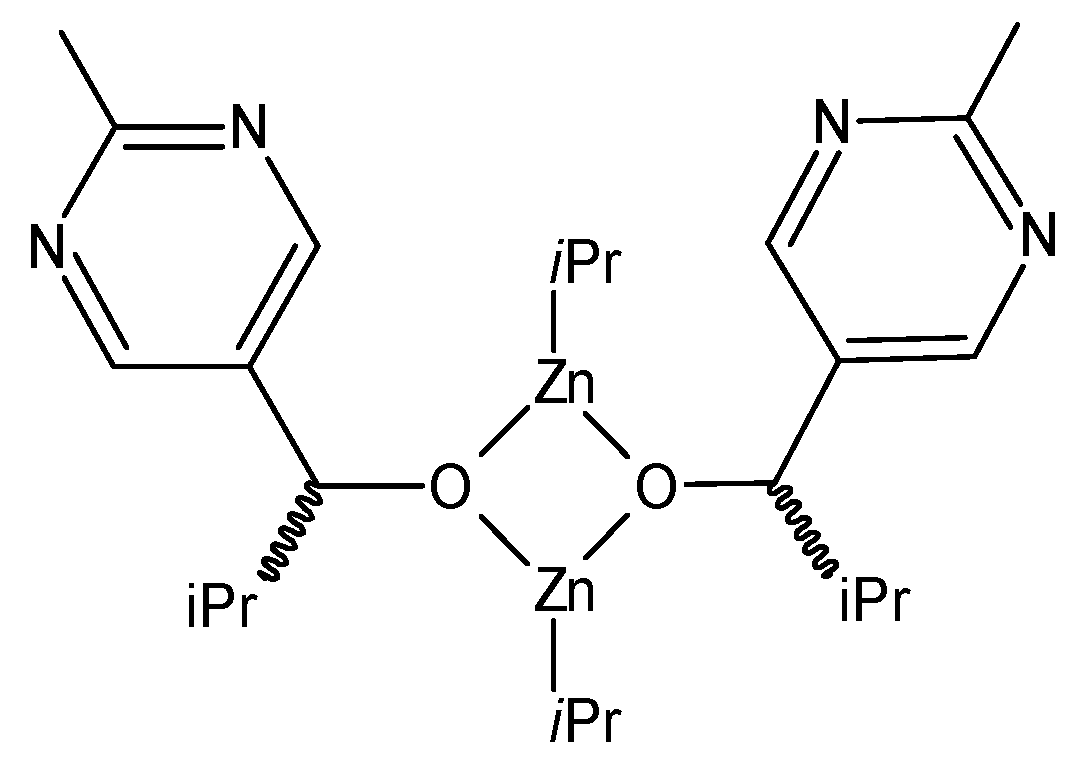
The reagents of the most efficient variant [
17] of the Soai reaction include an
N-heterocyclic aldehyde, 4-[2-(
tert.-butyl)ethynyl]pyrimidylaldehyde (
1), di(
iso-propyl)zinc (
2) and the autocatalyst, which is the alkylation product of
1 by
2, leading to the secondary alcohol (
3), as shown in
Figure 1 [
41,
42]. These molecules are formed from H, C, N, O and Zn, which are present in nature as mixtures of their stable isotopes; the H, C, N, and O with dominance of the lighter nuclei, while Zn as a more equable mixture of five isotopes [
12] (
Table S1). As can be seen from the structures in
Figure 1, the “isotopically prochiral” groups in compounds
1–
3 are the
tert.-Bu group in
1 and
3, as well as the
iso-Pr group in compounds
2 and
3. These groups consist of H and C, thus the isotopic substitution by these elements will be analyzed here. N is not included in our analysis, even if a very recent study has shown that chirality caused (only) by N-isotopes can control the enantiomeric outcome of the Soai reaction [
43]. Some fragments of hypothetic intermediates [
44,
45,
46] of the Soai autocatalysis show chiral arrangement of the Zn atoms. One of these fragments, the structure of which has been essentially supported by X-ray crystallography [
47], will also be included in the present study.
