Analyzing Turning Behavior after Repeated Lithium, Ketamine, or NaCl Injection and Chronic Stress Exposure in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Treatment and Chronic Stress Paradigm
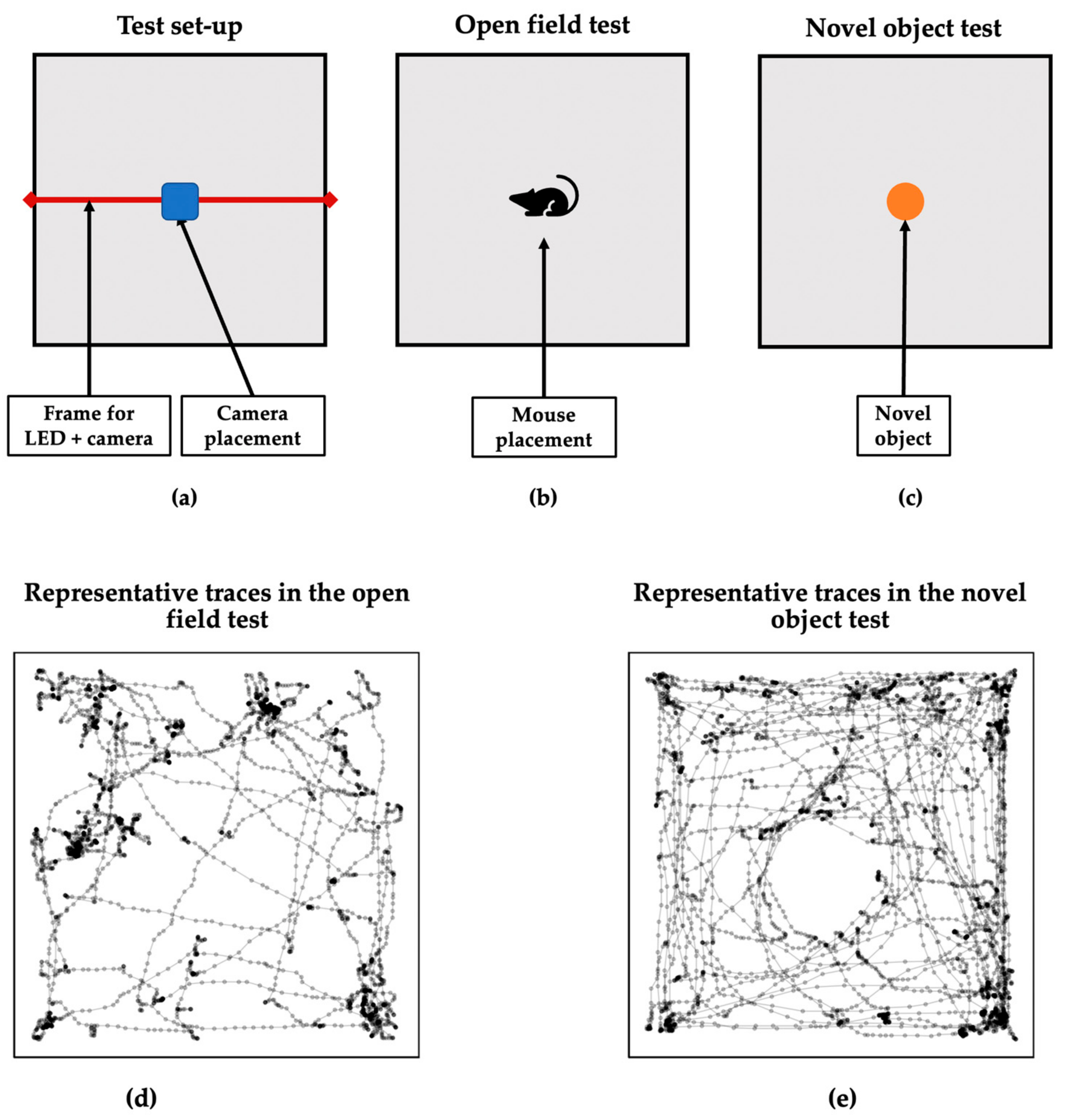
2.3. Behavioral Analysis
2.4. Laterality Measures
2.5. Statistical Analyses
3. Results
3.1. Behavioral Asymmetries
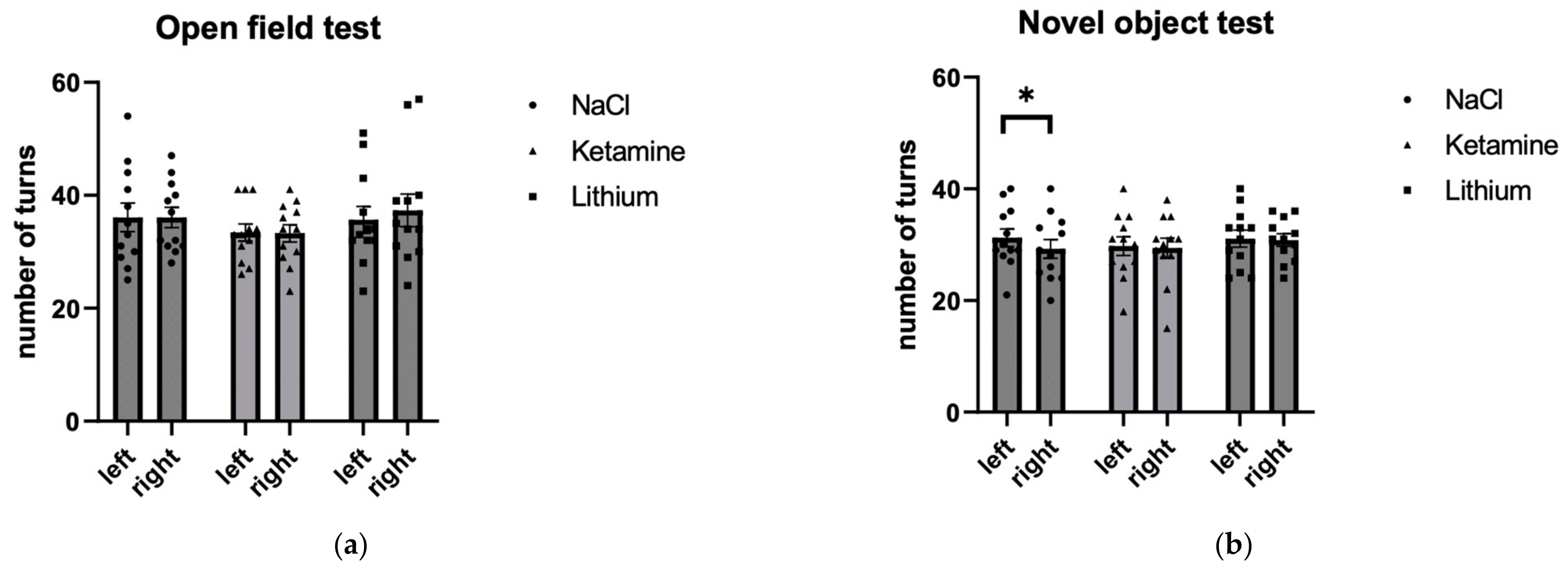
3.1.1. Number of Turns
3.1.2. Lateralization Quotient
3.1.3. Absolute Lateralization Quotient
3.1.4. Direction of Asymmetry
3.2. Behavioral Variability in the Novel Object Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Harbi, K.S. Treatment-Resistant Depression: Therapeutic Trends, Challenges, and Future Directions. Patient Prefer. Adherence 2012, 6, 369–388. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, D.H.; Rive, B.; Denee, T.R. The Humanistic and Economic Burden of Treatment-Resistant Depression in Europe: A Cross-Sectional Study. BMC Psychiatry 2019, 19, 247. [Google Scholar] [CrossRef] [PubMed]
- Zhdanava, M.; Pilon, D.; Ghelerter, I.; Chow, W.; Joshi, K.; Lefebvre, P.; Sheehan, J.J. The Prevalence and National Burden of Treatment-Resistant Depression and Major Depressive Disorder in the United States. J. Clin. Psychiatry 2021, 82, 20m13699. [Google Scholar] [CrossRef] [PubMed]
- Matveychuk, D.; Thomas, R.K.; Swainson, J.; Khullar, A.; MacKay, M.-A.; Baker, G.B.; Dursun, S.M. Ketamine as an Antidepressant: Overview of Its Mechanisms of Action and Potential Predictive Biomarkers. Ther. Adv. Psychopharmacol. 2020, 10, 2045125320916657. [Google Scholar] [CrossRef]
- Smith-Apeldoorn, S.Y.; Veraart, J.K.; Spijker, J.; Kamphuis, J.; Schoevers, R.A. Maintenance Ketamine Treatment for Depression: A Systematic Review of Efficacy, Safety, and Tolerability. Lancet Psychiatry 2022, 9, 907–921. [Google Scholar] [CrossRef]
- Molero, P.; Ramos-Quiroga, J.A.; Martin-Santos, R.; Calvo-Sánchez, E.; Gutiérrez-Rojas, L.; Meana, J.J. Antidepressant Efficacy and Tolerability of Ketamine and Esketamine: A Critical Review. CNS Drugs 2018, 32, 411–420. [Google Scholar] [CrossRef]
- Phillips, J.L.; Norris, S.; Talbot, J.; Birmingham, M.; Hatchard, T.; Ortiz, A.; Owoeye, O.; Batten, L.A.; Blier, P. Single, Repeated, and Maintenance Ketamine Infusions for Treatment-Resistant Depression: A Randomized Controlled Trial. Am. J. Psychiatry 2019, 176, 401–409. [Google Scholar] [CrossRef]
