Synchrotron Radiation XRD Investigation of the Fine Phase Transformation during Synthetic Chalcocite Acidic Ferric Sulfate Leaching
Abstract
1. Introduction
2. Materials and Methods
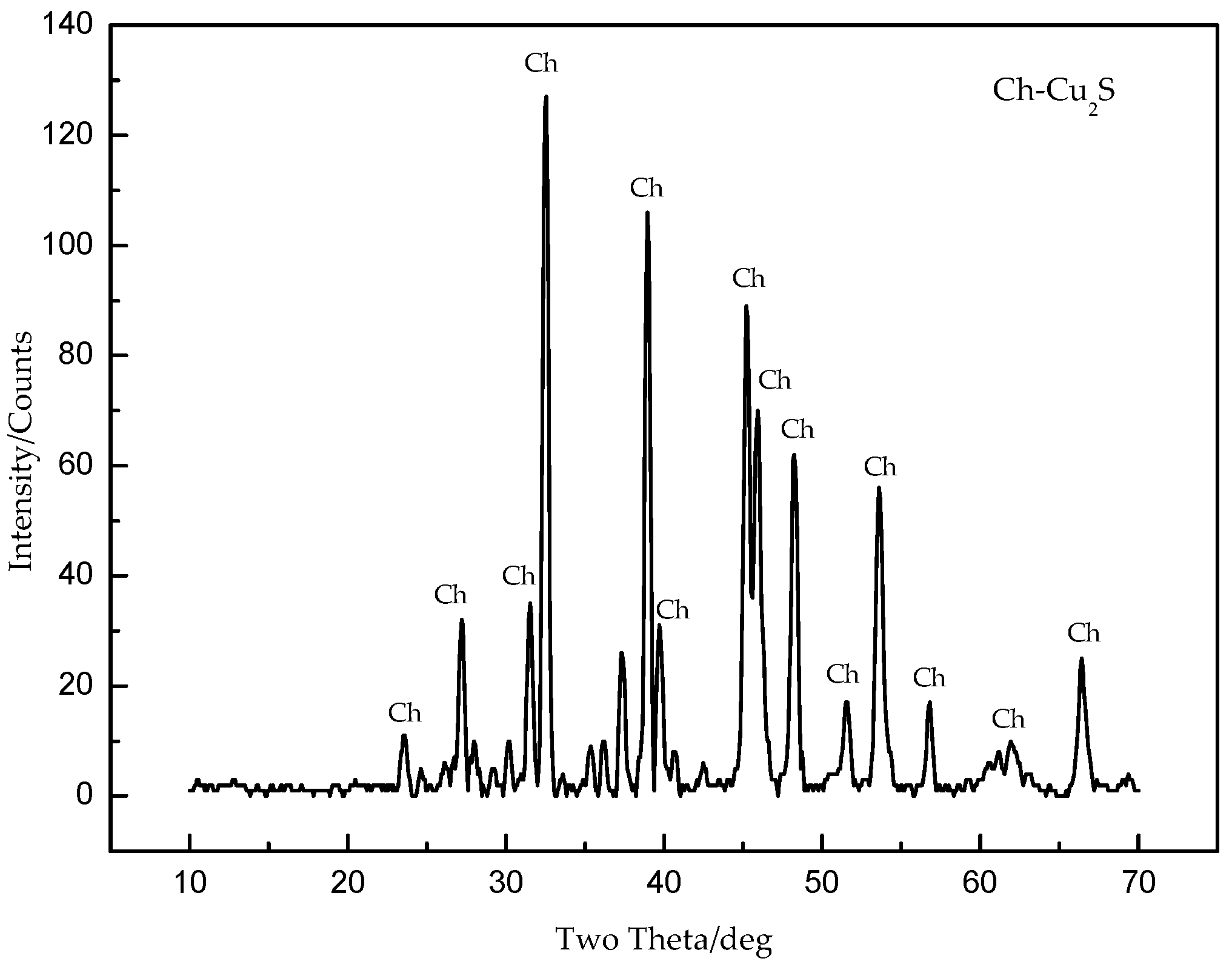
2.1. Mineral
2.2. Leaching Experiments
