Application of Mineral Sorbents for Removal of Petroleum Substances: A Review
Abstract
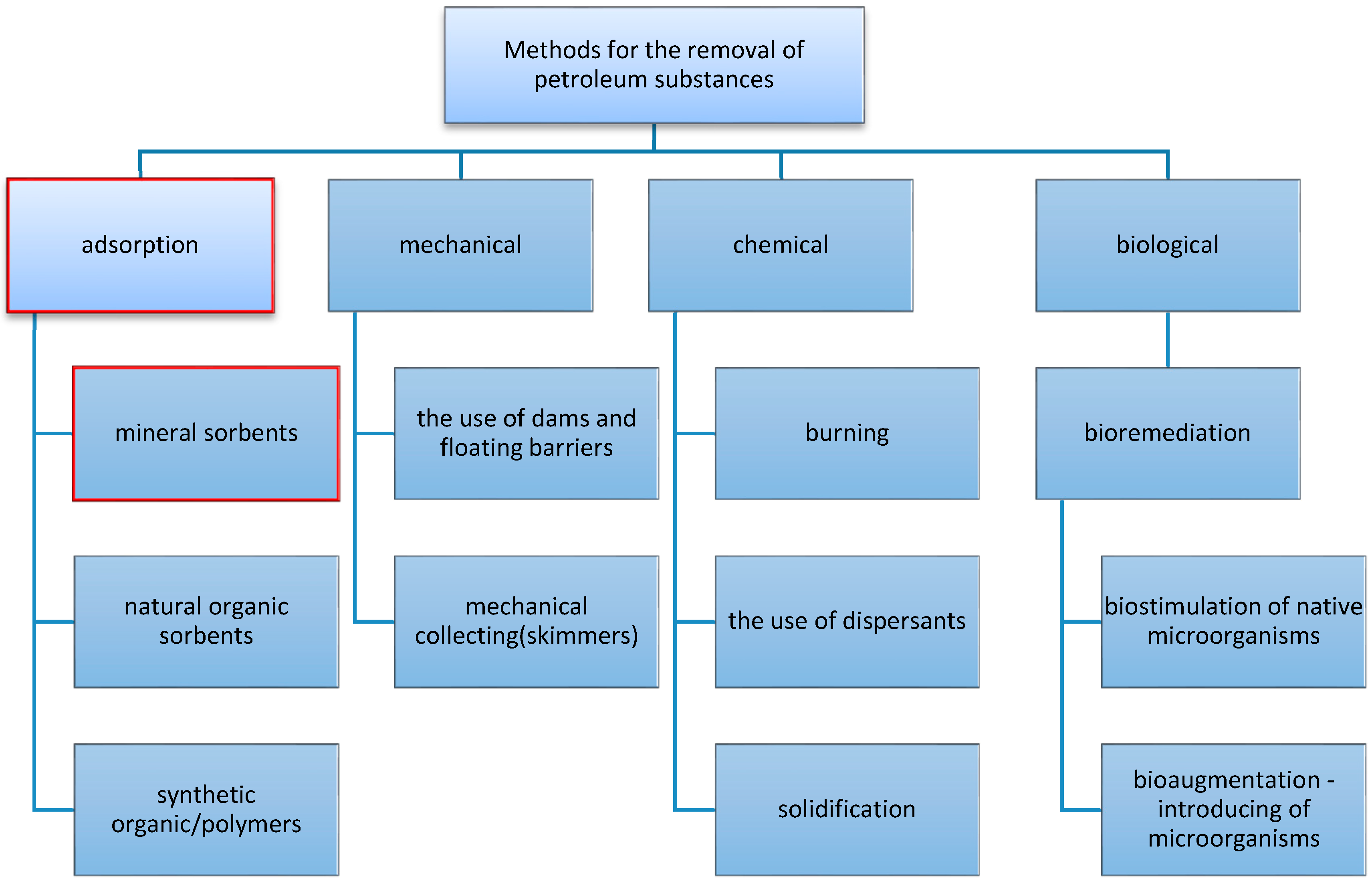
:1. Introduction
- atmospheric pollution caused by the evaporation of volatile components of petroleum products;
- pollution of soils;
- pollution of aquatic systems;
- environmental pollution caused by land-based spills of petroleum products.
2. Adsorbents in the Petroleum Spills Cleanup
- (1)
- inorganic mineral sorbents;
- (2)
- natural organic sorbents;
- (3)
- synthetic organic sorbents (synthetic polymers).
