Crystallization Conditions of Basaltic Lavas Based on Clinopyroxene and Olivine Phenocryst Petrology: A Case Study from the Neogene Lavarab Alkaline Basaltic Lavas (LABL), Eastern Iran
Abstract
1. Introduction
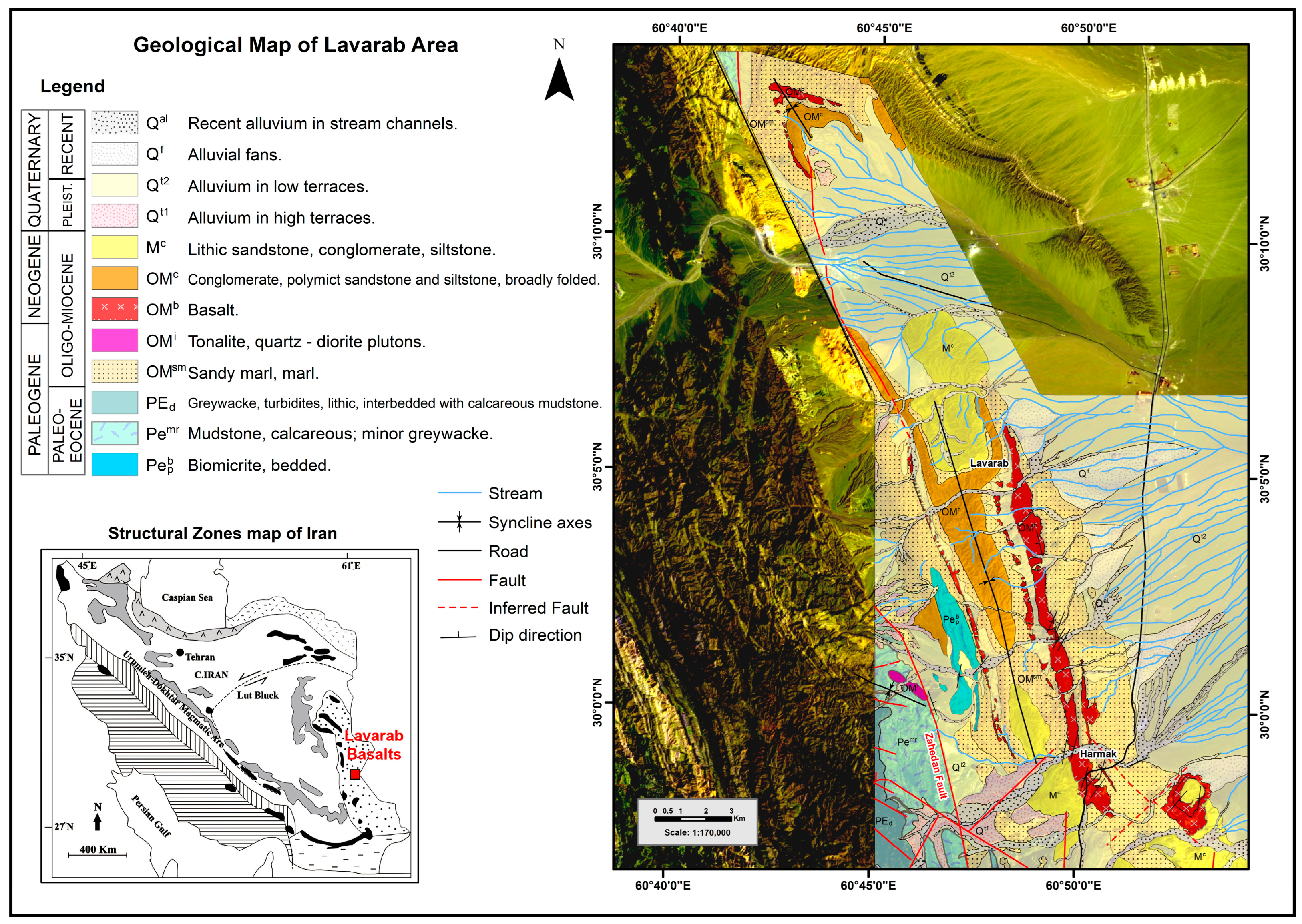
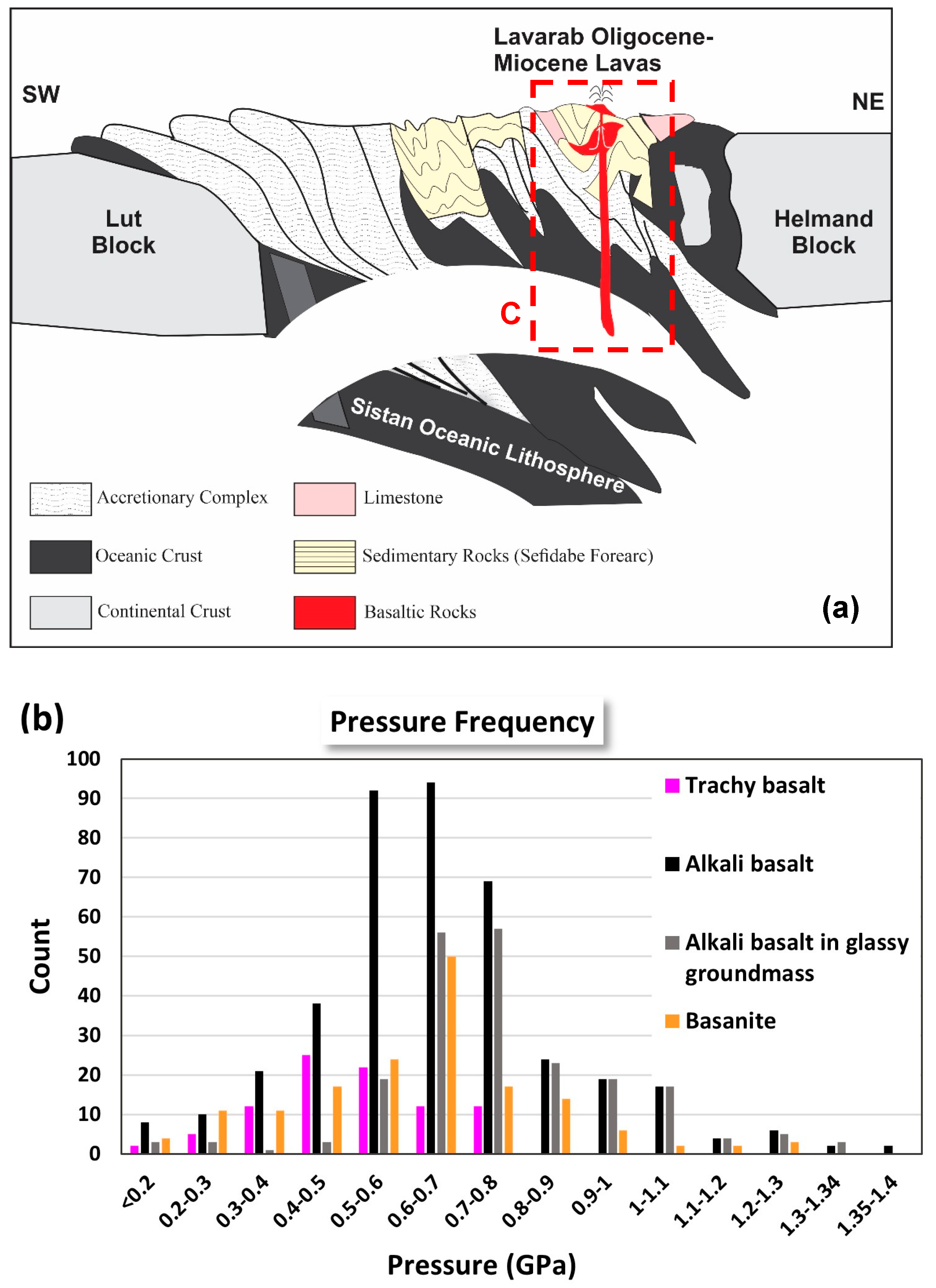
2. Geological Setting and Study Area
3. Field Relations and Petrography
3.1. Trachybasalt
3.2. Alkali Basalt
3.3. Basanite
4. Analytical Technique
4.1. Electron Probe Micro-Analyzer (EPMA)
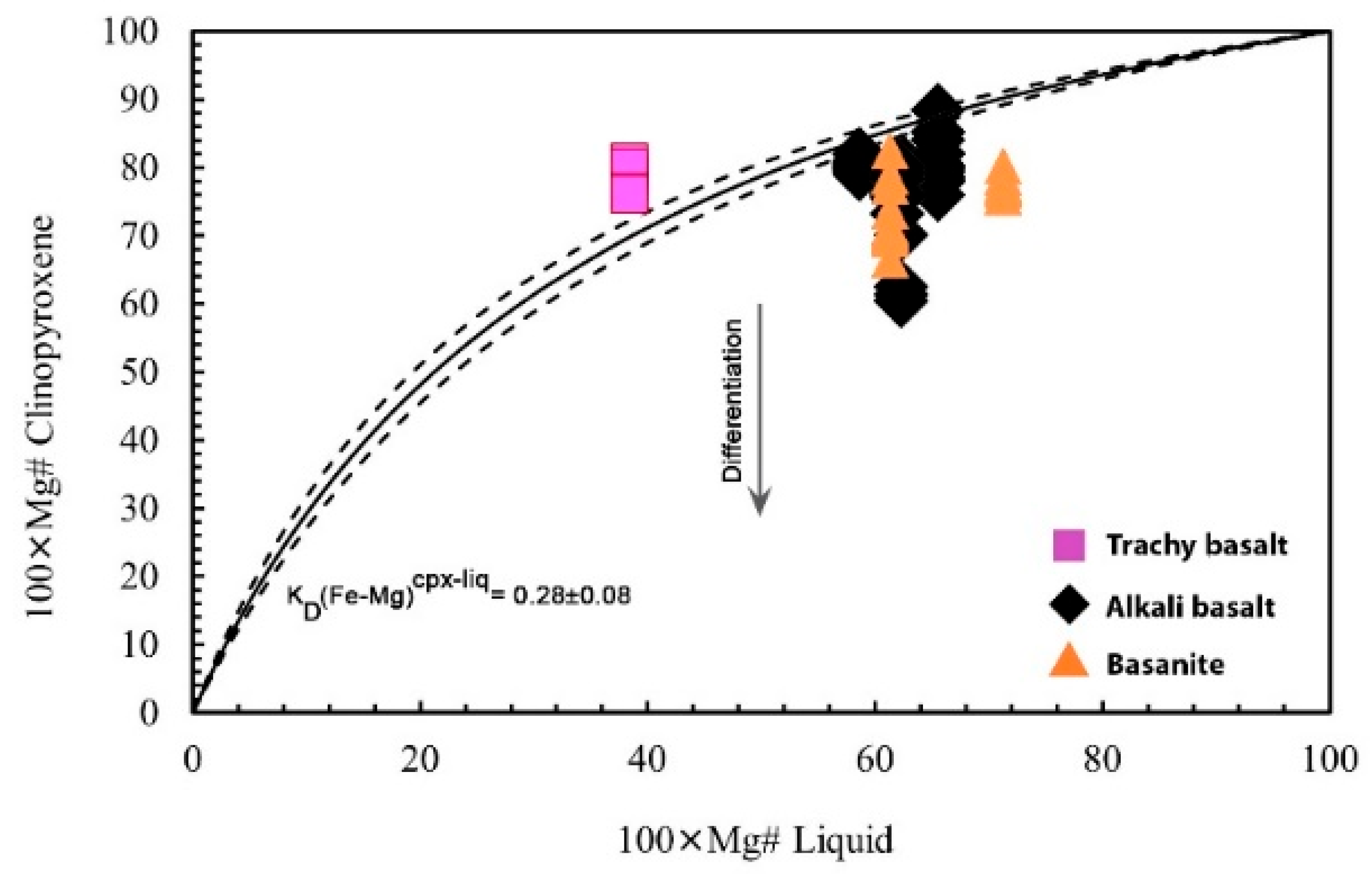
4.2. Test for Equilibrium in Mineral-Liquid Geothermobarometry
4.3. Programs to Calculate Temperature and Pressure
5. Results
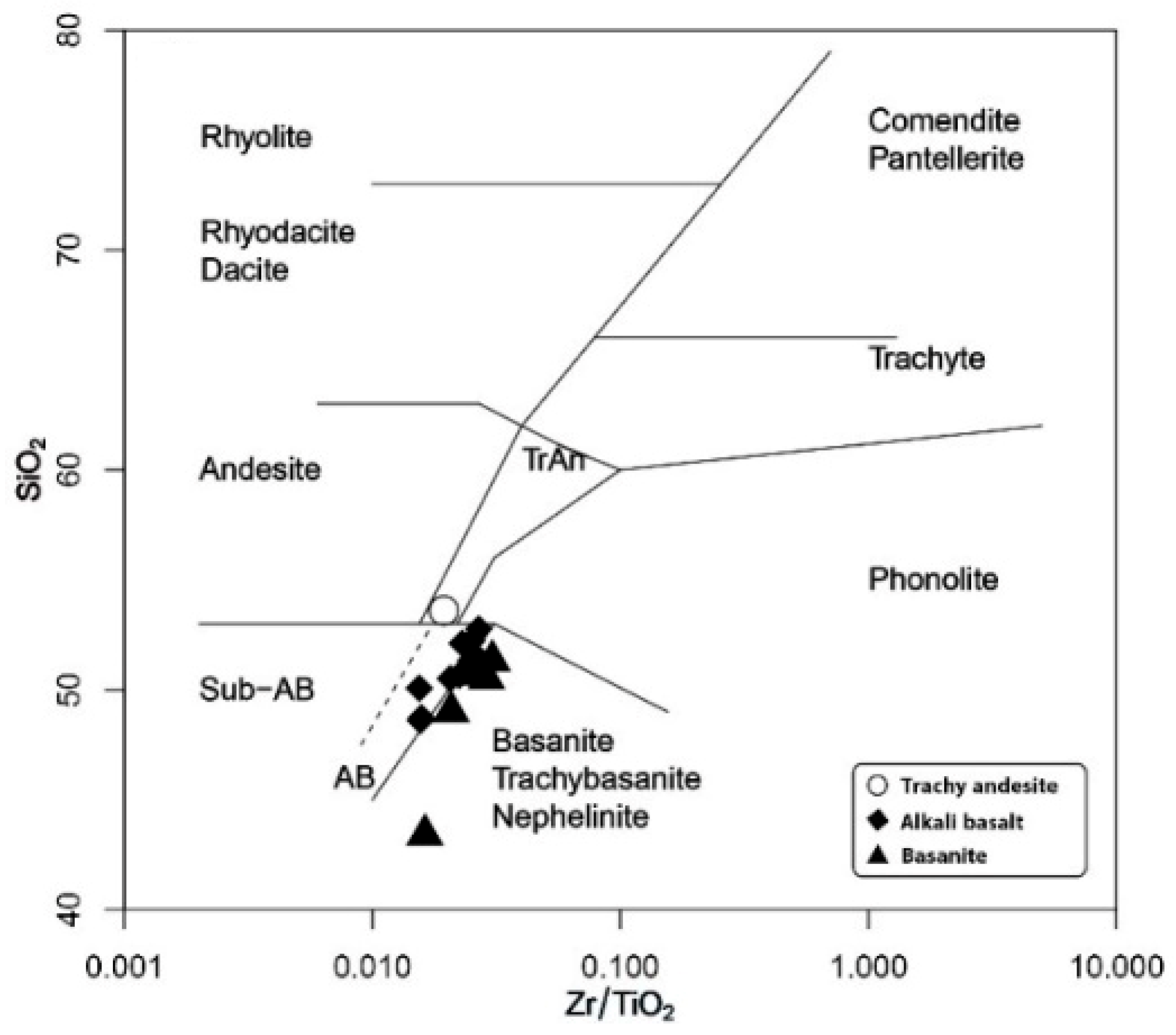
5.1. Whole-Rock Composition
5.2. Mineral Chemistry
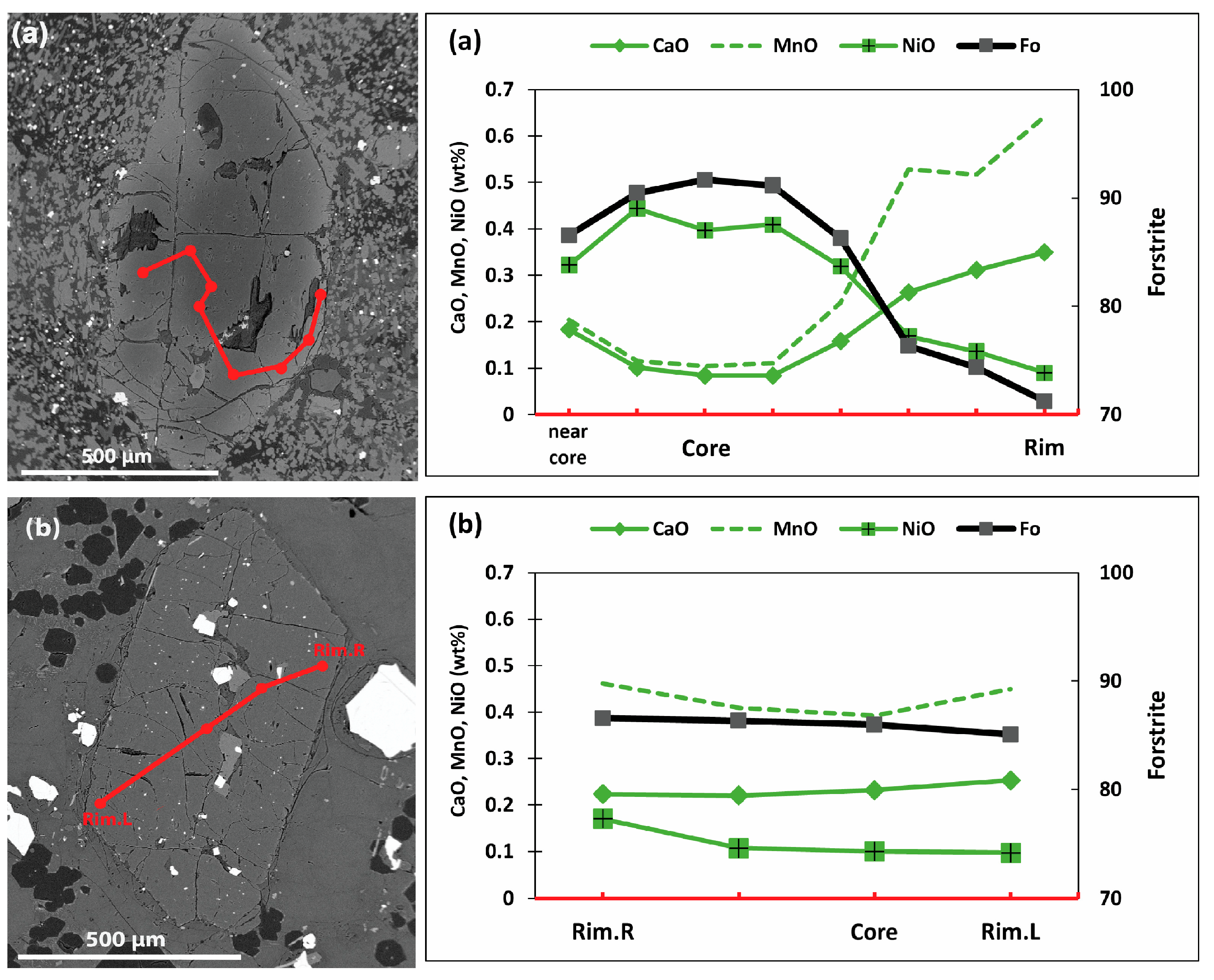
5.2.1. Olivine
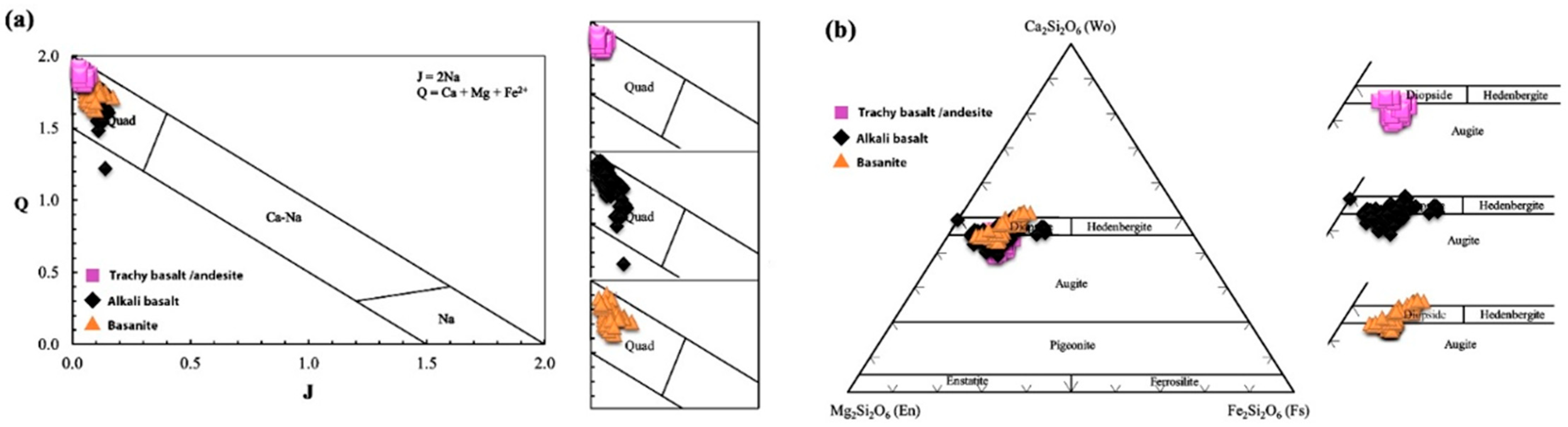
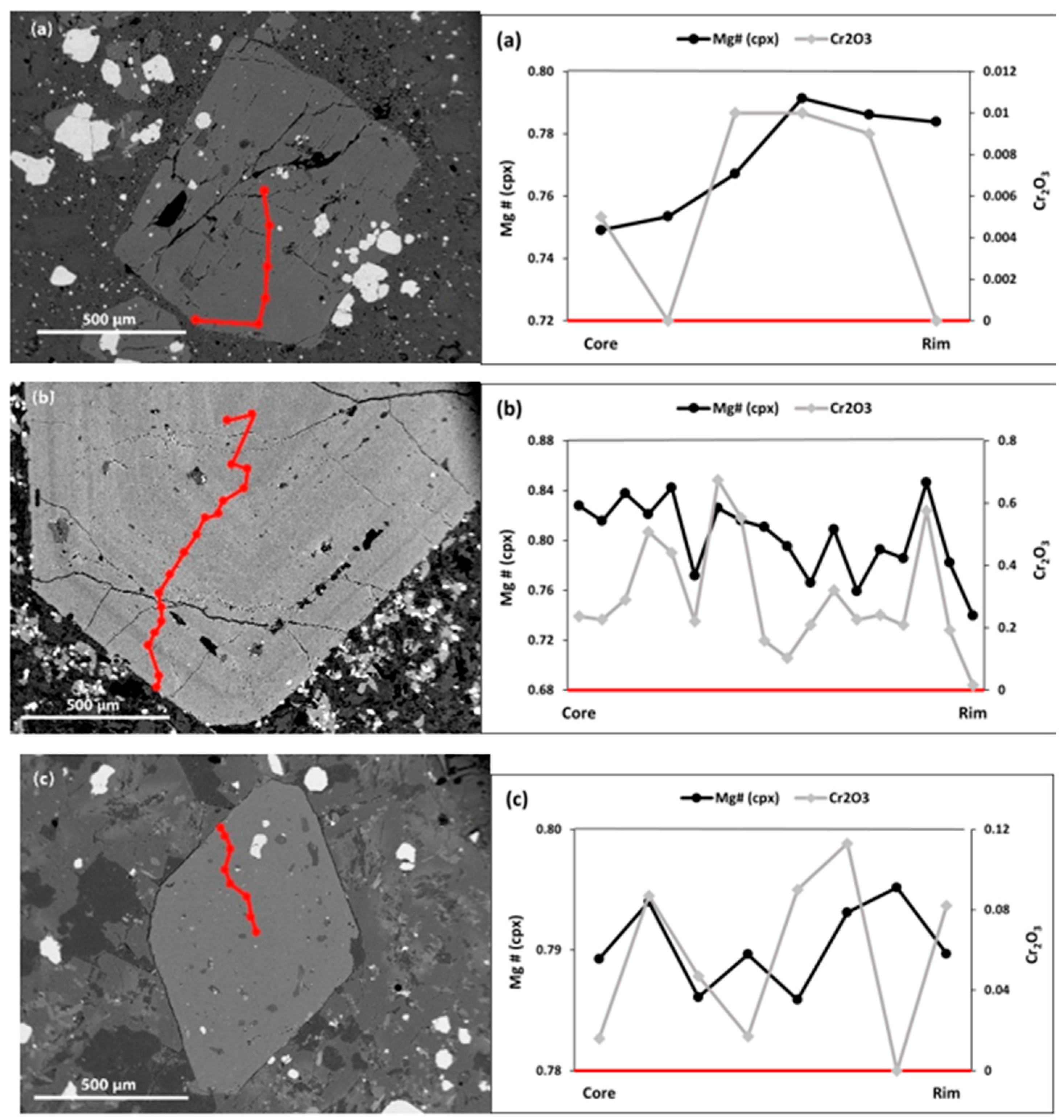
5.2.2. Clinopyroxene
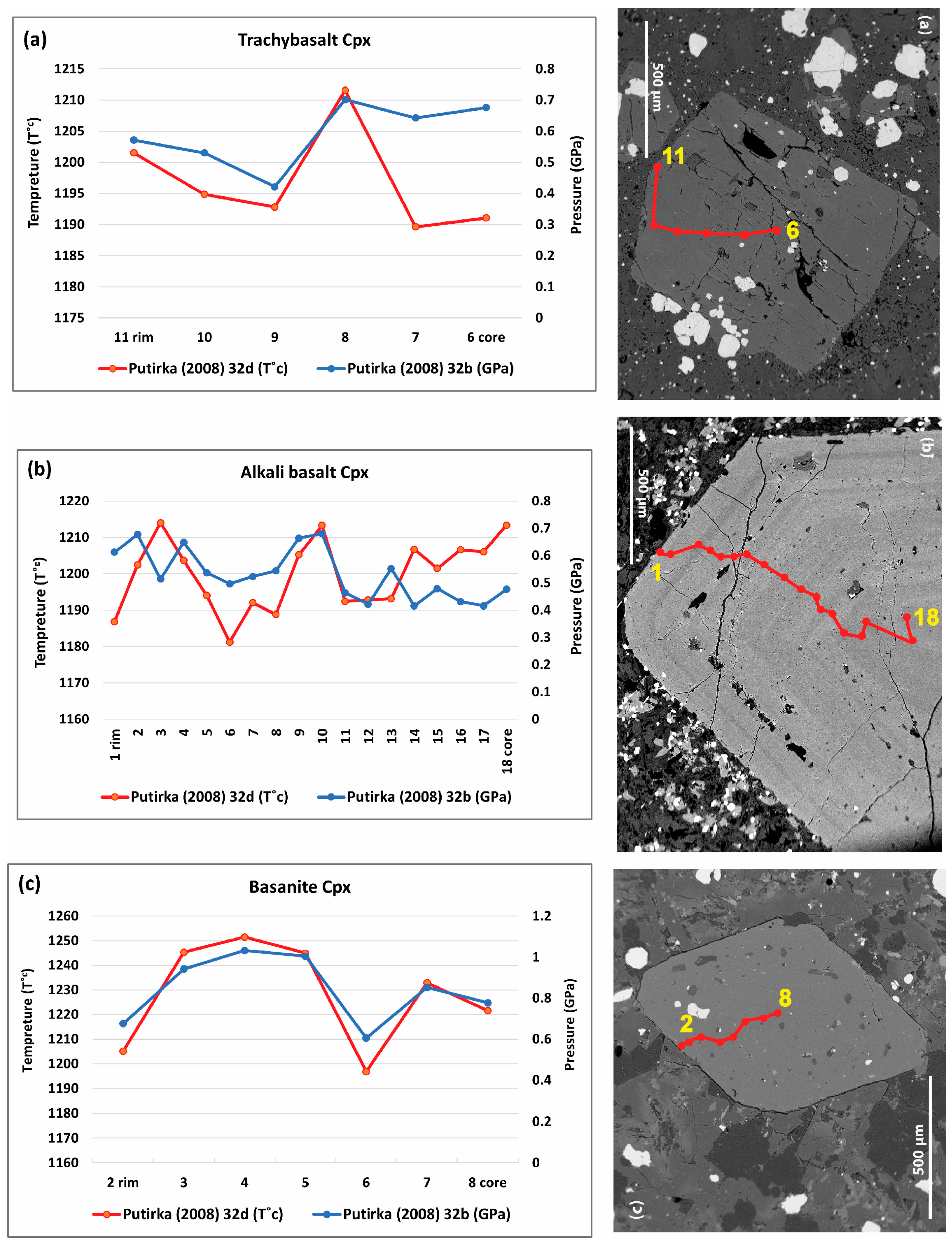
- Clinopyroxene micro-phenocrysts
- Clinopyroxene phenocrysts
- Clinopyroxene mega-phenocrysts
6. Discussion
6.1. Crystallization Conditions
6.1.1. Whole Rock/Glass Geothermometry
6.1.2. Olivine Geothermometry
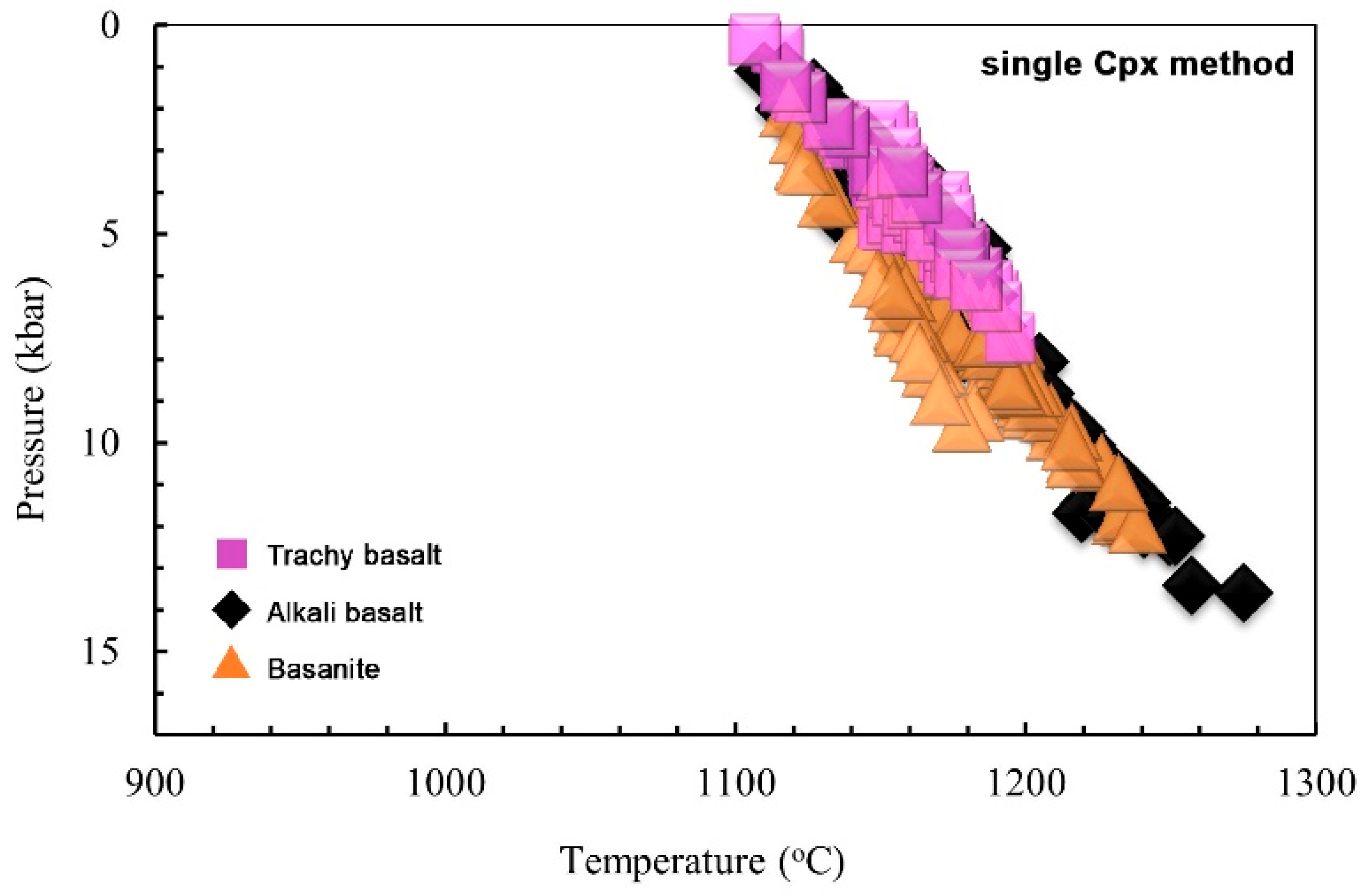
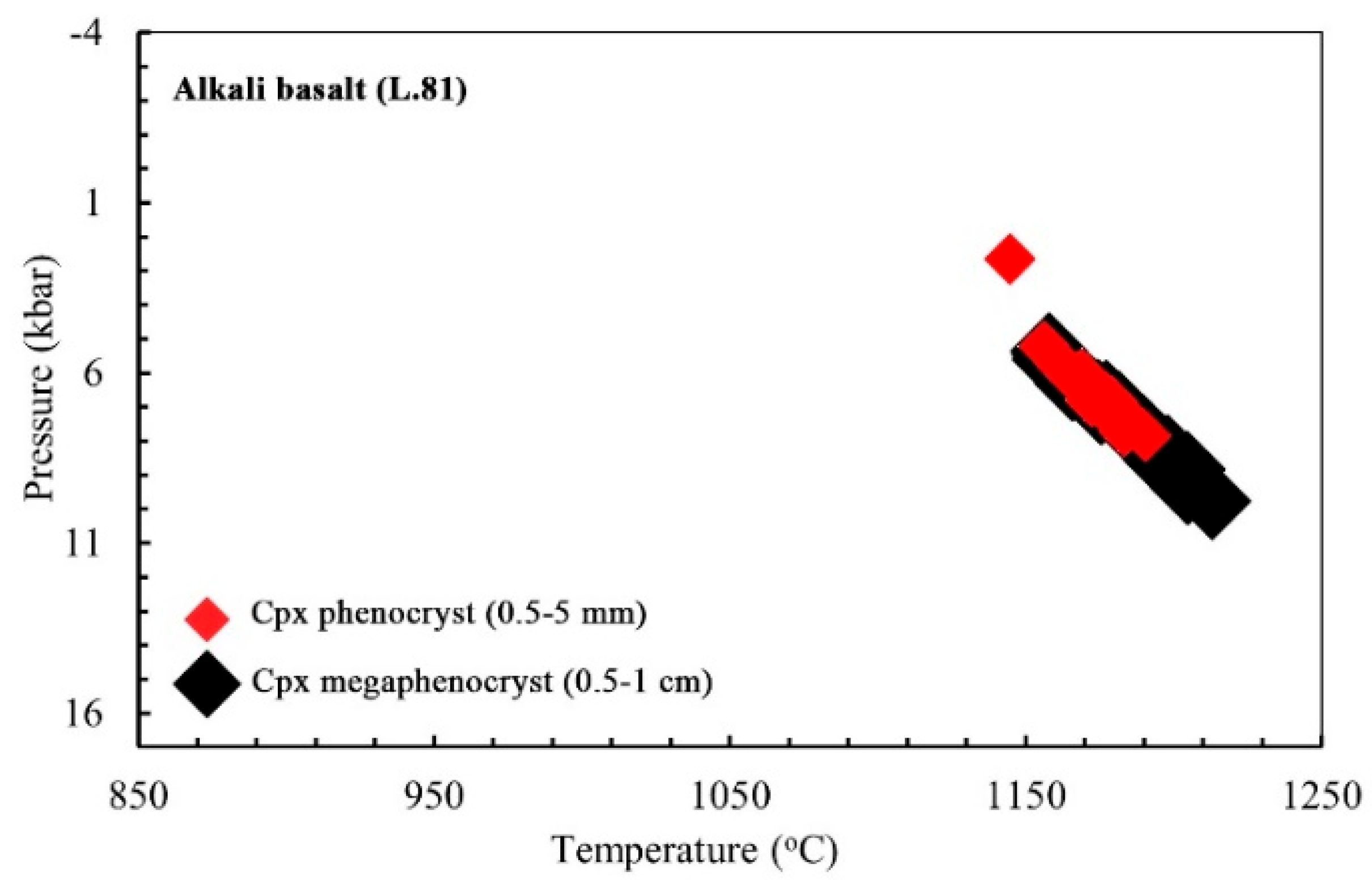
6.1.3. Clinopyroxene Geothermobarometry

| Clinopyroxene-Only Thermobarometry | |||||
|---|---|---|---|---|---|
| Soesoo (1997) [19] | Putirka (2008) [21] RiMG | ||||
| Equation (32a) (SEE = ±0.26 GPa) | Equation (32b) (SEE = ±0.26 GPa) | Equation (32d) (SEE = ±58 °C) | |||
| T (°C) | P (GPa) | P (GPa) | P (GPa) | T(°C) | |
| Trachybasalt n = 93 | 1150–1200 | 0.3–0.6 | 0.12–0.9 (avg = 0.55) | 0.13–0.9 (avg = 0.51) | 1156–1223 (avg = 1190) |
| Alkali basalts n = 418 | 1165–1270 | 0.4–1.45 | 0.03–1.99 (avg = 0.74) | 0.1–1.4 (avg = 0.65) | 1135–1287 (avg = 1209) |
| Basanites n = 169 | 1170–1230 | 0.5–1.25 | 0.1–1.57 (avg = 0.8) | 0.15–1.34 (avg = 0.71) | 934–1278 (avg = 1200) |
6.2. Thermobarometric Constraints and Limitations
6.2.1. Magma Composition and Mineral-Melt Equilibrium Test
6.2.2. Model-Specific Uncertainties
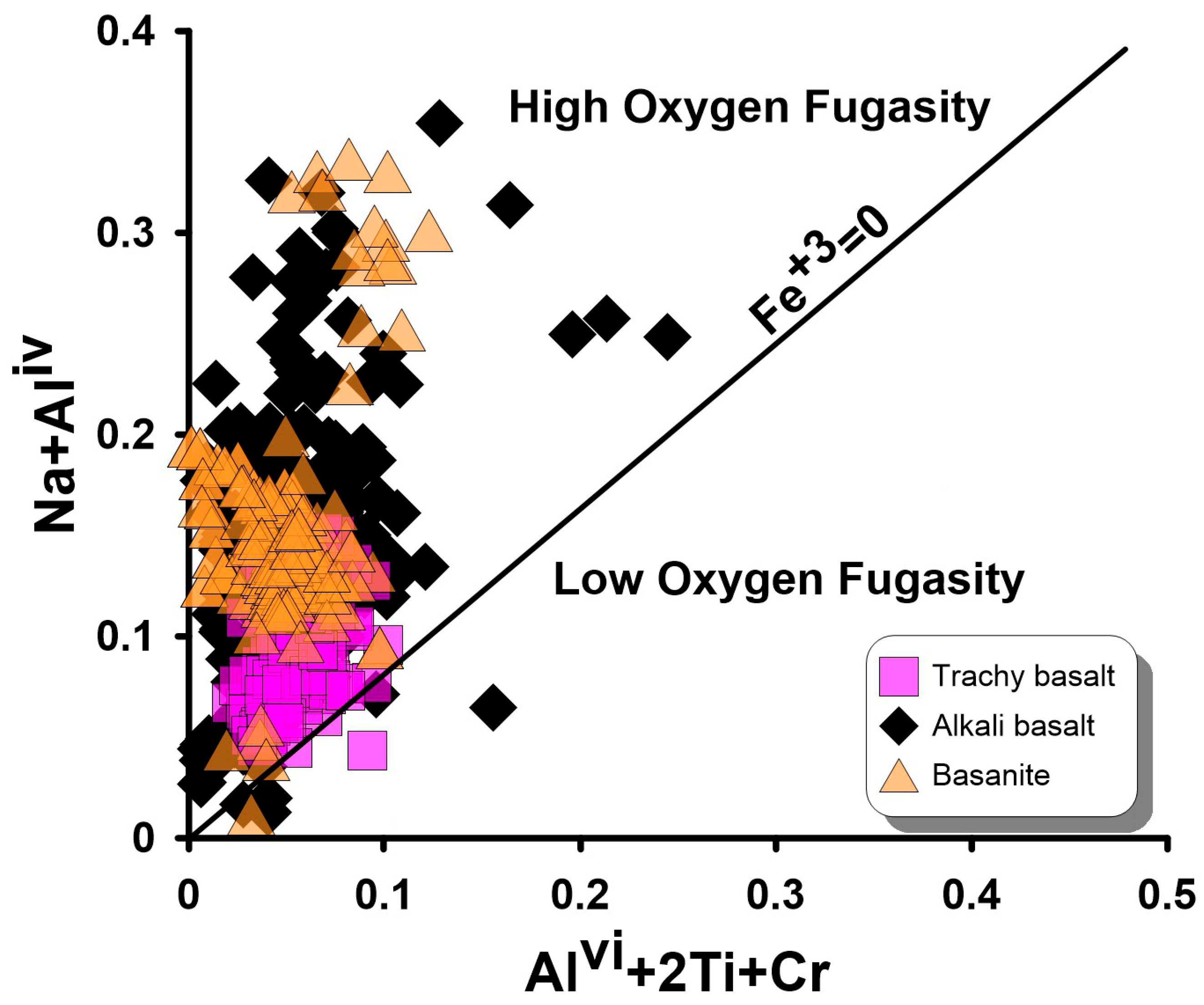
6.2.3. Redox Conditions and Their Thermobarometric Implications
6.3. Magma Type and Tectonic Setting Based on Clinopyroxene Composition

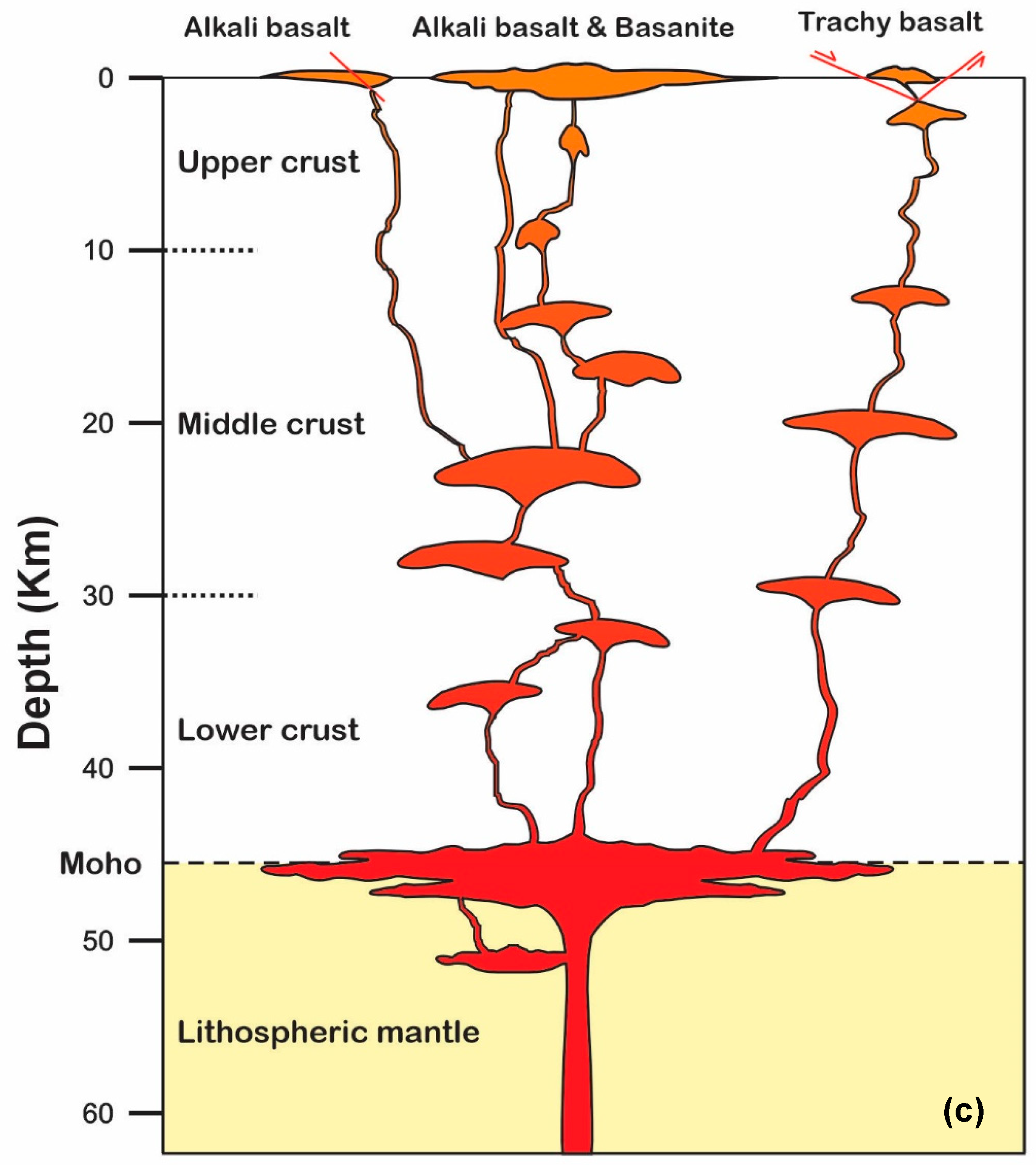
6.4. Magma Crystallization and Evolution Modeling
- Deep Storage (1.0–1.4 GPa; ~38–51 km):
- Mid-Crustal Storage (0.3–0.8 GPa; ~11–30 km):
- Shallow Conduits (<0.3 GPa; <11 km):