- Guarraci, F.A.; Gonzalez, C.M.F.; Lucero, D.; Womble, P.D.; Abdel-Rahim, H.; DeVore, J.; Kunkel, M.N.; Quadlander, E.; Stinnett, M.; Boyette-Davis, J. The Effects of Ketamine on Sexual Behavior, Anxiety, and Locomotion in Female Rats. Pharmacol. Biochem. Behav. 2018, 165, 36–44. [Google Scholar] [CrossRef]
- Franceschelli, A.; Sens, J.; Herchick, S.; Thelen, C.; Pitychoutis, P.M. Sex Differences in the Rapid and the Sustained Antidepressant-like Effects of Ketamine in Stress-Naïve and “Depressed” Mice Exposed to Chronic Mild Stress. Neuroscience 2015, 290, 49–60. [Google Scholar] [CrossRef]
- Ng, L.H.L.; Huang, Y.; Han, L.; Chang, R.C.-C.; Chan, Y.S.; Lai, C.S.W. Ketamine and Selective Activation of Parvalbumin Interneurons Inhibit Stress-Induced Dendritic Spine Elimination. Transl. Psychiatry 2018, 8, 272. [Google Scholar] [CrossRef]
- Okine, T.; Shepard, R.; Lemanski, E.; Coutellier, L. Sex Differences in the Sustained Effects of Ketamine on Resilience to Chronic Stress. Front. Behav. Neurosci. 2020, 14, 581360. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, P.J.; Yen, J.Y.; Watson, B.O. Stress-Sensitive Antidepressant-like Effects of Ketamine in the Mouse Forced Swim Test. PLoS ONE 2019, 14, e0215554. [Google Scholar] [CrossRef] [PubMed]
- Viktorov, M.; Wilkinson, M.P.; Elston, V.C.E.; Stone, M.; Robinson, E.S.J. A Systematic Review of Studies Investigating the Acute Effects of N-Methyl-D-Aspartate Receptor Antagonists on Behavioural Despair in Normal Animals Suggests Poor Predictive Validity. Brain Neurosci. Adv. 2022, 6, 23982128221081644. [Google Scholar] [CrossRef] [PubMed]
- Klein, P.S.; Melton, D.A. A Molecular Mechanism for the Effect of Lithium on Development. Proc. Natl. Acad. Sci. USA 1996, 93, 8455–8459. [Google Scholar] [CrossRef]
- Marcus, S.R.; Nadiger, H.A.; Chandrakala, M.V.; Rao, T.I.; Sadasivudu, B. Acute and Short-Term Effects of Lithium on Glutamate Metabolism in Rat Brain. Biochem. Pharmacol. 1986, 35, 365–369. [Google Scholar] [CrossRef]
- Petzold, J.; Bauer, M.; Severus, E. Update-Lithium in the Long-Term Treatment of Bipolar Disorders. Fortschr. Neurol. Psychiatr. 2018, 86, 745–753. [Google Scholar] [CrossRef]
- Silva, R.; Mesquita, A.R.; Bessa, J.; Sousa, J.C.; Sotiropoulos, I.; Leão, P.; Almeida, O.F.X.; Sousa, N. Lithium Blocks Stress-Induced Changes in Depressive-like Behavior and Hippocampal Cell Fate: The Role of Glycogen-Synthase-Kinase-3beta. Neuroscience 2008, 152, 656–669. [Google Scholar] [CrossRef]