2.1. Mineral Adsorbents
2.1.1. Zeolites
2.1.2. Clay Minerals
2.1.3. Silica Adsorbents
2.2. Modified Mineral Adsorbents
3. Overview of the Sorption of Petroleum Substances by Mineral Adsorbents
3.1. Sorption of Land-Based Oil Spills
3.2. Sorption of Benzene and Its Derivatives Present in Aqueous Solutions by Organo-Mineral Adsorbents
3.3. Sorption of Volatile Petroleum Derivatives
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paulauskienė, T.; Jucikė, I.; Juščenko, N.; Baziukė, D. The use of natural sorbents for spilled crude oil and diesel cleanup from the water surface. Water Air Soil Pollut. 2014, 225, 1959–1971. [Google Scholar] [CrossRef]
- Zhang, J.; Fan, S.; Yang, J.; Du, X.; Li, F.; Hou, H. Petroleum contamination of soil and water, and their effects on vegetables by statistically analyzing entire data set. Sci. Total Environ. 2014, 476–477, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Moro, A.M.; Brucker, N.; Charão, M.F.; Sauer, E.; Freitas, F.; Durgante, J.; Bubols, G.; Campanharo, S.; Linden, R.; Souza, A.P.; et al. Early hematological and immunological alterations in gasoline station attendants exposed to benzene. Environ. Res. 2015, 137, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, F.; Méndez, J.; Pásaro, E.; Laffon, B. Review on the effects of exposure to spilled oils on human health. J. Appl. Toxicol. 2010, 30, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Alvarez, C.; Pérez, C.; Velando, A. Effects of acute exposure to heavy fuel oil from the Prestige spill on a seabird. Aquat. Toxicol. 2007, 84, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Camacho, M.; Boada, L.D.; Orós, J.; Calabuig, P.; Zumbado, M.; Luzardo, O.P. Comparative study of polycyclic aromatic hydrocarbons (PAHs) in plasma of Eastern Atlantic juvenile and adult nesting loggerhead sea turtles (Caretta caretta). Mar. Pollut. Bull. 2012, 64, 1974–1980. [Google Scholar] [CrossRef] [PubMed]
- Zytner, R.G. Sorption of benzene, toluene, ethylbenzene and xylenes to various media. J. Hazard. Mater. 1994, 38, 113–126. [Google Scholar] [CrossRef]
- Półka, M.; Kukfisz, B.; Wysocki, P.; Polakovic, P.; Kvarcak, M. Efficiency analysis of the sorbents used to adsorb the vapors of petroleum products during rescue and firefighting actions. Przem. Chem. 2015, 1, 109–113. [Google Scholar]
- Chagas-Spinelli, A.C.O.; Kato, M.T.; de Lima, E.S.; Gavazza, S. Bioremediation of a tropical clay soil contaminated with diesel oil. J. Environ. Manag. 2012, 113, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Broje, V.; Keller, A.A. Improved mechanical oil spill recovery using an optimized geometry for the skimmer surface. Environ. Sci. Technol. 2006, 40, 7914–7918. [Google Scholar] [CrossRef] [PubMed]
- Saikia, R.R.; Deka, S. Removal of hydrocarbon from refinery tank bottom sludge employing microbial culture. Environ. Sci. Pollut. Res. Int. 2013, 20, 9026–9033. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Li, C.; Dong, X.; Li, Y.; Wang, D. Separation of oil from oily wastewater by sorption and coalescence technique using ethanol grafted polyacrylonitrile. J. Hazard. Mater. 2009, 164, 1346–1351. [Google Scholar] [CrossRef] [PubMed]
- Nikolajsen, K.; Kiwi-Minsker, L.; Renken, A. Structured fixed-bed adsorber based on zeolite/sintered metal fibre for low concentration VOC removal. Chem. Eng. Res. Des. 2006, 84, 562–568. [Google Scholar] [CrossRef]
- Mathur, A.K.; Majumder, C.B.; Chatterjee, S. Combined removal of BTEX in air stream by using mixture of sugar cane bagasse, compost and GAC as biofilter media. J. Hazard. Mater. 2007, 148, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.I.; Ghoshal, A.K. Removal of volatile organic compounds from polluted air. J. Loss Prev. Process Ind. 2000, 13, 527–545. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Adebayo, A.R.; Hossain, M.E. A sustainable approach to controlling oil spills. J. Environ. Manag. 2012, 113, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Fritt-Rasmussen, J.; Brandvik, P.J. Measuring ignitability for in situ burning of oil spills weathered under Arctic conditions: From laboratory studies to large-scale field experiments. Mar. Pollut. Bull. 2011, 62, 1780–1785. [Google Scholar] [CrossRef] [PubMed]
- Boglaienko, D.; Tansel, B. Partitioning of fresh crude oil between floating, dispersed and sediment phases: Effect of exposure order to dispersant and granular materials. J. Environ. Manag. 2016, 175, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liu, W.; Fu, J.; Cai, Z.; O’Reilly, S.E.; Zhao, D. Dispersion, sorption and photodegradation of petroleum hydrocarbons in dispersant-seawater-sediment systems. Mar. Pollut. Bull. 2016, 109, 526–538. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Zeng, G.; Huang, D.; Yang, C.; Lai, C.; Zhang, C.; Liu, Y. Advantages and challenges of Tween 80 surfactant-enhanced technologies for the remediation of soils contaminated with hydrophobic organic compounds. Chem. Eng. J. 2017, 314, 98–113. [Google Scholar] [CrossRef]
