The Separation of Carbonaceous Matter from Refractory Gold Ore Using Multi-Stage Flotation: A Case Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
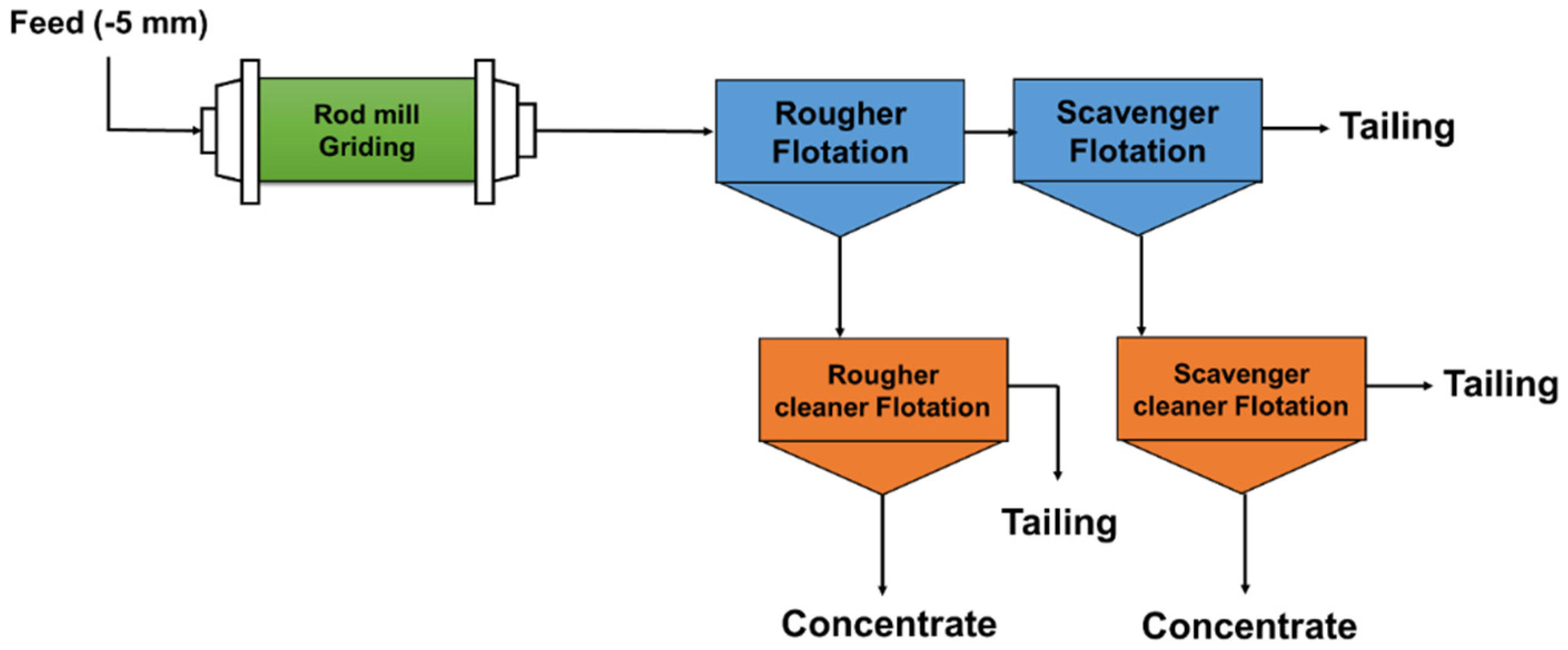
2.2. Method
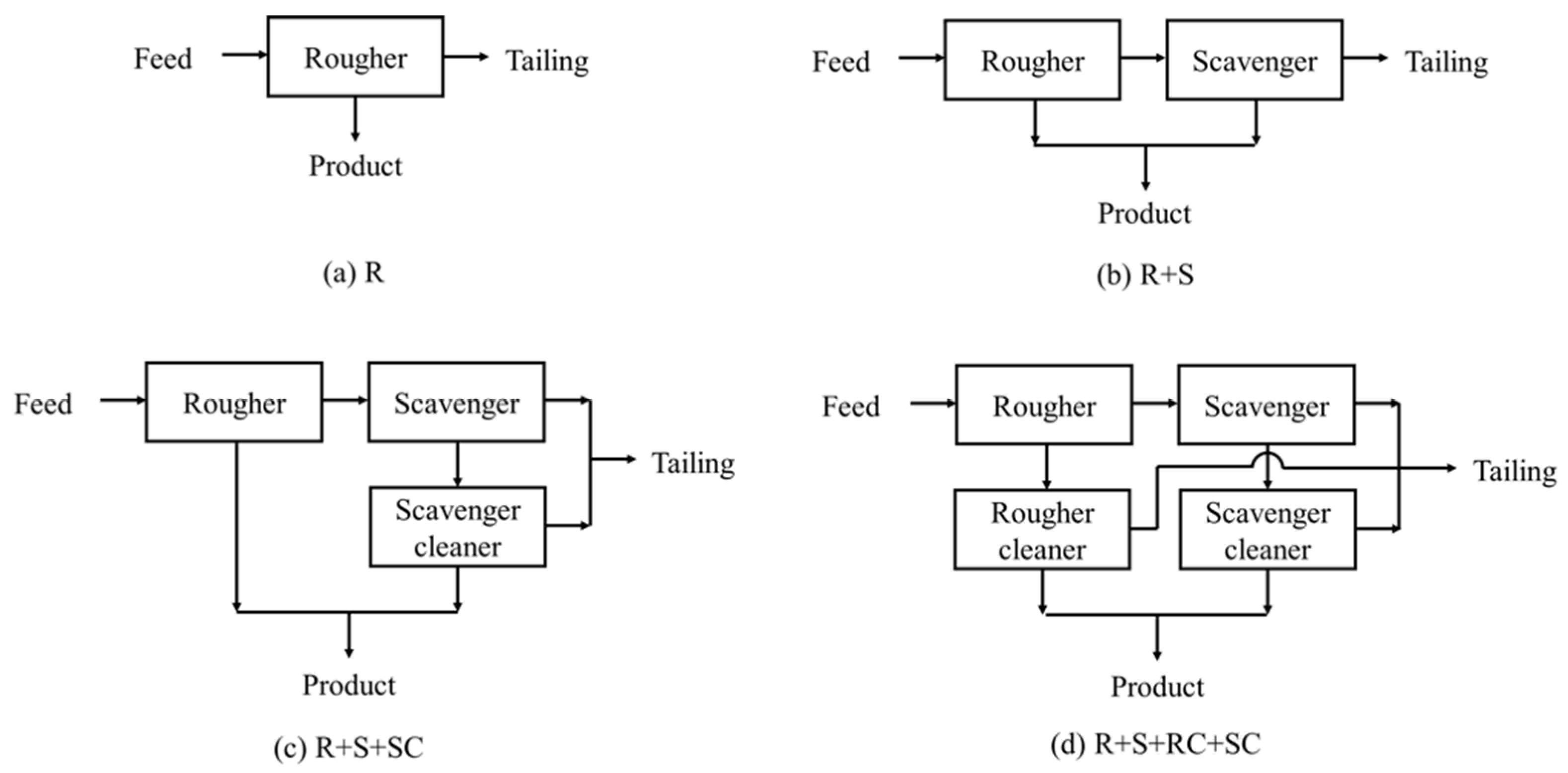
2.2.1. Rougher Flotation
2.2.2. Scavenger Flotation
2.2.3. Cleaner Flotation
3. Results and Discussion
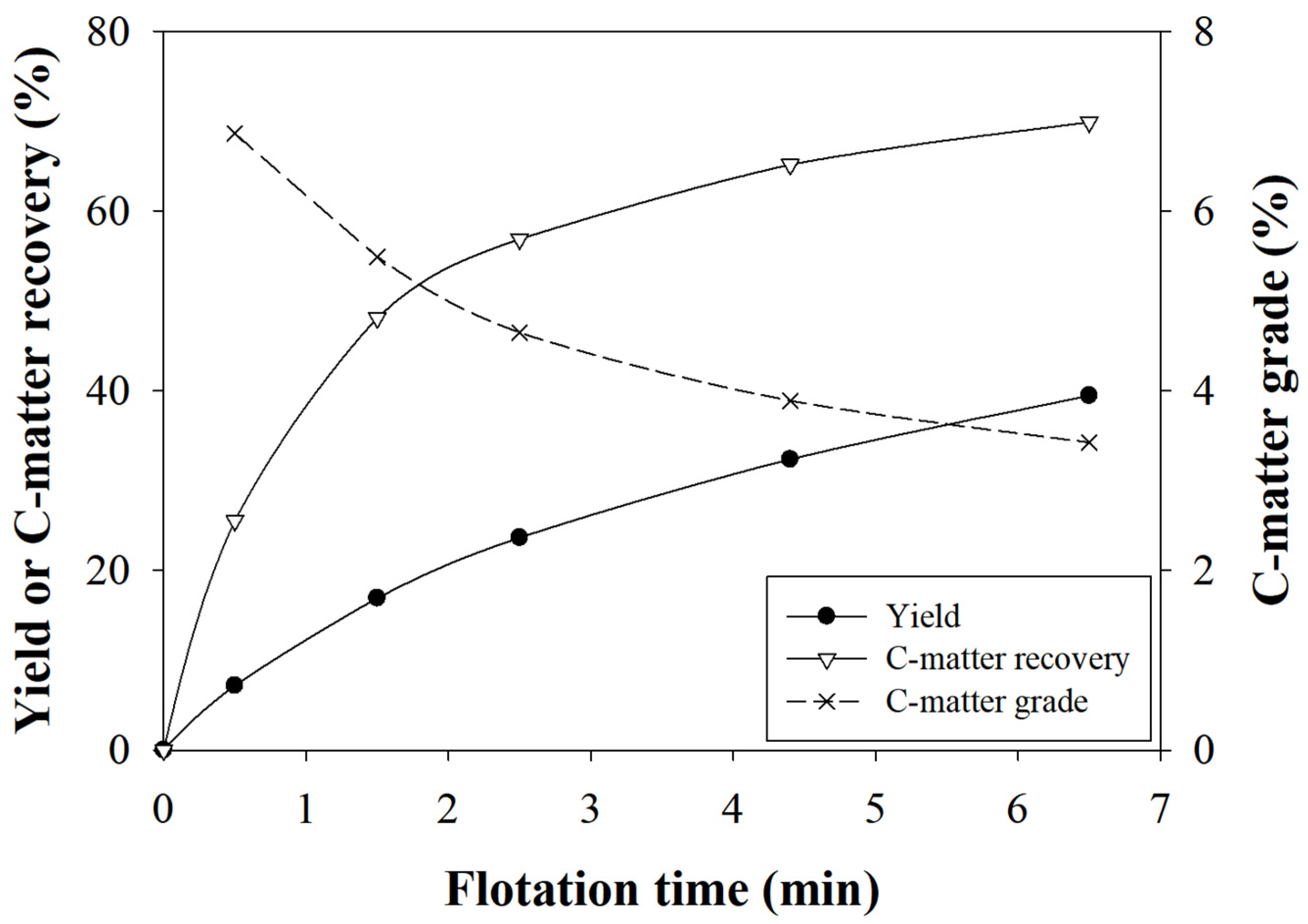
3.1. Rougher Flotation
3.2. Scavenger Flotation
3.3. Cleaner Flotation
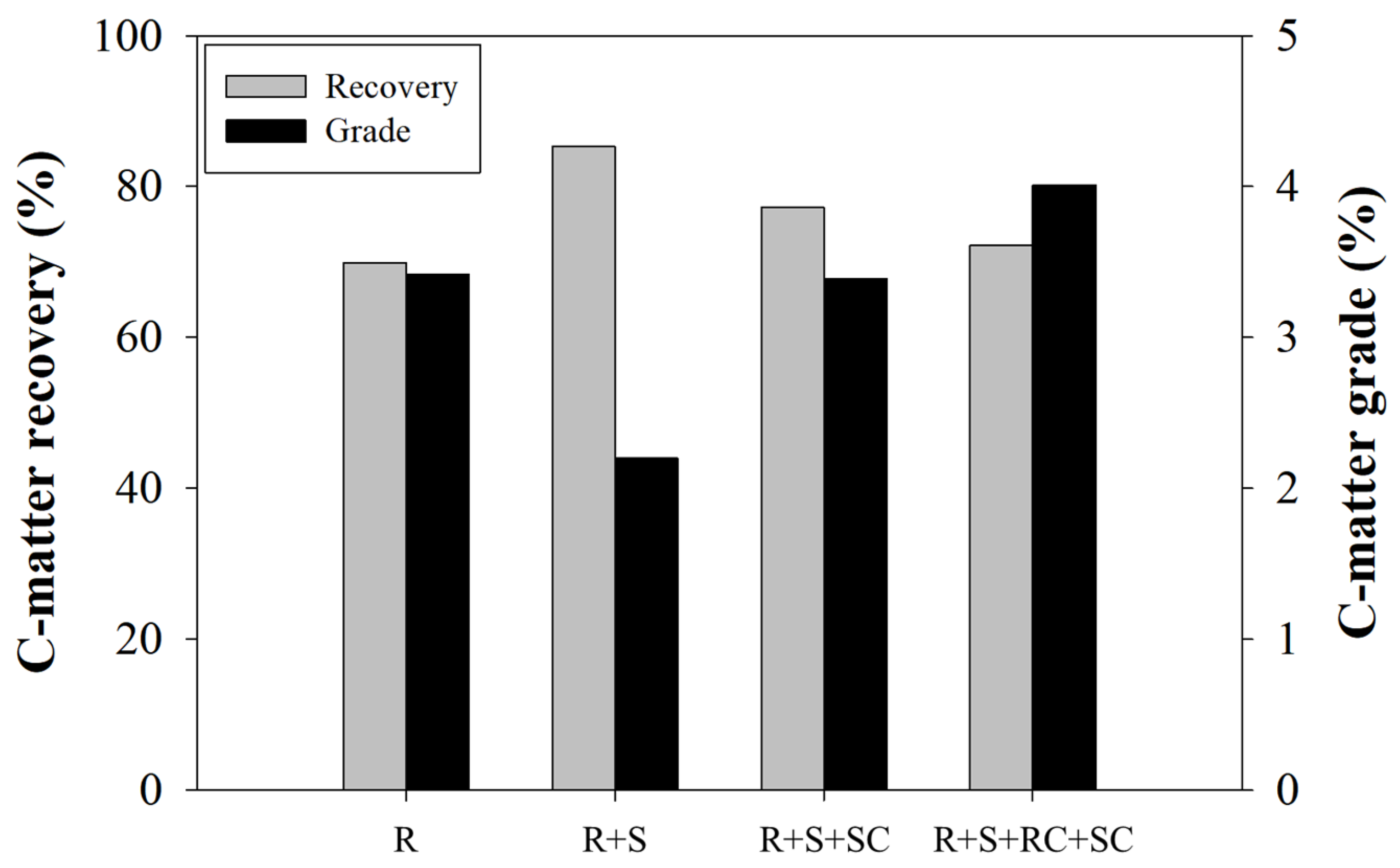
3.4. Comparison of Flotation Circuit Configurations
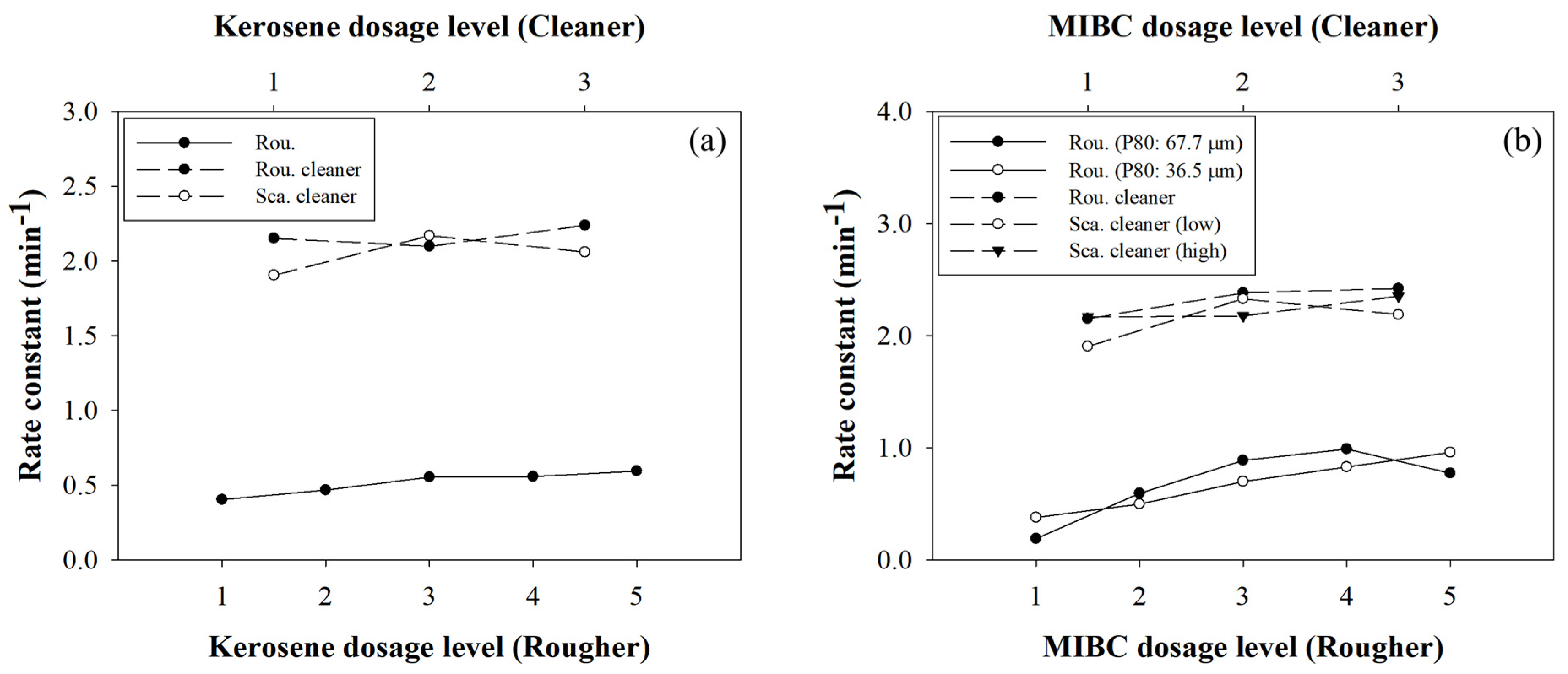
3.5. The Investigation of the Limited Flotation Performance
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Kerosene (g/t) | MIBC (g/t) | Collecting Time (min) | Cond. Speed (rpm) | Collect. Speed (rpm) | |
|---|---|---|---|---|---|
| Rougher | 500 | 120 | 4.5 | 1200 | 1200 |
| Scavenger 1 | 350 | 22 | 4.5 | 1200 | 1500 |
| Scavenger 2 | 175 | 11 | <10 | 1200 | 1500 |
| Cum. Yield (%) | Cum. C-matter (%) | Cum. S (%) | Cum. C as CO3 (%) | C-matter Grade (%) | S Grade (%) | C as CO3 Grade (%) | |
|---|---|---|---|---|---|---|---|
| Rougher | 9.84 | 47.41 | 13.61 | 9.7 | 9.48 | 1.61 | 4.39 |
| Scavenger 1 | 14.74 | 59.01 | 19.12 | 15.6 | 7.88 | 1.51 | 4.72 |
