Multi-Analytical Investigation of Stains on Dimension Stones in Master Valentim’s Fountain, Brazil
Abstract
1. Introduction
1.1. Master Valentim’s Fountain
1.2. Phacoidal Gneiss
1.3. Alterability in Dimension Stone Monuments
1.4. Importance of Technological Support in Restoration
2. Materials and Methods
2.1. Petrography
2.2. X-ray Fluorescence (XRF)
2.3. X-ray Diffraction (XRD)
2.4. Physical Properties
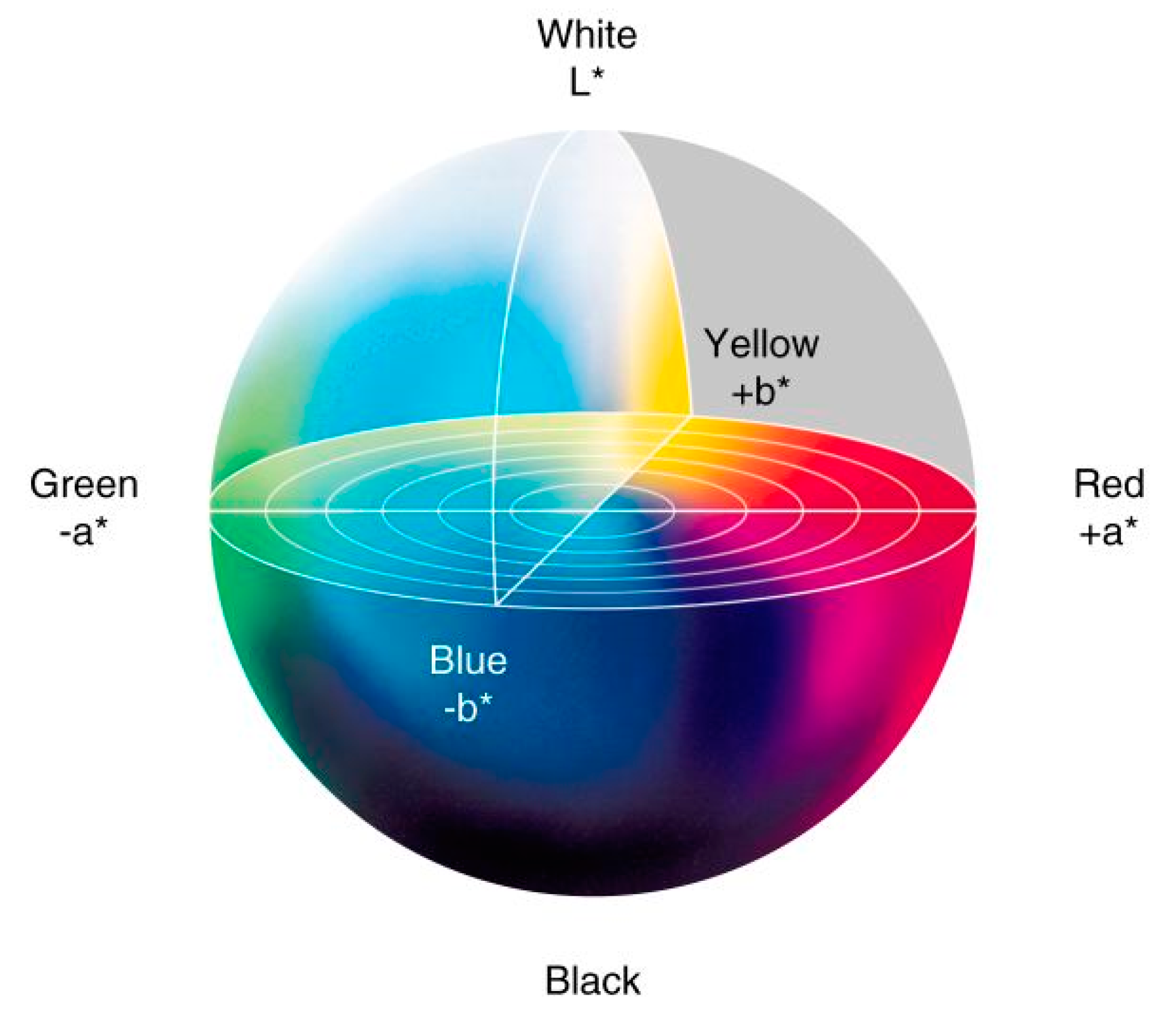
2.5. Colorimetry
2.6. Electrical Conductivity
2.7. Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES)
2.8. Scanning Electron Microscopy-Energy Dispersive X-ray (SEM-EDX)
2.9. Thermogravimetric Analysis (TGA)
3. Results and Discussion
3.1. Dimension Stones Characterization
3.2. Plate Characterization
3.3. Light Stains Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- ARQGUIARio. Available online: http://arqguia.com/obra/chafariz-do-mestre-valentim/?lang=en (accessed on 12 June 2018).
