Valorization of Kimberlite Tailings by Carbon Capture and Utilization (CCU) Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Kimberlite
2.2. Thin-Film Carbonation
2.3. Casting of Bricks
2.4. Initial Water Absorption Test
2.5. Compressive Strength Test
3. Results and Discussion
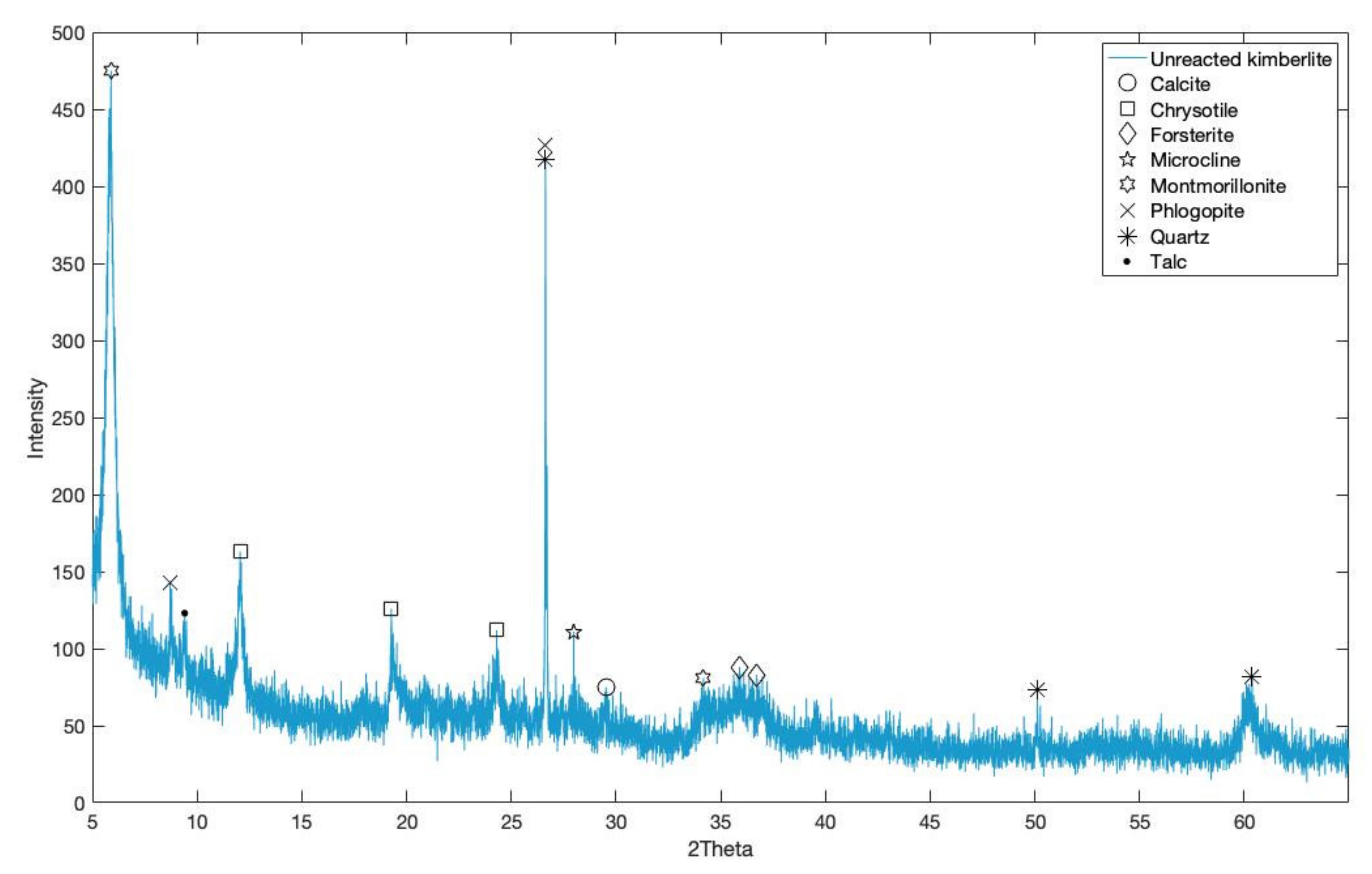
3.1. Kimberlite Carbonation
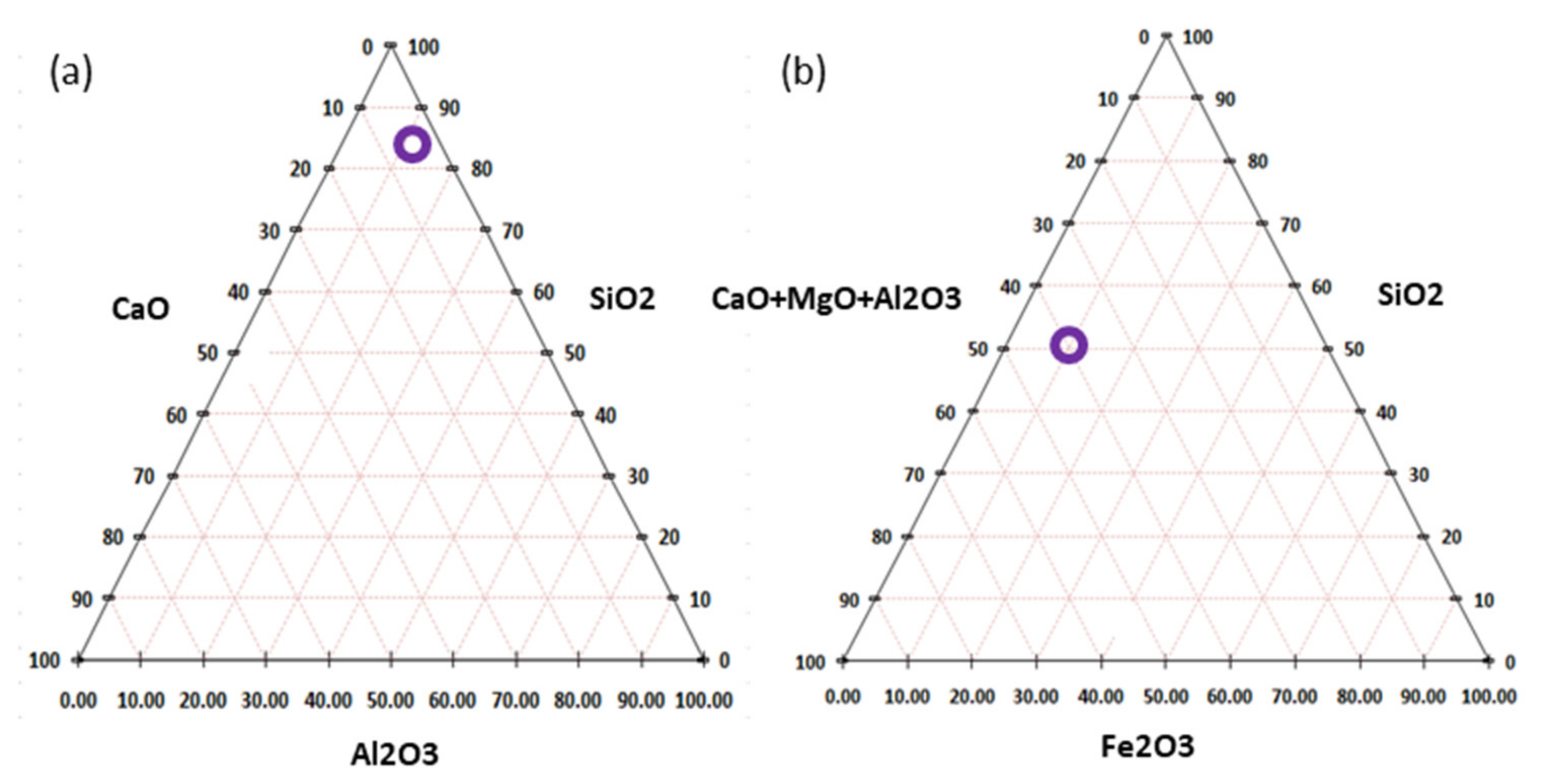
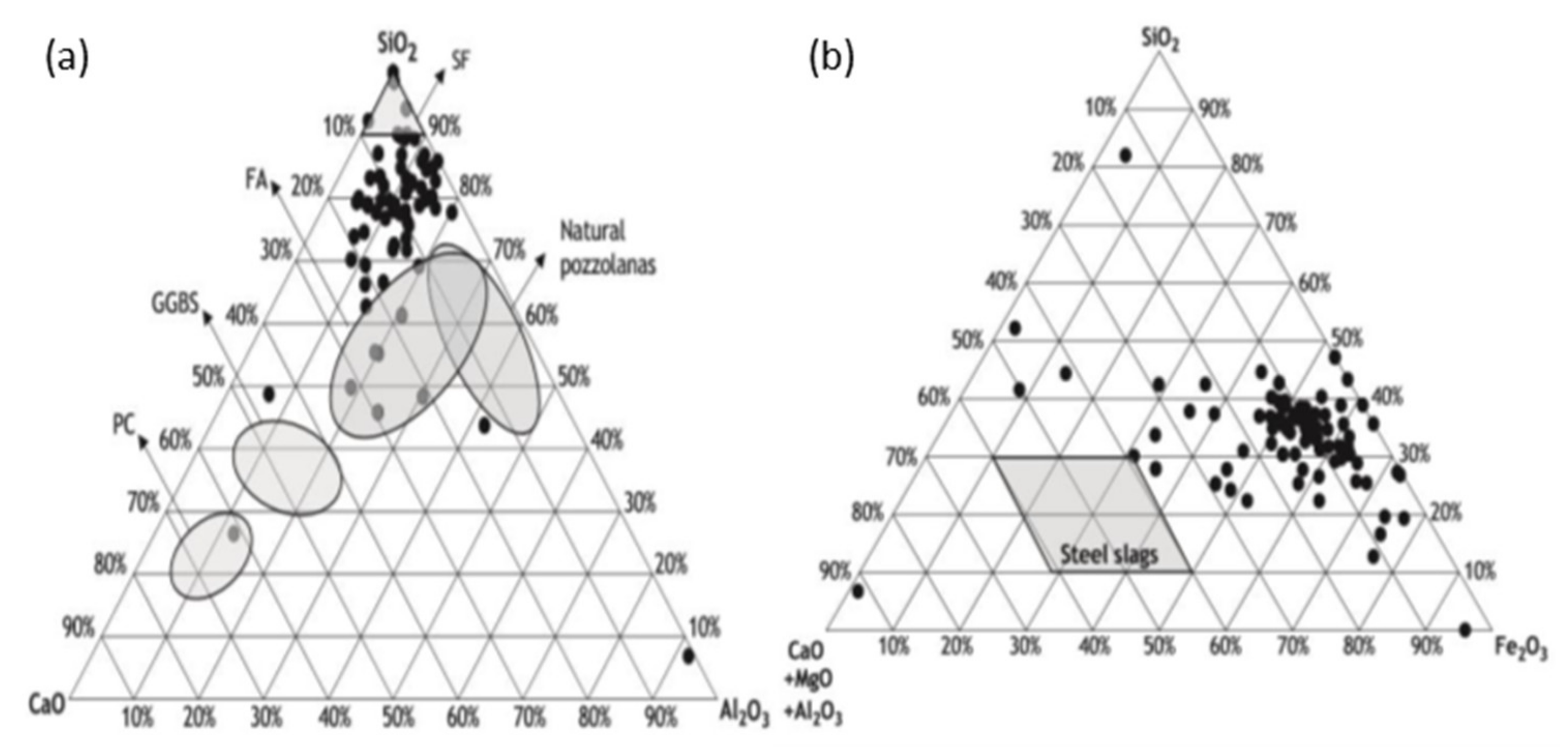
3.2. Binding Properties of Kimberlite
3.3. Initial Water Absorption of Bricks
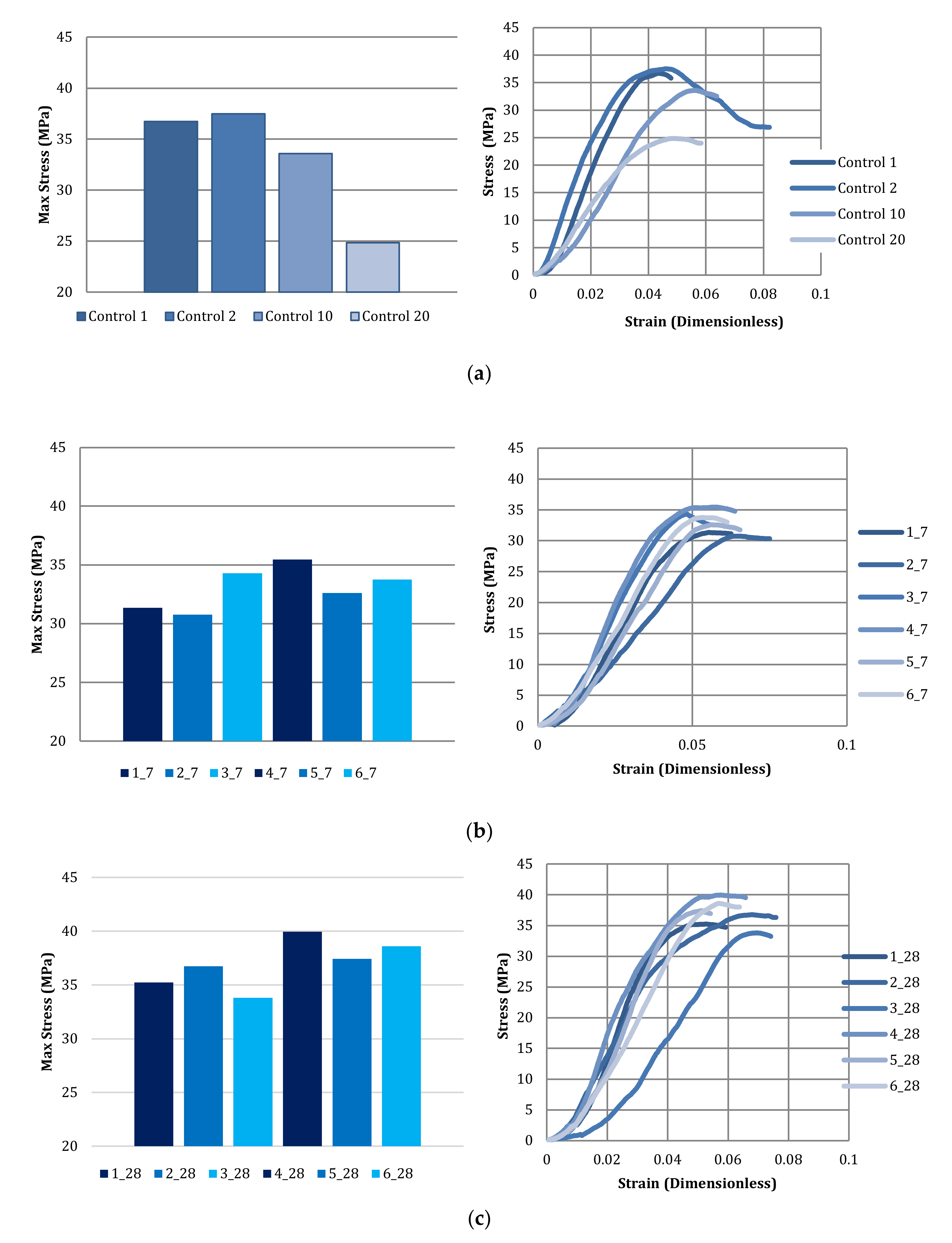
3.4. Compressive Strength of Bricks
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gibbs, M.J.; Soyka, P.; Conneely, D. Good Practice Guidance and Uncertainty Management in National Greenhouse Gas Inventories. 2001. Available online: https://www.ipcc-nggip.iges.or.jp/public/gp/english/ (accessed on 4 June 2020).
