Diffraction Features from (101¯4) Calcite Twins Mimicking Crystallographic Ordering
Abstract
:1. Introduction
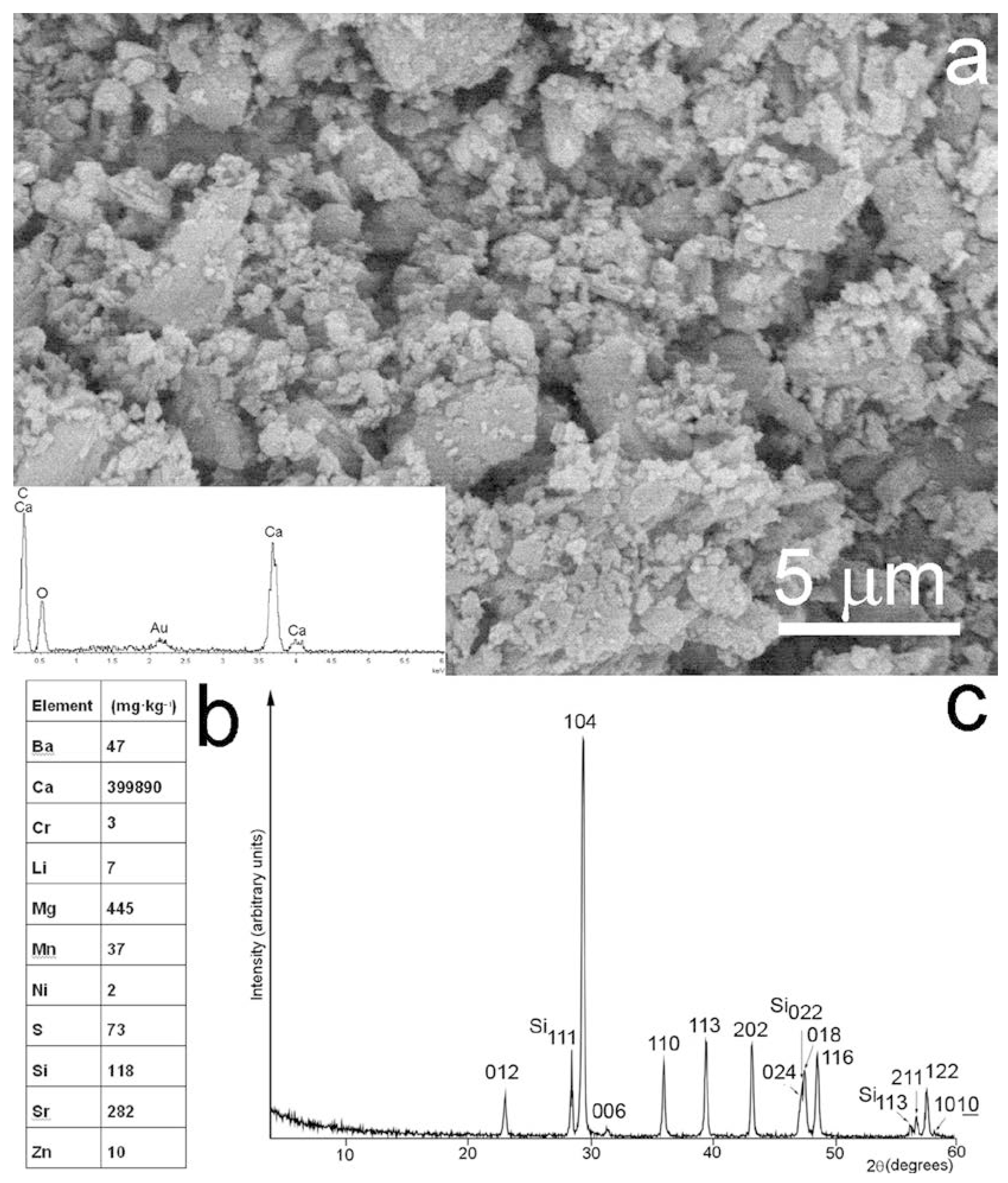
2. Materials and Methods
3. Results and Discussion
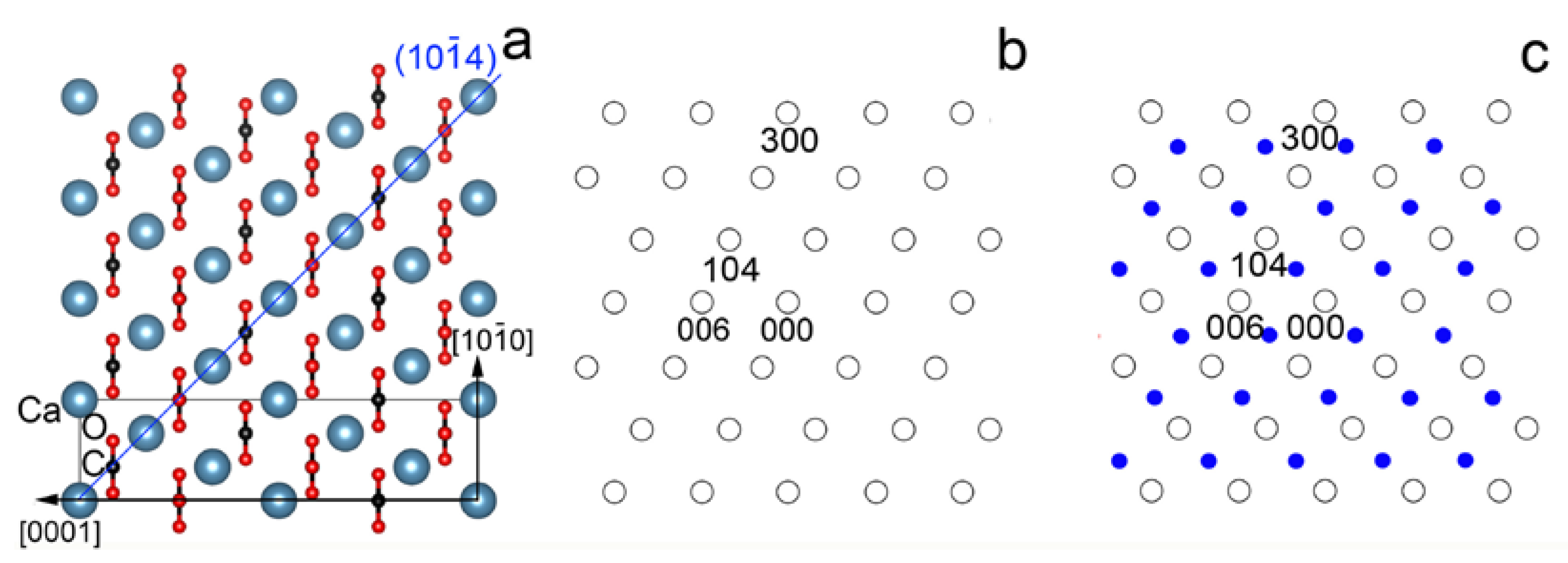
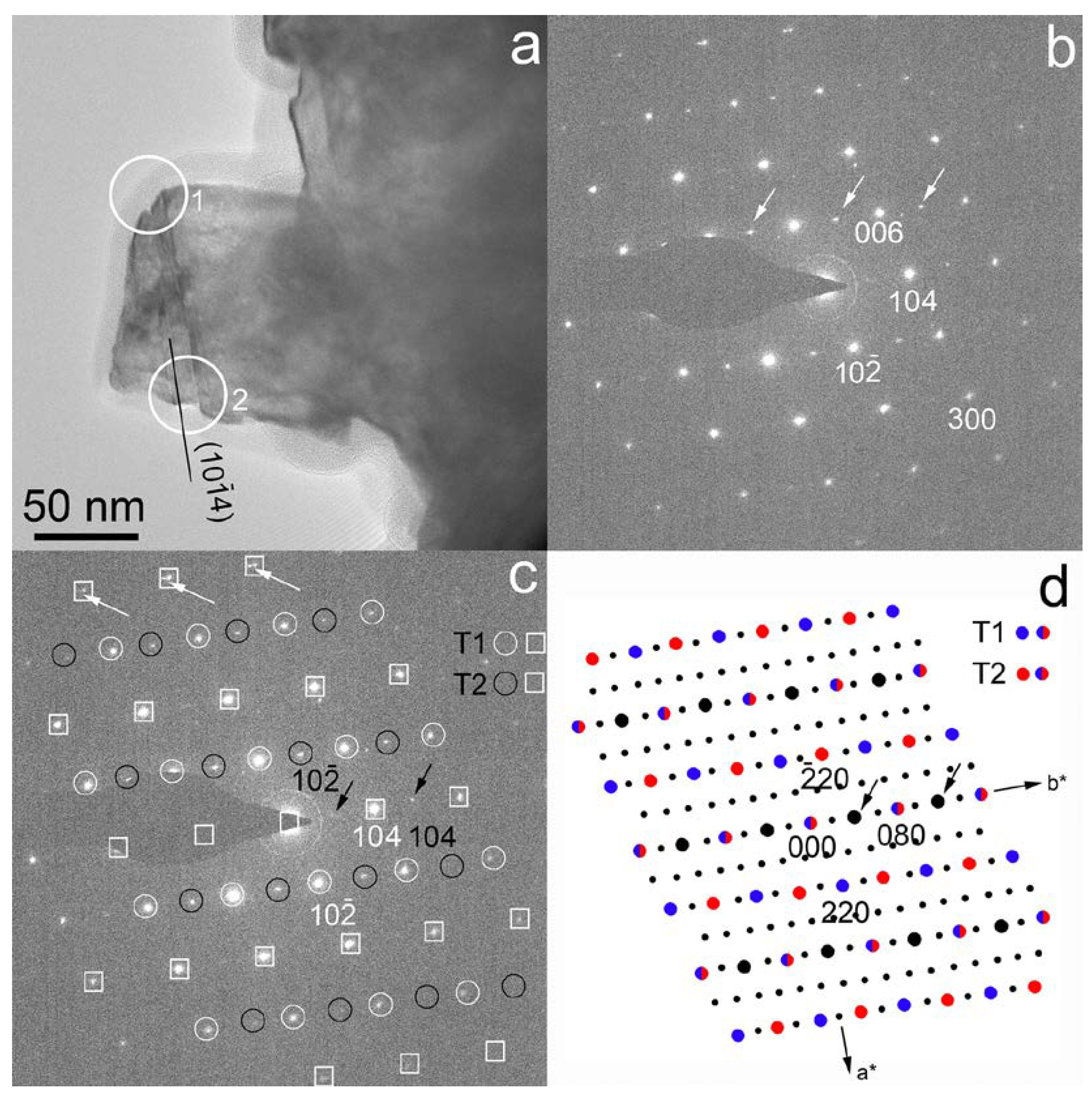
3.1. c-Type Reflections and the Twin Lattice
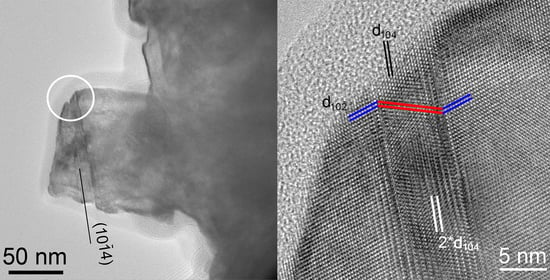
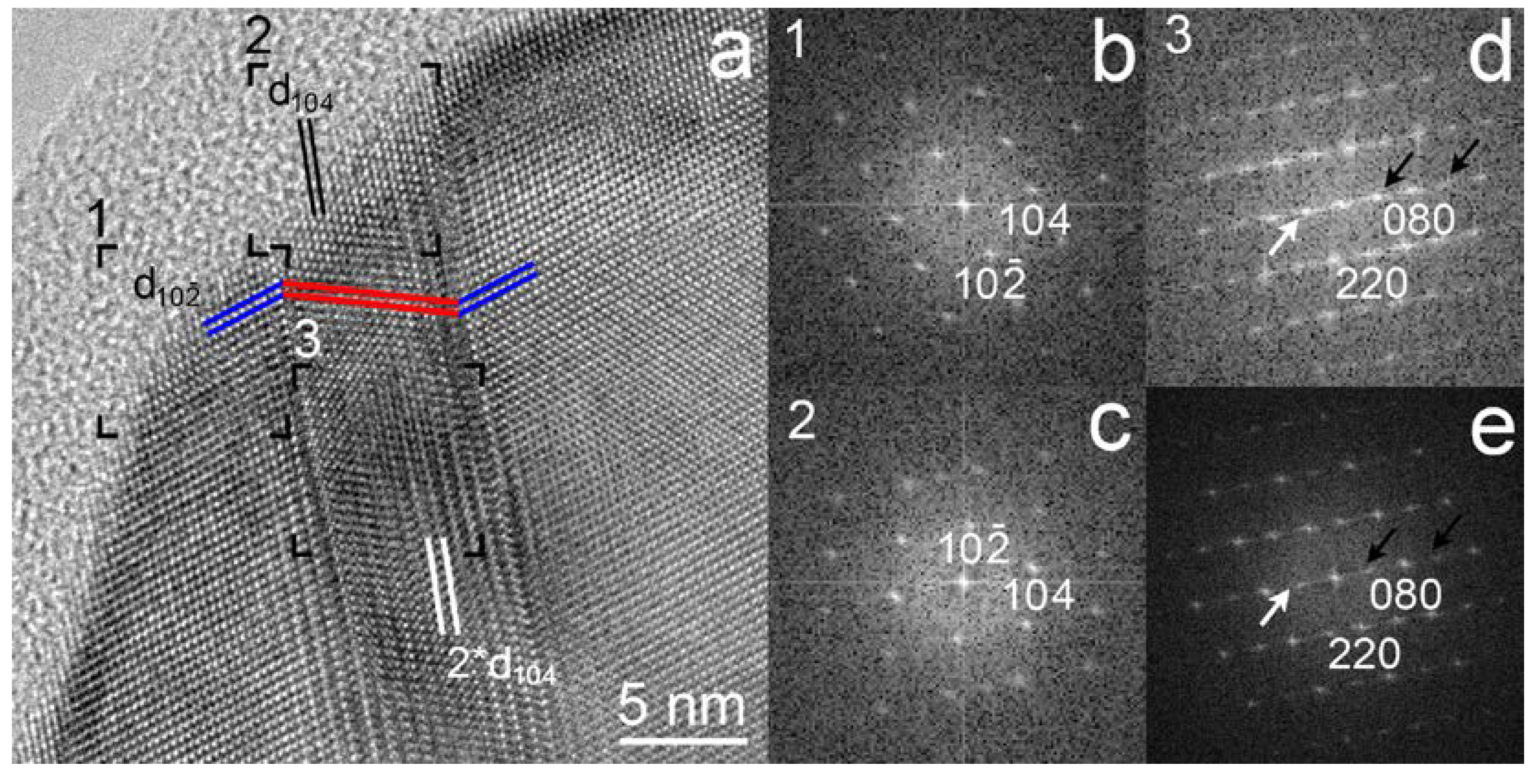
3.2. HRTEM Images of the (104) Calcite Twins and Doubled d104 Spacings
3.3. Crystal Structure of the (104) Calcite Twins
3.4. Possible Origin of the (104) Calcite Twins
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inkson, B.J. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) for materials characterization. In Materials Characterization Using Nondestructive Evaluation (NDE) Methods Editor(s): Gerhard Hübschen, Iris Altpeter, Ralf Tschuncky, Hans-Georg Herrmann; Woodhead Publishing: Cambridge, UK, 2016; pp. 17–43. [Google Scholar] [CrossRef]
- Jiang, Y.; Chen, Z.; Han, Y.; Deb, P.; Gao, H.; Xie, S.; Muller, D.A. Electron ptychography of 2D materials to deep sub-ångström resolution. Nature 2018, 559, 343–349. [Google Scholar] [CrossRef]
- Yan, X.; Liu, C.; Gadre, C.A.; Gu, L.; Aoki, T.; Lovejoy, T.C.; Pan, X. Single-defect phonons imaged by electron microscopy. Nature 2021, 589, 65–69. [Google Scholar] [CrossRef]
- Németh, P.; Dódony, I.; Pósfai, M.; Buseck, P.R. Complex defect in pyrite and its structure model derived from geometric phase analysis. Microsc. Microanal. 2013, 19, 1303–1307. [Google Scholar] [CrossRef] [PubMed]