- Rio de Janeiro Aqui. Available online: http://www.riodejaneiroaqui.com/pt/chafariz-da-piramide.html (accessed on 12 June 2018).
- IPHAN. Available online: http://portal.iphan.gov.br/ans.net/tema_consulta.asp?Linha=tc_hist.gif&Cod=2974 (accessed on 12 June 2018).
- Thought Co. Available online: https://www.thoughtco.com/rococo-art-architecture-4147980 (accessed on 12 June 2018).
- Telles, A.C.S. Atlas dos Monumentos Históricos e Artísticos do Brasil. Available online: http://portal.iphan.gov.br/uploads/publicacao/ColObrRef_AtlasMonumentosHistoricosArtisticosBrasil.pdf (accessed on 12 June 2018).
- Brusadin, L.; Quites, M. A técnica da escultura em madeira com máscara de chumbo policromada: A contingência dos Cristos da Paixão da Ordem Terceira do Carmo de Outro Preto (MG). Visualidades 2016, 14, 188–215. [Google Scholar] [CrossRef]
- Baptista, A.P. Debret’s Rio de Janeiro Castro Maya Collection; Museus Castro Maya: Rio de Janeiro, Brazil, 2015; p. 128. [Google Scholar]
- Mansur, K.L.; Carvalho, I.S.; Delphim, C.F.M.; Barroso, E.V. O gnaisse facoidal: A mais carioca das rochas. Anuário IGEO 2008, 31, 9–22. [Google Scholar]
- Öztrürk, I. Alkoxysilanes Consolidation of Stone and Earthen Building Materials. Master’s Thesis, University of Pennsylvania, Philadelphia, PA, USA, 1992. [Google Scholar]
- Winkler, E.M. Stone in Architecture Properties Durability, 3rd ed.; Springer: Berlin, Germany, 1997. [Google Scholar]
- Shiavon, N. BSEM study of decay mechanisms in urban limestone. Eur. Cult. Herit. 1992, 6, 35–46. [Google Scholar]
- Torök, A.; Rosgonyi, N. Morphology and mineralogy of weathering crusts on highly porous oolitic limestones, a case study from Budapest. Environ. Geol. 2004, 46, 333–349. [Google Scholar] [CrossRef]
- Sabbioni, C. Contribution of atmospheric deposit to the formation of damage layer. Sci. Total Environ. 1995, 167, 49–55. [Google Scholar] [CrossRef]
- Fronteau, G.; Thomachot, C.S.; Chopin, E.; Barbin, V.; Mouze, D.; Pascal, A. Black-Crust Growth and Interaction with Underling Limestone Microfacies. In Natural Stone Resources for Historical Monuments; Geological Society of London: London, UK, 2010; Volume 333, pp. 25–34. [Google Scholar]
- Matthieu, A.; Hérbert, R.; Menéndez, B.; David, C.; Bigas, J.P. Influence of Temperature and Salt Concentration on the Salt Weathering of A Sedimentary Stone With Sodium Sulphate. In Natural Stone Resources for Historical Monuments; Geological Society of London: London, UK, 2010; Volume 115, pp. 193–199. [Google Scholar]
- Bakolas, A.; Biscontin, G.; Moropoulou, A.; Zendri, E. Characterization of structural byzantine mortars by thermogravimetric analysis. Thermochim. Acta 1998, 321, 151–160. [Google Scholar] [CrossRef]
