The Influence of Common Monovalent and Divalent Chlorides on Chalcopyrite Flotation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Flotation Experiments
2.3. Zeta Potential Measurements
2.4. Contact Angle Measurements
2.5. X-ray Photoelectron Spectroscopy (XPS) Analysis
3. Results and Discussion
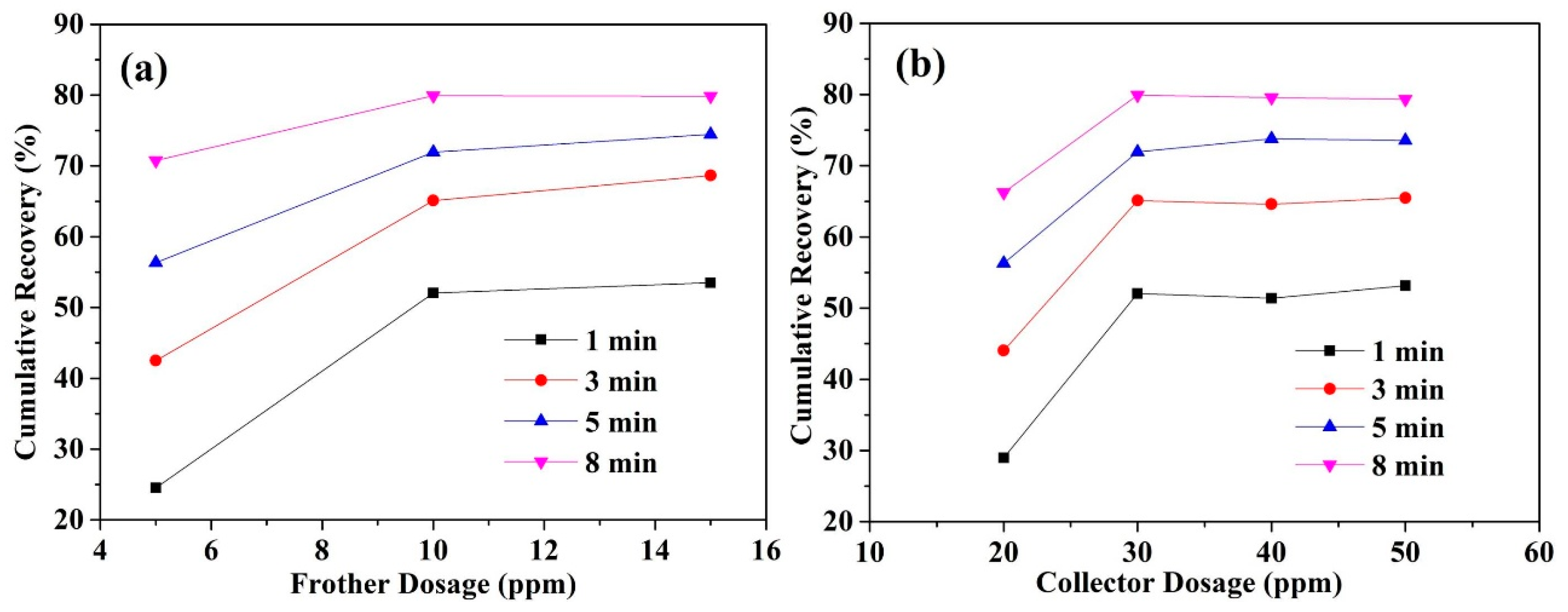
3.1. Effects of Reagent Dosages
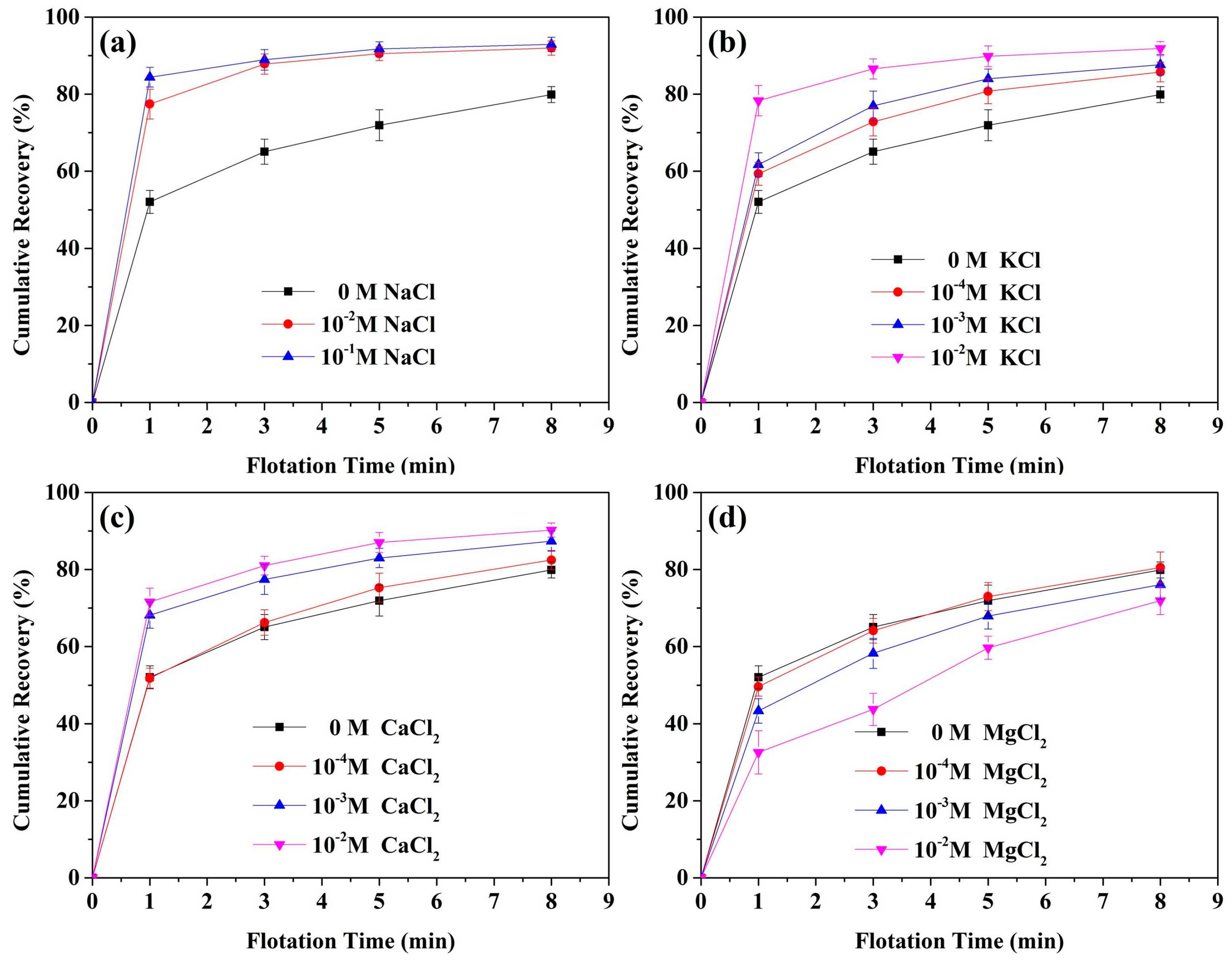
3.2. Effects of Salts
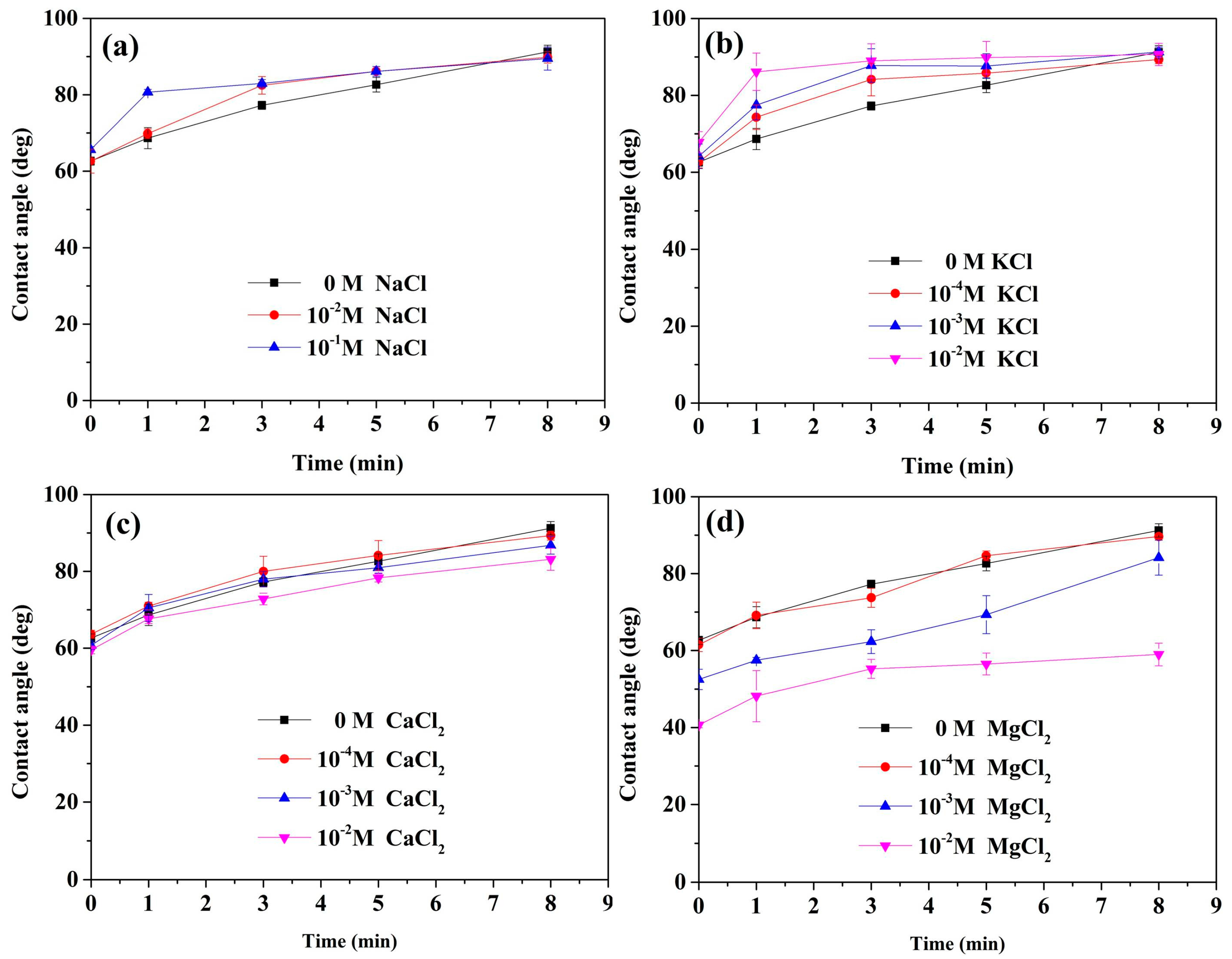
3.3. Contact Angle
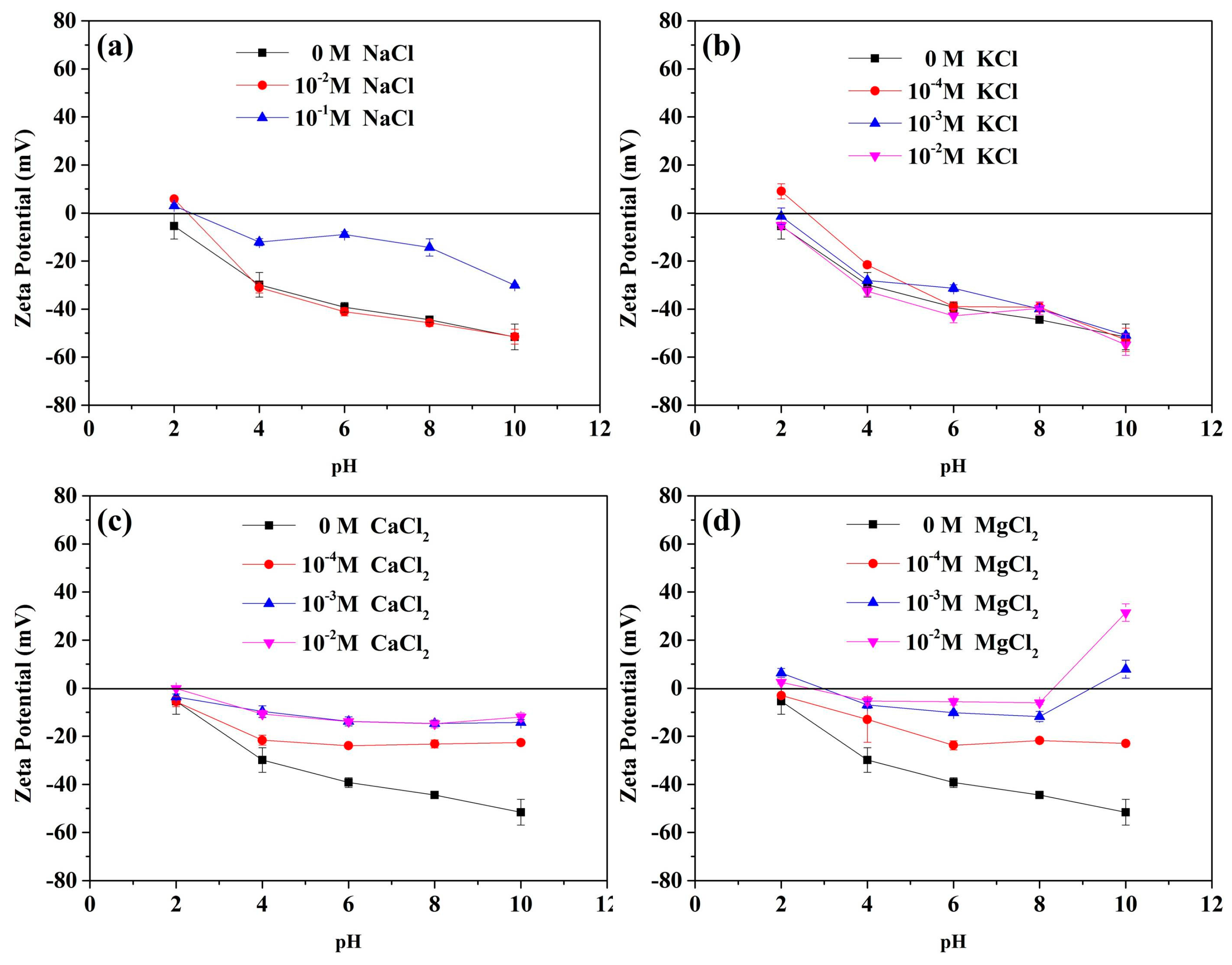
3.4. Zeta Potential
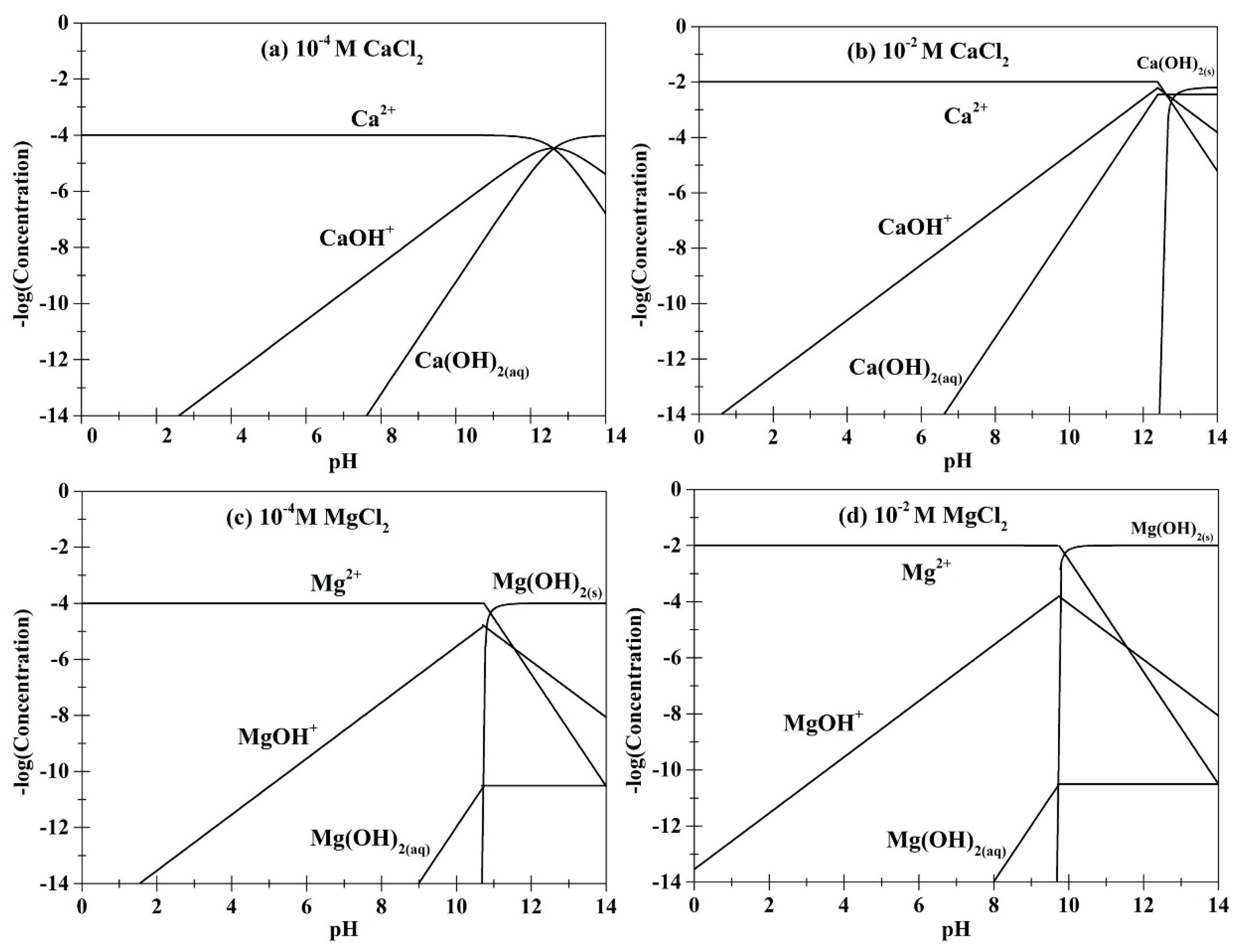
3.5. Solution Species
3.6. XPS Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93, 81–87. [Google Scholar] [CrossRef]
- Li, Y.; Kawashima, N.; Li, J.; Chandra, A.P.; Gerson, A.R. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite. Adv. Colloid Interface Sci. 2013, 197–198, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Hirajima, T.; Suyantara, G.P.W.; Ichikawa, O.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of Mg2+ and Ca2+ as divalent seawater cations on the floatability of molybdenite and chalcopyrite. Miner. Eng. 2016, 96–97, 83–93. [Google Scholar] [CrossRef]
