Up-Cycling of Iron-Rich Inorganic Waste in Functional Glass-Ceramics
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Effect of Firing Atmosphere on Microstructure and Properties of Fly Ash-Derived Glass-Ceramic Foams
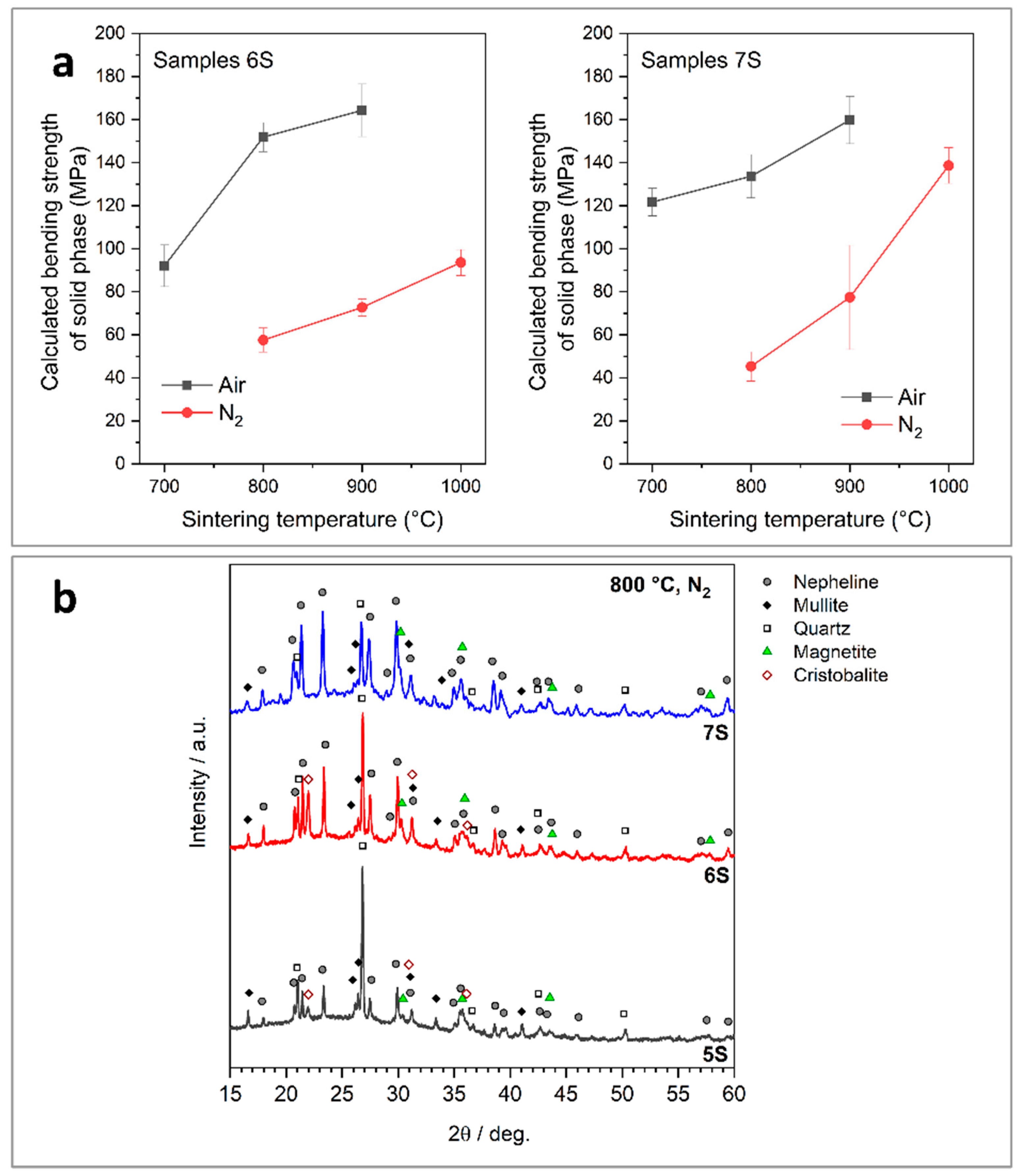
3.2. Effect of Glass Content on Microstructure and Properties of Fly Ash-Derived Glass-Ceramic Foams
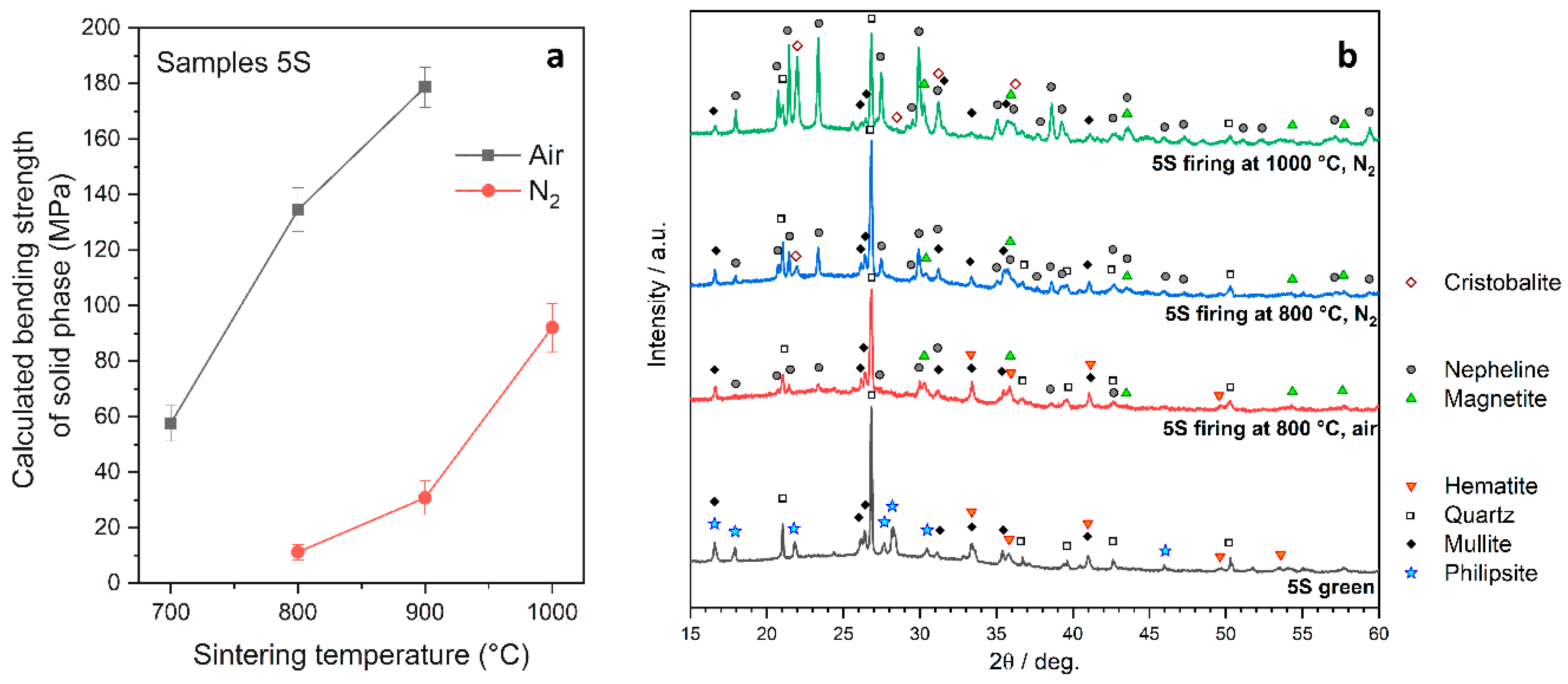
3.3. Effect of Firing Temperature on Microstructure and Properties of Fly Ash-Derived Glass-Ceramic Foams
3.4. Extension of the Approach to Red Mud-Derived Glass-Ceramic Foams
4. Conclusions
- The inclusion of fly ash (FA) powders mixed with soda-lime glass (SLG) waste in weakly alkaline solutions was confirmed as an excellent approach to produce glass-ceramic foams, allowing the incorporation of high proportions of fly ash.
- Gelation of FA/SLG suspensions was not a final stage, but it enabled the manufacturing of highly porous bodies by simple frothing.
- The decomposition of the gel and the SLG/FA interactions upon firing in N2 promoted the formation of new phases, among which nepheline was dominant; such a phase was stimulated by the separation of iron oxide into magnetite phase, in turn favored by the adoption of an inert atmosphere.
- The modifications in the phase assemblage, promoted by treatments in nitrogen, favored the chemical stability of the glass-ceramics as assessed by means of leaching tests.
- Firing in nitrogen, compared to firing in air, generally led to mechanically weaker samples according to the modifications in phase assemblage at low temperature; however, treatments at 1000 °C led to foams with remarkable strength.