- Chiarelli, N.; Miriello, D.; Bianchi, G.; Fichera, G.; Giamello, M.; Memmi, I.T. Characterization of ancient mortars from the S. Niccoló archaeological complex in Montieri (Tuscany Italy). Constr. Build. Mater. 2015, 96, 442–460. [Google Scholar] [CrossRef]
- Gleize, P.; Motta, E.; Silva, D.; Roman, H. Characterization of historical mortars from Santa Catarina (Brazil). Cem. Concr. Compos. 2009, 31, 342–346. [Google Scholar] [CrossRef]
- Moropoulou, A.; Bakolas, A.; Bisbikou, K. Characterization of ancient, byzantine and later historic mortars by thermal and X-ray diffraction techniques. Thermochim. Acta 1995, 269, 779–795. [Google Scholar] [CrossRef]
- Moropoulou, A.; Bakolas, A.; Bisbikou, K. Investigation of the technology of historic mortars. J. Cult. Herit. 2000, 1, 45–58. [Google Scholar] [CrossRef]
- Biscontin, G.; Birelli, M.P.; Zendri, E. Characterization of binders employed in the manufacture of Venetian historical mortars. J. Cult. Herit. 2002, 3, 31–37. [Google Scholar] [CrossRef]
- Freidin, C.; Meir, I. Byzantine mortars of the Negev Desert. Constr. Build. Mater. 2005, 19, 19–23. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhang, B.; Liang, X. A case study and mechanism investigation of typical mortars used on ancient architecture in China. Thermochim. Acta 2008, 473, 1–6. [Google Scholar] [CrossRef]
- Adriano, P.; Silva, A.S.; Veiga, R.; Mirao, J.; Candeias, A. Microscopic characterisation of old mortars from the Santa Maria Church in Évora. Mater. Charact. 2009, 60, 610–620. [Google Scholar] [CrossRef]
- Budak, M.; Akkurt, S.; Bke, H. Evaluation of heat treated clay for potential use in intervention mortars. Appl. Clay Sci. 2010, 49, 414–419. [Google Scholar] [CrossRef]
- Sanjurjo-Sánchez, J.; Trindade, M.; Blanco-Rotea, R.; Garcia, R.B.; Mosquera, D.F.; Burbidge, C.; Prudêncio, M.I.; Dias, M.I. Chemical and mineralogical characterization of historic mortars from the Santa Eulalia de Bóveda temple, NW Spain. J. Archaeol. Sci. 2010, 37, 2346–2351. [Google Scholar] [CrossRef]
- Martínez, I.; Castillo, A.; Martínez, E.; Castellote, M. Physico-chemical material characterization of historic unreinforced masonry buildings: The first step for a suitable intervention. Constr. Build. Mater. 2013, 40, 352–360. [Google Scholar] [CrossRef]
- Lezzerini, M.; Legnaioli, S.; Lorenzetti, G.; Palleschi, V.; Tamponi, M. Characterization of historical mortars from the bell tower of St. Nicholas Church (Pisa, Italy). Constr. Build. Mater. 2014, 69, 203–212. [Google Scholar] [CrossRef]
- Maria, S. Methods for porosity measurement in lime-based mortars. Constr. Build. Mater. 2010, 24, 2572–2578. [Google Scholar] [CrossRef]
- Nazdar Ink Technologies. Available online: https://www.nazdar.com/en-us/News-events/ArtMID/4165/ArticleID/224 (accessed on 12 June 2018).
