Critical Minerals from Post-Processing Tailing. A Case Study from Bangka Island, Indonesia
Abstract
1. Introduction
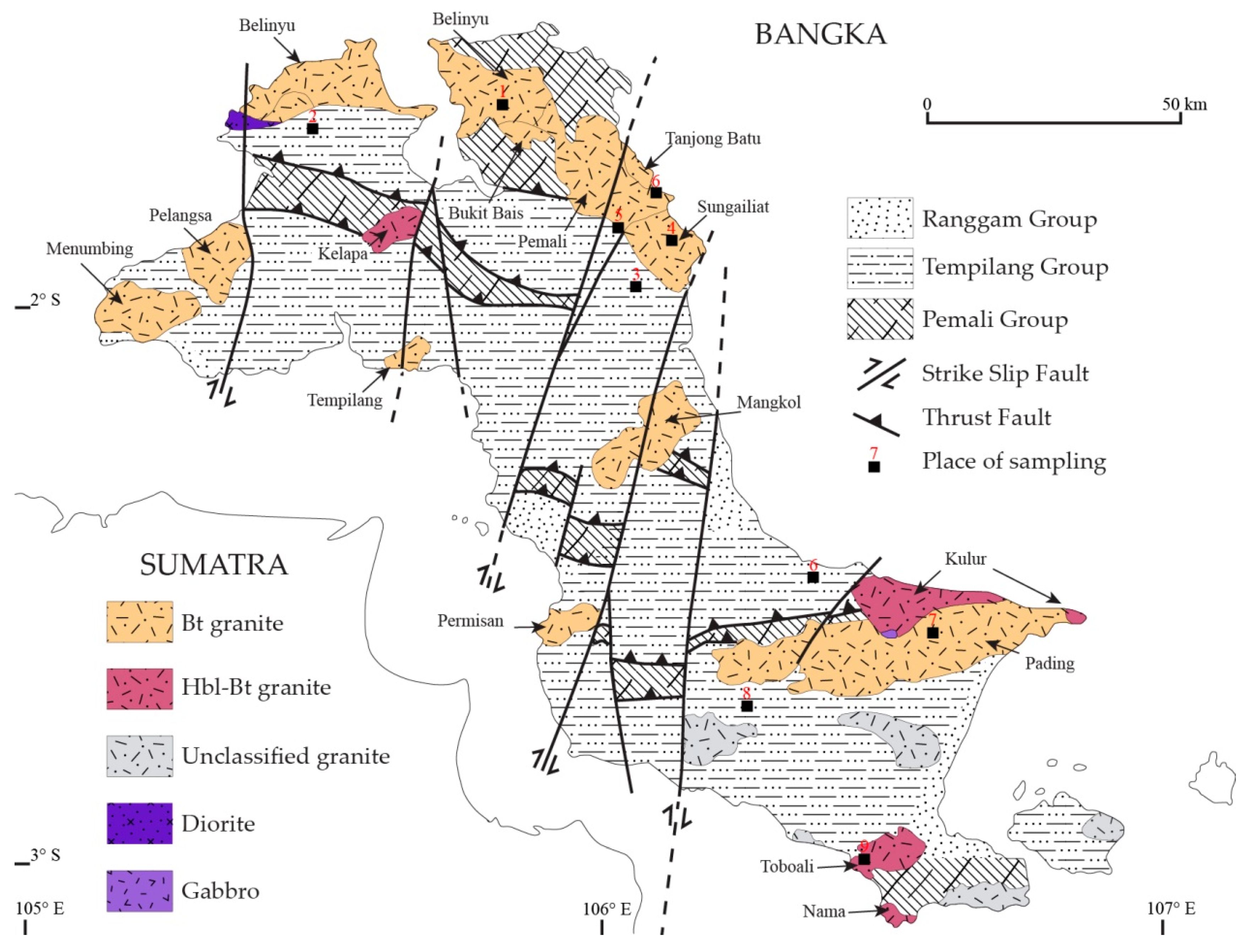
2. Geological Setting
3. Materials and Methods
3.1. Sampling
3.2. Analytical Methods
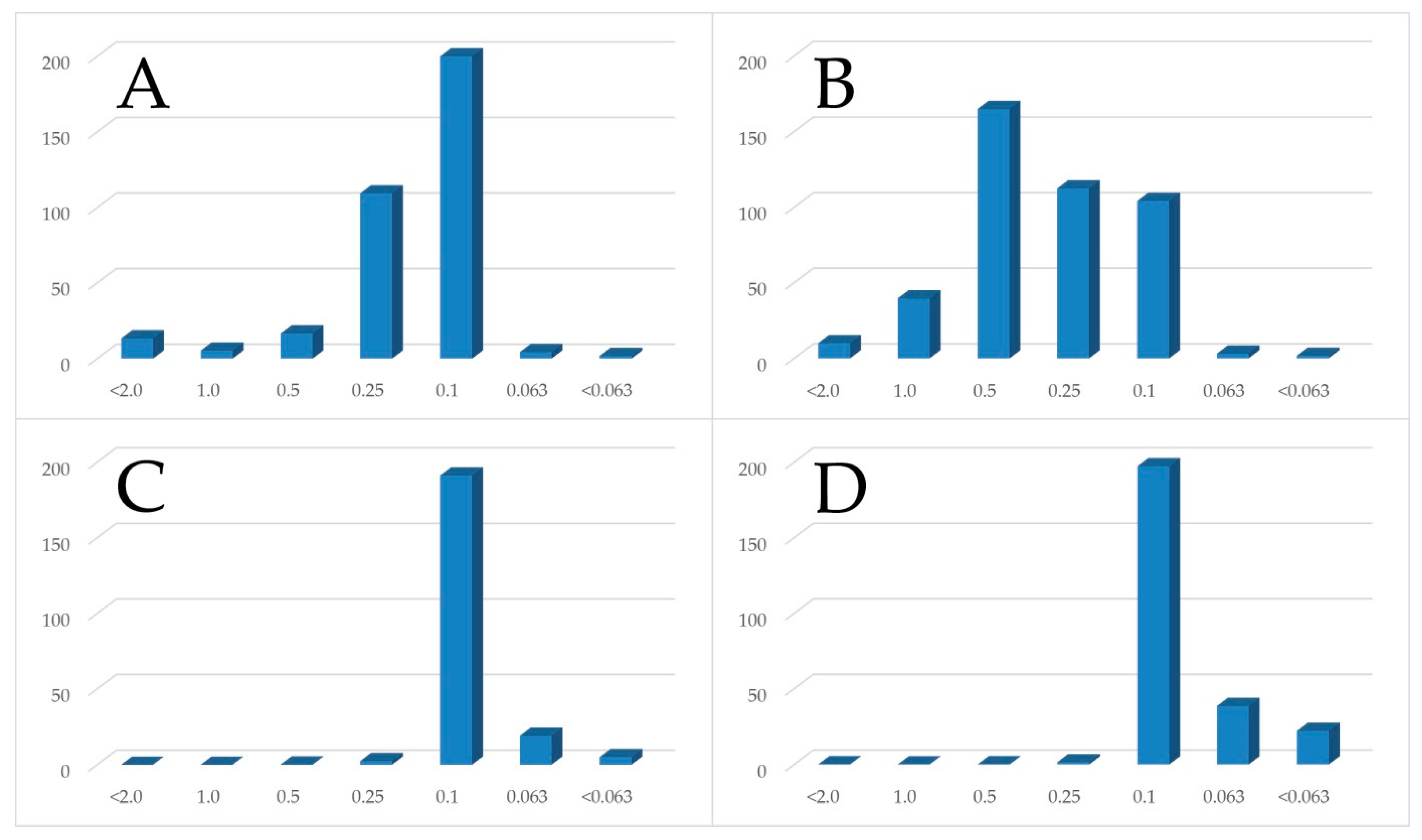
3.2.1. Grain Size Distribution
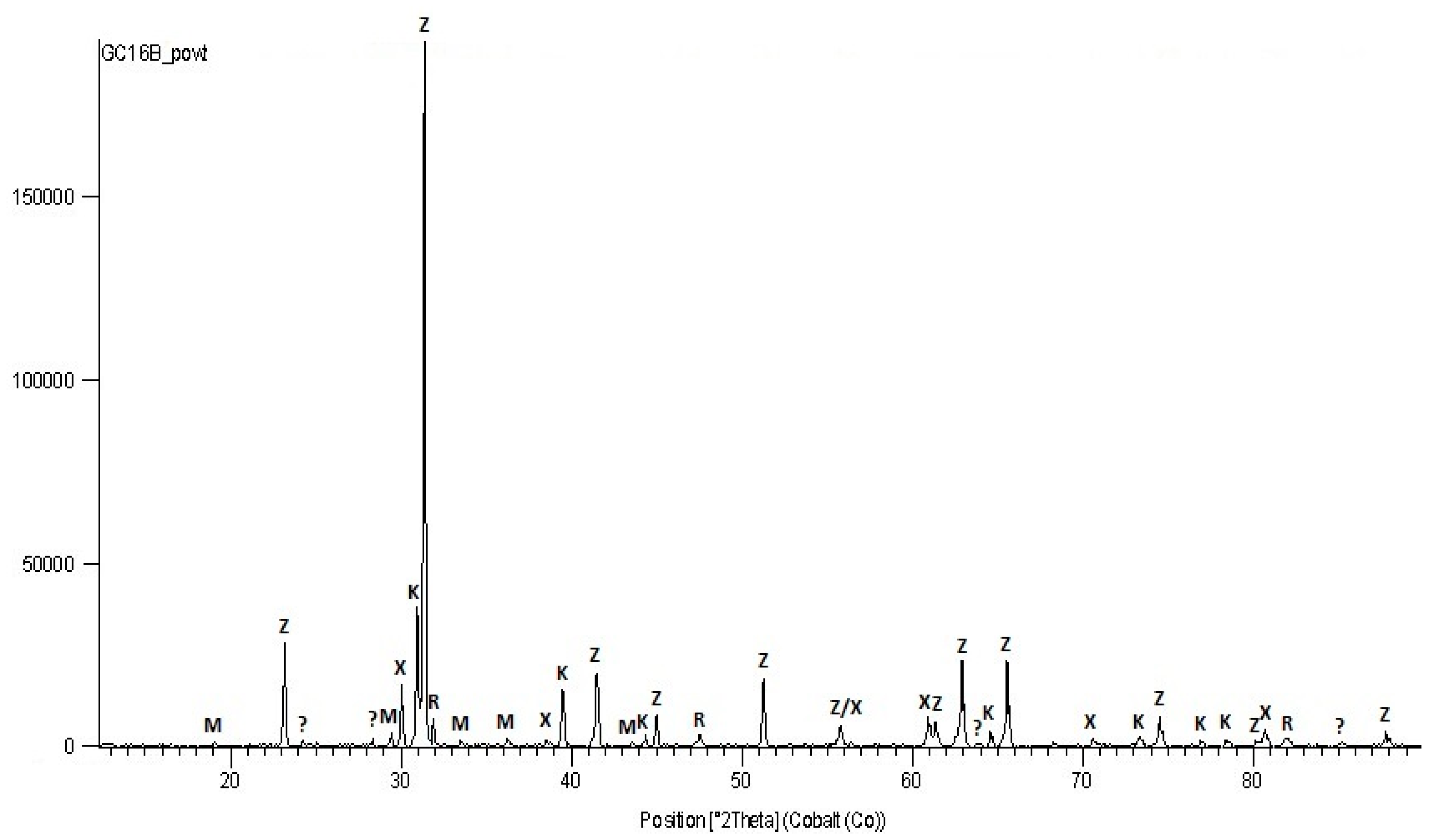
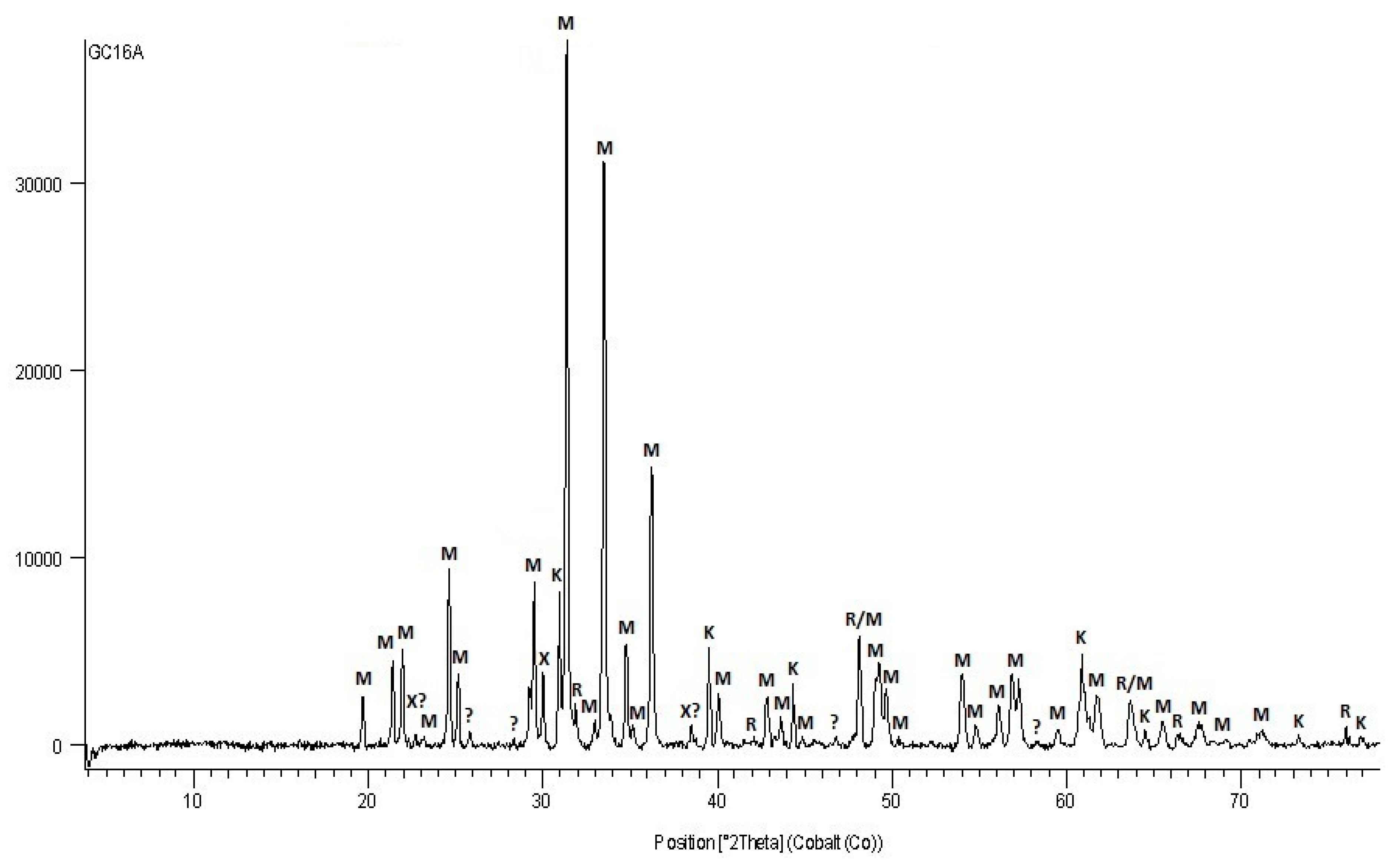
3.2.2. X-ray Diffraction
3.2.3. Microscopic Observations
3.2.4. Chemical Composition
4. Results
4.1. Grain Size and Macroscopic Description of Tailings Samples
4.2. Mineral Composition of Tailing
4.3. Chemical Composition of Tailing
5. Discussion
5.1. Critical and Other Raw Materials from Tailing
5.2. Potential Resources of Critical Raw Materials from Bangka Island
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goodenough, K.M.; Wall, F.; Merriman, D. The rare earth elements: Demand, global resources, and challenges for resourcing future generations. Nat. Resour. Res. 2018, 27, 201–216. [Google Scholar] [CrossRef]
- Zhou, B.; Li, Z.; Chen, C. Global potential of rare earth resources and rare earth demand from clean technologies. Minerals 2017, 7, 203. [Google Scholar] [CrossRef]
- European Commission. Report on Critical Raw Materials in the Circular Economy; European Commission: Brussels, Belgium, 2018. [Google Scholar]
- Galos, K.; Lewicka, E.; Burkowicz, A.; Guzik, K.; Kot-Niewiadomska, A.; Kamyk, J.; Szlugaj, J. Approach to identification and classification of the key, strategic and critical minerals important for the mineral security of Poland. Resour. Policy 2020, 101900. [Google Scholar] [CrossRef]
- European Commission. Communication from the Commission to the European Parlament, the Council, the European Economic and Social Committee and the Committee of the Regions. Tackling the Challenges in Commodity Markets and on Raw Materials; 2.2.2011 COM (2011) 25 Final; European Commission: Brussels, Belgium, 2011. [Google Scholar]