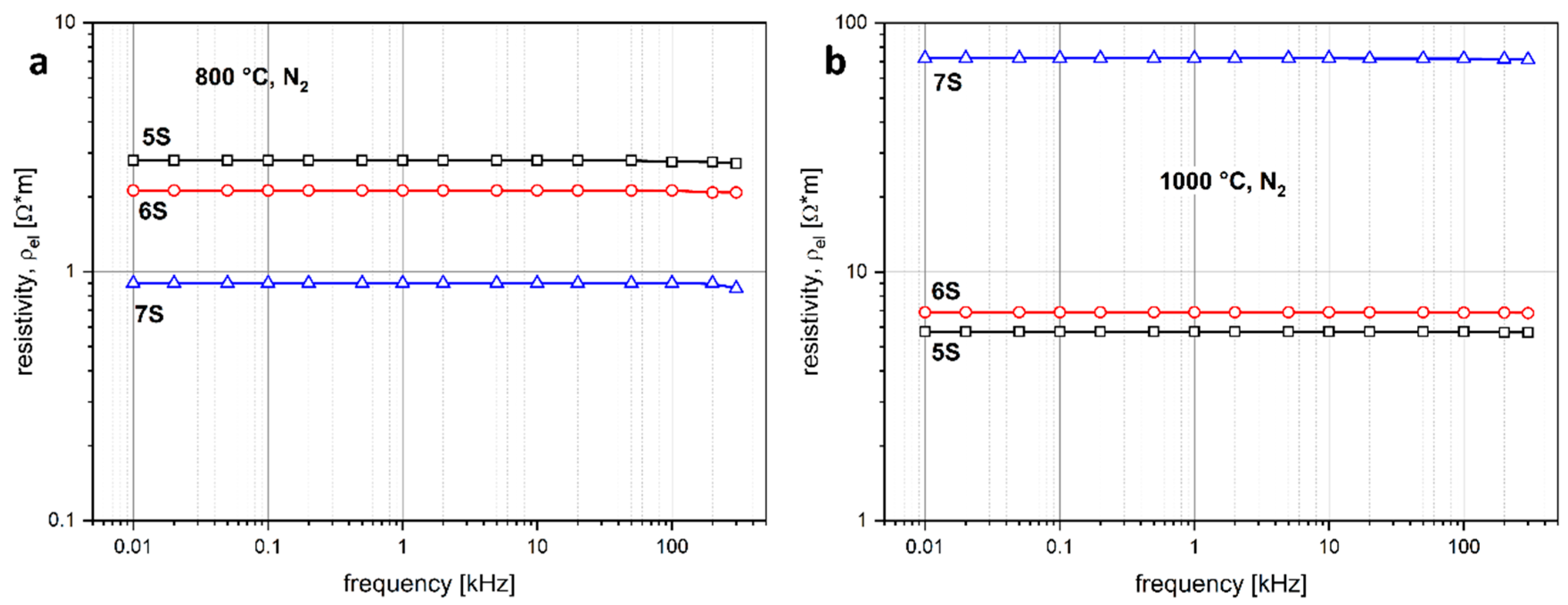
- Treatments in nitrogen atmosphere, although more expensive, bring advantages, besides in durability, in the development of electrical functionalities that could be tuned according to FA/SLG proportions and firing temperature. Semiconductive or highly polarizable foams could find applications in electrocatalysis and energy generation.
- The distribution of iron in the glass-ceramic and, in particular, its concentration in magnetite inclusions, controls the electrical functionality; the results from FA/SLG mixtures were substantially confirmed by additional glass-ceramic foams (still featuring nepheline) from red mud/SLG mixtures.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Korhonen, J.; Nuur, C.; Feldmann, A.; Birkie, S.E. Circular economy as an essentially contested concept. J. Clean. Prod. 2018, 175, 544–552. [Google Scholar] [CrossRef]
- Korhonen, J.; Honkasalo, A.; Seppälä, J. Circular Economy: The Concept and its Limitations. Ecol. Econ. 2018, 143, 37–46. [Google Scholar] [CrossRef]
- Smol, M.; Kulczycka, J.; Henclik, A.; Gorazda, K.; Wzorek, Z. The possible use of sewage sludge ash (SSA) in the construction industry as a way towards a circular economy. J. Clean. Prod. 2015, 95, 45–54. [Google Scholar] [CrossRef]
- Yao, Z.T.; Ji, X.S.; Sarker, P.K.; Tang, J.H.; Ge, L.Q.; Xia, M.S.; Xi, Y.Q. A comprehensive review on the applications of coal fly ash. Earth-Sci. Rev. 2015, 141, 105–121. [Google Scholar] [CrossRef]
- Lima, M.S.S.; Thives, L.P.; Haritonovs, V.; Bajars, K. Red mud application in construction industry: Review of benefits and possibilities. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; Volume 251, p. 012033. [Google Scholar]
- Al Bakri, M.M.; Mohammed, H.; Kamarudin, H.; Khairul Niza, I.; Zarina, Y. Review on fly ash-based geopolymer concrete without Portland Cement. J. Eng. Technol. Res. 2011, 3, 1–4. [Google Scholar]