- Gavali, H.R.; Bras, A.; Faria, P.; Ralegaonkar, R.V. Development of sustainable alkali-activated bricks using industrial wastes. Constr. Build. Mater. 2019, 215, 180–191. [Google Scholar] [CrossRef]
- Adiansyah, J.S.; Rosano, M.; Vink, S.; Keir, G. A framework for a sustainable approach to mine tailings management: Disposal strategies. J. Clean. Prod. 2015, 108, 1050–1062. [Google Scholar] [CrossRef] [Green Version]
- Edraki, M.; Baumgartl, T.; Manlapig, E.; Bradshaw, D.; Franks, D.M.; Moran, C.J. Designing mine tailings for better environmental, social and economic outcomes: A review of alternative approaches. J. Clean. Prod. 2014, 84, 411–420. [Google Scholar] [CrossRef]
- Poinot, T.; Laracy, M.E.; Aponte, C.; Jennings, H.M.; Ochsendorf, J.A.; Olivetti, E.A. Beneficial use of boiler ash in alkali-activated bricks. Resour. Conserv. Recycl. 2018, 128, 1–10. [Google Scholar] [CrossRef]
- Zhang, L. Production of bricks from waste materials—A review. Constr. Build. Mater. 2013, 47, 643–655. [Google Scholar] [CrossRef]
- WBCSD. Cement Technology Roadmap 2009—Carbon Emissions Reductions up to 2050. 2009. Available online: http://www.cbcsd.org.cn/sjk/nengyuan/roadmap/20130516/download/Cement_Roadmap.pdf (accessed on 4 June 2020).
- Balasubramanian, J.; Sabumon, P.C.; Lazar, J.U.; Ilangovan, R. Reuse of textile effluent treatment plant sludge in building materials. Waste Manag. 2006, 26, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Wolff, E.; Schwabe, W.K.; Conceição, S.V. Utilization of water treatment plant sludge in structural ceramics. J. Clean. Prod. 2015, 96, 282–289. [Google Scholar] [CrossRef]
- Ukwatta, A.; Mohajerani, A. Characterisation of fired-clay bricks incorporating biosolids and the effect of heating rate on properties of bricks. Constr. Build. Mater. 2017, 142, 11–22. [Google Scholar] [CrossRef]
- Rani, M.Y.; Bhagawan, D.; Himabindu, V.; Reddy, V.V.; Saritha, P. Preparation and characterization of green bricks using pharmaceutical industrial wastes. Environ. Sci. Pollut. Res. 2016, 23, 9323–9333. [Google Scholar] [CrossRef]
- Goel, G.; Kalamdhad, A.S. An investigation on use of paper mill sludge in brick manufacturing. Constr. Build. Mater. 2017, 148, 334–343. [Google Scholar] [CrossRef]
- Goel, G.; Kalamdhad, A.S. Degraded municipal solid waste as partial substitute for manufacturing fired bricks. Constr. Build. Mater. 2017, 155, 259–266. [Google Scholar] [CrossRef]
- Demir, I. An investigation on the production of construction brick with processed waste tea. Build. Environ. 2006, 41, 1274–1278. [Google Scholar] [CrossRef]
