Modes of Occurrence, Elemental Relationships, and Economic Viability of Rare Earth Elements in West Virginia Coals: A Statistical Approach
Abstract
:1. Introduction
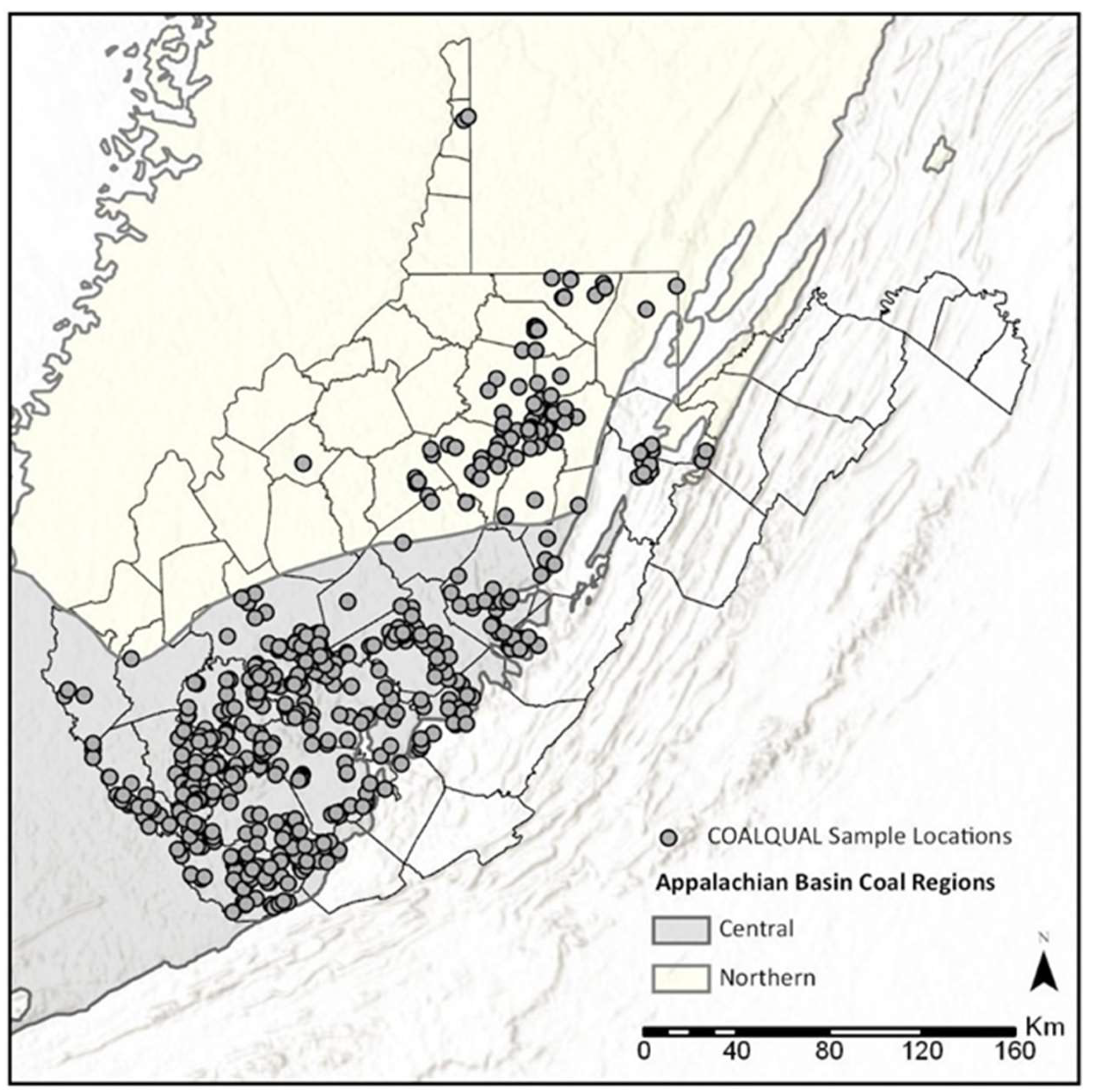
2. Materials & Methods
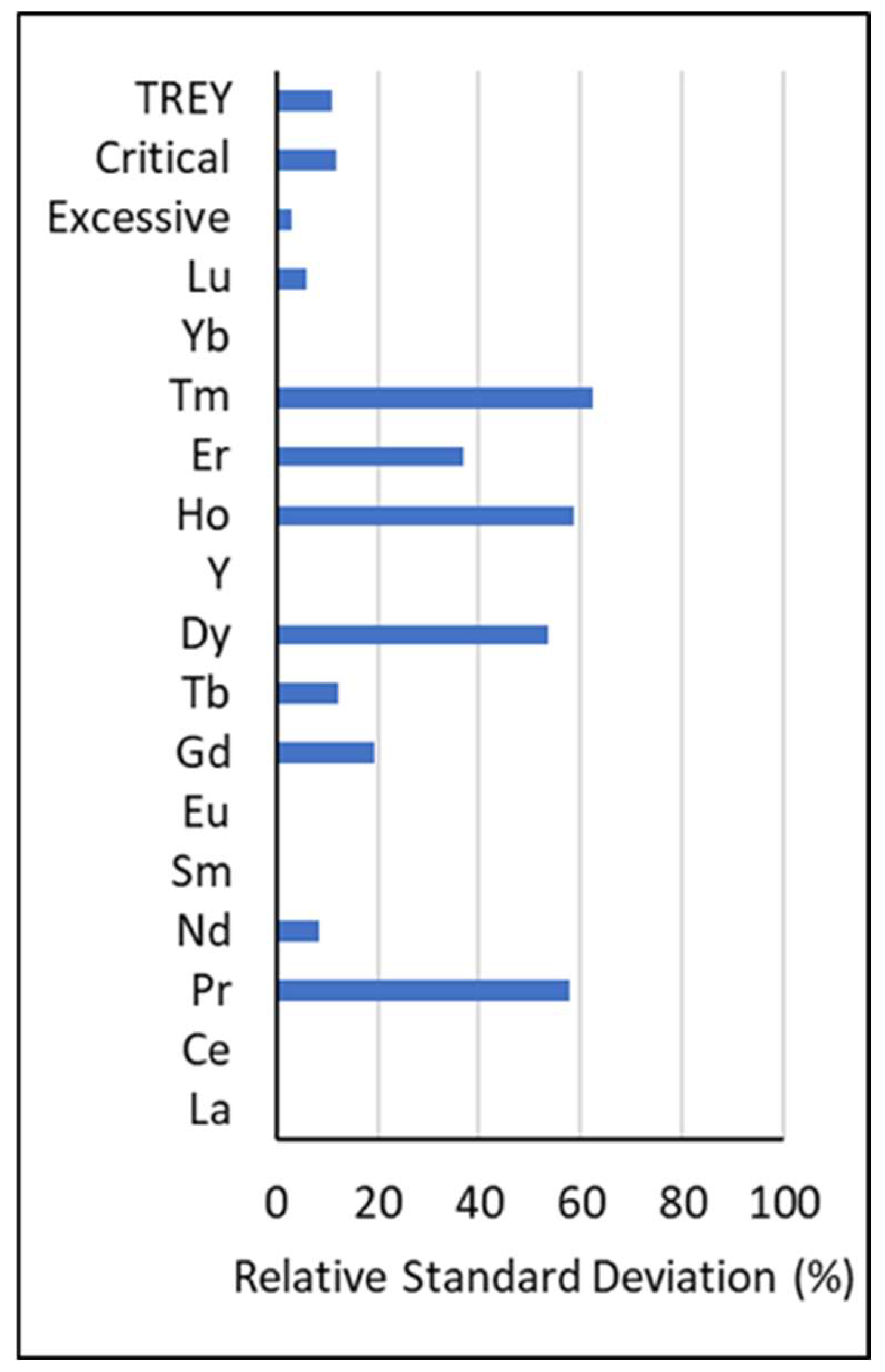
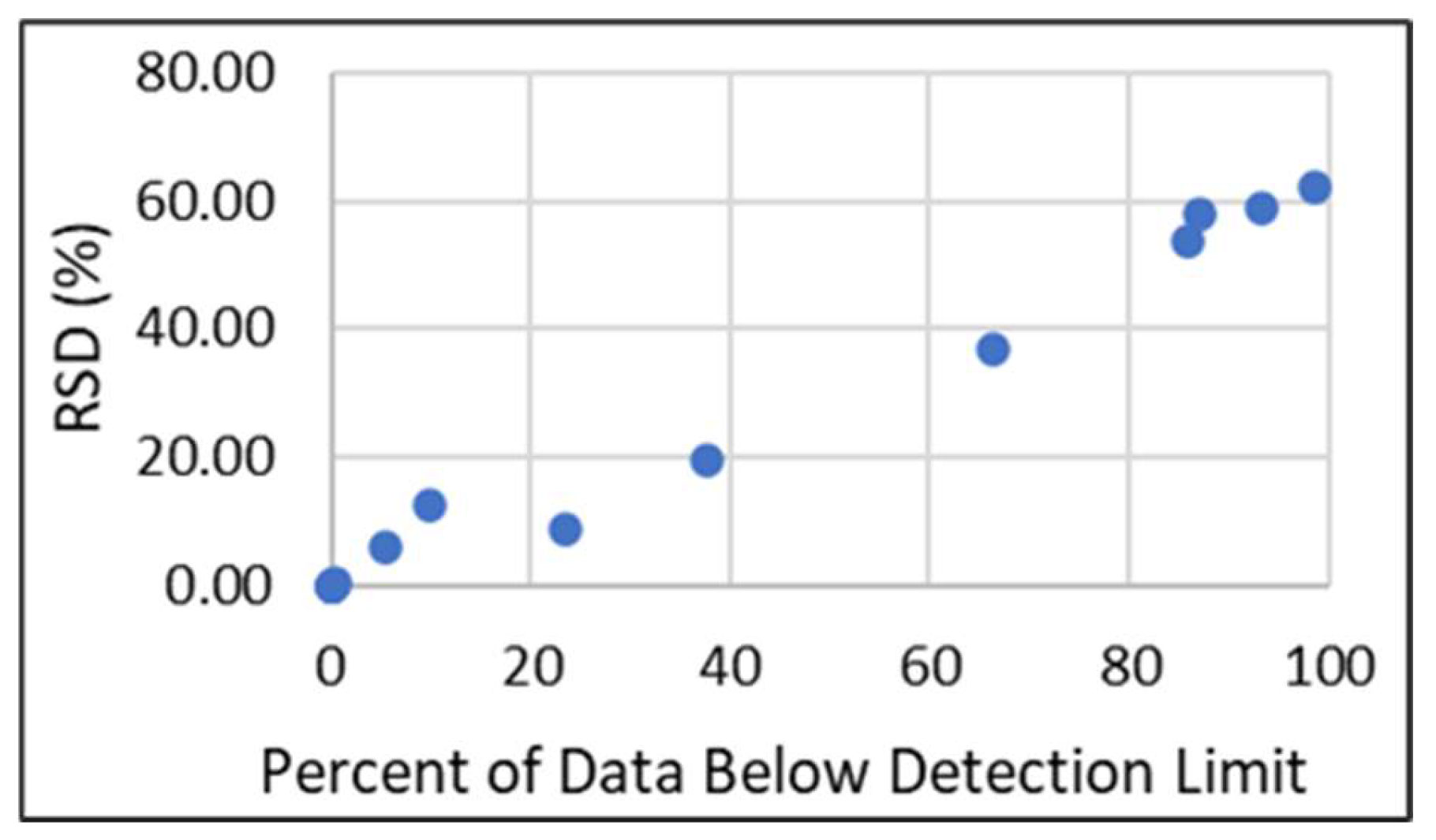
2.1. Data Substitutions
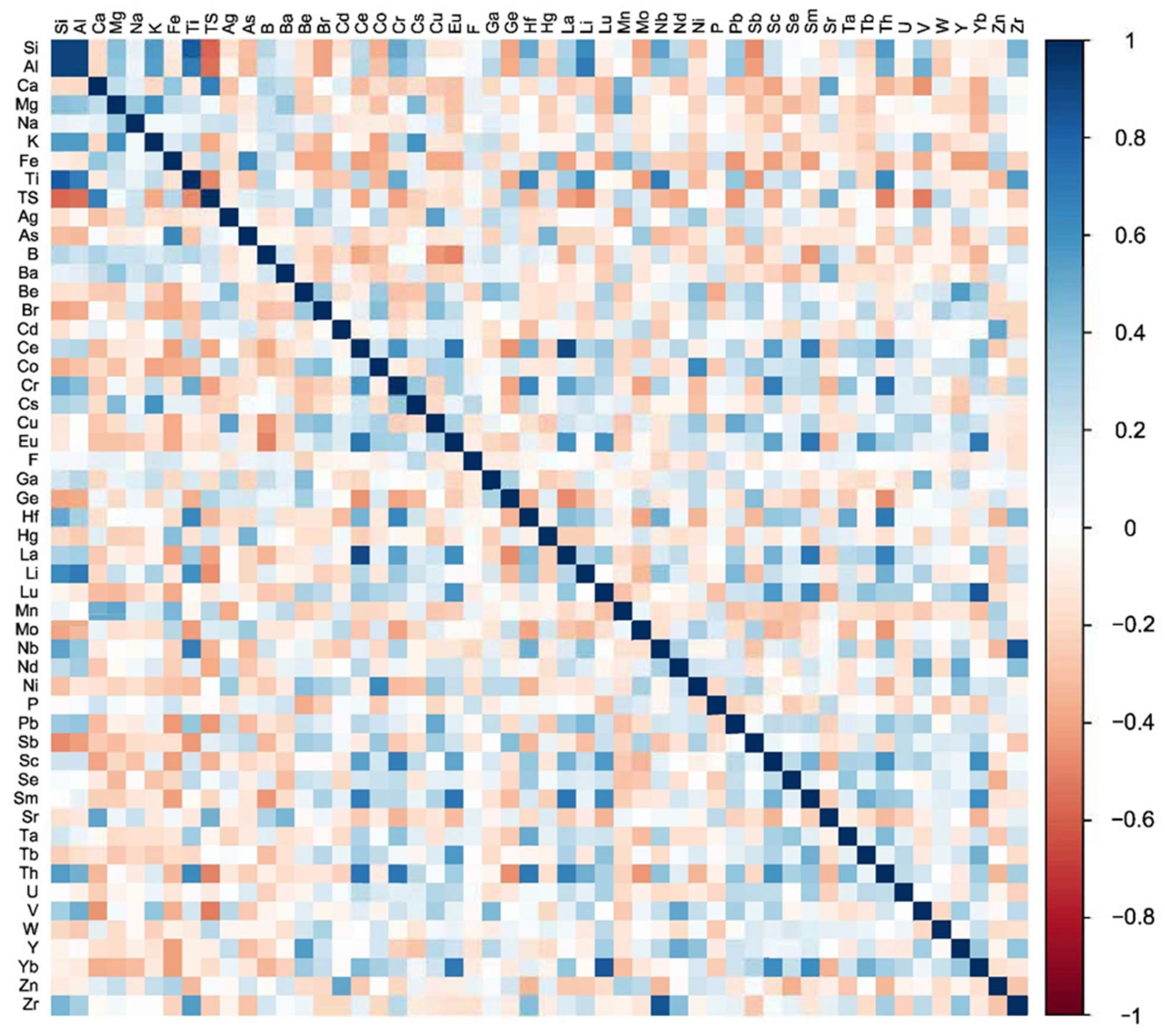
2.2. Pearson Correlation
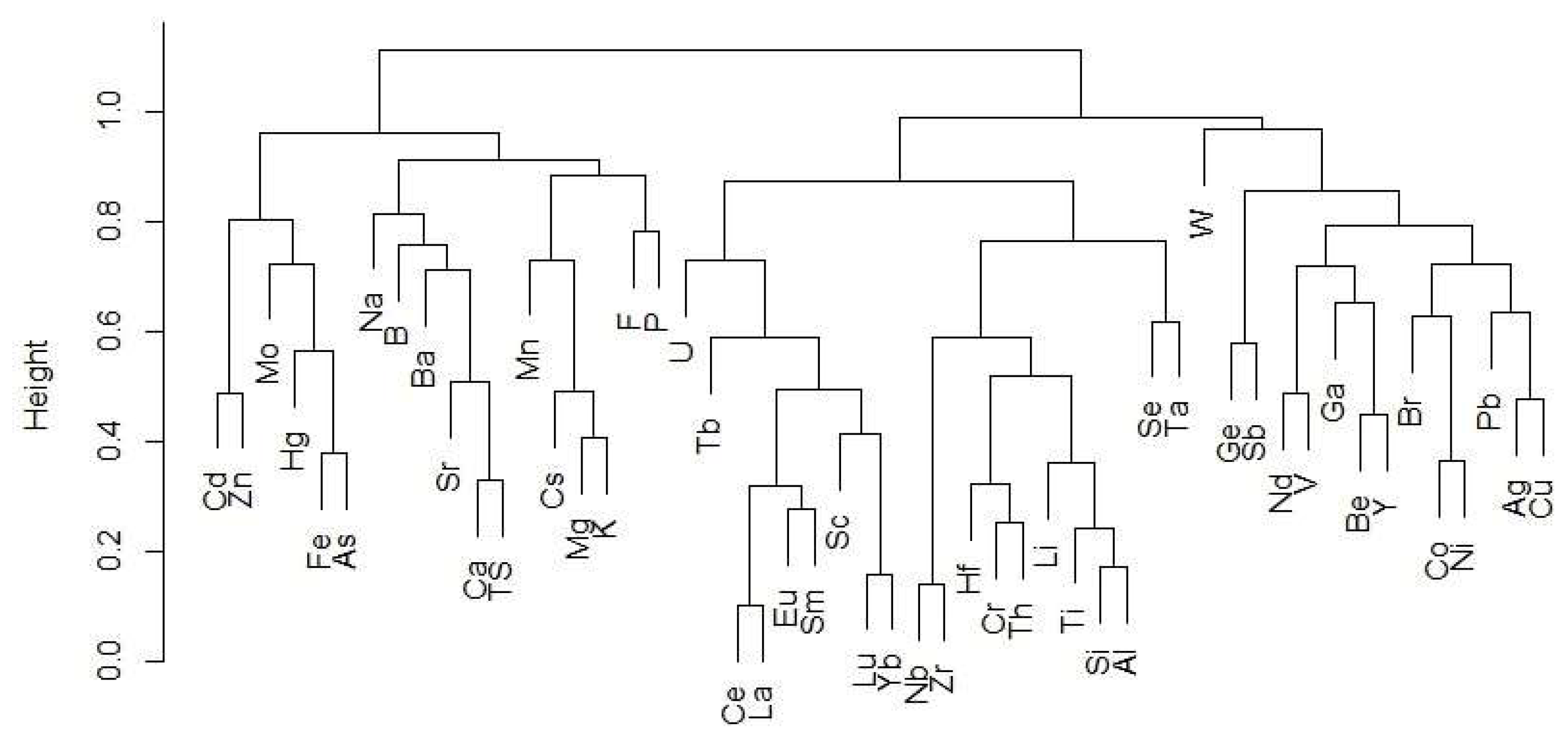
2.3. Hierarchical Clustering
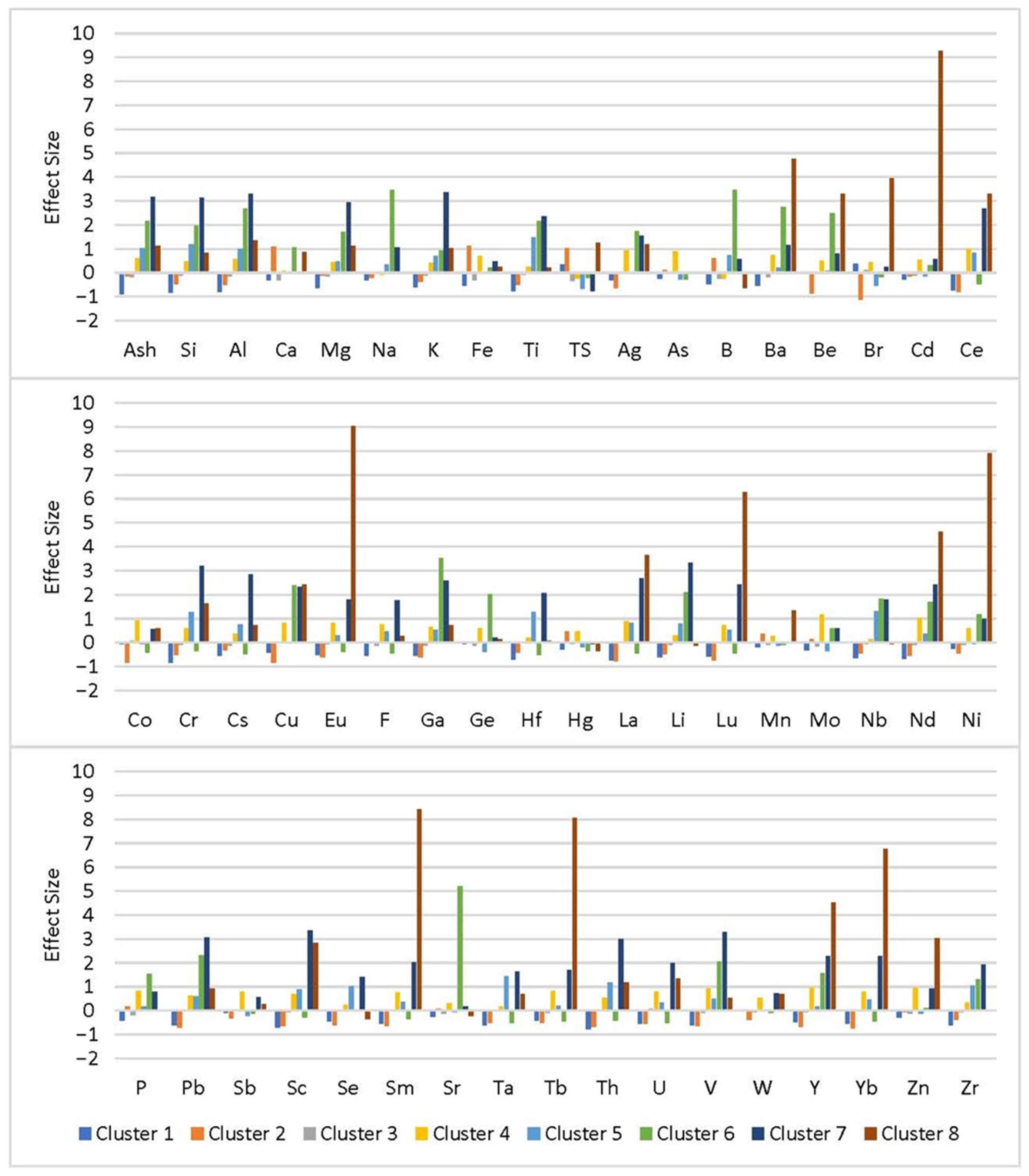
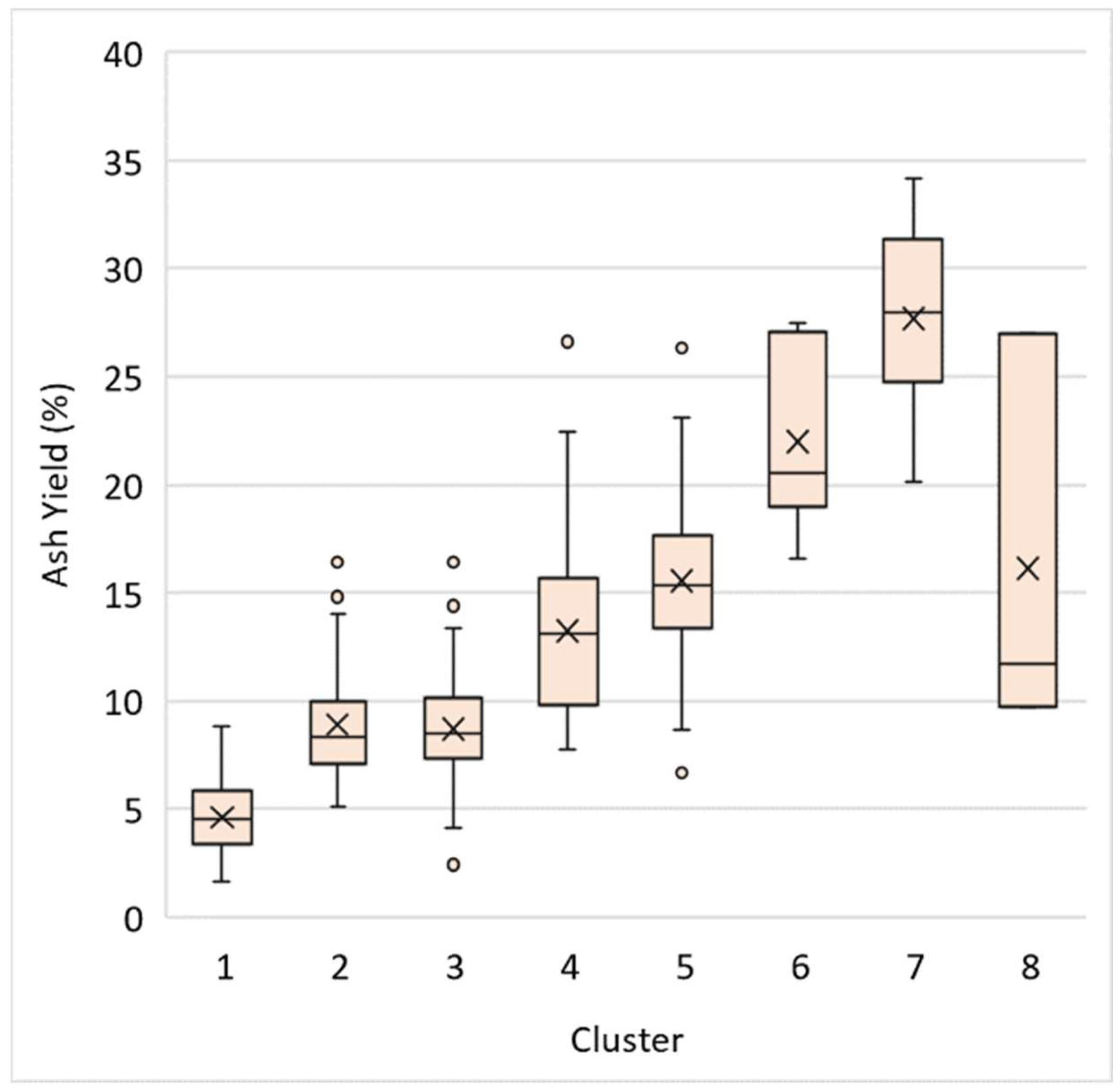
2.4. K-Means Clustering
2.5. REY Economic Assessment
3. Results and Discussion
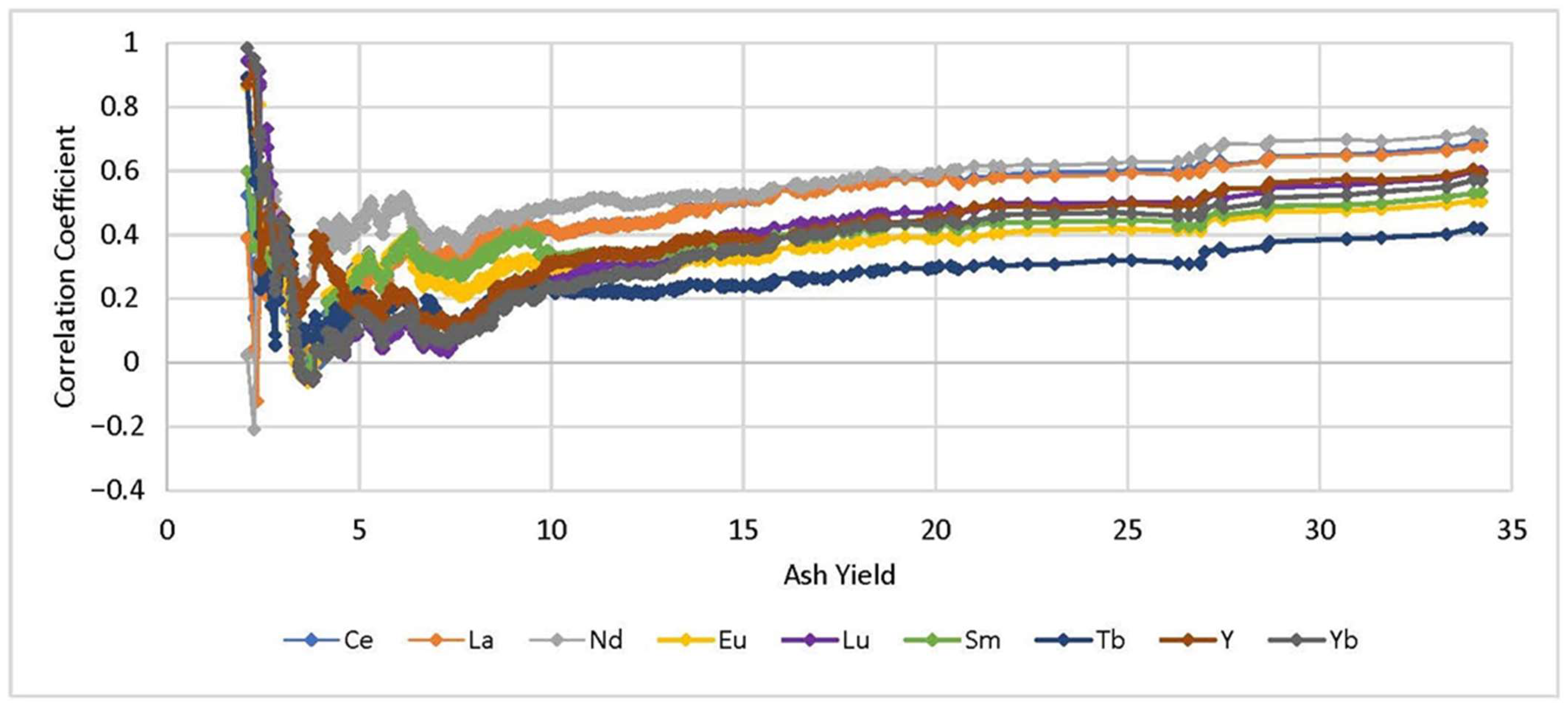
3.1. Pearson Correlation
3.2. Hierarchical Clustering
3.3. K-Means Clustering
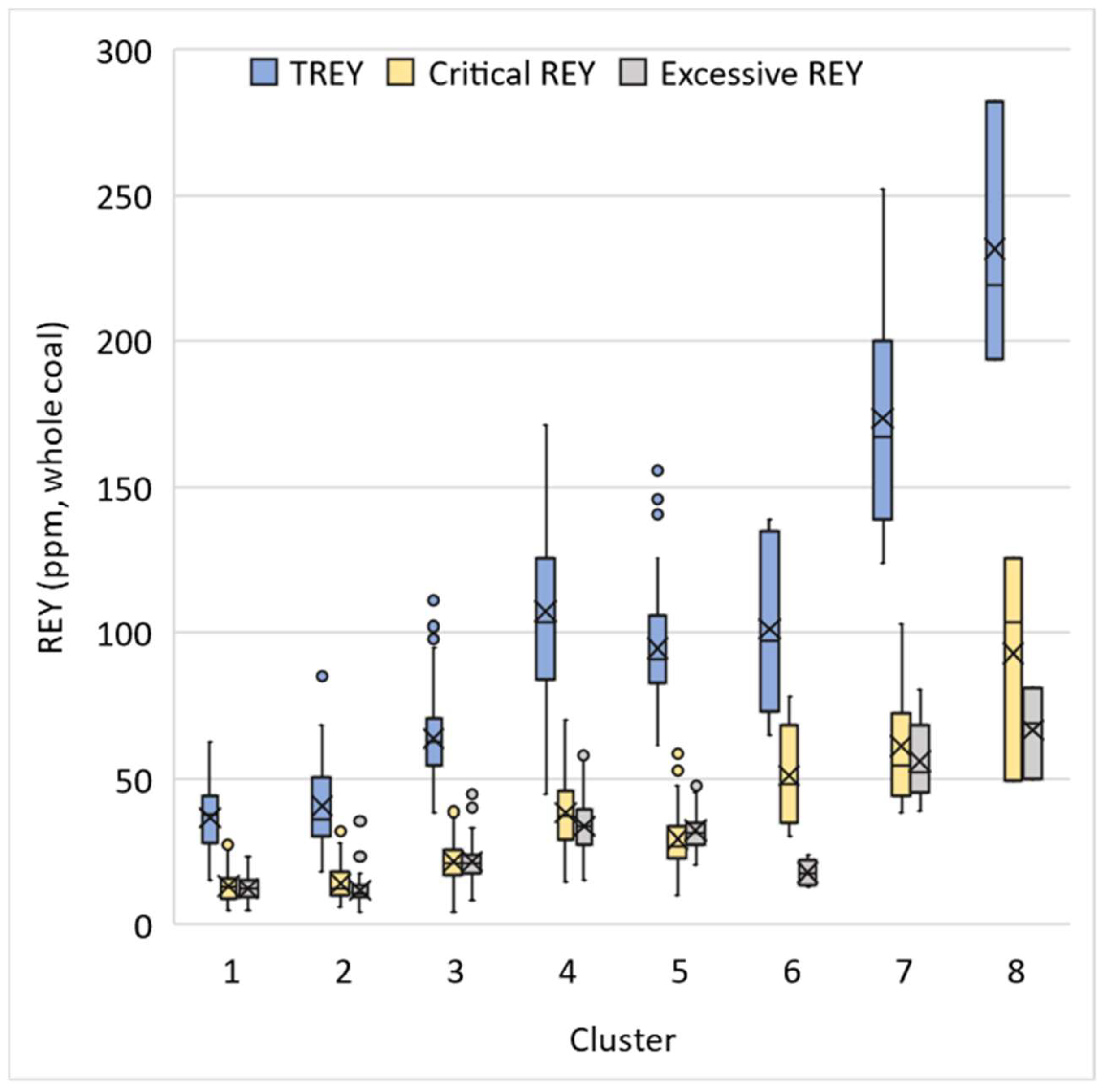
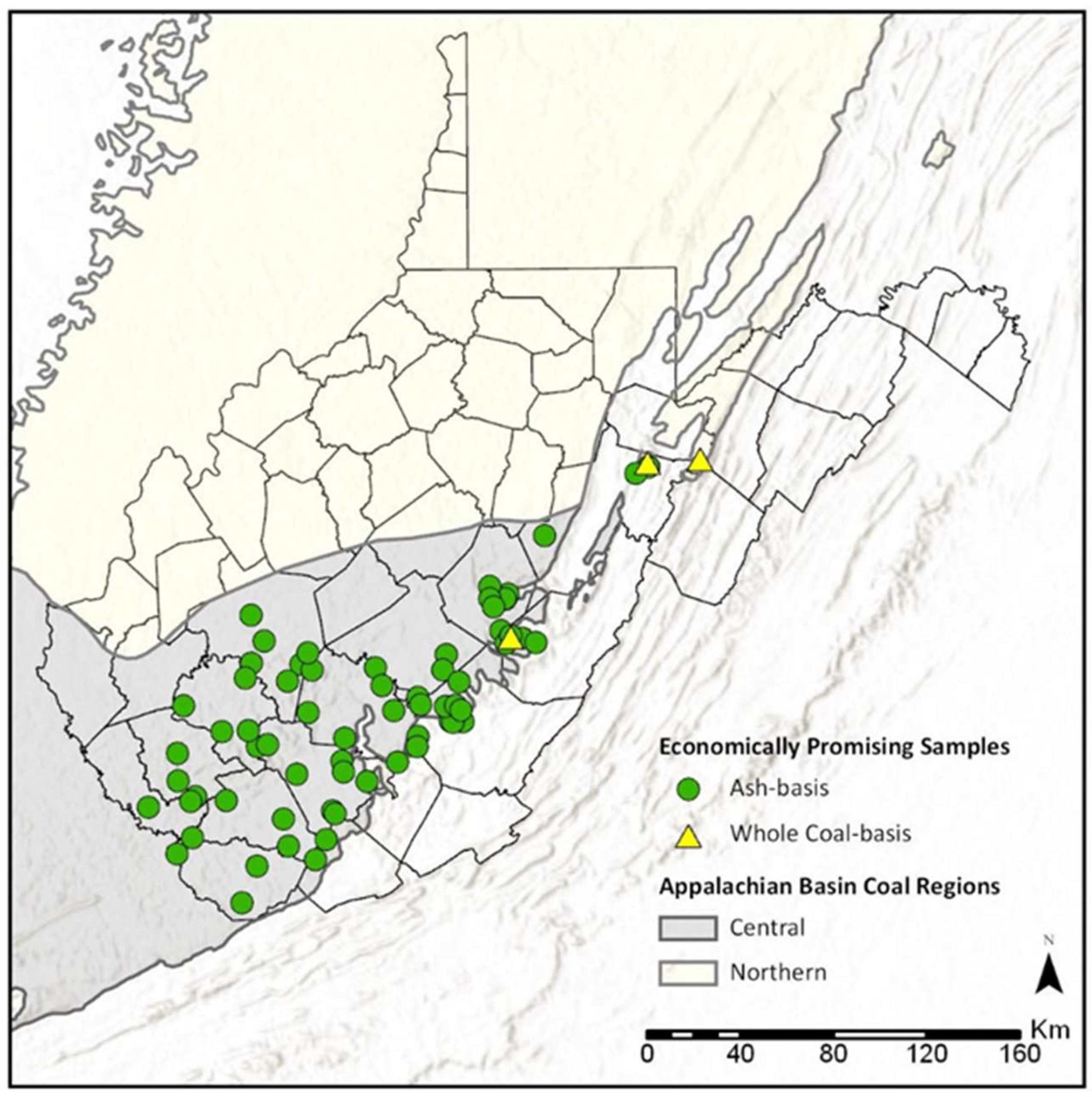
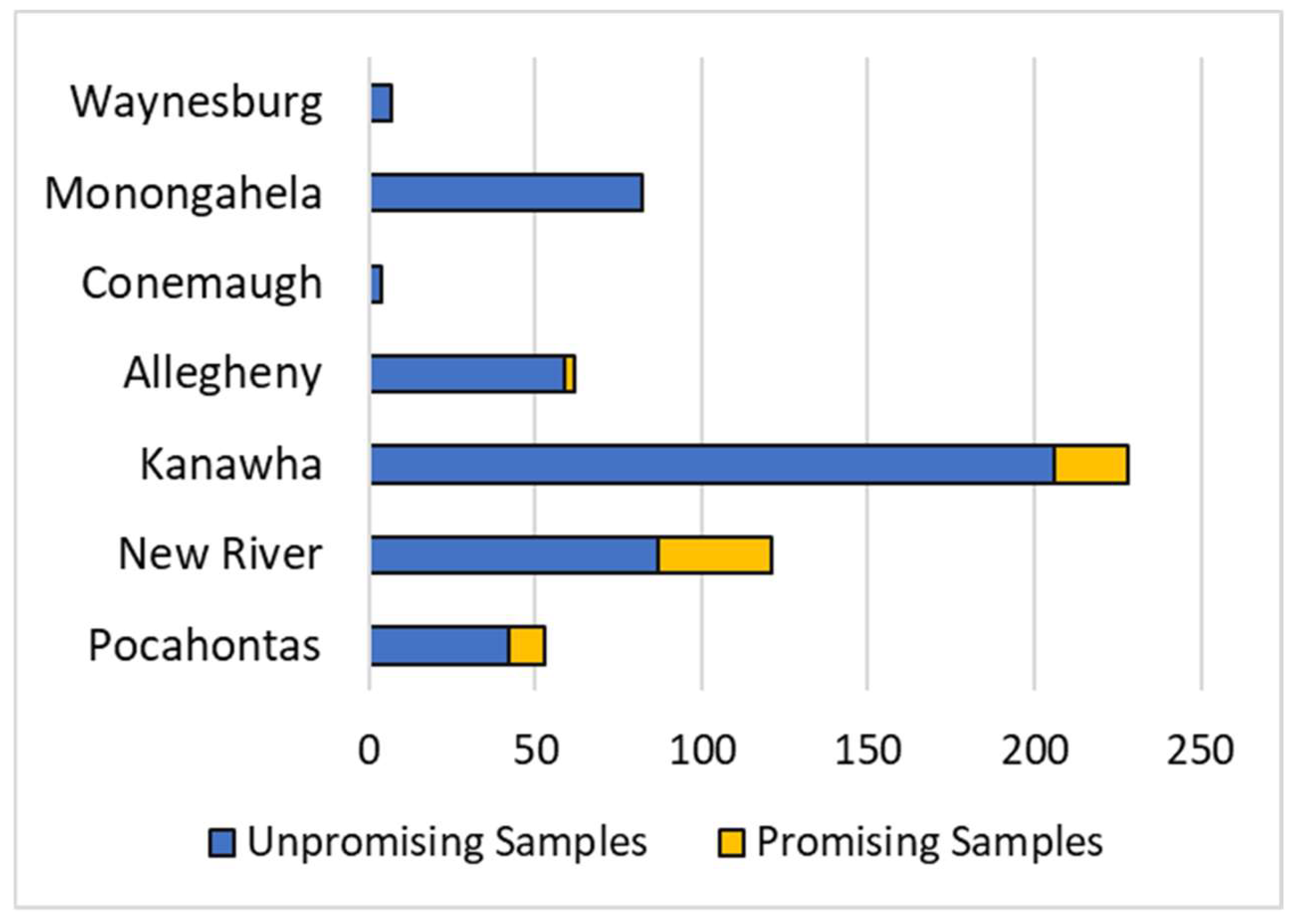
3.4. Economic Assessment of REY in WV Coals
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Williams-Jones, A.E.; Vasyukova, O.V. The Economic Geology of Scandium, the Runt of the Rare Earth Element Litter. Econ. Geol. Bull. Soc. Econ. Geol. 2018, 113, 973–988. [Google Scholar] [CrossRef]
- Dai, S.; Finkelman, R.B. Coal as a promising source of critical elements: Progress and future prospects. Int. J. Coal Geol. 2018, 186, 155–164. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. Rare Earth Elements: A Review of Production, Processing, Recycling, and Associated Environmental Issues; Engineering Technical Support Center, Land Remediation and Pollution Control Division, National Risk Management Research Laboratory, Office of Research and Development: Cincinnati, OH, USA, 2012. [Google Scholar]
- Seredin, V.V.; Dai, S. Coal deposits as potential alternative sources for lanthanides and yttrium. Int. J. Coal Geol. 2012, 94, 67–93. [Google Scholar] [CrossRef]
- Franus, W.; Wiatros-Motyka, M.M.; Wdowin, M. Coal fly ash as a resource for rare earth elements. Environ. Sci. Pollut. Res. 2015, 22, 9464–9474. [Google Scholar] [CrossRef]
- Balaram, V. Rare earth elements: A review of applications, occurrence, exploration, analysis, recycling, and environmental impact. Geosci. Front. 2019, 10, 1285–1303. [Google Scholar] [CrossRef]
