1. Introduction
Spent nuclear fuel (SNF) generates significant heat via radioactive decay. After a period of aboveground cooling, a deep geological repository (DGR) is considered the only suitable way to dispose of such high-level radioactive waste. For many DGR concepts, the temperature on the interface between disposal canisters and bentonite buffer is typically designed to be lower than 100 °C. If a higher temperature is allowed (beyond 100 °C), the cooling and storage period could shorten, or more SNF assemblies could be loaded in the disposal canister. Both would positively affect the economy of radioactive waste disposal, especially when considering size optimisation [
1]. However, possible changes in DGR designs must be justified, by proving that the performance targets for the engineered barrier safety functions are fulfilled, even at an elevated temperature, and confirming that the repository ensures post-closure radiation safety.
The thermal criterion of not exceeding a buffer peak temperature of 100 °C was chosen to avoid bentonite alteration [
2]. Alteration here refers to mineral transformations of montmorillonite, leading to low- or non-swelling clay structures, which may not meet the buffer safety criteria for swelling pressure and hydraulic conductivity. Several possible processes, such as atomic substitutions (eventually, iron reduction/oxidation) in the mineral structure, layer charge redistribution, balanced by the compensating cations and congruent dissolution, are involved in the montmorillonite transformation [
2]. The main alteration processes involve the transformation to illite/montmorillonite mixed layers, illite, beidellite, saponite, or chlorite [
2,
3].
Moreover, bentonite soluble components dissolve under hydrothermal conditions and the solutes may interact with silica released by the montmorillonite transformation, resulting in precipitation of new Si phases (so-called cementation), also contributing to a reduction of buffer plasticity and an increase of hydraulic conductivity [
2,
3]. Cementation can also be caused by non-Si compounds (e.g., sulphate and carbonate minerals), showing increased stability at higher temperatures [
3].
Based on the critical review of the performance of the bentonite barrier, at temperatures beyond 100 °C, Wersin et al. [
3] supported the idea of raising the thermal criterion to 120 °C; they identified higher thermal stability of bentonite under dry conditions (up to 300 °C) and noted the scarce data on hydraulic and mechanical properties of heat-exposed bentonites. The highest temperature yet applied in-situ was in the ABM5 experiment at Äspö Hard Rock Laboratory (Sweden). Temperatures up to 250 °C were achieved at the steel heater interface with compacted bentonites [
1,
4]. No significantly different chemical and mineralogical reactions were observed compared to the previous ABM tests or similar tests performed in the temperature range of 90–130 °C. In this test, the extent of magnesium enrichment, in the vicinity of the heater, observed in many large-scale tests, was small [
4]. No data on the characterisation of bentonites from the ABM5 experiment from the geotechnical point of view is yet available. Complete ABM experiments provided geotechnical data for MX-80, Asha 505, Deponit CAN, and Friedland clay. Slightly lower swelling pressure was determined for heat-exposed materials Asha 505, Deponit CAN, and Friedland clay. No changes in hydraulic conductivities between the above reference and in-situ exposed materials were observed [
5,
6,
7].
The influence of higher temperatures on bentonite performance and the lack of data from the thermo-hydro-mechanical (THM) behaviour of clay-based materials led to identifying this topic as a high priority subject within the framework of the EURAD project—WP7 HITEC. In general, two approaches are used to investigate thermally induced effects on clay-based materials.
The first approach examined HM properties at different temperatures. Geotechnical parameters of water-saturated compacted bentonites (FEBEX, MX-80, and Korean Ca-bentonite) were determined at temperatures ranging from 20 to 150 °C [
8,
9,
10,
11,
12]. Decreasing swelling pressure and suction pressure and increasing hydraulic conductivity with temperature were assigned to the temperature-dependent water properties and physicochemical interactions of water at the microscopic level [
9]. However, once corrected for temperature-dependent water density and viscosity, the intrinsic permeability was not significantly temperature dependent [
11,
12]. This approach demonstrates buffer safety functions (namely swelling pressure and hydraulic conductivity) at all thermal stages within the DGR evolution.
In the second approach, the material property changes of preheated material were analysed. The material was mainly provided from in-situ experiments, which were designed to simulate the heat released from waste canisters containing radioactive waste. MX-80 bentonite, the “reference” one, and the one exposed to the temperature of 130 °C at the copper heater in the form of parcel samples trimmed to the experimental cells, or re-compacted parcel samples to the original bulk density up to 2000 kg/m
3, was subjected to the determination of hydraulic conductivity (among other determinations). Slightly lower hydraulic conductivity in the trimmed samples in comparison to the re-compacted ones was observed. However, no significant difference between samples from different positions (warm, cool) in the parcel was observed; therefore, the decrease could not be related to temperature gradients [
13]. A mixture of FoCa clay with 35 wt.% sand and 5 wt.% graphite was used in OPHELIE mock-up experiment where a temperature up to 140 °C was maintained for 4.5 years [
14]. Slightly higher hydraulic conductivity of exposed material was determined. The authors explain this fact with thermally induced changes of the sample microstructure. An increase in intermediate pore sizes within the samples with temperature, which caused an increase in water and the cross-sectional area available for water to flow, was indicated by mercury intrusion porosimetry. Moreover, water retention curves were determined. Thermally loaded samples showed lower water retention capacity than the initial ones for the lower suction levels. For suction pressure over 150 MPa, no difference was observed. Thermally loaded samples showed less pronounced hysteresis than the initial ones, which was also attributed to the microstructural changes mentioned above [
14].
