Insights into Selection of the Auxiliary Collector and Its Applicability Analysis for Improving Molybdenite Flotation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Flotation Tests
2.3. Filtration Characteristic Tests of Flotation Concentrate
2.4. Crystallization Characteristics Analysis of Collectors
2.5. Crystallization Characteristic Measurements of Collectors on the Surface of Flotation Concentrate
3. Results
3.1. Analysis on the Selection Principle of the Main Collector and Auxiliary Collector
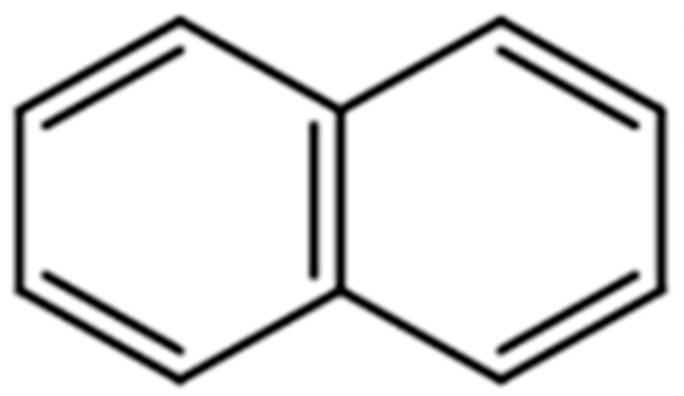
3.2. Surface Energy Analysis of Collectors
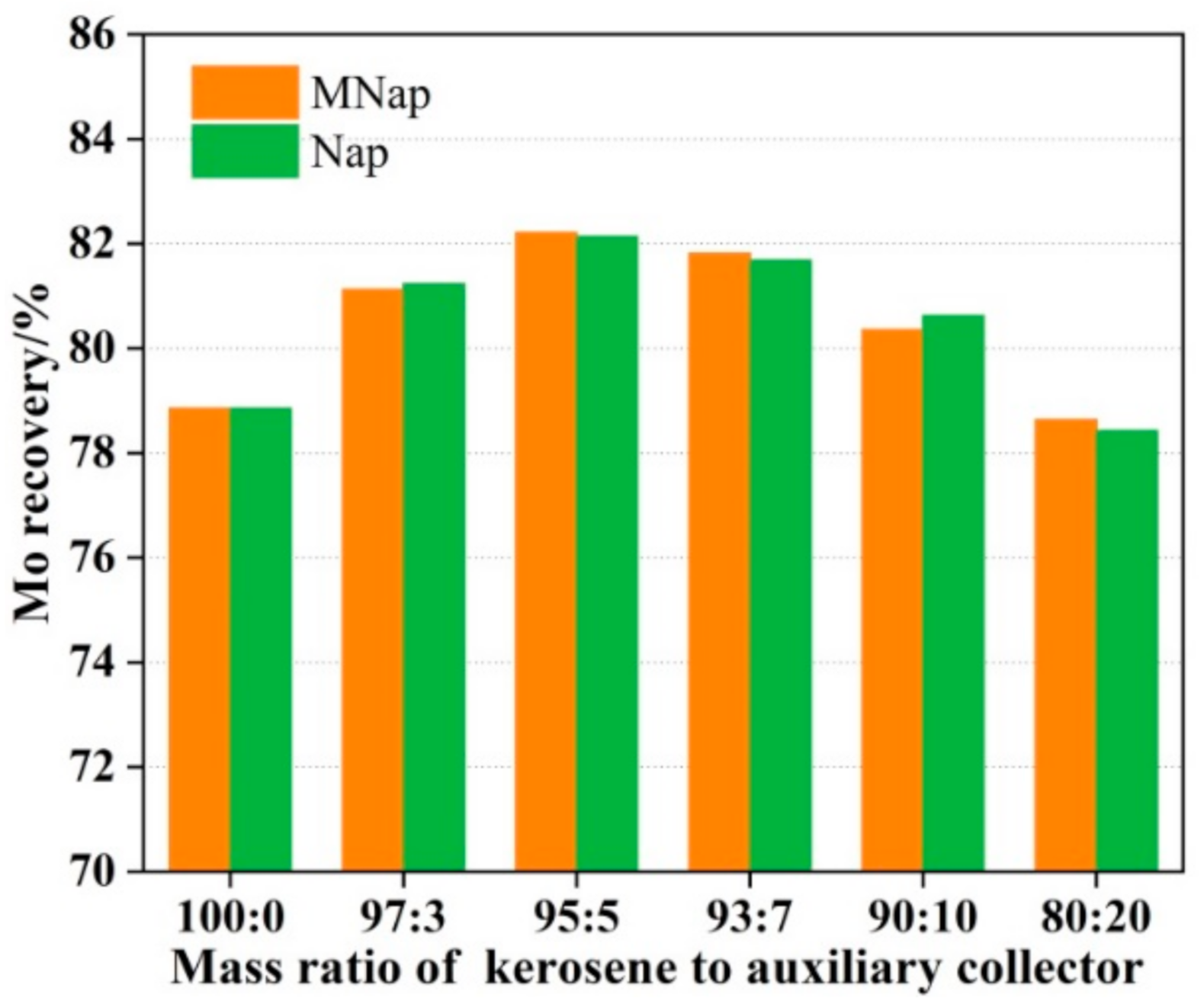
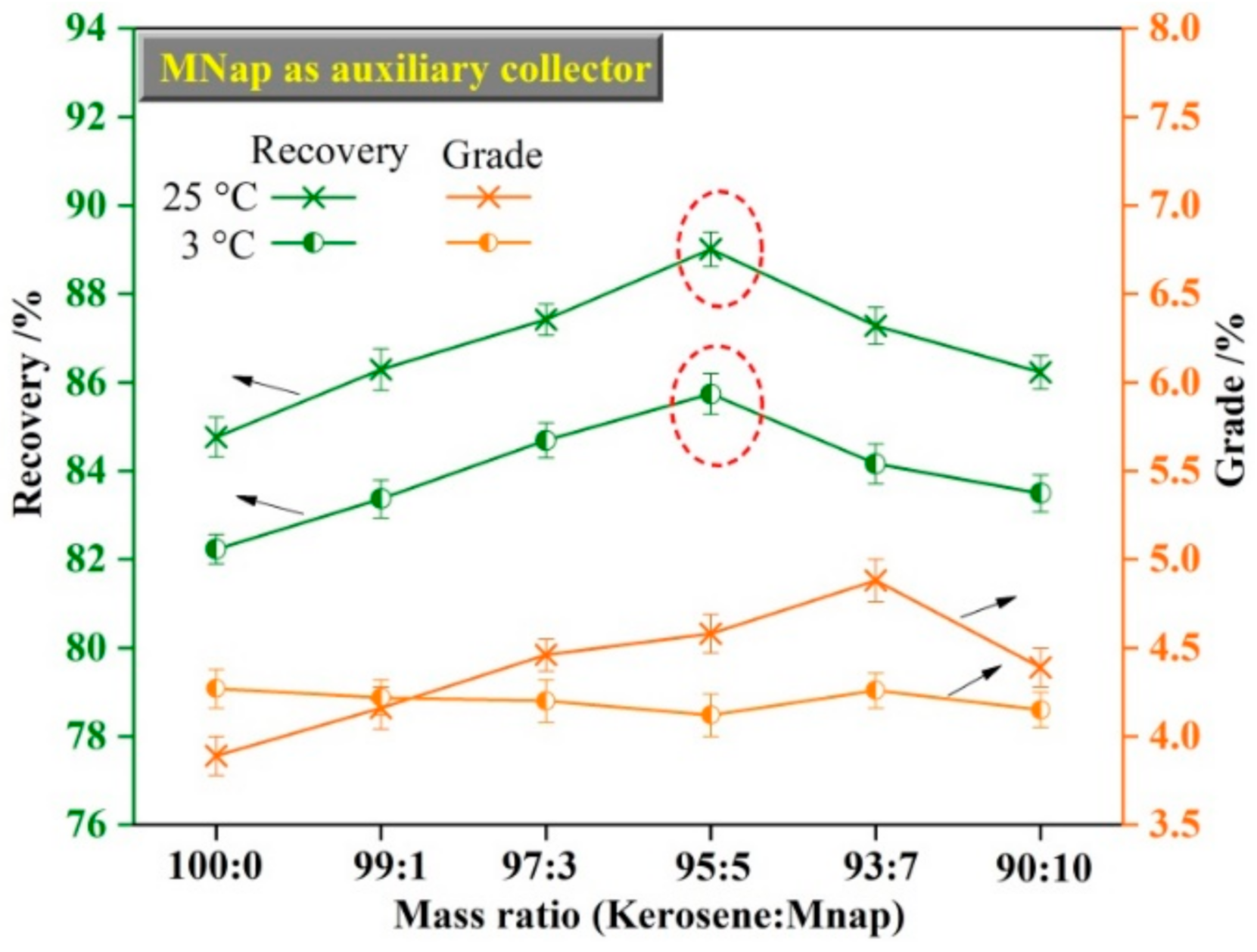
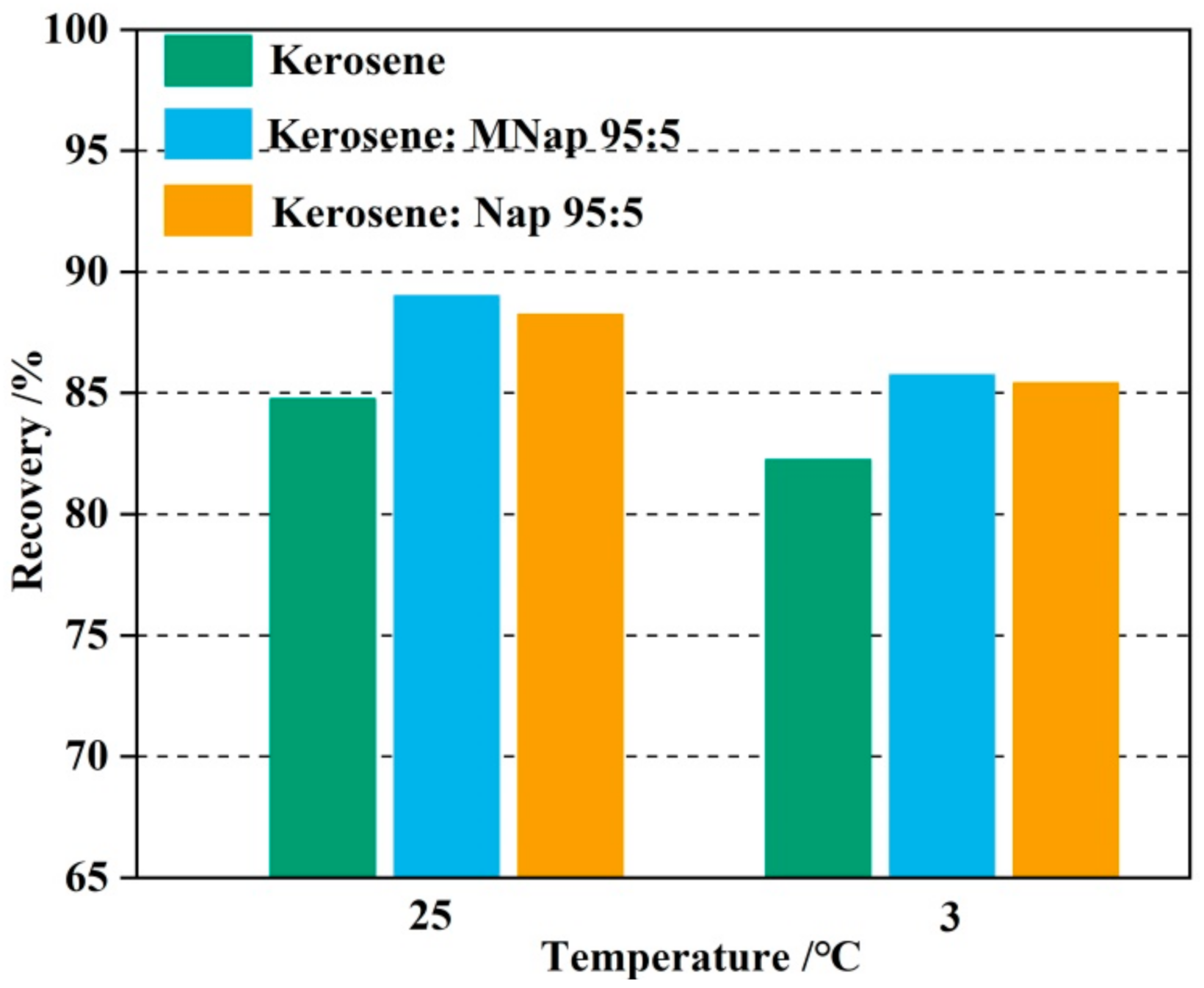
3.3. The Effect of Auxiliary Collector on Molybdenite Flotation