2.1. The Tert.-Butyl Group
The
tertiary-butyl group in compounds
1 and
3 is a highly symmetric species. Therefore, it needs at least two different substituents on at least two of its methyl groups for the generation of a center of chirality on the central C atom. Because of the presence of three identic (methyl) groups in this functionality, the number of possible isotopically substituted derivatives amounts to 112 (
Supporting Materials 1) if the isotopic substitution in the central carbon atom is neglected, and 224 if this is taken into account. Because of the necessity of (at least)
double isotopic substitution for obtaining a chiral isotopomer, the
probabilities of the formation of such structures with natural abundance
2H and/or
13C substituents is very low. Only four combinations (from 112) have probability >10
−8. The sum of the probabilities is 1.027 × 10
−5, deriving mostly from species with one
13C substitution.
We calculated the expectable
enantiomeric excesses (
e.e.50%, %) with the Pars–Mills equation [
40,
48] (with 50% confidence, see also
Supporting Materials 2) for sample sizes that could appear in usual micropreparative work (ranging from millimol to femtomol) (
Table 1).
Table 1.
Expectable enantiomeric excesses in the t-Bu group.
Table 1.
Expectable enantiomeric excesses in the t-Bu group.
| Sample Size | e.e.50% |
|---|
| millimol | 8.60 × 10−7 |
| micromole | 2.72 × 10−5 |
| nanomol | 8.60 × 10−4 |
| picomol | 2.72 × 10−2 |
| femtomol | 8.60 × 10−1 |
These data show that even if the expectable
e.e. values are very low, according to experimental evidence [
16,
17,
18,
19,
20] and theoretical considerations [
21,
22,
23,
24], the isotope chirality in the
tert.-butyl group in compounds
1 and/or
3 could influence the outcome of the most sensitive variant of the Soai reaction, however, taking into regard that it is separated from the pyrimidyl unit by the rigid C
2 moiety, and thus from the decisive molecular events around the new stereocenter, this option appears as scarcely probable, but cannot be excluded.
2.2. The Iso-Propyl Group(s)
The
i-Pr groups appear in two of the reactants:
2 and
3. There is, however, both quantitative and qualitative differences between these structures. Compound
2 contains
two i-Pr groups, in organometallic bond, while autocatalyst
3 contains only
one i-Pr moiety, linked by covalent C(sp
3)–C(sp
3) bond to the newly formed center of chirality. This is a fundamental difference, especially from the viewpoint that in
2 the
i-Pr groups are present
before the C–C bond making it an alkylation step, while the
i-Pr group in
3 represent a
final stage; that is, the stage when the fate of the asymmetry of the critical carbon atom has already been decided. On the other hand, compound
3 appears as autocatalyst in the subsequent cycle(s) of the reaction in intermediate(s), the structure of which is actually known only from theoretical studies [
44,
45,
46,
47,
49]
The analysis of the eventual role of isotopically chiral i-Pr group(s) will be deduced step-by-step starting with the methyl group.
For commodity, let us define the probabilities (P) on the basis of the natural abundances [
12] as follows: P(
12C) =
p = 0.98889, P(
13C) =
q = 0.01111, P(
1H) =
u = 0.999844 and P(
2D) =
v = 0.000156. Thus
| p1 = P(12C1H3) = pu3 | p5 = P(13C1H3) = qu3 |
| p2 = P(12C1H22D) = 3pu2v | p6 = P(13C1H22D) = 3qu2v |
| p3 = P(12C1H2D2) = 3puv2 | p7 = P(13C1H2D2) = 3quv2 |
| p4 = P(12C2D3) = pv3 | p8 = P(13C2D3) = qv3 |
amounting to eight cases, with ∑p
i = 1 (
i = 1–8). These cases, obviously, provide no chirality.
The iso-propyl group containing two methyl groups, gives 8 × 8 = 64 cases if the possible carbon or hydrogen isotope substitution in the central CH group is disregarded; if only the 12C/13C or 1H/2D exchange is taken into account, the number of cases is 2 × 64 = 128, while both of these exchanges yield 2 × 128 = 256 cases. We shall calculate below on the basis of 128 cases.
