The Influence of Ca2+ and pH on the Interaction between PAHs and Molybdenite Edges
Abstract
:1. Introduction
2. Materials and Methods
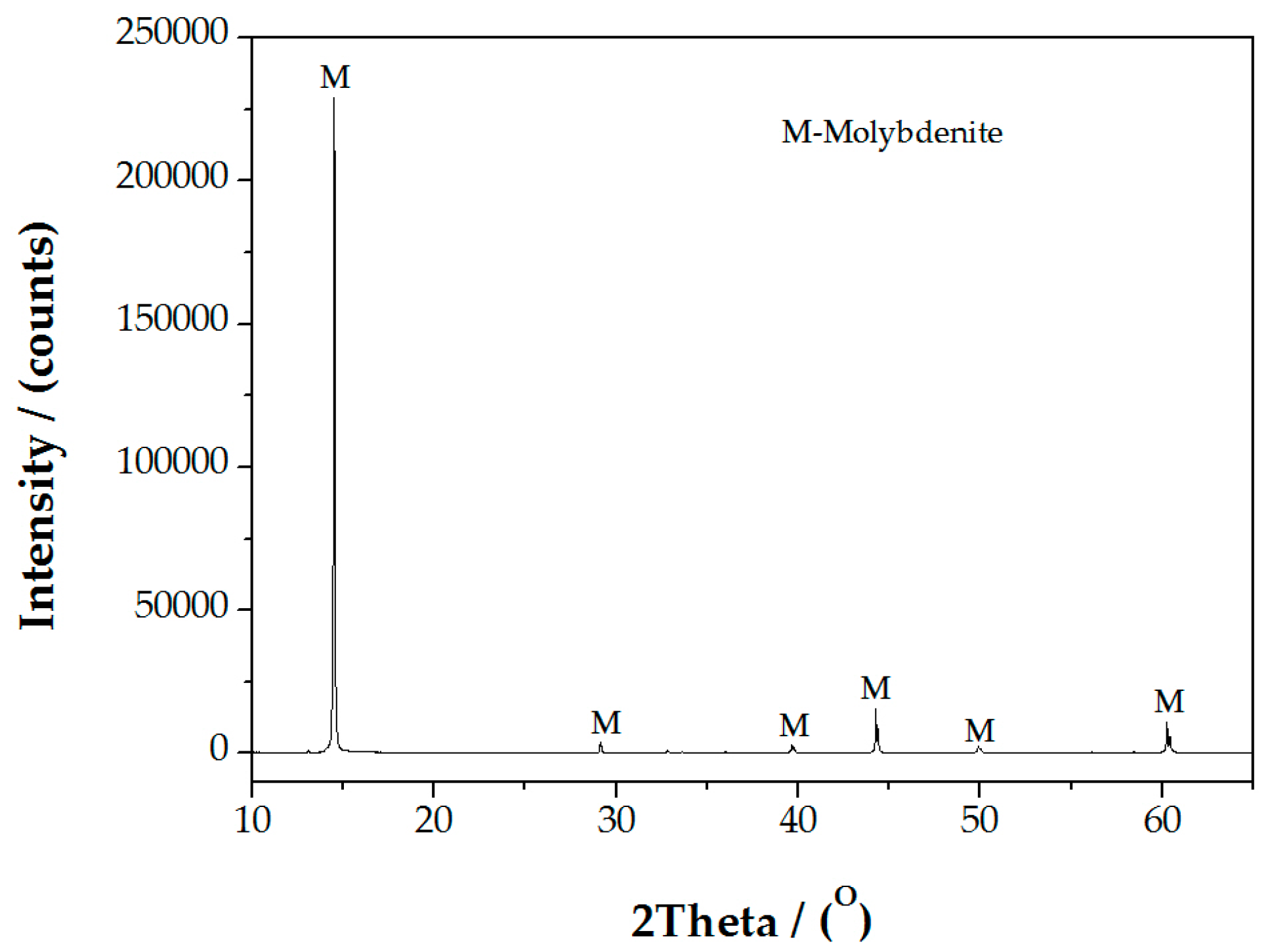
2.1. Materials and Reagents
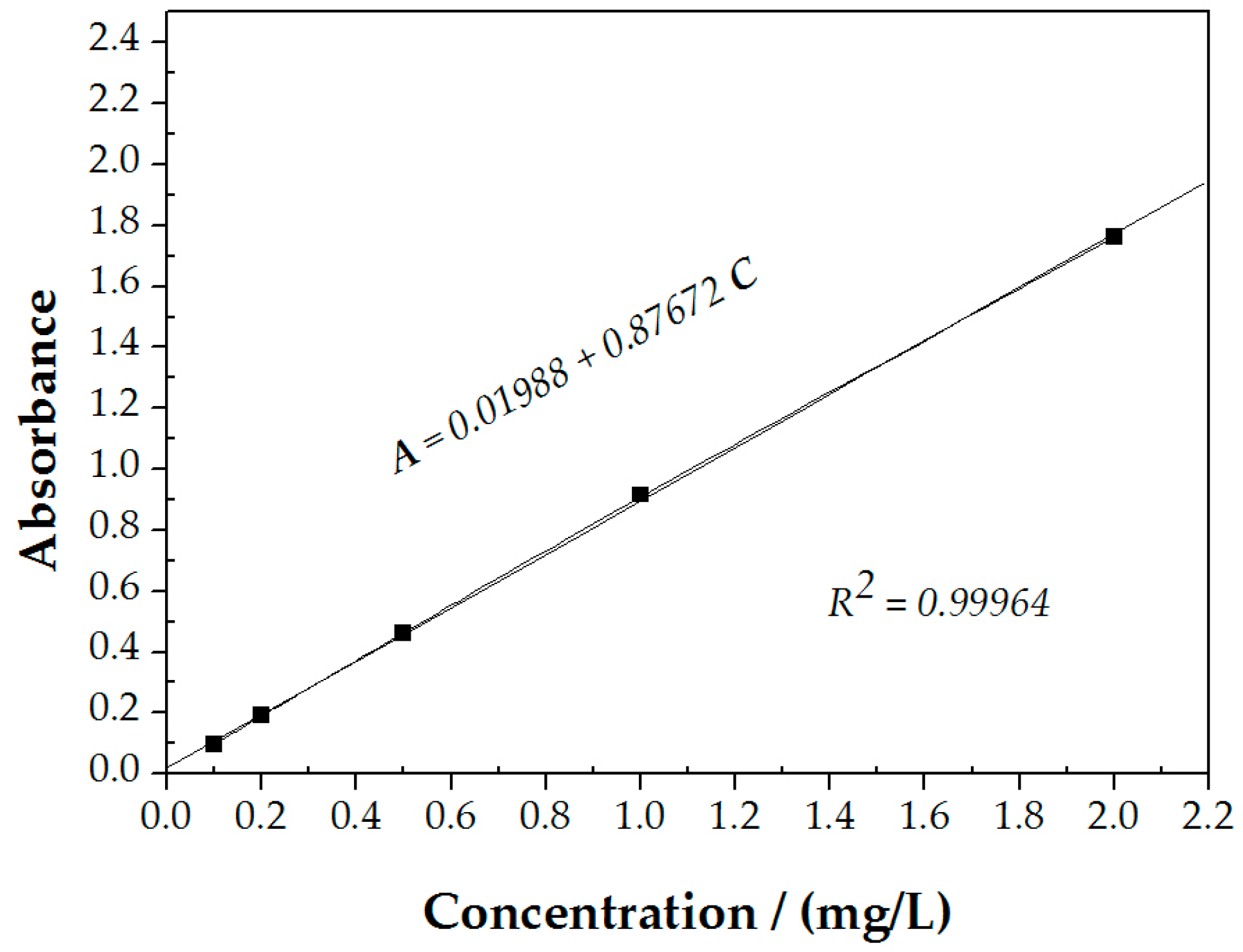
2.2. Ultraviolet-Visible Spectrophotometer Measurements
2.3. Zeta Potential Measurements
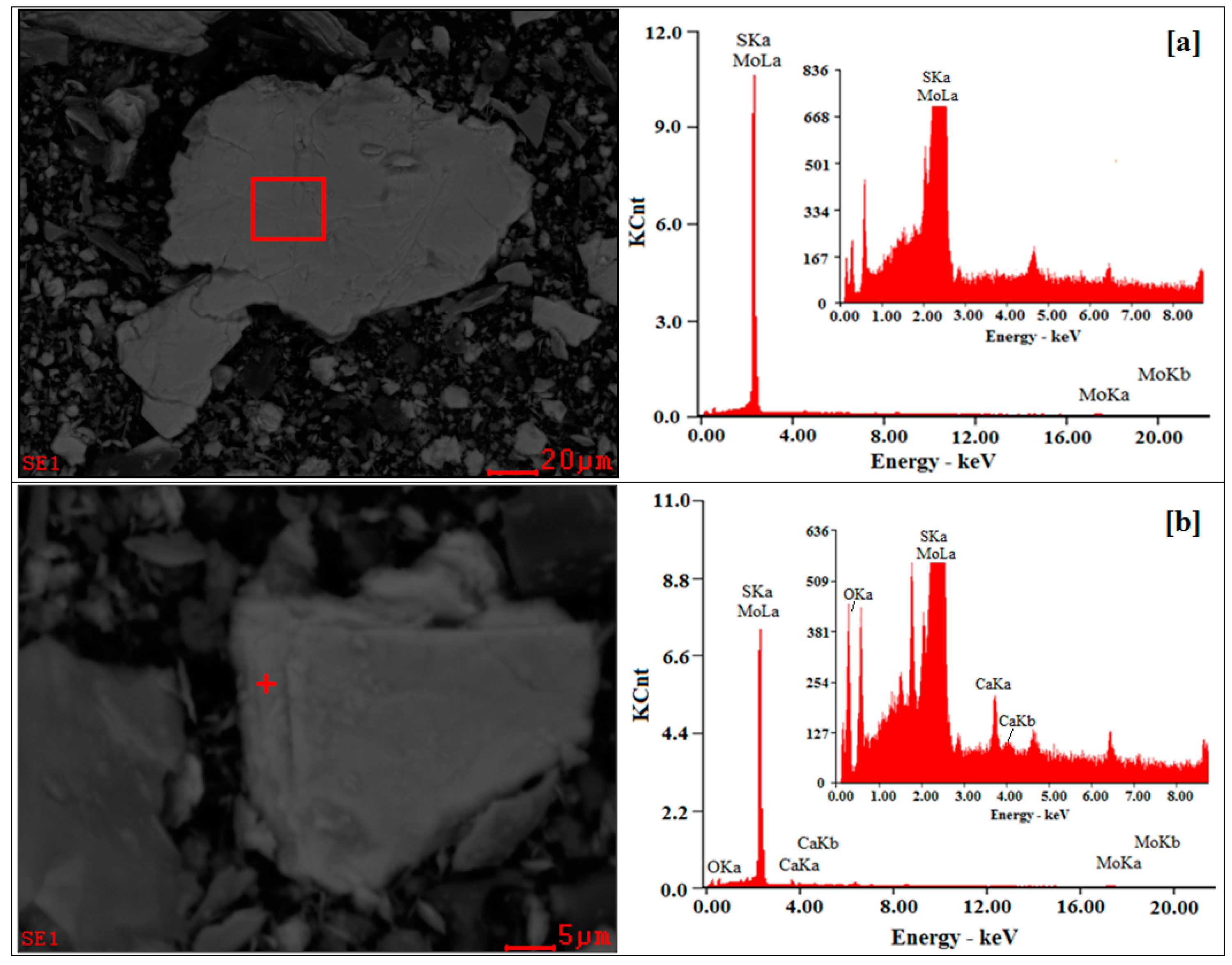
2.4. Scanning Electron Microscopy and Energy Dispersive X-Ray Spectrometer (SEM-EDS) Analysis
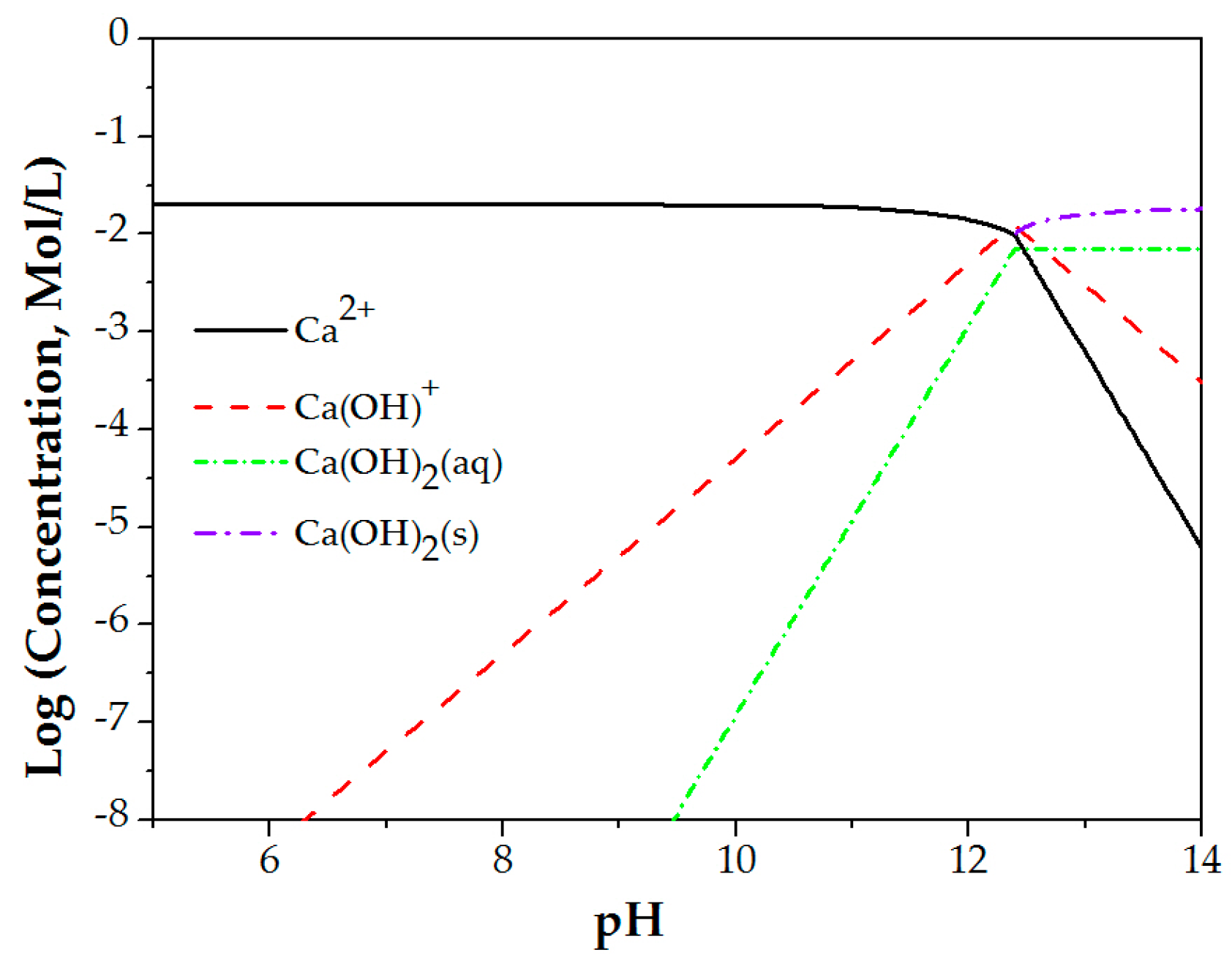
2.5. Calcium Speciation
3. Results and Discussion
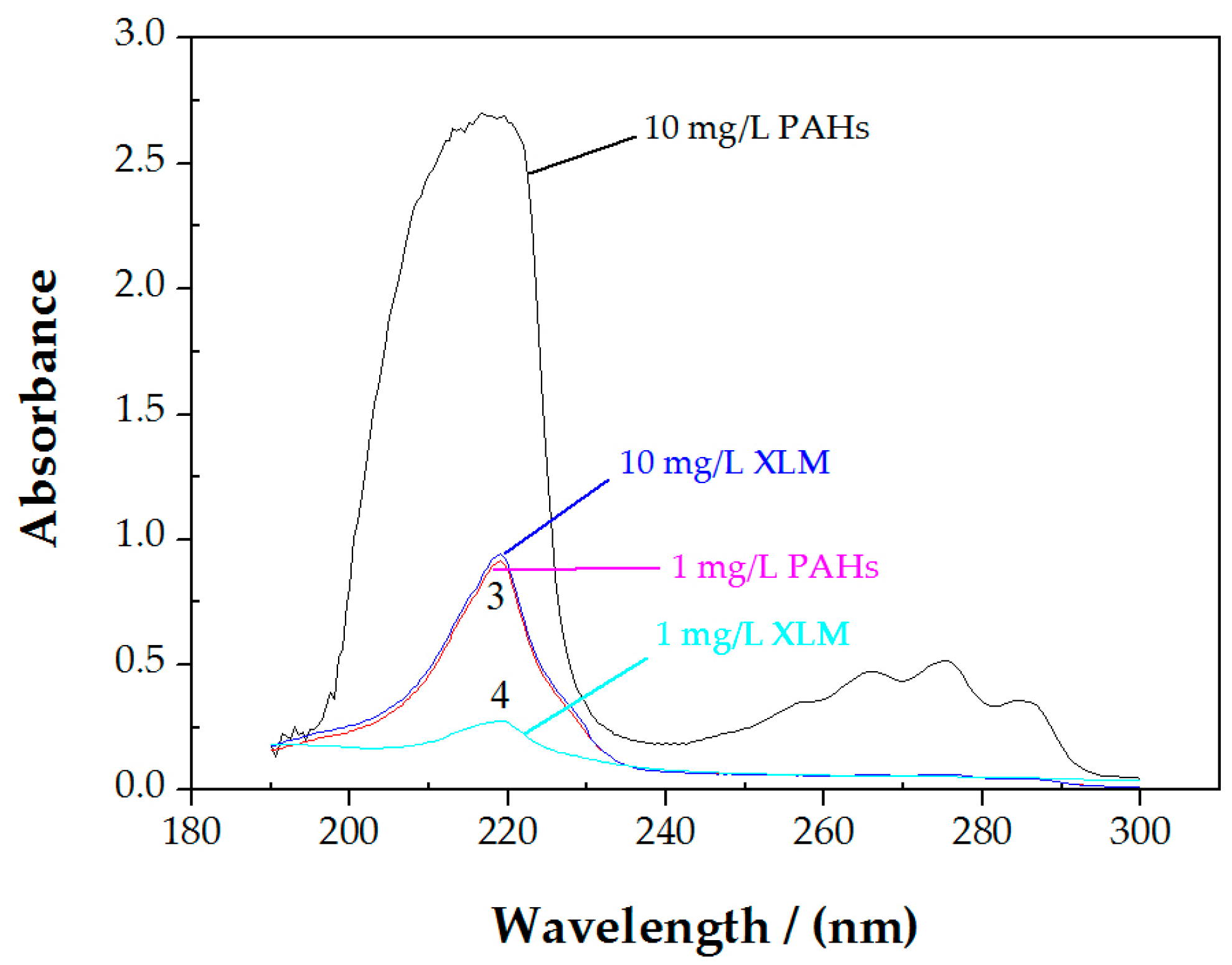
3.1. Ultraviolet-Visible Spectrophotometer Measurements
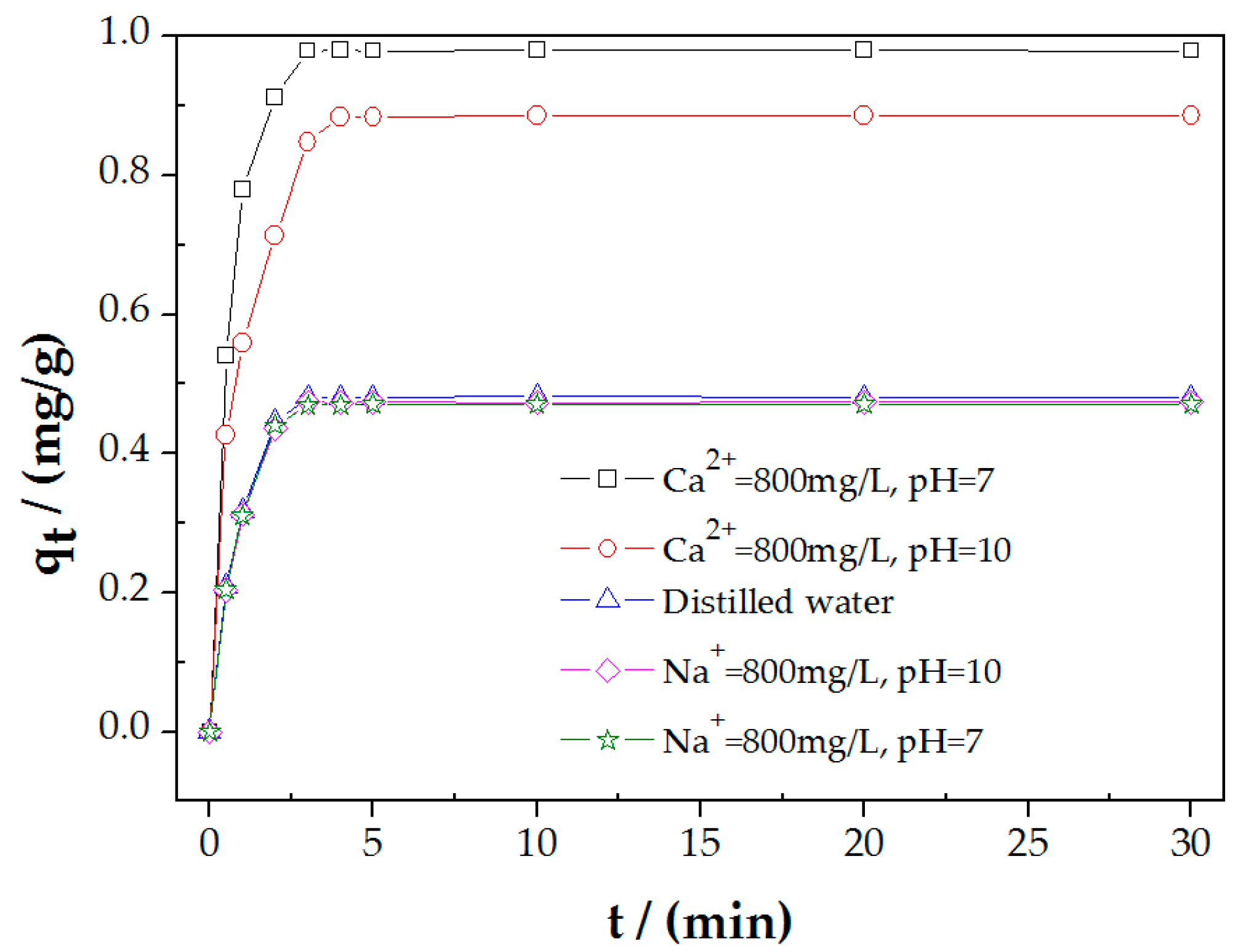
3.2. PAHs Adsorption on Molybdenite in the Presence of 800 mg/L Ca2+ or Na+
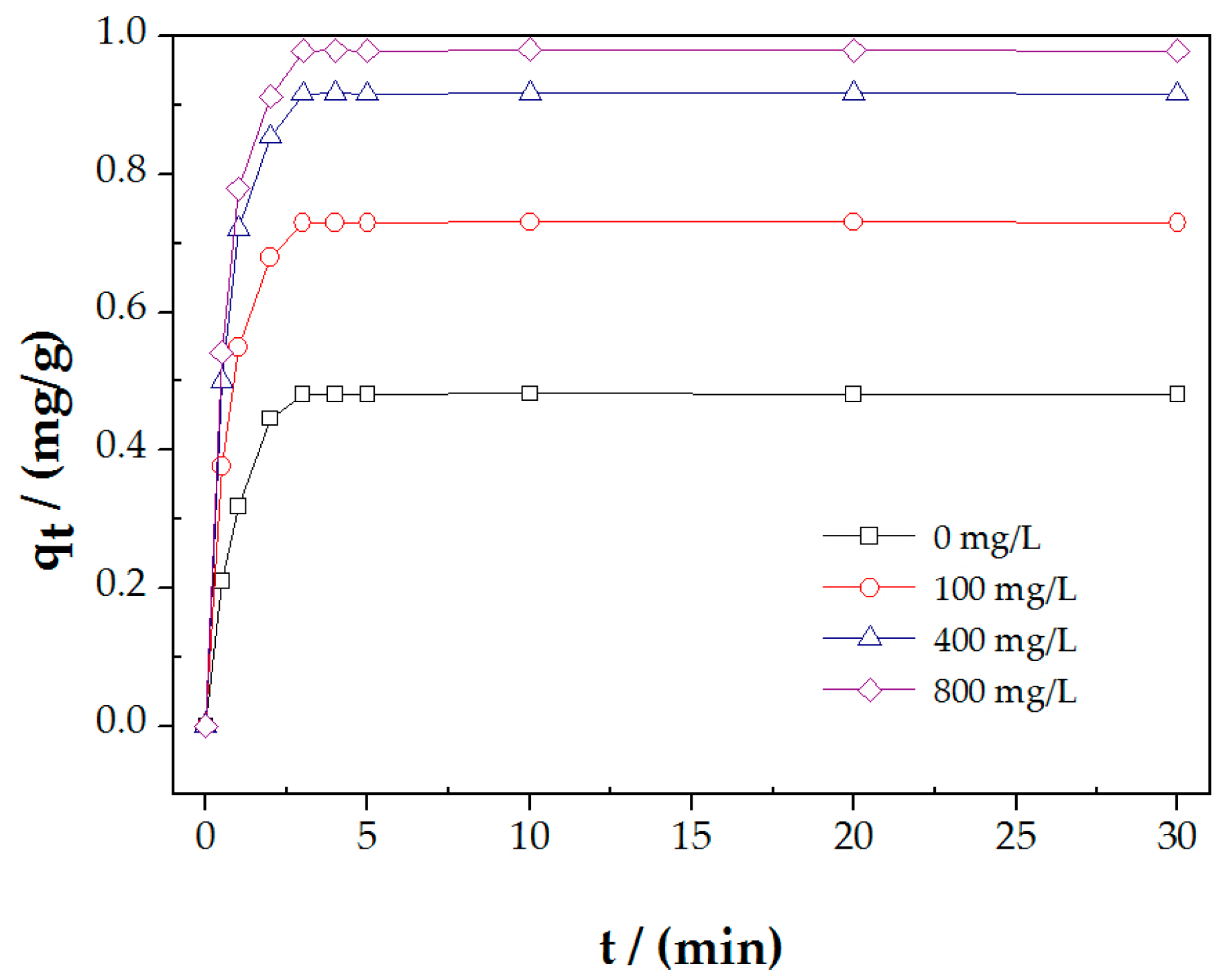
3.3. The Influence of Ca2+ Concentration on PAHs Adsoptionon Molybdenite
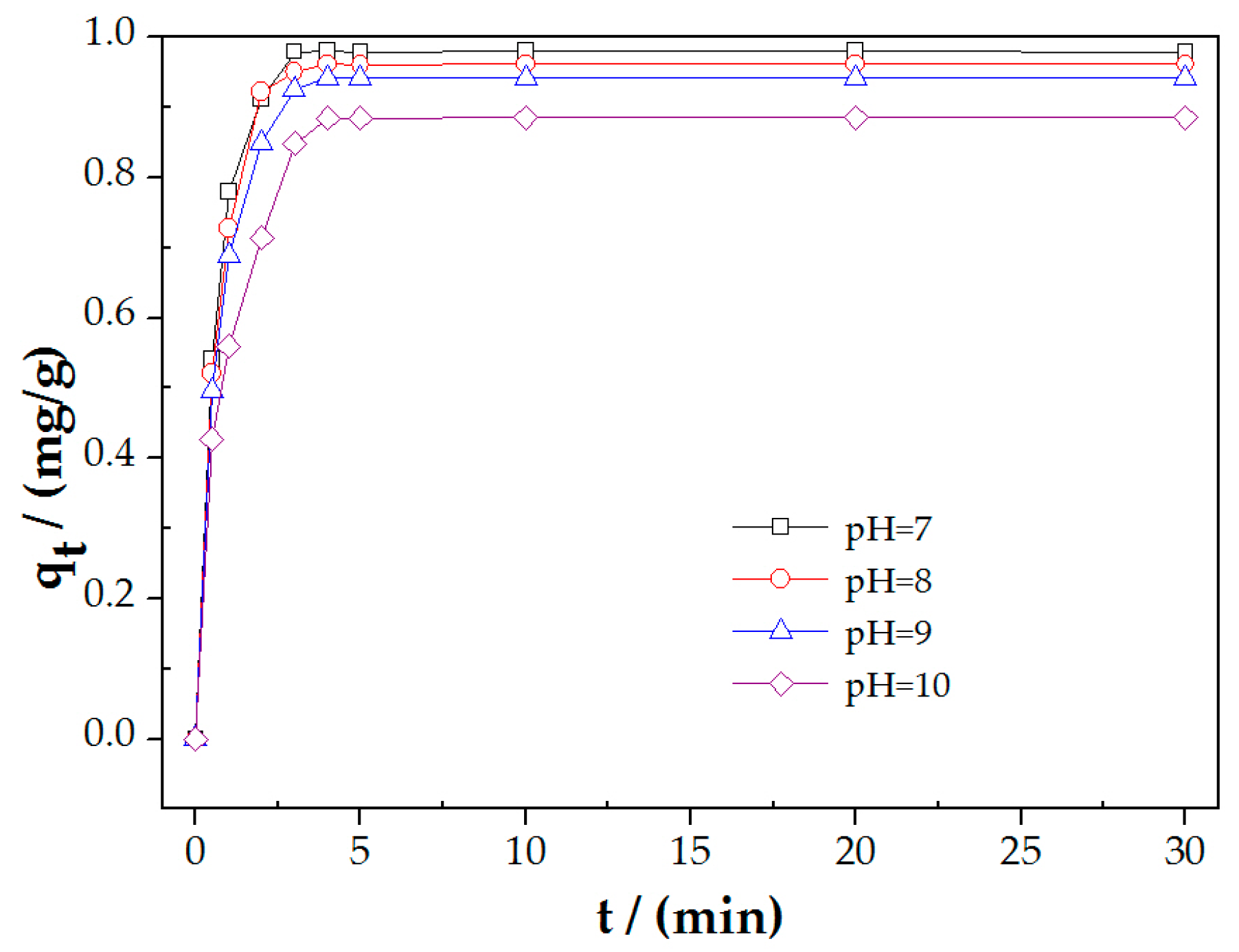
3.4. The Influence of pH on PAHs Adsoptionon Molybdenite
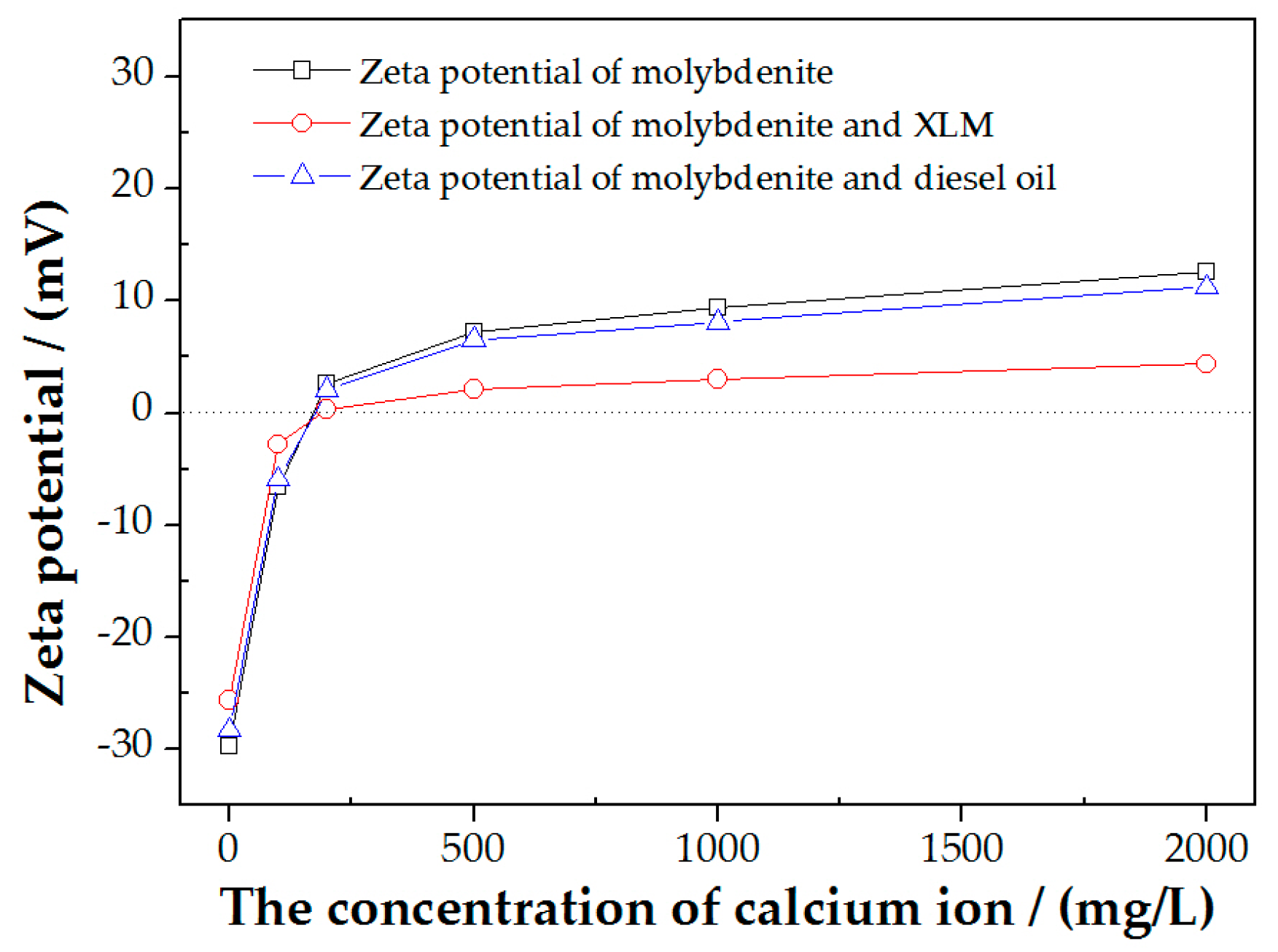
3.5. Zeta Potential Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kelebek, S. Critical surface tension of wetting and of floatability of molybdenite and sulfur. J. Colloid Interface Sci. 1988, 124, 504–514. [Google Scholar] [CrossRef]
- Castro, S.; Lopez-Valdivieso, A.; Laskowski, J.S. Review of the flotation of molybdenite. Part I: Surface properties and floatability. Int. J. Miner. Process. 2016, 148, 48–58. [Google Scholar] [CrossRef]
