Characterization of Fine Fractions from the Processing of Municipal Solid Waste Incinerator Bottom Ashes for the Potential Recovery of Valuable Metals
Abstract
1. Introduction
1.1. Municipal Solid Waste Incineration
1.2. Municipal Waste Incineration Bottom Ash
- (1)
- Mineral fraction (coarse): 32%;
- (2)
- Grit < 2 mm, slag: 27% each;
- (3)
- Metals: 11%;
- (4)
- Organics: 3%.
1.3. Processing—State of the Art
2. Materials and Methods
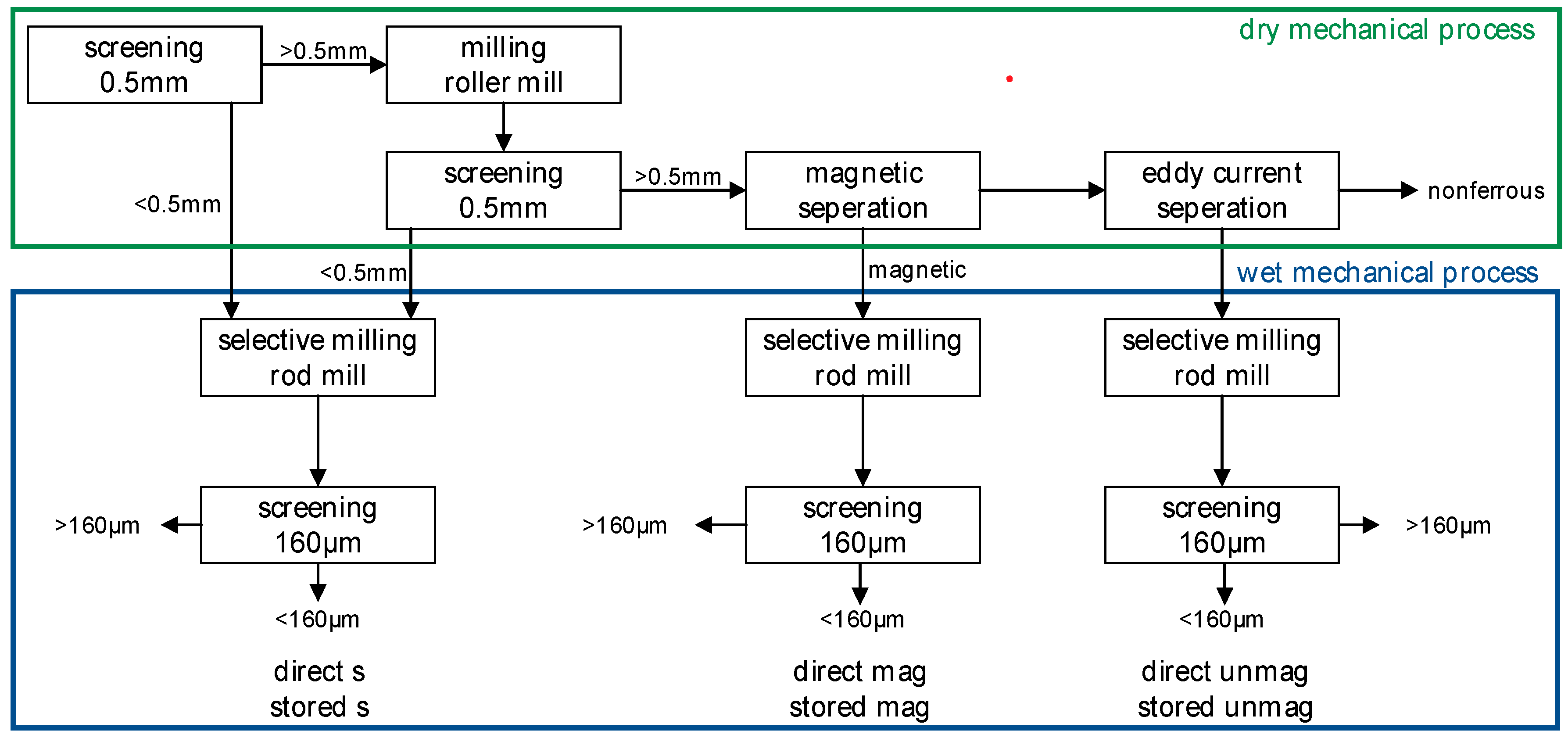
2.1. Material
- (1)
- Dry mechanical processing to separate coarse material;
- (2)
- Wet mechanical processing to separate finer metal particles;
- (3)
- Hydrometallurgical processing to recover the residual metal content.
