Heterotrophic Microbial Stimulation through Biosolids Addition for Enhanced Acid Mine Drainage Control
Abstract
:1. Introduction
2. Materials and Methods
2.1. Kinetic Leach Columns
2.2. Biosolids Leach Column Inoculation and Treatment Solutions
2.3. Microbiological Analysis
2.4. Solid Phase Analysis
2.5. Chemistry Analysis of Column Leachates
2.6. Data and Statistical Analyses
3. Results and Discussion
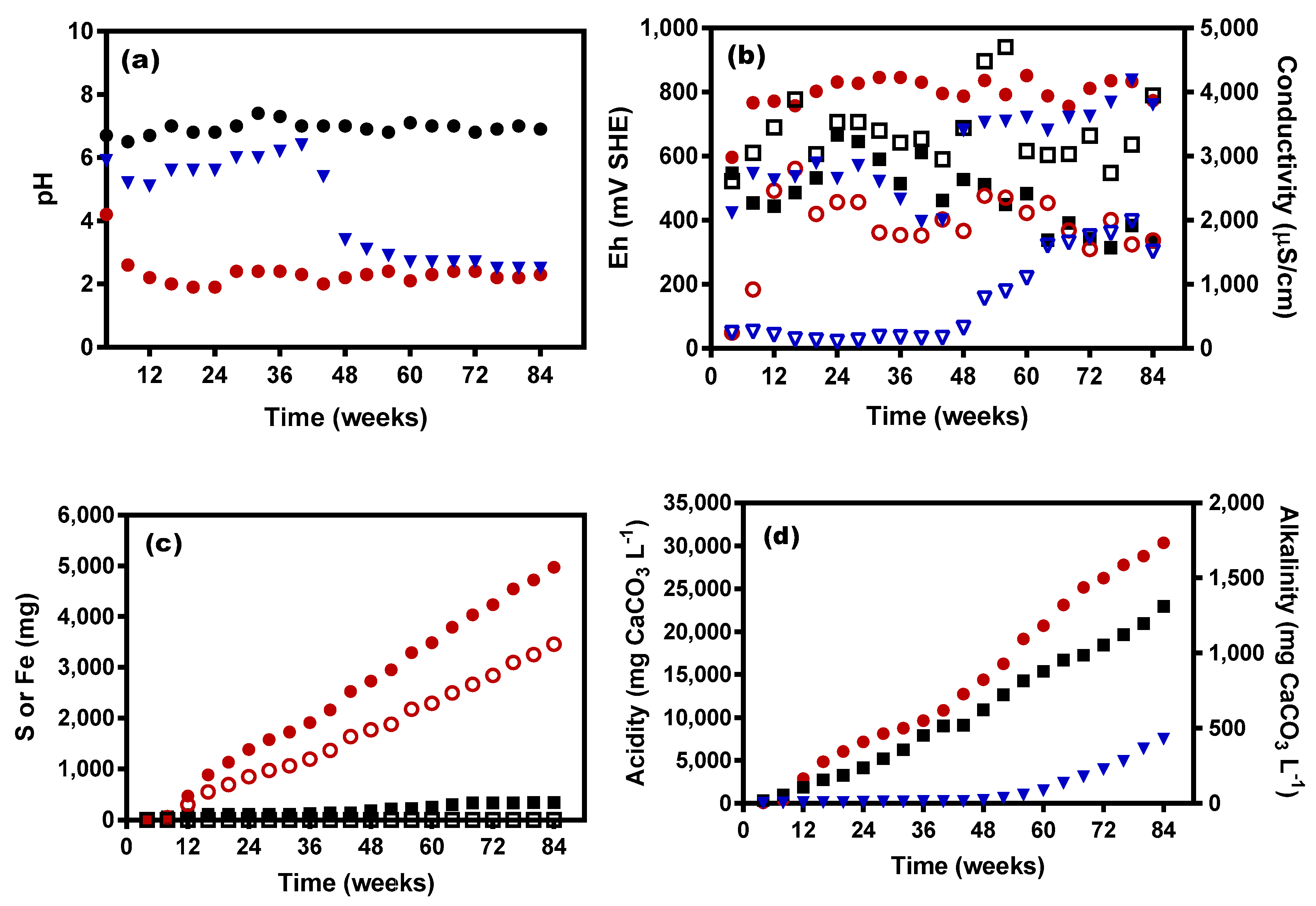
3.1. KLC Characteristics
3.2. Leachate Analyses
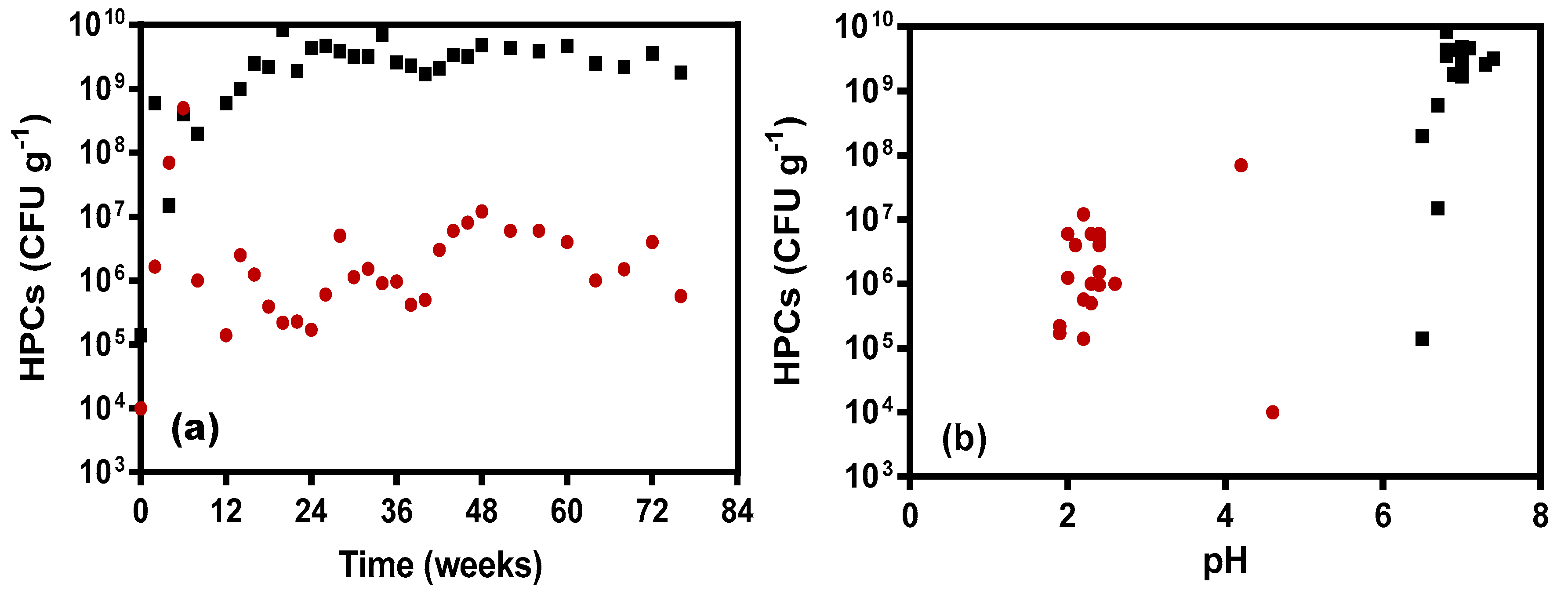
3.3. Microbial Response to Leachate Changes
3.4. Microbial O2 Demand and Reduced Acidity Levels
3.5. Pyrite Surface Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Singer, P.C.; Stumm, W. Acidic Mine Drainage: The Rate-Determining Step. Science 1970, 167, 1121–1123. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Neculita, C.M.; Zagury, G.J. Biological treatment of highly contaminated acid mine drainage in batch reactors: Long-term treatment and reactive mixture characterization. J. Hazard. Mater. 2008, 157, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.; Strongin, D. Surface reactivity of pyrite and related sulfides. Surf. Sci. Rep. 2009, 64, 1–45. [Google Scholar] [CrossRef]
- Chandra, A.P.; Gerson, A.R. The mechanisms of pyrite oxidation and leaching: A fundamental perspective. Surf. Sci. Rep. 2010, 65, 293–315. [Google Scholar] [CrossRef]
- Moreno, N.; Querol, X.; Ayora, C.; Pereira, C.F.; Janssen-Jurkovicova, M. Utilization of zeolites synthesized from coal fly ash for the purification of acid mine waters. Environ. Sci. Technol. 2001, 35, 3526–3534. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, K.B. New perspectives in acid mine drainage microbiology. Hydrometallurgy 2010, 104, 448–453. [Google Scholar] [CrossRef]
- Smart, R.S.C.; Miller, S.D.; Stewart, W.S.; Rusdinar, Y.; Schumann, R.E.; Kawashima, N.; Li, J. In situ calcite formation in limestone-saturated water leaching of acid rock waste. Sci. Total Environ. 2010, 408, 3392–3402. [Google Scholar] [CrossRef] [PubMed]
- Harries, J. Acid Mine Drainage in Australia: Its Extent and Potential Future Liability; Supervising Scientist Report; Office of the Supervising Scientist, Australian Government: Canberra, Austrlia, 1997; Volume 125, pp. 1–147.