2.3. Synchrotron Radiation X-ray Diffraction
3. Results and Discussion
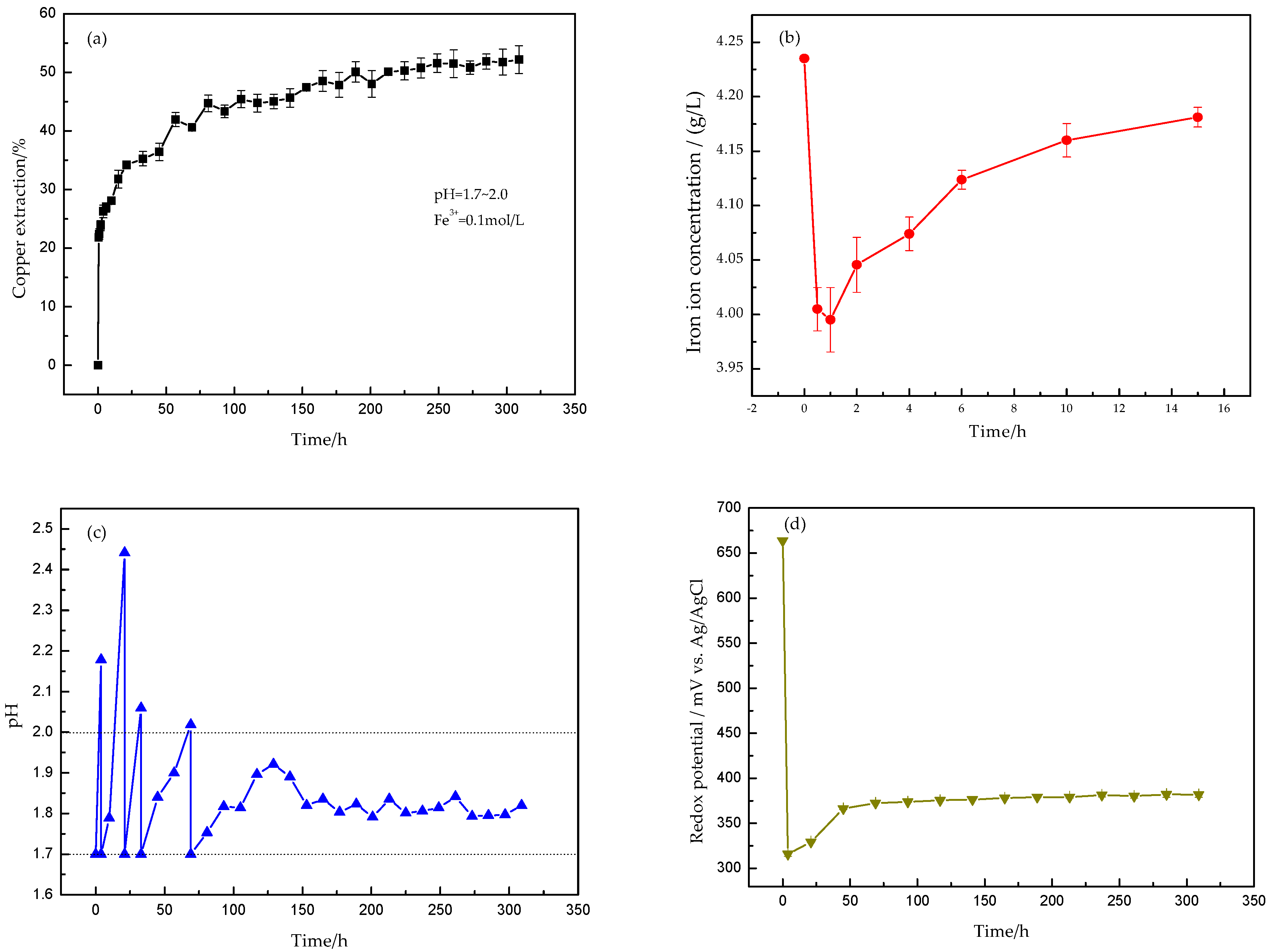
3.1. Leaching Behaviors
3.2. Synchrotron Radiation X-ray Diffraction Tests
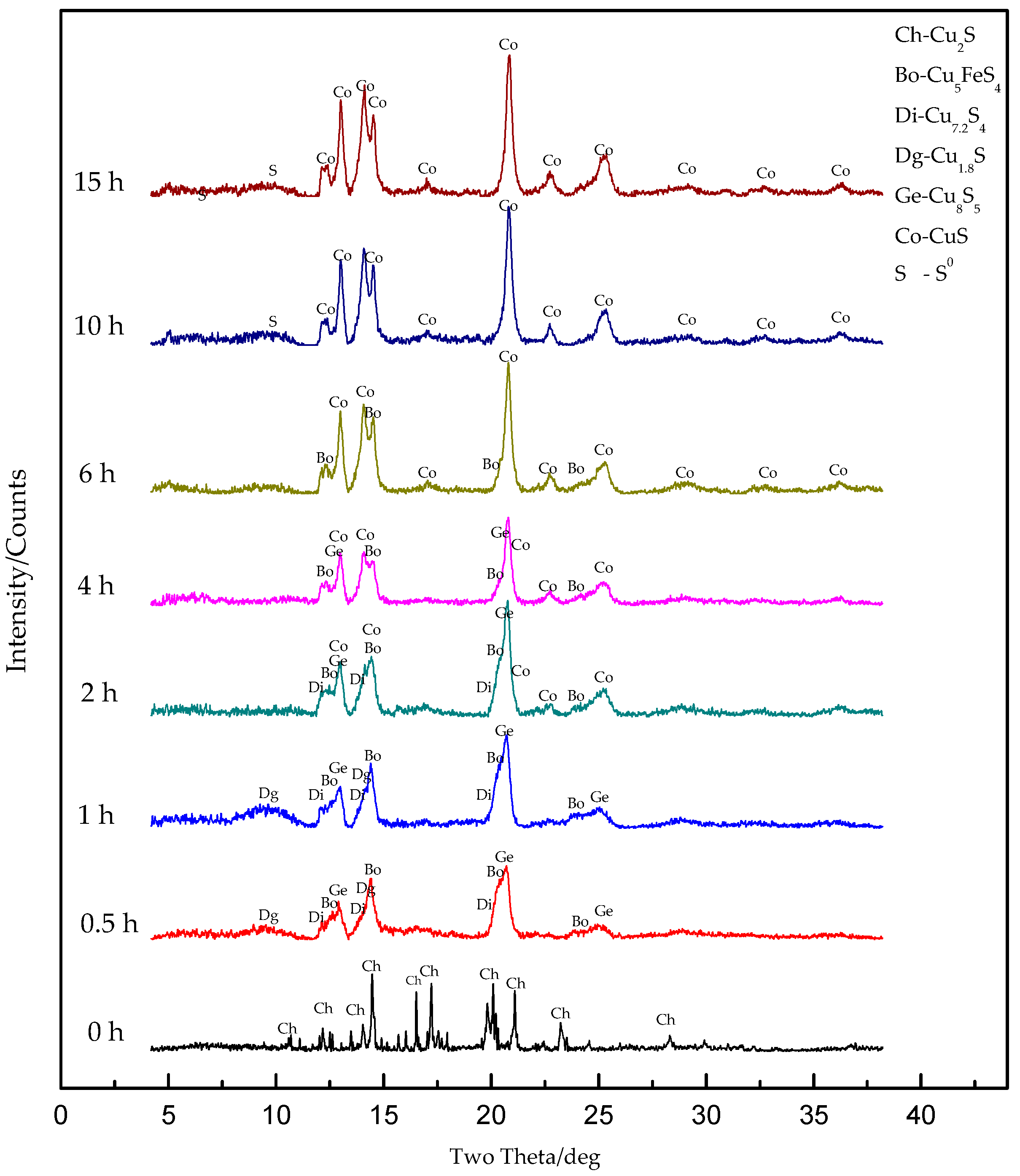