2.2. Analytical Methods
3. Results
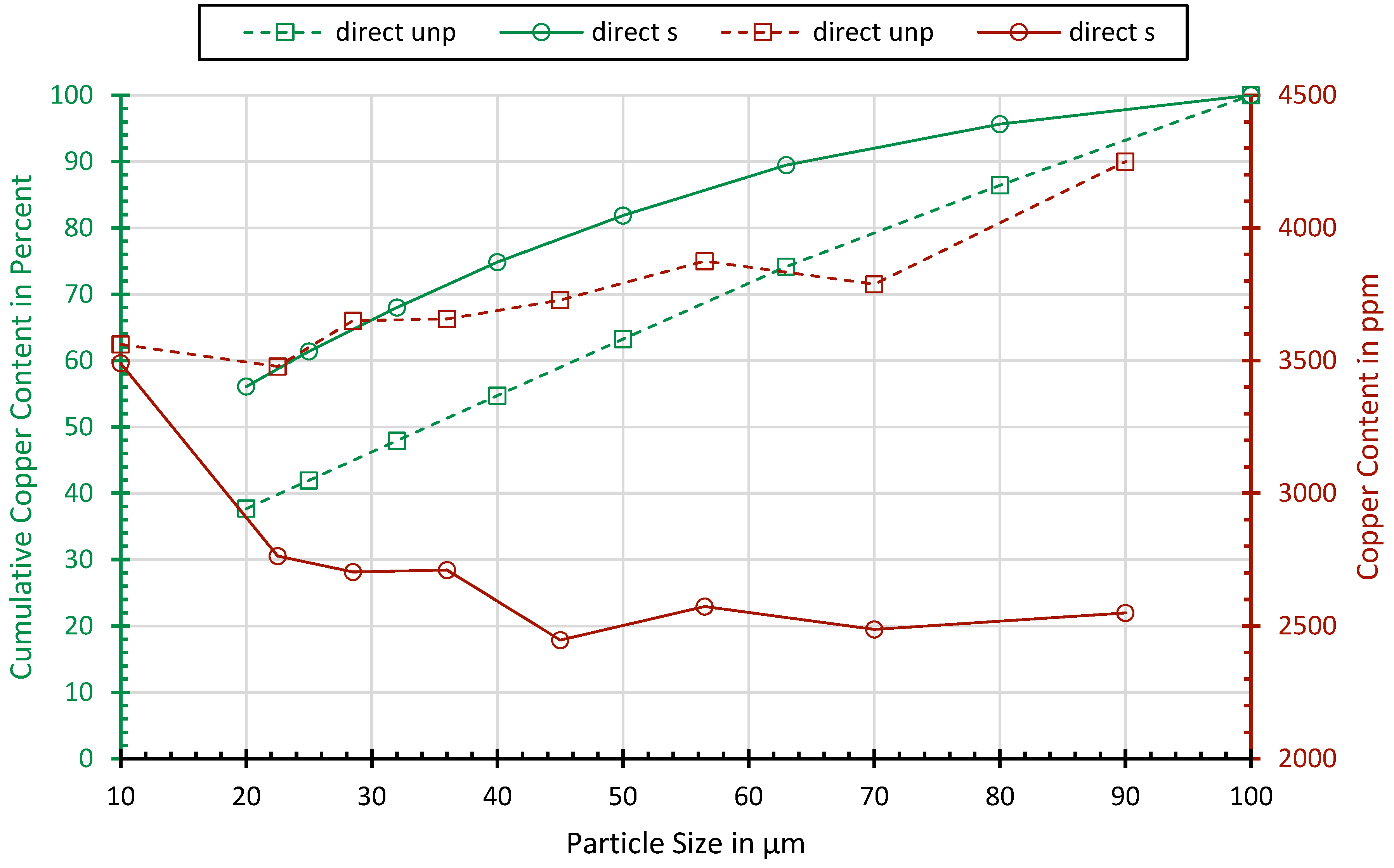
3.1. Particle Size Distribution
3.2. Bulk Chemistry
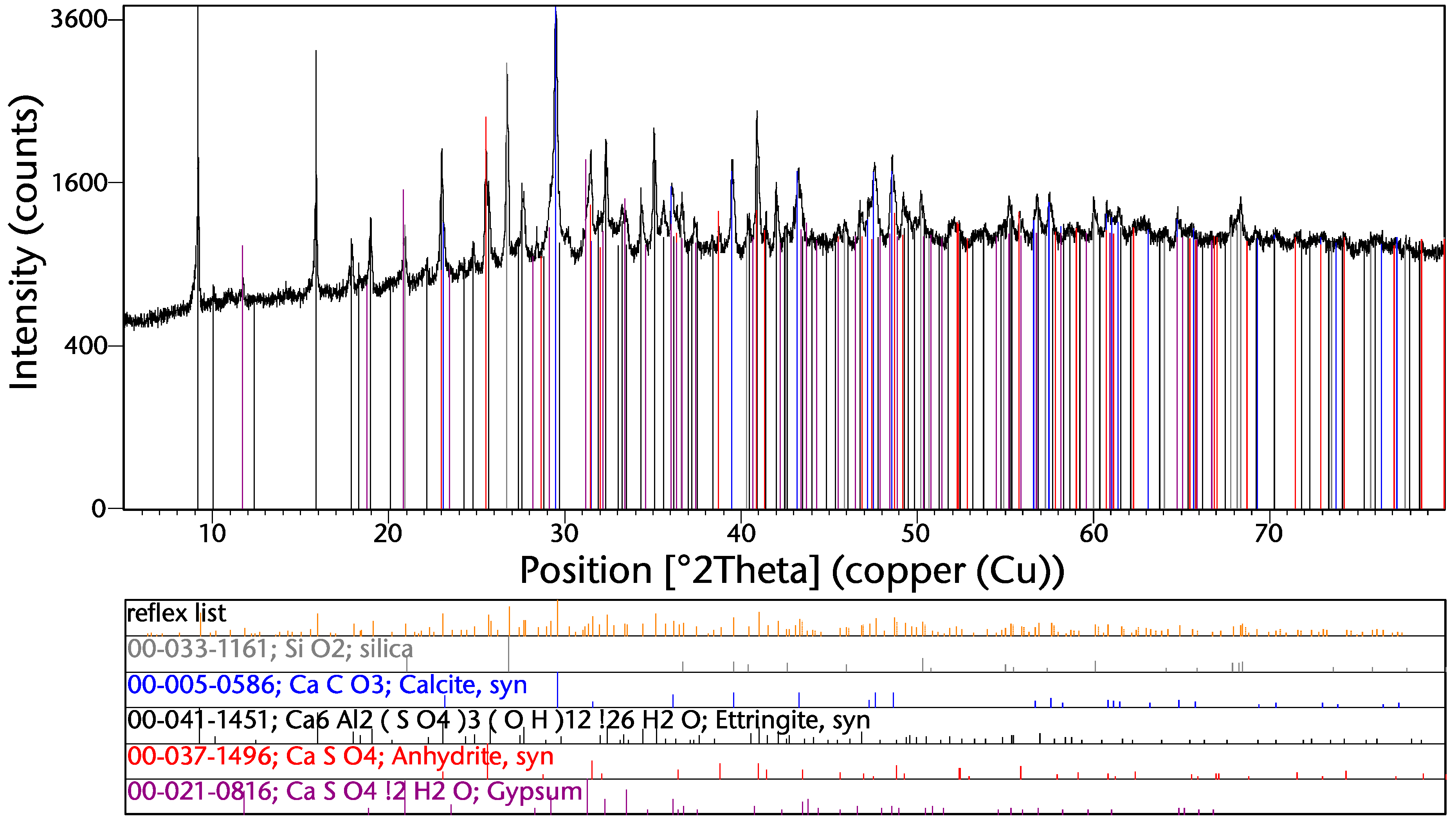
3.3. Bulk Phase Composition
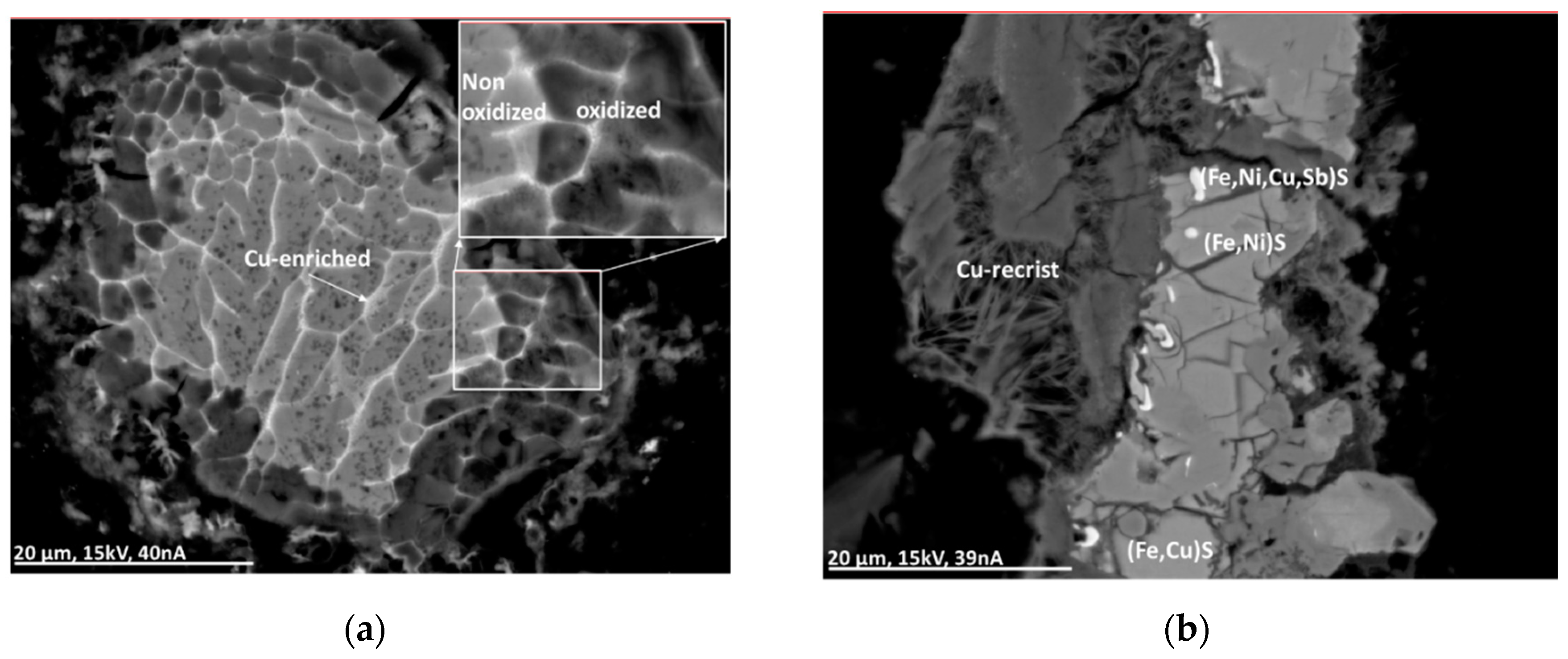
3.4. Electron Imaging and Electron Probe Microanalysis
3.4.1. Overview
3.4.2. Metals/Alloys
3.4.3. Sulfides
3.4.4. Oxides
3.4.5. Glass and Silicates
3.4.6. Carbonate, Phosphate, and Sulfate
3.4.7. Incorporation of Valuable Metals
3.4.8. Metal Enrichment in Different Fractions
3.4.9. Alteration
4. Discussion
4.1. Mineralogy and Chemistry
4.2. Waste Incineration Process
4.3. Processing
5. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- State-of-the-art for practitioners, analysts, and scientists. In Handbook of Recycling; Worrell, E., Reuter, M.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-0-12-396459-5. [Google Scholar]
- Schubert, H. Handbuch der Mechanischen Verfahrenstechnik; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 9783527305773. [Google Scholar]
- Deike, R.; Ebert, D.; Warnecke, R.; Vogell, M. Abschlussbericht zum Projekt „Recyclingpotenziale bei Rückständen aus der Müllverbrennung“. Available online: https://www.itad.de/wissen/studien/20130110_deike-hmvarecyclingpotentialabschlussbericht.pdf (accessed on 30 March 2020).