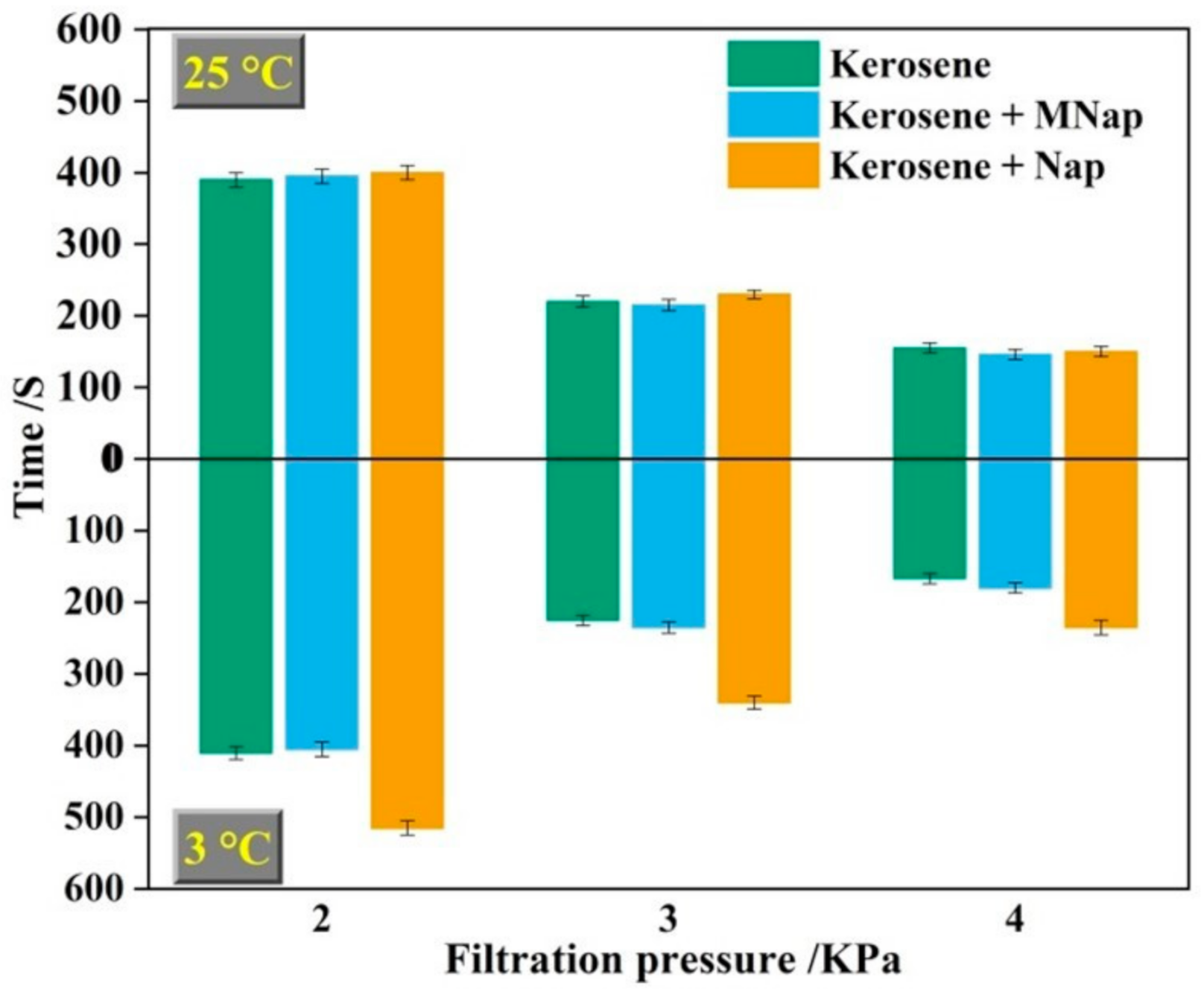
3.4. Filtration Characteristics of Flotation Concentrate with Different Collectors
3.5. Crystallization Characteristics of Different Collectors
3.6. Crystallization Characteristics of the Collectors on the Flotation Concentrate Surface at Different Temperatures
4. Conclusions
- (1)
- The mixed (kerosene/MNap or kerosene/Nap) collectors display a stronger collecting ability for molybdenite than the individual kerosene. We obtained a maximum recovery of 89.01% and 85.74% using the kerosene/MNap as the collector at temperatures of 25 °C and 3 °C, while using the kerosene/Nap as the collector at the temperature of 25 °C and 3 °C we obtained 88.23% and 85.41%. The molybdenum recovery was also increased by 3–4% both at room temperature and low temperature.