- Dimas, D.D.; Giannopoulou, I.P.; Panias, D. Utilization of alumina red mud for synthesis of inorganic polymeric materials. Min. Proc. Extr. Met. Rev. 2009, 30, 211–239. [Google Scholar] [CrossRef]
- Toniolo, N.; Boccaccini, A.R. Fly ash-based geopolymers containing added silicate waste. A review. Ceram. Int. 2017, 43, 14545–14551. [Google Scholar] [CrossRef]
- Provis, J.L. Geopolymers and other alkali activated materials: Why, how, and what? Mater. Struct. 2014, 47, 11–25. [Google Scholar] [CrossRef]
- Tchakouté, H.K.; Rüscher, C.H.; Kong, S.; Kamseu, E.; Leonelli, C. Geopolymer binders from metakaolin using sodium waterglass from waste glass and rice husk ash as alternative activators: A comparative study. Const. Build. Mat. 2016, 114, 276–289. [Google Scholar] [CrossRef]
- Toniolo, N.; Rincón, A.; Roether, J.A.; Ercole, P.; Bernardo, E.; Boccaccini, A.R. Extensive reuse of soda-lime waste glass in fly ash-based geopolymers. Const. Build. Mat. 2018, 188, 1077–1084. [Google Scholar] [CrossRef]
- Toniolo, N.; Rincón, A.; Avadhut, Y.S.; Hartmann, M.; Bernardo, E.; Boccaccini, A.R. Novel geopolymers incorporating red mud and waste glass cullet. Mater. Lett. 2018, 219, 152–154. [Google Scholar]
- Toniolo, N.; Taveri, G.; Hurle, K.; Roether, J.A.; Ercole, P.; Dlouhy, I.; Boccaccini, A.R. Fly-Ash-Based Geopolymers: How the Addition of Recycled Glass or Red Mud Waste Influences the Structural and Mechanical Properties. J. Ceram. Sci. Technol. 2017, 8, 411–420. [Google Scholar]
- Idir, R.; Cyr, M.; Pavoine, A. Investigations on the durability of alkali-activated recycled glass. Const. Build. Mat. 2020, 235, 117477. [Google Scholar]
- Rincón Romero, A.; Toniolo, N.; Boccaccini, A.R.; Bernardo, E. Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures. Materials 2019, 12, 588. [Google Scholar]
- Rabelo Monich, P.; Rincón Romero, A.; Desideri, D.; Bernardo, E. Waste-derived glass-ceramics fired in nitrogen: Stabilization and functionalization. Const. Build. Mat. 2020, 232, 117265. [Google Scholar]