- Ayora, C.; Macías, F.; Torres, E.; Lozano, A.; Carrero, S.; Nieto, J.M.; Pérez-López, R.; Fernández-Martínez, A.; Castillo-Michel, H. Recovery of Rare Earth Elements and Yttrium from Passive-Remediation Systems of Acid Mine Drainage. Environ. Sci. Technol. 2016, 50, 8255–8262. [Google Scholar] [CrossRef]
- Vass, C.R.; Noble, A.; Ziemkiewicz, P.F. The Occurrence and Concentration of Rare Earth Elements in Acid Mine Drainage and Treatment Byproducts. Part 2: Regional Survey of Northern and Central Appalachian Coal Basins. Min. Met. Explor. 2019, 36, 917–929. [Google Scholar] [CrossRef]
- Eskanazy, G.; Finkelman, R.B.; Chattarjee, S. Some considerations concerning the use of correlation coefficients and cluster analysis in interpreting coal geochemistry data. Int. J. Coal Geol. 2010, 83, 491–493. [Google Scholar] [CrossRef]
- Xu, N.; Finkelman, R.B.; Xu, C.; Dai, S. What do coal geochemistry statistics really mean? Fuel 2020, 267, 117084. [Google Scholar] [CrossRef]
- Eminagaoglu, M.; Oskay, R.G.; Karayigit, A.I. Evaluation of Elemental Affinities in Coal Using Agglomerative Hierarchical Clustering Algorithm: A Case Study in a Thick and Mineable Coal Seam (KM2) from Soma Basin (W. Turkey). Int. J. Coal Geol. 2022, 259, 104045. [Google Scholar] [CrossRef]
- Xu, N.; Xu, C.; Finkelman, R.B.; Engle, M.A.; Li, Q.; Peng, M.; He, L.; Huang, B.; Yang, Y. Coal Elemental (Compositional) Data Analysis with Hierarchical Clustering Algorithms. Int. J. Coal Geol. 2022, 249, 103892. [Google Scholar] [CrossRef]
- Crowley, S.S.; Stanton, R.W.; Ryer, T.A. The effects of volcanic ash on the maceral and chemical composition of the C coal bed, Emery Coal Field, Utah. Org. Geochem. 1989, 14, 315–331. [Google Scholar] [CrossRef]
- Fiket, Ž.; Medunić, G.; Turk, M.F.; Kniewald, G. Rare Earth Elements in Superhigh-Organic-Sulfur Raša Coal Ash (Croatia). Int. J. Coal Geol. 2018, 194, 1–10. [Google Scholar] [CrossRef]
- Karayigit, A.I.; Yerin, Ü.O.; Oskay, R.G.; Bulut, Y.; Córdoba, P. Enrichment and Distribution of Elements in the Middle Miocene Coal Seams in the Orhaneli Coalfield (NW Turkey). Int. J. Coal Geol. 2021, 247, 103854. [Google Scholar] [CrossRef]
- Mastalerz, M. Rare Earth Elements and Yttrium in Pennsylvanian Coals and Shales in the Eastern Part of the Illinois Basin. Int. J. Coal Geol. 2020, 20, 103620. [Google Scholar] [CrossRef]