- (2)
- The surface energy ( 44.50 mJ/m2) of polycyclic aromatic hydrocarbons was very close to that ( 42.55 mJ/m2) of the molybdenite {100} surface. Therefore, it can physically be adsorbed on the molybdenite {100} surface according to the principle of similar compatibility. The filtration velocity of flotation concentrate with kerosene mixed with auxiliary collector MNap as collector was basically the same as that obtained by using kerosene alone, and there was no crystallization phenomenon on the surface of flotation concentrate. This shows that MNap is suitable at various temperatures and its addition has no adverse effect for the use in industrial production in cold winter weather.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Castro, S.; Lopez-Valdivieso, A.; Laskowski, J.S. Review of the flotation of molybdenite. Part I: Surface properties and floatability. Int. J. Miner. Process. 2016, 148, 48–58. [Google Scholar] [CrossRef]
- Kelebek, S. Critical surface tension of wetting and of floatability of molybdenite and sulfur. J. Colloid Interface Sci. 1988, 124, 504–514. [Google Scholar] [CrossRef]
- Wan, H.; Yang, W.; He, T.S.; Yang, J.B.; Guo, L.; Peng, Y.J. The influence of Ca2+ and pH on the interaction between PAHs and molybdenite edges. Minerals 2017, 7, 104. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, H.J.; Qu, J.H.; Li, J.H. Two-Dimensional Layered Mos2: Rational Design, Properties and Electrochemical Applications. Energy Environ. Sci. 2016, 9, 1190–1209. [Google Scholar] [CrossRef]
- He, T.S.; Wan, H.; Song, N.P.; Guo, L. The influence of composition of nonpolar oil on flotation of molybdenite. Miner. Eng. 2011, 24, 1513–1516. [Google Scholar] [CrossRef]
- Smit, F.J.; Bhasin, A.K. Relationship of petroleum hydrocarbon characteristics and molybdenite flotation. Int. J. Miner. Process. 1985, 15, 19–40. [Google Scholar] [CrossRef]
- Crozier, R.D. Flotation reagent practice in primary and by-product molybdenite recovery. Min. Mag. 1979, 140, 174–178. [Google Scholar]
- Xia, Y.K.; Peng, F.F. Selection of frothers from residual organic reagents for copper-molybdenite sulfide flotation. Int. J. Miner. Process. 2007, 83, 68–75. [Google Scholar] [CrossRef]
- Kainuma, Y.; Uyeda, R. On the Structure of Adsorbed Organic Long-Chain Molecules on the Cleavage Surface of Molybdenite. J. Phys. Soc. Jpn. 1950, 5, 199–200. [Google Scholar] [CrossRef]
- Hoover, R.; Malhotra, D. Emulsion flotation of molybdenite. In Flotation—A. M. Gaudin Memorial; American Institute of Mining, Metallurgical, and Petroleum Engineers: New York, NY, USA, 1976; Volume 1, pp. 485–503. [Google Scholar]