The probabilities of these cases are the products of the individual probabilities:
p × pi × pj and q × pi × pj, where i = 1,2,…,8 and j = 1,2,…,8. If i = j, the i-Pr group becomes symmetric, which are 16 cases from 128, leaving 112 as asymmetric cases. From these, obviously, 56 are of R and 56 of S configuration. The sum of the probabilities of the symmetric (achiral) cases is ∑(p × pi2 + q × pi2) = 0.977131 (since i = j in the symmetric cases).
The probability of the remaining 112 chiral cases is Pchiral = 1 – 0.977131 = 0.022869. The abundance of the symmetric species with central 12C is thus 0.98889 × 0.977131 = 0.96627566, while with central 13C it is 0.01111 × 0.977131 = 0.01085593 and the sum of these (as it should be) equals 0.977131.
Consequently, the sum of the abundances of the 56S and 56R species is 0.022869 ≈ 2.3%.
The actual alkylation reagent, 2, of the Soai reaction, however, contains two iso-propyl groups. This generates 128 × 128 = 16,384 cases if only the 12C/13C exchange possibility on the central (CH) carbon is considered; if the 1H/2D exchange is also taken into account, this increases the number of cases to 4 × 128 × 128 = 65,536. We shall use the former number (128) in the following calculations.
We can approach the problem from two viewpoints:
- (i)
Each (of the two) i-Pr groups can be symmetric “internally”. In this case, each i-Pr group provides 16 (8 + 8) symmetric cases. Altogether, these are 162 = 256 cases. In the calculations with only one i-Pr group, we have seen that this leads to ∑(p × pi2 + q × pi2) = 0.977131 probability. The probability of that both i-Pr groups are of “internal” symmetry is the square of this value, 0.9771312 = 0.954785. This is then the probability of the achiral cases. Consequently, the probability of the chiral cases, which are 16,384 – 256 = 16,128 structures, is Pchiral = 1 – 0.954785 = 0.045215 ≈ 4.5%.
- (ii)
If the symmetry between the two i-Pr groups is considered, this requires a “corresponding pair” to each of the configurations in one of the groups: 64 cases to each of the 64 combinations in one i-Pr, that is 642 = 4096, with “unique” CH group, or 1282 = 16,384 with one isotope exchange in the methylene group, or 2562 = 65,536, if both C and H exchange is considered. In more explicit terms, Pachiral = ∑[(p × pi × pj)2 + (q × pi × pj)2] = p2∑(pi × pj)2 + q2∑(pi × pj)2 = (p2 + q2) ∑(pi × pj)2, which is numerically for the above mentioned (one isotope exchange in the CH group) case: 0.978 × 0.9771312 = 0.93380 ≈ 93.4%.
Consequently, while 128 cases are achiral, and 16,384 – 128 = 16,256 structures are chiral, but the overwhelming majority of abundance is on the side of the achiral structures.
The probability of the chiral cases is Pchiral = 1 – 0.93380 = 0.06620 ≈ 6.6%.
Table 2.
Expectable enantiomeric excesses for cases (i) and (ii).
Table 2.
Expectable enantiomeric excesses for cases (i) and (ii).
| Case (i) | Case (ii) |
|---|
| p = 0.045215 | p = 0.06662 |
| Sample size | e.e.50% | Sample size | e.e.50% |
| millimol | 1.29595 × 10−8 | millimol | 1.07 × 10−8 |
| micromole | 4.09814 × 10−7 | micromole | 3.39 × 10−7 |
| nanomol | 1.29595 × 10−5 | nanomol | 1.07 × 10−5 |
| picomol | 4.09814 × 10−4 | picomol | 3.39 × 10−4 |
| femtomol | 1.29595 × 10−2 | femtomol | 1.07 × 10−2 |
All supposed
intermediates of the Soai reaction, however, contain two kinds (both C-bound and Zn-bound)
i-Pr groups and more of these, than in the reagents shown in
Figure 1. We analyzed a relatively simple one, compound No. 4, according to the numbering in reference [
44] (
S-
E4), shown in
Figure 2. We considered that variant of the
i-Pr groups, where only
1H/
2D or
12C/
13C exchange occurs in the central CH groups. In this case, the number of all possibilities is 128
4 = 268,435,456. Several of these structures are “degenerated” (equivalent to each other).
