Slatecalculation—A Practical Tool for Deriving Norm Minerals in the Lowest-Grade Metamorphic Pelites and Roof Slates
Abstract
1. Introduction
2. Materials and Methods
2.1. Colors
2.2. Slate Samples
2.3. Algorithm Slatecalculation
Residual SiO2 → qz = quartz = SiO2
Residual H2O → water → aq
2.4. Hydro-Micas
aq (share K2O) → faill = 0.65K2O 1.05FeO 1.95Al2O3 6SiO2 2.35H2O (Mol. Wt of Mineral: 738.32)
or in combination:
aq (share K2O) → mfaill = 0.65K2O 0.525MgO 0.525FeO 1.95Al2O3 6SiO2 2.35H2O (Mol. Wt of Mineral: 721.77)
aq (share Na2O) → fabr = 0.65Na2O 1.05FeO 1.95Al2O3 6SiO2 2.35H2O (Mol. Wt of Mineral: 717.39)
or in combination:
aq (share Na2O) → mfabr = 0.65Na2O 0.525MgO 0.525FeO 1.95Al2O3 6SiO2 2.35H2O (Mol. Wt of Mineral: 700.83).
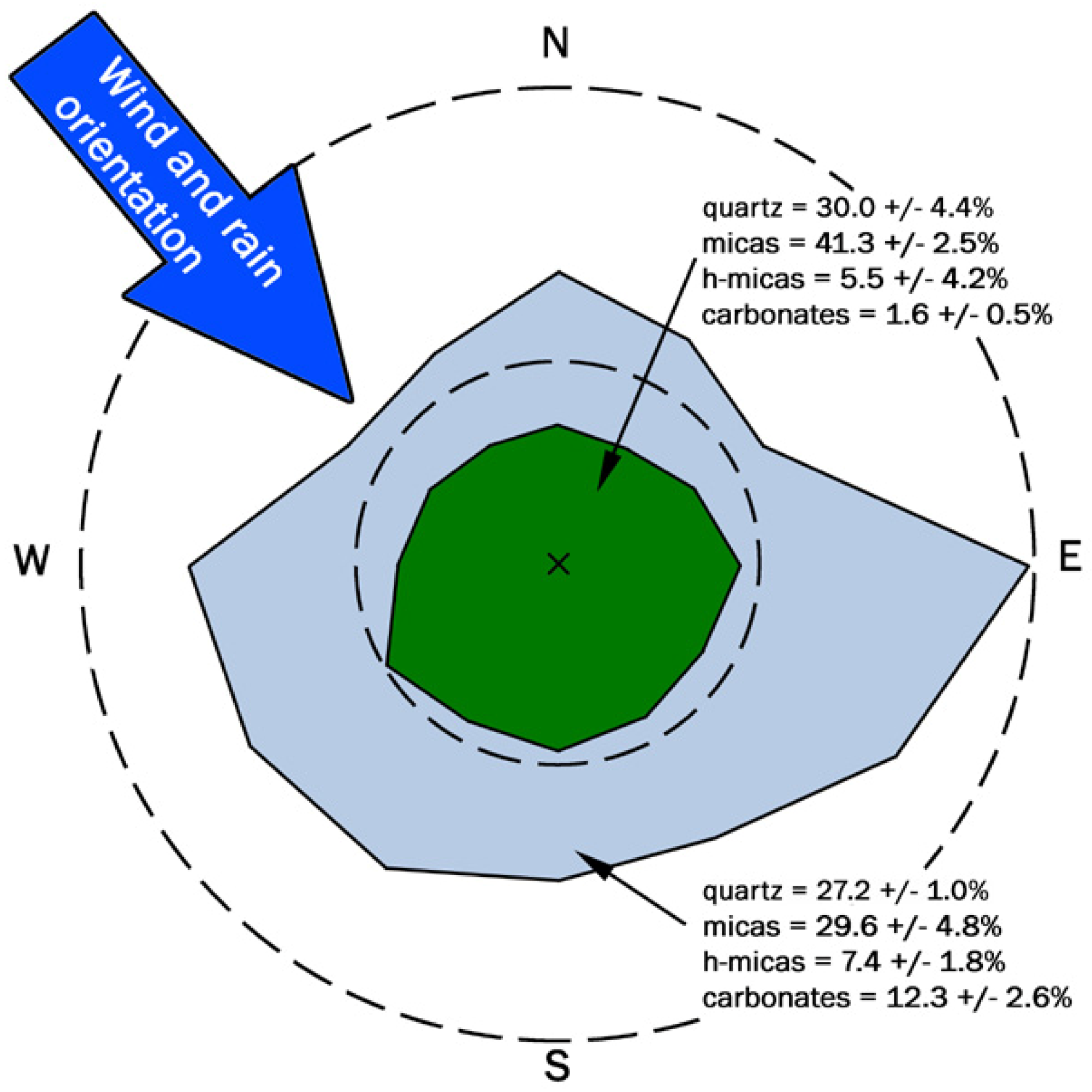
2.5. Analysis of the Fabric of Phyllosilicates
3. Results and Discussion
4. Conclusions
- carbonates (cc = calcite, dol = dolomite, sid = siderite),
- feldspars (an = anorthite, ab = albite, or = orthoclase),
- micas (mu = muskovite, pa = paragonite),
- hydro-micas (ill = illite, br = brammallite),
- chlorites (mac+mc = Mg-chlorites, fac+fc = Fe-chlorites)
- sulfides (pt = pyrite, pn = phyrrotite),
- other ore-minerals (tm = titanomagnetite, ilm = ilmenite, ru = rutile, he = hematite, lm = Limonit),
- others (ap = apatite, gr = graphite, ct = chloritoid),
- and qz = quartz.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Arkai, P.; Sassi, F.; Desmons, J. Very low- to low-grade Metamorphic rocks. Recomm. IUGS Subcomm. Syst. Metamorph. Rocks 2003, 5, 1–12. [Google Scholar]
- Passchier, C.W.; Trow, R.A.J. Microtectonics, 2nd ed.; Springer: Heidelberg, Germany, 2005. [Google Scholar]
- Wagner, H.W. The basics of test methods of slates for roofing and cladding. Grundlagen für die Prüfung von Dach- und Wandschiefern. Z. Dtsch. Ges. Geowiss. 2007, 158, 785–805. [Google Scholar]
- Wagner, H.W.; Le Bail, R.; Hacar, M.; Stanek, S. European roofing slates part 1: Remarks to the geology of mineral deposits. Z. Angew. Geol. 1994, 40, 68–74. [Google Scholar]
- Winter, J.D. (n. s.). CIPW Norm Excel Spreadsheet. Available online: http://webspace.pugetsound.edu/facultypages/jtepper/MIN-PET/Norm%20Calculation.XLS (accessed on 21 February 2020).
- Wagner, H.W.; Baumann, H.; Negendank, J.; Roschig, F. Geological, petrographic, geochemical and petrophysical investigations on roofing slates. Mainz. Geowiss. Mitt. 1997, 26, 131–184. Available online: https://www.uni-trier.de/fileadmin/fb6/prof/GEO/baumann/pdf_files/slate.pdf (accessed on 21 February 2020).