- Martinowich, K.; Schloesser, R.J.; Manji, H.K. Bipolar Disorder: From Genes to Behavior Pathways. J. Clin. Investig. 2009, 119, 726–736. [Google Scholar] [CrossRef]
- DelBello, M.P.; Adler, C.M.; Cerullo, M.A.; Fleck, D.E.; Strakowski, M.S. Bipolar Disorder. In Encyclopedia of Neuroscience; Squire, L.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Geddes, J.R.; Burgess, S.; Hawton, K.; Jamison, K.; Goodwin, G.M. Long-Term Lithium Therapy for Bipolar Disorder: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Am. J. Psychiatry 2004, 161, 217–222. [Google Scholar] [CrossRef]
- Cipriani, A.; Hawton, K.; Stockton, S.; Geddes, J.R. Lithium in the Prevention of Suicide in Mood Disorders: Updated Systematic Review and Meta-Analysis. BMJ 2013, 346, f3646. [Google Scholar] [CrossRef]
- Volkmann, C.; Bschor, T.; Köhler, S. Lithium Treatment Over the Lifespan in Bipolar Disorders. Front. Psychiatry 2020, 11, 377. [Google Scholar] [CrossRef] [PubMed]
- Bschor, P.D.T.; Bauer, M. Nebenwirkungs- und Risikoprofil von Lithium. Nervenarzt 2013, 84, 860–863. [Google Scholar] [CrossRef] [PubMed]
- McKnight, R.F.; Adida, M.; Budge, K.; Stockton, S.; Goodwin, G.M.; Geddes, J.R. Lithium Toxicity Profile: A Systematic Review and Meta-Analysis. Lancet 2012, 379, 721–728. [Google Scholar] [CrossRef]
- Giles, J.J.; Bannigan, J.G. Teratogenic and Developmental Effects of Lithium. Curr. Pharm. Des. 2006, 12, 1531–1541. [Google Scholar] [CrossRef]
- Dygalo, N.N.; Bannova, A.V.; Sukhareva, E.V.; Shishkina, G.T.; Ayriyants, K.A.; Kalinina, T.S. Effects of Short-Term Exposure to Lithium on Antiapoptotic Bcl-XL Protein Expression in Cortex and Hippocampus of Rats after Acute Stress. Biochem. Biokhimiia 2017, 82, 345–350. [Google Scholar] [CrossRef]
- Mundorf, A.; Knorr, A.; Mezö, C.; Klein, C.; Beyer, D.K.; Fallgatter, A.J.; Schwarz, M.; Freund, N. Lithium and Glutamine Synthetase: Protective Effects Following Stress. Psychiatry Res. 2019, 281, 112544. [Google Scholar] [CrossRef]
- Andersen, S.L.; Teicher, M.H. Stress, Sensitive Periods and Maturational Events in Adolescent Depression. Trends Neurosci. 2008, 31, 183–191. [Google Scholar] [CrossRef]
- Beyer, D.K.E.; Freund, N. Animal Models for Bipolar Disorder: From Bedside to the Cage. Int. J. Bipolar Disord. 2017, 5, 35. [Google Scholar] [CrossRef]