- Marques, E.A.G.; Barroso, E.V.; Menezes Filho, A.P.; Vargas, E.A., Jr. Weathering zones on metamorphic rocks from Rio de Janeiro—Physical, mineralogical and geomechanical characterization. Eng. Geol. 2010, 111, 1–18. [Google Scholar] [CrossRef]
- National Research Council. Conservation of Historic Stone Buildings and Monuments; The National Academies Press: Washington, DC, USA, 1982. [Google Scholar]
- Frazão, E.B.; Farjallat, J.E. Características tecnológicas das principais rochas silicáticas brasileiras usadas como pedras de revestimento. In I Congresso Internacional de Pedra Natural; FIL/AIP: Lisboa, Portugal, 1999; pp. 47–58. [Google Scholar]
- Delgado-Rodrigues, J. Conservation of granitic rocks with application to the megalithic monuments. conclusions report. In Degradation and Conservation of Granitic Rocks in Monuments; Vicente, M.A., Delgado-Rodrigues, J., Acevedo, J., Eds.; European Commission: Brussels, Belgium, 1996; pp. 161–242. [Google Scholar]
- Feilden, B.M. Conservation of Historic Buildings; Reed Edcuational and Porfessiornal Publish: Oxford, UK, 1994. [Google Scholar]
- Giacomelli, V.; Perego, G. II Degrade della Pietra in Basilica de San Pietro, Restauro e Conservazione; ENI: Roma, Italy, 1999; pp. 108–123. [Google Scholar]
- Gobbi, G.; Zappia, G.; Sarbbioni, C. Sulphite quantification on damaged stones and mortar. Atmos. Environ. 1998, 32, 783–798. [Google Scholar] [CrossRef]