- Ward, C.; Gómez-Fernandez, F. Quantitative mineralogical analysis of Spanish roofing slates using the Rietveld method and X-ray powder diffraction data. Eur. J. Mineral. 2003, 15, 1051–1062. [Google Scholar] [CrossRef]
- Jung, D.; Wagner, H.W. SlateNorm: Calculation of a normative mineral content of slates. Unpublished. 1998–2000. [Google Scholar]
- Cárdenes, V.; Rubio-Ordóñez, A.; Wichert, J.; Cnudde, J.P.; Cnudde, V. Petrography of roofing slates. Earth Sci. Rev. 2014, 138, 435–453. [Google Scholar] [CrossRef]
- Lorenz, W.; Gwodsdz, W. Chapter 3.2 Roofing slate. In Manual on the Geological-technical Assessment of Mineral Construction Materials; Geol. Jahrbuch, Sonderhefte Reihe H, Heft SH 15; BGR: Hannover, Germany, 2003; pp. 276–290. [Google Scholar]
- European Committee for Standardization (CEN). EN 12326-1 and -2 Slate and Stone Products for Discontinuous Roofing and Cladding—Part 1: Product Specification 2014, Part 2: Methods of Test 2010; Beuth: Berlin, Germany, 2010. [Google Scholar]
- Jung, D. Zur Frage der Qualitätsbeurteilung von Schiefer. Schr. Schiefer Fachverb. Dtschl. E. V. 2009, 10, 75–107. [Google Scholar]
- Wagner, H.W.; Schultheis, W. Römischer Dachschiefer—Neue Funde und neue Erkenntnisse. Anschnitt 2011, 63, 202–206. [Google Scholar]
- Wagner, H.W. Geologische Untersuchung und Materialprüfung an Dachschiefer-Altdeckungen. Mainz. Geowiss. Mitt. 2014, 42, 121–142. [Google Scholar]
- Morales-Demarco, M.; Oyhantçabal, P.; Stein, K.-J.; Siegesmund, S. Dolomitic slates from Uruguay: Petrophysical and petromechanical characterization and deposit evaluation. Environ. Earth Sci. 2013, 69, 1361–1395. [Google Scholar] [CrossRef]
- Franke, W.; Paul, J. Pelagic redbeds in the Devonian of Germany—Deposition and diagenesis. Sediment. Geol. 1980, 25, 231–256. [Google Scholar] [CrossRef]
- Cárdenes, V.; Prieto, B.; Sanmartín, P.; Ferrer, P.; Rubio, A.; Monterroso, C. Influence of chemical-mineralogical composition on the color and brightness of Iberian roofing slates. J. Mater. Civ. Eng. 2012, 24, 460–467. [Google Scholar] [CrossRef]
- Dale, T.N.; Eckel, E.C.; Hillebrand, W.F.; Coons, A.T. Slate Deposits and Slate Industry of the United States; Bulletin 275, Series A, Economic Geology; U.S. Geological Survey: Reston, VA, USA, 1906; Volume 63, pp. 1–154.