| Scavenger 2 | 15.49 | 60.45 | 19.87 | 16.6 | 7.68 | 1.50 | 4.77 |
| Kerosene (g/t) | MIBC (g/t) | Collecting Time (min) | Cond. Speed (rpm) | Collect. Speed (rpm) | |
|---|---|---|---|---|---|
| Rougher | 500 | 30 | 7 | 1200 | 1500 |
| Scavenger 1 | - | 22 | 3 | 1200 | 1500 |
| Scavenger 2 | - | 11 | 3 | 1200 | 1500 |
| Scavenger 3 | 175 | 5 | 3.5 | 1200 | 1500 |
| Cum. Yield (%) | Cum. C-matter (%) | Cum. S (%) | Cum. C as CO3 (%) | C-matter Grade (%) | S Grade (%) | C as CO3 Grade (%) | |
|---|---|---|---|---|---|---|---|
| Rougher | 3.92 | 22.80 | 5.95 | 3.2 | 11.30 | 1.61 | 3.67 |
| Scavenger 1 | 6.06 | 34.99 | 9.34 | 5.0 | 11.22 | 1.63 | 3.72 |
| Scavenger 2 | 9.76 | 48.17 | 14.46 | 8.7 | 9.59 | 1.57 | 4.01 |
| Scavenger 3 | 15.21 | 59.61 | 20.94 | 15.4 | 7.61 | 1.46 | 4.56 |
| Kerosene (g/t) | MIBC (g/t) | Collecting Time (min) | Cond. Speed (rpm) | Collect. Speed (rpm) | |
|---|---|---|---|---|---|
| Rougher | 500 | 86 | 0.5 | 1000 | 1200 |
| Scavenger 1 | - | 86 | 1 | 1200 | 1200 |
| Scavenger 2 | 250 | 43 | 2 | 1200 | 1200 |
| Scavenger 3 | 250 | 22 | 6 | 1200 | 1200 |
| Cum. Yield (%) | Cum. C-matter (%) | Cum. S (%) | Cum. C as CO3 (%) | C-matter Grade (%) | S grade (%) | C as CO3 Grade (%) | |