- Arbuzov, S.I.; Spears, D.A.; Vergunov, A.V.; Ilenok, S.S.; Mezhibor, A.M.; Ivanov, V.P.; Zarubina, N.A. Geochemistry, Mineralogy and Genesis of Rare Metal (Nb-Ta-Zr-Hf-Y-REE-Ga) Coals of the Seam XI in the South of Kuznetsk Basin, Russia. Ore Geol. Rev. 2019, 113, 103073. [Google Scholar] [CrossRef]
- Dai, S.; Hower, J.C.; Finkelman, R.B.; Graham, I.T.; French, D.; Ward, C.R.; Eskenazy, G.; Wei, Q.; Zhao, L. Organic Associations of Non-Mineral Elements in Coal: A Review. Int. J. Coal Geol. 2020, 218, 103347. [Google Scholar] [CrossRef]
- Dai, S.; Finkelman, R.B.; French, D.; Hower, J.C.; Graham, I.T.; Zhao, F. Modes of Occurrence of Elements in Coal: A Critical Evaluation. Earth-Sci. Rev. 2021, 222, 103815. [Google Scholar] [CrossRef]
- Lin, R.; Bank, T.L.; Roth, E.A.; Granite, E.J.; Soong, Y. Organic and Inorganic Associations of Rare Earth Elements in Central Appalachian Coal. Int. J. Coal Geol. 2017, 179, 295–301. [Google Scholar] [CrossRef]
- Laudal, D.A.; Benson, S.A.; Addleman, R.S.; Palo, D. Leaching Behavior of Rare Earth Elements in Fort Union Lignite Coals of North America. Int. J. Coal Geol. 2018, 191, 112–124. [Google Scholar] [CrossRef]
- Fernández-Caliani, J.C.; Grantcharova, M.M. Enrichment and Fractionation of Rare Earth Elements in an Estuarine Marsh Soil Receiving Acid Discharges from Legacy Sulfide Mine Wastes. Soil Syst. 2021, 5, 66. [Google Scholar] [CrossRef]
- Dai, S.; Graham, I.T.; Ward, C.R. A Review of Anomalous Rare Earth Elements and Yttrium in Coal. Int. J. Coal Geol. 2016, 159, 82–95. [Google Scholar] [CrossRef]
- Seredin, V.V. A New Method for Primary Evaluation of the Outlook for Rare Earth Element Ores. Geol. Ore Depos. 2010, 52, 428–433. [Google Scholar] [CrossRef]
- Dulong, F.T.; Cecil, C.B. Stratigraphic Variation in Bulk Sample Mineralogy of Pennsylvanian Underclays from the Central Appalachian Basin. In Paleoclimate Controls on Carboniferous Sedimentation and Cyclic Stratigraphy in the Appalachian Basin; US Geological Survey Open File Report; US Geological Survey: Williamsport, PA, USA, 1998. [Google Scholar]
- Palmer, C.A.; Oman, C.L.; Park, A.J.; Luppens, J.A. The US Geological Survey Coal Quality (COALQUAL) Database Version 3.0; US Geological Survey Open-file Report; US Geological Survey: Reston, VA, USA, 2015. [Google Scholar]
