Bioleaching of Mine Tailings by Mesophilic: Acidithiobacillus spp., Leptospirillum ferrooxidans, and Thermophilic: Sulfobacillus thermosulfidooxidans Cultures with the Addition of Ag+ Additive
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Characteristics
2.2. Applied Bacterial Cultures
2.3. Description of Experiments
2.4. Blank: Abiotic Conditions
2.5. Analytical Methods
3. Results and Discussion
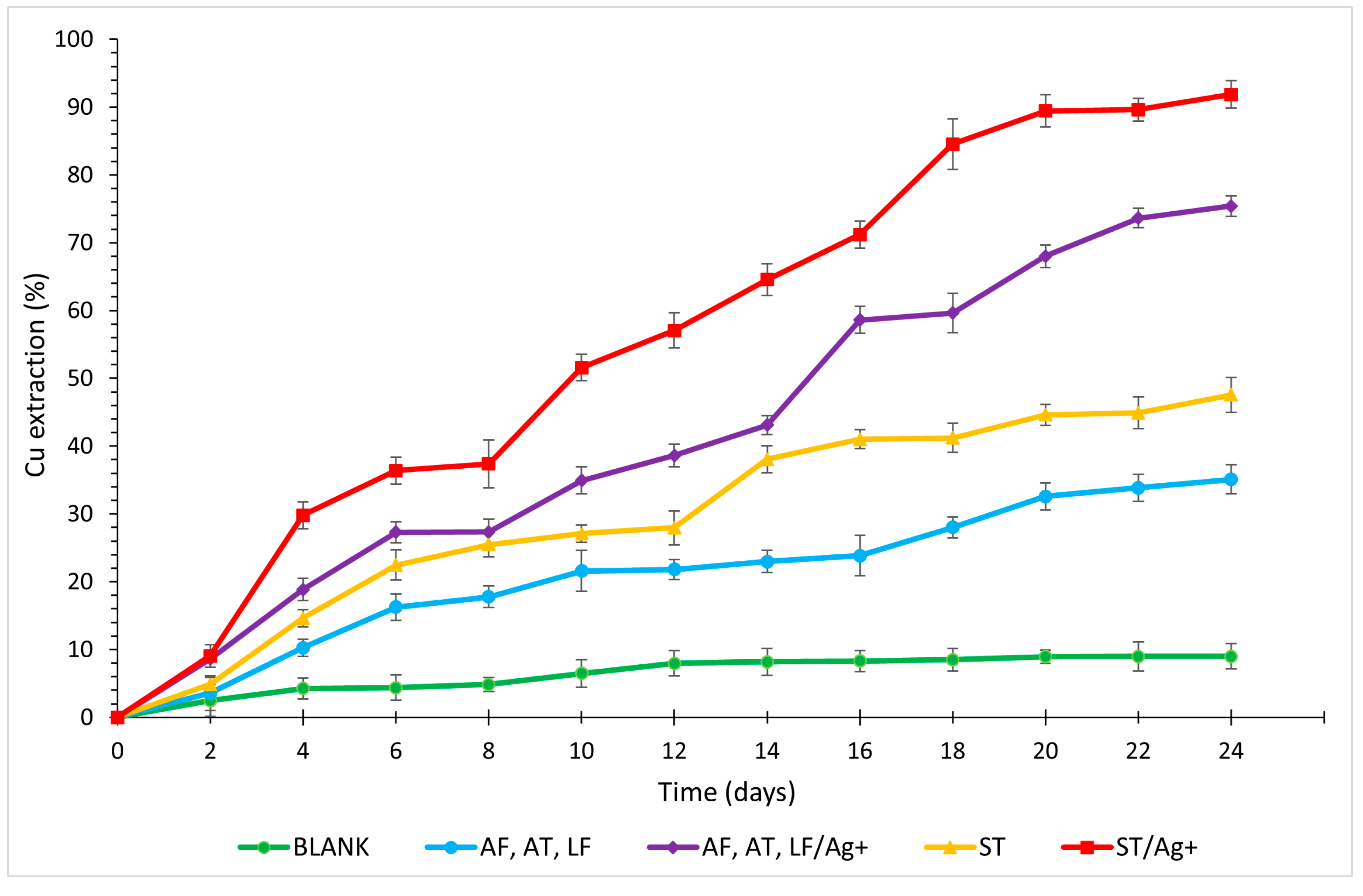