|---|---|---|---|---|---|---|---|
| Rougher | 4.27 | 33.35 | 9.34 | 2.54 | 14.25 | 2.07 | 2.66 |
| Scavenger 1 | 7.15 | 49.30 | 14.83 | 4.46 | 12.59 | 1.97 | 2.79 |
| Scavenger 2 | 13.03 | 67.54 | 23.83 | 9.49 | 9.46 | 1.73 | 3.26 |
| Scavenger 3 | 14.99 | 71.87 | 26.53 | 11.15 | 8.76 | 1.68 | 3.33 |
References
- Chen, T.; Cabri, L.; Dutrizac, J. Characterizing Gold in Refractory Sulfide Gold Ores and Residues. Jom 2002, 54, 20–22. [Google Scholar] [CrossRef]
- Komnitsas, C.; Pooley, F. Mineralogical Characteristics and Treatment of Refractory Gold Ores. Miner. Eng. 1989, 2, 449–457. [Google Scholar] [CrossRef]
- La Brooy, S.; Linge, H.; Walker, G. Review of Gold Extraction from Ores. Miner. Eng. 1994, 7, 1213–1241. [Google Scholar] [CrossRef]
- Fraser, K.; Walton, R.; Wells, J. Processing of Refractory Gold Ores. Miner. Eng. 1991, 4, 1029–1041. [Google Scholar] [CrossRef]
- Corrans, I.; Angove, J. Ultra Fine Milling for the Recovery of Refractory Gold. Miner. Eng. 1991, 4, 763–776. [Google Scholar] [CrossRef]
- Yang, Y.; Xie, Z.; Xu, B.; Li, Q.; Jiang, T. Gold Extraction from a High Carbon Low-Grade Refractory Gold Ore by Flotation-Roasting-Leaching Process. In Rare Metal Technology; Springer: Cham, Switzerland, 2015; Volume 2015, pp. 63–70. [Google Scholar] [CrossRef]