- Sundaravadivelu, D.; Suidan, M.T.; Venosa, A.D.; Rosales, P.I. Characterization of solidifiers used for oil spill remediation. Chemosphere 2016, 144, 1490–1497. [Google Scholar] [CrossRef] [PubMed]
- Sundaravadivelu, D.; Suidan, M.T.; Venosa, A.D. Parametric study to determine the effect of temperature on oil solidifier performance and the development of a new empirical correlation for predicting effectiveness. Mar. Pollut. Bull. 2015, 95, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Rosales, P.I.; Suidan, M.T.; Venosa, A.D. A laboratory screening study on the use of solidifiers as a response tool to remove crude oil slicks on seawater. Chemosphere 2010, 80, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Fingas, M. Review of Solidifiers: An Update 2013; Prince Williams Sound Regional Citizens’ Advisory Council: Edmonton, AB, Canada, 2013. [Google Scholar]
- Chebbi, R. Profile of oil spill confined with floating boom. Chem. Eng. Sci. 2009, 64, 467–473. [Google Scholar] [CrossRef]
- Agnello, A.C.; Bagard, M.; van Hullebusch, E.D.; Esposito, G.; Huguenot, D. Comparative bioremediation of heavy metals and petroleum hydrocarbons co-contaminated soil by natural attenuation, phytoremediation, bioaugmentation and bioaugmentation-assisted phytoremediation. Sci. Total Environ. 2016, 563–564, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Xu, P.; Zeng, G.; Yang, C.; Huang, D.; Zhang, J. Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting: Applications, microbes and future research needs. Biotechnol. Adv. 2015, 33, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Dick, W.A.; Li, W.; Wang, X.; Yang, Q.; Wang, T.; Xu, L.; Zhang, M.; Chen, L. Bioaugmentation and biostimulation of hydrocarbon degradation and the microbial community in a petroleum-contaminated soil. Int. Biodeterior. Biodegrad. 2016, 107, 158–164. [Google Scholar] [CrossRef]
- Atlas, R.M.; Hazen, T.C. Oil biodegradation and bioremediation: A tale of the two worst spills in U.S. history. Environ. Sci. Technol. 2011, 45, 6709–6715. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, A.; Viraraghavan, T. Oil removal from water using biomaterials. Bioresour. Technol. 2010, 101, 6594–6600. [Google Scholar] [CrossRef] [PubMed]
- Szarlip, P.; Stelmach, W.; Jaromin-Gleń, K.; Bieganowski, A.; Brzezińska, M.; Trembaczowski, A.; Hałas, S.; Łagód, G. Comparison of the dynamics of natural biodegradation of petrol and diesel oil in soil. Desalin. Water Treat. 2014, 52, 3690–3697. [Google Scholar] [CrossRef]
- Adebajo, M.O.; Frost, R.L.; Kloprogge, J.T.; Carmody, O.; Kokot, S. Porous materials for oil spill cleanup: A review of synthesis and absorbing properties. J. Porous Mater. 2003, 10, 159–170. [Google Scholar] [CrossRef]
- Tic, W. Characteristics of adsorbents used to remove petroleum contaminants from soil and wastewater. Przem. Chem. 2015, 1, 79–84. [Google Scholar]
- Wahi, R.; Chuah, L.A.; Choong, T.S.Y.; Ngaini, Z.; Nourouzi, M.M. Oil removal from aqueous state by natural fibrous sorbent: An overview. Sep. Purif. Technol. 2013, 113, 51–63. [Google Scholar] [CrossRef]
- Uzunov, I.; Uzunova, S.; Angelova, D.; Gigova, A. Effects of the pyrolysis process on the oil sorption capacity of rice husk. J. Anal. Appl. Pyrolysis 2012, 98, 166–176. [Google Scholar] [CrossRef]
- Kenes, K.; Yerdos, O.; Zulkhair, M.; Yerlan, D. Study on the effectiveness of thermally treated rice husks for petroleum adsorption. J. Non-Cryst. Solids 2012, 358, 2964–2969. [Google Scholar] [CrossRef]
- Abdullah, M.A.; Rahmah, A.U.; Man, Z. Physicochemical and sorption characteristics of Malaysian Ceiba pentandra (L.) Gaertn. as a natural oil sorbent. J. Hazard. Mater. 2010, 177, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Luo, M.; Zhao, C.J.; Li, C.Y.; Wang, W.; Zu, Y.G.; Fu, Y.J. Oil removal from water with yellow horn shell residues treated by ionic liquid. Bioresour. Technol. 2013, 128, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Shang, Y.; Ding, B.; Yang, J.; Yu, J.; Al-Deyab, S.S. Nanoporous polystyrene fibers for oil spill cleanup. Mar. Pollut. Bull. 2012, 64, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, L.; Yang, F. Hydrophobic modification of polyurethane foam for oil spill cleanup. Mar. Pollut. Bull. 2012, 64, 1648–1653. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Fang, L.; Qin, Y.; Wu, W.; Mao, C.; Zhu, H. Oil sorbents with high sorption capacity, oil/water selectivity and reusability for oil spill cleanup. Mar. Pollut. Bull. 2014, 84, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Pichór, W.; Mozgawa, W.; Król, M.; Adamczyk, A. Synthesis of the zeolites on the lightweight aluminosilicate fillers. Mater. Res. Bull. 2014, 49, 210–215. [Google Scholar] [CrossRef]