- Groszek, A.J. Preferential adsorption of long-chain normal paraffins on MoS2, WS2 and graphite from n-heptane. Nature 1964, 104, 680. [Google Scholar]
- Groszek, A.J. Preferential adsorption of compounds with long methylene chains on cast iron, graphite, boron nitride, and molybdenum disulfide. ASLE Trans. 1966, 9, 67–76. [Google Scholar] [CrossRef]
- Groszek, A.J. Selective adsorption at graphite/hydrocarbon interfaces. Proc. R. Soc. Lond. A Math. Phys. Sci 1970, 314, 473–498. [Google Scholar]
- Tabares, J.O.; Ortega, I.M.; Bahena, J.R.; López, A.S.; Pérez, D.V.; Valdivieso, A.L. Surface Properties and Floatability of Molybdenite. In Proceedings of the 2006 China-Mexico Workshop on Minerals Particle Technology, San Luis Potosi, Mexico, 11–13 August 2006; pp. 115–124. [Google Scholar]
- Triffett, B.; Bradshaw, D. The role of morphology and host rock lithology on the flotation behaviour of molybdenite at Kennecott Utah Copper. In Proceedings of the 9th International Congress for Applied Mineralogy: ICAM 2008, Brisbane, Australia, 8–10 September 2008; pp. 465–473. [Google Scholar]
- Zanin, M.; Ametov, I.; Grano, S.; Zhou, L.; Skinner, W. A study of mechanisms affecting molybdenite recovery in a bulk copper/molybdenum flotation circuit. Int. J. Miner. Process. 2009, 93, 256–266. [Google Scholar] [CrossRef]
- Yang, B.Q.; Song, S.X.; Lopez-Valdivieso, A. Effect of Particle Size on the Contact Angle of Molybdenite Powders. Miner. Process. Extr. Metall. Rev. 2014, 35, 208–215. [Google Scholar] [CrossRef]
- Ronzio, R.A. The role of surfactants in the flotation of molybdenite at climax. J. Am. Oil Chem. Soc. 1970, 47, 504A. [Google Scholar] [CrossRef]
- Nishkov, I.; Beas, E.; Henriquez, C. Effect of Surfactant Oil Emulsification on the Flotation of Molybdenite for Porphyry Copper Ores. In Proceedings of the X Balkan Mineral Processing Congress, Varna, Bulgaria, 15–20 June 2003; pp. 205–211. [Google Scholar]
- Nishkov, I.; Lazarov, D.; Marinov, M.; Beas, E.; Henriquez, C. Surfactant-Hydrocarbon Oil Emulsions for Molybdenite Flotation. In Proceedings of the IV Meeting of the Southern Hemisphere on Mineral Technology and III Latin-American Congress on Froth Flotation, Concepción, Chile, 20–23 November 1994; pp. 319–329. [Google Scholar]
- Chander, S.; Fuerstenau, D.W. Electrochemical study of the molybdenite–potassium diethyldithiophosphate system. Trans. SME 1974, 265, 193–197. [Google Scholar]
