Mineralogical Study of a Biologically-Based Treatment System That Removes Arsenic, Zinc and Copper from Landfill Leachate
Abstract
:1. Introduction
2. Experimental Section
2.1. Sampling Site Characterization
2.2. Core Solids Chemistry
2.3. Pretreatment of Core Samples: Organic Matter (OM) Removal
2.4. Mineralogical Analysis
2.4.1. XRD (X-Ray Diffraction)
2.4.2. QEMSCAN (Quantitative Evaluation of Minerals by Scanning Electron Microscopy)
2.5. Geochemical Modeling
2.6. Correlation with Microbial Communities
3. Results
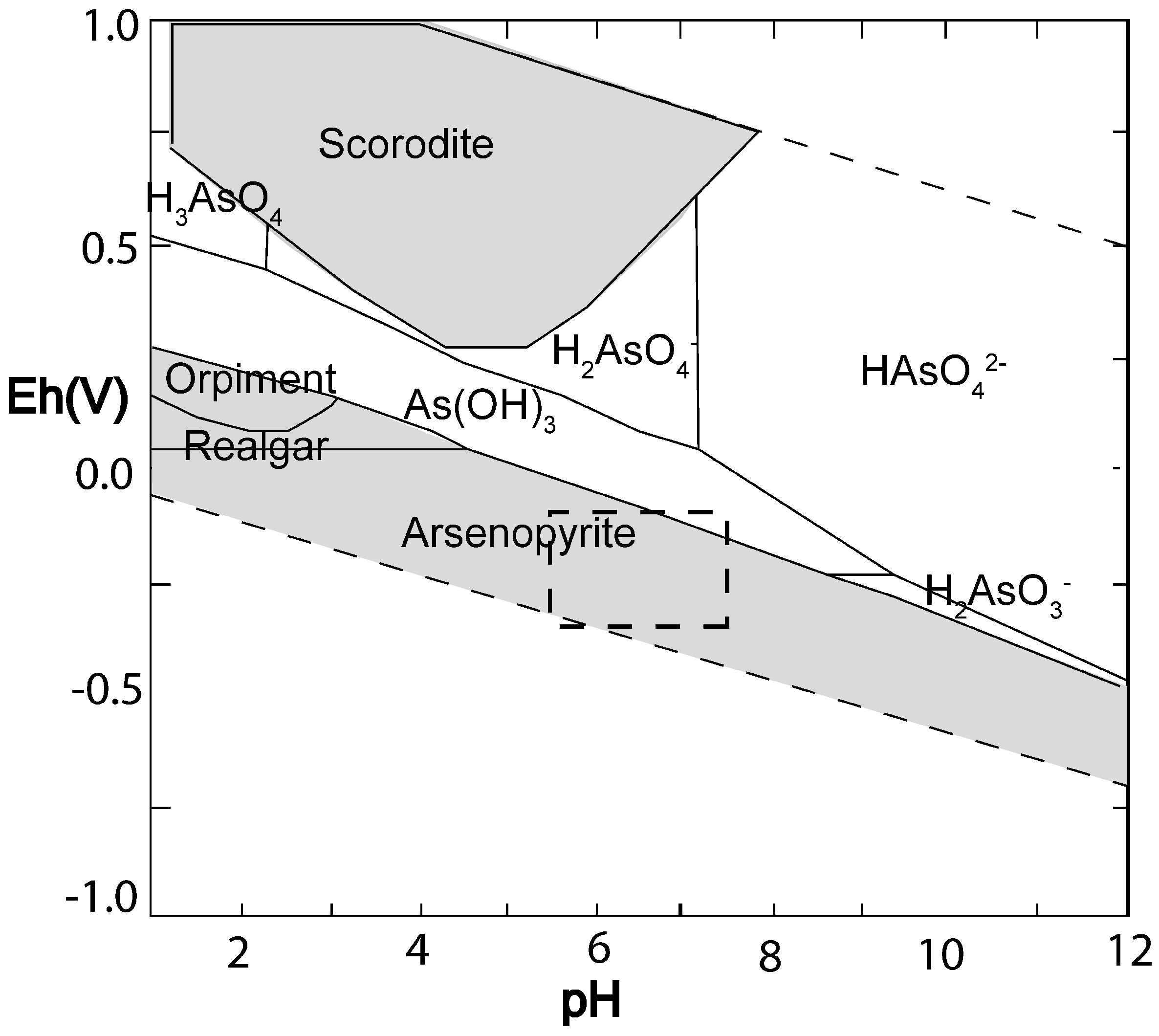
3.1. Geochemical Predictions
3.2. Mineral Species Based on XRD Analysis
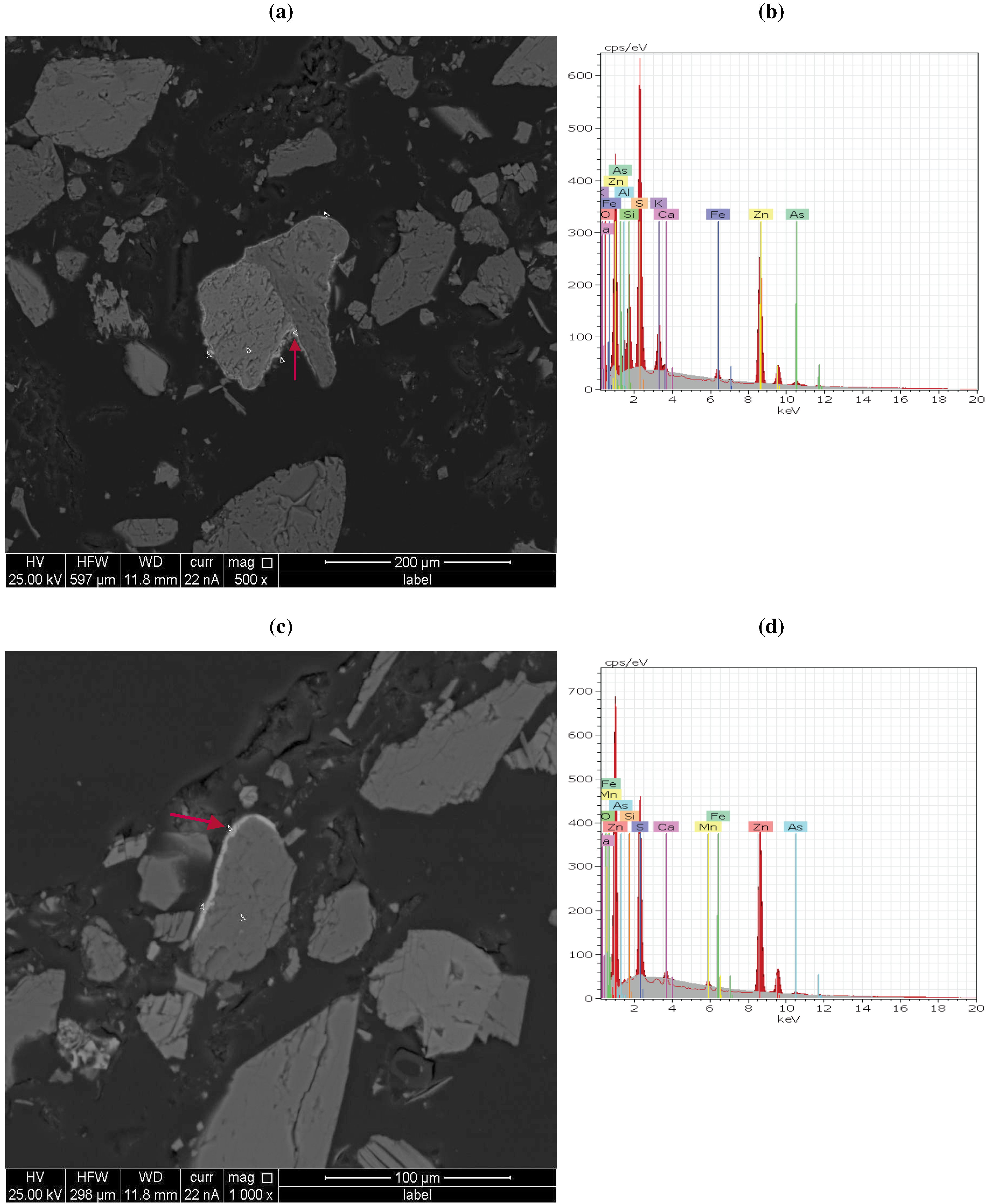
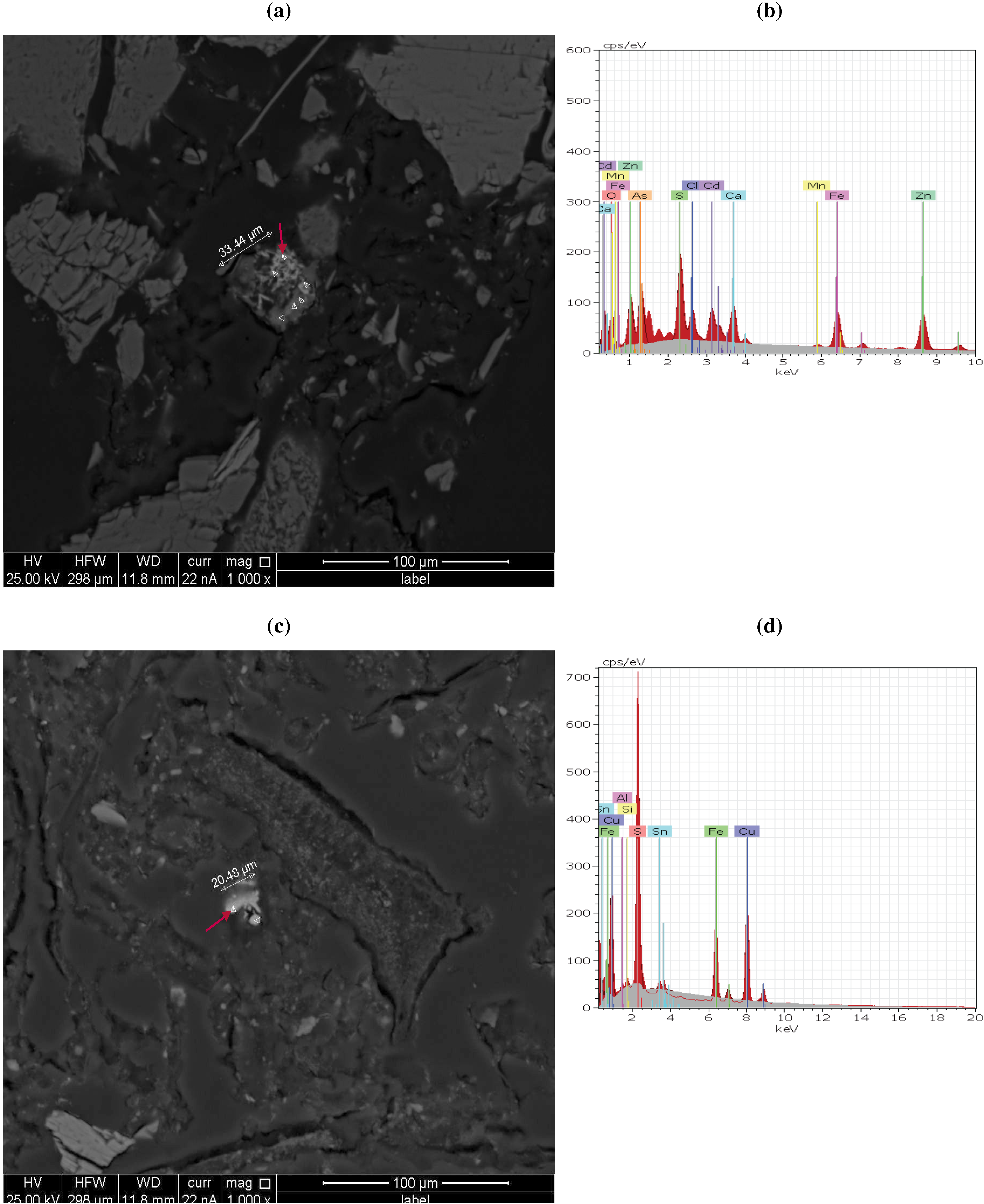