- European Commission. Communication from the Commission to the European Parlament, the Council, the European Economic and Social Committee and the Committee of the Regions. On the Review of the List of Critical Raw Materials for the EU and the Implementation of the Raw Materials Initiative; 26.5.2014 COM (2014) 297 Final; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- European Commission. Communication from the Commission to the European Parlament, the Council, the European Economic and Social Committee and the Committee of the Regions. On the 2017 List of Critical Raw Materials for the EU; 13.9.2017 COM (2017) 490 Final; European Commission: Brussels, Belgium, 2017. [Google Scholar]
- European Commission. Communication from the Commission to the European Parlament, the Council, the European Economic and Social Committee and the Committee of the Regions. Critical Raw Materials Resilience: Charting a Path Towards Greater Security and Sustainability; 3.9.2020 COM (2020) 474 Final; European Commission: Brussels, Belgium, 2020. [Google Scholar]
- European Commission. Communication from the Commission to the European Parlament, the Council, the European Economic and Social Committee and the Committee of the Regions. The European Green Deal; 11.12.2019 COM (2019) 640 Final; European Commission: Brussels, Belgium, 2019. [Google Scholar]
- EuroGeoSurveys. Strategic Research & Innovation Agenda; Draft; EuroGeoSurveys: Brussels, Belgium, 2021. [Google Scholar]
- Szamałek, K.; Konopka, G.; Zglinicki, K.; Marciniak-Maliszewska, B. New potential source of rare earth elements. Miner. Resour. Manag. 2013, 29, 59–76. [Google Scholar] [CrossRef]
- Grilli, M.L.; Bellezze, T.; Gamsjäger, E.; Rinaldi, A.; Novak, P.; Balos, S.; Piticescu, R.R.; Ruello, M.L. Solutions for critical raw materials under extreme conditions: A review. Materials 2017, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Edraki, M.; Baumgartl, T.; Manlapig, E.; Bradshaw, D.; Franks, D.M.; Moran, C.J. Designing mine tailings for better environmental, social and economic outcomes: A review of alternative approaches. J. Clean. Prod. 2014, 84, 411–420. [Google Scholar] [CrossRef]
- Naicker, K.; Cukrowska, E.; McCarthy, T.S. Acid mine drainage from gold mining activities in Johannesburg, South Africa and environs. Environ. Pollut. 2003, 122, 29–40. [Google Scholar] [CrossRef]