- Mekki, H.; Ammar, E.; Anderson, M.; Zina, M.B. The recycling of olive oil mill by-products in bricks. Ann. Chimie Sci. Materiaux 2003, 28, 109–127. [Google Scholar] [CrossRef]
- Mekki, H.; Anderson, M.; Amar, E.; Skerratt, G.R.; Zina, M.B. Olive oil mill waste water as a replacement for fresh water in the manufacture of fired clay bricks. J. Chem. Technol. Biotechnol. 2006, 81, 1419–1425. [Google Scholar] [CrossRef]
- Cotes Palomino, M.T.; Martínez García, C.; Iglesias Godino, F.J.; Eliche Quesada, D.; Corpas Iglesias, F.A. Study of the wet pomace as an additive in ceramic material. Desalin. Water Treat. 2016, 57, 2712–2718. [Google Scholar] [CrossRef]
- José, A.; Lorite, M.; Jiménez, J.; Castro, E. Valorisation of wastewater from two-phase olive oil extraction in fired clay brick production. J. Hazard. Mater. 2009, 169, 271–278. [Google Scholar]
- Santos, R.M.; Van Bouwel, J.; Vandevelde, E.; Mertens, G.; Elsen, J.; Van Gerven, T. Accelerated mineral carbonation of stainless steel slags for CO2 storage and waste valorization: Effect of process parameters on geochemical properties. Int. J. Greenh. Gas Control. 2013, 17, 32–45. [Google Scholar] [CrossRef] [Green Version]
- Baciocchi, R.; Costa, G.; Polettini, A.; Pomi, R. Influence of particle size on the carbonation of stainless steel slag for CO2 storage. Energy Procedia 2009, 1, 4859–4866. [Google Scholar] [CrossRef] [Green Version]
- Bodénan, F.; Bourgeois, F.; Petiot, C.; Augé, T.; Bonfils, B.; Julcour-Lebigue, C.; Guyot, F.; Boukary, A.; Tremosa, J.; Lassin, A.; et al. Ex situ mineral carbonation for CO2 mitigation: Evaluation of mining waste resources, aqueous carbonation processability and life cycle assessment (Carmex project). Miner. Eng. 2014, 59, 52–63. [Google Scholar] [CrossRef] [Green Version]
- Romanov, V.; Soong, Y.; Carney, C.; Rush, G.E.; Nielsen, B.; O’Connor, W. Mineralization of carbon dioxide: A literature review. ChemBioEng Rev. 2015, 2, 231–256. [Google Scholar] [CrossRef]
- Ostovari, H.; Sternberg, A.; Bardow, A. Rock ‘n’ use of CO2: Carbon footprint of carbon capture and utilization by mineralization. Sustain. Energy Fuels 2020. [Google Scholar] [CrossRef]
- O’Connor, W.K.; Dahlin, D.C.; Rush, G.E.; Gerdemann, S.J.; Penner, L.R.; Nilsen, D.N. Aqueous Mineral Carbonation; Final Report DOE/ARC-TR-04-002; US Department of Energy: Albany, OR, USA, 2005.