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LABL | Lavarab Alkaline Basaltic Lavas |
| GSI | Geological Survey of Iran |
| SEE | Standard error of estimate |
| RiMG | Reviews in Mineralogy and Geochemistry |
| Quad | Ca-Mg-Fe pyroxenes |
| N/A | Not available |
References
- Xi, J.; Yang, Y.; Xu, H.; Xian, H.; Pan, F.; Li, S.; Xue, S.; Cao, Y.; Zhu, J.; He, H. Reconstruction of Magma Plumbing System and Regional Magmatic Processes via Chemical and Structural Zoning of Biotite in Rhyolite from Long Valley, CA. JGR Solid Earth 2024, 129, e2024JB029205. [Google Scholar] [CrossRef]
- Blundy, J.; Cashman, K. Petrologic Reconstruction of Magmatic System Variables and Processes. Rev. Mineral. Geochem. 2008, 69, 179–239. [Google Scholar] [CrossRef]
- Annen, C.; Blundy, J.D.; Sparks, R.S.J. The Genesis of Intermediate and Silicic Magmas in Deep Crustal Hot Zones. J. Petrol. 2006, 47, 505–539. [Google Scholar] [CrossRef]
- Cashman, K.V.; Sparks, R.S.J.; Blundy, J.D. Vertically Extensive and Unstable Magmatic Systems: A Unified View of Igneous Processes | Science, 355(6331), Eaag3055. Available online: https://www.science.org/doi/abs/10.1126/science.aag3055 (accessed on 22 August 2025).
- Behruzi, A. Geological Quadrangle Map of Zahedan; Geological Survey of Iran: Tehran, Iran, 1973. [Google Scholar]
- Aghanabati, A. Geological Map of Kuh-e Do Poshti; Geological Survey of Iran: Tehran, Iran, 1987. [Google Scholar]
- Aghanabati, A. Geological Quadrangle Map of Daryacheh-Ye-Hamun; Geological Survey of Iran: Tehran, Iran, 1991. [Google Scholar]
- Camp, V.E.; Griffis, R.J. Character, Genesis and Tectonic Setting of Igneous Rocks in the Sistan Suture Zone, Eastern Iran. Lithos 1982, 15, 221–239. [Google Scholar] [CrossRef]
- Tirrul, R.; Bell, I.R.; Griffis, R.J.; Camp, V.E. The Sistan Suture Zone of Eastern Iran. GSA Bull. 1983, 94, 134–150. [Google Scholar] [CrossRef]
- Leterrier, J.; Maury, R.C.; Thonon, P.; Girard, D.; Marchal, M. Clinopyroxene Composition as a Method of Identification of the Magmatic Affinities of Paleo-Volcanic Series. Earth Planet. Sci. Lett. 1982, 59, 139–154. [Google Scholar] [CrossRef]
- Nisbet, E.G.; Pearce, J.A. Clinopyroxene Composition in Mafic Lavas from Different Tectonic Settings. Contrib. Mineral. Petrol. 1977, 63, 149–160. [Google Scholar] [CrossRef]
- Wood, B.J.; Banno, S. Garnet-Orthopyroxene and Orthopyroxene-Clinopyroxene Relationships in Simple and Complex Systems. Contr. Mineral. Petrol. 1973, 42, 109–124. [Google Scholar] [CrossRef]
