/10.3390/min8110487
Chicago/Turabian StyleKlemme, Stephan, Jasper Berndt, Constantinos Mavrogonatos, Stamatis Flemetakis, Ioannis Baziotis, Panagiotis Voudouris, and Stamatios Xydous. 2018. "On the Color and Genesis of Prase (Green Quartz) and Amethyst from the Island of Serifos, Cyclades, Greece" Minerals 8, no. 11: 487. https://doi.org/10.3390/min8110487
APA StyleKlemme, S., Berndt, J., Mavrogonatos, C., Flemetakis, S., Baziotis, I., Voudouris, P., & Xydous, S. (2018). On the Color and Genesis of Prase (Green Quartz) and Amethyst from the Island of Serifos, Cyclades, Greece. Minerals, 8(11), 487. https://doi.org/10.3390/min8110487