Natural clay materials (e.g., bentonites) also represent an important source of indigenous microorganisms [
5,
15]. The microbial activity might negatively affect the long-term safety of DGR as it can result in microbiologically influenced corrosion of the canister and possibly alter the bentonite mineralogy [
16,
17]. Microbial survival and growth are generally inhibited by thermal and radiation performance after deposition of the canister [
17]. However, bacterial endospores that are microbial survival forms with reduced water content, and undetectable metabolic activities, can tolerate adverse environmental conditions, such as desiccation, wet and dry heat (above 100 °C), and UV and gamma irradiation for extended periods [
18,
19]. Several studies in the bentonite environment have also demonstrated microbial survivability. Masurat et al. [
20,
21] found a loss of sulphate-reducing bacteria (SRB) viability after loading MX-80 bentonite at 120 °C for 20 h. However, another experiment with even more prolonged heating for one week failed to eradicate SRB [
22]. Similarly, the presence of indigenous SRB, which survived heat loading of Boom Clay at 120 °C for 48 h, was reported [
23]. Eleven compacted bentonites were exposed to groundwater saturation and high temperatures (90–130 °C) in the in-situ experiment—Alternative Buffer Material at the Äspö Hard Rock Laboratory—for approximately one year. Microbial analyses of three bentonites within the first test package showed a detrimental effect of heat on bacterial activity. The bacterial growth was scarce; only mesophilic aerobic heterotrophs in the range of 10
2–10
3 g
−1 wet weight were cultivated [
5].
It can be presumed that, during the early stage of disposal, when the buffer is exposed to an elevated temperature induced by the radioactive waste and, concurrently, is not yet saturated by the groundwater, the microbial activity would be insignificant. However, the safety functions of bentonite may be affected at this stage. Therefore, it is crucial to identify potential changes in the safety functions of bentonite when exposed to elevated temperatures. This work explores Czech Mg/Ca bentonite before and after medium-term thermal loading at 150 °C in a powder state. Several techniques and methods were chosen for geochemical and geotechnical characterisation (e.g., XRD, TA-EGA, aqueous leachates, CEC, SSA, free swelling, saturated hydraulic conductivity, WRC). In addition, microbiological methods were used to investigate bacterial survivability in thermally loaded bentonite and the ability to regenerate from spores in favourable cultivation conditions.
2. Materials and Methods
2.1. Bentonite Powder
It is anticipated that Czech bentonites will be used in the construction of engineered barriers in the future Czech DGR project [
24]. For this study, a commercial bentonite product originating from the Černý vrch deposit (Keramost PLC, Most, Czech Republic) was chosen. Bentonite Černý vrch (BCV) is dominated by montmorillonite, with prevailing divalent exchangeable cations (mainly magnesium). Basic characteristics of the BCV bentonite were summarised in an HITEC SotA report [
1].
Since December 2019, approximately 10 kg of bentonite powder in open stainless-steel vessels have been exposed to a 150 °C temperature in a heating chamber (Binder, Tuttlingen, Germany). This set-up was designed to simulate an early post-closure state of the DGR; when radioactive decay generates a significant amount of heat, oxygen is still available, and the buffer is not saturated by the groundwater. Overall, thermal loading of bentonite powder was planned for two years. This paper deals with the characterisation of the initial BCV bentonite (denoted as BCV_IN) and two subsamples taken from the thermally loaded bentonite after a year and a half (denoted as BCV_0.5_y and BCV_1.0_y, respectively).
The first sampling revealed a colour change of bentonite on the surface, from light brown to dark brown (see
Figure 1). The thin surface layer was sampled as a separate sample. The same procedure was then followed for the second sampling, after one year. This paper only focuses on the characterisation of the bentonite powder beneath the thin surface layer. This sample defines the barrier properties and allows evaluating the impact of the thermal loading. The effect of the potential thermally induced surface buffer layer near the containers on the DGR is expected to be negligible (regarding long-term safety) due to the minimal thickness. However, future research should focus on the properties of the thermally induced surface layer and the extent to which it will form in a compacted state.
2.2. Geochemical Characterisation
Bentonite powder samples were subjected to various determinations, providing insight into geochemical properties, such as the composition of major minerals, the identification of gaseous products evolved upon heating, or soluble phases released on contact with water. Cation exchange capacity (CEC), specific surface area (SSA), and caesium sorption determination were selected to characterise the clay component in bentonite.
Powder X-ray diffractometry (XRD) was conducted on an Empyrean third generation diffractometer (Malvern Panalytical B.V., Almelo, Netherlands) with the following measurement conditions: Co-K
α radiation, a PIXcel3D detector, a range 3.5–105° 2θ, a step of 0.026° 2θ, and a total counting time of 5.25 h. Randomly oriented powder mounts (with inner ZnO standard) and clay fraction (oriented specimens) were analysed. Minerals were identified using HighScore Plus version 4.8 (Malvern Panalytical B.V., Almelo, The Netherlands) software and PDF-4+ 2020 database. Quantitative analysis was performed using a graphical user interface Profex v.4.2.1 (Nicola Döbelin, Solothurn, Switzerland) [
25].