- Gibson, L.J.; Ashby, M.F. Cellular Solids: Structure and Properties, 2nd ed.; Cambridge University Press: Cambridge, UK, 2014; pp. 1–510. [Google Scholar]
- Cetin, S.; Marangoni, M.; Bernardo, E. Lightweight glass-ceramic tiles from the sintering of mining tailings. Ceram. Int. 2015, 41, 5294–5300. [Google Scholar]
- Chmelík, F.; Trník, A.; Štubňa, I.; Pešička, J. Creation of microcracks in porcelain during firing. J. Eur. Ceram. Soc. 2011, 31, 2205–2209. [Google Scholar]
- Shaharyar, Y.; Cheng, J.Y.; Han, E.; Maron, A.; Weaver, J.; Marcial, J.; McCloy, J.S.; Goel, A. Elucidating the Effect of Iron Speciation (Fe2+/Fe3+) on Crystallization Kinetics of Sodium Aluminosilicate Glasses. J. Am. Ceram. Soc. 2016, 99, 2306–2315. [Google Scholar]
- Duke, D.A.; MacDowell, J.F.; Karstetter, B.R. Crystallization and Chemical Strengthening of Nepheline Glass-Ceramics. J. Am. Ceram. Soc. 1967, 50, 67–74. [Google Scholar]
- Ahmadzadeh, M.; Marcial, J.; McCloy, J.S. Crystallization of iron-containing sodium aluminosilicate glasses in the NaAlSiO4-NaFeSiO4 join. JGR Solid Earth 2017, 122, 2504–2524. [Google Scholar]
- Onuma, K.; Iwai, T.; Yagi, K. Nepheline-”Iron-Nepheline” Solid Solutions. J. Fac. Sci. Hokkaido Univ. 1972, 15, 179–190. [Google Scholar]
- Sargin, I.; Lonergan, C.E.; Vienna, J.D.; McCloy, J.S.; Beckman, S.P. A data-driven approach for predicting nepheline crystallization in high-level waste glasses. J. Am. Ceram. Soc. 2020, 103, 4913–4924. [Google Scholar] [CrossRef]
- Ezz Eldin, F.M.; El Alaily, N.A. Electrical conductivity of some alkali silicate glasses. Mat. Chem. Phys. 1998, 52, 175–179. [Google Scholar] [CrossRef]
- Abdullah, M.H.; Yusoff, A.N. Complex impedance and dielectric properties of an Mg-Zn ferrite. J. Alloys Compd. 1996, 233, 129–135. [Google Scholar] [CrossRef]