- Grossi, J.H.; Chiodi, C.; Kistemann, D. Panorama do Setor de Ardósias do Estado de Minas Gerais; Textos e Anexos; Governo de Minas Gerais: Belo Horizonte, Brasil, 1998; Volume 1, pp. 1–90.
- Krosse, S.; Schreyer, W. Comparative geochemistry of coticules (spessartinequartzites) and their redschist country rocks in the Ordovician of the Ardennes Mountains, Belgium. Chem. Erde 1993, 53, 1–20. [Google Scholar]
- Ministerio de Industria y Energia. Pizarras de Espana; Instituto Geolocico y Minero de Espana: Madrid, Spain, 1977; p. 83. [Google Scholar]
- Zimmerle, W.; Stribrny, B. Organic carbon-rich pelitic sediments in the Federal Republic of Germany. Cour. Forsch. Inst. Senckenberg 1992, 152, 1–144. [Google Scholar]
- Jung, D. Mineralogisch-petrographische Untersuchung eines Schiefers aus Bhutan. Unpublished. 2001; 13. [Google Scholar]
- Stribny, B.; Urban, H. Classification of sedimentary rocks—Black shale series based on their normative minerals compositions. IGCP Proj. Newsl. 1989, 254, 30. [Google Scholar]
- Jung, D.; Geisler-Wierwille, T. Program “SlateNorm” Version 1.1. Unpublished. 1999–2000. [Google Scholar]
- Cardenes, V.; Rubio Ordonez, A.; Lopez Munguira, A.; Monteroso, C. Petrografía y mineralogía de las pizarras para cubiertas de la Península Ibérica en relación con su calidad. Trab. Geol. Univ. Oviedo 2010, 30, 412–420. [Google Scholar]
- Ferreiro Mählmann, R.; Bozkaya, Ö.; Potel SLe Bayon, R.; Šegvic, B.; Nieto, F. The pioneer work of Bernard Kübler and Martin Frey in very low-grade metamorphic terranes: Paleo-geothermal potential of variation in Kübler-Index/organic matter reflectance correlations. A review. Swiss J. Geosci. 2012, 105, 121–152. [Google Scholar] [CrossRef]
- Bushan, A. Dachschiefervorkommen Deutschlands-petrographisch-geochemische Charakterisierung mit besonderem Blick auf die Goslarer Dachschiefer. Masterarbeit Universität Potsdam. Unpublished. 2017; 1–112. [Google Scholar]