- Abraham, M.; Mundorf, A.; Brodmann, K.; Freund, N. Unraveling the Mystery of White Matter in Depression: A Translational Perspective on Recent Advances. Brain Behav. 2022, 12, e2629. [Google Scholar] [CrossRef]
- Güntürkün, O.; Ströckens, F.; Ocklenburg, S. Brain Lateralization: A Comparative Perspective. Physiol. Rev. 2020, 100, 1019–1063. [Google Scholar] [CrossRef]
- Niven, J.E.; Frasnelli, E. Insights into the Evolution of Lateralization from the Insects. Prog. Brain Res. 2018, 238, 3–31. [Google Scholar] [CrossRef]
- Vallortigara, G.; Rogers, L.J. A Function for the Bicameral Mind. Cortex J. Devoted Study Nerv. Syst. Behav. 2020, 124, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Ocklenburg, S.; Mundorf, A. Symmetry and Asymmetry in Biological Structures. Proc. Natl. Acad. Sci. USA 2022, 119, e2204881119. [Google Scholar] [CrossRef] [PubMed]
- Mundorf, A.; Peterburs, J.; Ocklenburg, S. Asymmetry in the Central Nervous System: A Clinical Neuroscience Perspective. Front. Syst. Neurosci. 2021, 15, 733898. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.-Z.; Postema, M.C.; Guadalupe, T.; de Kovel, C.; Boedhoe, P.S.W.; Hoogman, M.; Mathias, S.R.; van Rooij, D.; Schijven, D.; Glahn, D.C.; et al. Mapping Brain Asymmetry in Health and Disease through the ENIGMA Consortium. Hum. Brain Mapp. 2022, 43, 167–181. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Peterburs, J.; Mundorf, A. Hemispheric Asymmetries in the Amygdala: A Comparative Primer. Prog. Neurobiol. 2022, 214, 102283. [Google Scholar] [CrossRef]
- Berretz, G.; Wolf, O.T.; Güntürkün, O.; Ocklenburg, S. Atypical Lateralization in Neurodevelopmental and Psychiatric Disorders: What Is the Role of Stress? Cortex J. Devoted Study Nerv. Syst. Behav. 2020, 125, 215–232. [Google Scholar] [CrossRef]
- Mundorf, A.; Matsui, H.; Ocklenburg, S.; Freund, N. Asymmetry of Turning Behavior in Rats Is Modulated by Early Life Stress. Behav. Brain Res. 2020, 393, 112807. [Google Scholar] [CrossRef]
- Mundorf, A.; Kubitza, N.; Hünten, K.; Matsui, H.; Juckel, G.; Ocklenburg, S.; Freund, N. Maternal Immune Activation Leads to Atypical Turning Asymmetry and Reduced DRD2 MRNA Expression in a Rat Model of Schizophrenia. Behav. Brain Res. 2021, 414, 113504. [Google Scholar] [CrossRef]
- Pfeifer, L.S.; Heyers, K.; Ocklenburg, S.; Wolf, O.T. Stress Research during the COVID-19 Pandemic and Beyond. Neurosci. Biobehav. Rev. 2021, 131, 581–596. [Google Scholar] [CrossRef]