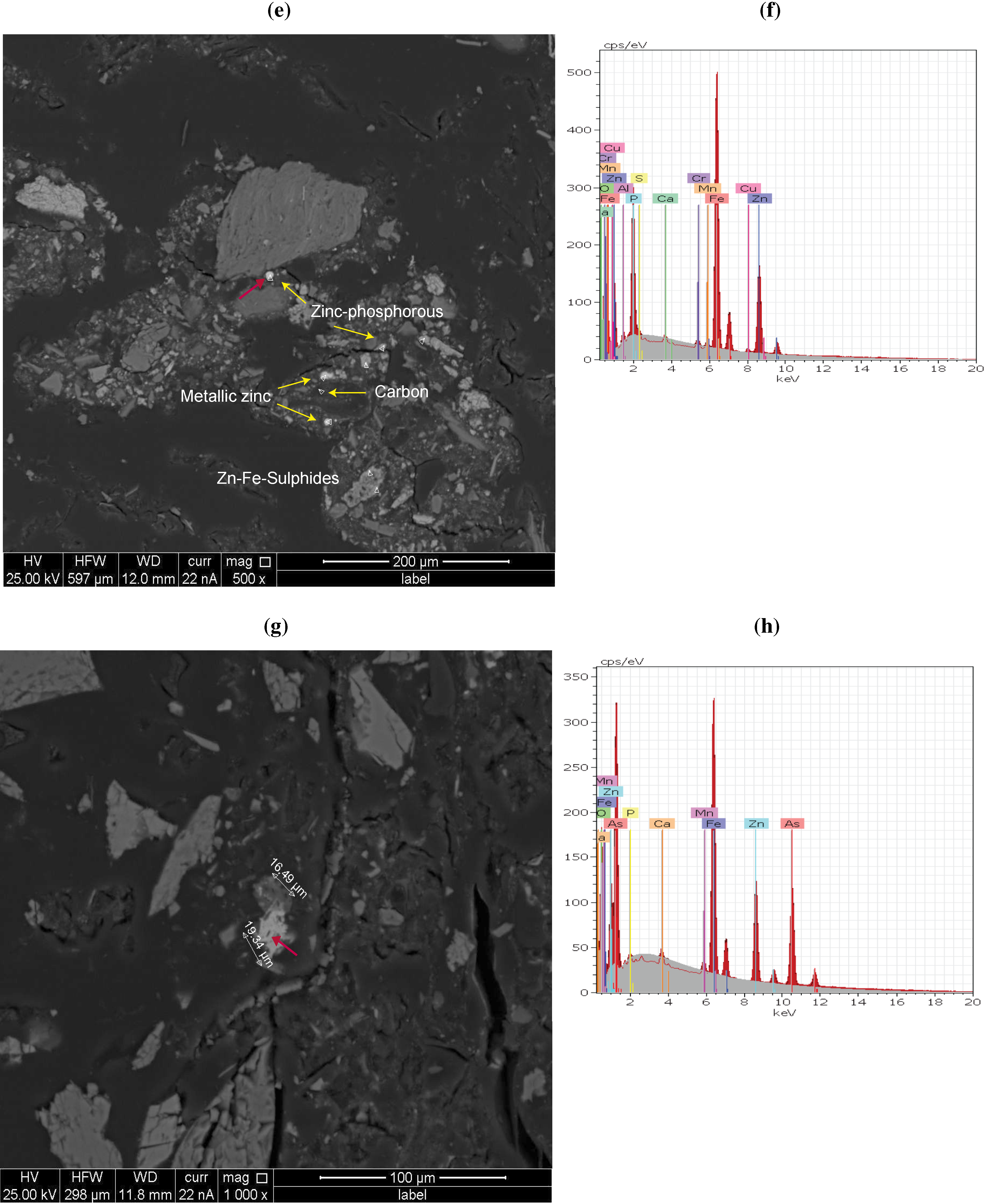
3.3. QEMSCAN and X-Ray Analysis Observations
3.4. BCR Microbial Groups Possibly Associated with Metal Removal
4. Discussion
4.1. Evidence for Biotic Mineralization
4.2. Other Possible Mechanisms for Mineral Formation in the BCR
5. Conclusions
Supplemental Information
minerals-03-00427-s001.pdfAcknowledgments
Conflicts of Interest
References
- Wieder, R.K. A survey of constructed wetlands for acid coal mine drainage treatment in the Eastern United States. Wetlands 1989, 9, 299–315. [Google Scholar]
- Gusek, J.J.; Gormley, J.T.; Scheetz, J.W. Design and construction aspects of pilot-scale passive treatment systems for acid rock drainage at metal mines. Hydrometallurgy ’94 1994, 1994, 777–793. [Google Scholar]
- Larratt, H.; Freburg, M.; Hamaguchi, R. Developing Tailings Ponds and Pit Lakes as Bioreactors and Habitat Cost-Effective Successes at Highland Valley Copper. In British Columbia’s 31st Annual Mine Reclamation Symposium; The British Columbia Technical and Research Committee on Reclamation (TRCR): Squamish, Canada, 2007. [Google Scholar]
- Reisman, D.; Ruttcowski, T.; Smart, P.; Gusek, J.J. The Construction and Instrumentation of a Pilot Treatment System at the Standard Mine Superfund Site, Crested Butte, CO. In Proceedings of the 2008 National Meeting of the American Society of Mining and Reclamation, Richmond, VA, USA, 14–19 June 2008.