- Larsson, A.-K.; Christy, A.G. On twinning and microstructures in calcite and dolomite. Am. Mineral. 2008, 93, 103–113. [Google Scholar] [CrossRef]
- Németh, P.; Garvie, L.A.J.; Buseck, P.R. Twinning of cubic diamond explains reported nanodiamond polymorphs. Sci. Rep. 2015, 5, 18381. [Google Scholar] [CrossRef] [PubMed]
- Cayron, C. Diffraction artefacts from twins and stacking faults, and the mirage of hexagonal, polytypes or other superstructures. Scr. Mater. 2020, 194, 113629. [Google Scholar] [CrossRef]
- Huggett, J.M.; Schultz, B.P.; Shearman, D.J.; Smith, A.J. The petrology of ikaite pseudomorphs and their diagenesis. Proc. Geol. Assoc. 2005, 116, 207–220. [Google Scholar] [CrossRef]
- De Lurio, J.L.; Frakes, L.A. Glendonites as a paleoenvironmental tool: Implications for early Cretaceous high latitude climates in Australia. Geochim. Cosmochim. Acta 1999, 633, 1039–1048. [Google Scholar] [CrossRef]
- Rogov, M.; Ershova, V.; Vereshchagin, O.; Vasileva, K.; Mikhailova, K.; Krylov, A. Database of global glendonite and ikaite records throughout the Phanerozoic. Earth Syst. Sci. Data 2021, 13, 343–356. [Google Scholar] [CrossRef]
- Tollefsen, E.; Balic-Zunic, T.; Mörth, C.M.; Brüchter, V.; Lee, C.C.; Skelton, A. Ikaite nucleation at 35 °C challenges the use of glendonite as a paleotemperature indicator. Sci. Rep. 2000, 10, 8141. [Google Scholar] [CrossRef]