- Afenya, P. Treatment of Carbonaceous Refractory Gold Ores. Miner. Eng. 1991, 4, 1043–1055. [Google Scholar] [CrossRef]
- Gredelj, S.; Zanin, M.; Grano, S. Selective Flotation of Carbon in the Pb–Zn Carbonaceous Sulphide Ores of Century Mine, Zinifex. Miner. Eng. 2009, 22, 279–288. [Google Scholar] [CrossRef]
- Zhang, W.; Honaker, R. Studies on Carbon Flotation from Fly Ash. Fuel Process. Technol. 2015, 139, 236–241. [Google Scholar] [CrossRef]
- Naik, P.K.; Reddy, P.S.R.; Misra, V.N. Interpretation of Interaction Effects and Optimization of Reagent Dosages for Fine Coal Flotation. Int. J. Miner. Process. 2005, 75, 83–90. [Google Scholar] [CrossRef]
- Polat, M.; Polat, H.; Chander, S. Physical and Chemical Interactions in Coal Flotation. Int. J. Miner. Process. 2003, 72, 199–213. [Google Scholar] [CrossRef] [Green Version]
- Calisaya, D.A.; López-Valdivieso, A.; Marcos, H.; Gálvez, E.E.; Cisternas, L.A. A Strategy for the Identification of Optimal Flotation Circuits. Miner. Eng. 2016, 96, 157–167. [Google Scholar] [CrossRef]
- Sutherland, D. A Study on the Optimization of the Arrangement of Flotation Circuits. Int. J. Miner. Process. 1981, 7, 319–346. [Google Scholar] [CrossRef]