- Chatwin, T.D. Business case: Gaining a social license to mine—ARD prevention. In Proceedings of the Sixth Australian Workshop on Acid Drainage; Bell, L.C., Barrie, B.M.D., Braddock, B., McLean, R.W., Eds.; ACMER, Sustainable Minerals Institute: Brisbane, Australia, 2008; pp. 1–11. ISBN 978-0-9750304-6-2. [Google Scholar]
- RoyChowdhury, A.; Sarkar, D.; Datta, R. Remediation of acid mine drainage-impacted water. Curr. Pollut. Rep. 2015, 1, 131–141. [Google Scholar] [CrossRef]
- United States. Acid Drainage from Mines on the National Forests: A Management Challenge; Program aid 1505; Forest Service: Washington, DC, USA; p. 12.
- USEPA. Technical Document: Acid Mine Drainage Prediction; EPA 530-R-94-036, NTIS PB94-201829; Office of Solid Waste: Washington, DC, USA, 1994.
- Jennings, S.R.; Blicker, P.S.; Neuman, D.R. Acid Mine Drainage and Effects on Fish Health and Ecology: A Review; Reclamation Research Group: Bozeman, MT, USA, 2008. [Google Scholar]
- USDA Forest Service. Wildland Waters. Issue 4. Winter 2005; FS-812. Available online: http://www.fs.fed.us (accessed on 1 May 2017).
- Kim, A.; Heisey, B.; Kleinmann, R.; Deul, M. Acid Mine Drainage: Control and Abatement Research. Information circular/1982; Pittsburgh Research Center: Pittsburgh, PA, USA, 1982. [Google Scholar]
- Benner, S.G.; Blowes, D.W.; Ptacek, C.J. A Full-Scale Porous Reactive Wall for Prevention of Acid Mine Drainage. Groundw. Monit. Remediat. 1997, 17, 99–107. [Google Scholar] [CrossRef]
- Adams, D.J.; Ogden, U.T.; Pennington, P.; Tachie-Menson, S.; Gutierrez, L.A.F. The role of microorganisms in acid rock drainage. In Proceedings of the SME Annual Meeting, Salt Lake City, UT, USA, 28 February–2 March 2005. [Google Scholar]
- Rohwerder, T.; Schippers, A.; Sand, W. Determination of reaction energy values for biological pyrite oxidation by calorimetry. Thermochim. Acta 1998, 309, 79–85. [Google Scholar] [CrossRef]
- Baker, B.J.; Banfield, J.F. Microbial communities in acid mine drainage. FEMS Microbiol. Ecol. 2003, 44, 139–152. [Google Scholar] [CrossRef]
- Lefebvre, R.; Hockley, D.; Smolensky, J.; Gélinas, P. Multiphase transfer processes in waste rock piles producing acid mine drainage: 1: Conceptual model and system characterization. J. Contam. Hydrol. 2001, 52, 137–164. [Google Scholar] [CrossRef]
- Silverman, M.P.; Ehrlich, H.L. Microbial Formation and Degradation of Minerals. In Advances in Applied Microbiology; Wayne, W.U., Ed.; Academic Press: Cambridge, MA, USA, 1964; pp. 153–206. [Google Scholar]