- Mattes, A.; Evans, L.J.; Douglas Gould, W.; Duncan, W.F.A.; Glasauer, S. The long term operation of a biologically based treatment system that removes As, S and Zn from industrial (smelter operation) landfill seepage. Appl. Geochem 2011, 26, 1886–1896. [Google Scholar]
- Blumenstein, E.P.; Gusek, J.J. Designing a Biochemical Reactor for Selenium and Thallium Removal, from Bench Scale Testing Through Pilot Construction. In Hydrometallurgy 2008: Proceedings of the Sixth International Symposium; Young Taylor, P.R., Anderson, C.G., Choi, Y., Eds.; Society for Mining, Metallurgy, Exploration: Phoenix, AZ, USA, 2008; pp. 117–129. [Google Scholar]
- Dann, A.L.; Cooper, R.S.; Bowman, J.P. Investigation and optimization of a passively operated compost-based system for remediation of acidic, highly iron- and sulfate-rich industrial waste water. Water Res 2009, 43, 2302–2316. [Google Scholar]
- Dvorak, D.H.; Hedin, R.S.; Edenborn, H.M.; McIntire, P.E. Treatment of metal-contaminated water using bacterial sulfate reduction: Results from pilot-scale reactors. Biotechnol. Bioeng 1992, 40, 609–616. [Google Scholar]
- Hammack, R.W.; Edenborn, H.M.; Dvorak, D.H. Treatment of water from an open-pit copper mine using biogenic sulfide and limestone: A feasibility study. Water Res 1994, 28, 2321–2329. [Google Scholar]
- Winch, S.; Mills, H.J.; Kostka, J.E.; Fortin, D.; Lean, D.R.S. Identification of sulfate-reducing bacteria in methylmercury-contaminated mine tailings by analysis of SSU rRNA genes. FEMS Microbiol. Ecol 2009, 68, 94–107. [Google Scholar]
- Regine, H.S.F.; Vieira, S.F.; Volesky, B. Biosorption: A solution to pollution. Int. Microbiol 2000, 3, 17–24. [Google Scholar]
- Churakov, S.V.; D¨ahn, R. Zinc adsorption on clays inferred from atomistic simulations and EXAFS spectroscopy. Environ. Sci. Technol 2012, 46, 5713–5719. [Google Scholar]
- Fortin, D.; Southam, G.; Beveridge, T.J. Nickel sulfide, iron-nickel sulfide and iron sulfide precipitation by a newly isolated Desulfotomaculum species and its relation to nickel resistance. FEMS Microbiol. Ecol 1994, 14, 121–132. [Google Scholar]
- Bridge, T.A.; White, C.; Gadd, G.M. Extracellular metal-binding activity of the sulphate-reducing bacterium Desulfococcus multivorans. Microbiology 1999, 145, 2987–2995. [Google Scholar]
- Frankel, R.B. Biologically induced mineralization by bacteria. Rev. Mineral. Geochem 2003, 54, 95–114. [Google Scholar]
- Glasauer, S.; Langley, S.; Beveridge, T.J. Sorption of Fe (hydr)oxides to the surface of Shewanella putrefaciens: Cell-bound fine-grained minerals are not always formed de novo. Appl. Environ. Microbiol 2001, 67, 5544–5550. [Google Scholar]
- Amoozegar, M.A.; Khoshnoodi, M.; Didari, M.; Hamedi, J.; Ventosa, A.; Baldwin, S.A. Tellurite removal by a tellurium-tolerant halophilic bacterial strain, Thermoactinomyces sp. QS-2006. Ann. Microbiol 2012, 62, 1031–1037. [Google Scholar]
- Fortin, D.; Praharaj, T. Role of microbial activity in Fe and S cycling in sub-Oxic to anoxic aulfide-rich mine tailings: A mini-review. J. Nucl. Radiochem. Sci 2005, 6, 39–42. [Google Scholar]
- Jiang, S.; Lee, J.H.; Kim, D.; Kanaly, R.A.; Kim, M.G.; Hur, H.G. Differential arsenic mobilization from As-bearing ferrihydrite by iron-respiring Shewanella strains with different arsenic-reducing activities. Environ. Sci. Technol 2013, 47, 8616–8623. [Google Scholar]
- Tufano, K.J.; Reyes, C.; Saltikov, C.W.; Fendorf, S. Reductive processes controlling arsenic retention: Revealing the relative importance of iron and arsenic reduction. Environ. Sci. Technol 2008, 42, 8283–8289. [Google Scholar]
- Kocar, B.D.; Herbel, M.J.; Tufano, K.J.; Fendorf, S. Contrasting effects of dissimilatory iron(III) and arsenic(V) reduction on arsenic retention and transport. Environ. Sci. Technol 2006, 40, 6715–6721. [Google Scholar]
- Labrenz, M.; Druschel, G.K.; Thomsen-Ebert, T.; Gilbert, B.; Welch, S.A.; Kemner, K.M.; Logan, G.A.; Summons, R.E.; Stasio, G.D.; Bond, P.L.; et al. Formation of sphalerite (ZnS) deposits in natural biofilms of sulfate-reducing bacteria. Science 2000, 290, 1744–1747. [Google Scholar]