- Wells, P.R.A. Pyroxene Thermometry in Simple and Complex Systems. Contr. Mineral. Petrol. 1977, 62, 129–139. [Google Scholar] [CrossRef]
- Schweitzer, E.L.; Papike, J.J.; Bence, A.E. Statistical Analysis of Clinopyroxenes from Deep-Sea Basalts. Am. Mineral. 1979, 64, 501–513. [Google Scholar] [CrossRef]
- Lindsley, D.H. Pyroxene Thermometry. Am. Mineral. 1983, 68, 477–493. [Google Scholar]
- Davidson, P.M. Thermodynamic Analysis of Quadrilateral Pyroxenes. Part 1: Derivation of the Ternary Nonconvergent Site-Disorder Model. Contr. Mineral. Petrol. 1985, 91, 383–389. [Google Scholar] [CrossRef]
- Davidson, P.M.; Lindsley, D.H. Thermodynamic Analysis of Quadrilateral Pyroxenes. Part II: Model Calibration from Experiments and Applications to Geothermometry. Contr. Mineral. Petrol. 1985, 91, 390–404. [Google Scholar] [CrossRef]
- Bertrand, P.; Mercier, J.-C.C. The Mutual Solubility of Coexisting Ortho- and Clinopyroxene: Toward an Absolute Geothermometer for the Natural System? Earth Planet. Sci. Lett. 1985, 76, 109–122. [Google Scholar] [CrossRef]
- Soesoo, A. A Multivariate Statistical Analysis of Clinopyroxene Composition: Empirical Coordinates for the Crystallisation PT-estimations. GFF 1997, 119, 55–60. [Google Scholar] [CrossRef]
- Nimis, P.; Taylor, W.R. Single Clinopyroxene Thermobarometry for Garnet Peridotites. Part I. Calibration and Testing of a Cr-in-Cpx Barometer and an Enstatite-in-Cpx Thermometer. Contrib Miner. Pet. 2000, 139, 541–554. [Google Scholar] [CrossRef]
- Putirka, K.D. Thermometers and Barometers for Volcanic Systems. Rev. Mineral. Geochem. 2008, 69, 61–120. [Google Scholar] [CrossRef]
- Wieser, P.E.; Kent, A.J.R.; Till, C.B.; Donovan, J.; Neave, D.A.; Blatter, D.L.; Krawczynski, M.J. Barometers Behaving Badly I: Assessing the Influence of Analytical and Experimental Uncertainty on Clinopyroxene Thermobarometry Calculations at Crustal Conditions. J. Petrol. 2023, 64, egac126. [Google Scholar] [CrossRef]
- Gill, R. Igneous Rocks and Processes: A Practical Guide; Wiley-Blackwell: Chichester, UK, 2010; ISBN 978-1-4443-3065-6. [Google Scholar]
- Şengör, A.M.C.; Altıner, D.; Cin, A.; Ustaömer, T.; Hsü, K.J. Origin and Assembly of the Tethyside Orogenic Collage at the Expense of Gondwana Land. Geol. Soc. Lond. Spec. Publ. 1988, 37, 119–181. [Google Scholar] [CrossRef]