- Mehta, N.; Dino, G.A.; Passarella, I.; Ajmone-Marsan, F.; Rossetti, P.; De Luca, D.A. Assessment of the possible reuse of extractive waste coming from abandoned mine sites: Case study in Gorno, Italy. Sustainability 2020, 12, 2471. [Google Scholar] [CrossRef]
- Dino, G.A.; Mehta, N.; Rossetti, P.; Ajmone-Marsan, F.; De Luca, D.A. Sustainable approach towards extractive waste management: Two case studies from Italy. Resour. Policy 2018, 59, 33–43. [Google Scholar] [CrossRef]
- Mehta, N.; Dino, G.A.; Ajmone-Marsan, F.; Lasagna, M.; Romè, C.; De Luca, D.A. Extractive waste management: A risk analysis approach. Sci. Total Environ. 2018, 622–623, 900–912. [Google Scholar] [CrossRef]
- Schwartz, M.O.; Surjono. The Pemali tin deposit, Bangka, Indonesia. Miner. Depos. 1991, 26, 18–25. [Google Scholar] [CrossRef]
- Schwartz, M.O.; Rajah, S.S.; Askury, S.K.; Putthapiban, P.; Djaswadi, S. The Southeast Asian Tin Belt. Earth-Sci. Rev. 1995, 38, 95–293. [Google Scholar] [CrossRef]
- Metcalfe, I. Permian tectonic framework and paleogeography of SE Asia. J. Asian Earth Sci. 2002, 20, 551–556. [Google Scholar] [CrossRef]
- Searle, M.P.; Whitehouse, M.J.; Robb, L.J.; Ghani, A.A.; Hutchison, C.S.; Sone, M.; Wai-Pan Ng, S.; Roselee, M.H.; Chung, S.-L.; Oliver, G.J.H. Tectonic evolution of the Sibumasu–Indochina terrane collision zone in Thailand and Malaysia: Constraints from new U–Pb zircon chronology of SE Asian tin granitoids. J. Geol. Soc. Lond. 2012, 169, 489–500. [Google Scholar] [CrossRef]
- Hutchison, C.S.; Taylor, D. Metallogenesis in SE Asia. J. Geol. Soc. Lond. 1978, 135, 405–428. [Google Scholar] [CrossRef]
- Cobbing, E.J.; Pitfield, P.E.J.; Darbyshire, D.P.F.; Mallick, D.I.J. The Granites of the South-East Asian Tin Belt. J. Geol. Soc. 1986, 143, 537–550. [Google Scholar] [CrossRef]
- Wai-Pan Ng, S.; Whitehouse, M.J.; Roselee, M.H.; Teschner, C.; Murtadha, S.; Oliver, G.J.H.; Ghani, A.A.; Chang, S. Late Triassic granites from Bangka, Indonesia: A continuation of the Main Range granite province of the South-East Asian Tin Belt. J. Asian Earth Sci. 2017, 138, 548–561. [Google Scholar]
- Ko, U.K. Preliminary synthesis of the geology of Bangka Island, Indonesia. Bull. Geol. Soc. Malays. 1986, 20, 81–96. [Google Scholar]