- Brooks, R.; Clark, L.M.; Thurston, E.F. Calcium carbonate and its hydrates. Philos. Trans. R. Soc. A 1950, 243, 145–167. [Google Scholar] [CrossRef]
- Swainson, I.P.; Hammond, R.P. Ikaite, CaCO3.6H2O: Cold comfort for glendonites as palaeothermometers. Am. Mineral. 2001, 86, 1530–1533. [Google Scholar] [CrossRef]
- Selleck, B.W.; Carr, P.F.; Jones, B.G. A Review and Synthesis of Glendonites (Pseudomorphs after Ikaite) with New Data: Assessing Applicability as Recorders of Ancient Coldwater Conditions. J. Sediment. Res. 2007, 77, 980–991. [Google Scholar] [CrossRef] [Green Version]
- Pauly, H. ‘Ikaite’, a new mineral from Greenland. Arctic Res. 1963, 16, 263–279. [Google Scholar] [CrossRef] [Green Version]
- Bischoff, J.L.; Fitzpatrick, J.A.; Rosenbauer, R.J. The solubility and stabilization of ikaite (CaCO3· 6H2O) from 0 to 25 C: Environmental and paleoclimatic implications for thinolite tufa. J. Geol. 1993, 101, 21–33. [Google Scholar] [CrossRef]
- Stein, E.S.; Smith, A.J. Authigenic carbonate nodules in the Nankai Trough, site 583. Initial. Rep. Deep. Sea Drill. Proj. 1985, 87, 659–668. [Google Scholar]
- Council, T.C.; Bennett, P.C. Geochemistry of ikaite formation at Mono Lake, California: Implications for the origin of tufa mounds. Geology 1993, 21, 971–974. [Google Scholar] [CrossRef]
- Zou, Z.; Bertinetti, L.; Habraken, W.; Fratzl, P. Reentrant phase transformation from crystalline ikaite to amorphous calcium carbonate. CrystEngComm 2018, 20, 2902–2906. [Google Scholar] [CrossRef] [Green Version]