- Moreau, J.W.; Webb, R.I.; Banfield, J.F. Ultrastructure, aggregation-state, and crystal growth of biogenic nanocrystalline sphalerite and wurtzite. Am. Mineral 2004, 89, 950–960. [Google Scholar]
- Castillo, J.; Perez-Lopez, R.; Caraballo, M.A.; Nieto, J.M.; Martins, M.; Costa, M.C.; Olias, M.; Ceron, J.C.; Tucoulou, R. Biologically-induced precipitation of sphalerite-wurtzite nanoparticles by sulfate-reducing bacteria: Implications for acid mine drainage treatment. Sci. Total Environ 2012, 423, 176–184. [Google Scholar]
- Niggemyer, A.; Spring, S.; Stackebrandt, E.; Rosenzweig, R.F. Isolation and characterization of a novel As(V)-reducing bacterium: Implications for arsenic mobilization and the genus Desulfitobacterium. Appl. Environ. Microbiol 2001, 67, 5568–5580. [Google Scholar]
- Santini, J.M.; Sly, L.I.; Wen, A.; Comrie, D.; Wulf-Durand, P.D.; Macy, J.M. New arsenite-oxidizing bacteria isolated from Australian gold mining environments—Phylogenetic relationships. Geomicrobiol. J 2002, 19, 67–76. [Google Scholar]
- Jackson, C.K.; Koch, I.; Reimer, K.J. Mechanisms of dissolved arsenic removal by biochemical reactors: A bench- and field-scale study. Appl. Geochem 2013, 29, 174–181. [Google Scholar]
- Pirrie, D.; Butcher, A.R.; Power, M.R.; Gottlieb, P.; Miller, G.L. Rapid quantitative mineral and phase analysis using automated scanning electron microscopy (QemSCAN); Potential applications in forensic geoscience. Geol. Soc 2004, 232, 123–136. [Google Scholar]
- Baldwin, S.A.; Rezadehbashi, M.; Taupp, M.; Mattes, A.; Hallam, S.J. The Genetic Diversity of Microbes in a Biochemical Reactor Treating Metal-Rich Landfill Leachate: What This Tells Us about Treatment Mechanisms. In Proceedings of 9th International Conference on Acid Rock Drainage, Ottawa, Canada, 20–24 May 2012.
- Adegoroye, A.; Uhlik, P.; Omotoso, O.; Xu, Z.; Masliyah, J. A comprehensive analysis of organic matter removal from clay-sized minerals extracted from oil sands using low temperature ashing and hydrogen peroxide. Energy Fuels 2009, 23, 3716–3720. [Google Scholar]
- Buckwalter-Davis, M.; Jaggardi, H.; Morman, S.; Plumlee, G.; Jamieson, H. Using automated mineralogy to evaluate bioaccessibility of Pb-bearing mine waste. Mineral. Mag 2012, 76, 1524. [Google Scholar]
- Chemical Equilibrium Diagrams. Available online: http://www.kth.se/che/medusa/ (accessed on 10 December 2013).
- Puigdomenech, I. MEDUSA: Make Equilibrium Diagrams Using Sophisticated Algorithms Programs; Royal Institute of Technology (KTH): Stockholm, Sweden, 2002. [Google Scholar]
- Puigdomenech, I. HYDRA: Hydrochemical Equilibrium-Constant Database Software; Royal Institute of Technology (KTH): Stockholm, Sweden, 2004. [Google Scholar]
- Lu, P.; Zhu, C. Arsenic Eh-pH diagrams at 25 °C and 1 bar. Environ. Earth Sci 2010, 62, 1673–1683. [Google Scholar]
- Rezadehbashi, M.; Khoshnoodi, M.; Taupp, M.; Hallam, S.; Baldwin, S.A. Microbial Diversity of A Biochemical Reactor Removing Metals from Smelter Waste Leachate. In Proceedings of the 2012 Canadian Society of Microbiologists Annual Conference, Vancouver, Canada, 20–23 June 2012.
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res 2013, 41, D590–D596. [Google Scholar]
- Pruitt, K.D.; Tatusova, T.; Brown, G.R.; Maglott, D.R. NCBI Reference Sequences (RefSeq): Current status, new features and genome annotation policy. Nucleic Acids Res 2012, 40, D130–D135. [Google Scholar]
- Lozupone, C.; Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol 2005, 71, 8228–8235. [Google Scholar]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar]
- Khoshnoodi, M.; Taupp, M.; Mewis, K.; Hallam, S.; Baldwin, S.A. Screening for Arsenate and Arsenite Resistant Microbes in Passive Treatment Systems for Metal Leachate Remediation. In Proceedings of the 9th International Conference on Acid Rock Drainage, Ottawa, Canada, 20–24 May 2012.
