Effects of Microwave Energy and MnO2 from Deep-Sea Polymetallic Nodules as an Oxidizing Agent on the Leaching of Chalcopyrite Concentrate
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Samples and Reagents
2.2. Analytical and Experimental Methods
2.3. Procedure of Leaching Experiments
3. Results and Discussion
3.1. Deep-Sea Polymetallic Nodules and Chalcopyrite Concentrate Characterization
3.1.1. Deep-Sea Polymetallic Nodules Analysis
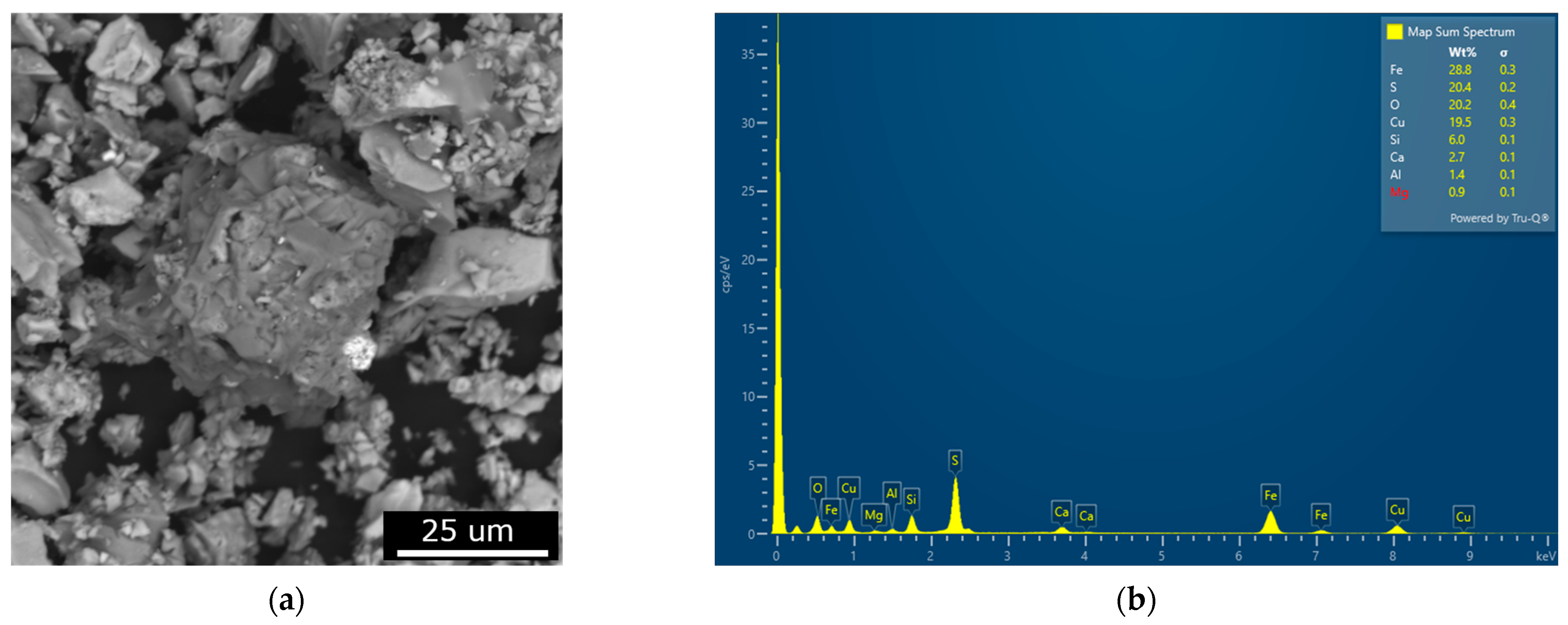
3.1.2. Chalcopyrite Concentrate Characterization
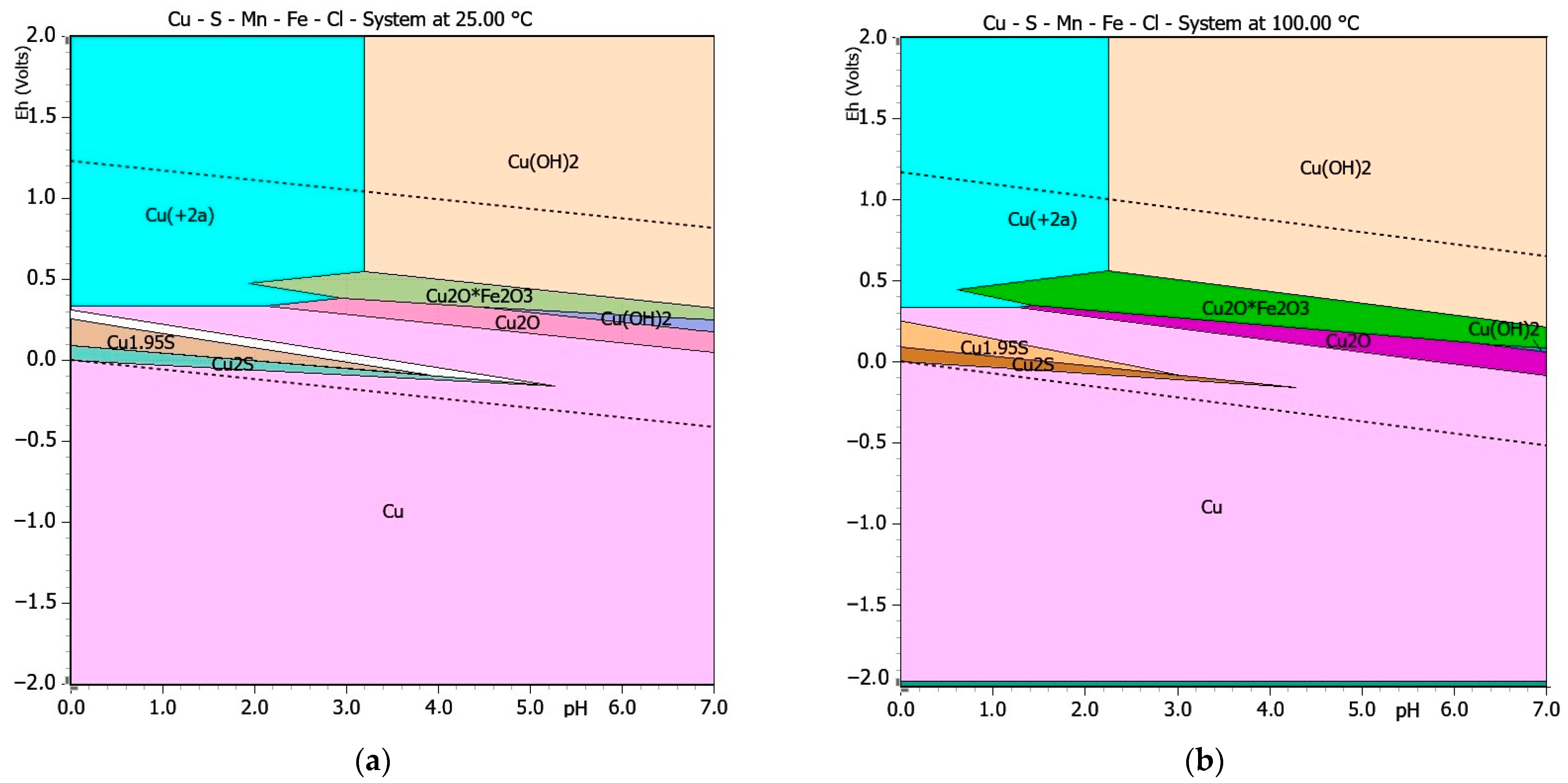
3.2. Thermodynamics Study of the Chalcopyrite Concentrates Leaching
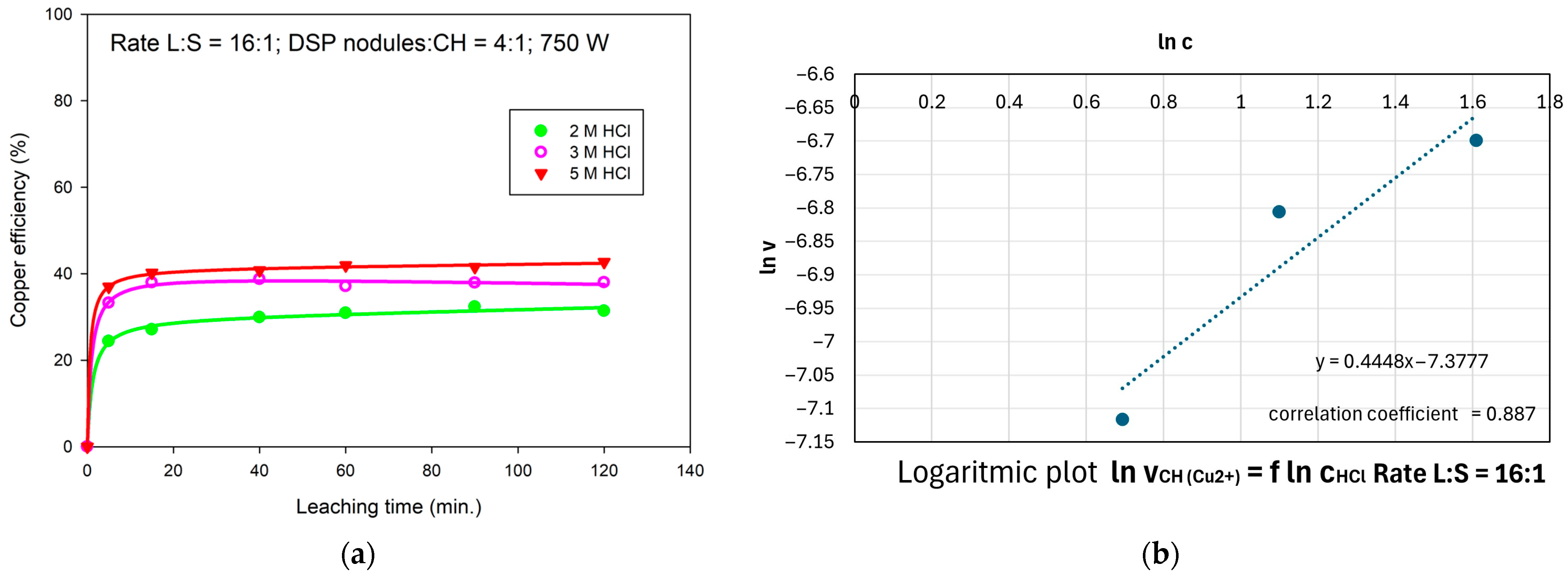
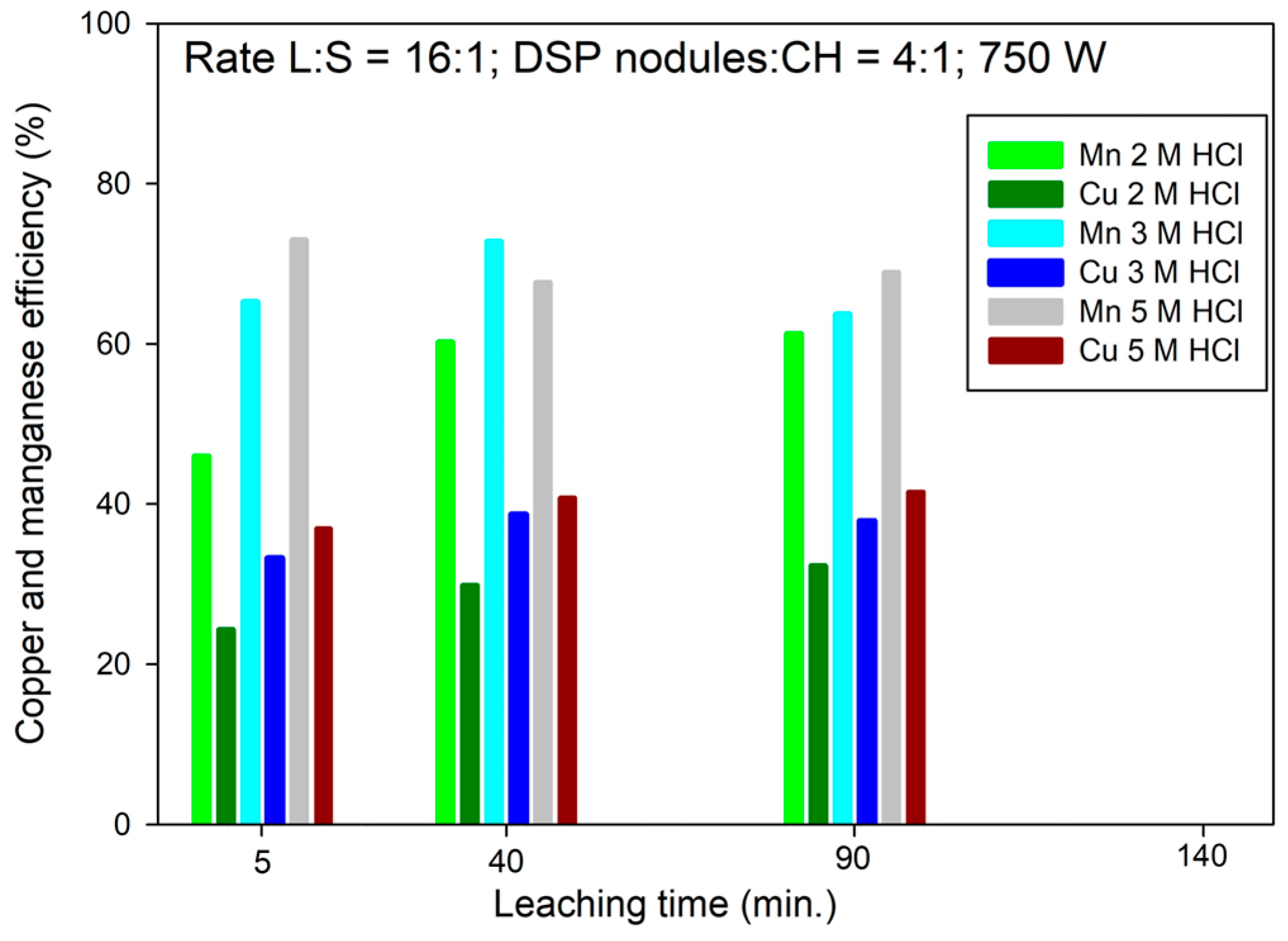
3.3. Effect of HCl Concentration on Cu Extraction Efficiency
- v—rate of reaction (4)
- k—rate constant
- c—the concentration of hydrochloric acid
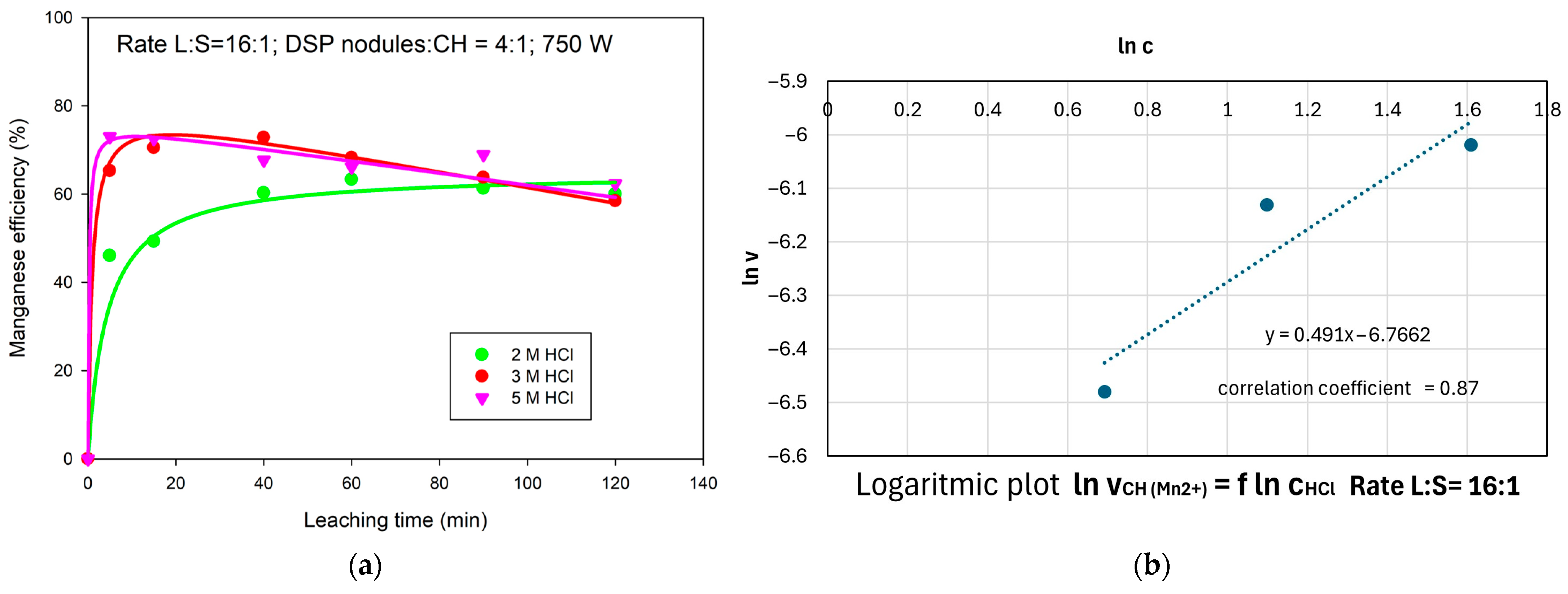
3.4. Effect of the HCl Concentration on Mn Extraction Efficiency