3.1. Copper
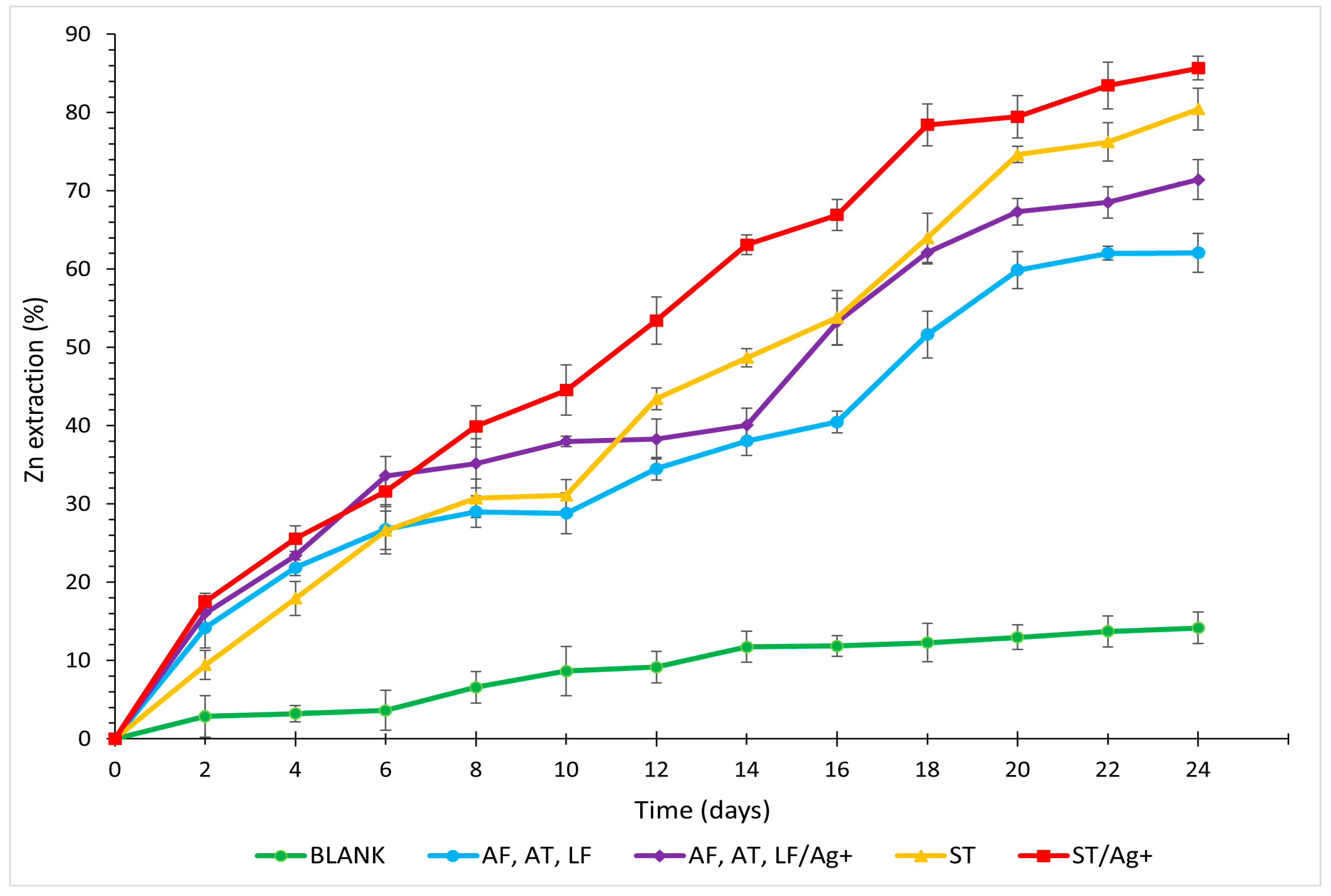
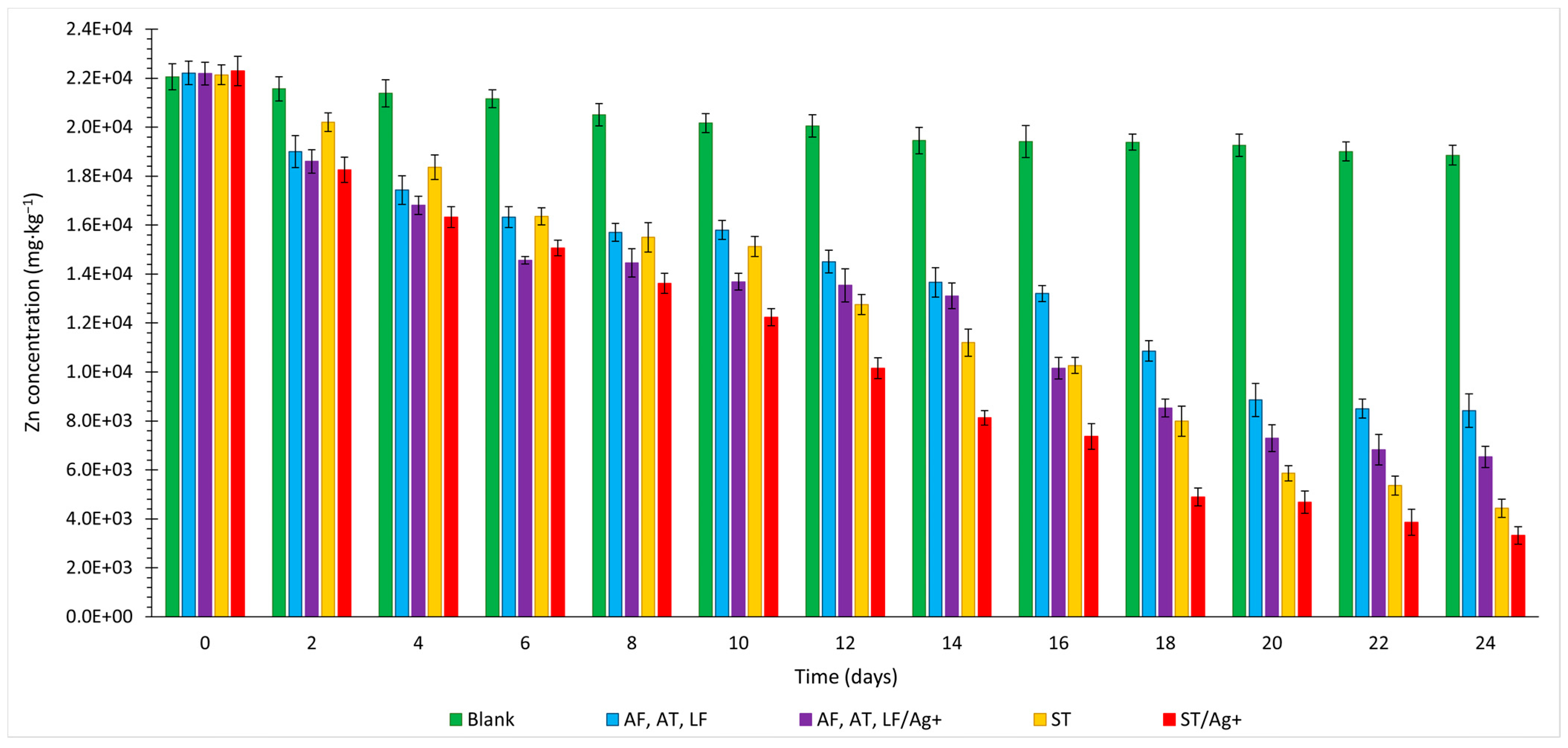
3.2. Zinc
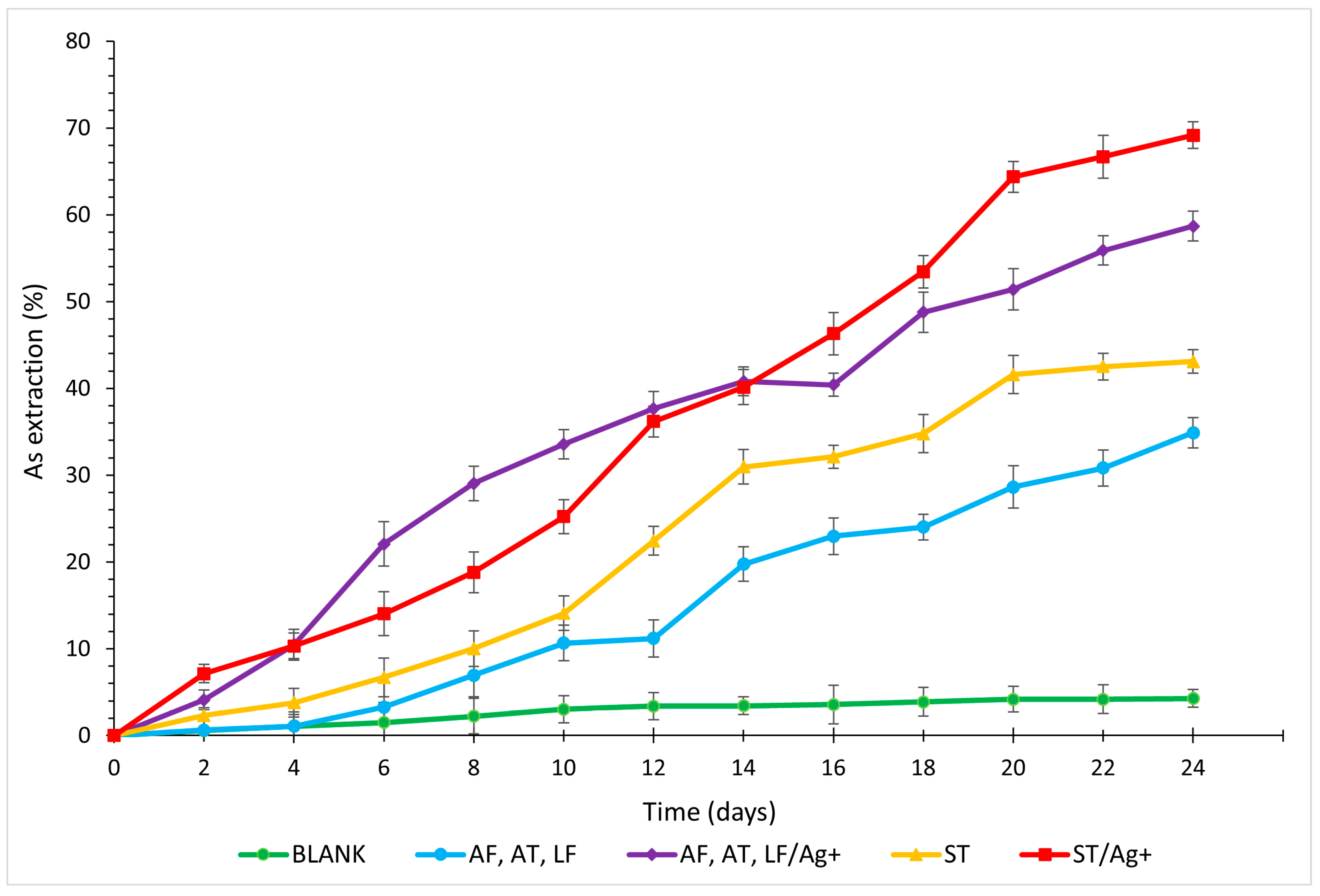
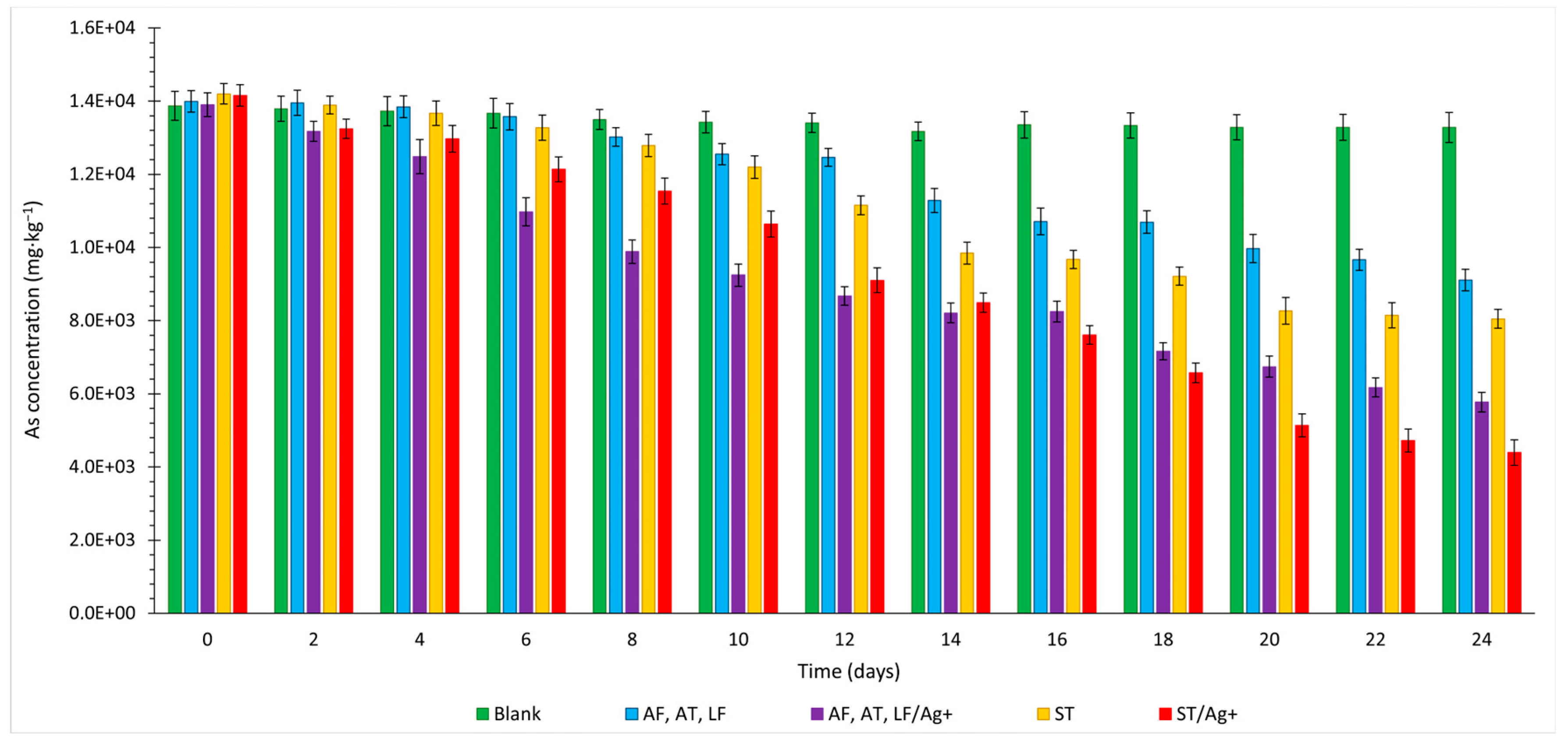
3.3. Arsenic
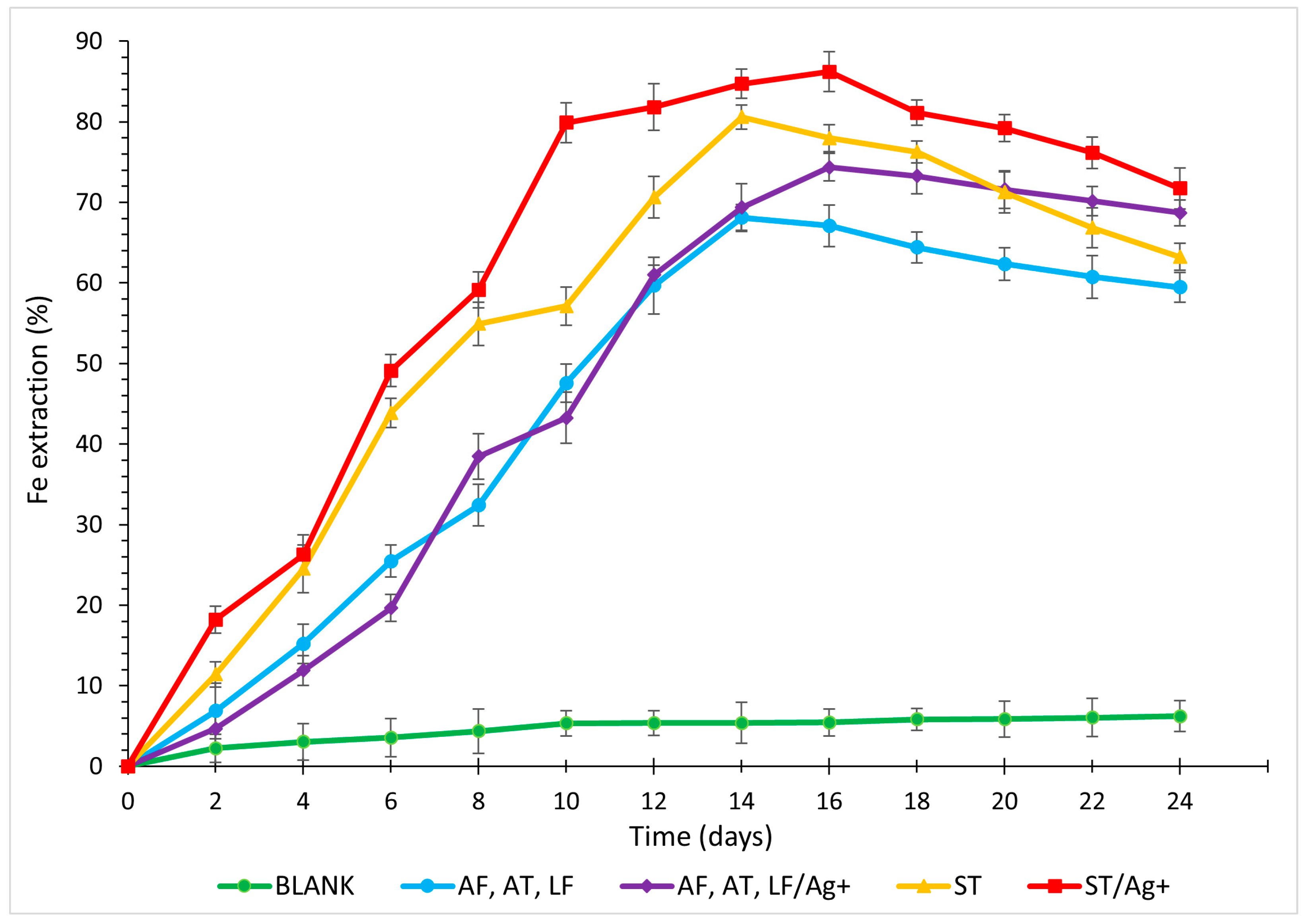
