Effects of Fluid Inclusion Component Release on Flotation Behavior of Fluorite Minerals
Abstract
1. Introduction
2. Materials and Methods
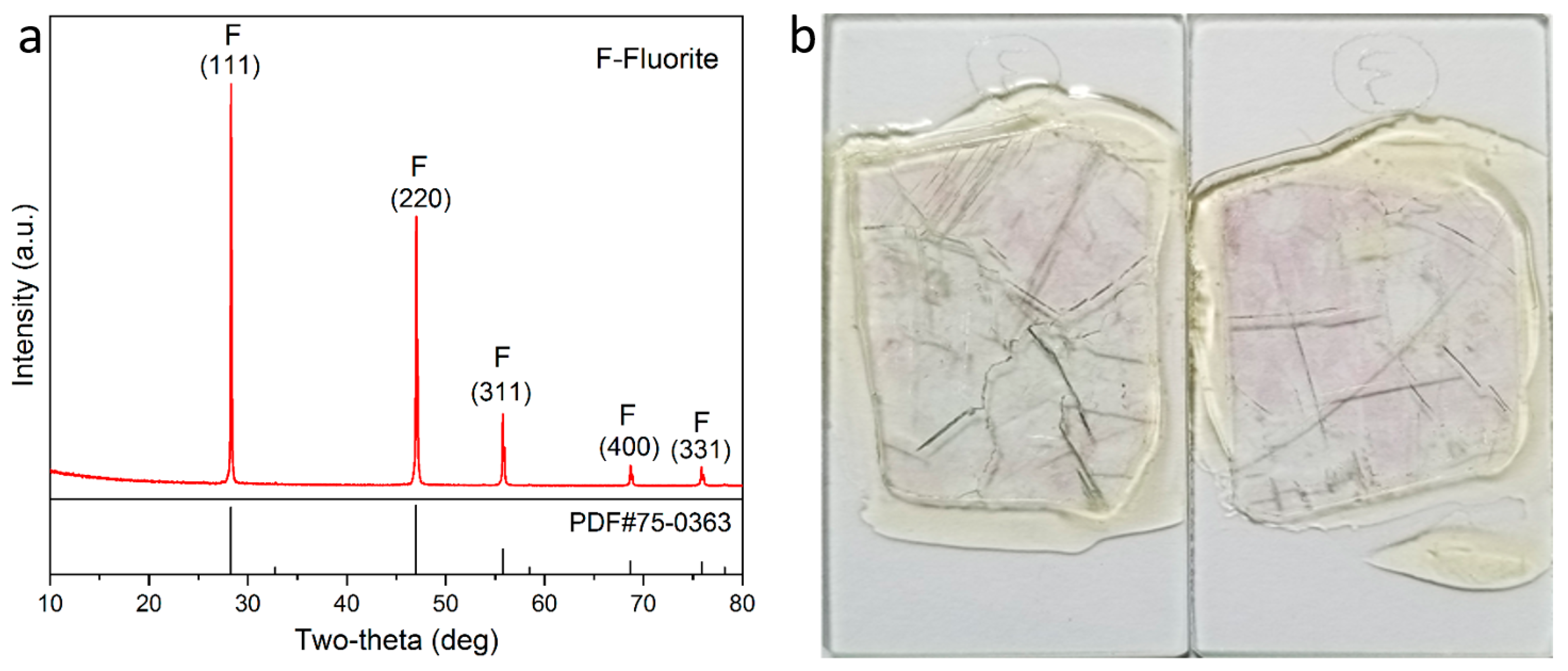
2.1. Materials
2.2. Petrographic Study of Fluid Inclusions
2.3. X-Ray Computed Tomography Test
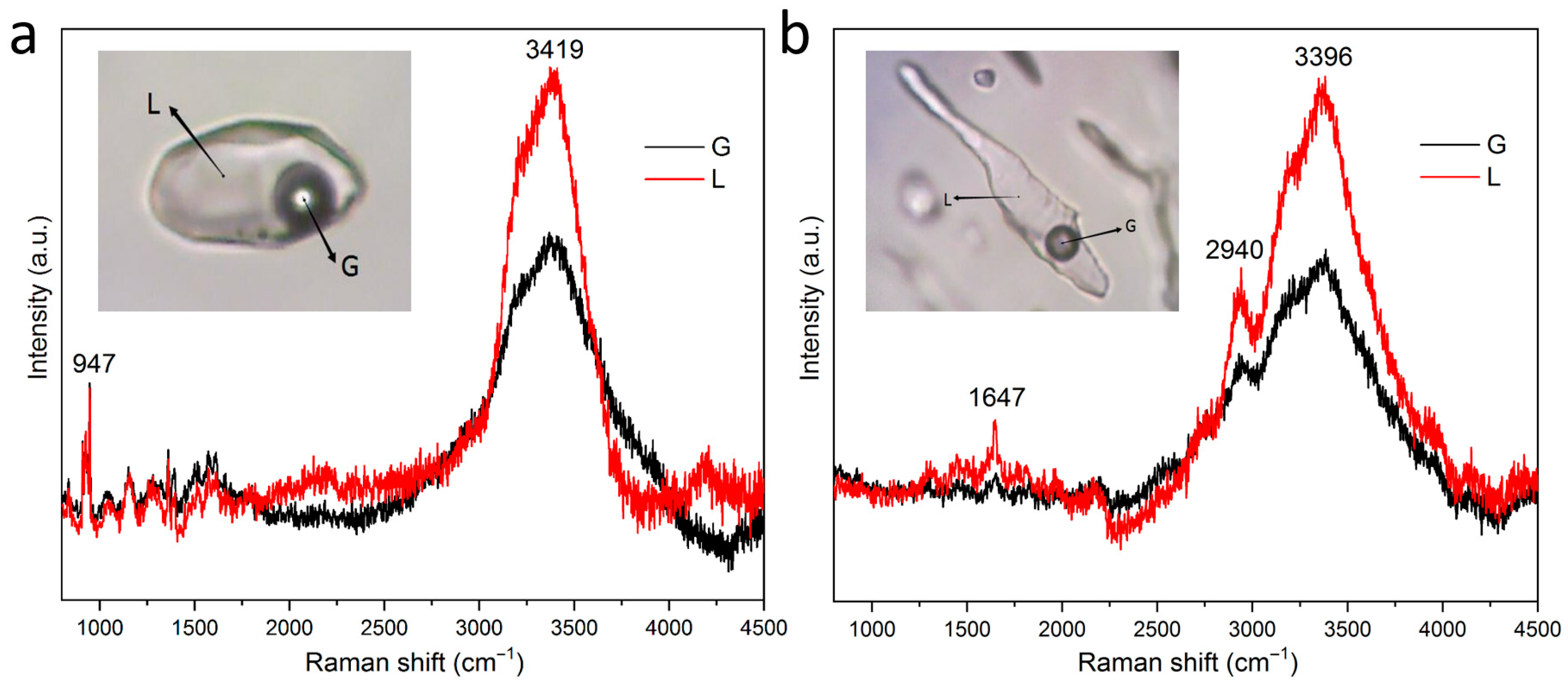
2.4. Laser Raman Spectroscopy Testing
2.5. Ion Component Test
2.6. Flotation Test
3. Results and Discussion
3.1. Morphological Characteristics of Fluorite Fluid Inclusions