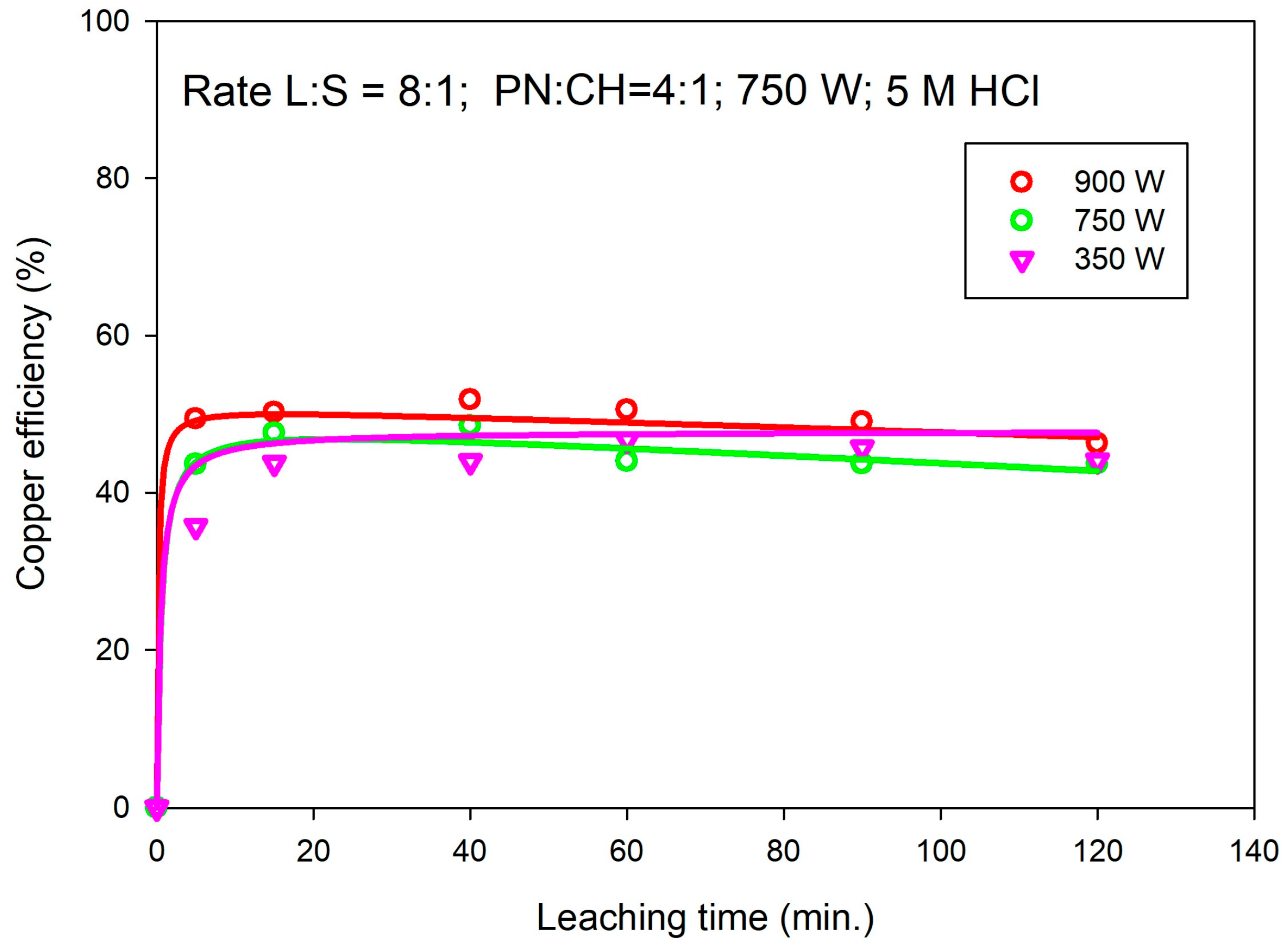
3.5. Effect of Microwave Energy Intensity on the Leaching Efficiency of Cu
4. Conclusions
- -
- Under optimized conditions (HCl concentration of 5 mol/L, L/S ratio of 16:1, and microwave power of 750 W), copper recovery reached a maximum of 40% within 40 min, while manganese recovery from DSP nodules increased by up to 30% compared to 2 mol/L HCl, confirming the synergistic oxidative role of MnO2.
- -
- Kinetic analysis revealed that the apparent reaction order with respect to HCl concentration was n = 0.4448 for Cu and n = 0.491 for Mn, indicating a decreasing sensitivity of the reaction rate to acid concentration at higher molarities and underscoring the growing importance of the oxidant (MnO2) in the leaching process.
- -
- The key scientific contribution of this work is the innovative use of deep-sea polymetallic nodules (DSP) as a leaching agent with a dual function, serving simultaneously as an oxidizing agent and as an additional source of valuable metals (Mn: 21.74 wt.%, Cu: 1.004 wt.%, Ni: 1.15 wt.%).