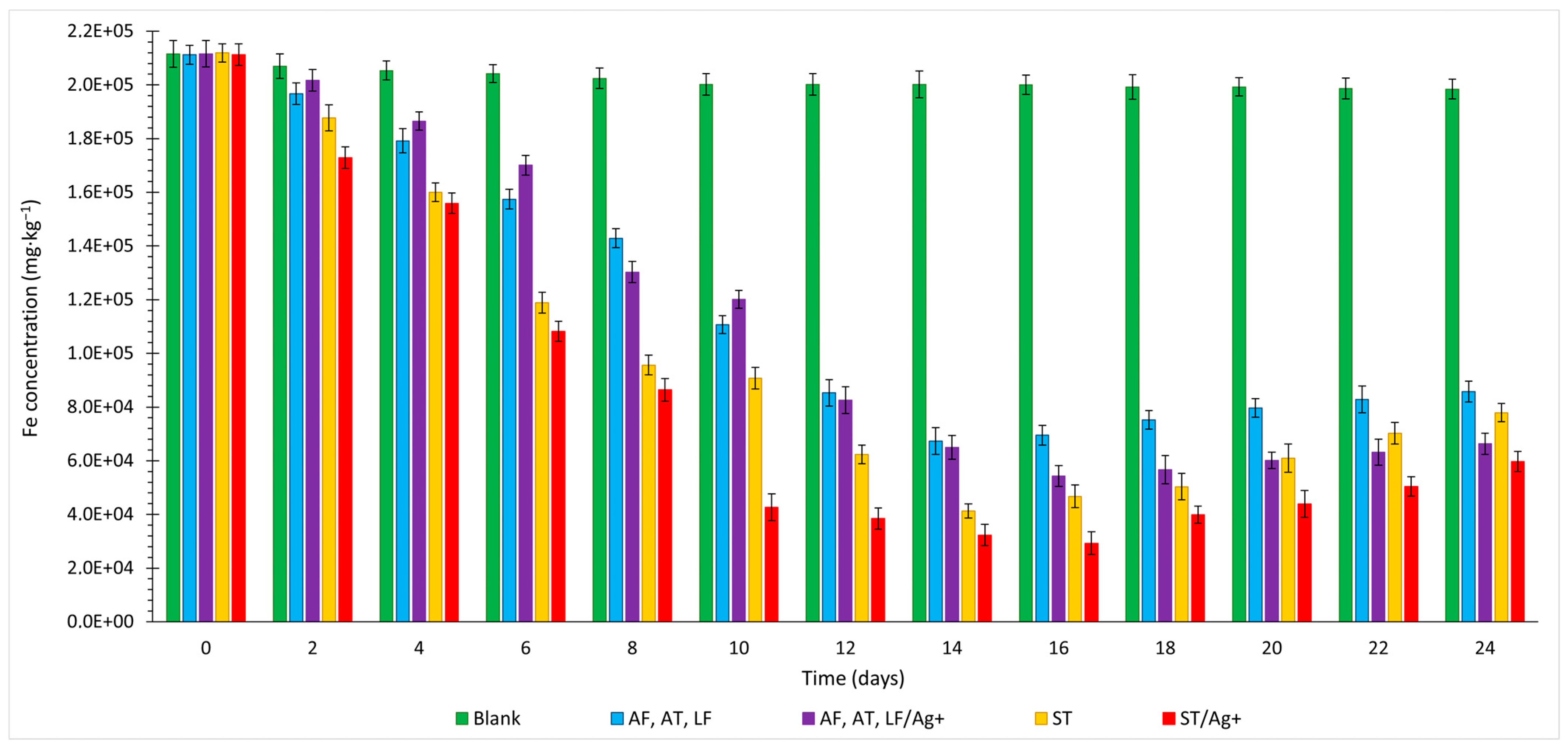
3.4. Iron
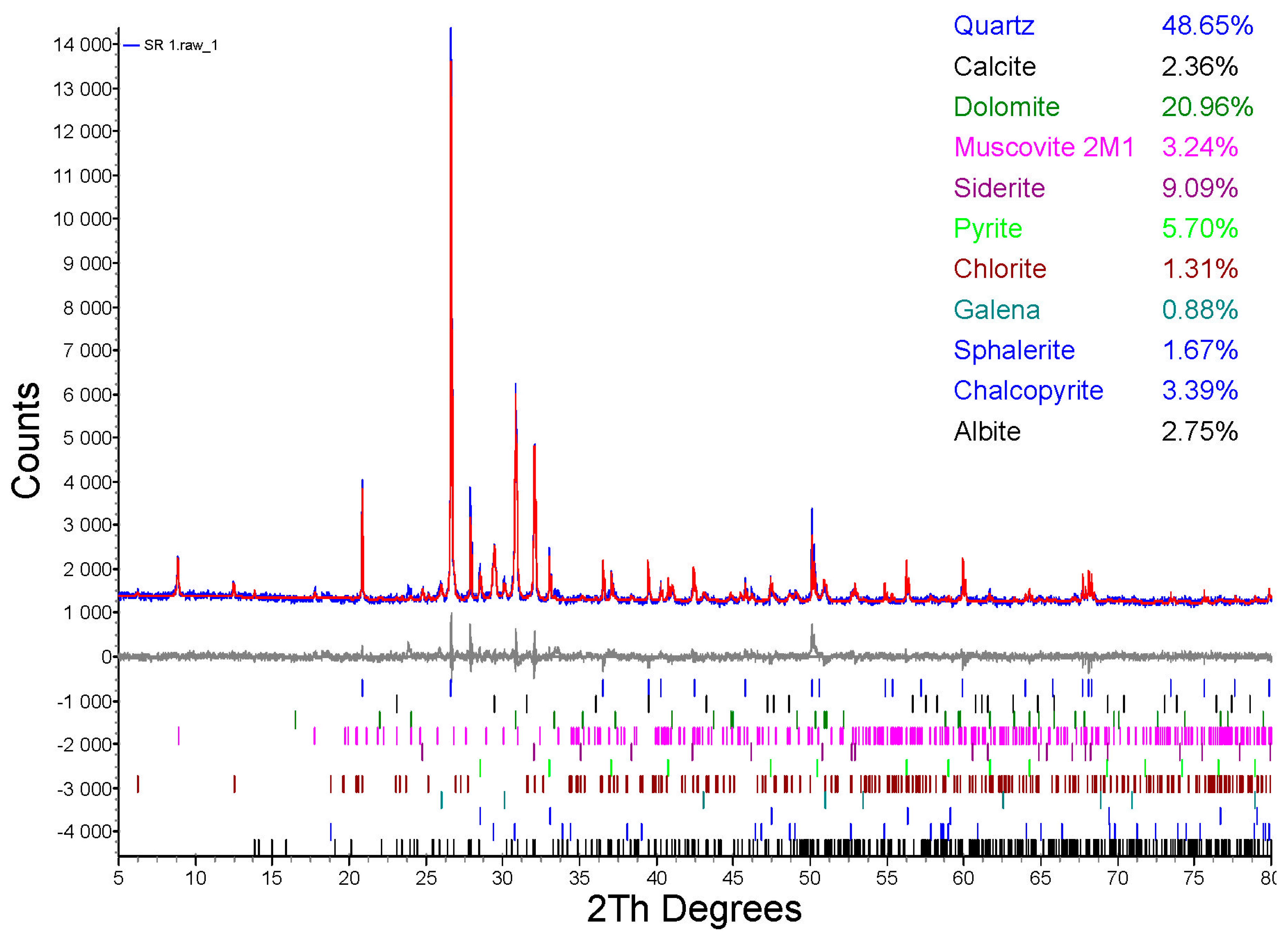
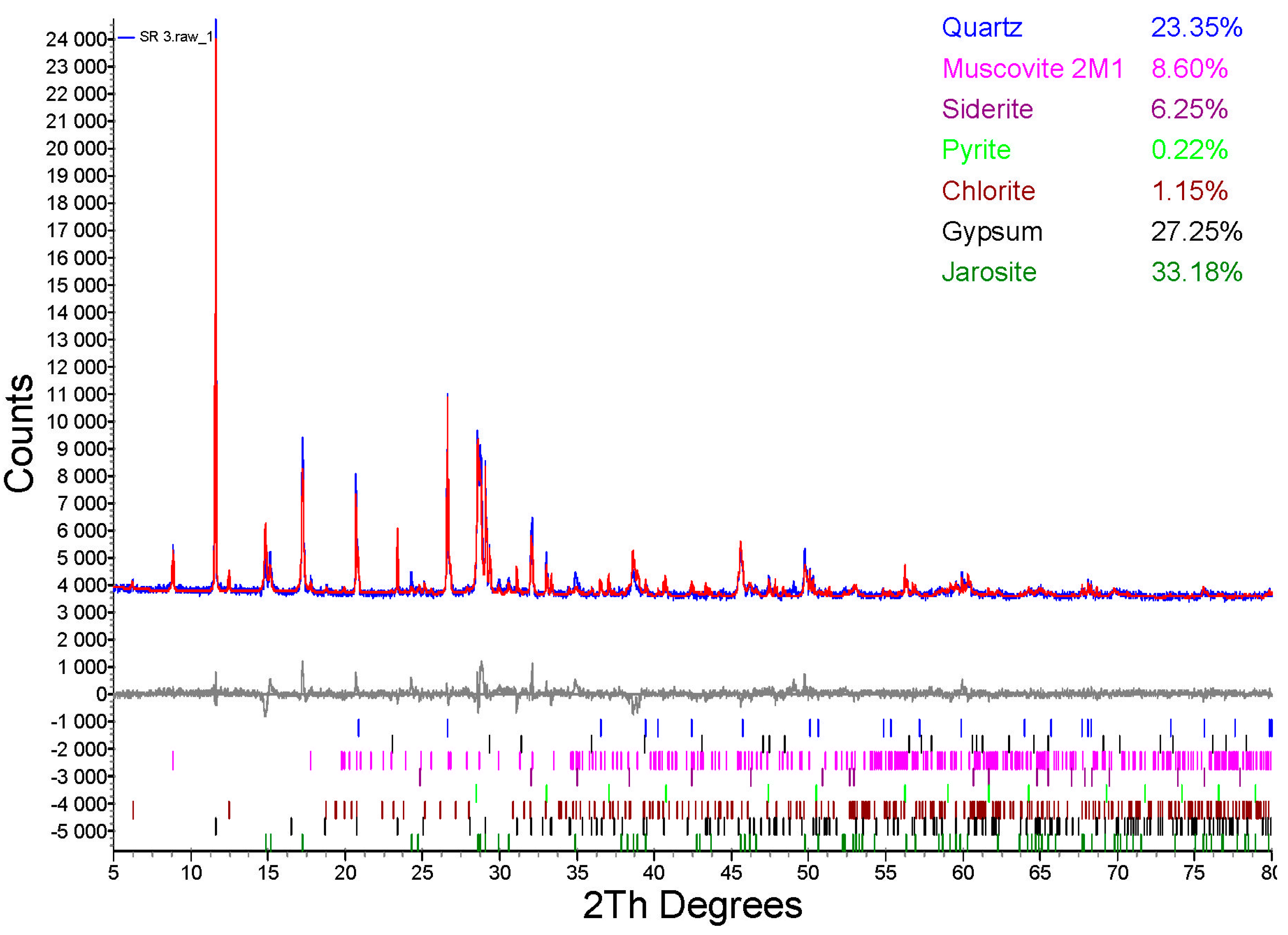
3.5. XRD Detection after Bioleaching
4. Conclusions
- Laboratory tests labelled ST/Ag+ were deemed the most optimal for the final recovery of metals and As for all analysed elements (Cu 91.93%, Zn 85.67%, As 69.16%, and Fe 71.72%, with the maximum solubilisation of Fe reaching 86.21% on day 16).