- Berberian, M. Generalized Tectonic Map of Iran. In Berberian M (Ed) Continental Deformation in the Iranian Plateau; Geological Survey of Iran: Tehran, Iran, 1983. [Google Scholar]
- Rezaei-Kahkhaei, M.; Corfu, F.; Galindo, C.; Rahbar, R.; Ghasemi, H. Adakite Genesis and Plate Convergent Process: Constraints from Whole Rock and Mineral Chemistry, Sr, Nd, Pb Isotopic Compositions and U-Pb Ages of the Lakhshak Magmatic Suite, East Iran. Lithos 2022, 426–427, 106806. [Google Scholar] [CrossRef]
- Damani Gol, S.; Bagheri, S. Paleogene Thrust System in the Eastern Iranian Ranges; Kharazmi University: Tehran, Iran, 2021. [Google Scholar]
- Houshmand-Mananvi, S.; Rezaei-Kahkhaei, M.; Ghasemi, H. Whole Rock Geochemistry and Tectonic Setting of Oligocene-Miocene Lavarab Lava (North Zahedan). Sci. Q. J. Geosci. 2024, 34, 67–86. [Google Scholar] [CrossRef]
- Rezaei-Kahkhaei, M.; Esmaeily, D.; Sahraei, H. Evaluation of Impact Processes in the Formation of Neshveh Volcanic Rocks, NW Saveh. Sci. Q. J. Geosci. 2018, 28, 285–294. [Google Scholar] [CrossRef]
- Gündüz, M.; Asan, K. MagMin_PT: An Excel-Based Mineral Classification and Geothermobarometry Program for Magmatic Rocks. MinMag 2023, 87, 1–9. [Google Scholar] [CrossRef]
- Roeder, P.L.; Emslie, R.F. Olivine-Liquid Equilibrium. Contr. Mineral. Petrol. 1970, 29, 275–289. [Google Scholar] [CrossRef]
- Putirka, K.D.; Mikaelian, H.; Ryerson, F.; Shaw, H. New Clinopyroxene-Liquid Thermobarometers for Mafic, Evolved, and Volatile-Bearing Lava Compositions, with Applications to Lavas from Tibet and the Snake River Plain, Idaho. Am. Mineral. 2003, 88, 1542–1554. [Google Scholar] [CrossRef]
- Putirka, K.; Ryerson, F.J.; Mikaelian, H. New Igneous Thermobarometers for Mafic and Evolved Lava Compositions, Based on Clinopyroxene + Liquid Equilibria. Am Miner. 2003, 88, 1542–1554. [Google Scholar] [CrossRef]
- Yavuz, F. WinPyrox: A Windows Program for Pyroxene Calculation Classification and Thermobarometry. Am. Mineral. 2013, 98, 1338–1359. [Google Scholar] [CrossRef]
- Peate, D.W.; Hawkesworth, C.J. Lithospheric to Asthenospheric Transition in Low-Ti Flood Basalts from Southern Paraná, Brazil. Chem. Geol. 1996, 127, 1–24. [Google Scholar] [CrossRef]
- Simon, S.B.; Sutton, S.R. Valences of Ti, Cr, and V in Apollo 17 High-Ti and Very Low-Ti Basalts and Implications for Their Formation. Meteorit. Planet. Sci. 2018, 53, 2138–2154. [Google Scholar] [CrossRef]