- Fuerstenau, M.; Jameson, G.; Yoon, R.H. Froth Flotation a Century of Innovation; SME: Littleton, CO, USA, 2007. [Google Scholar]
- Bulatovic, S.M. Handbook of Flotation Reagents Chemistry, Theory and Practice: Flotation of Sulfide Ores; Elsevier Science: Burlington, VT, USA, 2007. [Google Scholar]
- Wang, B.; Peng, Y. The effect of saline water on mineral flotation—A critical review. Miner. Eng. 2014, 66–68, 13–24. [Google Scholar] [CrossRef]
- Castro, S.; Laskowski, J. Froth flotation in saline water. KONA Power Part. J. 2011, 29, 4–15. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Zanin, M. An investigation into the effect of water quality on froth stability. Adv. Powder Technol. 2012, 23, 493–497. [Google Scholar] [CrossRef]
- Moreno, P.; Aral, H.; Cuevas, J.; Monardes, A.; Adaro, M.; Norgate, T.; Bruckard, W. The use of seawater as process water at Las luces copper–molybdenum beneficiation plant in Taltal (Chile). Miner. Eng. 2011, 24, 852–858. [Google Scholar] [CrossRef]
- Crastro, S. Challenges in flotation of Cu-Mo sulfide ores in sea water. In Water in Mineral Processing: Proceedings of the First International Symposium; Drelich, J., Ed.; Society for Mining, Metallurgy, and Exploration: Seattle, WA, USA, 2012. [Google Scholar]
- Hancer, M.; Celik, M.S.; Miller, J.D. The significance of interfacial water structure in soluble salt flotation systems. J. Colloid Interface Sci. 2001, 235, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Wang, X.; Miller, J.D.; Cheng, F.; Jiao, Y. Bubble attachment time and FTIR analysis of water structure in the flotation of sylvite, bischofite and carnallite. Miner. Eng. 2011, 24, 108–114. [Google Scholar] [CrossRef]
- Parsons, D.F.; Boström, M.; Maceina, T.J.; Salis, A.; Ninham, B.W. Why direct or reversed Hofmeister series interplay of hydration, non-electrostatic potentials, and ion size. Langmuir 2009, 26, 3323–3328. [Google Scholar] [CrossRef] [PubMed]
- Weissenborn, P.K.; Pugh, R.J. Surface tension of aqueous solutions of electrolytes: Relationship with ion hydration, oxygen solubility, and bubble coalescence. Colloid Interface Sci. 1996, 184, 550–563. [Google Scholar] [CrossRef]