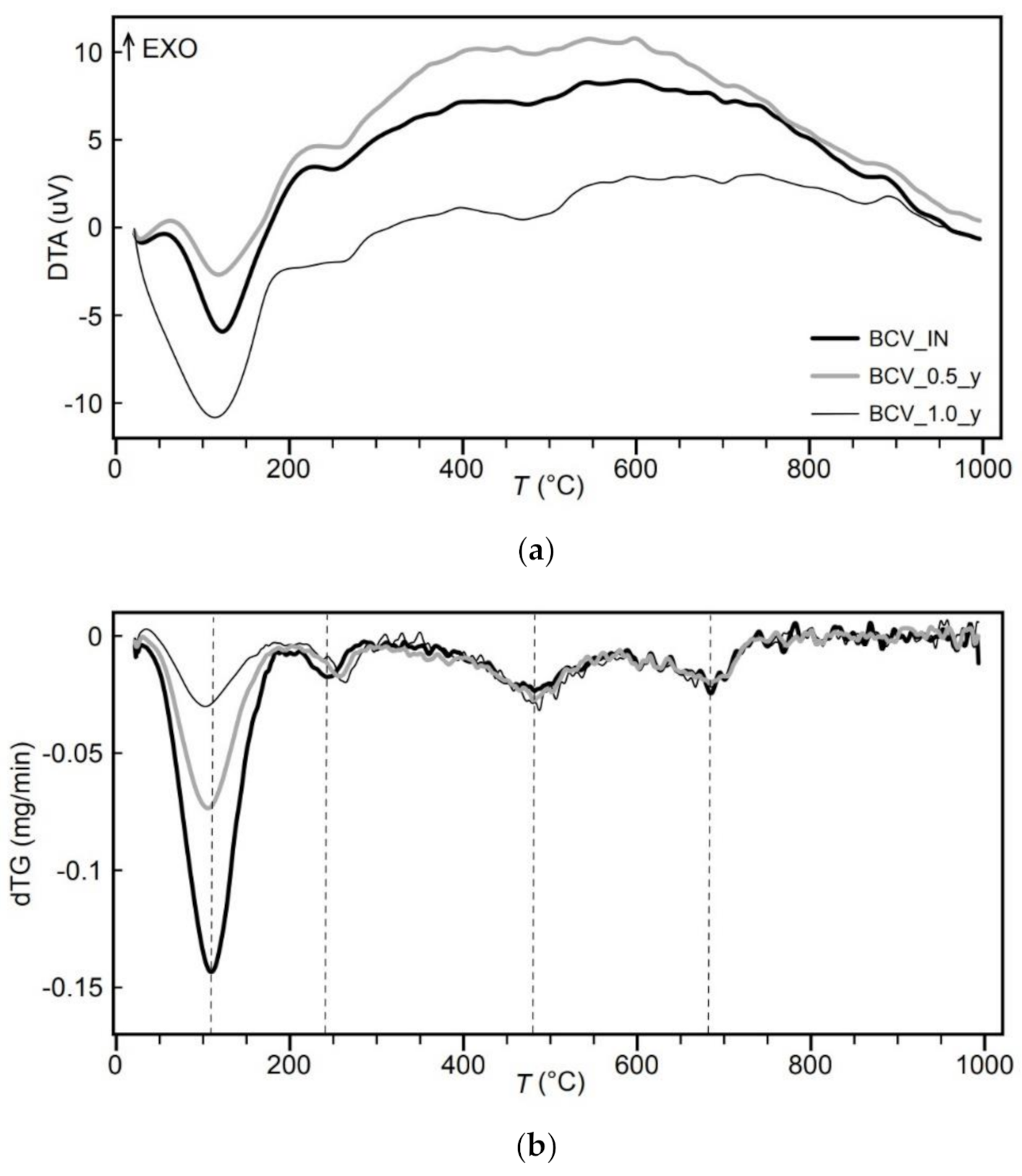
Thermal analysis with evolved gas analysis (TA-EGA) was performed on SetSys Evolution’s apparatus (Setaram, Calurie-et-Curie, France). The weight of the as-received samples was approximately 10 mg. Thermal degradations of bentonites were studied under an argon atmosphere (flow of 60 mL/min) in α-Al2O3 crucible. The heating rate was set to 10 °C/min in the temperature range of 30–1000 °C. Gases that evolved during heating were analysed with a quadrupole mass spectrometer QMG 700 (Pfeiffer Vacuum, Asslar, Germany) connected to a thermal analyser through a Supersonic System (Setaram, Calurie-et-Curie, France). Specific fragments corresponding to water, carbon dioxide, and sulphur oxides, were recorded.
The soluble salts were identified based on aqueous leachates. The dried (105 °C) samples were contacted with deionised water (40 mL) at three solid-to-liquid ratios for seven days. The suspensions were centrifuged, and supernatants were filtered using a 0.2 μm syringe filter. The main cations in the filtrates were analysed using atomic absorption spectrometer SavantAA (GBC Scientific Equipment, Braeside, Australia). The total alkalinity of the filtrates was determined by potentiometric titration using Titralab TIM800 (Radiometer Analytical, Loveland, USA). The concentrations of sulphates, chlorides, and fluorides were determined by ion chromatography (ALS Global, Prague, Czech Republic).
The highest solid-to-liquid ratio was also tested for caesium sorption. After seven days of shaking, the bentonite suspension of 200 g/L, aliquots of CsCl spiked with 134Cs were added and interacted for another seven days. Two initial caesium concentrations of spiking aliquots (0.001 mol/L and 6.4 mol/L) were chosen. Aliquots from supernatants, separated by centrifugation, were determined for 134Cs activity on gamma counter 1480 Wizard 3 (Perkin Elmer, Waltham, USA). Blank samples (without solid phase) were prepared and processed in the same way to evaluate the distribution coefficient of Cs. Each sorption experiment under the given conditions was performed in duplicate.
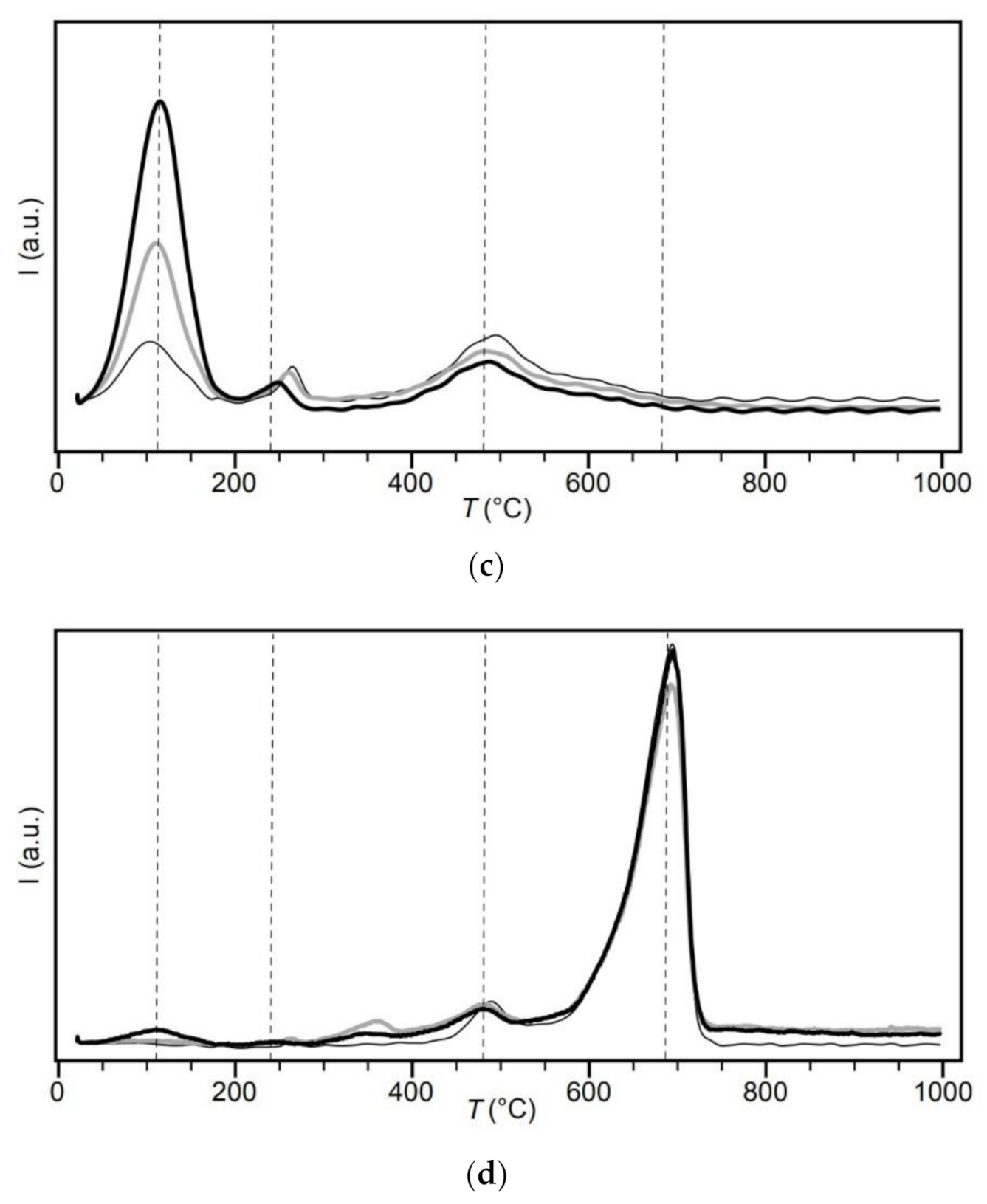
The CEC and exchangeable cations were determined by the Cu(II)-triethylenetetramine (Cu-trien) method [
26,
27]. Cu-trien (0.01 mol/L) was mixed with dried (105 °C) samples in the solid-to-liquid ratio of 25 g/L. After interaction for 30 min, the suspensions were centrifuged, and supernatants were analysed. The Cu
2+ concentration was determined by UV/Vis spectrophotometer Specord 205 (Analytik Jena, Jena, Germany). The concentration of displaced cations (Na
+, K
+, Ca
2+, and Mg
2+) was determined by atomic absorption spectrometer SavantAA (GBC Scientific Equipment, Braeside, Australia). Two values of cation exchange capacity were derived: CEC