- Gonzáles-Messones, F.L. La Interpretación de los Ensayos de Caracterización de la Piedra Natural, en el Marco de la Nueva Normativa Europea. In Curso de Rochas Ornamentais. Recife. CD-ROM. 2002. Available online: http://mineralis.cetem.gov.br/bitstream/cetem/1201/1/Cap.III.part.1.pdf (accessed on 12 October 2018).
- Gupta, A.S.; Rao, S. Weathering effects on the strength and deformational behavior of crystalline rocks under uniaxial compression state. Eng. Geol. 2000, 56, 357–374. [Google Scholar] [CrossRef]
- Perry, S.H.; Duffy, A.P. The short-term effects of mortar joints on salt movement in stone. Atmos. Environ. 1997, 31, 1297–1305. [Google Scholar] [CrossRef]
- Theoulakis, P.; Moropoulou, A. Microstructural and mechanical parameters determining the susceptibility of porous building stones to salt decay. Constr. Build. Mater. 1997, 11, 65–71. [Google Scholar] [CrossRef]
- Viles, H.A. Urban Air Pollution and the Deterioration of Buildings and Monuments. In The Global Environment: Science, Technology and Management; Brune, D., Chapman, D.V., Gruynne, M.D., Pacyna, J.M., Eds.; Wiley-VCH: Weinheim, Germany, 1997; pp. 599–609. [Google Scholar]
- Fitzner, B.J. Investigation of weathering damage on stone monuments. Geonomos 2016, 24, 1–15. [Google Scholar] [CrossRef]











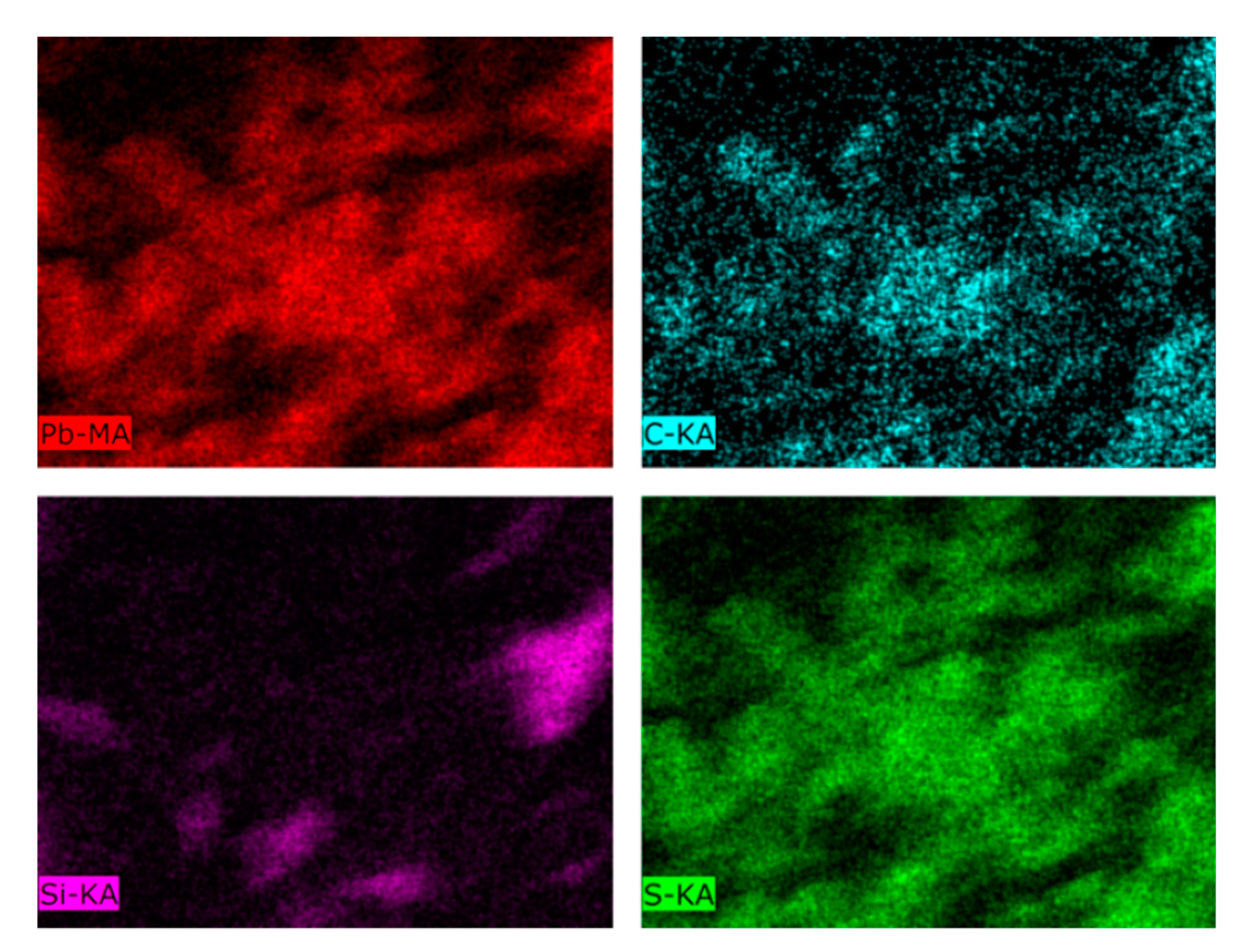
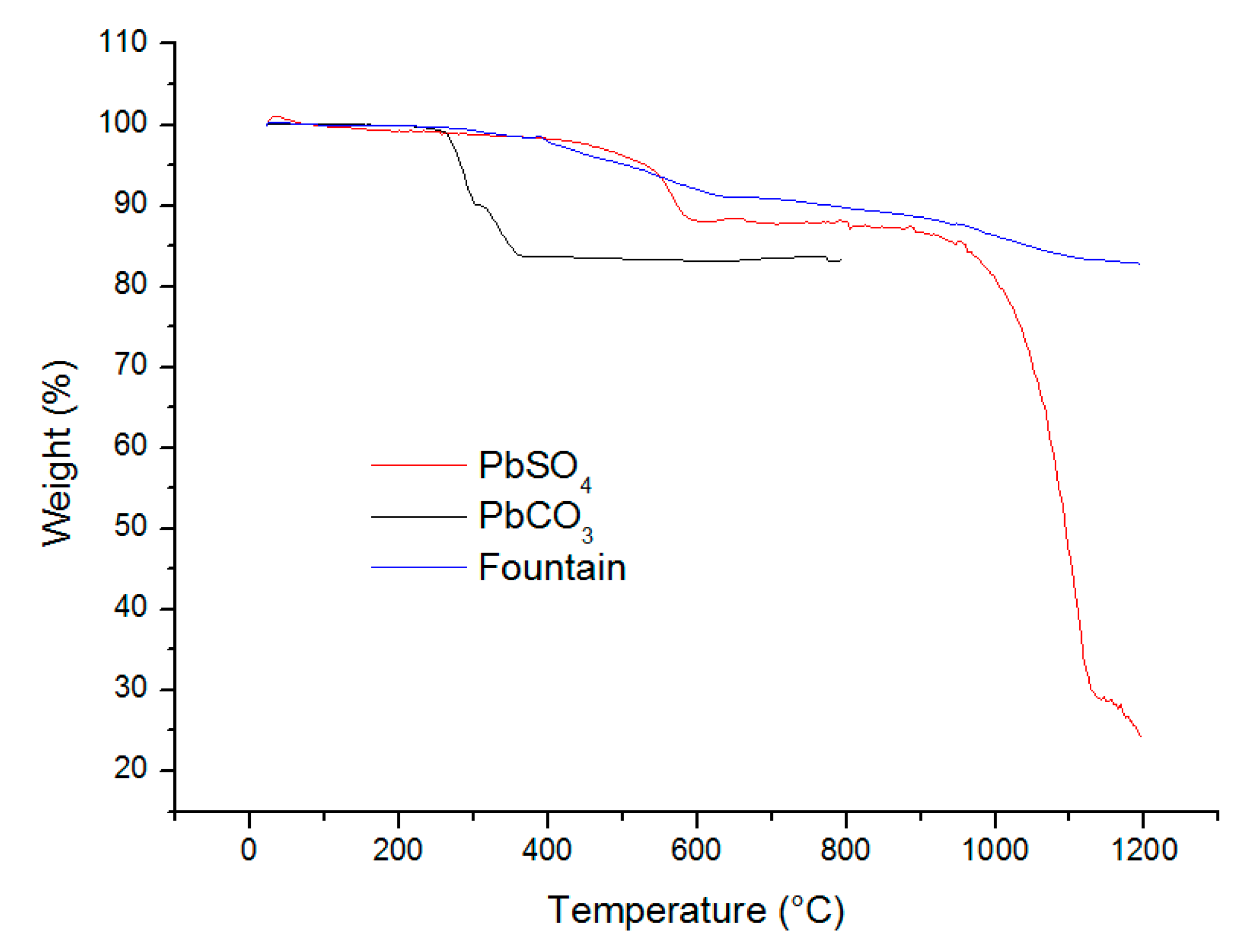
| Analytes | Na | Al | K | Ca | Fe | Mg | S |
|---|---|---|---|---|---|---|---|
| (mg L−1) | 2.1 | <0.007 | 0.81 | 3.5 | 0.01 | 0.43 | <0.01 |
| Samples | Altered/Substitute Stone Parameters | Standard Stone Parameters | ||||||
|---|---|---|---|---|---|---|---|---|
| L | a | b | G | L | a | b | G | |
| Substitute Gneiss (torch top) | 67.5 | 2.05 | 7.77 | 1.30 | 61.4 | 3.62 | 16.0 | 0.70 |