- Priem, H.N.A.; Boelrijk, N.A.I.M.; Bon, E.H.; Hebeda, E.H.; Verdurmen, E.A.T.; Verschure, R.H. Isotope Geochronology in the Indonesian Tin Belt. Geol. Mijnb. 1975, 54, 61–70. [Google Scholar]
- Aleva, G.J.J. The plutonic igneous rocks from Billiton. Indonesia. Geol. Mijnb. 1960, 39, 427–436. [Google Scholar]
- Wikarno, U.; Suyatna, D.A.D.; Sukardi, S. Granitoids of Sumatra and the Tin Islands. In Geology of Tin Deposits in Asia and the Pacific, Proceedings of the International Symposium on the Geology of Tin Deposits held in Nanning, Nanning, China, 26–30 October 1984; Hutchison, C.S., Ed.; Springer: Berlin/Heidelberg, Germany, 1984. [Google Scholar]
- Ishihara, S. The magnetite-series and ilmenite-series granitic rocks. Min. Geol. 1977, 27, 293–305. [Google Scholar] [CrossRef]
- Wentworth, C.K. A scale of grade and class terms for clastic sediments. J. Geol. 1922, 30, 377–392. [Google Scholar] [CrossRef]
- Blott, S.J.; Pye, K. GRADISTAT: A grain size distribution and statistics package for the analysis of unconsolidated sediments. Earth Surf. Process. Landf. 2001, 26, 1237–1248. [Google Scholar] [CrossRef]
- Lottermoser, B. Mine Wastes. Characterization, Treatment and Environmental Impacts, 3rd ed; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–400. [Google Scholar]
- BRGM. Management of Mining, Quarrying and Ore-Processing Waste in the European Union; European Commision: Brussels, Belgium, 2001; pp. 1–79. [Google Scholar]
- Nurtjahya, E.; Franklin, J.; Umroh; Agustina, F. The Impact of tin mining in Bangka Belitung and its reclamation studies. In Applied Technology for Sustainable Environment, Proceedings of the Sriwijaya International Conference on Engineering, Science and Technology (SICEST 2016), Bangka Island, Indonesia, 9–10 November 2016; Iskandar, I., Ismadji, S., Agustina, T.E., Yani, I., Komariah, L.N., Hasyim, S., Eds.; EDP Sciences: Les Ulis, France, 2017; pp. 1–6. [Google Scholar] [CrossRef]
- Irzon, R.; Sendjadja, P.; Kurnia; Imtihanah; Soebandrio, J. Kandungan rare earth elements dalam tailing tambang timah di pulau Singkep. J. Geol. Sumberd. Miner. 2014, 15, 143–151. [Google Scholar]
- Euratom Supply Agency. Annual Report 2019; Euratom Supply Agency: Luxemburg, 2019; p. 78. [Google Scholar] [CrossRef]