- Franus, M.; Wdowin, M.; Bandura, L.; Franus, W. Removal of environmental pollutions using zeolites from fly ash: A review. Fresenius Environ. Bull. 2015, 24, 854–866. [Google Scholar]
- Wdowin, M.; Franus, M.; Panek, R.; Badura, L.; Franus, W. The conversion technology of fly ash into zeolites. Clean Technol. Environ. Policy 2014, 16, 1217–1223. [Google Scholar] [CrossRef]
- Wdowin, M. Raw kaolin as a potential material for the synthesis of a-Type Zeolite. Gospodarka Surowcami Mineralnymi Miner. Resour. Manag. 2015, 31, 45–57. [Google Scholar]
- Belviso, C. EMT-type zeolite synthesized from obsidian. Microporous Mesoporous Mater. 2016, 226, 325–330. [Google Scholar] [CrossRef]
- Burham, N.; Sayed, M. Adsorption behavior of Cd2+ and Zn2+ onto natural Egyptian bentonitic clay. Minerals 2016, 6, 129. [Google Scholar] [CrossRef]
- Oueslati, W.; Ammar, M.; Chorfi, N. Quantitative XRD analysis of the structural changes of Ba-exchanged montmorillonite: Effect of an in situ hydrous perturbation. Minerals 2015, 5, 507–526. [Google Scholar] [CrossRef]
- Aivalioti, M.; Vamvasakis, I.; Gidarakos, E. BTEX and MTBE adsorption onto raw and thermally modified diatomite. J. Hazard. Mater. 2010, 178, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Bastani, D.; Safekordi, A.A.; Alihosseini, A.; Taghikhani, V. Study of oil sorption by expanded perlite at 298.15 K. Sep. Purif. Technol. 2006, 52, 295–300. [Google Scholar] [CrossRef]
- Teas, C.; Kalligeros, S.; Zanikos, F.; Stournas, S.; Lois, E.; Anastopoulos, G. Investigation of the effectiveness of absorbent materials in oil spills clean up. Desalination 2001, 140, 259–264. [Google Scholar] [CrossRef]
- Carmody, O.; Frost, R.; Xi, Y.; Kokot, S. Adsorption of hydrocarbons on organo-clays—implications for oil spill remediation. J. Colloid Interface Sci. 2007, 305, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.A.; Bowman, R.S. Nonequilibrium sorption and transport of volatile petroleum hydrocarbons in surfactant-modified zeolite. J. Contam. Hydrol. 2009, 108, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; McLaughlin, E.; Pfeffer, R.; Lin, Y.S. Adsorption of oils from pure liquid and oil–water emulsion on hydrophobic silica aerogels. Sep. Purif. Technol. 2012, 99, 28–35. [Google Scholar] [CrossRef]
- Emam, E.A. Modified activated carbon and bentonite used to adsorb petroleum hydrocarbons emulsified in aqueous solution. Am. J. Environ. Prot. 2013, 2, 161–169. [Google Scholar] [CrossRef]
- Sakthivel, T.; Reid, D.L.; Goldstein, I.; Hench, L.; Seal, S. Hydrophobic high surface area zeolites derived from fly ash for oil spill remediation. Environ. Sci. Technol. 2013, 47, 5843–5850. [Google Scholar] [CrossRef] [PubMed]
- Vidal, C.B.; Raulino, G.S.C.; Barros, A.L.; Lima, A.C.A.; Ribeiro, J.P.; Pires, M.J.R.; Nascimento, R.F. BTEX removal from aqueous solutions by HDTMA-modified Y zeolite. J. Environ. Manag. 2012, 112, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Szala, B.; Bajda, T.; Matusik, J.; Zięba, K.; Kijak, B. BTX sorption on Na-P1 organo-zeolite as a process controlled by the amount of adsorbed HDTMA. Microporous Mesoporous Mater. 2015, 202, 115–123. [Google Scholar] [CrossRef]
- Muir, B.; Matusik, J.; Bajda, T. New insights into alkylammonium-functionalized clinoptilolite and Na-P1 zeolite: Structural and textural features. Appl. Surf. Sci. 2016, 361, 242–250. [Google Scholar] [CrossRef]
- Muir, B.; Bajda, T. Organically modified zeolites in petroleum compounds spill cleanup—Production, efficiency, utilization. Fuel Process. Technol. 2016, 149, 153–162. [Google Scholar] [CrossRef]
- Moazed, H.; Viraraghavan, T. Removal of oil from water by bentonite organoclay. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2005, 9, 130–134. [Google Scholar] [CrossRef]
- Alther, G.R. Organically modified clay removes oil from water. Waste Manag. 1995, 15, 623–628. [Google Scholar] [CrossRef]
- Mowla, D.; Karimi, G.; Salehi, K. Modeling of the adsorption breakthrough behaviors of oil from salty waters in a fixed bed of commercial organoclay/sand mixture. Chem. Eng. J. 2013, 218, 116–125. [Google Scholar] [CrossRef]
- Sarkar, B.; Megharaj, M.; Shanmuganathan, D.; Naidu, R. Toxicity of organoclays to microbial processes and earthworm survival in soils. J. Hazard. Mater. 2013, 261, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.M. Sorpcja oleju na złożach mineralnych. The sorption of oil on the mineral beds. Sci. Rev. Eng. Environ. Sci. 2005, 1, 95–102. (In Polish) [Google Scholar]
- Bandura, L.; Franus, M.; Panek, R.; Woszuk, A.; Franus, W. Characterization of zeolites and their use as adsorbents of petroleum substances. Przem. Chem. 2015, 94, 323–327. [Google Scholar]
- Standard Test Method for Sorbent Performance of Adsorbents; ASTM F726-99; American Society for Testing and Materials: West Conshohocken, PA, USA, 1999.