- Baas Becking, L.G.M.; Moore, D. Biogenic sulfides. Econ. Geol 1961, 56, 259–272. [Google Scholar]
- Wang, S.; Mulligan, C.N. Natural attenuation processes for remediation of arsenic contaminated soils and groundwater. J. Hazard. Mater 2006, 138, 459–470. [Google Scholar]
- Bostick, B.C.; Fendorf, S. Arsenite sorption on troilite (FeS) and pyrite (FeS2). Geochim. Cosmochim. Acta 2003, 67, 909–921. [Google Scholar]
- Battaglia-Brunet, F.; Crouzet, C.; Burnol, A.; Coulon, S.; Morin, D.; Joulian, C. Precipitation of arsenic sulphide from acidic water in a fixed-film bioreactor. Water Res 2012, 46, 3923–3933. [Google Scholar]
- Rosenberg, P.E.; Foit, F.F. The stability of transition metal dolomites in carbonate systems: A discussion. Geochem. Cosmochim. Acta 1979, 43, 951–955. [Google Scholar]
- Cabala, J.; Zogala, B.; Dubiel, R. Geochemical and geophysical study of historical Zn-Pb ore processing waste dump areas (Southern Poland). Polish. J. Environ. Stud 2008, 17, 693–700. [Google Scholar]
- Vasconcelos, C.; McKenzie, J.A.; Bernasconi, S.; Grujic, D.; Tiens, A.J. Microbial mediation as a possible mechanism for natural dolomite formation at low temperatures. Nature 1995, 377, 220–222. [Google Scholar]
- Warthmann, R.; van Lith, Y.; Vasconcelos, C.; McKenzie, J.A.; Karpoff, A.M. Bacterially induced dolomite precipitation in anoxic culture experiments. Geology 2000, 28, 1091–1094. [Google Scholar]
- Chovatia, M.; Sikorski, J.; Schroder, M.; Lapidus, A.; Nolan, M.; Tice, H.; Glavina Del Rio, T.; Copeland, A.; Cheng, J.F.; Lucas, S.; et al. Complete genome sequence of Thermanaerovibrio acidaminovorans type strain (Su883T). Stand. Genomic Sci 2009, 1, 254–261. [Google Scholar]
- Labutti, K.; Mayilraj, S.; Clum, A.; Lucas, S.; Glavina Del Rio, T.; Nolan, M.; Tice, H.; Cheng, J.F.; Pitluck, S.; Liolios, K.; et al. Permanent draft genome sequence of Dethiosulfovibrio peptidovorans type strain (SEBR 4207T). Stand. Genomic Sci 2010, 3, 85–92. [Google Scholar]
- Pavissich, J.P.; Silva, M.; González, B. Sulfate reduction, molecular diversity, and copper amendment effects in bacterial communities enriched from sediments exposed to copper mining residues. Environ. Toxicol. Chem 2010, 29, 256–264. [Google Scholar]
- Singh, D.B.; Tiwary, R.K.; Tewary, B.K. Feldspar for the treatment of As(III) contaminated water. Water Qual. Res. J. Can 2002, 37, 757–771. [Google Scholar]
- Manning, B.A.; Goldberg, S. Adsorption and stability of Arsenic(III) at the clay mineral-water interface. Environ. Sci. Technol 1997, 31, 2005–2011. [Google Scholar]