- Tao, D.; Luttrell, G.; Yoon, R.-H. An Experimental Investigation on Column Flotation Circuit Configuration. Int. J. Miner. Process. 2000, 60, 37–56. [Google Scholar] [CrossRef]
- Honaker, R.; Mohanty, M. Enhanced Column Flotation Performance for Fine Coal Cleaning. Miner. Eng. 1996, 9, 931–945. [Google Scholar] [CrossRef]
- Lu, X.; Forssberg, E. Flotation Selectivity and Upgrading of Woxna Fine Graphite Concentrate. Miner. Eng. 2001, 14, 1541–1543. [Google Scholar] [CrossRef]
- Yianatos, J.; Contreras, F. Particle Entrainment Model for Industrial Flotation Cells. Powder Technol. 2010, 197, 260–267. [Google Scholar] [CrossRef]
- Neethling, S.; Cilliers, J. The Entrainment Factor in Froth Flotation: Model for Particle Size and Other Operating Parameter Effects. Int. J. Miner. Process. 2009, 93, 141–148. [Google Scholar] [CrossRef]
- Hoang, D.H.; Kupka, N.; Peuker, U.A.; Rudolph, M. Flotation Study of Fine Grained Carbonaceous Sedimentary Apatite Ore–Challenges in Process Mineralogy and Impact of Hydrodynamics. Miner. Eng. 2018, 121, 196–204. [Google Scholar] [CrossRef]
- Albijanic, B.; Subasinghe, G.N.; Bradshaw, D.J.; Nguyen, A.V. Influence of Liberation on Bubble–Particle Attachment Time in Flotation. Miner. Eng. 2015, 74, 156–162. [Google Scholar] [CrossRef]