3.2.1. SRXRD Results of the First Stage of Leaching
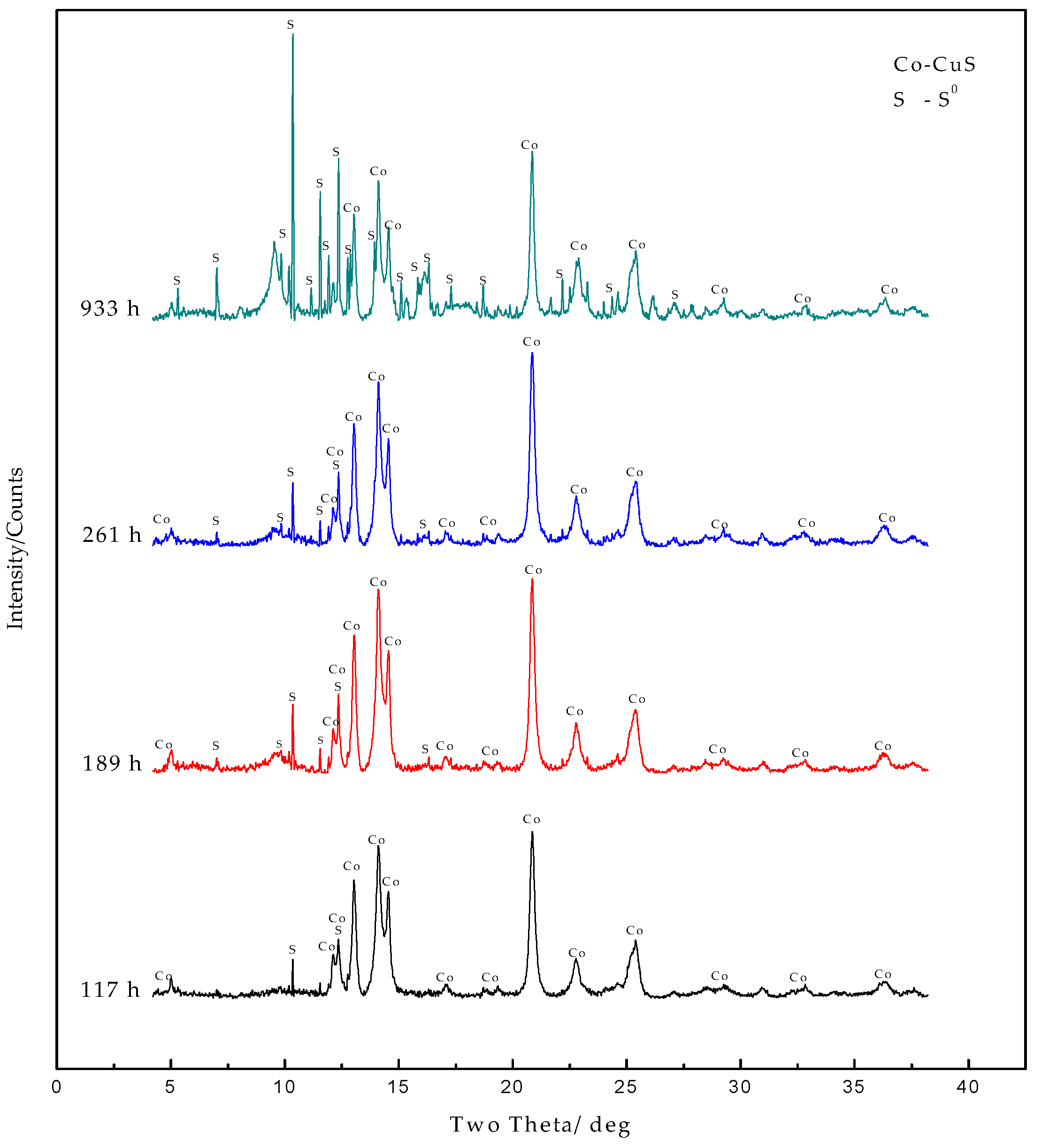
3.2.2. SRXRD Results of the Second Stage of Chalcocite Leaching
3.3. Thermodynamic Calculation of the Generation and Dissolution of Cu5FeS4 in Chalcocite Leaching Process