- Chemical Formulas of the Minerals with Changes after. Available online: http://webmineral.com (accessed on 21 February 2020).
- Molar Mass Calculator. Available online: http://www.chemeurope.com/en/tools/ (accessed on 21 February 2020).
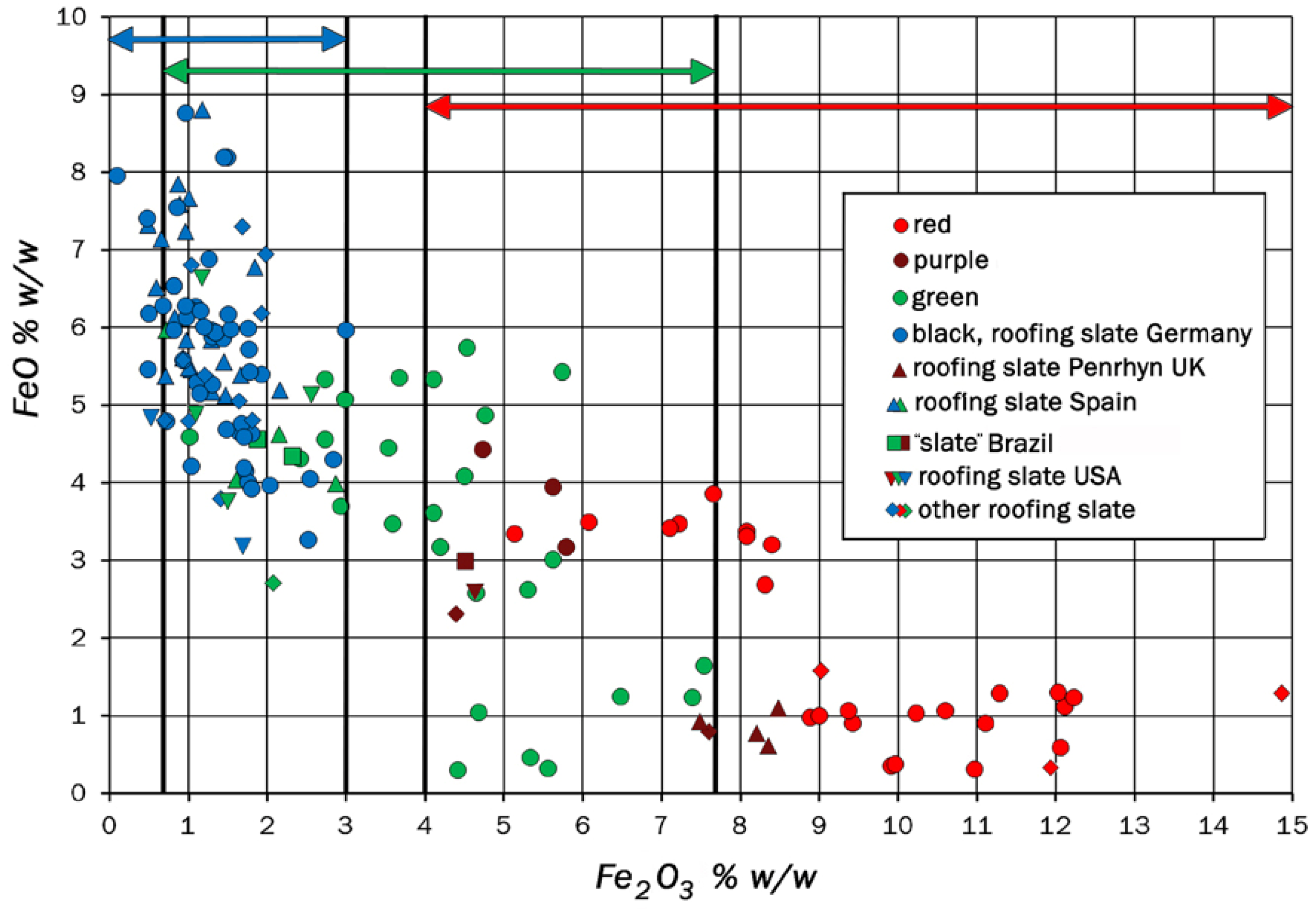







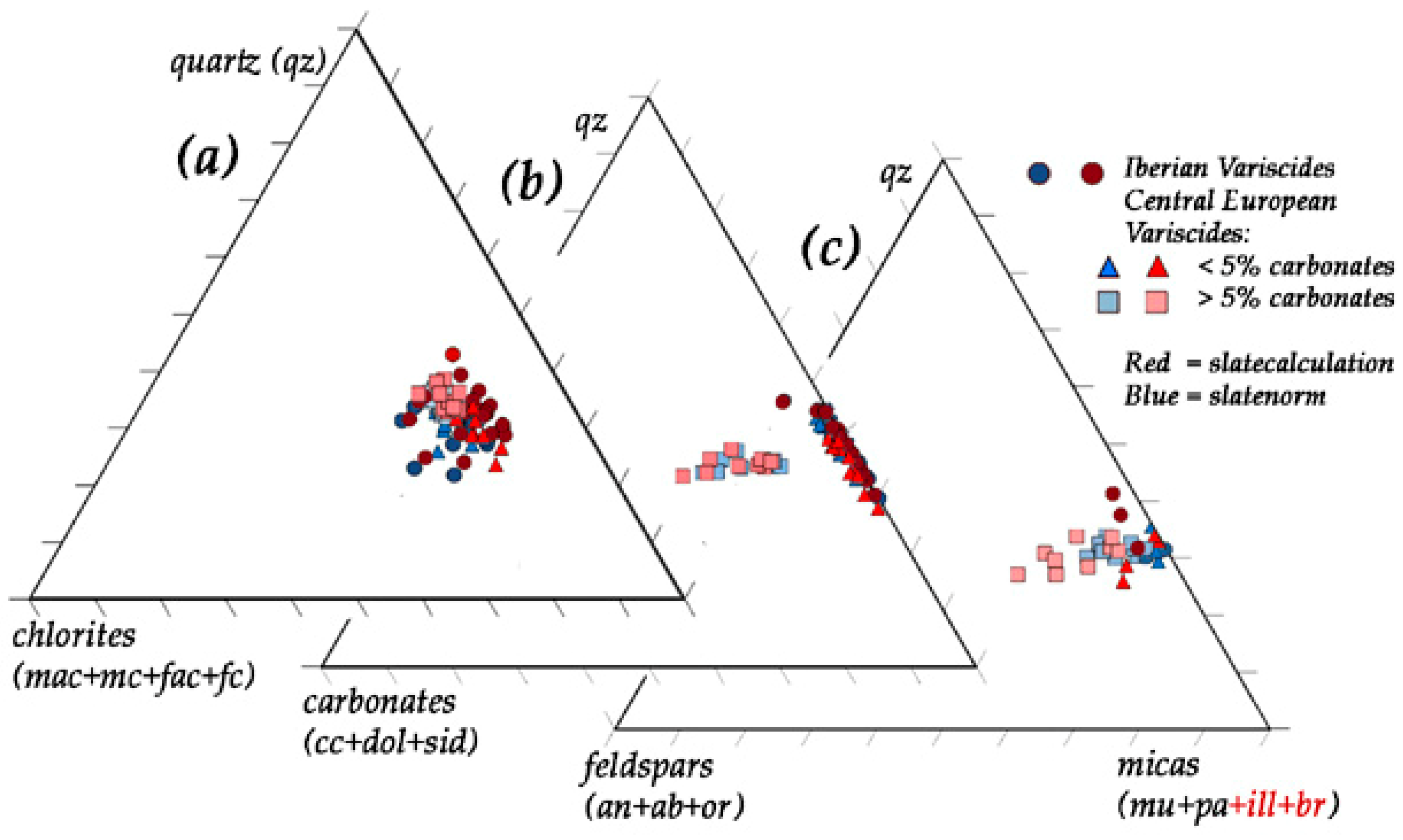
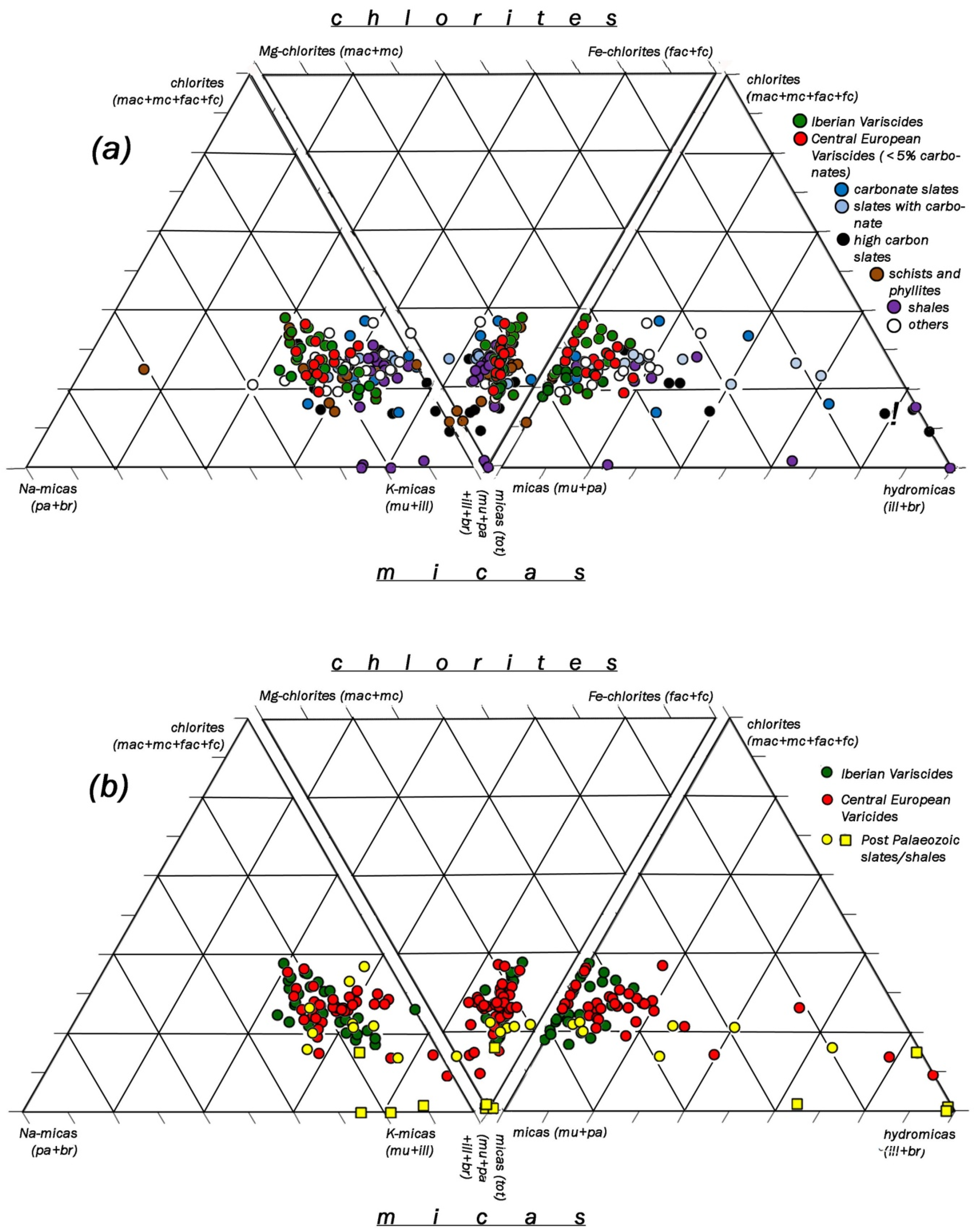
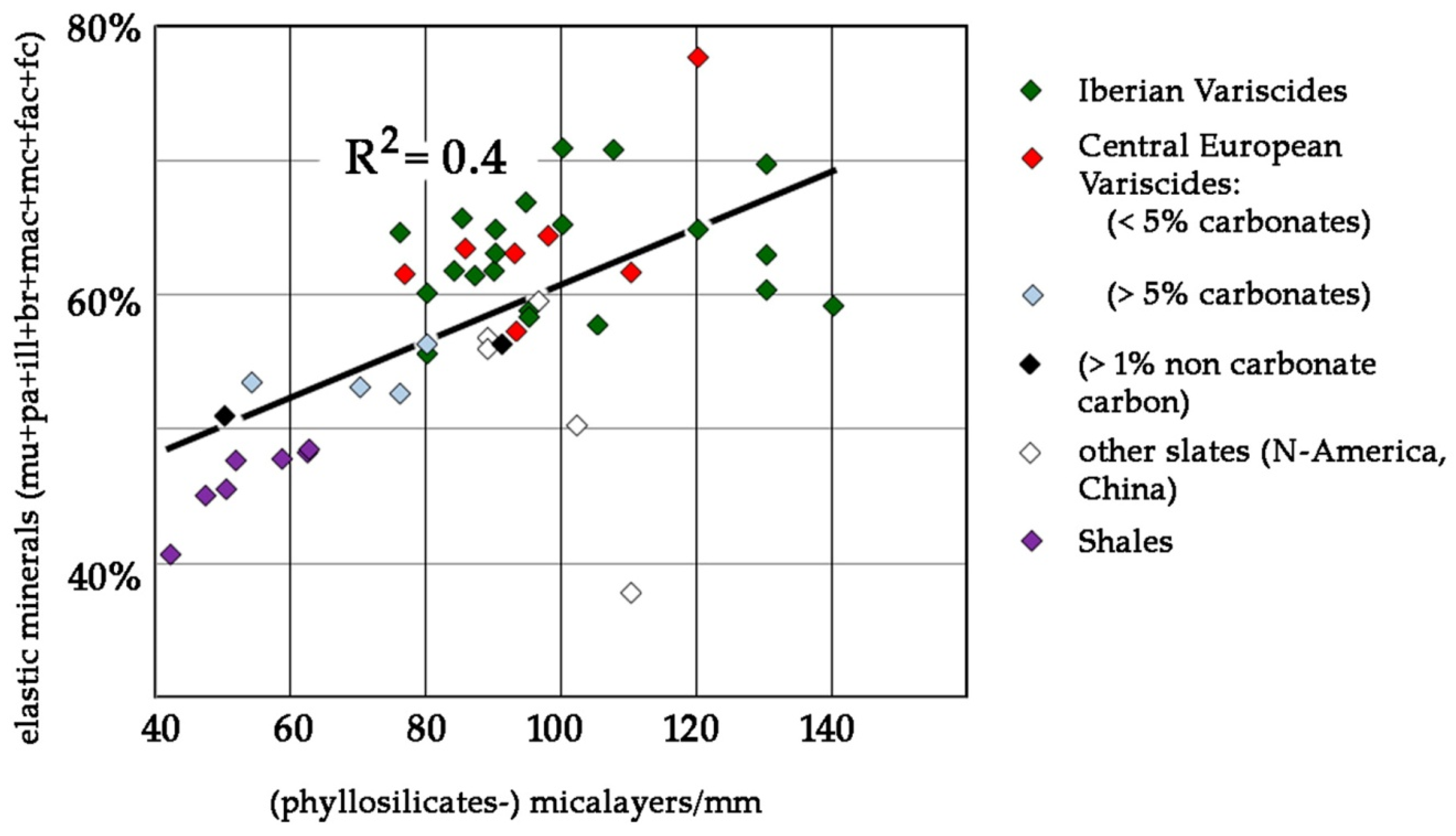
| Country | Province | Region/Location | Ø of Samples | Type of Rock | Formation | Series/System | ||
|---|---|---|---|---|---|---|---|---|
| “normal” slate, Iberian Varicides | ||||||||
| Spain | Asturia | Oscos | Vilarchao | 2 | slate | Luarca | Middle Ordovician | |
| Galicia/Orense | Alto Bierzo | Anllares | 2 | |||||
| Valdeorras | Mormeau | 8 | Argüeira | Casaio | Upper Ordovician | |||
| Los Molinos | 8 | |||||||
| Riodolas | 5 | |||||||
| Rozadais | 3 | Rozadais | ||||||
| Penedo Rayado | 6 | |||||||
| Los Campos | 2 | Losadilla | ||||||
| Castile and León | La Cabrera | San Pedro | 2 | Rozadais | ||||
| Benuza | 2 | |||||||
| La Baña | 2 | |||||||
| Spain | Galicia/Orense | San Vincente | 2 | Soldon | Middle Ordovician | |||
| Quiroga | Pacios gris | 3 | Luarca | |||||
| Pacios negra | 7 | |||||||
| La Campa negra | 1 | |||||||
| France | Anjou | Angers | Trélazé | 2 | Grand-Auverné (Trélazé Mb.) ≈ Luarca | |||
| Portugal | Porto | Valongo | 1 | Valongo ≈ Luarca | ||||
| Arouca | 1 | |||||||
| "normal" slate, Central European Varicides | ||||||||
| BeLux | Ardennes | Martelange | 2 | slate | Schiste ardoisier | Lower Devonian | ||
| Germany | North Rhine-Westphalia | Eifel | Venn | 21 | Revin 4/5 | Black shales | Upper Cambrian | |
| Rhineland-Palatinate | Katzenberg | 13 | Siegenian | Leutesdorf or Aubach | Lower Devonian | |||
| Bausberg | 2 | |||||||
| Margaretha | 7 | |||||||
| Xanten | from a Roman building | 2 | ||||||