- Lee, S.; Gibson, C.E.; Ghahreman, A. Flotation of Carbonaceous Matter from a Double Refractory Gold Ore: The Effect of MIBC on Flotation Performance and Kinetics. Minerals 2021, 11, 1021. [Google Scholar] [CrossRef]
- Tsatouhas, G.; Grano, S.; Vera, M. Case Studies on the Performance and Characterisation of the Froth Phase in Industrial Flotation Circuits. Miner. Eng. 2006, 19, 774–783. [Google Scholar] [CrossRef]
- Amankwah, R.; Pickles, C. Microwave Roasting of a Carbonaceous Sulphidic Gold Concentrate. Miner. Eng. 2009, 22, 1095–1101. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Dong, K.; Fu, Y.; Xie, F. Research on Leaching of Carbonaceous Gold Ore with Copper-Ammonia-Thiosulfate Solutions. Miner. Eng. 2019, 137, 232–240. [Google Scholar] [CrossRef]
- Nanthakumar, B.; Pickles, C.; Kelebek, S. Microwave Pretreatment of a Double Refractory Gold Ore. Miner. Eng. 2007, 20, 1109–1119. [Google Scholar] [CrossRef]
- Lin, Q.Q.; Gu, G.H.; Wang, H.; Liu, Y.C.; Fu, J.G.; Wang, C.Q. Flotation Mechanisms of Molybdenite Fines by Neutral Oils. Int. J. Miner. Metall. Mater. 2018, 25, 1–10. [Google Scholar] [CrossRef]
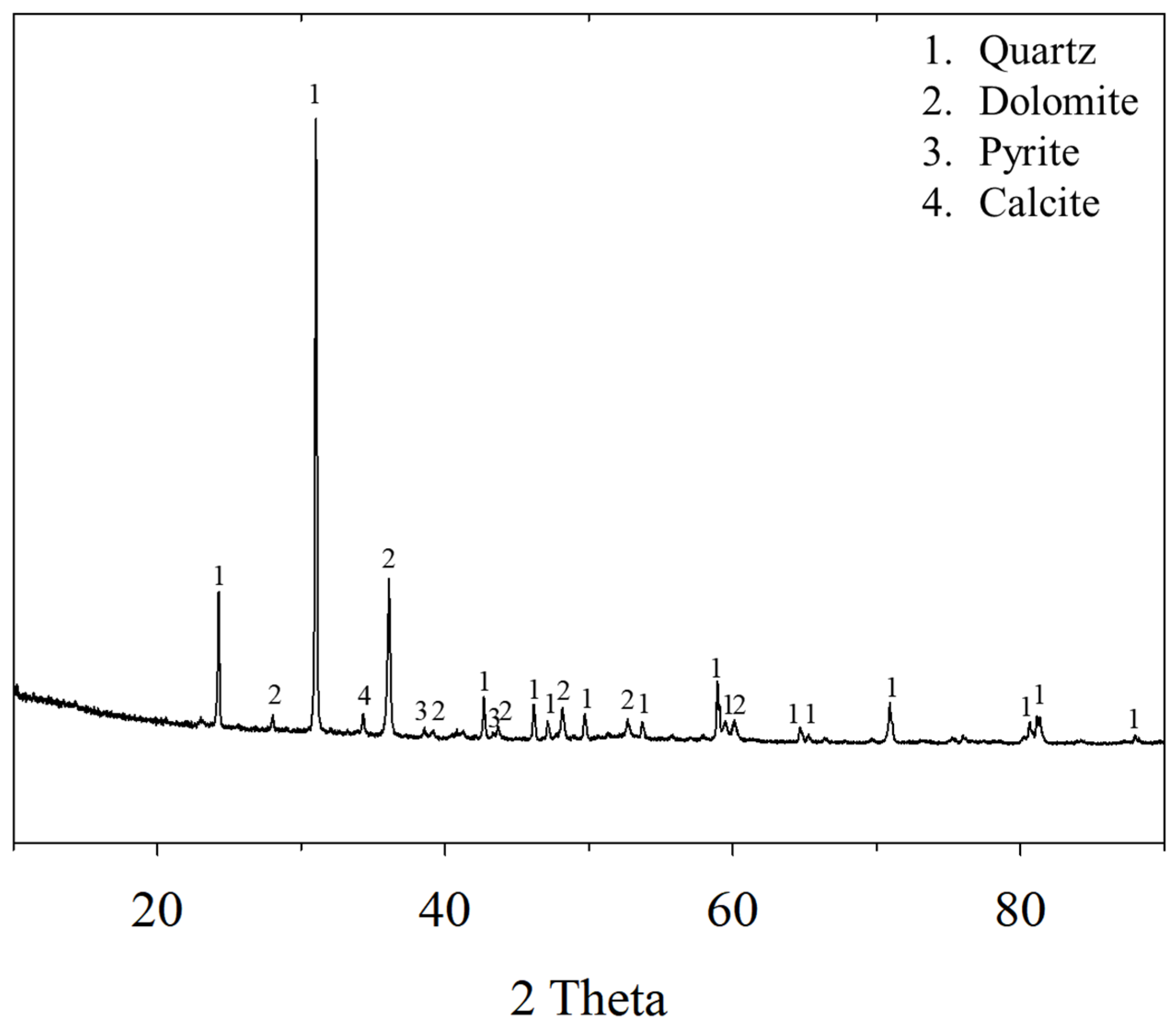