3.2. Petrographic Characteristics of Fluorite Fluid Inclusions
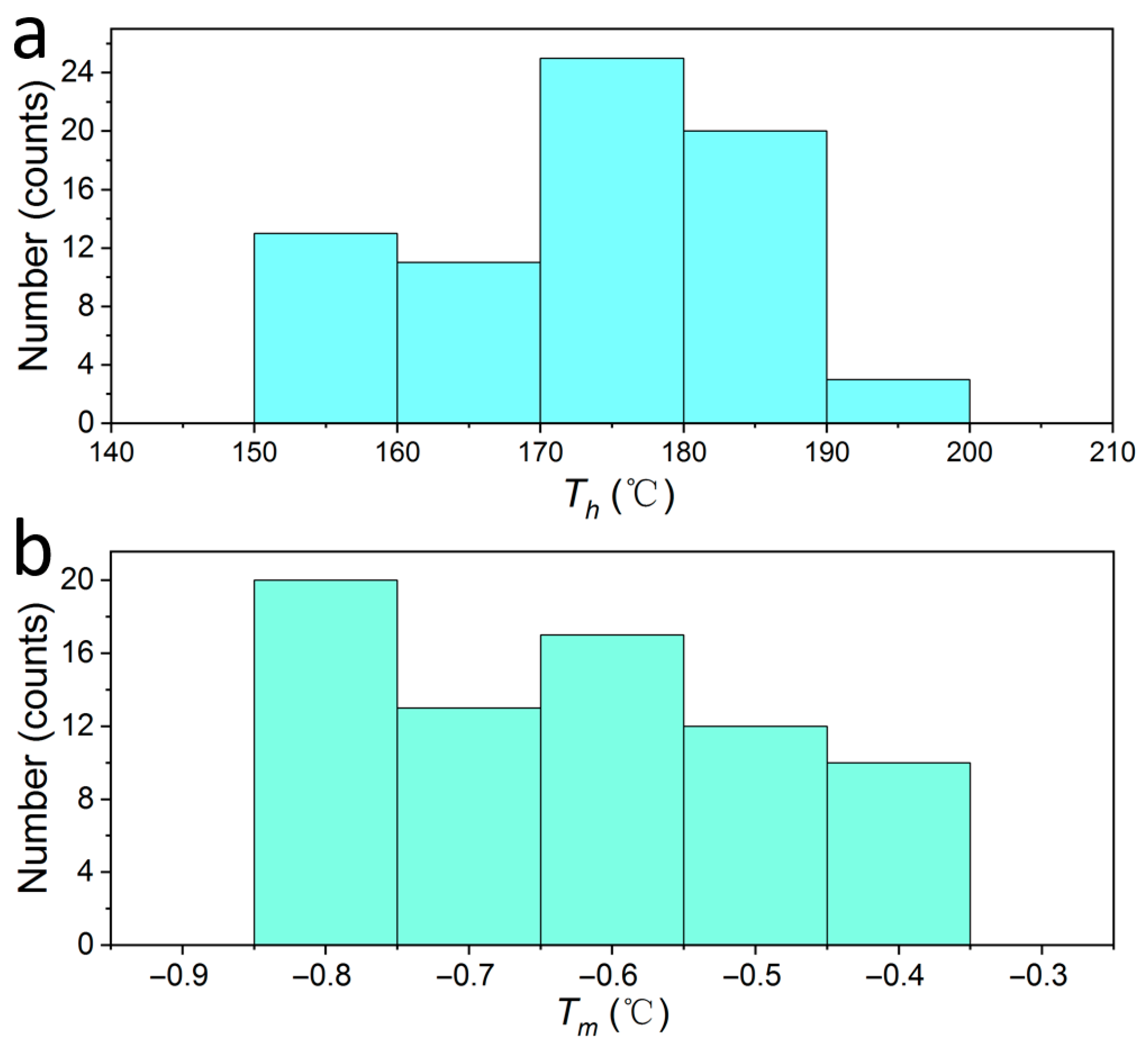
3.2.1. Homogenization Temperature and Freezing Point Temperature
3.2.2. Fluid Inclusion Salinity and Density Analysis
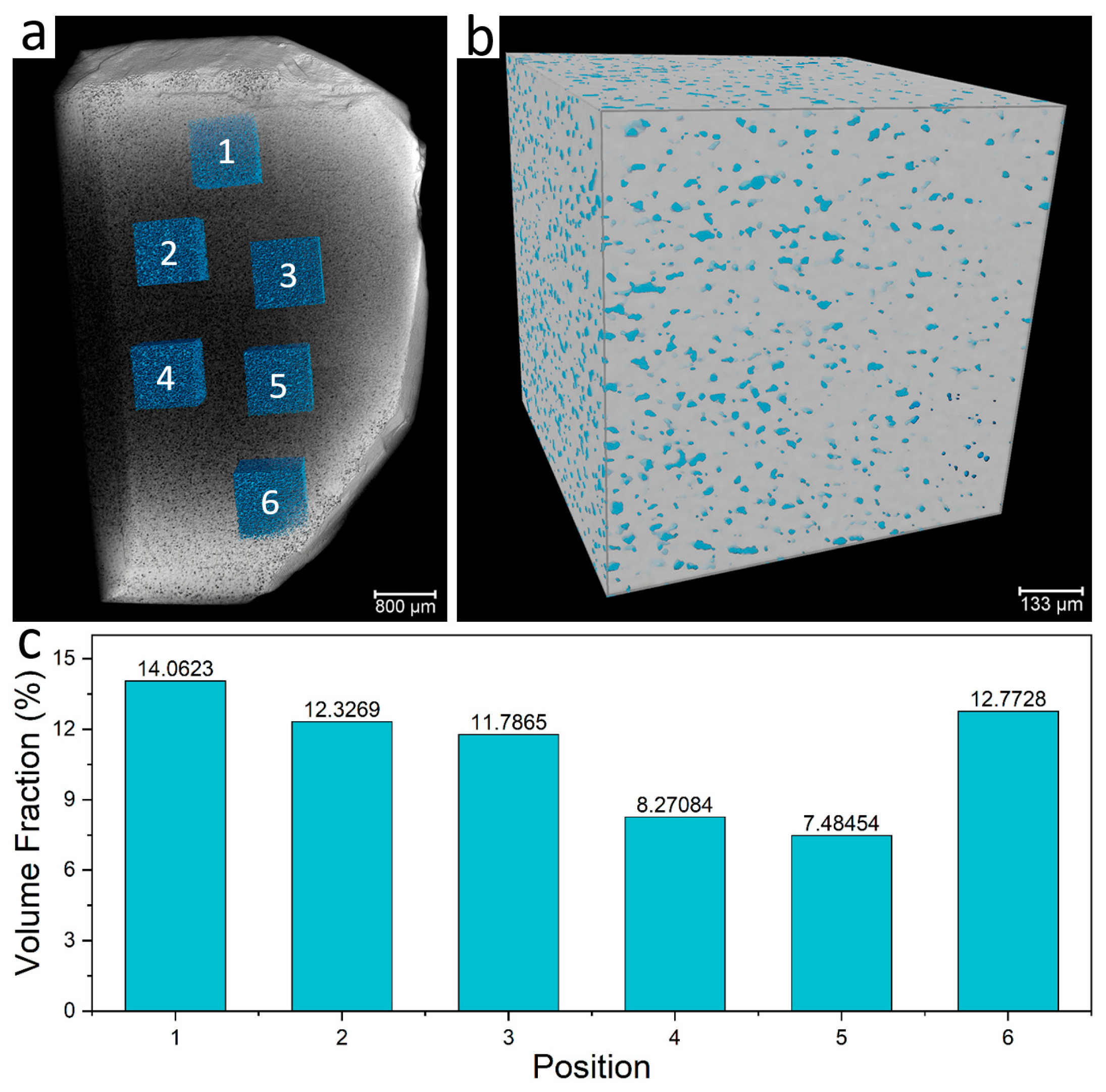
3.2.3. Spatial Distribution Characteristics of Fluid Inclusions
3.2.4. Chemical Composition Analysis of Fluid Inclusions
3.3. Release Behavior of Fluid Inclusion Compositions in Fluorite
3.4. Influence Mechanism of Fluid Inclusion Component Release on Fluorite Flotation
4. Conclusions
- Optical microscopy analysis identifies abundant and diverse fluid inclusions in fluorite, with gas–liquid two-phase inclusions being the dominant type, while single-liquid-phase inclusions are less common and three-phase inclusions containing daughter minerals are extremely rare. These inclusions exhibit variable morphologies and size distributions, and sizes vary from 4.5 to 20.4 μm.
- Microthermometric measurements show that the homogenization temperature of fluorite-hosted ore-forming fluids ranges from 155 to 195 °C, with salinities between 0.701% and 1.389 wt.% NaCl equivalent. Fluid density values fall within 0.878–0.927 g/cm3, and the spatial distribution density of inclusions reaches up to 14.1%. Laser Raman spectroscopy confirms that H2O constitutes the primary component of fluorite fluid inclusions, which belong to a low-temperature, low-salinity, low-density NaCl-H2O system.