- The addition of AgNO3 additive enhanced the process kinetics for all detected elements, with a fundamental increase in the solubilisation of Cu (AF, AT, LF/Ag+ by 40.33%; ST/Ag+ by 44.39%) and As (AF, AT, LF/Ag+ by 23.79%; ST/Ag+ by 26.08%).
- Regulating the temperature at 30 °C with the application of ST had a positive effect on the extraction mechanisms, particularly for Zn (ST by 18.36%; ST/Ag+ by 14.24%) compared to mixed mesophilic cultures.
- Two secondary minerals, CaSO4·2H2O and KFe3(SO4)2(OH)6, were detected in the solid fraction after 24 days of leaching.
- Due to the synthesis of the secondary compound, incorporating Fe3+ ions into its structure, the Fe extraction curves initially exhibited an increasing trend (with a peak in the range of 14–18 days), followed by a decline in metal recovery. When mixed mesophilic bacteria were employed, particularly with the representation of the species LF, a lesser reduction in total solubilisation was observed. The optimal ratio transformation of Fe3+/Fe2+ ions resulted in the synthesis of a smaller amount of jarosite-type compound in the reaction environment in the presence of cells of the mentioned species.
- Despite the detection of secondary minerals, the process was deemed highly effective for the recovery of Cu, Zn, and As.
- Comparative analysis of tests under biotic and abiotic conditions demonstrated the influence of bacterial cells and a significant enhancement in the kinetics of extraction processes facilitated by microorganisms.
- Based on the percentage recovery of metals, hydrometallurgical technology involving the cooperative action of microorganisms was assessed as optimal for mine tailings with a lower metal content. From both an environmental and economic perspective, the regeneration of acid leachates and their reuse as new input raw materials are viable.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AF | Acidithiobacillus ferrooxidans |
| AT | Acidithiobacillus thiooxidans |
| EDX | Energy dispersive X-ray spectroscopy |
| F-AAS | Atomic absorption spektrometry |
| LF | Leptospirillum ferrooxidans |
| LOI | Loss on ignition |
| SEM | Scanning electron microscope |
| ST | Sulfobacillus thermosulfidooxidans |
| w/v | weight by volume |
| XRD | X-ray diffraction |
| XRF | X-ray fluorescence |
References
- Vidal, O.; Rostom, F.; François, C.; Giraud, G. Global Trends in Metal Consumption and Supply: The Raw Material–Energy Nexus. Elements 2017, 13, 319–324. [Google Scholar] [CrossRef]
- Alvarenga, R.A.F.; Dewulf, J.; Guinée, J.; Schulze, R.; Weihed, P.; Bark, G.; Drielsma, J. Towards Product-Oriented Sustainability in the (Primary) Metal Supply Sector. Resour. Conserv. Recycl. 2019, 145, 40–48. [Google Scholar] [CrossRef]
- Elshkaki, A.; Graedel, T.E.; Ciacci, L.; Reck, B.K. Resource Demand Scenarios for the Major Metals. Environ. Sci. Technol. 2018, 52, 2491–2497. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Pan, J.; Dong, B.; Zhou, W.; Zhou, C. Bioleaching of Rare Earth Elements: Perspectives from Mineral Characteristics and Microbial Species. Minerals 2023, 13, 1186. [Google Scholar] [CrossRef]
- Mishra, S.; Panda, S.; Akcil, A.; Dembele, S. Biotechnological Avenues in Mineral Processing: Fundamentals, Applications and Advances in Bioleaching and Bio-Beneficiation. Miner. Process. Extr. Metall. Rev. 2023, 44, 22–51. [Google Scholar] [CrossRef]
- Srichandan, H.; Mohapatra, R.K.; Parhi, P.K.; Mishra, S. Bioleaching Approach for Extraction of Metal Values from Secondary Solid Wastes: A Critical Review. Hydrometallurgy 2019, 189, 105122. [Google Scholar] [CrossRef]
- Liu, X.; Li, Q.; Zhang, Y.; Jiang, T.; Yang, Y.; Xu, B.; He, Y. Electrochemical Behaviour of the Dissolution and Passivation of Arsenopyrite in 9K Culture Medium. Appl. Surf. Sci. 2020, 508, 145269. [Google Scholar] [CrossRef]
- Sepehri, A.; Sarrafzadeh, M.-H. Activity Enhancement of Ammonia-Oxidizing Bacteria and Nitrite-Oxidizing Bacteria in Activated Sludge Process: Metabolite Reduction and CO2 Mitigation Intensification Process. Appl. Water Sci. 2019, 9, 131. [Google Scholar] [CrossRef]
- Zhang, D.; Chen, H.; Xia, J.; Nie, Z.; Fan, X.; Liu, H.; Zheng, L.; Zhang, L.; Yang, H. Humic Acid Promotes Arsenopyrite Bio-Oxidation and Arsenic Immobilization. J. Hazard. Mater. 2020, 384, 121359. [Google Scholar] [CrossRef]
- Bogdanovic, G.; Stankovic, V.; Trumic, M.; Antic, D.; Trumic, M. Leaching of Low-Grade Copper Ores: A Case Study for “Kraku Bugaresku-Cementacija” Deposits (Eastern Serbia). J. Min. Met. A 2016, 52, 45–56. [Google Scholar] [CrossRef]
- Guler, E. Pressure Acid Leaching of Sphalerite Concentrate. Modeling and Optimization by Response Surface Methodology. Physicochem. Probl. Miner. Process. 2016, 52, 479–496. [Google Scholar] [CrossRef]
- Mejia, E.R.; Ospina, J.D.; Marquez, A.L.M. Bioleaching of Galena (PbS). In Fourier Transform–Materials Analysis; Salih, S., Ed.; InTech: London, UK, 2012; ISBN 978-953-51-0594-7. [Google Scholar]
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, 3rd ed.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2010; ISBN 978-3-642-12418-1. [Google Scholar]
- Pour, H.R.; Mostafavi, A.; Pur, T.S.; Pour, G.E.; Omran, A.H.Z. Removal of Sulfur and Phosphorous from Iron Ore Concentrate by Leaching. Physicochem. Probl. Miner. Process. 2016, 52, 845–854. [Google Scholar] [CrossRef]
- Ghassa, S.; Noaparast, M.; Shafaei, S.Z.; Abdollahi, H.; Gharabaghi, M.; Boruomand, Z. A Study on the Zinc Sulfide Dissolution Kinetics with Biological and Chemical Ferric Reagents. Hydrometallurgy 2017, 171, 362–373. [Google Scholar] [CrossRef]