| Al (%) | Ba (ppm) | Ca (%) | Mg (%) | Fe (%) | K (%) | Na (%) | P (ppm) | Total C (C-matter) (%) | Total S (%) | Au (g/t) |
|---|---|---|---|---|---|---|---|---|---|---|
| 2.21 | 926 | 7.92 | 3.96 | 1.32 | 0.95 | 0.04 | 1000 | 5.93 (1.95) | 1.33 | 2.14 |
| Test | Kerosene Dosage (g t−1) | MIBC Dosage (g t−1) | P80 (μm) | Recovery (%) | Grade (%) |
|---|---|---|---|---|---|
| 1 | 50 | 60 | 67.7 | 42.23 | 2.03 |
| 2 | 100 | 60 | 67.7 | 39.87 | 2.44 |
| 3 | 200 | 60 | 67.7 | 51.19 | 2.36 |
| 4 | 300 | 60 | 67.7 | 54.19 | 2.64 |
| 5 | 500 | 60 | 67.7 | 55.98 | 2.72 |
| 6 | 500 | 60 | 44.6 | 61.60 | 2.57 |
| 7 | 500 | 60 | 36.5 | 67.30 | 2.67 |
| 8 | 500 | 30 | 36.5 | 51.18 | 2.67 |
| 9 | 500 | 90 | 36.5 | 68.23 | 2.90 |
| 10 | 500 | 120 | 36.5 | 69.87 | 3.42 |
| 11 | 500 | 150 | 36.5 | 70.09 | 3.62 |
| 12 | 500 | 30 | 67.7 | 41.11 | 2.81 |
| 13 | 500 | 90 | 67.7 | 60.55 | 3.18 |
| 14 | 500 | 120 | 67.7 | 61.16 | 2.82 |
| 15 | 500 | 150 | 67.7 | 66.54 | 2.60 |
| Test | Kerosene Dosage (g t−1) | MIBC Dosage (g t−1) | Scavenger Overall Recovery (%) |
|---|---|---|---|
| 1 | 70 | 21 | 24.12 |
| 2 | 210 | 21 | 24.98 |
| 3 | 350 | 21 | 28.69 |
| 4 | 70 | 42 | 26.29 |
| 5 | 70 | 63 | 25.95 |
| 6 | 350 | 42 | 25.84 |