- -
- The use of microwave radiation at a power of 750 W proved effective in disrupting the formation of a passivating sulfur layer, which commonly occurs during conventional leaching processes. This facilitated a more effective interaction between the oxidizing agent and chalcopyrite, promoting copper extraction.
- -
- Further optimization of the leaching system could be achieved by reducing the particle size of the DSP nodules below 266 µm. Finer particles are expected to exhibit improved surface contact with chalcopyrite, enhancing Cl2 generation and thus intensifying the oxidative leaching process. Additionally, smaller DSP particles may absorb microwave energy more efficiently, leading to localized temperature increases that could accelerate reaction kinetics.
- -
- The practical significance lies in the fact that DSP nodules represent a sustainable source of oxidizing agents and metals, support the circular economy, and, under optimal leaching conditions, provide an efficient and easily scalable process for processing low-grade copper ores, which can be further improved by reducing particle size.
- -
- Future experiments should explore not only reducing the particle size of the feedstock and determining its limits, but also increasing the amount of DSP nodules in the batch. In addition to serving as a source of MnO2, these DSP nodules contain appreciable quantities of copper, offering a dual benefit in terms of oxidizing capability and metal recovery potential.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dutrizac, J.E. Elemental sulphur formation during the ferric chloride leaching of chalcopyrite. Hydrometallurgy 1990, 23, 153–176. [Google Scholar] [CrossRef]
- Havlik, T. Leaching of Copper Sulphides. In Hydrometallurgy: Principles and Application, 1st ed.; Woodhead Publishing Series in Metals and Surface Engineering; Woodhead Publishing: Sawston, UK, 2008; Chapter 12; ISBN 978-1-84569-407-4. [Google Scholar]
- Sokić, M.; Marković, B.; Stanković, S.; Kamberović, Ž.; Štrbac, N.; Manojlović, V.; Petronijević, N. Kinetics of chalcopyrite leaching by hydrogen peroxide in sulfuric acid. Metals 2019, 9, 1173. [Google Scholar] [CrossRef]
- Petrović, S.J.; Bogdanović, G.D.; Antonijević, M.M.; Vukčević, M.; Kovačević, R. The Extraction of Copper from Chalcopyrite Concentrate with Hydrogen Peroxide in Sulfuric Acid Solution. Metals 2023, 13, 1818. [Google Scholar] [CrossRef]
- Gok, O.; Anderson, C.G.; Cicekli, G.; Ilknur Cocen, E. Leaching kinetics of copper from chalcopyrite concentrate in nitrous-sulfuric acid. Physicochem. Probl. Miner. Process. 2014, 50, 399–413. [Google Scholar] [CrossRef]
- Tešinský, M.; Baláž, P. Copper leaching from chalcopyrite: Mechanochemical approach. Inz. Miner. 2017, 39, 1–6. Available online: http://www.potopk.com.pl/Full_text/2017_full/IM-1-2017-a1.pdf (accessed on 1 January 2017).