- Wang, X.; Maroto-Valer, M.M. Integration of CO2 capture and mineral carbonation by using recyclable ammonium salts. ChemSusChem 2011, 4, 1291–1300. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Hitch, M. Mechanical activation of magnesium silicates for mineral carbonation, a review. Miner. Eng. 2018, 128, 69–83. [Google Scholar]
- Ghoorah, M.; Dlugogorski, B.Z.; Balucan, R.D.; Kennedy, E.M. Selection of acid for weak acid processing of wollastonite for mineralisation of CO2. Fuel 2014, 122, 277–286. [Google Scholar] [CrossRef]
- Mervine, E.M.; Wilson, S.A.; Power, I.M.; Dipple, G.M.; Turvey, C.C.; Hamilton, J.L.; Vanderzee, S.; Raudsepp, M.; Southam, C.; Matter, J.M.; et al. Potential for offsetting diamond mine carbon emissions through mineral carbonation of processed kimberlite: An assessment of De Beers mine sites in South Africa and Canada. Mineral. Petrol. 2018, 112, S755–S765. [Google Scholar] [CrossRef]
- Bodor, M.; Santos, R.M.; Kriskova, L.; Elsen, J.; Vlad, M.; Van Gerven, T. Susceptibility of mineral phases of steel slags towards carbonation: Mineralogical, morphological and chemical assessment. Eur. J. Mineral. 2013, 25, 533–549. [Google Scholar] [CrossRef]
- Bukowski, J.M.; Berger, R.L. Reactivity and strength development of CO2 activated non-hydraulic calcium silicates. Cem. Concr. Res. 1979, 9, 57–68. [Google Scholar] [CrossRef]
- Van der Plas, G.; Limaye, P.; Loi, I.; Mercha, A.; Oprins, H.; Torregiani, C.; Thijs, S.; Linten, D.; Stucchi, M.; Katti, G.; et al. Design issues and considerations for low-cost 3-D TSV IC technology. IEEE J. Solid-State Circuits 2010, 46, 293–307. [Google Scholar] [CrossRef] [Green Version]
- Santos, R.M.; Van Gerven, T. Process intensification routes for mineral carbonation. Greenh. Gases Sci. Technol. 2010, 1, 287–293. [Google Scholar] [CrossRef] [Green Version]
- ASTM International. ASTM: C67-14, Standard Test Methods for Sampling and Testing Brick and Structural Clay Tile; ASTM: West Conshohocken, PA, USA, 2014. [Google Scholar]
- Morgan, B.; Wilson, S.A.; Madsen, I.C.; Gozukara, Y.M.; Habsuda, J. Increased thermal stability of nesquehonite (MgCO3·3H2O) in the presence of humidity and CO2: Implications for low-temperature CO2 storage. Int. J. Greenh. Gas Control. 2015, 39, 366–376. [Google Scholar] [CrossRef]
- Zhang, Z.; Zheng, Y.; Ni, Y.; Liu, Z.; Chen, J.; Liang, X. Temperature-and pH-dependent morphology and FT-IR analysis of magnesium carbonate hydrates. J. Phys. Chem. B 2006, 110, 12969–12973. [Google Scholar] [CrossRef]