| Netherland | Nijmwegen | from a Roman wreck | 2 | |||||
| Germany | Rhineland-Palatinate | Hunsrück | Moselberg | 2 | Lower Emsian | Hunsrück slates | ||
| Altlay | 1 | Altlay | ||||||
| Bundenbach | 5 | Hunsrück slates, Kaub | ||||||
| Middle Rhine | Kaub-Bacharach | 4 | ||||||
| Germany | Rhineland-Palatinate | Lahn | Singhofen | 1 | Lower Emsian | Singhofen | Lower Devonian | |
| Hesse | Langhecke | 1 | Adorf | Bänderschiefer | Upper Devonian | |||
| Bayern | Frankenwald | Lotharheil | 1 | Kulm | Bordenschiefer | Mississipian Carboniferous | ||
| Czech Rep. | Moravia | Olomouc | 5 | Moravice | ||||
| carbonate slate | ||||||||
| Germany | North Rhine-Westphalia | Nuttlar | 1 | carbonate slate | Flinz | Middle to Upper Devonian | ||
| Italy | Liguria | 3 | Val Lavagna | Upper Cretaceous | ||||
| Switzerland | Glarus | Engi | Landesplattenberg | 1 | Nordhelvetischer Flysch, Engi Slate | Eocene | ||
| China | Hubei | Daba Shan | Pingli | 1 | Pingli | Lower Ordovician | ||
| Uruguay | Lavalleja | Arr. Minas Viejas | 12 | Minas Viejas group | Paleoproterozoic | |||
| Arr. Mataojo | 3 | Mataojo group | ||||||
| high carbon content | ||||||||
| Germany | North Rhine-Westphalia | Eifel-Venn | Elise | 7 | slate | Salm 1/2 | Tremadoc | Lower Ordovician |
| Marsberg | 1 | Hangenberg | Upper Devonian to Lower Carboniferous | |||||
| 24 | Kulm | Lydit | Lower Carboniferous | |||||
| 10 | Posidonia | |||||||
| 15 | Clayshales (Marsfeld?) | |||||||
| 28 | Lower Alum | |||||||
| Thuringia | Ronneburg | 3 | Leder Slate | Upper Ordovician to Lower Silurian | ||||
| Lehesten Schmiedebach | 3 | Kulm Lehesten dunkelkiesig | Mississipian Carboniferous | |||||
| Bhutan | Wangdu Phodrang | 2 | above the Chekha | Triassic? | ||||
| Vietnam | Lai Chau/Dien Bien | Quang Chêng | 1 | schiste ardoisier | Triassic | |||
| slate with carbonate | ||||||||
| Germany | Hesse | Lahn-Dill | Haiger | 2 | slate | Eifelian | Wissenbach | Middle Devonian |