- Johnson, D. Recent Developments in Microbiological Approaches for Securing Mine Wastes and for Recovering Metals from Mine Waters. Minerals 2014, 4, 279–292. [Google Scholar] [CrossRef]
- Evangelou, V.P.; Zhang, Y.L. A review: Pyrite oxidation mechanisms and acid mine drainage prevention. Crit. Rev. Environ. Sci. Technol. 1995, 25, 141–199. [Google Scholar] [CrossRef]
- Afzal Ghauri, M.; Okibe, N.; Johnson, D.B. Attachment of acidophilic bacteria to solid surfaces: The significance of species and strain variations. Hydrometallurgy 2007, 85, 72–80. [Google Scholar] [CrossRef]
- Schumann, R. Passivating surface layer formation on pyrite in neutral rock drainage. In Proceedings of the 8th International Conference on Acid Rock Drainage (ICARD) and Securing the Future: Mining, Metals & the Environment in a Sustainable Society 2009, Skelleftea, Sweden, 22–26 June 2009. [Google Scholar]
- Miller, S.D.; Schumann, R.; Smart, R.; Rusdinar, Y. ARD Control by Limestone Induced Armouring and Passivation of Pyrite Minerals Surfaces. In Proceedings of the 8th International Conference on Acid Rock Drainage (ICARD) and Securing the Future: Mining, Metals & the Environment in a Sustainable Society 2009, Skelleftea, Sweden, 22–26 June 2009. [Google Scholar]
- Fan, R.; Schumann, R.; Smart, R.; Rusdinar, Y. The formation of surface passivating layers on pyrite for reduced acid rock drainage. Environ. Sci. Technol 2017. submitted. [Google Scholar]
- Zeng, S.; Li, J.; Schumann, R.; Smart, R. Effect of pH and dissolved silicate on the formation of surface passivation layers for reducing pyrite oxidation. Comput. Water Energy Environ. Eng. 2013, 2, 50. [Google Scholar] [CrossRef]
- Gerson, A.R.; Li, J.; Smart, R.S.C.; Saint, C.P.; Short, M.D.; Schumann, R.C.; Jarvie-Eggart, M.; Muga, H. Responsible Management of Acid Mine Wastes: Geochemical and Microbiological Resources; SME Publications: Littleton, CO, USA, 2014; pp. 519–524. [Google Scholar]
- Johnson, D.B. Biodiversity and ecology of acidophilic microorganisms. FEMS Microbiol. Ecol. 1998, 27, 307–317. [Google Scholar] [CrossRef]
- Lefevre, E.; Pereyra, L.P.; Hiibel, S.R.; Perrault, E.M.; De Long, S.K.; Reardon, K.F.; Pruden, A. Molecular assessment of the sensitivity of sulfate-reducing microbial communities remediating mine drainage to aerobic stress. Water Res. 2013, 47, 5316–5325. [Google Scholar] [CrossRef] [PubMed]