- Wilson, M. Igneous Petrogenesis: A Global Tectonic Approach; Springer: Dordrecht, The Netherlands, 1989. [Google Scholar]
- Winchester, J.A.; Floyd, P.A. Geochemical Discrimination of Different Magma Series and Their Differentiation Products Using Immobile Elements. Chem. Geol. 1977, 20, 325–343. [Google Scholar] [CrossRef]
- Sobolev, A.V.; Hofmann, A.W.; Kuzmin, D.V.; Yaxley, G.M.; Arndt, N.T.; Chung, S.L.; Danyushevsky, L.V.; Elliott, T.; Frey, F.A.; Garcia, M.O.; et al. The Amount of Recycled Crust in Sources of Mantle-Derived Melts. Science 2007, 316, 412–417. [Google Scholar] [CrossRef]
- Morimoto, N.; Kitamura, M. Q-J Diagram for Classification of pyroxenes. J. Jpn. Assoc. Mineral. Petrol. Econ. Geol. 1983, 78, 141. [Google Scholar]
- Morimoto, N.; Fabrise, J.; Ferguson, A.; Ginzburg, I.V.; Ross, M.; Seifert, F.A.; Zussman, J.; Akoi, K.; Gottardi, G. Nomenclature of Pyroxenes. Am. Mineral. 1988, 173, 1123–1133. [Google Scholar]
- Wang, X.; Hou, T.; Wang, M.; Zhang, C.; Zhang, Z.; Pan, R.; Marxer, F.; Zhang, H. A New Clinopyroxene Thermobarometer for Mafic to Intermediate Magmatic Systems. Eur. J. Mineral. 2021, 33, 621–637. [Google Scholar] [CrossRef]
- Zhang, L.; Hong, L.-B.; Qian, S.-P.; He, P.-L.; He, M.-H.; Yang, Y.-N.; Wang, J.-T.; Zhang, Y.-Q.; Ren, Z.-Y. The Effect of Elemental Diffusion on the Application of Olivine-Composition-Based Magmatic Thermometry, Oxybarometry, and Hygrometry: A Case Study of Olivine Phenocrysts from the Jiagedaqi Basalts, Northeast China. Am. Mineral. 2023, 108, 1449–1460. [Google Scholar] [CrossRef]
- Arabzadeh Baniasadi, M.; Ghasemi, H.; Angiboust, S.; Rezaei Kahkhaei, M.; Papadopoulou, L. Application of clinopyroxene geothermobarometers on the gabbro/dioritic rocks associated with the Gowd-e-Howz (Siah-Kuh) granitoid stock, Baft, Kerman. Sci. Q. J. Geosci. 2024, 34, 117–134. [Google Scholar] [CrossRef]
- Helz, R.T.; Thornber, C.R. Geothermometry of Kilauea Iki Lava Lake, Hawaii. Bull Volcanol 1987, 49, 651–668. [Google Scholar] [CrossRef]
- Montierth, C.; Johnston, A.D.; Cashman, K.V. An Empirical Glass-Composition-Based Geothermometer for Mauna Loa Lavas. In Mauna Loa Revealed: Structure, Composition, History, and Hazards; American Geophysical Union (AGU): Washington, DC, USA, 1995; pp. 207–217. ISBN 978-1-118-66433-9. [Google Scholar]
- Yang, H.-J.; Frey, F.A.; Clague, D.A.; Garcia, M.O. Mineral Chemistry of Submarine Lavas from Hilo Ridge, Hawaii: Implications for Magmatic Processes within Hawaiian Rift Zones. Contrib Miner. Pet. 1999, 135, 355–372. [Google Scholar] [CrossRef]