| Batzbach | 1 | |||||||
| Rhineland-Palatinate | Lahn | Rupbach | 2 | Rupbach (≈ Wissenbach) | ||||
| North Rhine-Westphalia | Bad Fredeburg | Magog | 12 | Fredeburg (≈ Wissenbach) | ||||
| Felicitas | 7 | |||||||
| Hessen | Willingen | Brilon | 2 | Asten (≈ Wissenbach) | ||||
| Switzerland | St Gallen | Calanda | Pfäfers-Vadura | 1 | Kalkthonschiefer | Eocene | ||
| U. S. A. | New York | Red | 1 | Poultney | Cambrian to Ordoviciann | |||
| Pennsylvania | Lehigh | 1 | Martinsburg | Middle to Upper Ordovician | ||||
| Other slate UK, N-America, China | ||||||||
| UK | Wales | Penrhyn | purple | 5 | slate | Llanberis | Cambrian | |
| Canada | Newfoundland | Trinity Bay | purple | 3 | Bonavista | |||
| green | 1 | |||||||
| U. S. A. | Vermont | Sea green | 1 | Poultney | Cambrian to Ordovician | |||
| Unfading green | 1 | |||||||
| variegated | Eureka | 1 | ||||||
| purple | 1 | |||||||
| black | 1 | Trenton | ||||||
| New York | Bright green | 1 | Poultney | |||||
| China | Hubei | Hongshigou | 3 | Daguiping | s 1ds | Lower Silurian | ||
| Shaanxi | Ziyang | 2 | ||||||
| shales | ||||||||
| Botswana | Southern District | Kanye | 1 | shale | Platberg Group | Rietgat | Neoarchean | |
| Nigeria | Ebonye | Southern Benue trough | 3 | Asu River Group | Abakaliki | Lower Cretaceous | ||
| Brazil | Minas Gerais | Papagaios, Paraopeba, Sete Lagoas | black | 1 | Bambui, Santa Helena | Neoproterozoic | ||
| oliv | 2 | |||||||
| green | 1 | |||||||
| purple | 1 | |||||||
| Santa Catarina | dark green, black | 2 | Tubarao | Mafra | Permian | |||
| Germany | Baden-Wuerttemberg | Dotternhausen | Tafelfleins | 1 | carbonate shale | Posidonia | Lias ɛ | Lower Jurassic |
| Norway | Finnmark | Vestertana | red | 1 | shale | Finnmark Supergroup | Vestertana Group | Riphaen Proterozoic |
| green | 1 | |||||||