- Krishnaswamy, P. Kinetics of the Aqueous Oxidation of Molybdenite and the Role of Crystal Anisotropy on the Electrochemical Mechanisms of the Process. Ph.D. Dissertation, University of California, Berkeley, CA, USA, 1981; pp. 11–12. [Google Scholar]
- Chander, S.; Fuerstenau, D.W. On the natural floatability of molybdenite. Trans. Am. Inst. Min. Metall. Eng. 1972, 252, 62–69. [Google Scholar]
- Yang, B.Q.; Song, S.X.; Lopez-Valdivieso, A. Effect of Particle Size on the Contact Angle of Molybdenite Powders. Miner. Process. Extr. Metall. Rev. 2014, 35, 208–215. [Google Scholar] [CrossRef]
- He, T.S.; Wan, H.; Song, N.P.; Guo, L. The influence of composition of nonpolar oil on flotation of molybdenite. Miner. Eng. 2011, 24, 1513–1516. [Google Scholar] [CrossRef]
- Smit, F.J.; Bhasin, A.K. Relationship of petroleum hydrocarbon characteristics and molybdenite flotation. Int. J. Miner. Process. 1985, 15, 19–40. [Google Scholar] [CrossRef]
- Crozier, R.D. Flotation reagent practice in primary and by-product molybdenite recovery. Min. Mag. 1979, 140, 174–178. [Google Scholar]
- Xia, Y.K.; Peng, F.F. Selection of frothers from residual organic reagents for copper-molybdenite sulfide flotation. Int. J. Miner. Process. 2007, 83, 68–75. [Google Scholar] [CrossRef]