- Gillot, B.; Bouton, F.; Chassagneux, F.; Rousset, A. Electrical conductivity of (Fe2+Al2−x3+Crx3+)O42− spinels and defect phases γ(Fe1/33+Al2/3−y3+Cry3+)2O32−. Phys. St. Sol. 1978, 50, 109–116. [Google Scholar] [CrossRef]
- Sastry, M.D.; Nagar, Y.C.; Bhushan, B.; Mishra, K.P.; Balaram, V.; Singhvi, A.K. An unusual radiation dose dependent EPR line at geff = 2.54 in feldspars: Possible evidence of Fe3+O2− ↔ Fe2+O− and exchange coupled Fe3+–Fe2+–nO−. J. Phys.: Cond. Mat. 2007, 20, 025224. [Google Scholar]
- Li, W.-H.; Lv, J.; Li, Q.; Xie, J.; Ogiwara, N.; Huang, Y.; Jiang, H.; Kitagawa, H.; Xu, G.; Wang, Y. Conductive metal–organic framework nanowire arrays for electrocatalytic oxygen evolution. J. Mater. Chem. A 2019, 7, 10431. [Google Scholar] [CrossRef]
- Canuto de Almeida e Silva, T.; Bhowmick, G.D.; Ghangrekar, M.M.; Wilhelm, M.; Rezwan, K. SiOC-based polymer derived-ceramic porous anodes for microbial fuel cells. Biochem. Eng. J. 2019, 148, 29–36. [Google Scholar] [CrossRef]
- Breneman, R.C.; Halloran, J.W. Effect of Cristobalite on the Strength of Sintered Fused Silica Above and Below the Cristobalite Transformation. J. Am. Ceram. Soc. 2015, 98, 1611–1617. [Google Scholar] [CrossRef]
- Li, Y.; Huang, X.; Hu, Z.; Jiang, P.; Li, S.; Tanaka, T. Large Dielectric Constant and High Thermal Conductivity in Poly (vinylidene fluoride)/Barium Titanate/Silicon Carbide Three-Phase Nanocomposites. ACS Appl. Mater. Interfaces 2011, 3, 4396–4403. [Google Scholar] [CrossRef]
- Prasad, B.V.; Rao, G.N.; Chen, J.W.; Babu, D.S. Relaxor ferroelectric like giant permittivity in PrCrO3 semiconductor ceramics. Mat. Chem. Phys. 2011, 126, 918–921. [Google Scholar] [CrossRef]
- Jonscher, A.K. Dielectric relaxation in solids. J. Phys. D Appl. Phys. 1999, 32, R57. [Google Scholar] [CrossRef]
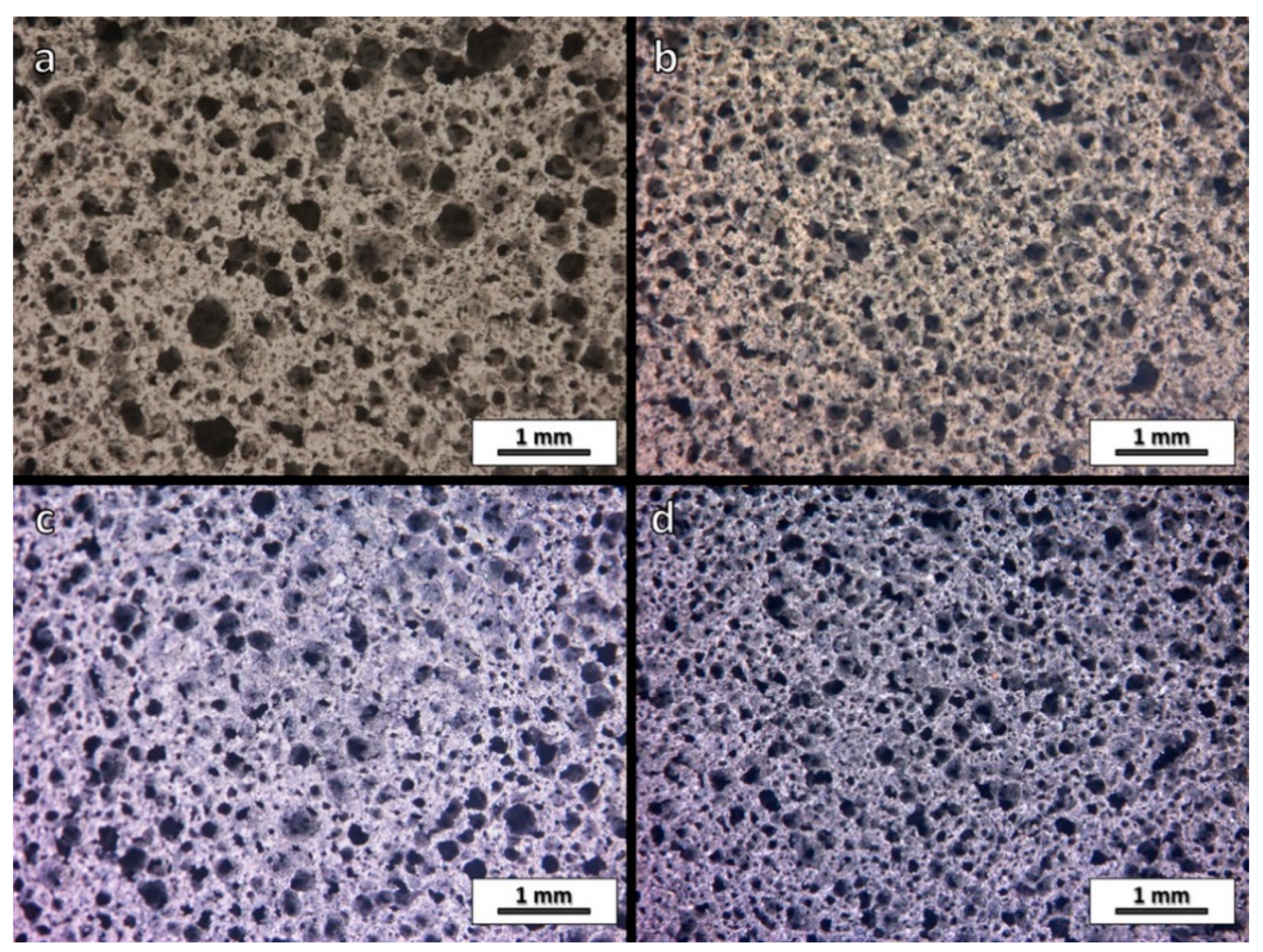
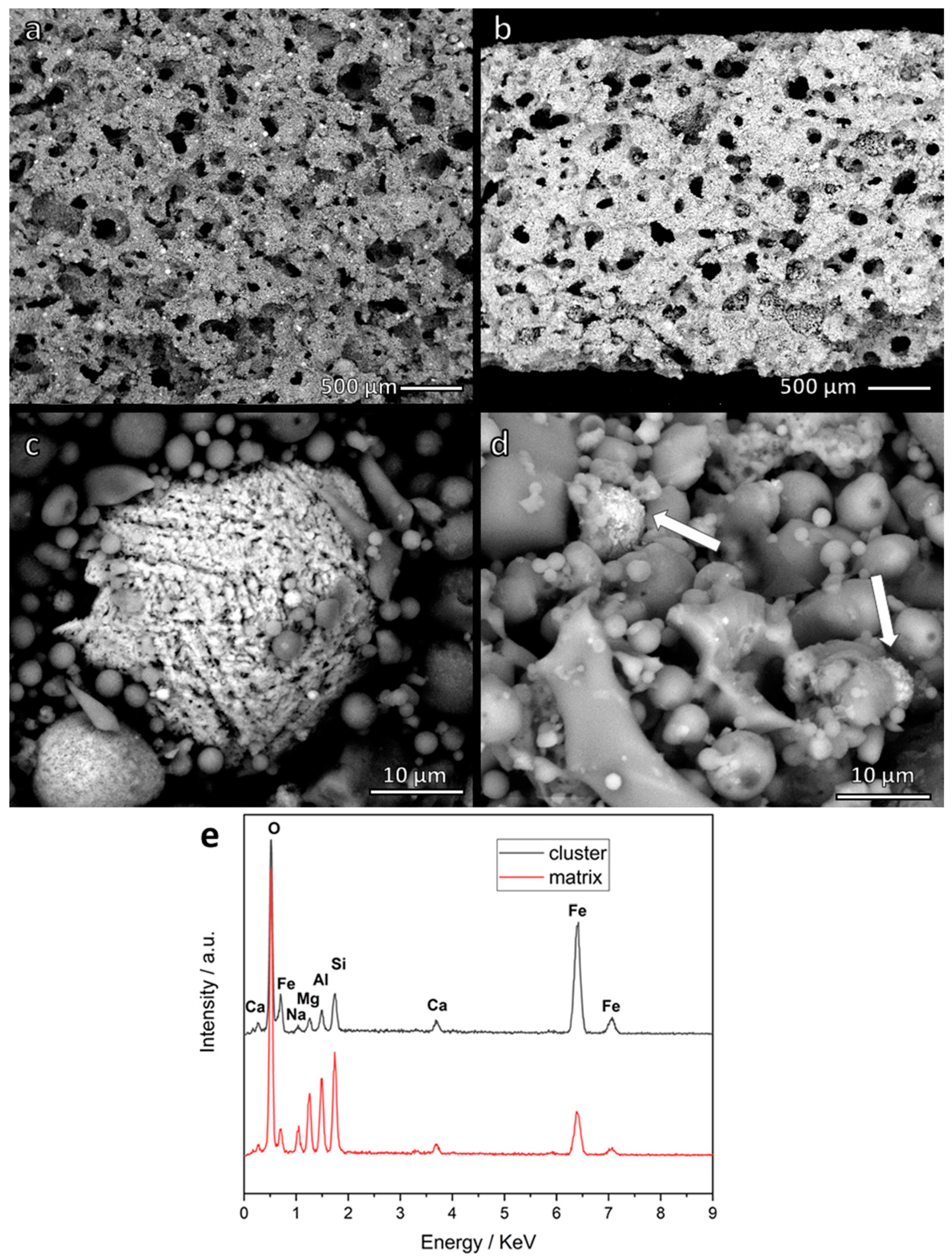
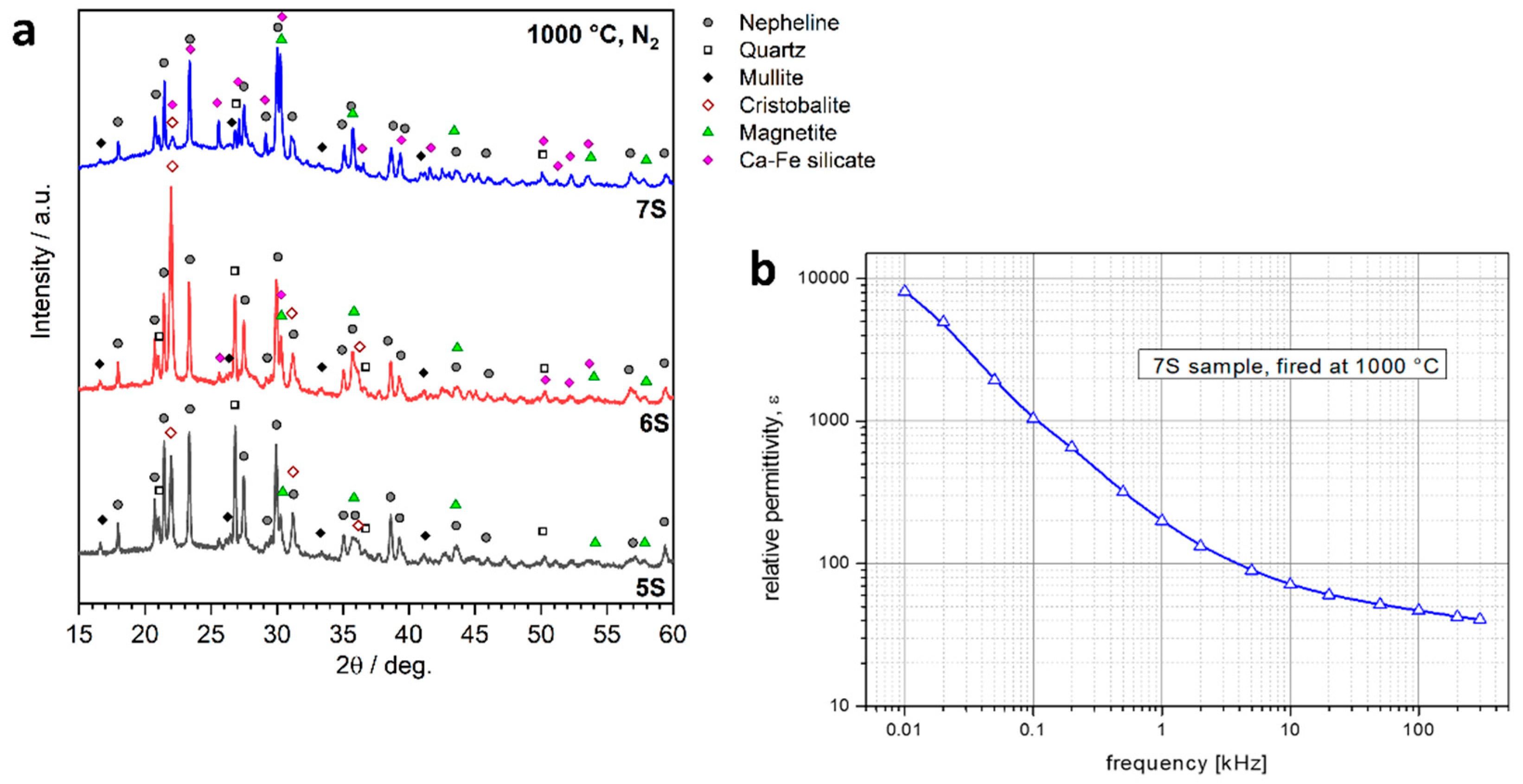



| Oxide (wt.%) | SiO2 | Al2O3 | Na2O | K2O | CaO | MgO | Fe2O3 | TiO2 |
|---|---|---|---|---|---|---|---|---|