Vis from copper depletion and CEC
SUM by summing equivalent concentrations of displaced cations.
The specific surface area (SSA) was determined based on the sorption of ethylene glycol monoethyl ether (EGME) [
28,
29]. Moreover, 1 g of bentonite powder (dried at 105 °C) was mixed with 2 mL of EGME and equilibrated in a desiccator with CaCl
2-EGME solvate. The applied procedure [
30] consisted of regular desiccator evacuation and monitoring of the samples until a constant weight was reached. The SSA was derived from the mass ratio of adsorbed EGME and dried bentonite sample.
2.3. Geotechnical Characterisation
Free swelling tests, determination of water retention curves (WRC), and saturated hydraulic conductivity were selected for geotechnical characterisation. Free swelling tests were performed with 10 mL deionised water and 0.6 g powders (previously dried at 105 °C). This solid-to-liquid ratio allows a better determination of the swell index in graduated cylinders for low swelling materials (such as Ca- and Mg-bentonites), highlighting the differences between the input and the thermally loaded bentonite samples. The standard ratio of 0.02 g/L [
31] did not allow the determination of the swell index nor the comparison between the samples.
Bentonite dry densities of 1400, 1600, and 1800 kg
/m
3 were chosen for determining saturated hydraulic conductivity and WRC. Bentonite powder was compacted in the cylindrical space of a cell body (diameter of 30 mm, thickness of 15 mm) using a hydraulic press MEGA 11-300 DM1S (FORM+TEST Seidner & Co., Riedlingen, Germany). In the WRC experiments, a hole was drilled for placing a wireless sensor (diameter of 17 mm, depth of 6 mm). The compacted sample was closed with end plates composed of stainless steel fabrics placed on the carbon composite percolator [
32]. Three-quarter section views of the experimental cells are shown in
Figure 2.
Saturated hydraulic conductivity of compacted bentonite was determined according to ISO 17892-11:2019. The cells were placed into a desiccator filled with deionised water. The desiccator was regularly vacuumed for at least two weeks. After water saturation, the cell was connected via pressure hoses to the assembly for measuring saturated hydraulic conductivity consisted of an input and output pressure controller (HPDPC and ELDPC, GDS Instruments, Hook, UK). Hydraulic heads of 5, 8, and 10 MPa were applied for the dry density of 1400, 1600, and 1800 kg/m3, respectively. The experiment was terminated when a steady-state water flow was reached, and the output water flow equalled the input water flow. The cell was dismantled, and the actual dry density was determined. The hydraulic conductivity was calculated according to Darcy’s law and converted to a referential temperature of 10 °C according to the ISO standard.