- Kainuma, Y.; Uyeda, R. On the Structure of Adsorbed Organic Long-Chain Molecules on the Cleavage Surface of Molybdenite. J. Phys. Soc. Jpn. 1950, 5, 199–200. [Google Scholar] [CrossRef]
- Hoover, R.; Malhotra, D. Emulsion flotation of molybdenite. In Flotation—A. M. Gaudin Memorial; American Institute of Mining, Metallurgical, and Petroleum Engineers: New York, NY, USA, 1976; Volume 1, pp. 485–503. [Google Scholar]
- Groszek, A.J. Preferential adsorption of long-chain normal paraffins on MoS2, WS2 and graphite from n-heptane. Nature 1964, 104, 680. [Google Scholar] [CrossRef]
- Groszek, A.J. Preferential adsorption of compounds with long methylene chains on cast iron, graphite, boron nitride, and molybdenum disulfide. Tribol. Trans. 1966, 9, 67–76. [Google Scholar] [CrossRef]
- Groszek, A.J. Selective adsorption at graphite/hydrocarbon interfaces. Proc. Royal Soc. Lon. A Math. Phys. Eng. Sci. 1970, 314, 473–498. [Google Scholar] [CrossRef]
- Tabares, J.O.; Ortega, I.M.; Bahena, J.R.; López, A.S.; Pérez, D.V.; Valdivieso, A.L. Surface Properties and Floatability of Molybdenite. Proceedings of 2006 China-Mexico Workshop on Minerals Particle Technology, San Luis Potosi, Mexico, August 2006; pp. 115–124. [Google Scholar]
- Triffett, B; Bradshaw, D. The role of morphology and host rock lithology on the flotation behaviour of molybdenite at Kennecott Utah Copper. Proceedings of 9th International Congress for Applied Mineralogy: ICAM 2008, Brisbane, Australia, 8–10 September 2008; pp. 465–473. [Google Scholar]