- Toplis, M.J. The Thermodynamics of Iron and Magnesium Partitioning between Olivine and Liquid: Criteria for Assessing and Predicting Equilibrium in Natural and Experimental Systems. Contrib Miner. Pet. 2005, 149, 22–39. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Dungan, M.A.; Blanchard, D.P.; Long, P.E. Magma Mixing at Mid-Ocean Ridges: Evidence from Basalts Drilled near 22° N on the Mid-Atlantic Ridge. Tectonophysics 1979, 55, 35–61. [Google Scholar] [CrossRef]
- Putirka, K.D.; Perfit, M.; Ryerson, F.J.; Jackson, M.G. Ambient and Excess Mantle Temperatures, Olivine Thermometry, and Active vs. Passive Upwelling. Chem. Geol. 2007, 241, 177–206. [Google Scholar] [CrossRef]
- Nimis, P. A Clinopyroxene Geobarometer for Basaltic Systems Based on Crystal-Structure Modeling. Contrib Miner. Pet. 1995, 121, 115–125. [Google Scholar] [CrossRef]
- Nimis, P.; Ulmer, P. Clinopyroxene Geobarometry of Magmatic Rocks Part 1: An Expanded Structural Geobarometer for Anhydrous and Hydrous, Basic and Ultrabasic Systems. Contrib Miner. Pet. 1998, 133, 122–135. [Google Scholar] [CrossRef]
- Neave, D.A.; Putirka, K.D. A New Clinopyroxene-Liquid Barometer, and Implications for Magma Storage Pressures under Icelandic Rift Zones. Am. Mineral. 2017, 102, 777–794. [Google Scholar] [CrossRef]
- Putirka, K.; Johnson, M.; Kinzler, R.; Longhi, J.; Walker, D. Thermobarometry of Mafic Igneous Rocks Based on Clinopyroxene-Liquid Equilibria, 0–30 Kbar. Contrib Miner. Pet. 1996, 123, 92–108. [Google Scholar] [CrossRef]
- Tormey, D.R.; Grove, T.L.; Bryan, W.B. Experimental Petrology of Normal MORB near the Kane Fracture Zone: 22°–25° N, Mid-Atlantic Ridge. Contr. Mineral. Petrol. 1987, 96, 121–139. [Google Scholar] [CrossRef]
- Berndt, J.; Koepke, J.; Holtz, F. An Experimental Investigation of the Influence of Water and Oxygen Fugacity on Differentiation of MORB at 200 MPa. J. Petrol. 2005, 46, 135–167. [Google Scholar] [CrossRef]
- Thy, P.; Lesher, C.E.; Nielsen, T.F.D.; Brooks, C.K. Experimental Constraints on the Skaergaard Liquid Line of Descent. Lithos 2006, 92, 154–180. [Google Scholar] [CrossRef]
- Botcharnikov, R.E.; Koepke, J.; Holtz, F.; McCammon, C.; Wilke, M. The Effect of Water Activity on the Oxidation and Structural State of Fe in a Ferro-Basaltic Melt. Geochim. Et Cosmochim. Acta 2005, 69, 5071–5085. [Google Scholar] [CrossRef]