- Wei, Y.; Shimaoka, T.; Saffarzadeh, A.; Takahashi, F. Mineralogical characterization of municipal solid waste incineration bottom ash with an emphasis on heavy metal-bearing phases. J. Hazard. Mater. 2011, 187, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Speiser, C. Exothermer Stoffumsatz in MVA-Schlackedeponien: Mineralogische und geochemische Charakterisierung von Müllverbrennungsschlacken, Stoff-und Wärmebilanz. Ph.D. Thesis, Technische Universität München, München, Germany, 2001. [Google Scholar]
- Elwert, T.; Goldmann, D.-I.D.; Schirmer, T.; Strauß, K. Affinity of Rare Earth Elements to Silico-Phosphate Phases in the System Al2O3-CaO-MgO-P2O5-SiO2. Chem. Ing. Tech. 2014, 86, 840–847. [Google Scholar] [CrossRef]
- Elwert, T.; Goldmann, D.; Schirmer, T.; Strauß, K. Phase composition of high lithium slags from the recycling of lithium ion batteries. Erzmetall 2012, 163–171. [Google Scholar]
- Friedrich, B.; Zander, M.; Kemper, C. Rückgewinnung von kupfer und kobalt aus schmelzflüssigen schlacken der ne-metallurgie. In Aschen, Schlacken, Stäube: Aus Abfallverbrennung und Metallurgie; Karl, J., Thomé-Kozmiensky, Eds.; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2013; pp. 599–614. [Google Scholar]
- Panda, S.; Mishra, S.; Rao, D.S.; Pradhan, N.; Mohapatra, U.; Angadi, S.; Mishra, B.K. Extraction of copper from copper slag: Mineralogical insights, physical beneficiation and bioleaching studies. Korean J. Chem. Eng. 2015, 32, 667–676. [Google Scholar] [CrossRef]
- Potysz, A.; van Hullebusch, E.D.; Kierczak, J.; Grybos, M.; Lens, P.N.L.; Guibaud, G. Copper Metallurgical Slags—Current Knowledge and Fate: A Review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 2424–2488. [Google Scholar] [CrossRef]
- Schlumberger, S.; Bühlrt, J. Metallrückgewinnung aus filterstäuben. In Aschen, Schlacken, Stäube: Aus Abfallverbrennung und Metallurgie; Thomé-Kozmiensky, Ed.; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2013; pp. 377–396. ISBN 978-3-935317-99-3. [Google Scholar]
- Brett, B.; Schrader, D.; Räuchle, K.; Heide, G.; Bertau, M. Wertstoffgewinnung aus Kraftwerksaschen Teil I: Charakterisierung von Braunkohlenkraftwerksaschen zur Gewinnung strategischer Metalle. Chem. Ing. Tech. 2015, 87, 1383–1391. [Google Scholar] [CrossRef]
- Martens, H.; Goldmann, D. Recyclingtechnik; Springer Fachmedien Wiesbaden: Wiesbaden, Germany, 2016; ISBN 978-3-658-02785-8. [Google Scholar]
- Huyen, P.T.; Dang, T.D.; Tung, M.T.; Huyen, N.T.T.; Green, T.A.; Roy, S. Electrochemical copper recovery from galvanic sludge. Hydrometallurgy 2016, 164, 295–303. [Google Scholar] [CrossRef]
- Pikhard, O.; Pretz, T. Schleifschlamm Quo vadis. Müll und ABfall 2009, 398–405. [Google Scholar] [CrossRef]
- Breitenstein, B. Verfahren zur Rückgewinnung von NE-Metallen aus feinkörnigen Rostaschen der thermischen Abfallbehandlung. Ph.D. Thesis, Technische Unversität Clausthal, Clausthal, Germany, 2017. [Google Scholar]
- Neuwahl, F.; Cusano, G.; Benavides, J.G.; Holbrook, S.; Roudler, S. Best Available Techniques (BAT) Reference Document for Waste Incineration. Available online: http://eippcb.jrc.ec.europa.eu/sites/default/files/2020-01/JRC118637_WI_Bref_2019_published_0.pdf (accessed on 6 February 2020).
- Siebzehnte Verordnung zur Durchführung des Bundes-Immissionsschutzgesetzes. 17. BImSchV. Available online: https://www.gesetze-im-internet.de/bimschv_17_2013/BJNR104400013.html (accessed on 2 May 2013).