- Zanin, M.; Ametov, I.; Grano, S.; Zhou, L.; Skinner, W. A study of mechanisms affecting molybdenite recovery in a bulk copper/molybdenum flotation circuit. Int. J. Miner. Process. 2009, 93, 256–266. [Google Scholar] [CrossRef]
- Muzenda, E. An investigation into the effect of water quality on flotation performance. World Acad. Sci. Eng. Technol. 2010, 69, 237–241. [Google Scholar]
- Levay, G.; Smart, R.S.C.; Skinner, W.M. The impact of water quality on flotation performance. J. South Afr. Inst. Min. Metall. 2001, 101, 69–75. [Google Scholar]
- Chen, J.M.; Liu, R.Q.; Sun, W.; Qiu, G.Z. Effect of mineral processing wastewater on flotation of sulfide mineral. Trans. Nonferrous Metal. Soc. China 2009, 19, 454–457. [Google Scholar] [CrossRef]
- Raghavan, S.; Hsu, L.L. Factors affecting the flotation recovery of molybdenite from porphyry copper ore. Int. J. Miner. Process. 1984, 12, 145–162. [Google Scholar] [CrossRef]
- Ronzio, R.A. The role of surfactants in the flotation of molybdenite at climax. J. Am. Oil Chem. Soc. 1970, 47, 504A. [Google Scholar] [CrossRef]
- Nishkov, I.; Beas, E.; Henriquez, C. Effect of Surfactant Oil Emulsification on the Flotation of Molybdenite for Porphyry Copper Ores. In Proceedings of the X Balkan Mineral Processing Congress, Varna, Bulgaria, 15–20 June 2003; pp. 205–211. [Google Scholar]