The number of the
chiral structures deriving from
S-E4 can be deduced as follows:
- (a)
Chirality caused by the Zn-bound i-Pr groups, and only by these. The number of the Zn-bound cases is 1282, subtracting the number of the symmetric cases and multiplying the rest with the number of the symmetric cases of the C-bound i-Pr groups we obtain: (1282 – 162) × 128 = 2,064,384. The probability of the symmetric (achiral) cases is the same, which was calculated above for case (i) of the instance where only the internal symmetry of two i-Pr groups was considered: 0.9771312 = 0.954785 ≈ 95.5%, while for the chiral cases (1 − 0.954785) = 0.045215 ≈ 4.5%.
- (b)
Chirality is caused by C-bound i-Pr groups, and only by these. In this case, each of the C-bound 128 i-Pr groups can be paired with 127 different configurations, choosing achiral Zn-bond partners we obtain 128 × 127 × 162 = 4,161,536 chiral cases. The probability of the C-bound achiral cases according to this approach is: 0.93380 ≈ 93.4%, and of the chiral cases (1 − 0.93380) = 0.06620 ≈ 6.6%.
- (c)
If the combination of both the chirality of the Zn-bound and the C-bound structures (i-Pr groups) is considered, the number of the chiral cases amounts: (1282 − 162) × 128 × 127 = 262,176,768. The probability of this case equals 0.00300 because the probability of chirality in case (a) is 0.045215 and in case (b) is 0.06620, thus the probability of joint event equals 0.045215 × 0.06620 = 0.00300.
Hence the probability of the chiral cases all together is P∑ = 04222 + 0.06320 + 0.00300 = 0.10842 ≈ 10.8%.
We can get the same result if we consider that the probability of the achiral structures in this set-up is the product of the achiral probabilities in cases (a) and (b): 0.954785 × 0.93380 = 0.891572 ≈ 89.2%, while for the probability of the chiral structures we obtain P∑ = (1 − 0.891572) = 0.108428 ≈ 10.8%.
- (d)
The number of the achiral cases is 128 for the C-bound structures and 16 × 16 = 256 for the Zn-bound ones. Thus the total number of the achiral cases is 128 × 256 = 32,768.
The sum of the number of chiral structures, from cases (a), (b) and (c) equals 268,402,688. This number plus the number of the achiral structures from case (d) gives exactly 268,435,456, as calculated from pure combinatorial considerations.
Figure 2.
Schematic structure of the dimeric intermediate of the Soai reaction, according to Schiaffino and Ercolani [
44] (
S-E4).
Figure 2.
Schematic structure of the dimeric intermediate of the Soai reaction, according to Schiaffino and Ercolani [
44] (
S-E4).
It should be noted that this number of combinations corresponds approximately to the number of molecules in an ~0.45 femtomol size sample, approaching to a situation, where only for statistical reasons not all of the possibilities can be “materialized”, similarly to the case described by Eschenmoser for “statistical origin” of DNA [
50].
It is remarkable that the huge numbers of possible chiral combinations generate only a fairly low percentage of chiral components (corresponding to the low probability values), while the large majority of components are of achiral structure. This, obviously, is due to the asymmetric natural abundance of the isotopes of H and C. However, even so, the percentages of expected enantiomeric excesses are much higher than the experimentally documented sensitivity threshold of the Soai reaction [
16,
17,
18,
19,
20,
37,
38,
39,
41,
42].
These results can be controlled by calculations on a somewhat different way, as shown in
Supporting Materials 3. The result is exactly the same.
We also calculated for compound
S-E4, with the Pars–Mills equation (with 50% confidence, see also
Supporting Materials 2) the expectable
e.e.50% values from sample sizes ranging from millimol to femtomol, for the chiral fraction of the molecules (
Table 3):
Table 3.
Expectable enantiomeric excesses for S-E4.
Table 3.
Expectable enantiomeric excesses for S-E4.
| Sample Size | e.e.50% (%) |
|---|
| millimol | 8.36900 × 10−9 |
| micromole | 2.64651 × 10−7 |
| nanomol | 8.36900 × 10−6 |
| picomol | 2.64651 × 10−4 |
| femtomol | 8.36900 × 10−3 |
It should be mentioned that the symmetries of compound S-E4 are valid only if the isotopic composition of the central O2Zn2 ring is disregarded. At the present level of our calculations we did so.