- Mundorf, A.; Schmitz, J.; Hünten, K.; Fraenz, C.; Schlüter, C.; Genç, E.; Ocklenburg, S.; Freund, N. MORC1 Methylation and BDI Are Associated with Microstructural Features of the Hippocampus and Medial Prefrontal Cortex. J. Affect. Disord. 2021, 282, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Manns, M.; Basbasse, Y.E.; Freund, N.; Ocklenburg, S. Paw Preferences in Mice and Rats: Meta-Analysis. Neurosci. Biobehav. Rev. 2021, 127, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Soyman, E.; Yilmaz, G.D.; Canbeyli, R. Head-Turning Asymmetry: A Novel Lateralization in Rats Predicts Susceptibility to Behavioral Despair. Behav. Brain Res. 2018, 338, 47–50. [Google Scholar] [CrossRef]
- Schwarting, R.K.W.; Borta, A. Analysis of Behavioral Asymmetries in the Elevated Plus-Maze and in the T-Maze. J. Neurosci. Methods 2005, 141, 251–260. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Berretz, G.; Packheiser, J.; Friedrich, P. Laterality 2020: Entering the next Decade. Laterality 2021, 26, 265–297. [Google Scholar] [CrossRef] [PubMed]
- Vallortigara, G. Laterality for the next Decade: Computational Ethology and the Search for Minimal Condition for Cognitive Asymmetry. Laterality 2021, 26, 303–306. [Google Scholar] [CrossRef]
- Anderson, D.J.; Perona, P. Toward a Science of Computational Ethology. Neuron 2014, 84, 18–31. [Google Scholar] [CrossRef]
- Mathis, A.; Mamidanna, P.; Cury, K.M.; Abe, T.; Murthy, V.N.; Mathis, M.W.; Bethge, M. DeepLabCut: Markerless Pose Estimation of User-Defined Body Parts with Deep Learning. Nat. Neurosci. 2018, 21, 1281–1289. [Google Scholar] [CrossRef]
- Mathis, M.W.; Mathis, A. Deep Learning Tools for the Measurement of Animal Behavior in Neuroscience. Curr. Opin. Neurobiol. 2020, 60, 1–11. [Google Scholar] [CrossRef]
- Nath, T.; Mathis, A.; Chen, A.C.; Patel, A.; Bethge, M.; Mathis, M.W. Using DeepLabCut for 3D Markerless Pose Estimation across Species and Behaviors. Nat. Protoc. 2019, 14, 2152–2176. [Google Scholar] [CrossRef]
- Hardin, A.; Schlupp, I. Using Machine Learning and DeepLabCut in Animal Behavior. Acta Ethologica 2022, 25, 125–133. [Google Scholar] [CrossRef]
- Josserand, M.; Rosa-Salva, O.; Versace, E.; Lemaire, B.S. Visual Field Analysis: A Reliable Method to Score Left and Right Eye Use Using Automated Tracking. Behav. Res. Methods 2022, 54, 1715–1724. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Korte, S.M.; Peterburs, J.; Wolf, O.T.; Güntürkün, O. Stress and Laterality-The Comparative Perspective. Physiol. Behav. 2016, 164, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Mundorf, A.; Ocklenburg, S. The Clinical Neuroscience of Lateralization; Routledge: London, UK, 2021; ISBN 978-1-00-308250-7. [Google Scholar]