| Substitute Gneiss (torch basis) | 72.1 | 1.99 | 9.29 | 1.10 | 61.4 | 3.62 | 16.0 | 0.70 |
| Substitute Gneiss (parapet) | 65.7 | 0.42 | 5.94 | 1.00 | 55.3 | 5.07 | 15.3 | 0.50 |
| Original Gneiss (external wall, light staining) | 56.8 | 0.22 | 4.83 | 0.50 | 57.4 | 1.43 | 12.9 | 0.80 |
| Lioz limestone (torch) | 74.3 | 1.77 | 6.98 | 1.50 | 73.4 | 1.99 | 8.44 | 0.80 |
| Lioz limestone (banister) | 76.1 | 1.86 | 7.74 | 0.90 | 73.9 | 2.54 | 9.80 | 1.10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Conceição Ribeiro, R.C.; Marques Ferreira de Figueiredo, P.; Silva Barbutti, D. Multi-Analytical Investigation of Stains on Dimension Stones in Master Valentim’s Fountain, Brazil. Minerals 2018, 8, 465. https://doi.org/10.3390/min8100465
Da Conceição Ribeiro RC, Marques Ferreira de Figueiredo P, Silva Barbutti D. Multi-Analytical Investigation of Stains on Dimension Stones in Master Valentim’s Fountain, Brazil. Minerals. 2018; 8(10):465. https://doi.org/10.3390/min8100465
Chicago/Turabian StyleDa Conceição Ribeiro, Roberto Carlos, Patrícia Marques Ferreira de Figueiredo, and Daniel Silva Barbutti. 2018. "Multi-Analytical Investigation of Stains on Dimension Stones in Master Valentim’s Fountain, Brazil" Minerals 8, no. 10: 465. https://doi.org/10.3390/min8100465
APA StyleDa Conceição Ribeiro, R. C., Marques Ferreira de Figueiredo, P., & Silva Barbutti, D. (2018). Multi-Analytical Investigation of Stains on Dimension Stones in Master Valentim’s Fountain, Brazil. Minerals, 8(10), 465. https://doi.org/10.3390/min8100465