The block/sensor method [
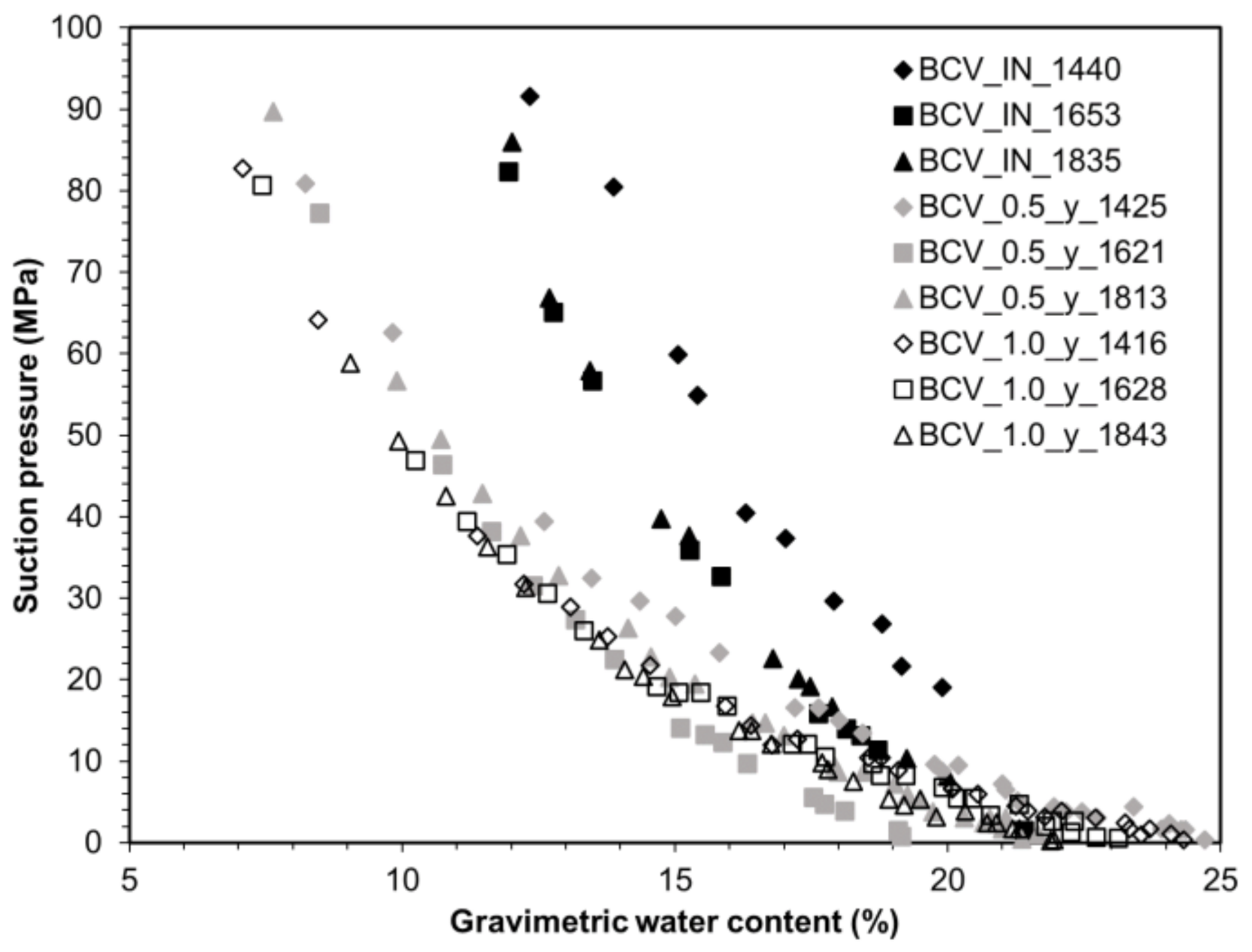
33] was used to determine WRC. A wireless sensor with its internal memory, Hygrochron iButton (Maxim Integrated, San Jose, CA, USA), was applied for measuring relative humidity and temperature inside the compacted sample exposed to saturated air with water vapour inside a desiccator filled with water. The cell was regularly weighed until a constant mass was reached. When constant mass was achieved, the cells were dismantled and the sensors removed. The measured data of relative humidity and temperature were used to calculate the suction pressure according to the Kelvin equation for each time interval. WRC were then constructed as a dependence between suction pressure and gravimetric water content.
2.4. Microbiological Characterisation
Cultivation of bentonite suspensions in an anaerobic atmosphere rich in H2, with a subsequent molecular genetic analysis, was used for studying microbial survivability in bentonite samples exposed to thermal loading. The bentonite suspensions were prepared from 2 g of naturally wet bentonite powders, mixed with 10 mL sterile Milli-Q water. Samples were incubated in an anaerobic workstation (Don Whitley Scientific, Bingley, United Kingdom) under an atmosphere containing 94% Ar and 6% H2 at room temperature. Suspensions were sampled at three different cultivation times, immediately after phases mixing (0 d), after 14 and 28 days, and kept in a freezer until DNA isolation. Zero-point (0 d) samples and BCV_IN samples were prepared in duplicate, whereas the heat-loaded samples in triplicate.
The DNA from the bentonite samples was extracted by DNeasy
® PowerMax
® Soil Kit (QIAGEN, Germantown, TN, USA) according to the manufacturer’s protocol. The extracted DNA was concentrated and purified by the DNA Clean & Concentrator Kit (Zymo Research, Irvine, CA, USA) and quantified by a Qubit 2.0 fluorometer (Life Technologies, Carlsbad, CA, USA). One background kit control without the sample was co-extracted together with samples in each DNA extraction batch to see the laboratory background and possible contaminations resulting from the kit or the laboratory. The extracted DNA was used to describe the microbial composition in the samples (16S rDNA amplicon sequencing, NGS) and for the relative quantification of microbial biomass in the samples (16S rDNA quantitative PCR). For these DNA-based analyses, protocols similar to [
34] were followed.
4. Discussion
The applied geochemical characterisation, targeting both the overall composition (in terms of mineralogy, thermolabile, and soluble phases) and the clay component, allowed investigating the effect of heating on individual components. The XRD analysis of BCV bentonite provided a substantial number of various phases, which may be affected by the exposition to the high temperatures, i.e., montmorillonite component, other clay minerals and non-clay minerals. Based on the XRD analysis, considering its uncertainties in detecting variations in the structural chemical composition of clay minerals, it is not possible to deduce any significant mineral transformation upon heating.
The processes that the bentonite underwent up to 150 °C are indicated by the results of thermal analysis. Besides the apparent loss of water content in as-received samples, TA-EGA identified a release of CO
2 from the BCV_IN sample. Several CO
2-related mass losses were recorded when heating the bentonite samples to higher temperatures, up to 1000 °C. The CO
2 release can be attributed mainly to the carbonate phases, which XRD and aqueous leachates detected. Assigning peaks to given minerals is difficult due to superimpositions of double carbonates with calcite [
39,
40]. The presence of various carbonates in BCV might be reflected in the broad CO
2 peak at 600–800 °C. The main difference in thermal behaviour between BCV_IN and thermally loaded samples was found in the shift of H
2O and CO
2 peaks. These shifts, and release of CO
2 from the BCV_IN sample up to 150 °C, indicate some alteration process during bentonite heating. Altered reactivity of carbonates was also detected by aqueous leachates. A significantly higher concentration of leachable HCO
3− from thermally loaded samples indicates an intense alteration of carbonate minerals, supported by the increased leaching of cations.