- Zhao, M.Q.; Huang, J.Q.; Zhang, Q.; Luo, W.L.; Wei, F. Improvement of oil adsorption performance by a sponge-like natural vermiculite-carbon nanotube hybrid. Appl. Clay Sci. 2011, 53, 1–7. [Google Scholar] [CrossRef]
- Ankowski, A. Wykorzystanie zeolitów z popiołów lotnych w sorpcji substancji ropopochodnych w warunkach rzeczywistych. In Proceedings of the Debata o Przyszłości Energetyki, Wysowa Zdrój, Poland, 4–7 May 2010. (In Polish)
- Zadaka-Amir, D.; Bleiman, N.; Mishael, Y.G. Sepiolite as an effective natural porous adsorbent for surface oil-spill. Microporous Mesoporous Mater. 2013, 169, 153–159. [Google Scholar] [CrossRef]
- Bandura, L.; Franus, M.; Józefaciuk, G.; Franus, W. Synthetic zeolites from fly ash as effective mineral sorbents for land-based petroleum spills cleanup. Fuel 2015, 147, 100–107. [Google Scholar] [CrossRef]
- Franus, M.; Jozefaciuk, G.; Bandura, L.; Lamorski, K.; Hajnos, M.; Franus, W. Modification of lightweight aggregates’ microstructure by used motor oil addition. Materials 2016, 9, 845. [Google Scholar] [CrossRef]
- Franus, W.; Jozefaciuk, G.; Bandura, L.; Franus, M. Use of spent zeolite sorbents for the preparation of lightweight aggregates differing in microstructure. Minerals 2017, 7, 25. [Google Scholar] [CrossRef]
- Boglaienko, D.; Tansel, B. Instantaneous stabilization of floating oils by surface application of natural granular materials (beach sand and limestone). Mar. Pollut. Bull. 2015, 91, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Boglaienko, D.; Tansel, B. Encapsulation of light hydrophobic liquids with fine quartz sand: Property based characterization and stability in aqueous media with different salinities. Chem. Eng. Sci. 2016, 145, 90–96. [Google Scholar] [CrossRef]
- Boglaienko, D.; Tansel, B.; Sukop, M.C. Granular encapsulation of light hydrophobic liquids (LHL) in LHL-salt water systems: Particle induced densification with quartz sand. Chemosphere 2016, 144, 1358–1364. [Google Scholar] [CrossRef] [PubMed]
- Boglaienko, D.; Tansel, B. Gravity induced densification of floating crude oil by granular materials: Effect of particle size and surface morphology. Sci. Total Environ. 2016, 556, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Carmody, O.; Frost, R.; Xi, Y.; Kokot, S. Surface characterisation of selected sorbent materials for common hydrocarbon fuels. Surf. Sci. 2007, 601, 2066–2076. [Google Scholar] [CrossRef]
- Carmody, O.; Frost, R.; Xi, Y.; Kokot, S. Selected adsorbent materials for oil-spill cleanup: A thermoanalytical study. J. Therm. Anal. Calorim. 2008, 91, 809–816. [Google Scholar] [CrossRef]
- Carvalho, M.N.; da Motta, M.; Benachour, M.; Sales, D.C.S.; Abreu, C.A.M. Evaluation of BTEX and phenol removal from aqueous solution by multi-solute adsorption onto smectite organoclay. J. Hazard. Mater. 2012, 239–240, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Nourmoradi, H.; Nikaeen, M.; Khiadani, M. Removal of benzene, toluene, ethylbenzene and xylene (BTEX) from aqueous solutions by montmorillonite modified with nonionic surfactant: Equilibrium, kinetic and thermodynamic study. Chem. Eng. J. 2012, 191, 341–348. [Google Scholar] [CrossRef]
- Aivalioti, M.; Pothoulaki, D.; Papoulias, P.; Gidarakos, E. Removal of BTEX, MTBE and TAME from aqueous solutions by adsorption onto raw and thermally treated lignite. J. Hazard. Mater. 2012, 207–208, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Standeker, S.; Novak, Z.; Knez, Z. Removal of BTEX vapours from waste gas streams using silica aerogels of different hydrophobicity. J. Hazard. Mater. 2009, 165, 1114–1118. [Google Scholar] [CrossRef] [PubMed]
- Sharmasarkar, S.; Jaynes, W.F.; Vance, G.F. BTEX sorption by montmorillonite organo-clays: TMPA, ADAM, HDTMA. Water Air Soil Pollut. 2000, 119, 257–273. [Google Scholar] [CrossRef]
- Simantiraki, F.; Aivalioti, M.; Gidarakos, E. Laboratory study on the remediation of BTEX contaminated groundwater using compost and Greek natural zeolite. In Proceedings of the CRETE 2012 3rd International Conference on Industrial and Hazardous Waste Management, Chania, Greece, 12–14 September 2012; pp. 1–8.