- Chander, S.; Fuerstenau, D.W. The effect of potassium diethyldithiophosphate on the interfacial properties of molybdenite. Trans. IMM 1974, 83, C180–C185. [Google Scholar]
- Zhang, B.Y.; Zhong, H. Review on Research Progress of Molybdenite Flotation and its Collectors. Conserv. Util. Miner. Resour. 2010, 3, 51–54. [Google Scholar]
- He, T.S.; Guo, Y.F.; Wang, Y.B.; Zhang, C.H.; Li, H.; Shi, X. Optimization on roughing reagent system of molybdenum ore plant by polycyclic aromatic hydrocarbon. Min. Sci. Technol. 2017, 37, 34–37. [Google Scholar]
- He, T.S.; Li, H.; Jin, J.P.; Peng, Y.J.; Wan, H. Improving fine molybdenite flotation using a combination of aliphatic hydrocarbon oil and polycyclic aromatic hydrocarbon. Results Phys. 2019, 3, 1050–1055. [Google Scholar] [CrossRef]
- Chibowski, E.; Terpilowski, K. Surface free energy of polypropylene and polycarbonate solidifying at different solid surfaces. Appl. Surf. Sci. 2009, 256, 1573–1581. [Google Scholar] [CrossRef]
- Yin, Z.X.; Kong, H.; Wang, H.C.; Zhang, J.; Liao, Z.Y.; Li, Y. Discussion on surface tension and free energy of surface. Univ. Chem. 2016, 31, 77–82. [Google Scholar] [CrossRef]
- Fowkes, F.M. Role of Acid-Base Interfacial Bonding in Adhesion. J. Adhes. Sci. Technol. 1987, 1, 7–27. [Google Scholar] [CrossRef]
- Van Oss, C.J.; Chaudhury, M.K.; Good, R.J. Interfacial Lifshitz-van der Waals and polar interactions in macroscopic systems. Chem. Rev. 1988, 88, 927–941. [Google Scholar] [CrossRef]
- Chaudhury, M.K. Interfacial interaction between low-energy surfaces. Mater. Sci. Eng. R 1996, 16, 97–159. [Google Scholar] [CrossRef]
- Jańczuk, B.; Wójcik, W.; Zdziennicka, A. Determination of the components of the surface tension of some liquids from interfacial liquid-liquid tension measurements. J. Colloid Interface Sci. 1993, 157, 384–393. [Google Scholar] [CrossRef]
- Holländer, A. On the Selection of Test Liquids for the Evaluation of Acid-Base Properties of Solid Surfaces by Contact Angle Goniometry. J. Colloid Interface Sci. 1995, 169, 493–496. [Google Scholar] [CrossRef]
- Smedley, G.T.; Coles, D.E. A refractive tilting-plate technique for measurement of dynamic contact angles. J. Colloid Interface Sci. 2005, 286, 310–311. [Google Scholar] [CrossRef]
- Wang, H.; Chen, L.; Fu, J.G.; Hao, Y. Interface thermodynamics of molybdenite floatation system. J. Cent. S. Univ. (Sci. Technol.) 2007, 38, 893–899. [Google Scholar]
- Wei, Z.L.; Li, Y.B. Anisotropy of molybdenite surface and its effects on flotation mechanism. Conserv. Util. Miner. Resour. 2018, 3, 31–36. [Google Scholar]