The impact of bentonite heating to 150 °C can also be inferred based on the leaching of fluorides, sulphates, and chlorides. A change, though to a minor extent, in sulphate content was also registered by TA-EGA. Nevertheless, no mineral phases containing fluorides, sulphates, and chlorides were detected by XRD. However, they may be present as impurities in minerals and/or in accessory amounts. For example, fluoride is commonly found in montmorillonites [
5,
41,
42]. It can substitute the hydroxyl groups in the octahedral sheets [
41]. The most significant increase in leachable concentration with exposure to elevated temperature has been determined for fluoride. An increased release of F
- into the solution might indicate montmorillonite lattice modification.
The effect of thermal loading on clay component was examined in detail using the determination of SSA and CEC and sorption experiments with caesium. The thermal loading caused a decrease in the SSA. A decrease in SSA upon heating up to 200 °C was also observed for Barmer bentonite [
43]. A lower specific surface area may indicate recrystallisation of minerals or montmorillonite collapse. Lower amorphous phases in thermally loaded samples may indicate recrystallisation of minerals, often leading to textural coarsening. Coarsening then may facilitate the release of species when contacted with water. Higher leachable concentrations were found for thermally loaded samples. However, since EGME penetrates in the montmorillonite interlayers due to its polarity, the SSA decrease can be more attributed to the montmorillonite alteration than to recrystallisation of minerals.
Montmorillonite lattice modification in thermally loaded samples can be deduced from the experiments with selectively interacting cations (such as Cu-trien cation or Cs
+). The
134Cs distribution coefficient at both studied carrier concentration decreased in the order: IN > 0.5_y > 1.0_y. The same trend was determined for the CEC determination with Cu-trien. Decreasing CEC due to extensive drying was observed in various bentonites and one illite/montmorillonite clay and was attributed to the type of exchangeable cations and their fixation [
44]. Generally, the origin of the CEC is a permanent negative charge of montmorillonite clay layers due to isomorphous substitutions [
45]. Altered reactivity of the applied selective cations, interacting by the cation exchange, clearly indicates a change in surface charge density of clay particles. Such a partial alteration can result in the formation of new phases (low-swelling or even non-swelling) at the expense of montmorillonite content.
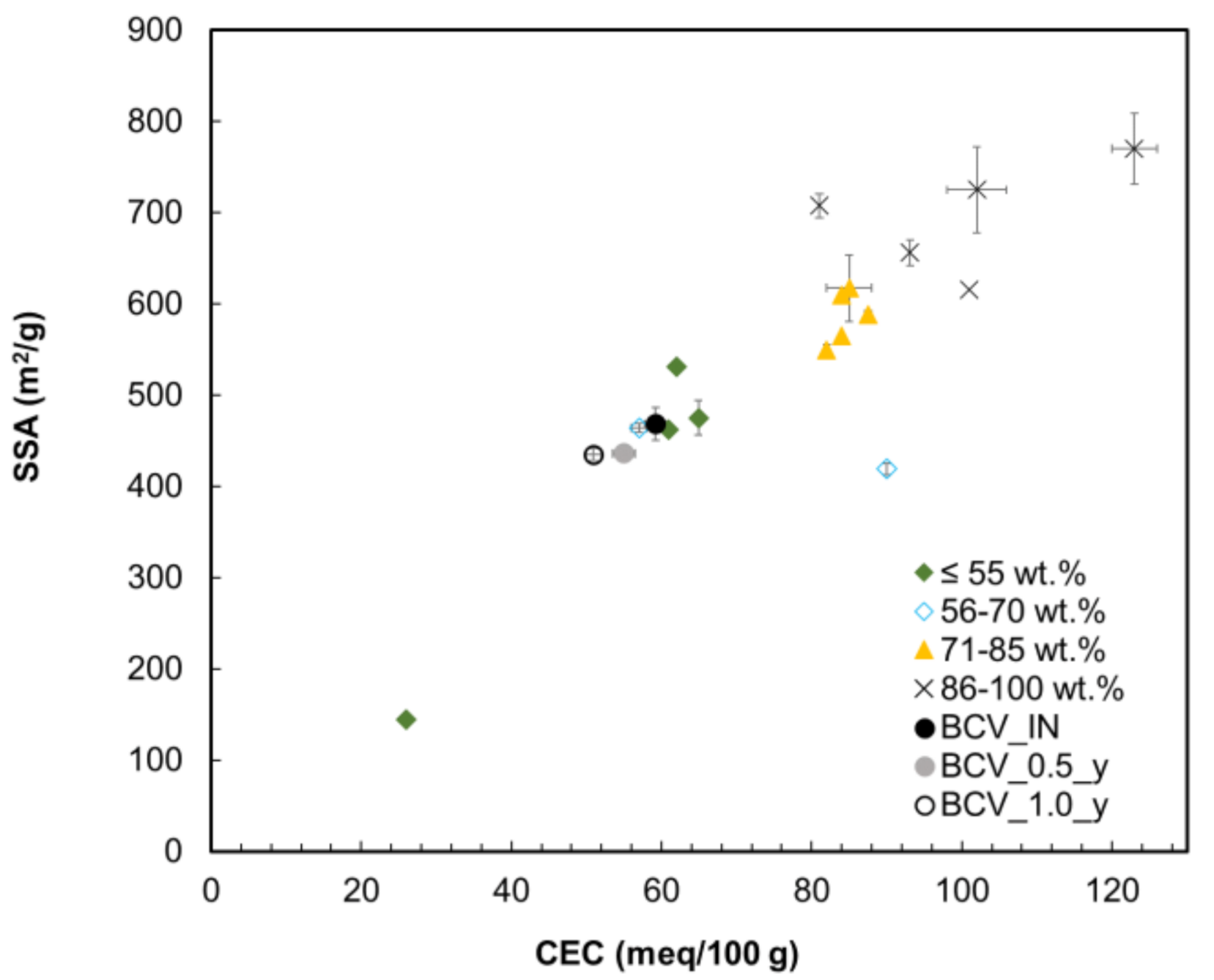
To support the finding of reduced montmorillonite content, which could not be confirmed using XRD, data analysis of CEC, SSA was performed. The interlocking of SSA, CEC and smectite content is illustrated in
Figure 8. Data selection (see
Table 6) was mainly restricted by existing SSA values measured according to the EGME method. Previously published data were compared with the results from the current study. Five groups of bentonites can be distinguished corresponding to their smectite content. The lowest SSA and CEC values are typical for bentonites containing up to 50 wt.% of smectite. The highest SSA and CEC values are typical for bentonites containing almost 100 wt.% of smectite. The CEC
Vis and SSA values of the BCV_IN sample correspond well with the correlation matching the group of bentonites containing 50–80 wt.% of smectite.