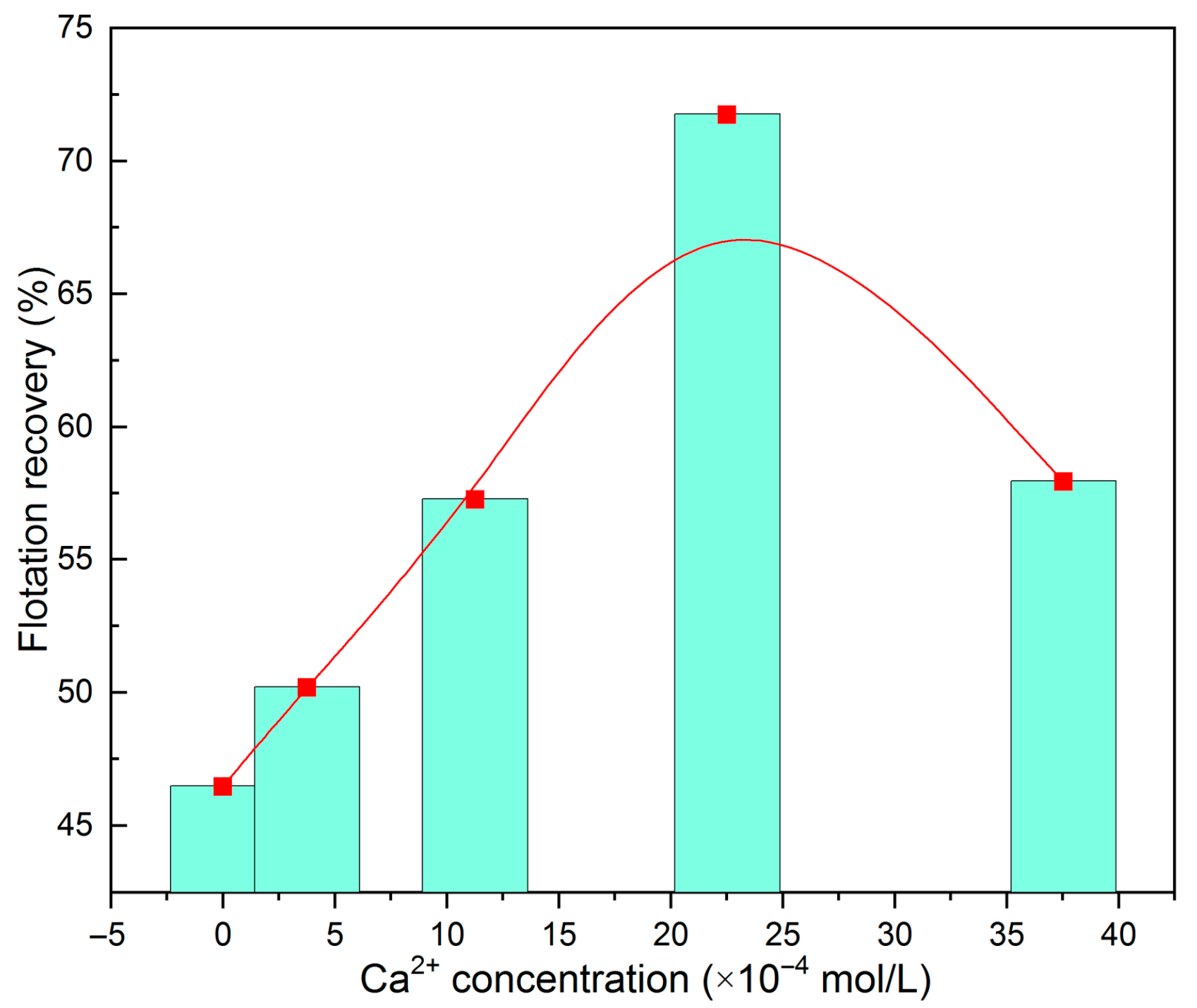
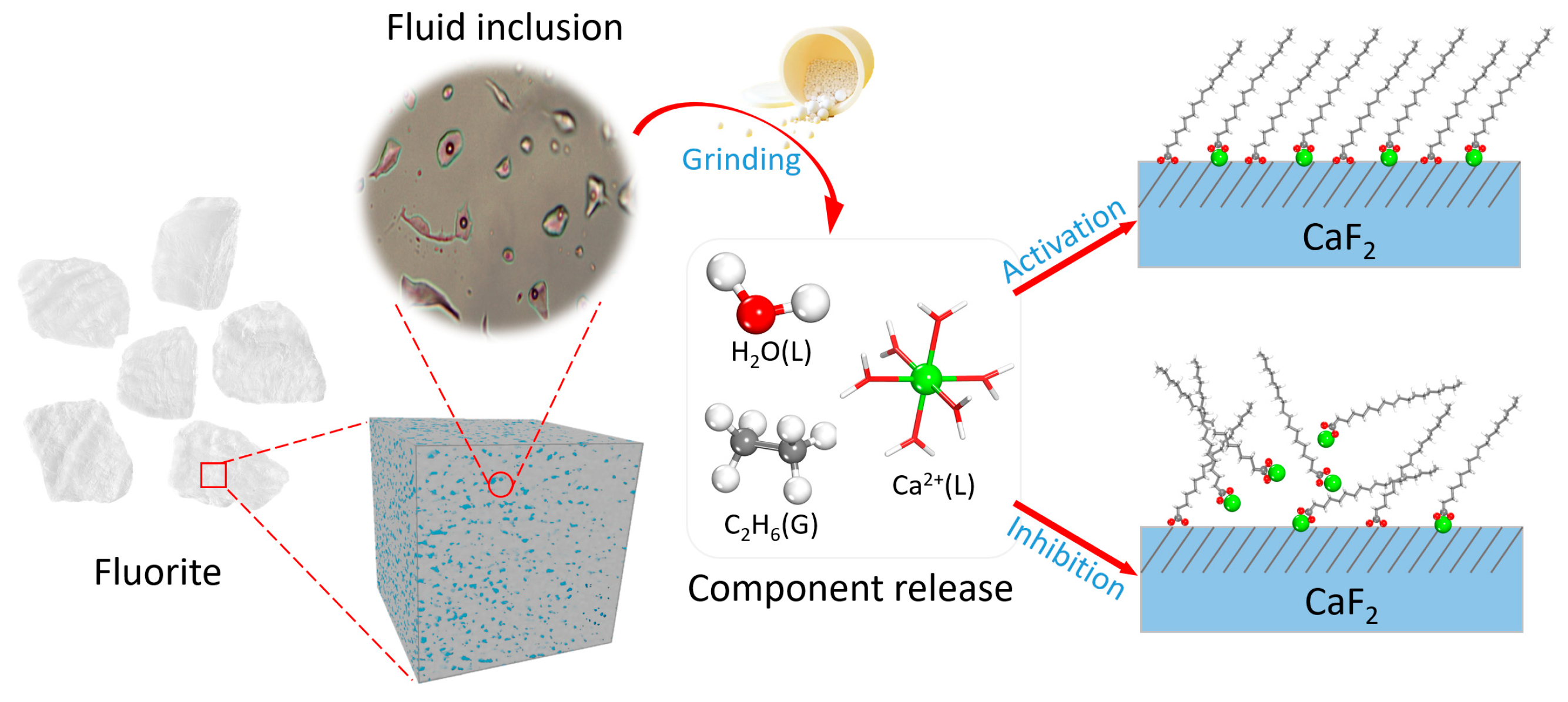
- Fluorite fluid inclusions contain significant ionic components such as Ca2+, F−, Cl−, and SO42−. Upon release into pulp solution, these ions act as unavoidable species that influence fluorite flotation behavior—most notably through the introduction of Ca2+. Low concentrations of Ca2+ activate fluorite flotation, whereas excessive amounts preferentially consume flotation reagents, thereby inhibiting the flotation process. This verifies that the release of components from fluorite fluid inclusions constitutes a non-negligible source of unavoidable ions in flotation pulp systems.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ministry of Land and Resources. National Plan for Mineral Resources (2016–2020) [EB/OL]; Ministry of Natural Resources of the People’s Republic of China: Beijing, China, 2016.