- Tang, C.C.; Thompson, S.P.; Parker, J.E.; Lennie, A.R.; Azough, F.; Kato, K. The ikaite-to-vaterite transformation: New evidence from diffraction and imaging. J. Appl. Crystallogr. 2009, 42, 225–233. [Google Scholar] [CrossRef]
- Németh, P.; Töchterle, P.; Dublyansky, Y.; Stalder, R.; Molnár, Z.S.; Klébert, S.Z. Spötl Tracing structural relicts of the ikaite-to-calcite transformation in cryogenic cave glendonite. Am. Mineral 2021. submitted. [Google Scholar]
- Reeder, R.J.; Wenk, H.R. Microstructures in low-temperature dolomites. Geophys. Res. Lett. 1979, 6, 77–80. [Google Scholar] [CrossRef]
- Redfern, S.A.T.; Salje, E.; Navrotsky, A. High-temperature enthalpy at the orientational order-disorder transition in calcite: Implications for the calcite/aragonite phase equilibrium. Contrib. Mineral. Petrol. 1989, 101, 479–484. [Google Scholar] [CrossRef]
- Van Tendeloo, G.; Wenk, H.R.; Gronsky, R. Modulated structures in calcium dolomite: A study by electron microscopy. Phys. Chem. Miner. 1985, 12, 333–341. [Google Scholar] [CrossRef]
- Wenk, H.R.; Meisheng, H.; Lindsey, T.; Morris, J.W. Superstructure in ankerite and calcite. Phys. Chem. Miner. 1991, 17, 527–539. [Google Scholar] [CrossRef]
- Shen, Z.; Konishi, H.; Brown, P.E.; Xu, H. STEM investigation of exsolution lamellae and “c” reflections in Ca-rich dolomite from the Platteville Formation, western Wisconsin. Am. Mineral. 2013, 4, 760–766. [Google Scholar] [CrossRef] [Green Version]