The most interesting finding of this study is in the composition of exchangeable cations. Magnesium dominated bentonite lost a significant amount of exchangeable Mg
2+ when heated. The proportion of Ca
2+ and K
+ increased with heating, while Na
+ remained unaffected (see
Figure 4). These changes in exchangeable cations took place in the solid state, without the influence of external fluids. In comparison, in the in situ experiments with access to groundwater, a gradually increasing proportion of the divalent cations towards the heater was observed [
13]. Only water originally present in BCV_IN and escaping due to thermal loading could act as a transport medium. From this point of view, it is necessary to analyse the top layers of bentonite, which were excluded from current research due to the small amount (see
Section 2.1). The loss of exchangeable Mg
2+ can be explained by increased fixation between charged surfaces [
44] or crystal lattice transformation. Magnesium in the montmorillonite structure is located in the octahedral sheet together with Al and Fe [
13]. It can be expected that when the montmorillonite lattice is damaged, Mg
2+ is released from the structure, and the surface charge is redistributed. A very slight decrease in the mean layer charge density for montmorillonite was detected in compacted bentonite (Almeria, Spain) exposed in situ to 100 °C [
52]. The change in surface charge density was deduced from our results of CEC, SSA and Cs sorption. Moreover, 1.5–2 times higher Mg concentrations were determined in the aqueous leachates from the thermally loaded samples compared to the input sample. Therefore, we are inclined to explain the changes in exchangeable cations to the partial lattice modifications. Plötze et al. [
52], however, observed Mg enrichment in samples from the heater region, as in many large-scale tests [
4,
13]. The mechanism behind the Mg enrichment is not yet clear [
4]. Both mechanisms, magnesium enrichment and depletion (as observed in this study), would be worth investigating thoroughly in future research.
The studied geotechnical parameters (free swelling, saturated hydraulic conductivity, and water retention curves) showed worsening with the thermal loading time. All these parameters are related to the behaviour of water in bentonite (maximal expansion of expandable structures in free swelling test, water uptake and transport through confined clay particles). As the water uptake ability of montmorillonite depends critically on the magnitude of the surface charge density of the particles [
45], the observed geotechnical behaviour indicates changes in surface charge distribution. The resulting reduction of expandable clay structures towards low- or non-swelling clay structures upon heating was therefore subjected to data analysis of saturated hydraulic conductivity.
Figure 9 displays hydraulic conductivities converted to a referential temperature of 10 °C, according to ISO 17892-11, for BCV bentonite and selected bentonites (from
Table 6) tested by Karnland et al. [
53]. A correlation between saturated hydraulic conductivity and montmorillonite content can be seen. The higher the
k10, the lower the montmorillonite content. The
k10 values for BCV can be categorised between Rokle bentonite and Friedland clay, which corresponds well to their low montmorillonite content. The saturated hydraulic conductivity of Deponit CAN, MX-80 and Asha is one order of magnitude smaller due to higher montmorillonite content.
A shift of measured saturated hydraulic conductivity for BCV towards clay material of lower montmorillonite content with thermal loading period can be clearly seen. Nevertheless, BCV meets an acceptance safety limit for saturated hydraulic conductivity of buffer lower than 10
−12 m/s [
17], even after the thermal loading. It can also be noticed in
Figure 9 that the differences between input and thermally loaded BCV samples were more dramatic for lower dry densities, whereas for dry densities over 1700 kg
/m
3, the differences induced by the thermal loading seem to be reduced. Based on this observation, it can be hypothesised that no significant effect of thermal loading on saturated hydraulic conductivity [
13] might be due to the high density of bentonite samples. On the other hand, the most notable difference between BCV samples was found in free swelling tests, where the dry density was very low. Therefore, it seems that the thermally induced changes manifest more in less dense systems. For further investigation, higher dry densities or, and vice-versa, lower ones than those used (e.g., 2000 kg
/m
3 and 1000 kg
/m
3) would be promising to determine saturated hydraulic conductivity and WRC to highlight the impact of the thermal loading on buffer mass.
Water retention ability for all dry densities decreased with the thermal loading period. This behaviour was more relevant for lower water content; for water content above 20%, the water retention ability of both thermally unloaded and thermally loaded samples became closer to each other. This trend was also observed by [
9]. The most significant difference is between the BCV_IN samples and the BCV_0.5_y ones; the difference between BCV_0.5_y and BCV_1.0_y is minor. It indicates that most of the material changes took place during the first half-year of thermal loading. This fact corresponds well with SSA values, where between BCV_IN and BCV_0.5_y, a crucial decrease was observed, whereas between BCV_0.5_y and BCV_1.0_y no visible change was detected.