- Silberman, E.K.; Weingartner, H. Hemispheric Lateralization of Functions Related to Emotion. Brain Cogn. 1986, 5, 322–353. [Google Scholar] [CrossRef]
- Quaranta, A.; Siniscalchi, M.; Vallortigara, G. Asymmetric Tail-Wagging Responses by Dogs to Different Emotive Stimuli. Curr. Biol. 2007, 17, R199–R201. [Google Scholar] [CrossRef] [PubMed]
- Simon, T.; Frasnelli, E.; Guo, K.; Barber, A.; Wilkinson, A.; Mills, D.S. Is There an Association between Paw Preference and Emotionality in Pet Dogs? Animals 2022, 12, 1153. [Google Scholar] [CrossRef]
- Farmer, K.; Krueger, K.; Byrne, R.W. Visual Laterality in the Domestic Horse (Equus caballus) Interacting with Humans. Anim. Cogn. 2010, 13, 229–238. [Google Scholar] [CrossRef]
- Tondo, L.; Alda, M.; Bauer, M.; Bergink, V.; Grof, P.; Hajek, T.; Lewitka, U.; Licht, R.W.; Manchia, M.; Müller-Oerlinghausen, B.; et al. Clinical Use of Lithium Salts: Guide for Users and Prescribers. Int. J. Bipolar Disord. 2019, 7, 16. [Google Scholar] [CrossRef]
- Fanciullacci, M.; Pietrini, U.; Boccuni, M.; Gatto, G.; Cangi, F. Does Lithium Balance the Neuronal Bilateral Asymmetries in Cluster Headache? Cephalalgia Int. J. Headache 1983, 3 (Suppl. S1), 85–87. [Google Scholar] [CrossRef]
- Tan, U.; Kara, I.; Tan, S. Lithium and Imipramin Effects on Paw Preference in Cats. Int. J. Neurosci. 1990, 52, 25–28. [Google Scholar] [CrossRef]
- Lebedev, S.V.; Petrov, S.V.; Blinov, D.V.; Lazarenko, I.P.; Chekhonin, V.P. Ketamine-Induced Rotational Asymmetry in Evaluation of Motor Disturbances in Rats with Middle Cerebral Artery Occlusion. Bull. Exp. Biol. Med. 2003, 135, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Lin, C.-T.; Ding, W.; Chen, M.-H.; Li, C.-T.; Su, T.-P. Identifying Ketamine Responses in Treatment-Resistant Depression Using a Wearable Forehead EEG. IEEE Trans. Biomed. Eng. 2019, 66, 1668–1679. [Google Scholar] [CrossRef] [PubMed]
- Rincón-Cortés, M.; Grace, A.A. Antidepressant Effects of Ketamine on Depression-Related Phenotypes and Dopamine Dysfunction in Rodent Models of Stress. Behav. Brain Res. 2020, 379, 112367. [Google Scholar] [CrossRef] [PubMed]

| Left | Right | Students t-Test | |||||