- Connor, J.J.; Keith, J.R.; Anderson, B.M. Trace-Metal Variation in Soils and Sagebrush in the Powder River Basin, Wyoming and Montana. J. Res. US Geol. Surv. 1976, 4, 49–59. [Google Scholar]
- Gluskoter, H.J.; Ruch, R.R.; Miller, W.G.; Cahill, R.A.; Dreher, G.B.; Kuhn, J.K. Trace Elements in Coal: Occurrence and Distribution; Illinois State Geological Survey: Champaign, IL, USA, 1977. [Google Scholar]
- Lin, R.; Soong, Y.; Granite, E.J. Evaluation of Trace Elements in U.S. Coals Using the USGS COALQUAL Database Version 3.0. Part I: Rare Earth Elements and Yttrium (REY). Int. J. Coal Geol. 2018, 192, 1–13. [Google Scholar] [CrossRef]
- Kynčlová, P.; Hron, K.; Filzmoser, P. Correlation Between Compositional Parts Based on Symmetric Balances. Math. Geosci. 2017, 49, 777–796. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Tang, Y.; Yue, M.; Hao, L. Concentration and Distribution of Elements in Late Permian Coals from Western Guizhou Province, China. Int. J. Coal Geol. 2005, 61, 119–137. [Google Scholar] [CrossRef]
- Spears, D. Role of Clay Minerals in UK Coal Combustion. Appl. Clay Sci. 2000, 16, 87–95. [Google Scholar] [CrossRef]
- Bryan, R.C.; Richers, D.; Andersen, H.T.; Gary, T. Assessment of Rare Earth Elemental Contents in Selected United States Coal-Basins; Document; Leonardo Technologies Inc.: St. Clairsville, OH, USA, 2015. [Google Scholar]
- Bragg, L.J.; Oman, J.K.; Tewalt, S.J.; Oman, C.L.; Rega, N.H.; Washington, P.M.; Finkelman, R.B. US Geological Survey Coal Quality (COALQUAL) Database: Version 2.0; US Geological Survey Open-file Report; US Geological Survey: Reston, VA, USA, 1998. [Google Scholar]
- Spiro, C.L.; Wong, J.; Maylotte, D.H.; Lamson, S.H.; Lytle, F.W.; Greegor, R.B. Forms of Potassium in Coal and Its Combustion Products. Fuel 1986, 65, 327–336. [Google Scholar] [CrossRef]
- Steinmetz, G.L.; Mohan, M.S.; Zingaro, R.A. Characterization of Titanium in United States Coals. Energy Fuels 1988, 2, 684–692. [Google Scholar] [CrossRef]
- Finkelman, R.B. Modes of Occurrence of Trace Elements in Coal; US Geological Survey Open-File Report; US Geological Survey: Reston, VA, USA, 1981. [Google Scholar]
- Palmer, C.A.; Filby, R.H. Distribution of Trace Elements in Coal from the Powhatan No. 6 Mine, Ohio. Fuel 1984, 63, 318–328. [Google Scholar] [CrossRef]