- Nishkov, I.; Lazarov, D.; Marinov, M.; Beas, E.; Henriquez, C. Surfactant-Hydrocarbon Oil Emulsions for Molybdenite Flotation. Proceedings of IV Meeting of the Southern Hemisphere on Mineral Techenology and III Latin-American Congreess on Froth Flotation, Concepción, Chile, 20–23 November 1994; pp. 319–329. [Google Scholar]
- Chander, S.; Fuerstenau, D.W. Electrochemical study of the molybdenite–potassium diethyldithiophosphate system. Trans. SME 1974, 265, 193–197. [Google Scholar]
- Chander, S.; Fuerstenau, D.W. The effect of potassium diethyldithiophosphate on the interfacial properties of molybdenite. Trans. IMM 1974, 83, C180–C185. [Google Scholar]
- Hirajim, T.; Suyantar, G.P.W.; Ichikaw, O.; Elmahdy, A.M.; Miki, H.; Sasaki, K. Effect of Mg2+ and Ca2+ as divalent seawater cations on the flotability of molybdenite and chalcopyrite. Miner. Eng. 2016, 96–97, 83–93. [Google Scholar] [CrossRef]
- Ogawa, K.; Harada, J.; Fujiwara, T.; Takahashi, H. UV–vis Absorption Spectra of Powdered Materials: Direct Measurements by Optical Waveguide Spectroscopy. Chem. Lett. 2004, 33, 1446–1447. [Google Scholar] [CrossRef]
- Hu, S.; Li, F.; Fan, Z. Preparation of Dihydroxy Naphthalene/TiO2 Complex via Surface Modification and Their Photocatalytic H2 Production Performances under Visible Light. Bull. Korean Chem. Soc. 2013, 34, 2056–2062. [Google Scholar] [CrossRef]