- Van der Hoek, E.E.; Bonouvrie, P.A.; Comans, R.N.J. Sorption of As and Se on mineral components of fly ash: Relevance for leaching processes. Appl. Geochem 1994, 9, 403–412. [Google Scholar]
- O’Day, P.A. Chemistry and mineralogy of arsenic. Elements 2006, 2, 77–83. [Google Scholar]
- Sharma, V.K.; Sohn, M. Aquatic arsenic: Toxicity, speciation, transformations, and remediation. Environ. Int 2009, 35, 743–759. [Google Scholar]
- Newman, D.K.; Ahmann, D.; Morel, F.M.M. A brief review of microbial arsenate respiration. Geomicrobiol. J 1998, 15, 255–268. [Google Scholar]
- Langmuir, D.; Mahoney, J.; Rowson, J. Solubility products of amorphous ferric arsenate and crystalline scorodite (FeAsO42H2O) and their application to arsenic behavior in buried mine tailings. Geochim. Cosmochim. Acta 2006, 70, 2942–2956. [Google Scholar]
- Praharaj, T.; Fortin, D. Seasonal variations of microbial sulfate and iron reduction in alkaline PbZn mine tailings (Ontario, Canada). Appl. Geochem 2008, 23, 3728–3740. [Google Scholar]
- Mattes, A.; Gould, D.; Taupp, M.; Glasauer, S. A novel autotrophic bacterium isolated from an engineered wetland system links nitrate-coupled iron oxidation to the removal of As, Zn and S. Water Air Soil Pollut 2013, 224. [Google Scholar] [CrossRef]
- Mattes, A.G. The Trail Biochemical Reactor the Removal of Arsenic with a Focus on the Specfic Role of Iron. Ph.D. Thesis, University of Guelph, Guelph, Canada, August 2013.
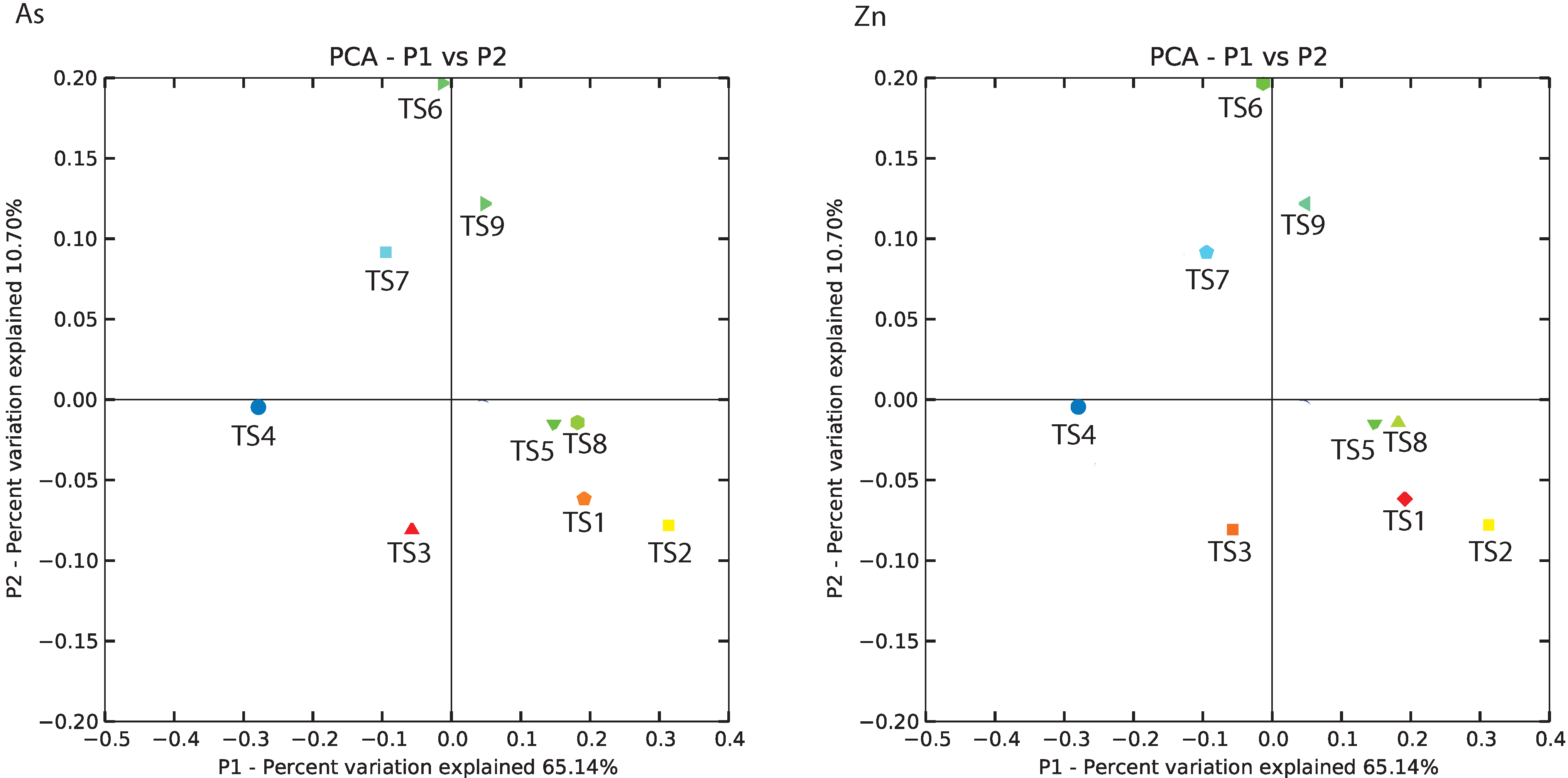
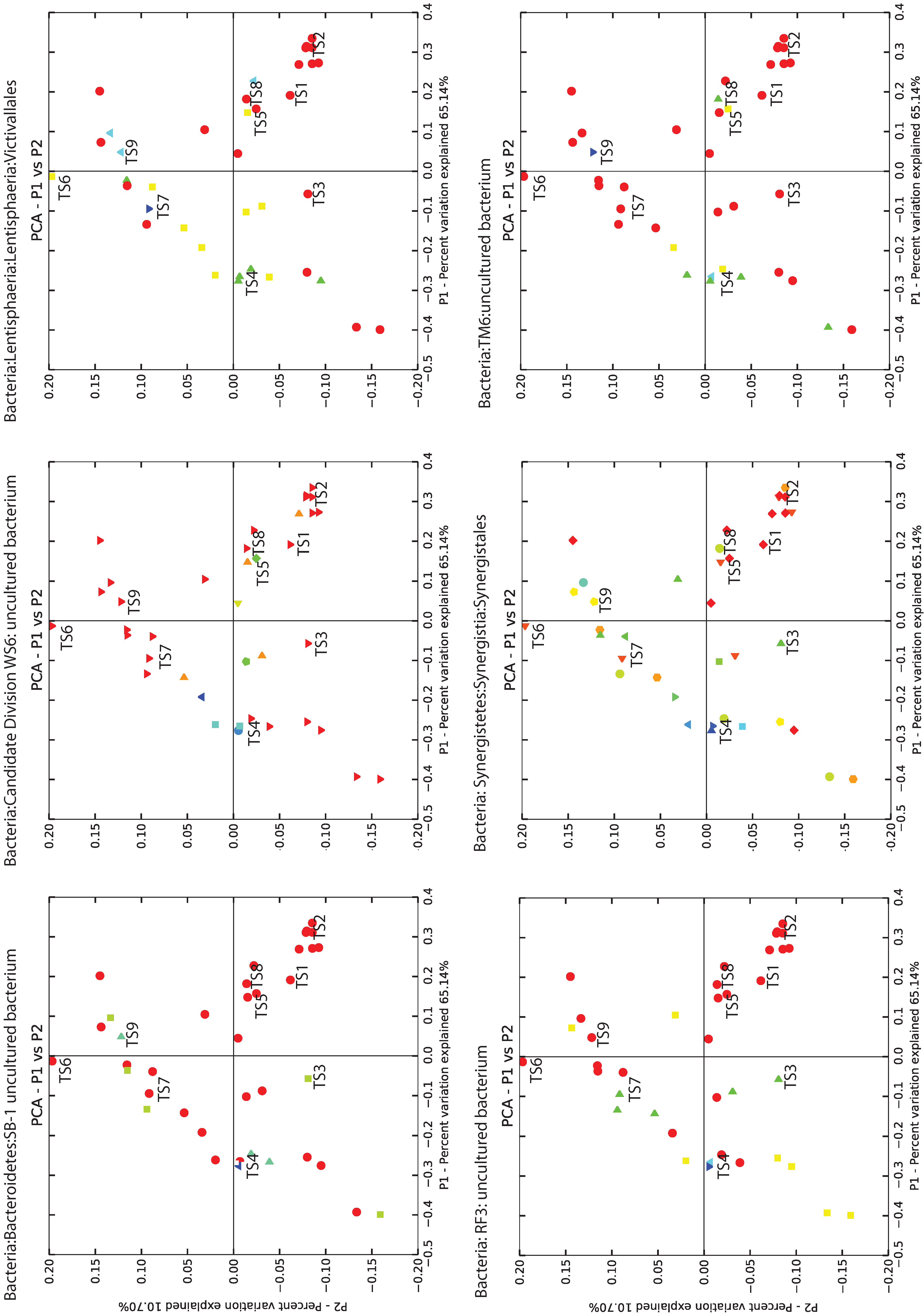
| SampleID | TS1 | TS2 | TS3 | TS4 | TS5 | TS6 | TS7 | TS8 | TS9 |
|---|---|---|---|---|---|---|---|---|---|
| Date | 22 July 2008 | 21 April 2009 | 20 October 2009 | ||||||
| Pore water chemistry | |||||||||
| pH | 5.6 | 5.6 | 6.9 | 6.9 | 6.9 | 7.0 | 6.7 | 6.2 | 7.5 |
| Eh (mV) | −133 | −112 | −238 | NA 1 | NA | NA | −118 | −244 | −130 |
| DO (mg/L) | 0 | 0 | 0 | 1.5 | 1.1 | 0.7 | 1.2 | 1.3 | 1.3 |
| T (°C) | 17.6 | 15.9 | 19.4 | 7.6 | 6.5 | 7.1 | 11.2 | 11.5 | 11.2 |
| Solids analysis | |||||||||
| As (ppm) | 2.7 | 2.9 | 1.8 | >250 | 16.6 | 18.9 | 45.8 | 9.3 | 18.9 |
| Zn (ppm) | 108 | 122 | 118 | 1735 | 180 | 177 | 705 | 127 | 380 |
| Cu (ppm) | 32 | 15 | 11 | 79 | 22 | 19 | 21 | 12 | 12 |
| Fe2O3 (%) | 1.58 | 1.36 | 1.86 | NA | 2.12 | 2.24 | 1.29 | 2.35 | 1.88 |
| S (%) | 0.24 | 0.19 | 0.11 | NA | 0.31 | 0.38 | 0.27 | 0.15 | 0.09 |
| TOC (mg/L) | 4.46 | 4.29 | 3.17 | 5.20 | 8.70 | 8.00 | 13.28 | 2.69 | 1.74 |