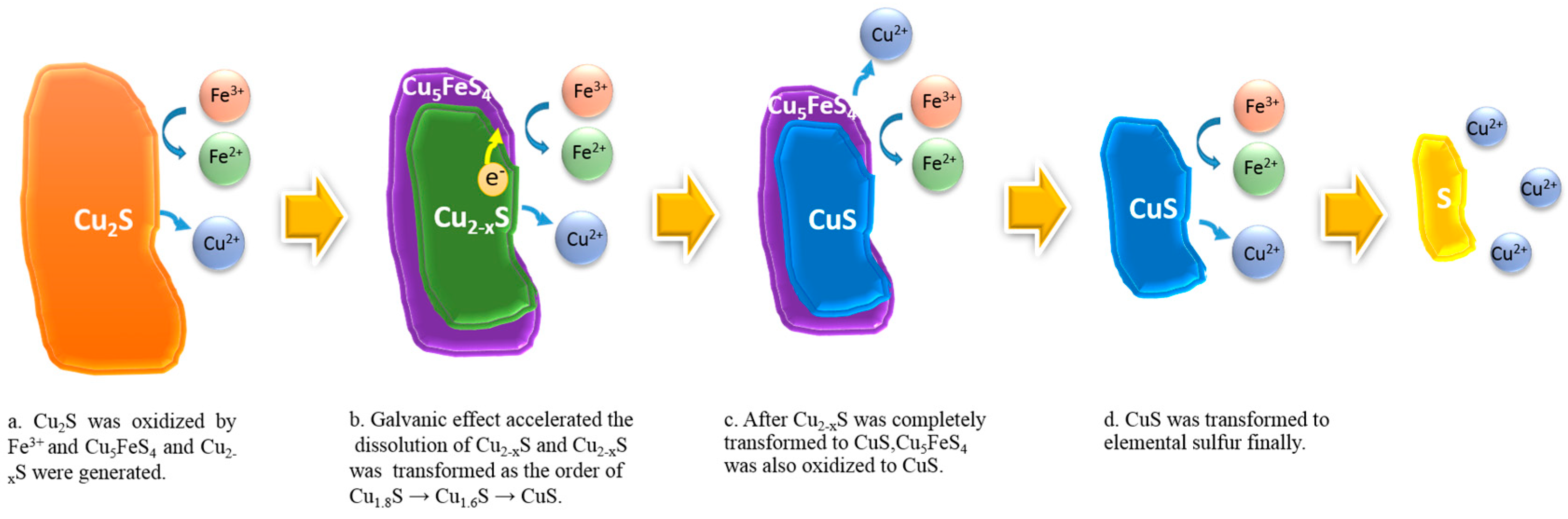
3.4. Disscussion of the Fine Phase Transformation of Chalcocite Acidic Ferric Sulfate Leaching
4. Conclusions
- (1)
- Firstly, chalcocite reacted with Fe3+ and was quickly transformed to Cu7.2S4, Cu1.8S, Cu8S5, and Cu5FeS4.