- Klassen, V.I.; Mokrousov, V.A. An Introduction to the Theory of Flotation, 2nd ed.; Butterworths: London, UK, 1963. [Google Scholar]
- Mancera, R.L. Computer simulation of the effect of salt on the hydrophobic effect. Chemistry 1998, 94, 3549–3559. [Google Scholar] [CrossRef]
- Medrzycka, K.B.; Zwierzykowski, W. The effect of the nature of inorganic ions on hydrocarbon flotation. Sep. Sci. Technol. 1988, 23, 719–729. [Google Scholar] [CrossRef]
- Henry, C.L.; Craig, V.S.J. The link between ion specific bubble coalescence and Hofmeister effects is the partitioning of ions within the interface. Langmuir 2010, 26, 6478–6483. [Google Scholar] [CrossRef] [PubMed]
- Castro, S.; Miranda, C.; Toledo, P.; Laskowski, J.S. Effect of frothers on bubble coalescence and foaming in electrolyte solutions and seawater. Int. J. Miner. Process. 2013, 124, 8–14. [Google Scholar] [CrossRef]
- Farrokhpay, S. The significance of froth stability in mineral flotation—A review. Adv. Colloid Interface Sci. 2011, 166, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Corin, K.C.; Reddy, A.; Miyen, L.; Wiese, J.G.; Harris, P.J. The effect of ionic strength of plant water on valuable mineral and gangue recovery in a platinum bearing ore from the Merensky reef. Miner. Eng. 2011, 24, 131–137. [Google Scholar] [CrossRef]
- Ramos, O.; Castro, S.; Laskowski, J.S. Copper–molybdenum ores flotation in sea water: Floatability and frothability. Miner. Eng. 2013, 53, 108–112. [Google Scholar] [CrossRef]
- Laskowski, J.S.; Castro, S.; Ramos, O. Effect of seawater main components on frothability in the flotation of Cu-Mo sulfide ore. Physicochem. Probl. Miner. Process. 2013, 50, 17–29. [Google Scholar]
- Quinn, J.J.; Sovechles, J.M.; Finch, J.A.; Waters, K.E. Critical coalescence concentration of inorganic salt solutions. Miner. Eng. 2014, 58, 1–6. [Google Scholar] [CrossRef]
- Quinn, J.J.; Kracht, W.; Gomez, C.O.; Gagnon, C.; Finch, J.A. Comparing the effect of salts and frother (MIBC) on gas dispersion and froth properties. Miner. Eng. 2007, 20, 1296–1302. [Google Scholar] [CrossRef]