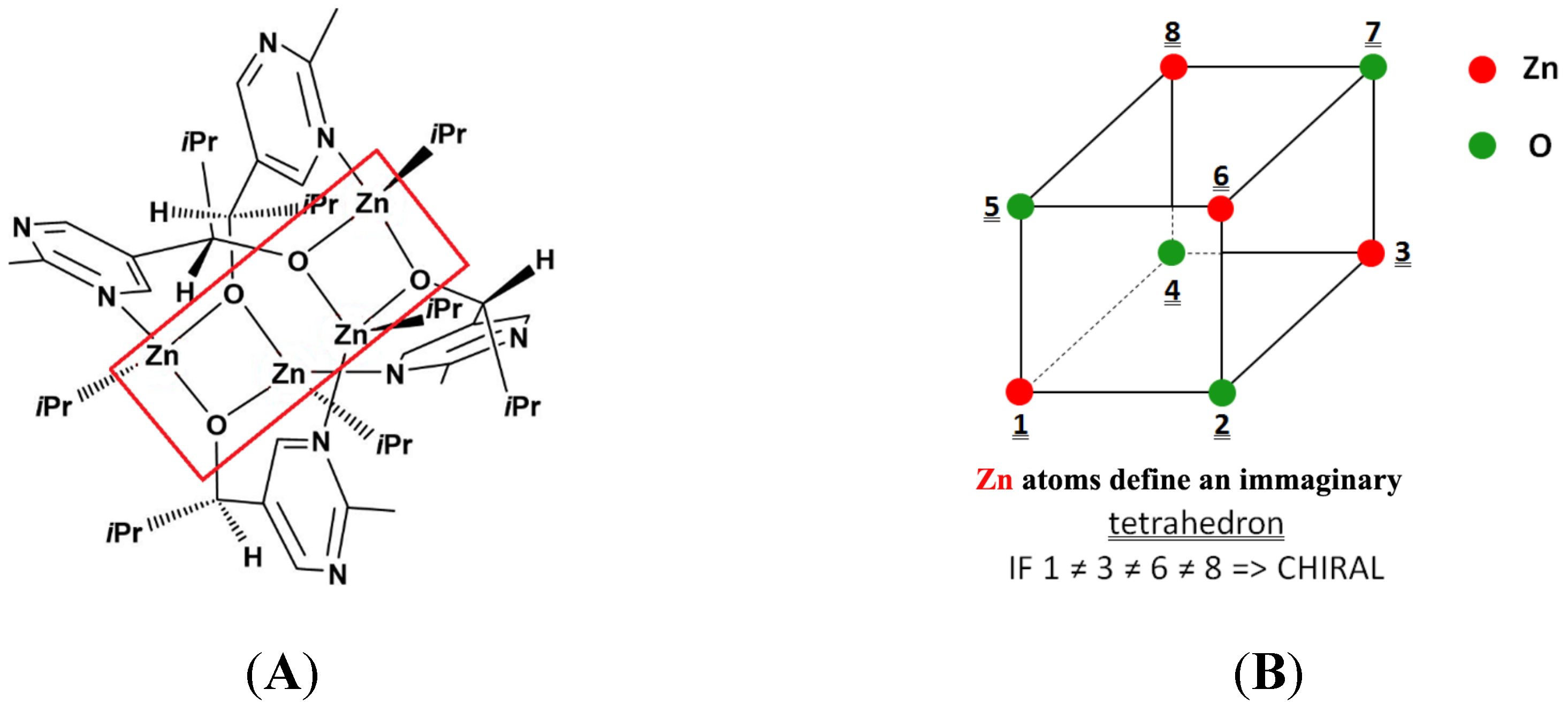
2.3. Zinc Isotopic Chirality?
Zinc is present in nature as a mixture of five stable isotopes, which are distributed much more evenly than those of H or C (see
Table S1). It appeared therefore challenging to identify (eventual) structural features in the polymetallic intermediates
hypothesized for the Soai reaction [
44,
45,
46] where the chirality is caused by spatial distribution of Zn isotopes. The most striking structural element of this kind was found in the number 9 intermediate suggested by Schiaffino and Ercolani [
44] (
S-E9), as shown in
Figure 3A. In this substructure, four Zn atoms and four O atoms form a virtual cube-like polyhedron, as shown in
Figure 3B. Interestingly this
hypothetical structural element also appears in the X-ray structure of the “enantiomeric” model compound of the Soai autocatalysis, reported very recently by Matsumoto
et al. [
47].