|---|---|---|---|---|---|---|---|
| Treatment | Mean | SE | Mean | SE | Cohen’s d | T | p |
| NaCl | 36.083 | 2.521 | 36.083 | 1.803 | 0.000 | 0.000 | 1.000 |
| Ketamine | 33.417 | 1.525 | 33.250 | 1.548 | 0.055 | 0.192 | 0.851 |
| Lithium | 35.667 | 2.369 | 37.333 | 2.909 | −0.447 | −1.550 | 0.149 |
| Left | Right | Students t-Test | |||||
|---|---|---|---|---|---|---|---|
| Treatment | Mean | SE | Mean | SE | Cohen’s d | T | p |
| NaCl | 31.250 | 1.567 | 29.250 | 1.688 | 0.816 | 2.828 | 0.016 |
| Ketamine | 29.750 | 1.684 | 29.417 | 1.764 | 0.188 | 0.650 | 0.529 |
| Lithium | 31.083 | 1.520 | 30.833 | 1.120 | 0.059 | 0.205 | 0.841 |
| Treatment | Left | Ambilateral | Right | Total |
|---|---|---|---|---|
| NaCl | 5 | 0 | 7 | 12 |
| Ketamine | 5 | 2 | 5 | 12 |
| Lithium | 3 | 2 | 7 | 12 |
| Total | 13 | 4 | 19 | 36 |
| Treatment | Left | Ambilateral | Right | Total |
|---|---|---|---|---|
| NaCl | 8 | 3 | 1 | 12 |
| Ketamine | 4 | 4 | 4 | 12 |
| Lithium | 7 | 0 | 5 | 12 |
| Total | 19 | 7 | 10 | 36 |
| Treatment | Angle Facing Object | Distance Traveled to Object | Proportion of Staying | |||
|---|---|---|---|---|---|---|
| Average | SE | Average | SE | Average | SE | |
| NaCl | 6.788 | ±3.477 | 77.207 | ±3.595 | 0.194 | ±0.034 |
| Ketamine | 5.094 | ±3.960 | 82.940 | ±2.977 | 0.148 | ±0.029 |
| Lithium | 7.599 | ±3.694 | 86.313 | ±3.211 | 0.121 | ±0.024 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mundorf, A.; Matsui, H.; Ocklenburg, S.; Freund, N. Analyzing Turning Behavior after Repeated Lithium, Ketamine, or NaCl Injection and Chronic Stress Exposure in Mice. Symmetry 2022, 14, 2352. https://doi.org/10.3390/sym14112352
Mundorf A, Matsui H, Ocklenburg S, Freund N. Analyzing Turning Behavior after Repeated Lithium, Ketamine, or NaCl Injection and Chronic Stress Exposure in Mice. Symmetry. 2022; 14(11):2352. https://doi.org/10.3390/sym14112352
Chicago/Turabian StyleMundorf, Annakarina, Hiroshi Matsui, Sebastian Ocklenburg, and Nadja Freund. 2022. "Analyzing Turning Behavior after Repeated Lithium, Ketamine, or NaCl Injection and Chronic Stress Exposure in Mice" Symmetry 14, no. 11: 2352. https://doi.org/10.3390/sym14112352
APA StyleMundorf, A., Matsui, H., Ocklenburg, S., & Freund, N. (2022). Analyzing Turning Behavior after Repeated Lithium, Ketamine, or NaCl Injection and Chronic Stress Exposure in Mice. Symmetry, 14(11), 2352. https://doi.org/10.3390/sym14112352