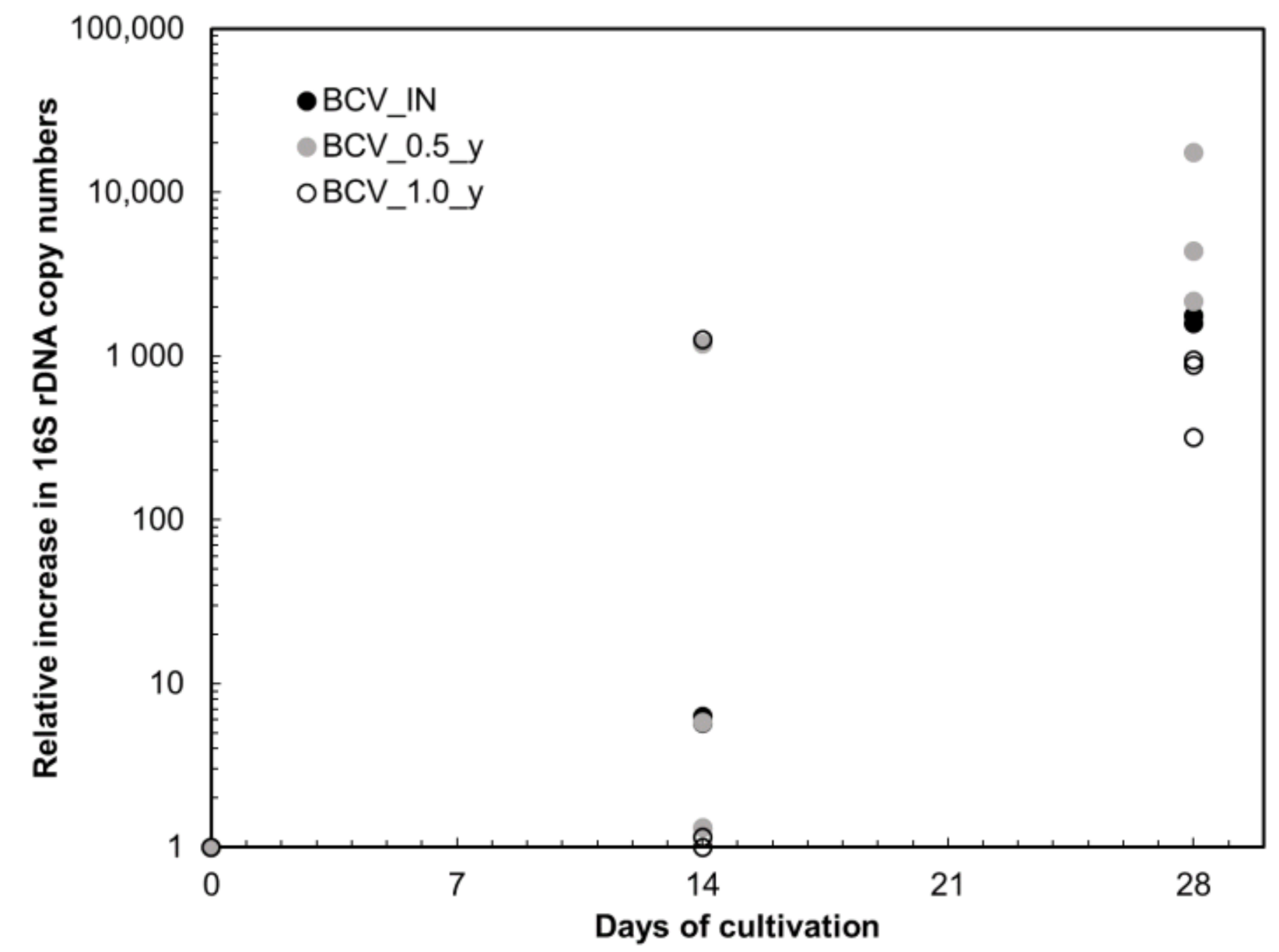
Microbiological experiments under very hospitable conditions in a suspended state (i.e., without space and water availability limitation) with continuous electron donor supply in the form of H2 present in an anaerobic atmosphere showed that heating of bentonite powder at 150 °C for up to one year did not result in bentonite sterilisation. However, a reduction of microbial load with increasing thermal loading time was observed. This reduction was especially true for the BCV_1.0_y samples, where microbial recovery was lower than BCV_IN and BCV_0.5_y. Our experiment further showed that microbial recovery from the dormant stages is a lengthy and probably partially stochastic procedure. After 14 days of cultivation, the signal in relative quantification seems to be rather random in the case of thermally loaded samples, while after 28 days of cultivation, the pattern was much more consistent, and this cultivation time proved to be more suitable for the detection of microbial activity restoration in bentonite samples at given environmental conditions.
Although we showed that bacteria could at least partially resume their activity after medium-term heat loading when cultivated at suitable conditions, the results imply that a substantial reduction in microbial load or even complete sterilisation of bentonite powder could be possible when the heating time exceeds one year. An equilibrium between an increase of the temperature limit at the canister surface resulting in bentonite sterilisation and the potential risk of losing favourable geochemical and geotechnical properties should therefore be addressed in the DGR performance assessment.
5. Conclusions
A medium-term thermal loading of bentonite powder resulted in detectable changes of properties, important directly or indirectly for the safety performance as a barrier material in a deep geological repository. No significant bentonite alteration, in terms of mineralogical composition, was observed by XRD. The thermal loading resulted in progressive disappearance of basal diffraction of montmorillonite, indicating increasing disorder of the montmorillonite layers. The impact of thermal loading on the overall bentonite composition was registered by aqueous leachates and TA-EGA mainly for carbonate phases, then for sulphates and fluorides.
Based on the determination of cation exchange capacity, total surface area and caesium distribution coefficient and interpretation of these parameters, it was concluded that montmorillonite had been partially altered in terms of the magnitude of the surface charge density of montmorillonite particles. Montmorillonite alteration towards low- or non-swelling clay structures was also supported by the determined geotechnical behaviour. Swelling ability and water uptake ability of thermally loaded samples were lower, and saturated hydraulic conductivity higher than for the input bentonite material.
This study has shown that XRD is a suitable method for mineralogical characterisation, but it was found to be insensitive for detecting slight variations of minerals in bentonite. In order to identify the altered montmorillonite fraction in more detail, a future investigation will focus on the clay fraction separation and its detailed analysis. We also plan to sample the heated bentonite powder after two years of thermal loading. Therefore, the current methods applied in this study to characterise bentonite properties will be amended to capture an alteration of clay minerals, especially by monitoring silica, aluminium, iron, and magnesium, which could confirm the hypothesis of thermal stress damage to the montmorillonite lattice.
The thermal loading also resulted in a reduction of microbial survivability and, thus, affected possible future microbial activity in bentonite. Thermal loading could play an important role in a permanent reduction of microbial activity in the bentonite buffer. However, more extended experiments focused on the stress-induced hindrances to microbial growth are needed.