- Behum, P.T.; Lefticariu, L.; Bender, K.S.; Segid, Y.T.; Burns, A.S.; Pugh, C.W. Remediation of coal-mine drainage by a sulfate-reducing bioreactor: A case study from the Illinois coal basin, USA. Appl. Geochem. 2011, 26, S162–S166. [Google Scholar] [CrossRef]
- Lindsay, M.B.; Blowes, D.W.; Condon, P.D.; Ptacek, C.J. Organic carbon amendments for passive in situ treatment of mine drainage: Field experiments. Appl. Geochem. 2011, 26, 1169–1183. [Google Scholar] [CrossRef]
- McMahon, M.J.L.; Daugulis, A.J. Enhancement of biogenic sulfide production in a packed-bed bioreactor via critical inoculum design and carrier material selection. Biotechnol. Bioeng. 2008, 100, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Neculita, C.-M.; Zagury, G.J.; Bussière, B. Passive treatment of acid mine drainage in bioreactors using sulfate-reducing bacteria. J. Environ. Qual. 2007, 36, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Postgate, J.R. The Sulphate Reducing Bacteria, 2nd ed.; Cambridge University Press: Cambridge, UK, 1984. [Google Scholar]
- Widdel, F. Microbiology and Ecology of Sulfate- and Sulfur-Reducing Bacteria. In Biology of Anaerobic Microorganisms; John Wiley & Sons: New York, NY, USA, 1988; pp. 469–585. [Google Scholar]
- Roetman, E.T. The sterilization of sewage by acid mine water. Master’s Thesis, West Virginia University, Morgantown, WV, USA, 1932. [Google Scholar]
- Deng, D.; Weidhaas, J.L.; Lin, L.-S. Kinetics and microbial ecology of batch sulfidogenic bioreactors for co-treatment of municipal wastewater and acid mine drainage. J. Hazard. Mater. 2016, 305, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.L.; Younger, P.L. The co-treatment of sewage and mine waters in aerobic wetlands. Eng. Geol. 2006, 85, 53–61. [Google Scholar] [CrossRef]
- McCullough, C.D.; Lund, M.A.; May, J.M. Field-scale demonstration of the potential for sewage to remediate acidic mine waters. Mine Water Environ. 2008, 27, 31–39. [Google Scholar] [CrossRef]
- Wei, X.; Viadero, R.C.; Bhojappa, S. Phosphorus removal by acid mine drainage sludge from secondary effluents of municipal wastewater treatment plants. Water Res. 2008, 42, 3275–3284. [Google Scholar] [CrossRef] [PubMed]
- Strosnider, W.H.; Winfrey, B.K.; Nairn, R.W. Novel passive co-treatment of acid mine drainage and municipal wastewater. J. Environ. Qual. 2011, 40, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Strosnider, W.; Winfrey, B.; Nairn, R. Alkalinity generation in a novel multi-stage high-strength acid mine drainage and municipal wastewater passive co-treatment system. Mine Water Environ. 2011, 30, 47–53. [Google Scholar] [CrossRef]