- Fomchenko, N.V.; Muravyov, M.I. Selective Leaching of Zinc from Copper-Zinc Concentrate. Appl. Biochem. Microbiol. 2017, 53, 73–77. [Google Scholar] [CrossRef]
- Conić, V.T.; Vujasinović, M.M.R.; Trujić, V.K.; Cvetkovski, V.B. Copper, Zinc, and Iron Bioleaching from Polymetallic Sulphide Concentrate. Trans. Nonferr. Met. Soc. China 2014, 24, 3688–3695. [Google Scholar] [CrossRef]
- Dew, D.W.; Van Buuren, C.; McEwan, K.; Bowker, C. Bioleaching of Base Metal Sulphide Concentrates: A Comparison of Mesophile and Thermophile Bacterial Cultures. In Process Metallurgy; Elsevier: Amsterdam, The Netherlands, 1999; Volume 9, pp. 229–238. ISBN 978-0-444-50193-6. [Google Scholar]
- Pradhan, N.; Nathsarma, K.C.; Srinivasa Rao, K.; Sukla, L.B.; Mishra, B.K. Heap Bioleaching of Chalcopyrite: A Review. Miner. Eng. 2008, 21, 355–365. [Google Scholar] [CrossRef]
- Li, Y.; Kawashima, N.; Li, J.; Chandra, A.P.; Gerson, A.R. A Review of the Structure, and Fundamental Mechanisms and Kinetics of the Leaching of Chalcopyrite. Adv. Colloid Interface Sci. 2013, 197–198, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Watling, H.R. The Bioleaching of Sulphide Minerals with Emphasis on Copper Sulphides–A Review. Hydrometallurgy 2006, 84, 81–108. [Google Scholar] [CrossRef]
- Gu, G.; Hu, K.; Zhang, X.; Xiong, X.; Yang, H. The Stepwise Dissolution of Chalcopyrite Bioleached by Leptospirillum ferriphilum. Electrochim. Acta 2013, 103, 50–57. [Google Scholar] [CrossRef]
- Feng, S.; Yang, H.; Xin, Y.; Gao, K.; Yang, J.; Liu, T.; Zhang, L.; Wang, W. A Novel and Highly Efficient System for Chalcopyrite Bioleaching by Mixed Strains of Acidithiobacillus. Bioresour. Technol. 2013, 129, 456–462. [Google Scholar] [CrossRef]
- Lee, E.; Han, Y.; Park, J.; Hong, J.; Silva, R.A.; Kim, S.; Kim, H. Bioleaching of Arsenic from Highly Contaminated Mine Tailings Using Acidithiobacillus Thiooxidans. J. Environ. Manag. 2015, 147, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Liu, J.; Xiao, L.; Zeng, J.; Li, B.; Geng, M.; Qiu, G. Single and Cooperative Bioleaching of Sphalerite by Two Kinds of Bacteria—Acidithiobacillus ferriooxidans and Acidithiobacillus thiooxidans. Trans. Nonferr. Met. Soc. China 2008, 18, 190–195. [Google Scholar] [CrossRef]
- Umanskii, A.B.; Klyushnikov, A.M. Bioleaching of Low Grade Uranium Ore Containing Pyrite Using A. ferrooxidans and A. thiooxidans. J. Radioanal. Nucl. Chem. 2013, 295, 151–156. [Google Scholar] [CrossRef]
- Silva, R.A.; Park, J.; Ilyas, S.; Borja, D.; Zhao, H.; Urík, M.; Rastegar, S.O.; Kim, H. Biodegradation Mechanism of Arsenopyrite Mine Tailing with Acidithiobacillus ferrooxidans and Influence of Ferric Supplements. Int. Biodeterior. Biodegrad. 2020, 153, 105042. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Q.; Liu, X.; Yin, H.; Yang, Y.; Xu, B.; Jiang, T.; He, Y. The Catalytic Effect of Copper Ion in the Bioleaching of Arsenopyrite by Acidithiobacillus ferrooxidans in 9K Culture Medium. J. Clean. Prod. 2020, 256, 120391. [Google Scholar] [CrossRef]
- Chang-Li, L.; Jin-Lan, X.; Zhen-Yuan, N.; Yi, Y.; Chen-Yan, M. Effect of Sodium Chloride on Sulfur Speciation of Chalcopyrite Bioleached by the Extreme Thermophile Acidianus Manzaensis. Bioresour. Technol. 2012, 110, 462–467. [Google Scholar] [CrossRef]
- Rahaman, M.S.; Basu, A.; Islam, M.R. The Removal of As(III) and As(V) from Aqueous Solutions by Waste Materials. Bioresour. Technol. 2008, 99, 2815–2823. [Google Scholar] [CrossRef]
- Wang, J.; Liao, R.; Tao, L.; Zhao, H.; Zhai, R.; Qin, W.; Qiu, G. A Comprehensive Utilization of Silver-Bearing Solid Wastes in Chalcopyrite Bioleaching. Hydrometallurgy 2017, 169, 152–157. [Google Scholar] [CrossRef]
- Hao, X.; Liu, X.; Zhu, P.; Chen, A.; Liu, H.; Yin, H.; Qiu, G.; Liang, Y. Carbon Material with High Specific Surface Area Improves Complex Copper Ores’ Bioleaching Efficiency by Mixed Moderate Thermophiles. Minerals 2018, 8, 301. [Google Scholar] [CrossRef]
- Akcil, A.; Ciftci, H.; Deveci, H. Role and Contribution of Pure and Mixed Cultures of Mesophiles in Bioleaching of a Pyritic Chalcopyrite Concentrate. Miner. Eng. 2007, 20, 310–318. [Google Scholar] [CrossRef]
- Deng, S.; Gu, G.; Wu, Z.; Xu, X. Bioleaching of Arsenopyrite by Mixed Cultures of Iron-Oxidizing and Sulfur-Oxidizing Microorganisms. Chemosphere 2017, 185, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, S.; Anwar, M.A.; Niazi, S.B.; Afzal Ghauri, M. Bioleaching of Metals from Electronic Scrap by Moderately Thermophilic Acidophilic Bacteria. Hydrometallurgy 2007, 88, 180–188. [Google Scholar] [CrossRef]
- Ghobadi, S.; Mehrabani, J.V.; Panda, S. Catalytic Effect of Graphite Promoting Zn Dissolution from Sphalerite Using a Leptospirillum ferrooxidans Dominated Mixed Culture. Geomicrobiol. J. 2022, 39, 199–209. [Google Scholar] [CrossRef]
- Rouchalova, D.; Rouchalova, K.; Janakova, I.; Cablik, V.; Janstova, S. Bioleaching of Iron, Copper, Lead, and Zinc from the Sludge Mining Sediment at Different Particle Sizes, pH, and Pulp Density Using Acidithiobacillus Ferrooxidans. Minerals 2020, 10, 1013. [Google Scholar] [CrossRef]
- Silverman, M.P.; Lundgren, D.G. Studies on the Chemoautotrophic Iron Bacterium Feroobacillus Ferrooxidans: I. An Improved Medium and a Harvesting Procedure for Securing High Cell Yields. J. Bacteriol. 1959, 78, 326–331. [Google Scholar] [CrossRef]
- ČSN 72 0103; Basic Analysis of Silicates–Determination of Loss on Ignition. UNMZ–Czech Office for Standards. Metrology and Testing: Prague, Czech Republic, 2009.