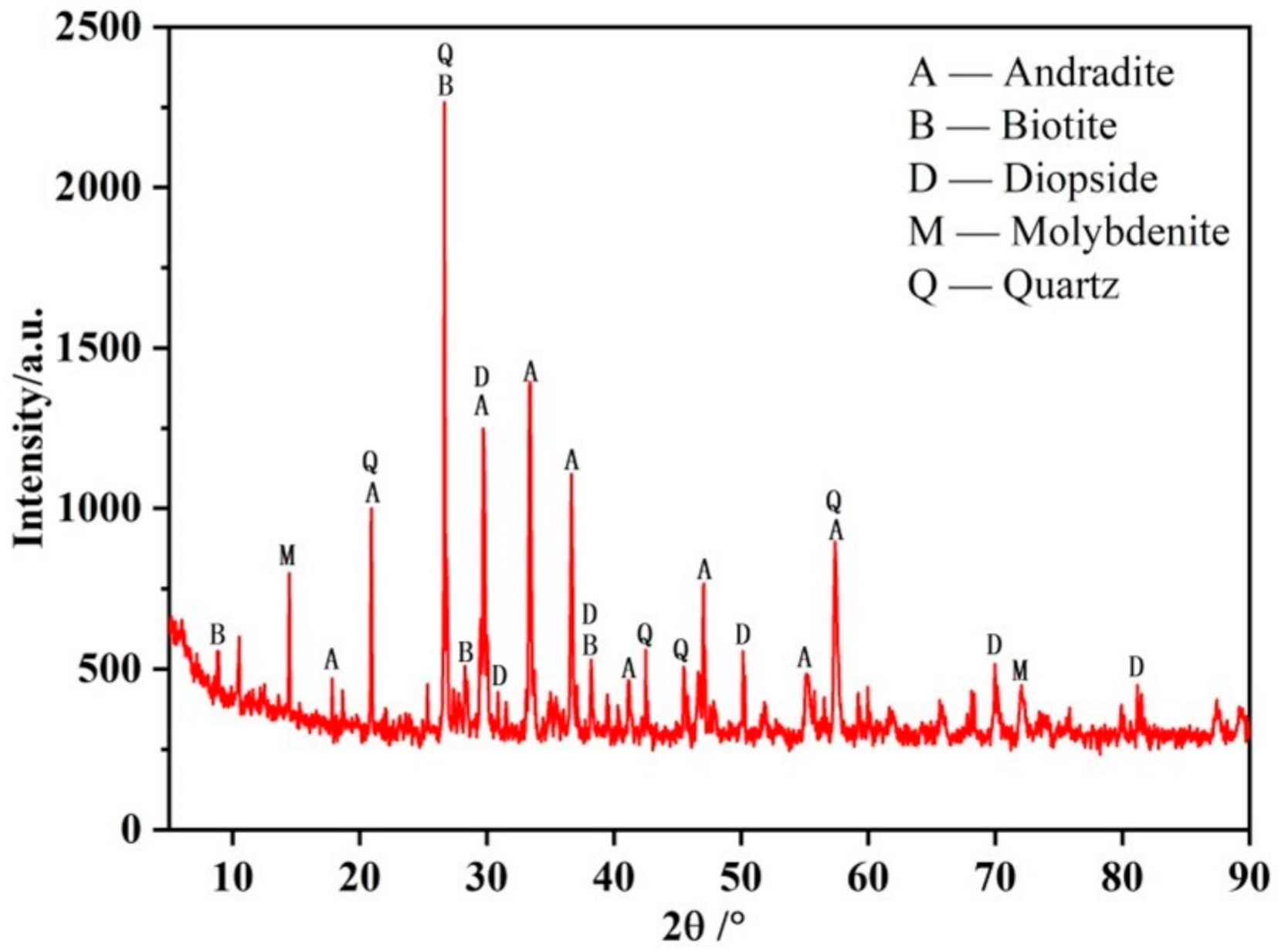

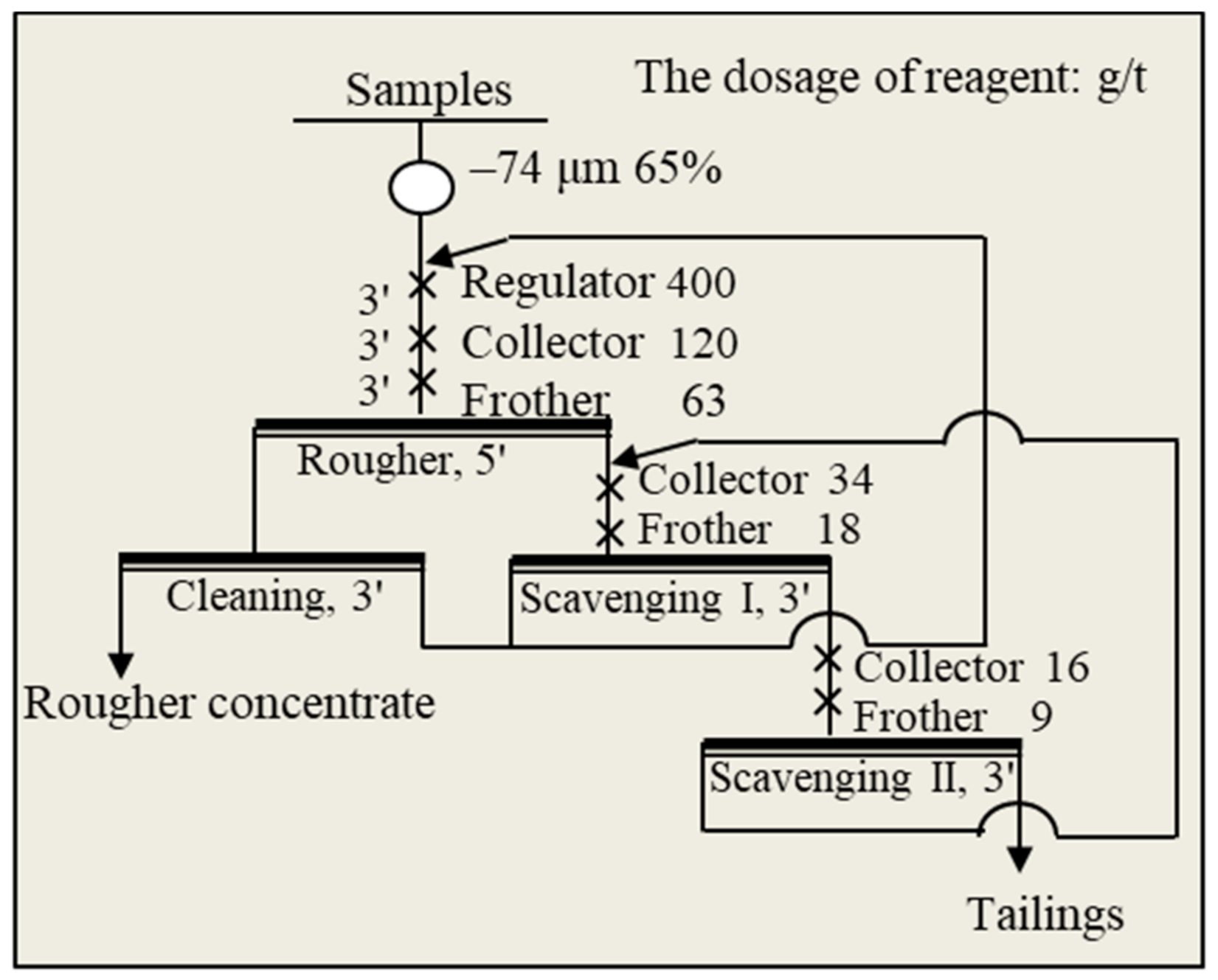



| Compositions | Mo | S | SiO2 | Cu | WO3 | TFe | Pb | Zn | Al2O3 | MgO |
|---|---|---|---|---|---|---|---|---|---|---|
| Content/% | 0.12 | 1.853 | 47.81 | 0.018 | 0.11 | 9.50 | 0.020 | 0.012 | 7.85 | 2.83 |
| Surface | Contact Angle/° | ||
|---|---|---|---|
| Distilled Water | Formamide | Di-Iodomethane | |
| {001} | 85.3 | 48.7 | 37.5 |
| {100} | 13.5 | 8.2 | 76.8 |
| Liquid for Detection | |||||
|---|---|---|---|---|---|
| Distilled water | 72.8 | 21.8 | 25.5 | 25.5 | 51.0 |
| Formamide | 58.0 | 39.0 | 2.28 | 39.6 | 19.0 |
| Di-iodomethane | 50.8 | 50.8 | 0.0 | 0.0 | 0.0 |
| Suface | |||||
|---|---|---|---|---|---|
| {001} | 42.55 | 40.84 | 1.28 | 0.57 | 1.71 |
| {100} | 195.18 | 19.16 | 39.72 | 195.01 | 176.02 |
| Surface | |||||
|---|---|---|---|---|---|
| Surface treated with kerosene | 44.50 | 41.97 | 0.12 | 4.62 | 2.53 |
| Surface treated with MNap | 198.53 | 18.38 | 41.46 | 195.69 | 180.15 |
| Type of Collectors | Product | Rate (%) | Grade (%) | Recovery (%) |
|---|---|---|---|---|
| Kerosene | Concentrate | 1.17 | 8.74 | 83.14 |
| Tailings | 98.83 | 0.021 | 16.86 | |
| Feed | 100.00 | 0.123 | 100.00 | |
| Kerosene:MNap 95:5 | Concentrate | 1.06 | 10.11 | 86.42 |
| Tailings | 98.94 | 0.017 | 13.58 | |
| Feed | 100.00 | 0.124 | 100.00 | |
| Kerosene:Nap 95:5 | Concentrate | 1.13 | 9.40 | 86.35 |
| Tailings | 98.87 | 0.017 | 13.65 | |
| Feed | 98.87 | 0.123 | 13.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; He, T.; Wan, H.; Han, Y.; Guo, Y.; Jin, J. Insights into Selection of the Auxiliary Collector and Its Applicability Analysis for Improving Molybdenite Flotation. Minerals 2021, 11, 528. https://doi.org/10.3390/min11050528
Li H, He T, Wan H, Han Y, Guo Y, Jin J. Insights into Selection of the Auxiliary Collector and Its Applicability Analysis for Improving Molybdenite Flotation. Minerals. 2021; 11(5):528. https://doi.org/10.3390/min11050528
Chicago/Turabian StyleLi, Hui, Tingshu He, He Wan, Yuexin Han, Yufeng Guo, and Jianping Jin. 2021. "Insights into Selection of the Auxiliary Collector and Its Applicability Analysis for Improving Molybdenite Flotation" Minerals 11, no. 5: 528. https://doi.org/10.3390/min11050528
APA StyleLi, H., He, T., Wan, H., Han, Y., Guo, Y., & Jin, J. (2021). Insights into Selection of the Auxiliary Collector and Its Applicability Analysis for Improving Molybdenite Flotation. Minerals, 11(5), 528. https://doi.org/10.3390/min11050528