- Tulsidas, H.; Gabriel, S.; Kiegiel, K.; Haneklaus, N. Uranium resources in EU phosphate rock imports. Resour. Policy 2019, 61, 151–156. [Google Scholar] [CrossRef]
- Soetopo, B.; Subiantoro, L.; Sularto, P.; dan Haryanto, D. The study of monazite and zircon in quarternary rocks in cerucuk belitung. Eksplorium 2015, 33, 25–40. [Google Scholar]
- Pearce, J.A.; Harris, N.B.W.; Tindle, A.G. Trace element discrimination diagrams for the tectonic interpretation of granitic rocks. J. Petrol. 1984, 25, 956–983. [Google Scholar] [CrossRef]
- Harjanto, S.; Virdhian, S.; Afrilinda, E. Characterization of Indonesia Rare Earth Minerals and Their Potential Processing Techniques. J. Rare Earth. 2013, 52, 99–108. [Google Scholar]
- Aryanto, N.C.D.; Kamiludin, U. The Content of Placer Heavy Mineral and Characteristics of REE at Toboali Coast and Its Surrounding Area. Bangka Belitung Province. Bull. Mar. Geol. 2016, 31, 45–54. [Google Scholar] [CrossRef][Green Version]
- Chesworth, W.; Dejou, J.; Larroque, P. The weathering of basalt and relative mobil-ities of the major elements at Belbex, France. Geochim. Cosmochim. Acta 1981, 45, 1235–1243. [Google Scholar] [CrossRef]
- Nesbitt, H.W.; Markovics, G. Weathering of granodioritic crust, long-term storage ofelements in weathering profiles, and petrogenesis of siliciclastic sediments. Geochim. Cosmochim. Acta 1997, 61, 1653–1670. [Google Scholar] [CrossRef]
- Nesbitt, H.W. Mobility and fractionation of rare earth elements during weatheringof a granodiorite. Nature 1979, 279, 206–210. [Google Scholar] [CrossRef]
- He, B.; Xu, Y.G.; Chung, S.L.; Xiao, L.; Wang, Y. Sedimentary evidence for a rapid, kilometer-scale crustal doming prior to the eruption of the Emeishan flood basalts. Earth Planet. Sci. Lett. 2003, 213, 391–405. [Google Scholar] [CrossRef]
- Zhang, Z.; Zheng, G.; Takahashi, Y.; Wu, C.; Zheng, C.; Yao, J.; Xiao, C. Extreme enrichment of rare earth elements in hard clay rocks and its potential as a resource. Ore Geol. Rev. 2016, 72, 191–212. [Google Scholar] [CrossRef]
- Estrade, G.; Marquis, E.; Smith, M.; Goodenough, K.; Nason, P. REE concentration processes in ion adsorption deposits: Evidence from the Ambohimirahavavy alkaline complex in Madagascar. Ore Geol. Rev. 2019, 112, 103027. [Google Scholar] [CrossRef]