- Ye, M.; Yan, P.; Sun, S.; Han, D.; Xiao, X.; Zheng, L.; Huang, S.; Chen, Y.; Zhuang, S. Bioleaching Combined Brine Leaching of Heavy Metals from Lead-Zinc Mine Tailings: Transformations during the Leaching Process. Chemosphere 2017, 168, 1115–1125. [Google Scholar] [CrossRef]
- Nguyen, V.K.; Lee, M.H.; Park, H.J.; Lee, J.-U. Bioleaching of Arsenic and Heavy Metals from Mine Tailings by Pure and Mixed Cultures of Acidithiobacillus spp. J. Ind. Eng. Chem. 2015, 21, 451–458. [Google Scholar] [CrossRef]
- Miller, J.D.; McDonough, P.J.; Portillo, H.Q. Electrochemistry in Silver Catalysed Ferric Sulfate Leaching of Chalcopyrite. Soc. Min. Metall. Explor. 1981, 27, 327–338. [Google Scholar]
- Parker, A.; Klauber, C.; Kougianos, A.; Watling, H.R.; Van Bronswijk, W. An X-ray Photoelectron Spectroscopy Study of the Mechanism of Oxidative Dissolution of Chalcopyrite. Hydrometallurgy 2003, 71, 265–276. [Google Scholar] [CrossRef]
- Nazari, G.; Dixon, D.G.; Dreisinger, D.B. The Mechanism of Chalcopyrite Leaching in the Presence of Silver-Enhanced Pyrite in the Galvanox™ Process. Hydrometallurgy 2012, 113–114, 122–130. [Google Scholar] [CrossRef]
- Ghahremaninezhad, A.; Radzinski, R.; Gheorghiu, T.; Dixon, D.G.; Asselin, E. A Model for Silver Ion Catalysis of Chalcopyrite (CuFeS2) Dissolution. Hydrometallurgy 2015, 155, 95–104. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, Y.; Deng, T.; Wang, K. Kinetic Modeling for the Bacterial Leaching of Chalcopyrite Catalyzed by Silver Ions. Miner. Eng. 2004, 17, 943–947. [Google Scholar] [CrossRef]
- Marrero, J.; Coto, O.; Schippers, A. 11 Metal Bioleaching: Fundamentals and Geobiotechnical Application of Aerobic and Anaerobic Acidophiles. In Biotechnological Applications of Extremophilic Microorganisms; Lee, N.M., Ed.; De Gruyter: Munich, Germany, 2020; pp. 261–288. ISBN 978-3-11-042433-1. [Google Scholar]
- Van Der Merwe, J.A.; Deane, S.M.; Rawlings, D.E. The Chromosomal Arsenic Resistance Genes of Sulfobacillus thermosulfidooxidans. Hydrometallurgy 2010, 104, 477–482. [Google Scholar] [CrossRef]
- Watkin, E.L.J.; Keeling, S.E.; Perrot, F.A.; Shiers, D.W.; Palmer, M.-L.; Watling, H.R. Metals Tolerance in Moderately Thermophilic Isolates from a Spent Copper Sulfide Heap, Closely Related to Acidithiobacillus caldus, Acidimicrobium ferrooxidans and Sulfobacillus thermosulfidooxidans. J. Ind. Microbiol. Biotechnol. 2009, 36, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Golyshina, O.V. Environmental, Biogeographic, and Biochemical Patterns of Archaea of the Family Ferroplasmaceae. Appl. Environ. Microbiol. 2011, 77, 5071–5078. [Google Scholar] [CrossRef] [PubMed]
- Fraser, K.S.; Walton, R.H.; Wells, J.A. Processing of Refractory Gold Ores. Miner. Eng. 1991, 4, 1029–1041. [Google Scholar] [CrossRef]
- Márquez, M.; Gaspar, J.; Bessler, K.E.; Magela, G. Process Mineralogy of Bacterial Oxidized Gold Ore in São Bento Mine (Brasil). Hydrometallurgy 2006, 83, 114–123. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, D.; Xia, J.; Nie, Z.; Liu, H.; Wang, N.; Xue, Z. Enhancement of Arsenopyrite Bioleaching by Different Fe(III) Compounds through Changing Composition and Structure of Passivation Layer. J. Mater. Res. Technol. 2020, 9, 12364–12377. [Google Scholar] [CrossRef]
- Cruz, L.F.; Cobine, P.A.; De La Fuente, L. Calcium Increases Xylella fastidiosa Surface Attachment, Biofilm Formation, and Twitching Motility. Appl. Environ. Microbiol. 2012, 78, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Coram, N.J.; Rawlings, D.E. Molecular Relationship between Two Groups of the Genus Leptospirillum and the Finding That Leptospirillum ferriphilum sp. Nov. Dominates South African Commercial Biooxidation Tanks That Operate at 40 °C. Appl. Environ. Microbiol. 2002, 68, 838–845. [Google Scholar] [CrossRef]
- Möller, D. Chemistry for Environmental Scientists; De Gruyter: Munich, Germany, 2022; ISBN 978-3-11-073517-8. [Google Scholar]


| Elements | Mass Fraction (%) | Elements | Mass Fraction (%) | ||
|---|---|---|---|---|---|
| Average | SD | Average | SD | ||
| As | 13.900 | 0.038 | P | 0.009 | 0.001 |
| Ca | 61.746 | 0.194 | S | 4.886 | 0.181 |
| Cu | 2.190 | 0.057 | Zn | 2.210 | 0.097 |
| Fe | 21.122 | 0.079 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rouchalová, D.; Rouchalová, K.; Čablík, V. Bioleaching of Mine Tailings by Mesophilic: Acidithiobacillus spp., Leptospirillum ferrooxidans, and Thermophilic: Sulfobacillus thermosulfidooxidans Cultures with the Addition of Ag+ Additive. Minerals 2024, 14, 255. https://doi.org/10.3390/min14030255
Rouchalová D, Rouchalová K, Čablík V. Bioleaching of Mine Tailings by Mesophilic: Acidithiobacillus spp., Leptospirillum ferrooxidans, and Thermophilic: Sulfobacillus thermosulfidooxidans Cultures with the Addition of Ag+ Additive. Minerals. 2024; 14(3):255. https://doi.org/10.3390/min14030255
Chicago/Turabian StyleRouchalová, Dana, Kamila Rouchalová, and Vladimír Čablík. 2024. "Bioleaching of Mine Tailings by Mesophilic: Acidithiobacillus spp., Leptospirillum ferrooxidans, and Thermophilic: Sulfobacillus thermosulfidooxidans Cultures with the Addition of Ag+ Additive" Minerals 14, no. 3: 255. https://doi.org/10.3390/min14030255
APA StyleRouchalová, D., Rouchalová, K., & Čablík, V. (2024). Bioleaching of Mine Tailings by Mesophilic: Acidithiobacillus spp., Leptospirillum ferrooxidans, and Thermophilic: Sulfobacillus thermosulfidooxidans Cultures with the Addition of Ag+ Additive. Minerals, 14(3), 255. https://doi.org/10.3390/min14030255