- (2)
- Then, the galvanic effect between Cu5FeS4 and Cu2-xS accelerated the dissolution of Cu2−xS, and the phase transformation order of Cu2−xS was Cu1.8S → Cu1.6S → CuS.
- (3)
- When Cu2−xS was completely transformed to CuS, Cu5FeS4 was transformed to CuS thereafter.
- (4)
- In the second stage, the generated CuS was oxidized to elemental sulfur, which should be the passivation layer inhibiting the efficient leaching of chalcocite.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, X.M.; Peng, Y.J.; Bradshaw, D. The separation of chalcopyrite and chalcocite from pyrite in cleaner flotation after regrinding. Miner. Eng. 2014, 58, 64–72. [Google Scholar] [CrossRef]
- Qin, W.Q.; Wu, J.J.; Jiao, F.; Zeng, J.M. Mechanism study on flotation separation of molybdenite from chalcocite using thioglycollic acid as depressant. Int. J. Min. Sci. Technol. 2017, 27, 1043–1049. [Google Scholar] [CrossRef]
- Gao, Y.S.; Gao, Z.Y.; Sun, W.; Yin, Z.G.; Wang, J.J.; Hu, Y.Y. Adsorption of a novel reagent scheme on scheelite and calcite causing an effective flotation separation. J. Colloid Interface Sci. 2018, 512, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Li, C.W.; Gao, Z.Y. Effect of grinding media on the surface property and flotation behavior of scheelite particles. Powder Technol. 2017, 322, 386–392. [Google Scholar] [CrossRef]
- Brierley, J.A.; Brierley, C.L. Present and future commercial applications in biohydrometallurgy. Hydrometallurgy 2001, 59, 233–240. [Google Scholar] [CrossRef]
- Zhao, H.B.; Wang, J.; Hu, M.H.; Qin, W.Q.; Zhang, Y.S.; Qiu, G.Z. Synergistic bioleaching of chalcopyrite and bornite in the presence of Acidithiobacillus ferrooxidans. Bioresour. Technol. 2013, 149, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.B.; Wang, J.; Gan, X.W.; Zheng, X.H.; Tao, L.; Hu, M.H.; Li, Y.N.; Qin, W.Q.; Zhang, Y.S.; Qiu, G.Z. Effects of pyrite and bornite on bioleaching of two different types of chalcopyrite in the presence of Leptospirillum ferriphilum. Bioresour. Technol. 2015, 194, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Lan, Z.Y.; Hu, Y.H.; Liu, J.S.; Wang, J. Solvent extraction of copper and zinc from bioleaching solutions with LIX984 and D2EHPA. J. Cent. South Univ. Technol. 2005, 12, 45–49. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, H.B.; Zhuang, T.; Qin, W.Q.; Zhu, S.; Qiu, G.Z. Bioleaching of Pb–Zn–Sn chalcopyrite concentrate in tank bioreactor and microbial community succession analysis. Trans. Nonferrous Met. Soc. China 2013, 23, 3758–3762. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, S.; Zhang, Y.S.; Zhao, H.B.; Hu, M.H.; Yang, C.R.; Qin, W.Q.; Qiu, G.Z. Bioleaching of low-grade copper sulfide ore by Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans. J. Cent. South Univ. 2014, 21, 728–734. [Google Scholar] [CrossRef]
- Zhao, H.B.; Huang, X.T.; Hu, M.H.; Zhang, C.Y.; Zhang, Y.S.; Wang, J.; Qin, W.Q.; Qiu, G.Z. Insights into the surface transformation and electrochemical dissolution process of bornite in bioleaching. Minerals 2018, 8, 173. [Google Scholar] [CrossRef]
- Michael, N.; Petrus, B. The anodic behavior of covellite in chloride solutions. Hydrometallurgy 2017, 172, 60–68. [Google Scholar]