| Sample | Bngk2 | Bngk4 | Bngk6 | Bngk7 | Bngk11 | Bngk12 | Bngk13 | Bngk14 | Bngk16 | Bngk18 | Bngk21 | Bngk23 | Bngk25 | Bngk27 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oxide | in wt% | |||||||||||||
| SiO2 | 26.51 | 20.43 | 47.33 | 20.33 | 50.36 | 31.39 | 14.23 | 3.75 | 17.82 | 25.37 | 2.81 | 27.48 | 18.65 | 32.66 |
| Al2O3 | 2.00 | 9.41 | 16.18 | 0.40 | 12.80 | 1.15 | 1.64 | 1.34 | 2.67 | 1.89 | 1.16 | 10.14 | 1.00 | 4.64 |
| Fe2O3 | 14.79 | 19.03 | 12.30 | 4.77 | 12.58 | 12.13 | 16.27 | 30.40 | 21.12 | 15.08 | 5.20 | 15.41 | 11.48 | 13.26 |
| MgO | 0.20 | 0.16 | 0.40 | 0.03 | 4.42 | 0.13 | 0.03 | 0.05 | 0.11 | 0.17 | <0.01 | 0.32 | 0.45 | 0.12 |
| CaO | 0.06 | 0.03 | 0.14 | 0.05 | 0.02 | 0.06 | 0.02 | 0.02 | 0.03 | 0.06 | 0.28 | 0.11 | 0.05 | 0.04 |
| Na2O | 0.08 | 0.35 | 1.05 | 0.01 | 0.05 | 0.04 | 0.01 | 0.01 | 0.06 | 0.07 | 0.02 | 0.66 | 0.02 | 0.17 |
| K2O | 0.03 | 0.03 | 0.07 | <0.01 | 0.01 | <0.01 | <0.01 | <0.01 | <0.01 | 0.02 | <0.01 | 0.10 | 0.07 | 0.07 |
| TiO2 | 23.97 | 41.82 | 9.31 | 6.04 | 7.68 | 18.65 | 48.51 | 51.26 | 25.50 | 25.43 | 3.74 | 20.89 | 13.48 | 32.68 |
| P2O5 | 1.76 | 0.58 | 0.39 | 0.34 | 0.42 | 1.15 | 1.91 | 1.73 | 2.26 | 1.89 | 17.23 | 1.91 | 2.01 | 1.08 |
| MnO | 1.16 | 1.85 | 0.47 | 0.10 | 0.39 | 0.92 | 1.35 | 2.46 | 1.11 | 1.20 | 0.15 | 1.00 | 0.19 | 0.96 |
| Cr2O3 | 0.27 | 0.02 | 0.03 | 0.20 | 3.63 | 0.17 | 0.07 | 0.08 | 0.08 | 0.26 | 0.43 | 0.12 | 1.63 | 0.08 |
| LOI | 2.10 | 2.00 | 3.70 | 1.90 | 0.70 | 4.20 | 2.18 | 1.40 | 11.50 | 1.70 | 3.70 | 1.80 | 3.40 | 2.00 |
| Total | 72.94 | 95.78 | 91.41 | 34.17 | 93.16 | 70.02 | 86.21 | 92.58 | 82.25 | 73.17 | 34.77 | 87.76 | 78.36 | 96.10 |
| As | 0.02 | 0.02 | <0.01 | 0.03 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.03 | 0.23 | 0.03 | <0.01 | 0.01 |
| Pb | <0.03 | <0.03 | <0.03 | 0.07 | 0.05 | <0.03 | 0.04 | <0.03 | 0.05 | <0.03 | 0.06 | <0.03 | <0.03 | <0.03 |
| Zn | 0.02 | <0.01 | 0.06 | 0.01 | 0.03 | 0.03 | 0.01 | <0.01 | 0.05 | 0.02 | 0.01 | <0.01 | 0.02 | <0.01 |
| S | 0.23 | 0.07 | 0.50 | 1.70 | 9.15 | 2.41 | 0.02 | 0.20 | 2.78 | 0.26 | 1.72 | 0.21 | 3.34 | 0.24 |