- Strosnider, W.; Winfrey, B.; Nairn, R. Biochemical oxygen demand and nutrient processing in a novel multi-stage raw municipal wastewater and acid mine drainage passive co-treatment system. Water Res. 2011, 45, 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Ruihua, L.; Lin, Z.; Tao, T.; Bo, L. Phosphorus removal performance of acid mine drainage from wastewater. J. Hazard. Mater. 2011, 190, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Winfrey, B.; Strosnider, W.; Nairn, R.; Strevett, K. Highly effective reduction of fecal indicator bacteria counts in an ecologically engineered municipal wastewater and acid mine drainage passive co-treatment system. Ecol. Eng. 2010, 36, 1620–1626. [Google Scholar] [CrossRef]
- Strosnider, W.; Winfrey, B.; Peer, R.; Nairn, R. Passive co-treatment of acid mine drainage and sewage: Anaerobic incubation reveals a regeneration technique and further treatment possibilities. Ecol. Eng. 2013, 61, 268–273. [Google Scholar] [CrossRef]
- Younger, P.L.; Henderson, R. Synergistic wetland treatment of sewage and mine water: Pollutant removal performance of the first full-scale system. Water Res. 2014, 55, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Smart, R.; Skinner, B.; Levay, G.; Gerson, A.; Thomas, J.; Sobieraj, H.; Schumann, R.; Weisener, C.; Weber, P.; Miller, S. ARD Test Handbook; AMIRA International: Melbourne, Austrila, 2002. [Google Scholar]
- Pepper, I.L.; Zerzghi, H.G.; Bengson, S.A.; Iker, B.C.; Banerjee, M.J.; Brooks, J.P. Bacterial populations within copper mine tailings: Long-term effects of amendment with Class A biosolids. J. Appl. Microbiol. 2012, 113, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Rietveld, H. Line profiles of neutron powder-diffraction peaks for structure refinement. Acta Crystallogr. 1967, 22, 151–152. [Google Scholar] [CrossRef]
- Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Kirby, C.S.; Cravotta, C.A. Net alkalinity and net acidity 2: Practical considerations. Appl. Geochem. 2005, 20, 1941–1964. [Google Scholar] [CrossRef]
- Wagner, B.A.; Venkataraman, S.; Buettner, G.R. The rate of oxygen utilization by cells. Free Radic. Biol. Med. 2011, 51, 700–712. [Google Scholar] [CrossRef] [PubMed]


| Components | Phase Impurities | Quantity in KLC | S | C | Fe | Si | HPC (CFU g−1) |