- Essington, M.E. Formation of calcium and magnesium molybdate complexes in dilute aqueous solutions. Soil Sci. Soc. Am. 1992, 56, 1124–1127. [Google Scholar] [CrossRef]
- Balan, A.; Santacruz, C.P.; Moutran, A.; Souza Ferreira, L.C.; Neshich, G.; Barbosa, J.A.R.G. Crystallographic structure and substrate-binding interactions of the molybdate-binding protein of the phytopathogen Xanthomonas axonopodis pv. citri. Biochim. Biophys. Acta 2008, 1784, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Balan, A.; Santacruz, C.P.; Moutran, A.; Ferreira, R.C.C.; Medrano, F.J.; Pérez, C.A.; Ramos, C.H.I.; Ferreira, L.C.S. The molybdate-binding protein (ModA) of the plant pathogen Xanthomonas axonopodis pv. citri. Protein Expr. Purif. 2006, 50, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Amunugama, R.; Rodgers, M.T. Cation–π interactions with a model for an extended π network absolute binding energies of alkali metal cation–naphthalene complexes determined by threshold collision-induced dissociation and theoretical studies. Int. J. Mass Spectrum. 2003, 227, 1–20. [Google Scholar] [CrossRef]
- Gong, Z.; Shen, H.Y.; Zhu, W.L.; Luo, X.M.; Chen, K.X.; Jiang, H.L. A computational study on electron transfer mechanism between alkaline earth metal atoms and cyclooctatetraene to form cation–π bonded complexes. Chem. Phys. Lett. 2006, 423, 339–343. [Google Scholar] [CrossRef]