- Finkelman, R.B.; Palmer, C.A.; Wang, P. Quantification of the Modes of Occurrence of 42 Elements in Coal. Int. J. Coal Geol. 2018, 185, 138–160. [Google Scholar] [CrossRef]
- Finkelman, R.B.; Dai, S.; French, D. The Importance of Minerals in Coal as the Hosts of Chemical Elements: A Review. Int. J. Coal Geol. 2019, 212, 103251. [Google Scholar] [CrossRef]
- Sutcu, E.C.; Karayigit, A.I. Mineral Matter, Major and Trace Element Content of the Afşin–Elbistan Coals, Kahramanmaraş, Turkey. Int. J. Coal Geol. 2015, 144–145, 111–129. [Google Scholar] [CrossRef]
- Querol, X.; Klika, Z.; Weiss, Z.; Finkelman, R.; Alastuey, A.; Juan, R.; López-Soler, A.; Plana, F.; Kolker, A.; Chenery, S.R. Determination of Element Affinities by Density Fractionation of Bulk Coal Samples. Fuel 2001, 80, 83–96. [Google Scholar] [CrossRef]
- Ruppert, L.; Finkelman, R.; Boti, E.; Milosavljevic, M.; Tewalt, S.; Simon, N.; Dulong, F. Origin and Significance of High Nickel and Chromium Concentrations in Pliocene Lignite of the Kosovo Basin, Serbia. Int. J. Coal Geol. 1996, 29, 235–258. [Google Scholar] [CrossRef]
- Arbuzov, S.I.; Volostnov, A.V.; Mezhibor, A.M.; Rybalko, V.I.; Ilenok, S.S. Scandium (Sc) Geochemistry in Coals (Siberia, Russian Far East, Mongolia, Kazakhstan, and Iran). Int. J. Coal Geol. 2014, 125, 22–35. [Google Scholar] [CrossRef]
- Montross, S.N.; Yang, J.; Britton, J.; McKoy, M.; Verba, C. Leaching of Rare Earth Elements from Central Appalachian Coal Seam Underclays. Minerals 2020, 10, 577. [Google Scholar] [CrossRef]
- Davis, B.A.; Rodrigues, S.; Esterle, J.S.; Rintoul, L.; Duxbury, A.J.; Golding, S.D. In Situ Techniques for Classifying Apatite in Coal. Int. J. Coal Geol. 2020, 230, 103572. [Google Scholar] [CrossRef]
- Yang, J.; Montross, S.; Verba, C.; Britton, J.; Stuckman, M.; Lopano, C. Microanalytical Approaches to Characterizing REE in Appalachian Basin Underclays. Minerals 2020, 10, 546. [Google Scholar] [CrossRef]
- Spiro, B.F.; Liu, J.; Dai, S.; Zeng, R.; Large, D.; French, D. Marine Derived 87Sr/86Sr in Coal, A New Key to Geochronology and Palaeoenvironment: Elucidation of the India-Eurasia and China-Indochina Collisions in Yunnan, China. Int. J. Coal Geol. 2019, 215, 103304. [Google Scholar] [CrossRef]