- Dakkoune, A.; Bourgeois, F.; Po, A.; Joulian, C.; Hubau, A.; Touzé, S.; Julcour, C.; Guezennec, A.-G.; Cassayre, L. Hydrometallurgical Processing of Chalcopyrite by Attrition-Aided Leaching. ACS Eng. Au 2023, 3, 195–209. [Google Scholar] [CrossRef]
- Wei, Z.; Yang, X.; Li, W.; Ma, Q.; Wu, X.; Li, Y. An Improved Understanding of Chalcopyrite Leaching Mechanisms: The Influence of Anisotropic Crystal Planes. Minerals 2023, 13, 1461. [Google Scholar] [CrossRef]
- Karimi, S.; Mohammadpour, P.; Esmailzadeh, M.; Izadi, M. Sustainable synthesis and application of green deep eutectic solvent in chalcopyrite leaching: A combined experimental and molecular dynamic simulation approach. Nano-Struct. Nano-Objects 2025, 42, 101481. [Google Scholar] [CrossRef]
- Behnajady, B.; Najafi, M.; Karimi, S. A new approach to direct chemical leaching of Sungun chalcopyrite concentrate via green deep eutectic solvent choline chloride-ρ-toluenesulfonic acid and MD simulation. J. Taiwan Inst. Chem. Eng. 2025, 172, 106118. [Google Scholar] [CrossRef]
- Ren, M.; Zhang, J.; Xu, Z.; Wang, J.; Qiu, J.; Xie, Y. Accelerated metal separation from chalcopyrite assisted with ozone. Chem. Eng. Process.-Process Intensif. 2025, 212, 110263. [Google Scholar] [CrossRef]
- Wen, T.; Zhao, Y.; Xiao, Q.; Ma, Q.; Kang, S.; Li, H.; Song, S. Effect of microwave-assisted heating on chalcopyrite leaching of kinetics, interface temperature and surface energy. Results Phys. 2017, 7, 2594–2600. [Google Scholar] [CrossRef]
- Havlik, T.; Popovičová, M.; Ukašík, M. Use of microwave energy for chalcopyrite leaching. Metall 2001, 56, 131–134. [Google Scholar]
- Ortega, F.; Petersen, J.; Velásquez-Yévenes, L. A preliminary study of electro-reduction pretreatment preceding chalcopyrite leaching in ammonium chloride media in the presence of copper ions. Hydrometallurgy 2025, 236, 106508. [Google Scholar] [CrossRef]
- Zhong, S.; Li, Y. An improved understanding of chalcopyrite leaching kinetics and mechanisms in the presence of NaCl. J. Mater. Res. Technol. 2019, 8, 3487–3494. [Google Scholar] [CrossRef]
- Toro, N.; Pérez, K.; Saldaña, M.; Jeldres, R.I.; Jeldres, M.; Cánovas, M. Dissolution of pure chalcopyrite with manganese nodules and waste water. J. Mater. Res. Technol. 2020, 9, 798–805. [Google Scholar] [CrossRef]
- Sea-Floor Mining. Available online: https://worldoceanreview.com/wp-content/downloads/wor3/WOR3_en_chapter_2.pdf (accessed on 20 June 2025).
- Wang, X.; Qin, W.; Li, M.; Liu, X.; Cheng, Y.; Chen, S.; Yang, C. Mineralogy of Deep-Sea Manganese Nodules and Advances in Extraction Technology of Valuable Elements from Manganese Nodules. Metals 2024, 14, 1359. [Google Scholar] [CrossRef]
- Li, J.; Hu, Y.; Yue, L.; Zeng, L.; Wu, S.; Wang, M.; Guan, W.; Cao, Z.; Li, Q.; Zhang, G. Recovery of critical battery metals from cobalt-rich deep-sea polymetallic nodules: Selective carbothermal reduction based on kinetics. J. Environ. Chem. Eng. 2024, 12, 111631. [Google Scholar] [CrossRef]
- Blistan, P.; Kršák, B.; Blistanová, M.; Ferencz, V. The seabed-an important mineral resource of Slovakia in the future. Acta Montan. Slovaca 2015, 20, 334–341. [Google Scholar] [CrossRef]
- The Interoceanmetal Joint Organization (IOM). Available online: https://iom.gov.pl/ (accessed on 20 June 2025).