| Switzerland | Zurich | Weiach | 4 | shale (high carbon) | Lower Rotliegendes, lakustrine series | Permian | ||
| 1 | Carbonate shale | |||||||
| schists and phyllites | ||||||||
| Spain | Castile and León | Segovia | Bernardos | 4 | phyllite | Schists-Metagraywacke Complex | Neoproterozoic to Lower Cambrian | |
| Galicia | A Terra Cha | Verde Lugo | 2 | Cándana Group | Lower Cambrian | |||
| Portugal | Trás-os-Montes e Alto Douro | Foz Coa | 1 | Desejosa | ||||
| Norway | Finnmark | Alta | 1 | Quar-tzite | Alta Group | Riphaen Proterozoic | ||
| Friarfjord | 2 | phyllite | Laksefjord group | |||||
| Belgium | Wallonia | Ardennes | Vielsalm | 1 | Salm Group | Early to Middle Ordovician | ||
| Ottré redschist | 8 | |||||||
| Ottré coticule | 25 | |||||||
| Germany | Saxony | Vogtland | Theuma, Oelsnitz | 1 | Phycodendachschiefer | Lower Ordovician | ||
| Czech Rep. | Plzeňský kraj | Rapštejn | 2 | Kralupy-Zbraslav group | Proterozoic | |||
| Liberecký kraj | Zelezny Brod | 1 | Radčice Group | Cambrian to Ordovician | ||||
| India | Himachal Pradesh | Himachal | 1 | Chamba and Katarigali | Proterozoic | |||
| Haryana | Bectel | 1 | Delhi Supergroup | Ajabgarh Group | Neo- to Mesoproterozoic | |||
| Argentina | Cuyo | San Luis | 2 | San Luis | Ordovician | |||
| Norm Minerals | Positive | Negative | Color | |||||
|---|---|---|---|---|---|---|---|---|
| Quartz | qz | Ridged minerals | mechanical strength, hardness | resistance to weathering | > 32% w/w brittleness, sometimes worse cleavability, punching quality and finishing quality in round forms | |||
| feldspars | Orthoclase | or | (have a cleavability) | |||||
| Albite | ab | |||||||
| Anorthite | an | |||||||
| micas | Muscovite | mu | Elastic minerals | cause the cleavability, punching quality and finishing quality also in round forms | resistance to weathering | |||
| Paragonite | pa | |||||||
| hydro-micas | Illite | ill | product of weathering | |||||