| FA | 54.36 | 24.84 | 0.83 | 3.03 | 2.56 | 2.06 | 8.28 | 1.07 |
| SLG | 70.50 | 3.20 | 12.00 | 1.00 | 10.00 | 2.30 | 0.42 | 0.07 |
| RM | 5.21 | 15.21 | 2.40 | 0.63 | 2.95 | 0.38 | 52.94 | 8.05 |
| Property | 5S | 6S | 7S | RM | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Firing temperature (°C) | 800 | 900 | 1000 | 800 | 900 | 1000 | 800 | 900 | 1000 | 800 |
| Density (g/cm3) | 0.51 ± 0.01 | 0.54 ± 0.01 | 0.81 ± 0.01 | 0.52 ± 0.01 | 0.54 ± 0.01 | 0.78 ± 0.01 | 0.61 ± 0.01 | 0.67 ± 0.01 | 0.94 ± 0.01 | 1.18 ± 0.04 |
| Total porosity (vol%) | 80 ± 2 | 81 ± 2 | 69 ± 1 | 80 ± 2 | 81 ± 2 | 70 ± 1 | 77 ± 6 | 76 ± 5 | 64 ± 1 | 65 ± 1 |
| Open porosity (vol%) | 79 ± 2 | 78 ± 2 | 68 ± 1 | 78 ± 2 | 77 ± 2 | 68 ± 1 | 76 ± 5 | 73 ± 2 | 62 ± 1 | 64 ± 1 |
| Compressive strength (MPa) | 0.2 ± 0.1 | 0.5 ± 0.1 | 3.2 ± 0.3 | 1.0 ± 0.1 | 1.2 ± 0.1 | 2.4 ± 0.1 | 1.0 ± 0.1 | 1.8 ± 0.3 | 6.0 ± 0.4 | 4.8 ± 0.6 |
| Element | Leachates (ppm) | Limits (ppm) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 5S | 6S | 7S | RM | ||||||
| Air [15] | N2 | Air [15] | N2 | Air [15] | N2 | N2 | Inert | Non-Hazardous | |
| As | 0.0316 | 0.0061 | 0.068 | 0.0521 | 0.0491 | 0.0475 | 0.4876 | 0.5 | 2 |
| Ba | >Al | <0.000 | >Al | <0.000 | 0.0672 | <0.000 | 0.0131 | 20 | 100 |
| Cd | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | 0.04 | 1 |
| Cr | 0.3406 | 0.0008 | 0.0255 | 0.0004 | 0.0689 | 0.0009 | 0.0303 | 0.5 | 10 |
| Cu | 0.0029 | 0.0048 | 0.0024 | 0.0023 | 0.0053 | 0.0034 | 0.0579 | 2 | 50 |
| Hg | 0.0032 | <0.0004 | 0.0006 | <0.0004 | 0.0020 | <0.0004 | <0.0004 | 0.01 | 0.2 |
| Mo | 0.5324 | 0.4542 | 0.2107 | 0.0158 | 0.1973 | 0.4925 | 0.0188 | 0.5 | 10 |
| Ni | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | 0.0146 | 0.4 | 10 |
| Pb | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | 0.0084 | 0.5 | 10 |
| Se | 0.0133 | 0.0168 | 0.0255 | 0.0194 | 0.0226 | 0.0177 | 0.0362 | 0.1 | 0.5 |