| 7 | 350 | 63 | 27.70 |
| Rou. Conc. | Rou. Tail. | Sca. Conc. | Sca. Tail. | Tail. | |
|---|---|---|---|---|---|
| P80 (μm) | 30.3 | 42.5 | 38.5 | 43.0 | 46.6 |
| Test 1 | Test 2 | Test 3 | |
|---|---|---|---|
| Cum. C-matter recovery (%) | 60.45 | 59.61 | 71.81 |
| Cum. C-matter grade (%) | 7.68 | 7.61 | 8.76 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Gibson, C.E.; Ghahreman, A. The Separation of Carbonaceous Matter from Refractory Gold Ore Using Multi-Stage Flotation: A Case Study. Minerals 2021, 11, 1430. https://doi.org/10.3390/min11121430
Lee S, Gibson CE, Ghahreman A. The Separation of Carbonaceous Matter from Refractory Gold Ore Using Multi-Stage Flotation: A Case Study. Minerals. 2021; 11(12):1430. https://doi.org/10.3390/min11121430
Chicago/Turabian StyleLee, Sugyeong, Charlotte E. Gibson, and Ahmad Ghahreman. 2021. "The Separation of Carbonaceous Matter from Refractory Gold Ore Using Multi-Stage Flotation: A Case Study" Minerals 11, no. 12: 1430. https://doi.org/10.3390/min11121430
APA StyleLee, S., Gibson, C. E., & Ghahreman, A. (2021). The Separation of Carbonaceous Matter from Refractory Gold Ore Using Multi-Stage Flotation: A Case Study. Minerals, 11(12), 1430. https://doi.org/10.3390/min11121430