- Wang, Y.; Peng, Y.; Nicholson, T.; Lautern, R.A. The role of cations in copper flotation in the presence of bentonite. Miner. Eng. 2016, 96–97, 108–112. [Google Scholar] [CrossRef]
- Suyantara, G.P.W.; Hirajima, T.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of kerosene emulsion in MgCl2 solution on the kinetics of bubble interactions with molybdenite and chalcopyrite. Colloids Surf. A Physicochem. Eng. Asp. 2016, 501, 98–113. [Google Scholar] [CrossRef]
- Craig, V.S.J. Bubble coalescence and specific-ion effects. Curr. Opin. Colloid Interface Sci. 2004, 9, 178–184. [Google Scholar] [CrossRef]
- Jeldres, R.I.; Arancibia-Bravo, M.P.; Reyes, A.; Aguirre, C.E.; Cortes, L.; Cisternas, L.A. The impact of seawater with calcium and magnesium removal for the flotation of copper-molybdenum sulphide ores. Miner. Eng. 2017, 109, 10–13. [Google Scholar] [CrossRef]
- Aral, H.; Mead, S.; Cuevas, J.; Davey, K.; Bruckard, W. Desperate times call for desperate measures—The use of seawater in mining and mineral processing. In Proceedings of the AusIMM Sustainable Mining Conference, Kalgoorlie, Australia, 17–19 August 2010. [Google Scholar]