- Nespolo, M.; Ferraris, G. Applied geminography—Symmetry analysis of twinned crystals and definition of twinning by reticular polyholohedry. Acta Crystallogr. A 2004, 60, 89–95. [Google Scholar] [CrossRef]
- Dublyansky, Y.; Moseley, G.E.; Lyakhnitsky, Y.; Cheng, H.; Edwards, R.L.; Scholz, D.; Spötl, C. Late Palaeolithic cave art and permafrost in the Southern Ural. Sci. Rep. 2018, 8, 12080. [Google Scholar] [CrossRef] [PubMed]
- Seto, Y.D.; Hamane, T.N.; Sata, N. Development of a software suite on X-ray diffraction experiments. Rev. High Press. Sci. Technol. 2010, 20, 269–276. [Google Scholar] [CrossRef] [Green Version]
- Zsigmond, A.R.; Kántor, I.; May, Z.; Urák, I.; Héberger, K. Elemental composition of Russula cyanoxantha along an urbanization gradient in Cluj-Napoca (Romania). Chemosphere 2020, 238, 124566. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Nespolo, M.; Ferraris, G. The derivation of twin laws in non-merohedric twins—Application to the analysis of hybrid twins. Acta Crystallogr. A 2006, 62, 336–349. [Google Scholar] [CrossRef]
- Németh, P.; Garvie, L.A.J.; Aoki, T.; Dubrovinskaia, N.; Dubrovinsky, L.; Buseck, P.R. Lonsdaleite is faulted and twinned cubic diamond and does not exist as a discrete material. Nat. Commun. 2014, 5, 5447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daulton, T.L.; Bernatowicz, T.J.; Lewis, R.S.; Messenger, S.; Stadermann, F.J.; Amari, S. Polytype distribution in circumstellar silicon carbide: Microstructural characterization by transmission electron microscopy. Geochim. Cosmochim. Acta 2003, 67, 4743–4767. [Google Scholar] [CrossRef]