- Senanayake, G. Acid leaching of metals from deep-sea manganese nodules—A critical review of fundamentals and applications. Miner. Eng. 2011, 24, 1379–1396. [Google Scholar] [CrossRef]
- Toro, N.; Rodríguez, F.; Rojas, A.; Robles, P.; Ghorbani, Y. Leaching manganese nodules with iron-reducing agents—A critical review. Miner. Eng. 2021, 163, 106748. [Google Scholar] [CrossRef]
- Abramovski, T.; Stefanova, V.P.; Causse, R.; Romanchuk, A. Technologies for the processing of polymetallic nodules from Clarion Clipperton Zone in the Pacific Ocean. J. Chem. Technol. Metall. 2017, 52, 258–269. [Google Scholar]
- Havlik, T.; Laubertova, M.; Miskufova, A.; Kondas, J.; Vranka, F. Extraction of copper, zinc, nickel and cobalt in acid oxidative leaching of chalcopyrite at the presence of deep-sea manganese nodules as oxidant. Hydrometallurgy 2005, 77, 51–59. [Google Scholar] [CrossRef]
- Devi, N.B.; Madhuchhanda, M.; Rath, P.C.; Rao, K.S.; Paramguru, R.K. Simultaneous leaching of a deep-sea manganese nodule and chalcopyrite in hydrochloric acid. Metall. Mater. Trans. B Process Metall. Mater. Process. Sci. 2001, 32, 777–784. [Google Scholar] [CrossRef]
- Torres, D.; Ayala, L.; Jeldres, R.I.; Cerecedo-sáenz, E.; Salinas-rodríguez, E.; Robles, P.; Toro, N. Leaching chalcopyrite with high MnO2 and chloride concentrations. Metals 2020, 10, 107. [Google Scholar] [CrossRef]
- Xiao, L.; Liu, J.S.; Fang, Z.; Qiu, G.-Z. Mechanism of electro-generating leaching of chalcopyrite-MnO2 in presence of Acidithiobacillus thiooxidans. Trans. Nonferrous Met. Soc. China (Eng. Ed.) 2008, 18, 1458–1462. [Google Scholar] [CrossRef]
- Souza, R.; Queiroz, C.; Brant, J.; Brocchi, E. Pyrometallurgical processing of a low copper content concentrate based on a thermodynamic assessment. Miner. Eng. 2019, 130, 156–164. [Google Scholar] [CrossRef]
- Sitlhou, L.; Chakraborty, P. Comparing deep-sea polymetallic nodule mining technologies and evaluating their probable impacts on deep-sea pollution. Mar. Pollut. Bull. 2024, 206, 116762. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, K.; Du, Y.; Liu, X.; Shen, Y. Status of Sustainability Development of Deep-Sea Mining Activities. J. Mar. Sci. Eng. 2022, 10, 1508. [Google Scholar] [CrossRef]
- Szabó, L. Deep-Sea Mining and the Sustainability Paradox: Pathways to Balance Critical Material Demands and Ocean Conservation. Sustainability 2025, 17, 6580. [Google Scholar] [CrossRef]
- Onol, K.; Saridede, M.N. Investigation on microwave heating for direct leaching of chalcopyrite ores and concentrates. Int. J. Miner. Metall. Mater. 2013, 20, 228–233. [Google Scholar] [CrossRef]
- Laubertova, M.; Havlik, T.; Parilak, L.; Derin, B.; Trpcevska, J. The effects of microwave-assisted leaching on the treatment of electric arc furnace dusts (EAFD). Arch. Metall. Mater. 2020, 65, 321–328. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.; Kingman, S.; Bradshaw, S. Scale up possibilities for microwave leaching of chalcopyrite in ferric sulphate. Int. J. Miner. Process. 2006, 80, 198–204. [Google Scholar] [CrossRef]
- Znamenáčková, I.; Dolinská, S.; Kováčová, M.; Lovás, M.; Čablík, V.; Čablíková, L. Innovative Method of Material Treatmentby Microwave Energy. Procedia Earth Planet. Sci. 2015, 15, 855–860. [Google Scholar] [CrossRef][Green Version]
- Štyriaková, D.; Štyriaková, I.; Šuba, J.; Baláž, P.; Abramowski, T. Bioleaching Test of Polymetallic Nodule Samples from the IOM Exploration Area. Minerals 2022, 12, 1373. [Google Scholar] [CrossRef]
- Xu, J.; Wang, H.; Gao, C.; Zhang, L.; Yang, H.; Sai, M.; Hu, J.; Huang, Q.; Luo, H. Effect of Different Crushing Methods on Chalcopyrite Liberation and Heavy Media Preconcentration. Minerals 2025, 15, 179. [Google Scholar] [CrossRef]
- Long, F.; Chen, W.; Zhang, Z.; Chi, R. Effects of Ore Particle Size Distribution on Rare Earth Leaching Process of Weathered Crust Elution-Deposited Rare Earth Ores. Minerals 2025, 15, 149. [Google Scholar] [CrossRef]
- Tombal, T.D.; Kursun, I.; Terzi, M.; Gungoren, C. Optimization and Kinetics of Chalcopyrite Leaching in the Presence of Hydrochloric and Nitric Acids, along with a Particle Shape-Based Evaluation. JOM 2025, 77, 3589–3599. [Google Scholar] [CrossRef]
- Hikov, A.; Milakovska, Z.; Peytcheva, I.; Stoyanova, V.; Stefanova, E.; Abramowski, T.; Kadiyski, M.; Chavdarova, S.; Stavrev, M.; Dimitrova, D. Geochemistry of REE and Other Critical Elements in Deep-Sea Polymetallic Nodules from Interoceanmetal (IOM) Exploration Area in Eastern Part of Clarion–Clipperton Fracture Zone, NE Pacific. Minerals 2025, 15, 154. [Google Scholar] [CrossRef]
- Roine, A. Outokumpu HSC Chemistry for Windows: Chemical Reaction and Equilibrium Software with Extensive Thermo-chemical Database. In User’s Guide Outokumpu HSC Chemistry® Windows; Outokumpu Research Oy: Helsinki, Finland, 2002. [Google Scholar]
- Sminčáková, E.; Trpčevská, J.; Pirošková, J. Kinetic Aspects of Leaching Zinc from Waste Galvanizing Zinc by Using Hydrochloric Acid Solutions. JOM 2017, 69, 1869–1875. [Google Scholar] [CrossRef]
- Znamenackova, I.; Lovas, M.; Hredzak, S.; Dolinska, S. An application of microwave heating in treatment of selected minerals. In Proceedings of the International Multidisciplinary Scientific GeoConference Surveying Geology and Mining Ecology Management, SGEM, Albena, Bulgaria, 17–26 June 2014; Volume 3, pp. 965–972. Available online: https://www.proceedings.com/content/026/026859webtoc.pdf (accessed on 5 June 2025).