| Brammalite | br | |||||||
| chlorites | Mg-Al-Chlorite | mac | help with cleavability, punching quality and finishing quality | |||||
| Mg-Chlorite | mc | |||||||
| Fe-Al-Chlorite | fac | blue, green | ||||||
| Fe-Chlorite | fc | |||||||
| carbonates | Calcite | cc | (have a cleavability) | > 5% w/w after EN 12326-1 thickness increase | with higher contents > 5% w/w less resistant to weathering | light grey with oxidation colors weathering ("semiweathering") | ||
| Dolomite | dol | |||||||
| Siderite | sid | |||||||
| ore minerals | Pyrrhotite | pn | > 0.3% w/w and in accumulations oxidation risks | |||||
| Pyrite | pt | in accumulations of micro pyrites oxidation risks | ||||||
| Hematite | he | > 2% w/w red, purple | ||||||
| Limonite | lm | product of weathering | ||||||
| Graphite | gr | 2% w/w after EN 12326-1 does not permit | black | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wagner, H.W.; Jung, D.; Wagner, J.-F.; Wagner, M.P. Slatecalculation—A Practical Tool for Deriving Norm Minerals in the Lowest-Grade Metamorphic Pelites and Roof Slates. Minerals 2020, 10, 395. https://doi.org/10.3390/min10050395
Wagner HW, Jung D, Wagner J-F, Wagner MP. Slatecalculation—A Practical Tool for Deriving Norm Minerals in the Lowest-Grade Metamorphic Pelites and Roof Slates. Minerals. 2020; 10(5):395. https://doi.org/10.3390/min10050395
Chicago/Turabian StyleWagner, Hans Wolfgang, Dieter Jung, Jean-Frank Wagner, and Matthias Patrick Wagner. 2020. "Slatecalculation—A Practical Tool for Deriving Norm Minerals in the Lowest-Grade Metamorphic Pelites and Roof Slates" Minerals 10, no. 5: 395. https://doi.org/10.3390/min10050395
APA StyleWagner, H. W., Jung, D., Wagner, J.-F., & Wagner, M. P. (2020). Slatecalculation—A Practical Tool for Deriving Norm Minerals in the Lowest-Grade Metamorphic Pelites and Roof Slates. Minerals, 10(5), 395. https://doi.org/10.3390/min10050395