- Syc, M.; Simon, F.-G.; Biganzolli, L.; Grosso, M.; Hyks, J. Recource Recorvery from Incineration Bottom Ash: Basics, Concepts, Principles. In Removal, Treatment and Utilisation of Waste Incineration Bottom Ash; Holm, O., Ed.; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2018; pp. 1–9. ISBN 978-3-944310-44-2. [Google Scholar]
- Bunge, R. Recovery of Metals from Waste Incinerator Bottom Ash. Available online: https://www.umtec.ch/fileadmin/user_upload/umtec.hsr.ch/Dokumente/Metals_from_MWIBA_6_2019.pdf (accessed on 6 February 2020).
- Alba, N.; Gassó, S.; Lacorte, T.; Baldasano, J.M. Characterization of Municipal Solid Waste Incineration Residues From Facilities with Different Air Pollution Control Systems. J. Air Waste Manag. Assoc. 1997, 47, 1170–1179. [Google Scholar] [CrossRef]
- Seifert, S.; Thome, V.; Karlstetter, C.; Maier, M. Elektrodynamische fragmentierung von mva-schlacken: Zerlegung der schlacken und abscheidung von chloriden und sulfaten. In Aschen, Schlacken, Stäube: Aus Abfallverbrennung und Metallurgie; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2013; pp. 353–365. ISBN 978-3-935317-99-3. [Google Scholar]
- Alwast, H.; Riemann, A. Verbesserung der Umweltrelevanten Qualitäten von Schlacken aus Abfallverbrennungsanlagen. Available online: http://www.uba.de/uba-info-medien/4025.html (accessed on 6 February 2020).
- Bayuseno, A.P.; Schmahl, W.W. Understanding the chemical and mineralogical properties of the inorganic portion of MSWI bottom ash. Waste Manag. 2010, 30, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Böni, D.; Morf, L. Thermo-Recycling: Efficient Recovery of Valuable Materials from Dry Bottom Ash. In Removal, Treatment and Utilisation of Waste Incineration Bottom Ash; Holm, O., Ed.; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2018; pp. 25–37. ISBN 978-3-944310-44-2. [Google Scholar]
- Stockinger, G. Direct Wet Treatment of Fresh, Wet-Discharged Grate Ash from a Waste Incineration Plant. In Removal, Treatment and Utilisation of Waste Incineration Bottom Ash; Holm, O., Ed.; Thomé-Kozmiensky Verlag GmbH: Neuruppin, Germany, 2018; pp. 47–52. ISBN 978-3-944310-44-2. [Google Scholar]
- Breitenstein, B.; Elwert, T.; Goldmann, D.; Haas, A.; Schirmer, T.; Vogt, V. Froth Flotation of Copper and Copper Compounds from Fine Fractions of Waste Incineration Bottom Ashes. Chem. Ing. Tech. 2017, 89, 97–107. [Google Scholar] [CrossRef]
- Wills, B.A.; Finch, J.A. Wills’ Mineral Processing Technology; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 9780080970530. [Google Scholar]
- Bulatovic, S.M. Flotation of industrial minerals. In Handbook of Flotation Reagents. Chemistry, Theory and Practice; Elsevier: Kidlington, UK, 2015; Volume 3, ISBN 978-0-444-53083-7. [Google Scholar]
- Bulatovic, S.M. Handbook of Flotation Reagents. Chemistry, Theory and Practice, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2010; ISBN 978-0-444-53082-0. [Google Scholar]
- Xiong, K.; Wen, S.M.; Zheng, G.S.; Bai, S.J.; Shen, H.Y. Flotation Research on Cuprite-Type Oxide Copper in XinJiang. AMR 2012, 524–527, 987–992. [Google Scholar] [CrossRef]
- Cytec Industries Inc. Mining Chemicals Handbook; Arnold, D., Ed.; CYTEC: Woodland Park, NJ, USA, 2010. [Google Scholar]