- Shirley, D.A. High-resolution X-ray photoemission spectrum of the valence bands of gold. Phys. Rev. B 1972, 5, 4709–4714. [Google Scholar] [CrossRef]
- Metson, J.B. Charge compensation and binding energy referencing in XPS analysis. Surf. Interface Anal. 1999, 27, 1069–1072. [Google Scholar] [CrossRef]
- Nagaraj, D.R.; Farinato, R. Chemical factor effects in saline and hypersaline waters in the flotation of Cu and Cu-Mo ores. In Proceedings of the Processing Congress Presented at the XXVII International Mineral, Santiago, Chile, 20–24 October 2014. [Google Scholar]
- Blake, T.D.; Kitchener, J.A. Stability of aqueous films on hydrophobic methylated silica. J. Chem. Soc. 1972, 68, 1435–1442. [Google Scholar] [CrossRef]
- Laskowski, J.S. Coal flotation in solution with a raised concentration of inorganic salts. Colliery Guard. 1965, 211, 361–366. [Google Scholar]
- Li, C.; Somasundaran, P. Reversal of bubble charge in multivalent inorganic salt solutions—Effect of aluminum. J. Colloid Interface Sci. 1992, 148, 587–591. [Google Scholar] [CrossRef]
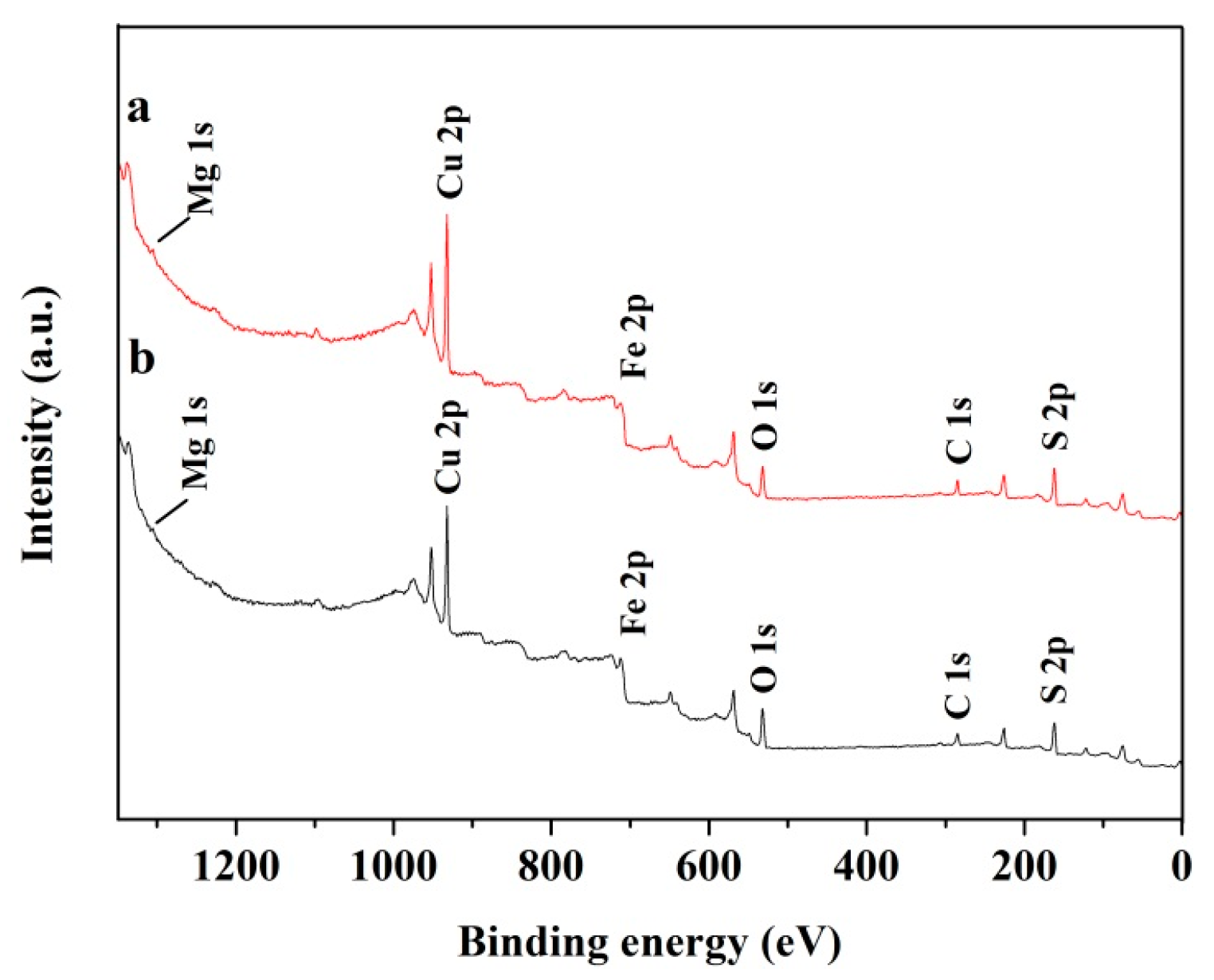
| MgCl2 Concentration (M) | at % | ||||