- Hower, J.C.; Ruppert, L.F.; Eble, C.F. Lanthanide, Yttrium, and Zirconium Anomalies in the Fire Clay Coal Bed, Eastern Kentucky. Int. J. Coal Geol. 1999, 39, 141–153. [Google Scholar] [CrossRef]
- Hower, J.C.; Eble, C.F.; Dai, S.; Belkin, H.E. Distribution of Rare Earth Elements in Eastern Kentucky Coals: Indicators of Multiple Modes of Enrichment? Int. J. Coal Geol. 2016, 160–161, 73–81. [Google Scholar] [CrossRef]
- Schatzel, S.J.; Stewart, B.W. A Provenance Study of Mineral Matter in Coal from Appalachian Basin Coal Mining Regions and Implications Regarding the Respirable Health of Underground Coal Workers: A Geochemical and Nd Isotope Investigation. Int. J. Coal Geol. 2012, 94, 123–136. [Google Scholar] [CrossRef]
- Messinger, T. Water-Quality Assessment of the Kanawha-New River Basin, West Virginia, Virginia, and North Carolin-Review of Water-Quality Literature Through 1996; US Geological Survey Water-Resources Investigations Report; US Geological Survey: Reston, VA, USA, 1997. [Google Scholar]
- McNutt, R.H.; Frape, S.K.; Dollar, P. A Strontium, Oxygen and Hydrogen Isotopic Composition of Brines, Michigan and Appalachian Basins, Ontario and Michigan. Appl. Geochem. 1987, 2, 495–505. [Google Scholar] [CrossRef]
- Beaton, A.P.; Goodarzi, F.; Potter, J. The Petrography, Mineralogy and Geochemistry of a Paleocene Lignite from Southern Saskatchewan, Canada. Int. J. Coal Geol. 1991, 17, 117–148. [Google Scholar] [CrossRef]
- Boyd, R.J. The Partitioning Behaviour of Boron from Tourmaline during Ashing of Coal. Int. J. Coal Geol. 2002, 53, 43–54. [Google Scholar] [CrossRef]
- Karayigit, A.I.; Atalay, M.; Oskay, R.G.; Córdoba, P.; Querol, X.; Bulut, Y. Variations in Elemental and Mineralogical Compositions of Late Oligocene, Early and Middle Miocene Coal Seams in the Kale-Tavas Molasse Sub-Basin, SW Turkey. Int. J. Coal Geol. 2020, 218, 103366. [Google Scholar] [CrossRef]
- Çelik, Y.; Karayiğit, A.İ.; Querol, X.; Oskay, R.G.; Mastalerz, M.; Kayseri Özer, M.S. Coal Characteristics, Palynology, and Palaeoenvironmental Interpretation of the Yeniköy Coal of Late Oligocene Age in the Thrace Basin (NW Turkey). Int. J. Coal Geol. 2017, 181, 103–123. [Google Scholar] [CrossRef]