|---|---|---|---|---|---|---|---|
| Quartz | - | 81.3 | 0.04 | 0.13 | 0.16 | 42.7 | - |
| K-feldspar (microcline) | 1 quartz | 10.0 | 0.04 | 0.15 | 0.06 | 31.6 | - |
| Chlorite | 5 phlogopite | 5.0 | 0.06 | 0.17 | 18.00 | 13.0 | - |
| Pyrite | - | 3.7 | 51.50 | 0.16 | 42.80 | 0.0 | - |
| Calcite | 1 quartz | - | 0.31 | 11.84 | 0.03 | 0.0 | - |
| Control KLC | - | - | 2.00 | - | - | - | 1 × 104 |
| BS + CSW KLC | - | - | 2.00 | - | - | - | 1.4 × 105 |
| CSW KLC | - | - | 2.00 | - | - | - | - |
| Parameter | pH | Eh | Conductivity | Acidity | S | Fe | HPCs |
|---|---|---|---|---|---|---|---|
| Time | −0.283 ns | 0.385 ns | 0.131 ns | 0.344 ns | 0.167 ns | 0.353 ns | 0.314 ns |
| pH | −0.776 *** | −0.788 *** | −0.476 * | −0.646 ** | −0.558 ** | −0.391 ns | |
| S | −0.646 ** | 0.250 ns | 0.825 *** | 0.840 *** | 0.941 *** | −0.013 ns |
| Parameter | pH | Eh | Conductivity | Alkalinity | S | Fe | HPCs |
|---|---|---|---|---|---|---|---|
| Time | 0.272 ns | −0.663 ** | 0.112 ns | 0.528 * | −0.030 ns | 0 | 0.571 ** |
| pH | 0.169 ns | −0.010 ns | 0.268 ns | −0.208 ns | 0 | 0.526 * | |
| S | −0.208 ns | −0.218 ns | −0.044 ns | 0.133 ns | - | 0 | −0.072 ns |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogbughalu, O.T.; Gerson, A.R.; Qian, G.; Smart, R.S.C.; Schumann, R.C.; Kawashima, N.; Fan, R.; Li, J.; Short, M.D. Heterotrophic Microbial Stimulation through Biosolids Addition for Enhanced Acid Mine Drainage Control. Minerals 2017, 7, 105. https://doi.org/10.3390/min7060105
Ogbughalu OT, Gerson AR, Qian G, Smart RSC, Schumann RC, Kawashima N, Fan R, Li J, Short MD. Heterotrophic Microbial Stimulation through Biosolids Addition for Enhanced Acid Mine Drainage Control. Minerals. 2017; 7(6):105. https://doi.org/10.3390/min7060105
Chicago/Turabian StyleOgbughalu, Omy T., Andrea R. Gerson, Gujie Qian, Roger St. C. Smart, Russell C. Schumann, Nobuyuki Kawashima, Rong Fan, Jun Li, and Michael D. Short. 2017. "Heterotrophic Microbial Stimulation through Biosolids Addition for Enhanced Acid Mine Drainage Control" Minerals 7, no. 6: 105. https://doi.org/10.3390/min7060105
APA StyleOgbughalu, O. T., Gerson, A. R., Qian, G., Smart, R. S. C., Schumann, R. C., Kawashima, N., Fan, R., Li, J., & Short, M. D. (2017). Heterotrophic Microbial Stimulation through Biosolids Addition for Enhanced Acid Mine Drainage Control. Minerals, 7(6), 105. https://doi.org/10.3390/min7060105