- Farrokhpay, S.; Filippov, L.; Fornasiero, D. Flotation of Fine Particles: A Review. Miner. Process. Extr. Metall. Rev. 2020, 31, 1–11. [Google Scholar] [CrossRef]






| Element | Unit | Content |
|---|---|---|
| Carbon | % DS | 18–40 |
| Oxygen | % DS | 15–22 |
| Sulfur | % DS | 0.1–0.5 |
| Lead | ppm DS | 100–2000 |
| Cadmium | ppm DS | 1–15 |
| Copper | ppm DS | 200–700 |
| Zinc | ppm DS | 400–1400 |
| Nickel | ppm DS | 30–50 |
| Carbon | % DS | 18–40 |
| Sample | Origin | Preparation |
|---|---|---|
| Direct unp | direct from the processing plant | Sample division and screening at 160 µm. |
| Stored unp | from the storage area | |
| Validation unp | direct from the processing plant | |
| Direct s | direct from the processing plant | Sample division and processing with the RENE-adapt process. |
| Direct mag | ||
| Direct nomag | ||
| Stored s | from the storage area | Sample division and processing with the RENE-adapt process. |
| Stored mag | ||
| Stored nomag |
| Element | Ag | Al | Cu | Fe | Ni | Pb | Sn | Zn | Ca | Si |
|---|---|---|---|---|---|---|---|---|---|---|
| Unit | ppm | % | ppm | % | ppm | ppm | ppm | ppm | % | % |
| Direct unp | n.a. | 3.58 | 3125 | 4.44 | 262 | 799 | 150 | 7788 | 14.25 | 8.64 |
| Stored unp | 573 | 4.03 | 2744 | 4.12 | 201 | 726 | n.a. | n.a. | 725.71 | n.a. |
| Validation unp | n.a. | n.a. | 2944 | 3.31 | n.a. | 1272 | n.a. | 9753 | 19.72 | 7.48 |
| Direct s | n.a. | 3.73 | 3510 | 5.94 | 579 | 877 | 165 | 6803 | 13.19 | 15.42 |
| Direct mag | n.a. | 3.99 | 2771 | 15.87 | 715 | 647 | 194 | 4214 | 12.52 | 14.86 |
| Direct nomag | n.a. | 3.79 | 2526 | 2.14 | 405 | 1249 | 195 | 4978 | 11.76 | 22.59 |
| Stored s | 22 | 4.23 | 2661 | 5.73 | 478 | 876 | n.a. | n.a. | 876.21 | n.a. |
| Stored mag | <5 | 4.01 | 2889 | 14.93 | 558 | 8823 | n.a. | n.a. | 8823.35 | n.a. |
| Stored nomag | 74 | 4.04 | 2604 | 2.37 | 331 | 4486 | n.a. | n.a. | 4486.14 | n.a. |
| Cu2O | CuO | (Co,Cu)2O | (Cu,Zn)O | (Mg,Fe,Zn)O | (Mn,Fe,Cr,Ni,Cu,Zn)2 (Al,Ti,Fe)O4 | CuS | (Cu,Fe)S | Glass | |
|---|---|---|---|---|---|---|---|---|---|
| Al | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 9.9 | 0.0 | 0.0 | 2.7 |
| Ca | 0.2 | 0.4 | 0.1 | 0.1 | 0.1 | 0.2 | 0.1 | 0.3 | 26.9 |
| Co | 0.0 | 0.0 | 71.9 | 0.0 | 0.1 | 0.0 | 0.0 | 0.1 | 0.0 |
| Cr | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 |
| Cu | 86.7 | 78.9 | 12.6 | 4.0 | 2.1 | 4.1 | 66.0 | 45.2 | 1.1 |
| Fe | 0.2 | 0.5 | 0.2 | 0.3 | 51.9 | 12.2 | 0.9 | 22.5 | 2.8 |
| K | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Mg | 0.0 | 0.0 | 0.1 | 0.0 | 4.0 | 0.2 | 0.1 | 0.0 | 5.6 |
| Mn | 0.0 | 0.0 | 0.0 | 0.0 | 1.1 | 2.6 | 0.0 | 0.0 | 0.2 |
| Na | 0.0 | 0.0 | 0.0 | 0.0 | 0.5 | 0.6 | 0.0 | 0.0 | 1.1 |
| Ni | 0.0 | 0.0 | 0.1 | 0.0 | 0.1 | 7.9 | 0.0 | 0.2 | 0.1 |
| P | 0.1 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| S | 0.3 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 | 31.0 | 31.2 | 0.0 |
| Si | 0.0 | 0.3 | 0.1 | 0.0 | 0.0 | 1.4 | 0.0 | 0.1 | 19.3 |
| Ti | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 3.9 | 0.0 | 0.0 | 0.1 |
| Zn | 0.1 | 0.0 | 0.0 | 75.6 | 16.7 | 26.1 | 0.0 | 0.2 | 0.3 |