- Torabian, A.; Kazemian, H.; Seifi, L.; Bidhendi, G.N.; Azimi, A.A.; Ghadiri, S.K. Removal of petroleum aromatic hydrocarbons by surfactant-modified natural zeolite: The effect of surfactant. Clean Soil Air Water 2010, 38, 77–83. [Google Scholar] [CrossRef]
- Seifi, L.; Torabian, A.; Kazemian, H.; Bidhendi, G.N.; Azimi, A.A.; Farhadi, F.; Nazmara, S. Kinetic study of BTEX removal using granulated surfactant-modified natural zeolites nanoparticles. Water Air Soil Pollut. 2011, 219, 443–457. [Google Scholar] [CrossRef]
- Ho, Y.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Long, C.; Li, Y.; Yu, W.; Li, A. Removal of benzene and methyl ethyl ketone vapor: Comparison of hypercrosslinked polymeric adsorbent with activated carbon. J. Hazard. Mater. 2012, 203–204, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, S.; Valdés, H.; Manéro, M.H.; Zaror, C.A. Oxidative regeneration of toluene-saturated natural zeolite by gaseous ozone: the influence of zeolite chemical surface characteristics. J. Hazard. Mater. 2014, 274, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Valdés, H.; Solar, V.A.; Cabrera, E.H.; Veloso, A.F.; Zaror, C.A. Control of released volatile organic compounds from industrial facilities using natural and acid-treated mordenites: The role of acidic surface sites on the adsorption mechanism. Chem. Eng. J. 2014, 244, 117–127. [Google Scholar] [CrossRef]
- Cosseron, A.F.; Daou, T.J.; Tzanis, L.; Nouali, H.; Deroche, I.; Coasne, B.; Tchamber, V. Adsorption of volatile organic compounds in pure silica CHA, BEA, MFI and STT-type zeolites. Microporous Mesoporous Mater. 2013, 173, 147–154. [Google Scholar] [CrossRef]
- Kim, K.J.; Ahn, H.G. The effect of pore structure of zeolite on the adsorption of VOCs and their desorption properties by microwave heating. Microporous Mesoporous Mater. 2012, 152, 78–83. [Google Scholar] [CrossRef]
- Serrano, D.P.; Calleja, G.; Botas, J.A.; Gutierrez, F.J. Characterization of adsorptive and hydrophobic properties of silicalite-1, ZSM-5, TS-1 and Beta zeolites by TPD techniques. Sep. Purif. Technol. 2007, 54, 1–9. [Google Scholar] [CrossRef]
- Bandura, L.; Panek, R.; Rotko, M.; Franus, W. Synthetic zeolites from fly ash for an effective trapping of BTX in gas stream. Microporous Mesoporous Mater. 2016, 223, 1–9. [Google Scholar] [CrossRef]
| Sorbent | SC (g/g) | SBET (m2/g) | Source |
|---|---|---|---|
| Zeolites | |||
| clinoptilolite (Sokirnica Mine) | 0.47–0.65 | 15.88 | [60] |
| 0.23–0.38 | 18.3 | [66,71] | |
| clinoptilolite (Nižný Hrabovec) | 0.19–0.22 | 29 | [65] |
| Na-X | 0.75–0.79 | 236.4 | [71] |
| 0.91–1.13 | [66] | ||
| Na-P1 | 0.86–0.91 | 75.6 | [71] |
| 1.24–1.40 | [66] | ||
| 0.89–1.18 | 74.9 | [60] | |
| zeolites from fly ashes | 0.6–0.9 | - | [52] |
| zeolites X | 0.37, 1.33 | 40, 404 | [56] |
| Clay minerals | |||
| vermiculite | 1.3 | - | [68] |
| sepiolite | 0.97–1.2 | 258 | [70] |
| talc | 0.33 | 17 | [70] |
| Silica rocks | |||
| diatomite | 0.17–0.26 | 30 | [65] |
| chalcedonite | 1.15–1.18 | 3 | [65] |
| quartz sand | 0.2–0.3 | - | [52] |
| 0.03–0.05 | - | [65] | |
| 0.17 | - | [70] | |
| Modified minerals/organo-minerals | |||
| PTMA-montmorillonite | 0.30 | - | [70] |
| ODTMA-montmorillonite | 0.37 | - | [70] |
| 1.2–1.6 | - | [52] | |
| DDDMA-monmorillonite | 3.6–5.2 | - | [52] |
| DDDMA-bentonite | 2.1–3.5 | - | [52] |
| zeolite X modified by propyl-, octyl-, octadecyl-trimethoxysilane and stearic acid | 1.10, 1.02, 0.86, 1.15 | - | [56] |
| Na-P1 from fly ash and clinoptilolite, both modified with ODTMA, HDTMA, TDTMA, DDTMA, DODDMA, DHDDMA, DTDDMA, DDDDMA | 0.80–1.19 | 20–64 | [60] |
| 0.36–0.75 | 4.7–7.0 | ||
| Commercial mineral adsorbents | |||
| sodium aluminosilicate | 0.27–0.43 | - | [69] |
| Eco-Dry (based on diatomaceous earth) | 1.11 | - | [69] |
| Absodan (diatomite) | 0.80–0.89 | 24 | [66] |
| 0.41–0.52 | [71] | ||