| TOT/C | 0.06 | 0.05 | 0.06 | 0.05 | 0.07 | 0.04 | 0.10 | 0.05 | 0.10 | 0.04 | 0.06 | 0.04 | 0.06 | 0.04 |
| TOT/S | 0.40 | 0.08 | 0.69 | 1.69 | 0.03 | 1.76 | 0.04 | 0.20 | 6.12 | 0.25 | 1.65 | 0.22 | 0.96 | 0.23 |
| Element | in ppm/ppb | |||||||||||||
| Cd [ppm] | 0.03 | 0.09 | 0.04 | 0.06 | 0.02 | 0.02 | 0.01 | 0.10 | 0.22 | 0.03 | 0.04 | 0.12 | 0.02 | 0.04 |
| Sb [ppm] | 3.62 | 1.48 | 1.02 | 10.53 | 0.26 | 1.85 | 4.56 | 3.50 | 2.34 | 3.00 | 3.63 | 2.11 | 1.82 | 2.72 |
| Hg [ppb] | 169 | 203 | 131 | 188 | 43 | 173 | 3121 | 439 | 774 | 183 | 426 | 312 | 102 | 432 |
| in wt% | ||||||||||||||
| Ga | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0.00 | 0.03 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Hf | 0.36 | 0.03 | 0.04 | 0.61 | 0.08 | 0.42 | 0.14 | 0.04 | 0.23 | 0.36 | 0.01 | 0.19 | 0.64 | 0.07 |
| Nb | 0.09 | 0.23 | 0.12 | 0.01 | 0.03 | 0.05 | 0.16 | 0.17 | 0.15 | 0.09 | 0.04 | 0.06 | 0.04 | 0.14 |
| Ta | 0.01 | 0.02 | 0.08 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.03 | 0.01 | 0.02 |
| Sn | 3.89 | 0.50 | 2.96 | 11.64 | 6.72 | 3.98 | 1.58 | 0.87 | 1.03 | 3.68 | 5.89 | 1.23 | 5.14 | 0.83 |
| Th | 0.16 | 0.17 | 0.03 | 0.17 | 0.03 | 0.14 | 0.42 | 0.28 | 0.40 | 0.17 | 1.00 | 0.30 | 0.05 | 0.12 |
| U | 0.06 | 0.02 | 0.02 | 0.04 | 0.01 | 0.04 | 0.04 | 0.03 | 0.08 | 0.07 | 0.13 | 0.06 | 0.04 | 0.02 |
| W | 0.02 | 0.14 | 0.03 | 0.03 | 0.01 | 0.01 | 0.10 | 0.08 | 0.05 | 0.02 | 0.06 | 0.02 | 0.02 | 0.08 |
| Zr | >5.0 | 0.90 | 0.90 | >5.0 | 2.95 | >5.0 | >5.0 | 1.15 | >5.0 | >5.0 | 0.61 | >5.0 | >5.0 | 2.27 |
| Sc | 0.01 | 0.01 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| Y | 1.05 | 0.27 | 0.24 | 0.46 | 0.17 | 0.40 | 0.49 | 0.50 | 0.51 | 1.20 | 1.10 | 1.06 | 0.57 | 0.44 |
| La | 0.30 | 0.15 | 0.04 | 0.34 | 0.07 | 0.33 | 0.66 | 0.57 | 0.72 | 0.27 | >5.0 | 0.71 | 0.07 | 0.22 |
| Ce | 0.58 | 0.33 | 0.09 | 0.72 | 0.15 | 0.68 | 1.35 | 1.13 | 1.55 | 0.54 | >5.0 | 1.61 | 0.14 | 0.46 |
| Pr | 0.06 | 0.04 | 0.01 | 0.08 | 0.02 | 0.08 | 0.14 | 0.13 | 0.16 | 0.06 | >1.0 | 0.16 | 0.02 | 0.05 |
| Nd | 0.23 | 0.13 | 0.04 | 0.028 | 0.07 | 0.25 | 0.47 | 0.44 | 0.53 | 0.21 | >1.0 | 0.58 | 0.06 | 0.18 |
| Sm | 0.06 | 0.03 | 0.01 | 0.06 | 0.02 | 0.05 | 0.08 | 0.09 | 0.09 | 0.06 | >1.0 | 0.12 | 0.02 | 0.04 |