| O * | 11.2 | 20.7 | 11.8 | 20.3 | 22.7 | 30.6 | 0.0 | 0.0 | 40.6 |
| Sum | 98.7 | 101.1 | 97.0 | 100.4 | 99.3 | 100.0 | 98.3 | 99.9 | 100.8 |
| Ferroalloy | Al-Ferroalloy | Zn-Al-Alloy | Si/Al-Si-Alloy | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Al | 0.0 | 0.0 | 0.0 | 0.0 | 36.2 | 50.0 | 64.4 | 22.5 | 65.6 | 0.4 | 72.0 | 76.4 |
| Ca | 0.2 | 0.1 | 0.1 | 0.3 | 0.1 | 0.1 | 0.0 | 0.2 | 0.2 | 0.1 | 0.1 | 0.2 |
| Co | 0.1 | 0.1 | 0.1 | 0.3 | 0.0 | 0.1 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 |
| Cr | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.3 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 |
| Cu | 23.1 | 4.7 | 1.8 | 1.6 | 36.9 | 14.4 | 3.7 | 1.7 | 1.9 | 27.4 | 3.7 | 1.3 |
| Fe | 40.9 | 41.9 | 58.7 | 60.9 | 17.9 | 27.0 | 22.6 | 64.5 | 0.1 | 0.9 | 2.9 | 0.2 |
| K | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Mg | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.5 | 1.5 | 0.1 | 0.2 |
| Mn | 0.0 | 1.5 | 0.1 | 0.4 | 0.2 | 0.0 | 0.1 | 0.4 | 0.0 | 0.0 | 0.2 | 0.0 |
| Na | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.7 | 0.1 | 0.0 | 0.1 |
| Ni | 0.5 | 0.1 | 0.1 | 0.3 | 0.5 | 0.8 | 0.1 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 |
| P | 0.0 | 0.8 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 |
| S | 34.3 | 0.0 | 36.3 | 37.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Si | 0.0 | 42.1 | 0.0 | 0.0 | 5.9 | 2.9 | 3.6 | 10.3 | 0.6 | 64.7 | 18.4 | 18.3 |
| Ti | 0.0 | 5.6 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.1 | 0.2 | 0.1 |
| Zn | 0.7 | 2.1 | 0.0 | 0.1 | 0.1 | 0.0 | 0.1 | 0.1 | 26.9 | 1.7 | 0.4 | 1.1 |
| Sum | 100.0 | 99.4 | 97.3 | 101.1 | 98.0 | 95.3 | 95.0 | 100.0 | 96.6 | 97.3 | 98.1 | 98.0 |
| Phase | Formula | Density in g⁄cm3 |
|---|---|---|
| Calcite | CaCO3 | 1.7102 |
| Ettringite | Ca6Al2[(OH)12|(SO4)3]∙26H2O | 1.77 |
| Gypsum | CaSO4∙2H2O | 2.322 |
| Glass | ~2.5 | |
| Quartz | SiO2 | 2.66 |
| Anhydrite | CaSO4 | 2.98 |
| Tenorite | CuO | 6.45 |
| Covellite | CuS | 4.6 |
| Cuprite | Cu2O | 6.14 |
| Copper | Cu | 8.92 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keber, S.; Schirmer, T.; Elwert, T.; Goldmann, D. Characterization of Fine Fractions from the Processing of Municipal Solid Waste Incinerator Bottom Ashes for the Potential Recovery of Valuable Metals. Minerals 2020, 10, 838. https://doi.org/10.3390/min10100838
Keber S, Schirmer T, Elwert T, Goldmann D. Characterization of Fine Fractions from the Processing of Municipal Solid Waste Incinerator Bottom Ashes for the Potential Recovery of Valuable Metals. Minerals. 2020; 10(10):838. https://doi.org/10.3390/min10100838
Chicago/Turabian StyleKeber, Sebastian, Thomas Schirmer, Tobias Elwert, and Daniel Goldmann. 2020. "Characterization of Fine Fractions from the Processing of Municipal Solid Waste Incinerator Bottom Ashes for the Potential Recovery of Valuable Metals" Minerals 10, no. 10: 838. https://doi.org/10.3390/min10100838
APA StyleKeber, S., Schirmer, T., Elwert, T., & Goldmann, D. (2020). Characterization of Fine Fractions from the Processing of Municipal Solid Waste Incinerator Bottom Ashes for the Potential Recovery of Valuable Metals. Minerals, 10(10), 838. https://doi.org/10.3390/min10100838