- Akgün, A.; Teğin, İ.; Ziyadanoğulları, R. Enrichment of molybdenum and fluorite by flotation of fluorite ore containing molybdenum. J. Miner. Mater. Charact. Eng. 2006, 05, 103–107. [Google Scholar] [CrossRef]
- Chandra, A.P.; Gerson, A.R. A review of the fundamental studies of the copper activation mechanisms for selective flotation of the sulfide minerals, sphalerite and pyrite. Adv. Colloid Interface Sci. 2009, 145, 97–110. [Google Scholar] [CrossRef]
- Zheng, R.; Ren, Z.; Gao, H.; Chen, Z.; Qian, Y.; Li, Y. Effects of crystal chemistry on sodium oleate adsorption on fluorite surface investigated by molecular dynamics simulation. Miner. Eng. 2018, 124, 77–85. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, Q.; Jiang, T.; Chen, M.; Wang, C.; Wang, S.; Li, H.; Chen, F. Enhancing the flotation separation of fluorite and calcite: Insights into the mechanism of differential surface regulation by various metallic ions. Appl. Surf. Sci. 2025, 696, 162964. [Google Scholar] [CrossRef]
- Zheng, Q.; Dong, L.; Shen, P.; Liu, D. A novel activation scheme for fine cassiterite flotation by using a metal ion modified collector. Colloids Surf. A Physicochem. Eng. Asp. 2025, 710, 136310. [Google Scholar] [CrossRef]
- Zhang, B.; Sun, N.; Sun, W.; Jiang, F.; Wang, L. Enhanced spodumene flotation through Zn(Ⅱ) ions activation using sodium oleate as collector. Miner. Eng. 2025, 230, 109414. [Google Scholar] [CrossRef]
- Yang, X.; Li, Y.; Chen, J.; Xie, S.; Chen, P.; Song, S. Enhanced flotation separation of fluorite and F−-activated calcite by sodium gluconate and sodium silicate. Miner. Eng. 2025, 229, 109375. [Google Scholar] [CrossRef]
- Rao, S.R.; Finch, J.A. A review of water re-use in flotation. Miner. Eng. 1989, 2, 65–85. [Google Scholar] [CrossRef]
- Muzenda, E. An investigation into the effect of water quality on flotation performance. Sci. Rep. 2010, 6, 26172. [Google Scholar]
- Wei, Y.; Sandenbergh, R. Effects of grinding environment on the flotation of Rosh Pinah complex Pb/Zn ore. Miner. Eng. 2007, 20, 264–272. [Google Scholar] [CrossRef][Green Version]
- Liu, J.; Wen, S.; Xian, Y.; Deng, J.; Huang, Y. Dissolubility and surface properties of a natural sphalerite in aqueous solution. Miner. Metall. Process. 2012, 29, 113–120. [Google Scholar] [CrossRef]
- Stanton, M.R.; Gemery-Hill, P.A.; Shanks, W.C.; Taylor, C.D. Rates of zinc and trace metal release from dissolving sphalerite at pH 2.0–4.0. Appl. Geochem. 2008, 23, 136–147. [Google Scholar] [CrossRef]
- Acero, P.; Cama, J.; Ayora, C. Sphalerite dissolution kinetics in acidic environment. Appl. Geochem. 2007, 22, 1872–1883. [Google Scholar] [CrossRef]
- Lu, H.; Fan, H.; Ni, P.; Ou, G.X. Fluid Inclusion; Science Press: Beijing, China, 2004; pp. 1–444. [Google Scholar]
- Ai, G.; Cai, X.; Bi, K.; Li, J.; Tian, Y.; Zhang, R. Research progress on the effect of metal ions on mineral flotation behavior. Nonferrous Met. Sci. Eng. 2017, 8, 70–74. [Google Scholar]
- Deng, J.; Wen, S.; Xian, Y.; Liu, J.; Bai, S. New discovery of unavoidable ions source in chalcopyrite flotation pulp: Fluid inclusions. Miner. Eng. 2013, 42, 22–28. [Google Scholar] [CrossRef]
- Liu, J.; Wen, S.; Wu, D.; Bai, S.; Liu, D. Determination of the concentrations of calcium and magnesium released from fluid inclusions of sphalerite and quartz. Miner. Eng. 2013, 45, 41–43. [Google Scholar] [CrossRef]