| Chemical form | Mineral (retrieved from XRD) | Formula (retrieved from EDX) | BMA(QEMSCAN) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TS1 | TS2 | TS3 | TS4 | TS5 | TS6 | TS7 | TS8 | TS9 | |||
| Carbonates | Calcite | CaCO3 | 16.3 | 21.9 | 16.0 | 24.3 | 15.3 | 15.0 | 28.1 | 29.9 | 10.2 |
| Minrecordite/dolomite | Ca(Zn, Mg)(CO3)2 | 0.3 | 0.3 | 0.6 | 0.6 | 0.6 | 0.5 | 0.8 | 1.3 | 0.2 | |
| Oxides, Hydroxides | Iron oxides 1 | FexOx | 0.2 | 0.2 | 0.5 | 0.2 | 1.1 | 0.5 | 0.2 | 0.2 | 0.3 |
| not detected | ZnAsOx | ||||||||||
| Sulphate | Zinc woodwardite-3R | ((Zn1−xAlx)(OH)2)(SO4)x/2) | P | P | P | P | |||||
| Guildite | CuFe+3(SO4)(OH)4H2O | P | P | P | |||||||
| not detected | Zn − SO4 | 1.8 2 | 2.2 2 | 1.3 2 | 2.2 2 | 1.4 2 | 0.9 2 | 3.1 2 | 1.3 2 | 1.3 2 | |
| Zn − PbSOx | |||||||||||
| Sulphide | Sphalerite | ZnS | 1.9 2 | 8.3 2 | 1.9 2 | 0.8 2 | 3.6 2 | 1.5 2 | 1 2 | 0.5 2 | 1.0 2 |
| Arsenopyrite | FeAsS | 0.0 3 | 34.6 3 | 4.1 3 | 0.0 3 | 25.7 3 | 0.0 3 | 0.0 3 | 7.4 3 | 0.0 3 | |
| Pyrite | FeS2 | <0.1 | 0.2 | <0.1 | <0.1 | <0.1 | <0.1 | 0.1 | 0.1 | <0.1 | |
| Iron sulphides 4 | FeS2 or (Fe, Ni)S0.9 | ||||||||||
| not detected | FeAsS-Zn or FeAsS-Zn-Cd | ||||||||||
| Tennantite | (Cu, Fe)12As4S13 | 2.2 3 | 7.4 3 | ||||||||
| Chalcopyrite | CuFeS2 | ||||||||||
| not detected | CuCoNiZnS | ||||||||||
| Chalcocite | Cu2S | ||||||||||
| Phosphates, Arsenates, Vanadate | Wilhelmkleinite | ZnFe2(AsO4)2(OH)2 | P | P | P | P | |||||
| not detected | ZnPOx or ZnFe − POx | ||||||||||
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khoshnoodi, M.; Dipple, G.; Baldwin, S.A. Mineralogical Study of a Biologically-Based Treatment System That Removes Arsenic, Zinc and Copper from Landfill Leachate. Minerals 2013, 3, 427-449. https://doi.org/10.3390/min3040427
Khoshnoodi M, Dipple G, Baldwin SA. Mineralogical Study of a Biologically-Based Treatment System That Removes Arsenic, Zinc and Copper from Landfill Leachate. Minerals. 2013; 3(4):427-449. https://doi.org/10.3390/min3040427
Chicago/Turabian StyleKhoshnoodi, Maryam, Gregory Dipple, and Susan A. Baldwin. 2013. "Mineralogical Study of a Biologically-Based Treatment System That Removes Arsenic, Zinc and Copper from Landfill Leachate" Minerals 3, no. 4: 427-449. https://doi.org/10.3390/min3040427
APA StyleKhoshnoodi, M., Dipple, G., & Baldwin, S. A. (2013). Mineralogical Study of a Biologically-Based Treatment System That Removes Arsenic, Zinc and Copper from Landfill Leachate. Minerals, 3(4), 427-449. https://doi.org/10.3390/min3040427