- Bolorunduro, S.A. Kinetics of Leaching of Chalcocite in Acid Ferric Sulfate Media: Chemical and Bacterial Leaching. Master’s Thesis, University of Brithsh Columbia, Vancouver, BC, Canada, 1999. [Google Scholar]
- Brierley, C.L. A perspective on developments in biohydrometallurgy. Hydrometallurgy 2008, 94, 2–7. [Google Scholar] [CrossRef]
- Brierley, C.L. Biohydrometallurgical prospects. Hydrometallurgy 2010, 104, 324–328. [Google Scholar] [CrossRef]
- Buerger, M.J.; Wuensch, B.J. Distribution of atoms in high chalcocite, Cu2S. Science 1963, 141, 276–277. [Google Scholar] [CrossRef] [PubMed]
- Howard, T.; Evans, J. The crystal structure of low chalcocite and djurleite. Z. Krist. 1979, 150, 299–320. [Google Scholar]
- Evans, J.; Howard, T. Djurleite (Cu1.94S) and Low Chalcocite (Cu2S): New Crystal Structure Studies. Science 1979, 203, 356–358. [Google Scholar] [CrossRef] [PubMed]
- Howard, T.; Evans, J. Copper coordination in low chalcocite and djurleite and other copper-rich sulfides. Am. Miner. 1981, 66, 807–818. [Google Scholar]
- Sullivan, J.D. Chemistry of Leaching Chalcocite; US Bureau of Mines: Washington, DC, USA, 1930; p. TP 473.
- Fisher, W.W.; Flores, F.A.; Henderson, J.A. Comparison of chalcocite dissolution in the oxygenated, aqueous sulfate and chloride systems. Miner. Eng. 1992, 7, 817–834. [Google Scholar] [CrossRef]
- Peterson, J.; Dixon, D.G. The dynamics of chalcocite heap bioleaching. In Hydrometallurgy 2003: Fifth International Conference in Honor of Professor Lan M. Ritchie; TMS: Pittsburgh, PA, USA, 2003; pp. 351–364. [Google Scholar]
- Ruiz, M.C.; Abarzua, E.; Padilla, R. Oxygen pressure leaching of white metal. Hydrometallurgy 2007, 86, 131–139. [Google Scholar] [CrossRef]
- Miki, H.; Nicol, M.; Velásquez-Yévenes, L. The kinetics of dissolution of synthetic covellite, chalcocite and digenite in dilute chloride solutions at ambient temperatures. Hydrometallurgy 2011, 105, 321–327. [Google Scholar] [CrossRef]
- Ruan, R.M.; Zou, G.; Zhong, S.P.; Wu, Z.L.; Chan, B.; Wang, D.Z. Why Zijinshan copper bioheapleaching plant works efficiently at low microbial activity—Study on leaching kinetics of copper sulfides and its implications. Miner. Eng. 2013, 48, 36–43. [Google Scholar] [CrossRef]
- Niu, X.P.; Ruan, R.M.; Tan, Q.Y.; Jia, Y.; Sun, H.Y. Study on the second stage of chalcocite leaching in column with redox potential control and its implications. Hydrometallurgy 2015, 155, 141–152. [Google Scholar] [CrossRef]
- Cavallotti, P.; Salvago, G. Electronic Behavior of Copper Sulfides in Aqueous Solutions. Electrochim. Metallorum 1969, 4, 181–210. [Google Scholar]
- Moh, G.H. Blue remaining covellite and its relations to phases in the sulfur rich portion of the copper-sulfur System at low temperatures. Miner. Soc. Jpn. 1971, 1, 180–188. [Google Scholar]
- Whiteside, L.S.; Goble, R.J. Structural and compositional changes in copper sulfides during leaching and dissolution. Can. Mineral. 1986, 24, 247–258. [Google Scholar]
- Cheng, C.Y.; Lawson, F. The kinetics of leaching chalcocite in acidic oxygenated sulphat-chloride solutions. Hydrometallurgy 1991, 27, 249–268. [Google Scholar] [CrossRef]