- Xian, Y.; Wen, S.; Bai, S.; Chen, C. Metal ions released from fluid inclusions of quartz associated with sulfides. Miner. Eng. 2013, 50–51, 1–3. [Google Scholar] [CrossRef]
- Bai, S.; Wen, S.; Xian, Y.; Liu, J.; Deng, J. New source of unavoidable ions in galena flotation pulp: Components released from fluid inclusions. Miner. Eng. 2013, 45, 94–99. [Google Scholar] [CrossRef]
- Barton, P.B.; I-Ming, C. Calculation of the vapor-saturated liquidus for the NaCl-CO2-H2O system. Geochim. Et Cosmochim. Acta 1993, 57, 2715–2723. [Google Scholar] [CrossRef]
- Bodnar, R.J.; Bethke, P.M. Systematics of stretching of fluid inclusions I: Fluorite and sphalerite at 1 atmosphere confining pressure. Econ. Geol. 1984, 79, 141–161. [Google Scholar] [CrossRef]
- Zeng, Z.; Cao, H.; Gao, F.; Gao, Y.; Zou, H.; Li, D. Fluid inclusion study of fluorite deposits in linxi region, Inner Mongolia. Geochimica 2013, 42, 73–81. [Google Scholar]
- Zhang, S.; Cao, H.; Zheng, L.; Ma, Y.; Fang, J.; Ju, H. Characteristics of ore- forming fluids and mineralization processes of the Shuitou fluorite deposit in Linxi, Inner Mongolia Autonomous Region. Earth Sci. Front. 2014, 21, 31–40. [Google Scholar]
- Pei, Q.; Zhang, S.; Santosh, M.; Cao, H.; Zhang, W.; Hu, X.; Wang, L. Geochronology, geochemistry, fluid inclusion and C, O and Hf isotope compositions of the Shuitou fluorite deposit, Inner Mongolia, China. Ore Geol. Rev. 2017, 83, 174–190. [Google Scholar] [CrossRef]


| F | Ca | Al− | Si | P | S | Fe | Sr | L.O.I. |
|---|---|---|---|---|---|---|---|---|
| 48.429 | 49.647 | 0.149 | 0.961 | 0.002 | 0.005 | 0.073 | 0.016 | 0.717 |
| Sample | Th/°C | Tm/°C | Salinity/wt.% | Density/g/cm3 |
|---|---|---|---|---|
| Fluorite | 155–195 | −0.8–−0.4 | 0.701–1.389 | 0.878–0.927 |
| Grinding Time/min | Ion Concentration/×10−4 mol/L | ||||
|---|---|---|---|---|---|
| Ca2+(T) | F− | Cl− | SO42− | Ca2+(R) | |
| 8 | 14.21 | 6.42 | 0.20 | 0.11 | 11.00 |
| 15 | 30.17 | 26.98 | 0.29 | 0.21 | 16.68 |
| 25 | 56.41 | 42.74 | 0.60 | 0.45 | 35.04 |
| 40 | 97.17 | 50.14 | 0.96 | 0.71 | 72.10 |
| 60 | 114.57 | 52.66 | 1.10 | 0.81 | 88.24 |
| Grinding Time/min | Dissolution Time/h | Ion Concentration/×10−4 mol/L | |||
|---|---|---|---|---|---|
| Ca2+ | F− | Cl− | SO42− | ||
| 60 | 7 | 3.12 | 1.18 | 0.02 | n.a. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, R.; Hong, S.; Wang, S.; Tang, H.; Gao, Z. Effects of Fluid Inclusion Component Release on Flotation Behavior of Fluorite Minerals. Minerals 2025, 15, 912. https://doi.org/10.3390/min15090912
Zheng R, Hong S, Wang S, Tang H, Gao Z. Effects of Fluid Inclusion Component Release on Flotation Behavior of Fluorite Minerals. Minerals. 2025; 15(9):912. https://doi.org/10.3390/min15090912
Chicago/Turabian StyleZheng, Renji, Shilin Hong, Sheng Wang, Honghu Tang, and Zhiyong Gao. 2025. "Effects of Fluid Inclusion Component Release on Flotation Behavior of Fluorite Minerals" Minerals 15, no. 9: 912. https://doi.org/10.3390/min15090912
APA StyleZheng, R., Hong, S., Wang, S., Tang, H., & Gao, Z. (2025). Effects of Fluid Inclusion Component Release on Flotation Behavior of Fluorite Minerals. Minerals, 15(9), 912. https://doi.org/10.3390/min15090912







