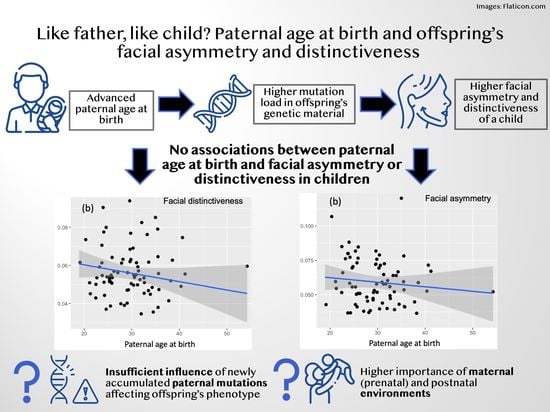
Like Father, Like Child? Paternal Age at Birth and Offspring’s Facial Asymmetry and Distinctiveness
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Group
2.2. Geometric Morphometrics
2.3. Statistical Analysis
3. Results
3.1. Facial Distinctiveness
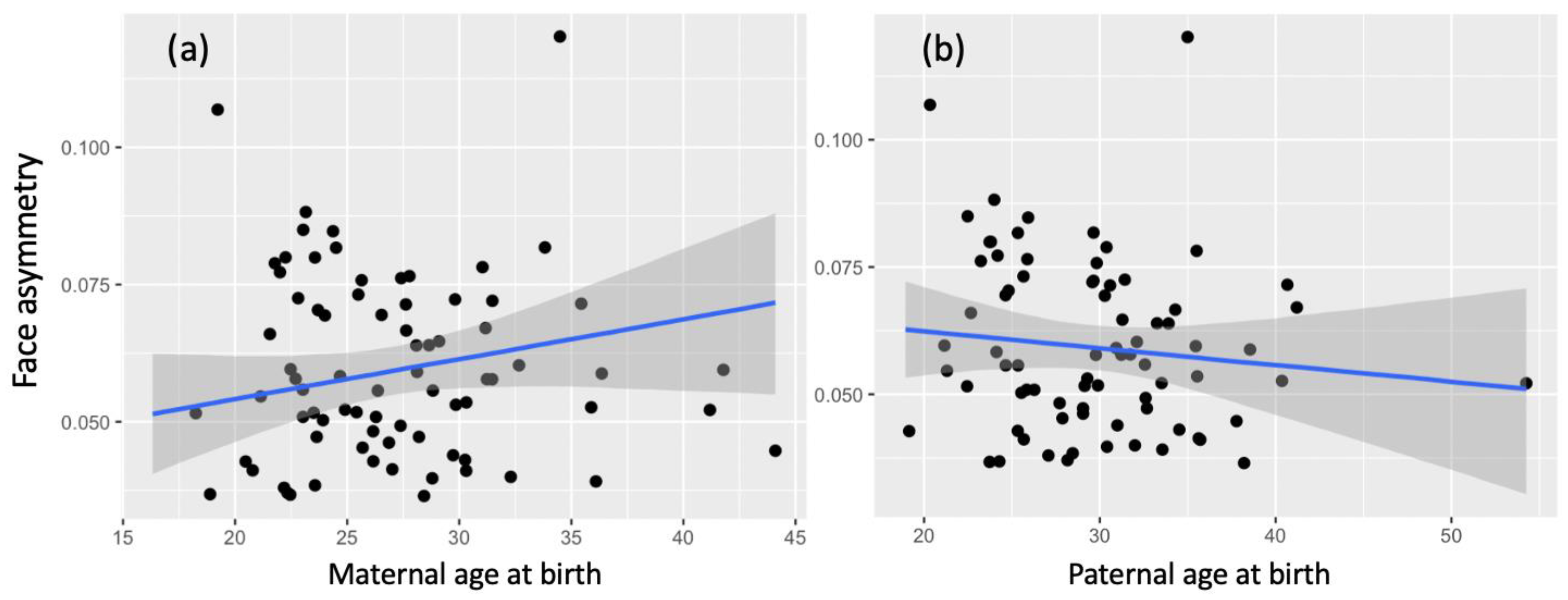
3.2. Facial Asymmetry
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharp, G.C.; Schellhas, L.; Richardson, S.S.; Lawlor, D.A. Time to cut the cord: Recognizing and addressing the imbalance of DOHaD research towards the study of maternal pregnancy exposures. J. Dev. Orig. Health Dis. 2019, 10, 509–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sartorius, G.A.; Nieschlag, E. Paternal age and reproduction. Hum. Reprod. Update 2010, 16, 65–79. [Google Scholar] [CrossRef] [Green Version]
- Pang, T.Y.; Short, A.K.; Bredy, T.W.; Hannan, A.J. Transgenerational paternal transmission of acquired traits: Stress-induced modification of the sperm regulatory transcriptome and offspring phenotypes. Curr. Opin. Behav. Sci. 2017, 14, 140–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eberle, C.; Kirchner, M.F.; Herden, R.; Stichling, S. Paternal metabolic and cardiovascular programming of their offspring: A systematic scoping review. PLoS ONE 2020, 15, e0244826. [Google Scholar] [CrossRef]
- Braun, J.M.; Messerlian, C.; Hauser, R. Fathers matter: Why it’s time to consider the impact of paternal environmental exposures on children’s health. Curr. Epidemiol. Rep. 2017, 4, 46–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soubry, A. POHaD: Why we should study future fathers. Environ. Epigenetics 2018, 4, dvy007. [Google Scholar] [CrossRef] [PubMed]
- Kong, A.; Frigge, M.L.; Masson, G.; Besenbacher, S.; Sulem, P.; Magnusson, G.; Gudjonsson, S.A.; Sigurdsson, A.; Jonasdottir, A.; Jonasdottir, A. Rate of de novo mutations and the importance of father’s age to disease risk. Nature 2012, 488, 471–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crow, J.F. The origins, patterns and implications of human spontaneous mutation. Nat. Rev. Genet. 2000, 1, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Janecka, M.; Mill, J.; Basson, M.; Goriely, A.; Spiers, H.; Reichenberg, A.; Schalkwyk, L.; Fernandes, C. Advanced paternal age effects in neurodevelopmental disorders—Review of potential underlying mechanisms. Transl. Psychiatry 2017, 7, e1019. [Google Scholar] [CrossRef] [Green Version]
- Curley, J.P.; Mashoodh, R.; Champagne, F.A. Epigenetics and the origins of paternal effects. Horm. Behav. 2011, 59, 306–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simard, M.; Laprise, C.; Girard, S.L. Impact of paternal age at conception on human health. Clin. Chem. 2019, 65, 146–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fieder, M.; Huber, S. Paternal age predicts offspring chances of marriage and reproduction. Am. J. Hum. Biol. 2015, 27, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Arslan, R.C.; Willführ, K.P.; Frans, E.M.; Verweij, K.J.; Bürkner, P.