- Zhao, H.B.; Wang, J.; Yang, C.R.; Hu, M.H.; Gan, X.W.; Tao, L.; Qin, W.Q.; Qiu, G.Z. Effect of redox potential on bioleaching of chalcopyrite by moderately thermophilic bacteria: An emphasis on solution compositions. Hydrometallurgy 2015, 151, 141–150. [Google Scholar] [CrossRef]
- Dutrizac, J. Elemental sulphur formation during the ferric sulphate leaching of chalcopyrite. Can. Metall. Q. 1989, 28, 337–344. [Google Scholar] [CrossRef]
- Dutrizac, J. Elemental sulphur formation during the ferric chloride leaching of chalcopyrite. Hydrometallurgy 1990, 23, 153–176. [Google Scholar] [CrossRef]
- Klauber, C. A critical review of the surface chemistry of acidic ferric sulphate dissolution of chalcopyrite with regards to hindered dissolution. Int. J. Miner. Process. 2008, 86, 1–17. [Google Scholar] [CrossRef]
- Zhao, H.B.; Hu, M.H.; Li, Y.N.; Zhu, S.; Qin, W.Q.; Qiu, G.Z.; Wang, J. Comparison of electrochemical dissolution of chalcopyrite and bornite in acid culture medium. Trans. Nonferrous Met. Soc. China 2015, 25, 303–313. [Google Scholar] [CrossRef]
- Zhao, H.B.; Wang, J.; Gan, X.W.; Hu, M.H.; Tao, L.; Qin, W.Q.; Qiu, G.Z. Role of pyrite in sulfuric acid leaching of chalcopyrite: An elimination of polysulfide by controlling redox potential. Hydrometallurgy 2016, 164, 159–165. [Google Scholar] [CrossRef]
- Zhao, H.B.; Wang, J.; Qin, W.Q.; Hu, M.H.; Zhu, S.; Qiu, G.Z. Electrochemical dissolution process of chalcopyrite in the presence of mesophilic microorganisms. Miner. Eng. 2015, 71, 159–169. [Google Scholar] [CrossRef]
- Qin, W.Q.; Wang, J.; Zhang, Y.S.; Zhen, S.J.; Shang, H.; Liu, Q.; Qiu, G.Z. Electrochemical behavior of massive bornite bioleached electrodes in the presence of Acidithiobacillus ferrooxidans and Acidithiobacillus caldus. Adv. Mater. Res. 2009, 71, 417–420. [Google Scholar] [CrossRef]
- Liu, H.C.; Xia, J.L.; Nie, Z.Y. Comparative study of S, Fe and Cu speciation transformation during chalcopyrite bioleaching by mixed mesophiles and mixed thermophiles. Miner. Eng. 2017, 106, 22–32. [Google Scholar] [CrossRef]
- Yang, Y.; Harmer, S.; Chen, M. Synchrotron X-ray photoelectron spectroscopic study of the chalcopyrite leached by moderate thermophiles and mesophiles. Miner. Eng. 2014, 69, 185–195. [Google Scholar] [CrossRef]
- Acres, R.G.; Harmer, S.L.; Beattie, D.A. Synchrotron XPS studies of solution exposed chalcopyrite, bornite, and heterogeneous chalcopyrite with bornite. Int. J. Miner. Process. 2010, 94, 43–51. [Google Scholar] [CrossRef]
- Majuste, D.; Ciminelliv, S.T.; Eng, P.J. Applications of in situ synchrotron XRD in hydrometallurgy: Literature review and investigation of chalcopyrite dissolution. Hydrometallurgy 2013, 131, 54–66. [Google Scholar] [CrossRef]
- Fang, J.H.; Liu, Y.; He, W.L.; Qin, W.Q.; Qiu, G.Z.; Wang, J. Transformation of iron in pure culture process of extremely acidophilic microorganisms. Trans. Nonferrous Met. Soc. China 2017, 27, 1150–1155. [Google Scholar] [CrossRef]
- Wang, X.X.; Liao, R.; Zhao, H.B.; Hong, M.X.; Wang, J. Synergetic effect of pyrite on strengthening bornite bioleaching by Leptospirillum ferriphilum. Hydrometallurgy 2017, 176, 9–16. [Google Scholar] [CrossRef]