|---|---|---|---|---|---|
| Cu 2p | S 2p | Fe 2p | O 1s | Mg 1s | |
| 10−2 | 24 | 38 | 6 | 30 | 2 |
| 10−4 | 21 | 35 | 8 | 36 | <1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Li, W.; Xiao, Q.; He, N.; Ren, Z.; Lartey, C.; Gerson, A.R. The Influence of Common Monovalent and Divalent Chlorides on Chalcopyrite Flotation. Minerals 2017, 7, 111. https://doi.org/10.3390/min7070111
Li Y, Li W, Xiao Q, He N, Ren Z, Lartey C, Gerson AR. The Influence of Common Monovalent and Divalent Chlorides on Chalcopyrite Flotation. Minerals. 2017; 7(7):111. https://doi.org/10.3390/min7070111
Chicago/Turabian StyleLi, Yubiao, Wanqing Li, Qing Xiao, Nan He, Zijie Ren, Clement Lartey, and Andrea R. Gerson. 2017. "The Influence of Common Monovalent and Divalent Chlorides on Chalcopyrite Flotation" Minerals 7, no. 7: 111. https://doi.org/10.3390/min7070111
APA StyleLi, Y., Li, W., Xiao, Q., He, N., Ren, Z., Lartey, C., & Gerson, A. R. (2017). The Influence of Common Monovalent and Divalent Chlorides on Chalcopyrite Flotation. Minerals, 7(7), 111. https://doi.org/10.3390/min7070111