-C.; Myrskylä, M.; Voland, E.; Almqvist, C.; Zietsch, B.P.; Penke, L. Older fathers’ children have lower evolutionary fitness across four centuries and in four populations. Proc. R. Soc. B Biol. Sci. 2017, 284, 20171562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eisenberg, D.T.; Kuzawa, C.W. The paternal age at conception effect on offspring telomere length: Mechanistic, comparative and adaptive perspectives. Philos. Trans. R. Soc. B 2018, 373, 20160442. [Google Scholar] [CrossRef] [Green Version]
- Gangestad, S.W.; Thornhill, R. Facial masculinity and fluctuating asymmetry. Evol. Hum. Behav. 2003, 24, 231–241. [Google Scholar] [CrossRef]
- Huber, S.; Fieder, M. Advanced paternal age is associated with lower facial attractiveness. Evol. Hum. Behav. 2014, 35, 298–301. [Google Scholar] [CrossRef]
- Woodley of Menie, M.A.; Kanazawa, S. Paternal age negatively predicts offspring physical attractiveness in two, large, nationally representative datasets. Personal. Individ. Differ. 2017, 106, 217–221. [Google Scholar] [CrossRef]
- Lee, A.J.; Mitchem, D.G.; Wright, M.J.; Martin, N.G.; Keller, M.C.; Zietsch, B.P. Facial averageness and genetic quality: Testing heritability, genetic correlation with attractiveness, and the paternal age effect. Evol. Hum. Behav. 2016, 37, 61–66. [Google Scholar] [CrossRef] [Green Version]
- Grammer, K.; Thornhill, R. Human (Homo sapiens) facial attractiveness and sexual selection: The role of symmetry and averageness. J. Comp. Psychol. 1994, 108, 233. [Google Scholar] [CrossRef]
- Rhodes, G.; Yoshikawa, S.; Clark, A.; Lee, K.; McKay, R.; Akamatsu, S. Attractiveness of facial averageness and symmetry in non-Western cultures: In search of biologically based standards of beauty. Perception 2001, 30, 611–625. [Google Scholar] [CrossRef]
- Apicella, C.L.; Little, A.C.; Marlowe, F.W. Facial averageness and attractiveness in an isolated population of hunter-gatherers. Perception 2007, 36, 1813–1820. [Google Scholar] [CrossRef]
- Valentine, T.; Darling, S.; Donnelly, M. Why are average faces attractive? The effect of view and averageness on the attractiveness of female faces. Psychon. Bull. Rev. 2004, 11, 482–487. [Google Scholar] [CrossRef] [Green Version]
- Danel, D.; Dziedzic-Danel, A.; Kleisner, K. Does age difference really matter? Facial markers of biological quality and age difference between husband and wife. Homo 2016, 67, 337–347. [Google Scholar] [CrossRef]
- Rohlf, F.J. The tps series of software. Hystrix 2015, 26, 9–12. [Google Scholar] [CrossRef]
- Kleisner, K.; Pokorný, Š.; Saribay, S.A. Toward a new approach to cross-cultural distinctiveness and typicality of human faces: The cross-group typicality/distinctiveness metric. Front. Psychol. 2019, 10, 124. [Google Scholar] [CrossRef] [Green Version]
- Adams, D.C.; Otárola-Castillo, E. geomorph: An R package for the collection and analysis of geometric morphometric shape data. Methods Ecol. Evol. 2013, 4, 393–399. [Google Scholar] [CrossRef]