- Bruno, M.; Massaro, F.R.; Rubbo, M.; Prencipe, M.; Aquilano, D. The (10.4), (01.8), (01.2) and (00.1) twin laws of calcite (CaCO3): Equilibrium geometry of the twin boundary interfaces and twinning energy. Cryst. Growth Des. 2010, 10, 3102–3109. [Google Scholar] [CrossRef]
- Aquilano, D.; Benages-Vilau, R.; Bruno, M.; Rubbo, M.; Massaro, F. Positive {hk.l} and negative {hk.l} forms of calcite (CaCO3) crystal. New open questions from the evaluation of their surface energies. CrystEngComm 2013, 15, 4465. [Google Scholar] [CrossRef]
- Wenk, H.R.; Barber, D.J.; Reeder, R.J. Microstructures in Carbonates. In Carbonates: Mineralogy and Chemistry; Reeder, R.J., Ed.; Reviews in Mineralogy; Mineralogical Society of America: Blacksburg, VA, USA, 1983; Volume 11, pp. 301–367. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Németh, P. Diffraction Features from (101¯4) Calcite Twins Mimicking Crystallographic Ordering. Minerals 2021, 11, 720. https://doi.org/10.3390/min11070720
Németh P. Diffraction Features from (101¯4) Calcite Twins Mimicking Crystallographic Ordering. Minerals. 2021; 11(7):720. https://doi.org/10.3390/min11070720
Chicago/Turabian StyleNémeth, Péter. 2021. "Diffraction Features from (101¯4) Calcite Twins Mimicking Crystallographic Ordering" Minerals 11, no. 7: 720. https://doi.org/10.3390/min11070720
APA StyleNémeth, P. (2021). Diffraction Features from (101¯4) Calcite Twins Mimicking Crystallographic Ordering. Minerals, 11(7), 720. https://doi.org/10.3390/min11070720