- Power, I.M.; Wilson, S.A.; Harrison, A.L.; Dipple, G.M.; Mccutcheon, J.; Southam, G.; Kenward, P.A. A depositional model for hydromagnesite–magnesite playas near Atlin, British Columbia, Canada. Sedimentology 2014, 61, 1701–1733. [Google Scholar] [CrossRef]
- Hwang, K.-Y.; Kim, J.Y.; Phan, H.Q.H.; Ahn, J.-Y.; Kim, T.Y.; Hwang, I. Effect of CO2 concentration on strength development and carbonation of a MgO-based binder for treating fine sediment. Environ. Sci. Pollut. Res. 2018, 25, 22552–22560. [Google Scholar] [CrossRef] [PubMed]
- Obe, R.K.D.; De Brito, J.; Mangabhai, R.; Lye, C.Q. Sustainable Construction Materials: Copper Slag; Woodhead Publishing: Cambridge, UK, 2016. [Google Scholar] [CrossRef]
- ASTM International. ASTM: C618-19, Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete; ASTM: West Conshohocken, PA, USA, 2012. [Google Scholar]
- Salman, M.; Cizer, Ö.; Pontikes, Y.; Santos, R.M.; Snellings, R.; Vandewalle, L.; Blanpain, B. Van Balen, K. Effect of accelerated carbonation on AOD stainless steel slag for its valorisation as a CO2-sequestering construction material. Chem. Eng. J. 2014, 246, 39–52. [Google Scholar] [CrossRef] [Green Version]
- Canadian Concrete Mansory Producers Association. Physical Properties, Metric Technical Manual. 2020. Available online: https://ccmpa.ca/wp-content/uploads/2012/02/Final2013Sec4.pdf (accessed on 23 June 2013).
- Kuranchie, F.A.; Shukla, S.K.; Habibi, D. Utilisation of iron ore mine tailings for the production of geopolymer bricks. Int. J. Min. Reclam. Environ. 2016, 30, 92–114. [Google Scholar] [CrossRef]




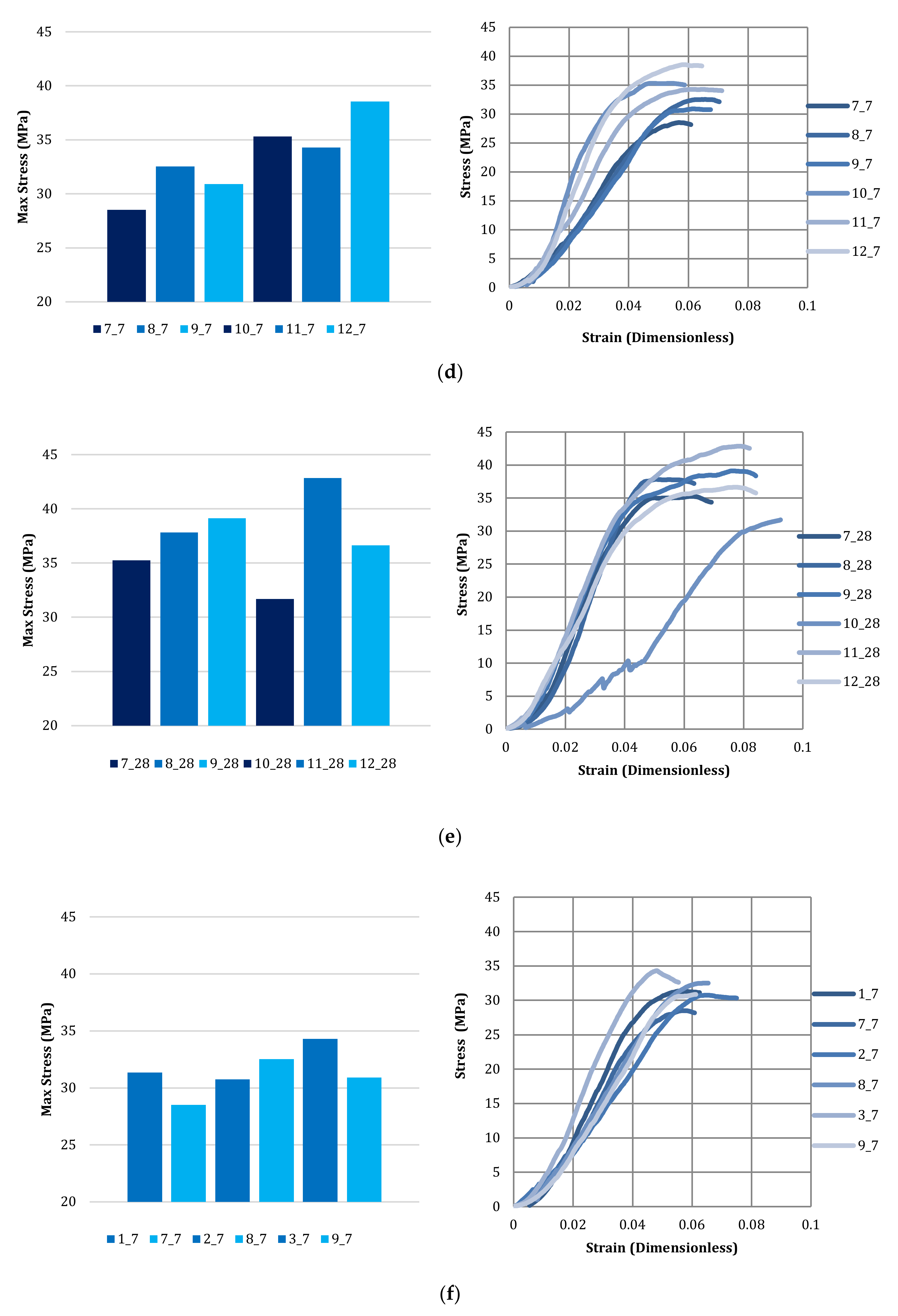
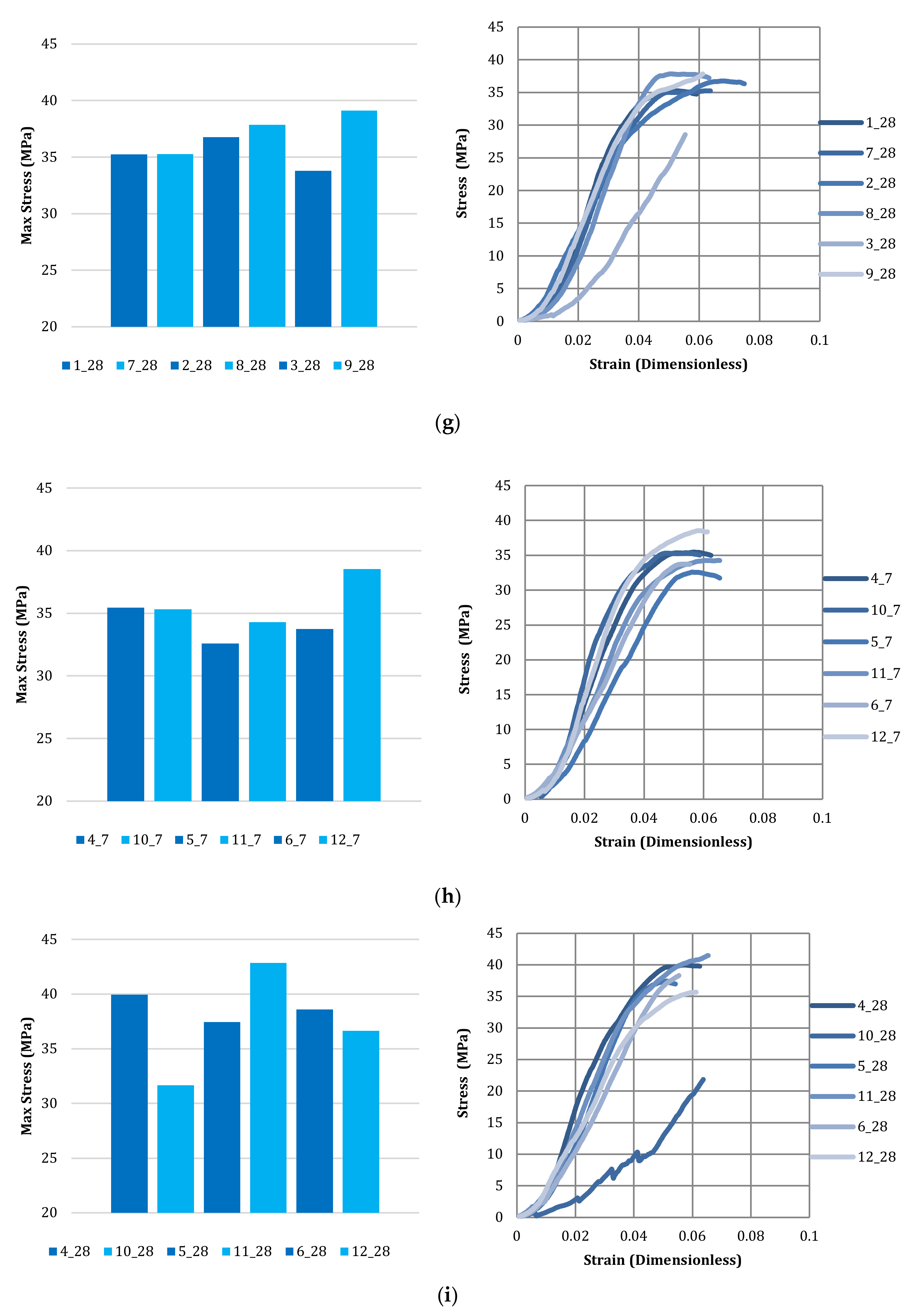
| Brick Number | Carbonated Kimberlite Material—10% Replacement |
|---|---|
| 1 (7 days) | 10% Moisture Content, 10% CO2, 35 °C |
| 1 (28 days) | |
| 2 (7 days) | 15% Moisture Content, 10% CO2, 35 °C |
| 2 (28 days) | |
| 3 (7 days) | 20% Moisture Content, 10% CO2, 35 °C |
| 3 (28 days) | |
| 4 (7 days) | 10% Moisture Content, 20% CO2, 35 °C |
| 4 (28 days) | |
| 5 (7 days) | 15% Moisture Content, 20% CO2, 35 °C |
| 5 (28 days) | |
| 6 (7 days) | 20% Moisture Content, 20% CO2, 35 °C |
| 6 (28 days) | |
| 7 (7 days) | 10% Moisture Content, 10% CO2, 50 °C |
| 7 (28 days) | |
| 8 (7 days) | 15% Moisture Content, 10% CO2, 50 °C |
| 8 (28 days) | |
| 9 (7 days) | 20% Moisture Content, 10% CO2, 50 °C |
| 9 (28 days) | |
| 10 (7 days) | 10% Moisture Content, 20% CO2, 50 °C |
| 10 (28 days) | |
| 11 (7 days) | 15% Moisture Content, 20% CO2, 50 °C |
| 11 (28 days) | |
| 12 (7 days) | 20% Moisture Content, 20% CO2, 50 °C |
| 12 (28 days) |
| Carbonation Conditions | Carbonated Kimberlite | ||