- Bookstein, F.L. Landmark methods for forms without landmarks: Morphometrics of group differences in outline shape. Med. Image Anal. 1997, 1, 225–243. [Google Scholar] [CrossRef]
- Bürkner, P.-C. Advanced Bayesian multilevel modeling with the R package brms. arXiv 2018, arXiv:1705.11123. [Google Scholar] [CrossRef]
- Barr, D.J. Random effects structure for testing interactions in linear mixed-effects models. Front. Psychol. 2013, 4, 328. [Google Scholar] [CrossRef] [Green Version]
- Lemoine, N.P. Moving beyond noninformative priors: Why and how to choose weakly informative priors in Bayesian analyses. Oikos 2019, 128, 912–928. [Google Scholar] [CrossRef] [Green Version]
- Girard-Buttoz, C.; Tkaczynski, P.J.; Samuni, L.; Fedurek, P.; Gomes, C.; Löhrich, T.; Manin, V.; Preis, A.; Valé, P.F.; Deschner, T. Early maternal loss leads to short-but not long-term effects on diurnal cortisol slopes in wild chimpanzees. eLife 2021, 10, e64134. [Google Scholar] [CrossRef]
- Gerhart, J.; Kirschner, M. The theory of facilitated variation. Proc. Natl. Acad. Sci. USA 2007, 104, 8582–8589. [Google Scholar] [CrossRef] [Green Version]
- Fang, Y.; Wang, Y.; Peng, M.; Xu, J.; Fan, Z.; Liu, C.; Zhao, K.; Zhang, H. Effect of paternal age on offspring birth defects: A systematic review and meta-analysis. Aging 2020, 12, 25373. [Google Scholar] [CrossRef]
- Zadzińska, E.; Kozieł, S.; Kurek, M.; Spinek, A. Mother’s trauma during pregnancy affects fluctuating asymmetry in offspring’s face. Anthropol. Anz. 2013, 70, 427–437. [Google Scholar] [CrossRef]
- King, S.; Mancini-Marïe, A.; Brunet, A.; Walker, E.; Meaney, M.J.; Laplante, D.P. Prenatal maternal stress from a natural disaster predicts dermatoglyphic asymmetry in humans. Dev. Psychopathol. 2009, 21, 343–353. [Google Scholar] [CrossRef] [Green Version]
- Hope, D.; Bates, T.; Penke, L.; Gow, A.J.; Starr, J.M.; Deary, I.J. Symmetry of the face in old age reflects childhood social status. Econ. Hum. Biol. 2013, 11, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Özener, B.; Fink, B. Facial symmetry in young girls and boys from a slum and a control area of Ankara, Turkey. Evol. Hum. Behav. 2010, 31, 436–441. [Google Scholar] [CrossRef]
- Pound, N.; Lawson, D.W.; Toma, A.M.; Richmond, S.; Zhurov, A.I.; Penton-Voak, I.S. Facial fluctuating asymmetry is not associated with childhood ill-health in a large British cohort study. Proc. R. Soc. B Biol. Sci. 2014, 281, 20141639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuna, B.; Galbarczyk, A.; Klimek, M.; Nenko, I.; Jasienska, G. Age difference between parents influences parity and number of sons. Am. J. Hum. Biol. 2018, 30, e23095. [Google Scholar] [CrossRef] [PubMed]
- Jasienska, G. Reproduction and lifespan: Trade-offs, overall energy budgets, intergenerational costs, and costs neglected by research. Am. J. Hum. Biol. 2009, 21, 524–532. [Google Scholar] [CrossRef]
- Little, A.C.; Jones, B.C.; DeBruine, L.M. The many faces of research on face perception. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 1634–1637. [Google Scholar] [CrossRef] [PubMed]