- Husen, A.; Almeev, R.R.; Holtz, F. The Effect of H2O and Pressure on Multiple Saturation and Liquid Lines of Descent in Basalt from the Shatsky Rise. J. Petrol. 2016, 57, 309–344. [Google Scholar] [CrossRef]
- Sundermeyer, C.; Gätjen, J.; Weimann, L.; Wörner, G. Timescales from Magma Mixing to Eruption in Alkaline Volcanism in the Eifel Volcanic Fields, Western Germany. Contrib Miner. Pet. 2020, 175, 77. [Google Scholar] [CrossRef]
- Carmichael, I.S.E. The Redox States of Basic and Silicic Magmas: A Reflection of Their Source Regions? Contr. Mineral. Petrol. 1991, 106, 129–141. [Google Scholar] [CrossRef]
- Chinner, G.A. Pelitic Gneisses with Varying Ferrous/Ferric Ratios from Glen Clova, Angus, Scotland. J. Petrol. 1960, 1, 178–217. [Google Scholar] [CrossRef]
- Rumble, D. Fe-Ti Oxide Minerals from Regionally Metamorphosed Quartzites of Western New Hampshire. Contr. Mineral. Petrol. 1973, 42, 181–195. [Google Scholar] [CrossRef]
- Powell, R.; Sandiford, M. Sapphirine and Spinel Phase Relationships in the System FeO-MgO-Al2O3-SiO2-TiO2-O2 in the Presence of Quartz and Hypersthene. Contr. Mineral. Petrol. 1988, 98, 64–71. [Google Scholar] [CrossRef]
- Clarke, G.L.; Powell, R.; Guiraud, M. Low-Pressure Granulite Facies Metapelitic Assemblages and Corona Textures from MacRobertson Land, East Antarctica: The Importance of Fe203and TiOz in Accounting for Spinel-Bearing Assemblages. J. Metamorph. Geol. 1989, 7, 323–335. [Google Scholar] [CrossRef]
- Ague, J.J.; Baxter, E.F.; Eckert, J.O. JR High fO2 During Sillimanite Zone Metamorphism of Part of the Barrovian Type Locality, Glen Clova, Scotland. J. Petrol. 2001, 42, 1301–1320. [Google Scholar] [CrossRef]
- Evans, K.A. Redox Decoupling and Redox Budgets: Conceptual Tools for the Study of Earth Systems. Geology 2006, 34, 489–492. [Google Scholar] [CrossRef]
- Diener, J.F.A.; Powell, R. Influence of Ferric Iron on the Stability of Mineral Assemblages. J. Metamorph. Geol. 2010, 28, 599–613. [Google Scholar] [CrossRef]
- LeBas, M.J. The Role of Aluminium in Igneous Clinopyroxenes with Relation to Their Parentage. Am. J. Sci. 1962, 260, 267–288. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Z.; Zhang, R.; Reichow, M.K.; Cheng, Z.; Zhang, Z.; Santosh, M. A Machine-Learning-Based Approach Using Clinopyroxene Data to Improve Accuracy and Efficiency in Predicting Tectonic Settings: Implications for Rodinia Supercontinent Breakup Triggered by Mantle Plume Events. Am. Mineral. 2025, 110, 996–1011. [Google Scholar] [CrossRef]