| Sorbent | SBET (m2/g) | Adsorbate | T (°C) | qe (mg/g) | C (mg/L) | Kinetic Model | k Parameter | Isotherm Model | Isotherm Constants | Source | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Kf | 1/n | ||||||||||
| diatomite raw | 38.4 | benzene | 20 | 0.10 | 50 | Pseudo-second-order, k2 (g·(mg·h)−1) | 3.3263 | Freundlich | 3.68 × 10−5 | 2.47 | [49] |
| toluene | 0.15 | 50 | 1.8276 | 1.92 × 10−3 | 1.33 | ||||||
| ethylbenzene | 0.30 | 50 | 0.9134 | 4.44 × 10−2 | 0.55 | ||||||
| p-xylene | 0.34 | 50 | 0.9214 | 6.11 × 10−2 | 0.48 | ||||||
| o-xylene | 0.20 | 50 | 0.5226 | 4.6 × 10−2 | 0.46 | ||||||
| MTBE | 0.08 | 100 | 2.1956 | 5.92 × 10−10 | 4.38 | ||||||
| diatomite calcinated at 550 °C | 43.3 | benzene | 20 | 0.06 | 50 | Pseudo-second-order, k2 (g·(mg·h)−1) | 18.0996 | Freundlich | 1.55 × 10−13 | 8.13 | [49] |
| toluene | 0.21 | 50 | 7.2447 | 6.61 × 10−12 | 7.29 | ||||||
| ethylbenzene | 0.62 | 50 | 4.7319 | 7.41 × 10−11 | 6.07 | ||||||
| p-xylene | 0.82 | 50 | 2.1946 | 2.74 × 10−10 | 5.71 | ||||||
| o-xylene | 0.64 | 50 | 2.6614 | 6.02 × 10−13 | 7.25 | ||||||
| MTBE | 0.01 | 100 | 48.9326 | 4.36 × 10−11 | 4.82 | ||||||
| diatomite calcinated at 750 °C | 31.8 | benzene | 20 | 0.21 | 50 | Pseudo-second-order, k2 (g·(mg·h)−1) | 0.3069 | Freundlich | 1.35 × 10−7 | 4.45 | [49] |
| toluene | 0.22 | 50 | 0.1868 | 9.82 × 10−9 | 5.50 | ||||||
| ethylbenzene | 0.58 | 50 | 0.0557 | 1.95 × 10−6 | 3.63 | ||||||
| p-xylene | 0.75 | 50 | 0.1581 | 3.18 × 10−6 | 3.46 | ||||||
| o-xylene | 0.50 | 50 | 0.3335 | 1.02 × 10−9 | 5.46 | ||||||
| MTBE | 0.21 | 100 | 0.4441 | 48.42 | 1.53 | ||||||
| diatomite calcinated at 950 °C | 7.7 | benzene | 20 | 0.10 | - | Pseudo-second-order, k2 (g·(mg·h)−1) | 2.0324 | - | - | [49] | |
| toluene | 0.11 | 0.7896 | |||||||||
| ethylbenzene | 0.38 | 0.2091 | |||||||||
| p-xylene | 0.42 | 0.5710 | |||||||||
| o-xylene | 0.23 | 1.2817 | |||||||||
| MTBE | 0.05 | 0.0002 | |||||||||
| HDTMA smectite | 61.7 | benzene | 23 | 0.52 | 10.82 | Langmuir-Freundlich, ka × 102 [Ln/(g·min·mgn−1)] | 5.19 | - | - | - | [80] |
| toluene | 0.69 | 29.06 | 9.30 | ||||||||
| ethylbenzene | 0.72 | 8.58 | 5.04 | ||||||||
| p-xylene | 0.75 | 8.55 | 6.05 | ||||||||
| m-xylene | 0.76 | 8.52 | 5.97 | ||||||||
| phenol | 0.51 | 10.00 | 0.34 | ||||||||
| Kf | 1/n | ||||||||||
| PEG montmorillonite | 27.8 | benzene | 25 | 3.47 | 37.5 | Pseudo-second-order, k2 (g·(mg·h)−1) | 0.264 | Freundlich | 0.024 | 0.763 | [81] |
| toluene | 4.18 | 37.5 | 0.241 | 0.043 | 0.725 | ||||||
| ethylbenzene | 5.12 | 37.5 | 0.199 | 0.027 | 0.637 | ||||||
| xylene | 6.00 | 37.5 | 0.153 | 0.016 | 0.719 | ||||||
| HDTMA clinoptilolite | - | benzene | 20 | 0.22 | 9 | Pseudo-second-order, k2 (g·(mg·h)−1) | 4.46 | - | - | [87] | |
| toluene | 0.20 | 9 | 6.49 | ||||||||
| ethylbenzene | 0.16 | 9 | 5.60 | ||||||||
| xylene | 0.13 | 9 | 4.25 | ||||||||
| CPB clinoptilolite | - | benzene | 20 | 0.35 | 9 | Pseudo-second-order, k2 (g·(mg·h)−1) | 1.94 | - | - | [87] | |
| toluene | 0.30 | 9 | 1.64 | ||||||||
| ethylbenzene | 0.27 | 9 | 1.47 | ||||||||
| xylene | 0.26 | 9 | 1.42 | ||||||||
| HDTMA clinoptilolite granulated | - | benzene | 20 | 1.07 | 9 | Pseudo-second-order, k2 (g·(mg·h)−1) | 5.12 | - | - | [87] | |
| toluene | 0.83 | 9 | 2.87 | ||||||||
| ethylbenzene | 0.67 | 9 | 6.46 | ||||||||
| xylene | 0.62 | 9 | 8.82 | ||||||||
| CPB clinoptilolite granulated | - | benzene | 20 | 1.16 | 9 | Pseudo-second-order, k2 (g·(mg·h)−1) | 1.94 | - | - | [87] | |
| toluene | 0.89 | 9 | 4.46 | ||||||||
| ethylbenzene | 0.73 | 9 | 7.27 | ||||||||
| xylene | 0.66 | 9 | 4.88 | ||||||||
| Zeolite Y | 650.3 | benzene | 20 | 0.01 | 10 | Pseudo-second-order, k2 (g·(mg·h)−1) | - | - | - | [57] | |
| toluene | 0.10 | 10 | |||||||||
| ethylbenzene | 0.05 | 10 | |||||||||
| o-xylene | 0.10 | 10 | |||||||||
| m,p-xylene | 0.02 | 10 | |||||||||
| Kt | b | ||||||||||