- Hallowita, N.; Udonkang, E.; Ruan, C.H.; Frieler, C.E.; Rodgers, M.T. Inductive effects on cation–π interactions: Structures and bond dissociation energies of alkali metal cation–halobenzene complexes. Int. J. Mass Spectrum. 2009, 283, 35–47. [Google Scholar] [CrossRef]
- He, L.Y.; Cheng, J.G.; Wang, T.; Li, C.; Gong, Z.; Liu, H.; Zeng, B.B.; Jiang, H.L.; Zhu, W.L. Cation–π complexes formed between cyclooctatetraene and alkaline earth metals: Predicted and recorded NMR features. Chem. Phys. Lett. 2008, 462, 45–48. [Google Scholar] [CrossRef]
- Klippenstein, S.J.; Yang, C.N. Density functional theory predictions for the binding of transition metal cations to π systems: from acetylene to coronene and tribenzocyclyne. Int. J. Mass Spectrum. 2000, 201, 253–267. [Google Scholar] [CrossRef]
- Ruan, C.H.; Yang, Z.B.; Rodgers, M.T. Cation–π interactions with a π-excessive nitrogen heterocycle: Structures and absolute binding energies of alkali metal cation–pyrrole complexes. Int. J. Mass Spectrum. 2007, 267, 233–247. [Google Scholar] [CrossRef]
- Szumna, A.; Jurczak, J.; Urbańczyk-Lipkowska, Z. Competition between π–π stacking and hydrogen bonding in (1:2) picrates of 1,5-diamino-3-oxapentane, 1,8-diamino-3,6-dioxaoctane and 1,5-diamino-3-azapentane-solid state studies. J. Mol. Struct. 2000, 526, 165–175. [Google Scholar] [CrossRef]