| Zn | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | 4 | 50 |
| Element | Leachates (ppm) | Limits (ppm) | ||||||
|---|---|---|---|---|---|---|---|---|
| 5S | 6S | 7S | ||||||
| 800 °C | 1000 °C | 800 °C | 1000 °C | 800 °C | 1000 °C | Inert | Non-Hazardous | |
| As | 0.0061 | <0.0049 | 0.0521 | 0.0521 | 0.0475 | 0.0188 | 0.5 | 2 |
| Ba | <0.000 | 0.0028 | <0.000 | <0.000 | <0.000 | <0.000 | 20 | 100 |
| Cd | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | 0.04 | 1 |
| Cr | 0.0008 | 0.0007 | 0.0004 | 0.0004 | 0.0009 | 0.0009 | 0.5 | 10 |
| Cu | 0.0048 | 0.0074 | 0.0023 | 0.0054 | 0.0034 | 0.0072 | 2 | 50 |
| Hg | <0.0004 | <0.0004 | <0.0004 | <0.0004 | <0.0004 | <0.0004 | 0.01 | 0.2 |
| Mo | 0.4542 | 0.0129 | 0.0158 | <0.0033 | 0.4925 | 0.0735 | 0.5 | 10 |
| Ni | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | 0.4 | 10 |
| Pb | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | 0.5 | 10 |
| Se | 0.0168 | 0.0187 | 0.0194 | 0.0230 | 0.0177 | <0.0122 | 0.1 | 0.5 |
| Zn | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | 4 | 50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rincón Romero, A.; Desideri, D.; Boccaccini, A.R.; Bernardo, E. Up-Cycling of Iron-Rich Inorganic Waste in Functional Glass-Ceramics. Minerals 2020, 10, 959. https://doi.org/10.3390/min10110959
Rincón Romero A, Desideri D, Boccaccini AR, Bernardo E. Up-Cycling of Iron-Rich Inorganic Waste in Functional Glass-Ceramics. Minerals. 2020; 10(11):959. https://doi.org/10.3390/min10110959
Chicago/Turabian StyleRincón Romero, Acacio, Daniele Desideri, Aldo R. Boccaccini, and Enrico Bernardo. 2020. "Up-Cycling of Iron-Rich Inorganic Waste in Functional Glass-Ceramics" Minerals 10, no. 11: 959. https://doi.org/10.3390/min10110959
APA StyleRincón Romero, A., Desideri, D., Boccaccini, A. R., & Bernardo, E. (2020). Up-Cycling of Iron-Rich Inorganic Waste in Functional Glass-Ceramics. Minerals, 10(11), 959. https://doi.org/10.3390/min10110959