| HDTMA-Y 100 | - | benzene | 28 | 12.13 | 10 | Pseudo-second-order, k2 (g·(mg·h)−1) | 7.114 | Temkin | 0.463 | 2.848 | [57] |
| toluene | 13.75 | 10 | 3.825 | 0.368 | 1.362 | ||||||
| ethylbenzene | 13.86 | 10 | 1.891 | 5.125 | 0.429 | ||||||
| o-xylene | 13.98 | 10 | 1.460 | 0.869 | 0.554 | ||||||
| m,p-xylene | 13.98 | 10 | 2.092 | 0.924 | 0.694 | ||||||
| Na-P1 | 88 | benzene | 20 | 1.38–6.11 | - | - | - | - | - | [58] | |
| toluene | 1.44–8.64 | ||||||||||
| p-xylene | 1.81–11.41 | ||||||||||
| HDTMA-NaP1 | - | benzene | 20 | 1.36–10.28 | - | - | - | - | - | [58] | |
| toluene | 1.40–9.99 | ||||||||||
| p-xylene | 1.79–11.47 | ||||||||||
| Sorbent | SBET (m2/g) | Adsorbate | T (°C) | qe (mg/g) | Source | ||
|---|---|---|---|---|---|---|---|
| 623 K | 823 K | 623 K | 823 K | ||||
| natural zeolite—NZ | 205 | 170 | toluene | 20 | 0.028 | 0.009 | [90] |
| acid treated NZ | 434 | 369 | toluene | 20 | 0.131 | 0.104 | [90] |
| ion exchanged NZ | 181 | 222 | toluene | 20 | 0.035 | 0.056 | [90] |
| double ion exchanged NZ | 171 | 261 | toluene | 20 | 0.082 | 0.074 | [90] |
| natural mordenite | 20 | benzene | 20 | 3.6 | [91] | ||
| toluene | 1.9 | ||||||
| p-xylene | 1.9 | ||||||
| acid-treated mordenite | 128 | benzene | 20 | 21.9 | [91] | ||
| toluene | 25.8 | ||||||
| p-xylene | 11.4 | ||||||
| MFI | 377 | acetone | 25 | 105.71 | [92] | ||
| p-xylene | 135.88 | ||||||
| n-hexane | 110.31 | ||||||
| *BEA | 493 | acetone | 25 | 124.87 | [92] | ||
| p-xylene | 124.21 | ||||||
| n-hexane | 106.00 | ||||||
| STT | 536 | acetone | 25 | 141.12 | [92] | ||
| p-xylene | 102.98 | ||||||
| n-hexane | 97.38 | ||||||
| CHA | 803 | acetone | 25 | 5.808 | [92] | ||
| p-xylene | 4.25 | ||||||
| n-hexane | 15.51 | ||||||
| faujasite Y HY901 | 591 | toluene | 25 | 1068.82 | [93] | ||
| MEK | 757.155 | ||||||
| faujasite X MS13X | 582 | toluene | 25 | 340.92 | [93] | ||
| MEK | 778.79 | ||||||
| ZSM-5 | 390 | toluene | 40 | 65.5 | [94] | ||
| isopentane | 32.3 | ||||||
| ethanol | 81.3 | ||||||
| beta zeolite | 446 | toluene | 40 | 39.5 | [94] | ||
| isopentane | 4.0 | ||||||
| ethanol | 10.5 | ||||||
| TS-1 | 414 | toluene | 40 | 74.0 | [94] | ||
| isopentane | 29.5 | ||||||
| ethanol | 15.8 | ||||||
| silicate-1 | 356 | toluene | 40 | 28.9 | [94] | ||
| isopentane | 11.5 | ||||||
| ethanol | 18.4 | ||||||
| Na-P1 zeolite from fly ash | 94.49 | benzene | 40 | 0.11 | [95] | ||
| toluene | 0.97 | ||||||
| o-xylene | 2.78 | ||||||
| m-xylene | 2.76 | ||||||
| p-xylene | 2.18 | ||||||
| Na-X zeolite from fly ash | 157.43 | benzene | 40 | 29.97 | [95] | ||
| toluene | 48.38 | ||||||
| o-xylene | 57.88 | ||||||
| m-xylene | 59.86 | ||||||
| p-xylene | 61.88 | ||||||
| clinoptilolite | 18.33 | benzene | 40 | 0.05 | [95] | ||
| toluene | 0.27 | ||||||
| o-xylene | 0.77 | ||||||
| m-xylene | 0.41 | ||||||
| p-xylene | 0.30 | ||||||
| diatomite | 23.51 | benzene | 40 | 0.02 | [95] | ||
| toluene | 0.09 | ||||||
| o-xylene | 0.10 | ||||||
| m-xylene | 0.10 | ||||||
| p-xylene | 0.10 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bandura, L.; Woszuk, A.; Kołodyńska, D.; Franus, W. Application of Mineral Sorbents for Removal of Petroleum Substances: A Review. Minerals 2017, 7, 37. https://doi.org/10.3390/min7030037
Bandura L, Woszuk A, Kołodyńska D, Franus W. Application of Mineral Sorbents for Removal of Petroleum Substances: A Review. Minerals. 2017; 7(3):37. https://doi.org/10.3390/min7030037
Chicago/Turabian StyleBandura, Lidia, Agnieszka Woszuk, Dorota Kołodyńska, and Wojciech Franus. 2017. "Application of Mineral Sorbents for Removal of Petroleum Substances: A Review" Minerals 7, no. 3: 37. https://doi.org/10.3390/min7030037
APA StyleBandura, L., Woszuk, A., Kołodyńska, D., & Franus, W. (2017). Application of Mineral Sorbents for Removal of Petroleum Substances: A Review. Minerals, 7(3), 37. https://doi.org/10.3390/min7030037