- Cheng, J.G.; Luo, X.M.; Yan, X.H.; Li, Z.; Tang, Y.; Jiang, H.L.; Zhu, W.L. Research progress in cation–π interactions. Sci. China Ser. B Chem. 2008, 51, 709–717. [Google Scholar] [CrossRef]
| Property | Basal Plane [0001] | Edge [0100] |
|---|---|---|
| Cleavage | Perfect | None |
| Hardness | 32 Kg/mm2 | 900 Kg/mm2 |
| Surface energy | 24 erg/cm2 | 700 erg/cm2 |
| Contact angle | 80° | 35° |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wan, H.; Yang, W.; He, T.; Yang, J.; Guo, L.; Peng, Y. The Influence of Ca2+ and pH on the Interaction between PAHs and Molybdenite Edges. Minerals 2017, 7, 104. https://doi.org/10.3390/min7060104
Wan H, Yang W, He T, Yang J, Guo L, Peng Y. The Influence of Ca2+ and pH on the Interaction between PAHs and Molybdenite Edges. Minerals. 2017; 7(6):104. https://doi.org/10.3390/min7060104
Chicago/Turabian StyleWan, He, Wei Yang, Tingshu He, Jianbo Yang, Lin Guo, and Yongjun Peng. 2017. "The Influence of Ca2+ and pH on the Interaction between PAHs and Molybdenite Edges" Minerals 7, no. 6: 104. https://doi.org/10.3390/min7060104
APA StyleWan, H., Yang, W., He, T., Yang, J., Guo, L., & Peng, Y. (2017). The Influence of Ca2+ and pH on the Interaction between PAHs and Molybdenite Edges. Minerals, 7(6), 104. https://doi.org/10.3390/min7060104