- Goodarzi, F.; Swaine, D.J. Paleoenvironmental and Environmental Implications of the Boron Content of Coals; Geological Survey of Canada: Ottawa, ON, Canada, 1994. [Google Scholar]
- Bau, M. Controls on the Fractionation of Isovalent Trace Elements in Magmatic and Aqueous Systems: Evidence from Y/Ho, Zr/Hf, and Lanthanide Tetrad Effect. Contrib. Mineral. Petrol. 1996, 123, 323–333. [Google Scholar] [CrossRef]
- Mastalerz, M.; Drobniak, A. Gallium and Germanium in Selected Indiana Coals. Int. J. Coal Geol. 2012, 94, 302–313. [Google Scholar] [CrossRef]
- Bladh, K.W. The Formation of Goethite, Jarosite, and Alunite during the Weathering of Sulfide-Bearing Felsic Rocks. Econ. Geol. Bull. Soc. Econ. Geol. 1982, 77, 176–184. [Google Scholar] [CrossRef]
- Grawunder, A.; Merten, D.; Büchel, G. Origin of Middle Rare Earth Element Enrichment in Acid Mine Drainage-Impacted Areas. Environ. Sci. Pollut. Res. 2014, 21, 6812–6823. [Google Scholar] [CrossRef]
- Merovich, G.T.; Stiles, J.M.; Petty, J.T.; Ziemkiewicz, P.F.; Fulton, J.B. Water Chemistry-Based Classification of Streams and Implications for Restoring Mined Appalachian Watersheds. Environ. Toxicol. Chem. 2007, 26, 1361–1369. [Google Scholar] [CrossRef]
- Hedin, B.C.; Hedin, R.S.; Capo, R.C.; Stewart, B.W. Critical Metal Recovery Potential of Appalachian Acid Mine Drainage Treatment Solids. Int. J. Coal Geol. 2020, 231, 103610. [Google Scholar] [CrossRef]
- Cecil, C.B. Paleoclimate Controls on Carboniferous Sedimentation and Cyclic Stratigraphy in the Appalachian Basin; US Geological Survey Open File Report; US Geological Survey: Knoxville, TN, USA, 1998. [Google Scholar]
- Greber, N.D.; Mäder, U.; Nägler, T.F. Experimental Dissolution of Molybdenum-Sulphides at Low Oxygen Concentrations: A First-Order Approximation of Late Archean Atmospheric Conditions. Earth Space Sci. 2015, 2, 173–180. [Google Scholar] [CrossRef]
- Diehl, S.F.; Goldhaber, M.B.; Koenig, A.E.; Lowers, H.A.; Ruppert, L.F. Distribution of Arsenic, Selenium, and Other Trace Elements in High Pyrite Appalachian Coals: Evidence for Multiple Episodes of Pyrite Formation. Int. J. Coal Geol. 2012, 94, 238. [Google Scholar] [CrossRef]
- West Virginia Geological and Economic Survey Selenium (Se). Available online: http://www.wvgs.wvnet.edu/www/datastat/te/sehome.htm (accessed on 8 May 2022).