Figure 3.
(
A) Schematic structure of a tetrameric intermediate of the Soai reaction according to Schiaffino and Ercolani [
44] (
S-E9) and (
B) schematic structure of the O
4Zn
4 “twisted cube” fragment in compound
S-E9.
Figure 3.
(
A) Schematic structure of a tetrameric intermediate of the Soai reaction according to Schiaffino and Ercolani [
44] (
S-E9) and (
B) schematic structure of the O
4Zn
4 “twisted cube” fragment in compound
S-E9.
The possibilities of developing chiral structure(s) in the O4Zn4 “cube” of compound S-E9 can be deduced as follows.
Simple case (all O atoms are regarded equal, or isotopic differences in these O atoms are disregarded). This approach enables calculating the contribution of the metal atoms to chirality.
In this case, chirality develops if all four Zn atoms are different isotopes, which are thus on the points of an imaginary tetrahedron centered in the center of the “cube”. Let us denote 64Zn as a, 65Zn as b, 66Zn as c, 67Zn as d, and 68Zn or 70Zn as e (we shall later use similar notation for oxygen isotopes: 16O u, 17O v, and 18O w).
Under these conditions, 10 chiral cases can evolve: abcd two times (since one point is a and the other three can be bcd or bdc), similarly abce, abde, acde or bcde all two times. Taking into regard the natural abundances, one obtains for the sum of the probabilities of all chiral cases P∑ = 0.002530375 ≈ 0.25%.
Utilizing the Pars–Mills equation [
40,
48] for calculation of the expected
enantiomeric excesses (with 50% confidence, see also
Supporting Materials 2), we obtain (
Table 4).
Table 4.
Expectable enantiomeric excesses in the O4Zn4 “cube” (simple case).
Table 4.
Expectable enantiomeric excesses in the O4Zn4 “cube” (simple case).
| Sample Size | e.e.50% (%) |
|---|
| millimol | 5.47817 × 10−8 |
| micromole | 1.73235 × 10−6 |
| nanomol | 5.47817 × 10−5 |
| picomol | 1.73235 × 10−3 |
| femtomol | 5.47817 × 10−2 |
Complex case (isotopes of the oxygen atoms are considered). This situation can be approached by the following partial calculations:
- (i)
The “cube” contains four different Zn isotopes, no matter which oxygen isotopes are present. This is equal to the “simple” case. The “cube” is chiral, 10 structures can be identified, the sum of the probabilities is P(i) = 0.002530375 = 0.2530375 ≈ 0.25%.
- (ii)
Three different Zn isotopes in the “cube”; that is to say, within the four Zn atoms, two are of the same isotope. The two equal isotopes are placed in any case on one of the diagonals of one of the faces. Let us turn the cube in such position that the two equal isotopes occupy positions 1 and 3 (
Figure 3b). Taking into regard that Zn has five isotopes, this results in 60 cases. If only the Zn atoms are regarded, this situation results in a symmetry plane in plane [2-6-8-4]. The “cube” becomes chiral if this symmetry is destroyed by putting different O isotopes to positions 5 and 7. Let us now consider the possibilities given by the three different O isotopes. This results in 54 additional cases coupled with each of the 60 cases derived from combinations of the Zn isotopes. Thus. the total number of chiral structures is 60 × 54 = 3240. The highest probability is provided by the combination (
aabc +
uuuw). This taken two times gives P
(ii) = 1.10662 × 10
−5 ≈ 0.0011%.
- (iii)
Two different Zn isotopes, pairwise, that is 2 + 2 equal Zn isotopes, aabb and so on. This gives 10 cases, considering the three O isotopes, one obtains 36 additional possibilities for each of the Zn-derived 10, thus the total number of combinations is 10 × 36 = 360. The most probable case is the combination (aabb + uuww) two times, which gives P(iii) = 1.53122 × 10−7 ≈ 0.000015%.