|---|---|---|---|
| Temperature (°C) | CO2 Concentration (vol%) | Moisture Content (wt% w.b.) | CO2 Content (wt% d.b.) |
| Unreacted | - | - | 4.32 |
| 35 | 10 | 10 | 7.67 |
| 15 | 7.92 | ||
| 20 | 7.76 | ||
| 35 | 20 | 10 | 7.94 |
| 15 | 7.96 | ||
| 20 | 8.04 | ||
| 50 | 10 | 10 | 7.52 |
| 15 | 7.53 | ||
| 20 | 7.35 | ||
| 50 | 20 | 10 | 7.75 |
| 15 | 7.62 | ||
| 20 | 7.47 | ||
| Brick Number | Initial Weight (g) | Weight after 24 h (g) | Initial Water Absorption (%) |
|---|---|---|---|
| 1 (7 days) | 2445.15 | 2471.95 | 1.096 |
| 1 (28 days) | 2464.87 | 2494.95 | 1.220 |
| 2 (7 days) | 2490.21 | 2523.64 | 1.342 |
| 2 (28 days) | 2512.21 | 2544.84 | 1.299 |
| 3 (7 days) | 2463.27 | 2497.16 | 1.376 |
| 3 (28 days) | 2479.84 | 2514.87 | 1.413 |
| 4 (7 days) | 2558.10 | 2597.15 | 1.526 |
| 4 (28 days) | 2587.64 | 2623.46 | 1.384 |
| 5 (7 days) | 2561.48 | 2590.66 | 1.139 |
| 5 (28 days) | 2489.00 | 2518.87 | 1.200 |
| 6 (7 days) | 2503.73 | 2539.11 | 1.413 |
| 6 (28 days) | 2551.33 | 2581.85 | 1.196 |
| 7 (7 days) | 2581.85 | 2605.44 | 0.914 |
| 7 (28 days) | 2581.57 | 2606.61 | 0.970 |
| 8 (7 days) | 2566.82 | 2591.55 | 0.963 |
| 8 (28 days) | 2553.44 | 2578.07 | 0.965 |
| 9 (7 days) | 2471.97 | 2500.82 | 1.167 |
| 9 (28 days) | 2541.44 | 2570.81 | 1.156 |
| 10 (7 days) | 2679.80 | 2718.45 | 1.442 |
| 10 (28 days) | 2515.91 | 2549.81 | 1.347 |
| 11 (7 days) | 2536.43 | 2568.78 | 1.275 |
| 11 (28 days) | 2580.04 | 2611.12 | 1.205 |
| 12 (7 days) | 2554.70 | 2584.45 | 1.164 |
| 12 (28 days) | 2563.73 | 2597.85 | 1.331 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakravarthy, C.; Chalouati, S.; Chai, Y.E.; Fantucci, H.; Santos, R.M. Valorization of Kimberlite Tailings by Carbon Capture and Utilization (CCU) Method. Minerals 2020, 10, 611. https://doi.org/10.3390/min10070611
Chakravarthy C, Chalouati S, Chai YE, Fantucci H, Santos RM. Valorization of Kimberlite Tailings by Carbon Capture and Utilization (CCU) Method. Minerals. 2020; 10(7):611. https://doi.org/10.3390/min10070611
Chicago/Turabian StyleChakravarthy, Cibi, Salma Chalouati, Ye Eun Chai, Hugo Fantucci, and Rafael M. Santos. 2020. "Valorization of Kimberlite Tailings by Carbon Capture and Utilization (CCU) Method" Minerals 10, no. 7: 611. https://doi.org/10.3390/min10070611
APA StyleChakravarthy, C., Chalouati, S., Chai, Y. E., Fantucci, H., & Santos, R. M. (2020). Valorization of Kimberlite Tailings by Carbon Capture and Utilization (CCU) Method. Minerals, 10(7), 611. https://doi.org/10.3390/min10070611