- Yudovich, Y.E.; Ketris, M.P. Selenium in Coal: A Review. Int. J. Coal Geol. 2006, 67, 112–126. [Google Scholar] [CrossRef]
- Belogub, E.V.; Ayupova, N.R.; Novoselov, K.A.; Blinov, I.A.; Krivovichev, V.G.; Charykova, M.V. Se Minerals in the Continental and Submarine Oxidation Zones of the South Urals Volcanogenic-Hosted Massive Sulfide Deposits: A Review. Ore Geol. Rev. 2020, 122, 103500. [Google Scholar] [CrossRef]
- Borst, A.M.; Smith, M.P.; Finch, A.A.; Estrade, G.; Villanova-de-Benavent, C.; Nason, P.; Marquis, E.; Horsburgh, N.J.; Goodenough, K.M.; Xu, C.; et al. Adsorption of Rare Earth Elements in Regolith-Hosted Clay Deposits. Nat. Commun. 2020, 11, 4386. [Google Scholar] [CrossRef]
- Fuller, A.J.; Shaw, S.; Ward, M.B.; Haigh, S.J.; Mosselmans, J.F.W.; Peacock, C.L.; Stackhouse, S.; Dent, A.J.; Trivedi, D.; Burke, I.T. Caesium Incorporation and Retention in Illite Interlayers. Appl. Clay Sci. 2015, 108, 128–134. [Google Scholar] [CrossRef]
- Lukman, S.; Essa, M.H.; Bukhari, A.; Mu’azu, N.D.; Basheer, C. Adsorption and Desorption of Heavy Metals onto Natural Clay Material: Influence of Initial PH. J. Environ. Sci. Technol. 2013, 6, 1–15. [Google Scholar] [CrossRef]
- Rozelle, P.L.; Khadilkar, A.B.; Pulati, N.; Soundarrajan, N.; Klima, M.S.; Mosser, M.M.; Miller, C.E.; Pisupati, S.V. A Study on Removal of Rare Earth Elements from US Coal Byproducts by Ion Exchange. Metall. Mater. Trans. E Mater. Energy Syst. 2016, 3, 6–17. [Google Scholar]
- US Department of Energy. Report on Rare Earth Elements from Coal and Coal Byproducts; US Department of Energy: Washington, DC, USA, 2017. [Google Scholar]
- American Coal Ash Association. 2019 Coal Combustion Product (CCP) Production & Use Survey Report. 2019. Available online: https://acaa-usa.org/wp-content/uploads/coal-combustion-products-use/2019-Survey-Results.pdf (accessed on 5 June 2022).
- Bagdonas, D.A.; Enriquez, A.J.; Coddington, K.A.; Finnoff, D.C.; McLaughlin, J.F.; Bazilian, M.D.; Phillips, E.H.; McLing, T.L. Rare Earth Element Resource Evaluation of Coal Byproducts: A Case Study from the Powder River Basin, Wyoming. Renew. Sustain. Energy Rev. 2022, 158, 112148. [Google Scholar] [CrossRef]
| Data below Detection Limit | Data above Detection Limit | Adjusted Data | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Element | No. of Samples | Percent of Data | Min. | Mean | Max. | S.D. | No. of Samples | Min. | Mean | Max. | S.D. | Q Factor | Min. | Mean | Max. | S.D. |
| La | 0 | 0.00 | n/a | n/a | n/a | n/a | 569 | 1.04 | 10.15 | 45.40 | 6.54 | n/a | 1.04 | 10.15 | 45.40 | 6.54 |
| Ce | 0 | 0.00 | n/a | n/a | n/a | n/a | 569 | 3.04 | 18.56 | 75.00 | 10.98 | n/a | 3.04 | 18.56 | 75.00 | 10.98 |
| Pr | 496 | 87.17 | 1.10 | 8.29 | 43.00 | 5.83 | 73 | 0.43 | 3.68 | 28.00 | 4.06 | 0.30 | 0.43 | 3.57 | 28 | 2.74 |
| Nd | 134 | 23.55 | 1.60 | 7.34 | 30.00 | 4.56 | 435 | 1.18 | 10.20 | 62.10 | 7.37 | 0.70 | 1.18 | 9.53 | 62.10 | 6.92 |
| Sm | 0 | 0.00 | n/a | n/a | n/a | n/a | 569 | 0.10 | 1.70 | 14.30 | 1.28 | n/a | 0.10 | 1.70 | 14.30 | 1.28 |
| Eu | 2 | 0.35 | 0.51 | 0.61 | 0.71 | 0.14 | 567 | 0.10 | 0.37 | 3.22 | 0.28 | 0.70 | 0.10 | 0.37 | 3.22 | 0.28 |
| Gd | 214 | 37.61 | 0.37 | 1.77 | 8.90 | 1.27 | 355 | 0.28 | 1.72 | 13.40 | 1.45 | 0.70 | 0.28 | 1.74 | 13.40 | 1.38 |
| Tb | 56 | 9.84 | 0.26 | 0.93 | 6.10 | 0.94 | 513 | 0.02 | 0.30 | 3.06 | 0.24 | 0.70 | 0.02 | 0.36 | 6.10 | 0.41 |
| Dy | 488 | 85.76 | 0.23 | 3.22 | 20.00 | 2.55 | 81 | 0.45 | 2.45 | 9.98 | 1.74 | 0.50 | 0.16 | 2.32 | 14.29 | 1.81 |
| Y | 0 | 0.00 | n/a | n/a | n/a | n/a | 569 | 0.92 | 7.84 | 44.30 | 5.52 | n/a | 0.92 | 7.84 | 44.30 | 5.52 |
| Ho | 530 | 93.15 | 0.12 | 0.86 | 8.00 | 0.74 | 39 | 0.19 | 0.64 | 2.24 | 0.48 | 0.50 | 0.09 | 0.61 | 5.71 | 0.52 |
| Er | 377 | 66.26 | 0.21 | 1.27 | 6.20 | 0.81 | 192 | 0.28 | 1.26 | 5.13 | 0.84 | 0.70 | 0.21 | 1.27 | 6.20 | 0.82 |
| Tm | 561 | 98.59 | 0.07 | 0.52 | 3.00 | 0.36 | 8 | 0.10 | 0.36 | 0.93 | 0.25 | 0.50 | 0.05 | 0.37 | 2.14 | 0.26 |
| Yb | 1 | 0.18 | 0.42 | 0.42 | 0.42 | n/a | 568 | 0.19 | 0.87 | 5.00 | 0.56 | 0.70 | 0.19 | 0.87 | 5.00 | 0.56 |
| Lu | 31 | 5.45 | 0.08 | 0.35 | 0.71 | 0.17 | 538 | 0.01 | 0.13 | 0.80 | 0.09 | 0.70 | 0.01 | 0.15 | 0.80 | 0.10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yesenchak, R.; Sharma, S.; Maxwell, A.E. Modes of Occurrence, Elemental Relationships, and Economic Viability of Rare Earth Elements in West Virginia Coals: A Statistical Approach. Minerals 2022, 12, 1060. https://doi.org/10.3390/min12081060
Yesenchak R, Sharma S, Maxwell AE. Modes of Occurrence, Elemental Relationships, and Economic Viability of Rare Earth Elements in West Virginia Coals: A Statistical Approach. Minerals. 2022; 12(8):1060. https://doi.org/10.3390/min12081060
Chicago/Turabian StyleYesenchak, Rachel, Shikha Sharma, and Aaron E. Maxwell. 2022. "Modes of Occurrence, Elemental Relationships, and Economic Viability of Rare Earth Elements in West Virginia Coals: A Statistical Approach" Minerals 12, no. 8: 1060. https://doi.org/10.3390/min12081060
APA StyleYesenchak, R., Sharma, S., & Maxwell, A. E. (2022). Modes of Occurrence, Elemental Relationships, and Economic Viability of Rare Earth Elements in West Virginia Coals: A Statistical Approach. Minerals, 12(8), 1060. https://doi.org/10.3390/min12081060