- Fang, C.J.; Chang, Z.Y.; Feng, Q.M.; Xiao, W.; Yu, S.C.; Qiu, G.Z.; Wang, J. The influence of backwater Al3+ on diaspore bauxite flotation. Minerals 2017, 7, 195. [Google Scholar] [CrossRef]
- Zhao, H.B.; Wang, J.; Tao, L.; Cao, P.; Yang, C.R.; Qin, W.Q.; Qiu, G.Z. Roles of oxidants and reductants in bioleaching system of chalcopyrite at normal atmospheric pressure and 45 °C. Int. J. Miner. Process. 2017, 162, 81–91. [Google Scholar] [CrossRef]
- Liang, Y.; Che, Y. Inorganic Thermodynamic Data Manual, 1st ed.; Northeastern University Press: Shenyang, China, 1993; pp. 39–441. [Google Scholar]
- Dean, J.A. Lange’s Handbook of Chemistry, 12th ed.; MvGrew-Hill: New York, NY, USA, 1979; pp. 9–94. [Google Scholar]
- Wu, B.; Yang, X.L.; Cai, L.L.; Yao, G.C.; Wen, J.K.; Wang, D.Z. The influence of pyrite on galvanic assisted leaching of chalcocite concentrates. Adv. Mater. Res. 2013, 825, 459–463. [Google Scholar] [CrossRef]
- Javad Koleini, S.M.; Jafarian, M.; Abdollahy, M.; Aghazadeh, V. Galvanic leaching of chalcopyrite in atmospheric pressure and sulfate media: Kinetic and surface studies. Ind. Eng. Chem. Res. 2010, 49, 5997–6002. [Google Scholar] [CrossRef]
- Huai, Y.Y.; Plackowski, C.; Peng, Y.J. The galvanic interaction between gold and pyrite in the presence of ferric ions. Miner. Eng. 2018, 119, 236–243. [Google Scholar] [CrossRef]
| Sample | Cu | S | Fe | Ca | Si |
|---|---|---|---|---|---|
| Chalcocite | 78.61 | 18.95 | 0.018 | 0.017 | 0.053 |
| Time/h | Cu2S | Cu5FeS4 | Cu1.8S | Cu7.2S4 | Cu8S5 | CuS | S |
|---|---|---|---|---|---|---|---|
| 0 | √ | ||||||
| 0.5 | √ | √ | √ | √ | |||
| 1 | √ | √ | √ | √ | |||
| 2 | √ | √ | √ | √ | |||
| 4 | √ | √ | √ | ||||
| 6 | √ | √ | |||||
| 10 | √ | √ | |||||
| 15 | √ | √ |
| Components | Enthalpy (KJ/mol) | Entropy (J/mol/K) | Gibbs Free Energy (KJ/mol) (298.15 K) |
|---|---|---|---|
| Cu2S | −79.496 | 120.918 | −115.548 |
| Cu5FeS4 | −380.326 | 362.334 | −488.356 |
| CuS | −48.530 | 66.530 | −68.366 |
| Cu2+ (aq) | 64.768 | −99.579 | 94.458 |
| Fe3+ (aq) | −48.534 | −315.892 | 45.649 |
| Fe2+ (aq) | −89.119 | −137.654 | −48.078 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, C.; Yu, S.; Wang, X.; Zhao, H.; Qin, W.; Qiu, G.; Wang, J. Synchrotron Radiation XRD Investigation of the Fine Phase Transformation during Synthetic Chalcocite Acidic Ferric Sulfate Leaching. Minerals 2018, 8, 461. https://doi.org/10.3390/min8100461
Fang C, Yu S, Wang X, Zhao H, Qin W, Qiu G, Wang J. Synchrotron Radiation XRD Investigation of the Fine Phase Transformation during Synthetic Chalcocite Acidic Ferric Sulfate Leaching. Minerals. 2018; 8(10):461. https://doi.org/10.3390/min8100461
Chicago/Turabian StyleFang, Chaojun, Shichao Yu, Xingxing Wang, Hongbo Zhao, Wenqing Qin, Guanzhou Qiu, and Jun Wang. 2018. "Synchrotron Radiation XRD Investigation of the Fine Phase Transformation during Synthetic Chalcocite Acidic Ferric Sulfate Leaching" Minerals 8, no. 10: 461. https://doi.org/10.3390/min8100461
APA StyleFang, C., Yu, S., Wang, X., Zhao, H., Qin, W., Qiu, G., & Wang, J. (2018). Synchrotron Radiation XRD Investigation of the Fine Phase Transformation during Synthetic Chalcocite Acidic Ferric Sulfate Leaching. Minerals, 8(10), 461. https://doi.org/10.3390/min8100461