- Holzleitner, I.J.; Lee, A.J.; Hahn, A.C.; Kandrik, M.; Bovet, J.; Renoult, J.P.; Simmons, D.; Garrod, O.; DeBruine, L.M.; Jones, B.C. Comparing theory-driven and data-driven attractiveness models using images of real women’s faces. J. Exp. Psychol. Hum. Percept. Perform. 2019, 45, 1589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Toole, A.J.; Castillo, C.D.; Parde, C.J.; Hill, M.Q.; Chellappa, R. Face space representations in deep convolutional neural networks. Trends Cogn. Sci. 2018, 22, 794–809. [Google Scholar] [CrossRef] [PubMed]
- Renoult, J.P.; Bovet, J.; Raymond, M. Beauty is in the efficient coding of the beholder. R. Soc. Open Sci. 2016, 3, 160027. [Google Scholar] [CrossRef] [Green Version]
- Bray, I.; Gunnell, D.; Smith, G.D. Advanced paternal age: How old is too old? J. Epidemiol. Community Health 2006, 60, 851–853. [Google Scholar] [CrossRef] [Green Version]

| Mean | SD | Max | Min | |
|---|---|---|---|---|
| Facial distinctiveness | 0.06 | 0.01 | 0.13 | 0.03 |
| Facial asymmetry | 0.06 | 0.02 | 0.16 | 0.03 |
| Age | 11.66 | 4.23 | 21.56 | 3.01 |
| Birth order | 2.41 | 1.18 | 6 | 1 |
| Maternal age at birth (years) | 26.84 | 5.19 | 44.10 | 16.32 |
| Paternal age at birth (years) | 29.74 | 5.90 | 54.25 | 18.94 |
| Estimate | SE | 95% CI Low | 95% CI High | |
|---|---|---|---|---|
| Intercept | −0.26 | 0.12 | −0.49 | −0.04 |
| Sex (reference: male) | 0.45 | 0.16 | 0.13 | 0.76 |
| Age | −0.27 | 0.11 | −0.48 | −0.06 |
| Birth order | −0.26 | 0.13 | −0.51 | −0.01 |
| Maternal age at birth (years) | 0.36 | 0.14 | 0.10 | 0.64 |
| Paternal age at birth (years) | −0.18 | 0.14 | −0.44 | 0.09 |
| Estimate | SE | 95% CI Low | 95% CI High | |
|---|---|---|---|---|
| Intercept | −0.08 | 0.12 | −0.32 | 0.15 |
| Sex (reference: male) | 0.11 | 0.17 | −0.22 | 0.44 |
| Age | −0.26 | 0.11 | −0.47 | −0.06 |
| Birth order | −0.29 | 0.13 | −0.56 | −0.02 |
| Maternal age at birth (years) | 0.20 | 0.14 | −0.07 | 0.48 |
| Paternal age at birth (years) | −0.10 | 0.14 | −0.36 | 0.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klimek, M.; Marcinkowska, U.M.; Fedurek, P.; Kleisner, K.; Danel, D.P. Like Father, Like Child? Paternal Age at Birth and Offspring’s Facial Asymmetry and Distinctiveness. Symmetry 2022, 14, 344. https://doi.org/10.3390/sym14020344
Klimek M, Marcinkowska UM, Fedurek P, Kleisner K, Danel DP. Like Father, Like Child? Paternal Age at Birth and Offspring’s Facial Asymmetry and Distinctiveness. Symmetry. 2022; 14(2):344. https://doi.org/10.3390/sym14020344
Chicago/Turabian StyleKlimek, Magdalena, Urszula M. Marcinkowska, Pawel Fedurek, Karel Kleisner, and Dariusz P. Danel. 2022. "Like Father, Like Child? Paternal Age at Birth and Offspring’s Facial Asymmetry and Distinctiveness" Symmetry 14, no. 2: 344. https://doi.org/10.3390/sym14020344
APA StyleKlimek, M., Marcinkowska, U. M., Fedurek, P., Kleisner, K., & Danel, D. P. (2022). Like Father, Like Child? Paternal Age at Birth and Offspring’s Facial Asymmetry and Distinctiveness. Symmetry, 14(2), 344. https://doi.org/10.3390/sym14020344