- Znamenácková, I.; Dolinská, S.; Hredzák, S.; Cablík, V. Effect of Microwave Pretreatment on Leaching of Tetrahedrite. Proc. IOP Conf. Ser. Earth Environ. Sci. 2021, 906, 012111. [Google Scholar] [CrossRef]
- Castellón, C.I.; Taboada, M.E. Leaching of Copper Concentrate with Iodized Salts in a Saline Acid Medium: Part 1—Effect of Concentrations. Materials 2023, 16, 2312. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Chao, C.; Krishnamoorthy, P.; Asselin, E.; Dixon, D.G.; Mora, N. The overlooked mechanism of chalcopyrite passivation. Acta Mater. 2022, 236, 118111. [Google Scholar] [CrossRef]
- Wen, T.; Zhao, Y.; Ma, Q.; Xiao, Q.; Zhang, T.; Chen, J.; Song, S. Microwave improving copper extraction from chalcopyrite through modifying the surface structure. J. Mater. Res. Technol. 2020, 9, 263–270. [Google Scholar] [CrossRef]


| Content (wt.%) | Mn | Fe | Cu | Zn | Ni | Co | Mo | Mg | Residue |
|---|---|---|---|---|---|---|---|---|---|
| Average | 21.74 | 3.282 | 1.004 | 0.14 | 1.15 | 0.156 | 0.242 | 1.336 | 70.95 |
| Standard Deviation | 1.041 | 0.153 | 0.015 | 0 | 0.026 | 0.008 | 0.004 | 0.035 | - |
| Variance | 1.085 | 0.234 | 0.22·10−3 | 0 | 0.68·10−3 | 0.64·10−4 | 0.16·10−5 | 0.12·10−2 | - |
| Content (wt.%) | Fe | Cu | Zn | Pb | As | Ba | Ag | Residue |
|---|---|---|---|---|---|---|---|---|
| Average | 29.7 | 29.1 | 2.4 | 0.43 | 0.03 | 0.002 | 0.02 | 38.18 |
| Standard Deviation | 0.1 | 0.15 | 0.88 | 0.01 | 0.01 | 0 | 0.02 | - |
| Variance | 6.6·10−3 | 1.55·10−2 | 0.1302 | 2.22·10−5 | 6.6·10−5 | 0 | 2.2·10−5 | - |
| Predicted Chemical Reaction | ΔGT [kJ/mol] | Equation | |
|---|---|---|---|
| 25 °C | 100 °C | ||
| −201.778 | −220.863 | (1) | |
| −305.401 | −282.813 | (2) | |
| −396.733 | −340.173 | (3) | |
| + | −901.179 | −892.330 | (4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laubertová, M.; Velgosová, O.; Pirošková, J.; Briančin, J. Effects of Microwave Energy and MnO2 from Deep-Sea Polymetallic Nodules as an Oxidizing Agent on the Leaching of Chalcopyrite Concentrate. Minerals 2025, 15, 914. https://doi.org/10.3390/min15090914
Laubertová M, Velgosová O, Pirošková J, Briančin J. Effects of Microwave Energy and MnO2 from Deep-Sea Polymetallic Nodules as an Oxidizing Agent on the Leaching of Chalcopyrite Concentrate. Minerals. 2025; 15(9):914. https://doi.org/10.3390/min15090914
Chicago/Turabian StyleLaubertová, Martina, Oksana Velgosová, Jana Pirošková, and Jaroslav Briančin. 2025. "Effects of Microwave Energy and MnO2 from Deep-Sea Polymetallic Nodules as an Oxidizing Agent on the Leaching of Chalcopyrite Concentrate" Minerals 15, no. 9: 914. https://doi.org/10.3390/min15090914
APA StyleLaubertová, M., Velgosová, O., Pirošková, J., & Briančin, J. (2025). Effects of Microwave Energy and MnO2 from Deep-Sea Polymetallic Nodules as an Oxidizing Agent on the Leaching of Chalcopyrite Concentrate. Minerals, 15(9), 914. https://doi.org/10.3390/min15090914