- (iv)
Two different Zn isotopes in the manner that three Zn isotopes are equal, that is the structure is of aaab type, which gives 20 cases, which is of relatively high symmetry; this can be spoiled in 54 ways for each structure, giving a total of 20 × 54 = 1080 cases. The highest probability is at the (aaab + uuuw) combination, taken two times P(iv) = 1.31637 × 10−4 ≈ 0.013%.
- (V)
If all Zn atoms are of the same isotope, a “fourth” O isotope would be needed to obtain chiral structure, which is impossible, providing, consequently 0 cases and 0 probability.
The total number of chiral structures is 4690. The sum of the probabilities of these structures is P∑(i)–(iv) = 0.002672827 ≈ 0.27%.
It should be noted, that the majority (0.25% from 0.27%, that is 92.6%!) of the amount (probability) of these 4690 chiral structures comes from the variations in the distribution of the metal isotopes (“simple” case, or case (i), 10 structures) obviously because of the more even distribution of the natural abundances of these isotopes.
We calculated with the Pars–Mills equation [
40,
48] (see also
Supporting Materials 2) the expectable
enantiomeric excesses for reasonable microchemical sample sizes (
Table 5).
Table 5.
Expectable enantiomeric excesses in the O4Zn4 “cube” (complex case).
Table 5.
Expectable enantiomeric excesses in the O4Zn4 “cube” (complex case).
| Sample Size | e.e.50% (%) |
|---|
| millimol | 5.33019 × 10−8 |
| micromole | 1.68555 × 10−6 |
| nanomol | 5.33019 × 10−5 |
| picomol | 1.68555 × 10−3 |
| femtomol | 5.33019 × 10−2 |
Similar to the probability dates, the expectable e.e. levels also show only a slight difference between the “simple” and the “complex” treatment.
It is an interesting feature of the mechanistic picture elaborated by Schiaffino and Ercolani [
44] for the Soai reaction that some (3 of 7) of the intermediates suggested by these authors contain Zn atoms with four different substituents of deformed tetrahedral geometry. This means that these Zn atoms are
chiral. To the best of our knowledge this aspect of the Schiaffino–Ercolani mechanism has not yet been discussed . We shall come back to this point in a forthcoming publication. The last two intermediates of this mechanism (2 of 7, one of these is the compound
S-E9 discussed above, see also
Supporting Materials 5), contain such Zn atoms, which are linked to two different ligands and to two oxygen atoms of equal “chemical environment”. These Zn atoms become chiral only if the two O atoms are different isotopes. Since all four Zn atoms in the intermediate
S-E9 are of this kind, we analyzed this aspect too.
The probability of the evolution of a stereocenter on one of the Zn atoms can easily be calculated on the basis of the natural abundances of O (
Table S1). The probability of achiral combinations is P
achiral =
0.995190 ≈ 99.5%, while that of the chiral ones is P
chiral = 1 – P
achiral =
0.0048099 ≈ 0.5%. The probability that one of the four zinc atoms in the imaginary “cube” becomes chiral is: P
”cube” = 4 × P
chiral =
0.01924 ≈ 2%. The probability that all four Zn atoms in the “cube” become chiral is, however, very low: 1.37 × 10
−4%. Whether mutual induction effects are influencing the configuration of these Zn atoms is not yet (?) clear.
The Pars–Mills [
40,
48] (see also
Supporting Materials 2) equation for these probabilities, in reasonable microchemical sample size ranges, gives the following
expectable enantiomeric excesses (
Table 6).
Table 6.
Expectable enantiomeric excesses deriving from Zn atoms, chiral by O isotopes.
Table 6.
Expectable enantiomeric excesses deriving from Zn atoms, chiral by O isotopes.
| Sample Size | e.e.50% (%, one Zn) | e.e.50 % (%, four Zn) |
|---|
| millimol | 3.97338 × 10−8 | 1.98669 × 10−8 |
| micromole | 1.25649 × 10−6 | 6.28247 × 10−7 |
| nanomol | 3.97338 × 10−5 | 1.98669 × 10−5 |
| picomol | 1.25649 × 10−3 | 6.28247 × 10−4 |
| femtomol | 3.97338 × 10−2 | 1.98669 × 10−2 |