- Hossein-Zadeh, S. Estimation of Moho Depth in Iran by Combination of Gravity and Seismic Data; Institude for Advanced Studies in Basic Sceineces, Gava Zang: Znajan, Iran, 2012. [Google Scholar]
- Sepahvand, M.R. Variations of Moho Depth and Vp/Vs Ratio in Central and Eastern Iran Using the Zhu and Kanamori Method; Kerman Graduate University of Technology (KGUT): Kerman, Iran, 2016. [Google Scholar]





| Clinopyroxene-Whole-Rock Equilibrium Thermobarometry | ||||
|---|---|---|---|---|
| Putirka et al. (2003) [32] | Putirka (2008) [21] RiMG MODELS | |||
| T (°C) | P (GPa) | P (GPa) | T (°C) | |
| (SEE * = ±61 °C) | (SEE = ±0.48 GPa) | Equation (31) (SEE = ±0.3 GPa) | Equation (33) (SEE = ±46 °C) | |
| Trachybasalt n = 6 | 1092–1112 (avg = 1104) | 0.00–0.2 (avg = 0.1) | 0.5–0.7 (avg = 0.6) | 1103–1141 (avg = 1125) |
| Alkali basalt n = 11 | 1156–1198 (avg = 1182) | 0.3–0.8 (avg = 0.6) | 0.2–0.8 (avg = 0.56) | 1115–1185 (avg = 1155) |
| Basanite | N/A | N/A | N/A | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Houshmand-Manavi, S.; Rezaei-Kahkhaei, M.; Klötzli, U.; Ghasemi, H. Crystallization Conditions of Basaltic Lavas Based on Clinopyroxene and Olivine Phenocryst Petrology: A Case Study from the Neogene Lavarab Alkaline Basaltic Lavas (LABL), Eastern Iran. Minerals 2025, 15, 915. https://doi.org/10.3390/min15090915
Houshmand-Manavi S, Rezaei-Kahkhaei M, Klötzli U, Ghasemi H. Crystallization Conditions of Basaltic Lavas Based on Clinopyroxene and Olivine Phenocryst Petrology: A Case Study from the Neogene Lavarab Alkaline Basaltic Lavas (LABL), Eastern Iran. Minerals. 2025; 15(9):915. https://doi.org/10.3390/min15090915
Chicago/Turabian StyleHoushmand-Manavi, Sara, Mehdi Rezaei-Kahkhaei, Urs Klötzli, and Habibollah Ghasemi. 2025. "Crystallization Conditions of Basaltic Lavas Based on Clinopyroxene and Olivine Phenocryst Petrology: A Case Study from the Neogene Lavarab Alkaline Basaltic Lavas (LABL), Eastern Iran" Minerals 15, no. 9: 915. https://doi.org/10.3390/min15090915
APA StyleHoushmand-Manavi, S., Rezaei-Kahkhaei, M., Klötzli, U., & Ghasemi, H. (2025). Crystallization Conditions of Basaltic Lavas Based on Clinopyroxene and Olivine Phenocryst Petrology: A Case Study from the Neogene Lavarab Alkaline Basaltic Lavas (LABL), Eastern Iran. Minerals, 15(9), 915. https://doi.org/10.3390/min15090915