| Eu (ppm) | 8.4 | 0.9 | 1.6 | 7.4 | 1.4 | 5.3 | 4.6 | 4.4 | 5.2 | 8.9 | 61.8 | 8.0 | 6.1 | 3.0 |
| Gd | 0.10 | 0.03 | 0.02 | 0.06 | 0.02 | 0.05 | 0.07 | 0.09 | 0.08 | 0.12 | 0.75 | 0.13 | 0.04 | 0.05 |
| Tb | 0.02 | 0.01 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.03 | 0.08 | 0.03 | 0.01 | 0.01 |
| Dy | 0.17 | 0.04 | 0.04 | 0.08 | 0.03 | 0.06 | 0.08 | 0.09 | 0.08 | 0.19 | 0.34 | 0.18 | 0.09 | 0.07 |
| Ho | 0.04 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 | 0.02 | 0.02 | 0.02 | 0.05 | 0.04 | 0.04 | 0.02 | 0.02 |
| Er | 0.13 | 0.03 | 0.03 | 0.06 | 0.02 | 0.05 | 0.06 | 0.06 | 0.06 | 0.15 | 0.10 | 0.13 | 0.07 | 0.05 |
| Tm | 0.02 | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 |
| Yb | 0.14 | 0.03 | 0.04 | 0.06 | 0.03 | 0.05 | 0.06 | 0.05 | 0.07 | 0.15 | 0.07 | 0.14 | 0.08 | 0.05 |
| Lu | 0.02 | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 |
| ∑REE+Y | 2.93 | 1.10 | 0.60 | 2.23 | 0.62 | 2.04 | 3.50 | 3.20 | 3.92 | 3.08 | >15.51 | 4.93 | 1.20 | 1.66 |
| LREE | 1.23 | 0.67 | 0.20 | 1.47 | 0.32 | .38 | 2.70 | 2.35 | 3.06 | 1.15 | >13.00 | 3.18 | 0.29 | 0.95 |
| HREE+Y | 1.70 | 0.43 | 0.40 | 0.76 | 0.30 | 0.66 | 0.80 | 0.84 | 0.85 | 1.93 | 2.51 | 1.74 | 0.91 | 0.71 |
| LREE:HREE | 0.72 | 1.57 | 0.50 | 1.93 | 1.09 | 2.10 | 3.39 | 2.80 | 3.59 | 0.60 | >5.17 | 1.83 | 0.32 | 1.35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zglinicki, K.; Szamałek, K.; Wołkowicz, S. Critical Minerals from Post-Processing Tailing. A Case Study from Bangka Island, Indonesia. Minerals 2021, 11, 352. https://doi.org/10.3390/min11040352
Zglinicki K, Szamałek K, Wołkowicz S. Critical Minerals from Post-Processing Tailing. A Case Study from Bangka Island, Indonesia. Minerals. 2021; 11(4):352. https://doi.org/10.3390/min11040352
Chicago/Turabian StyleZglinicki, Karol, Krzysztof Szamałek, and Stanisław Wołkowicz. 2021. "Critical Minerals from Post-Processing Tailing. A Case Study from Bangka Island, Indonesia" Minerals 11, no. 4: 352. https://doi.org/10.3390/min11040352
APA StyleZglinicki, K., Szamałek, K., & Wołkowicz, S. (2021). Critical Minerals from Post-Processing Tailing. A Case Study from Bangka Island, Indonesia. Minerals, 11(4), 352. https://doi.org/10.3390/min11040352