Brain Symmetry in Alpha Band When Watching Cuts in Movies
Abstract
:1. Introduction
1.1. Neural Processing of Visual Content
1.2. Neural Processing of Audiovisual Cuts
1.3. Brain Asymmetry in Visual Perception
2. Materials and Methods
2.1. Participants
2.2. Stimuli
2.3. Data Acquisition
2.4. Data Analysis
3. Results
3.1. Alpha Power
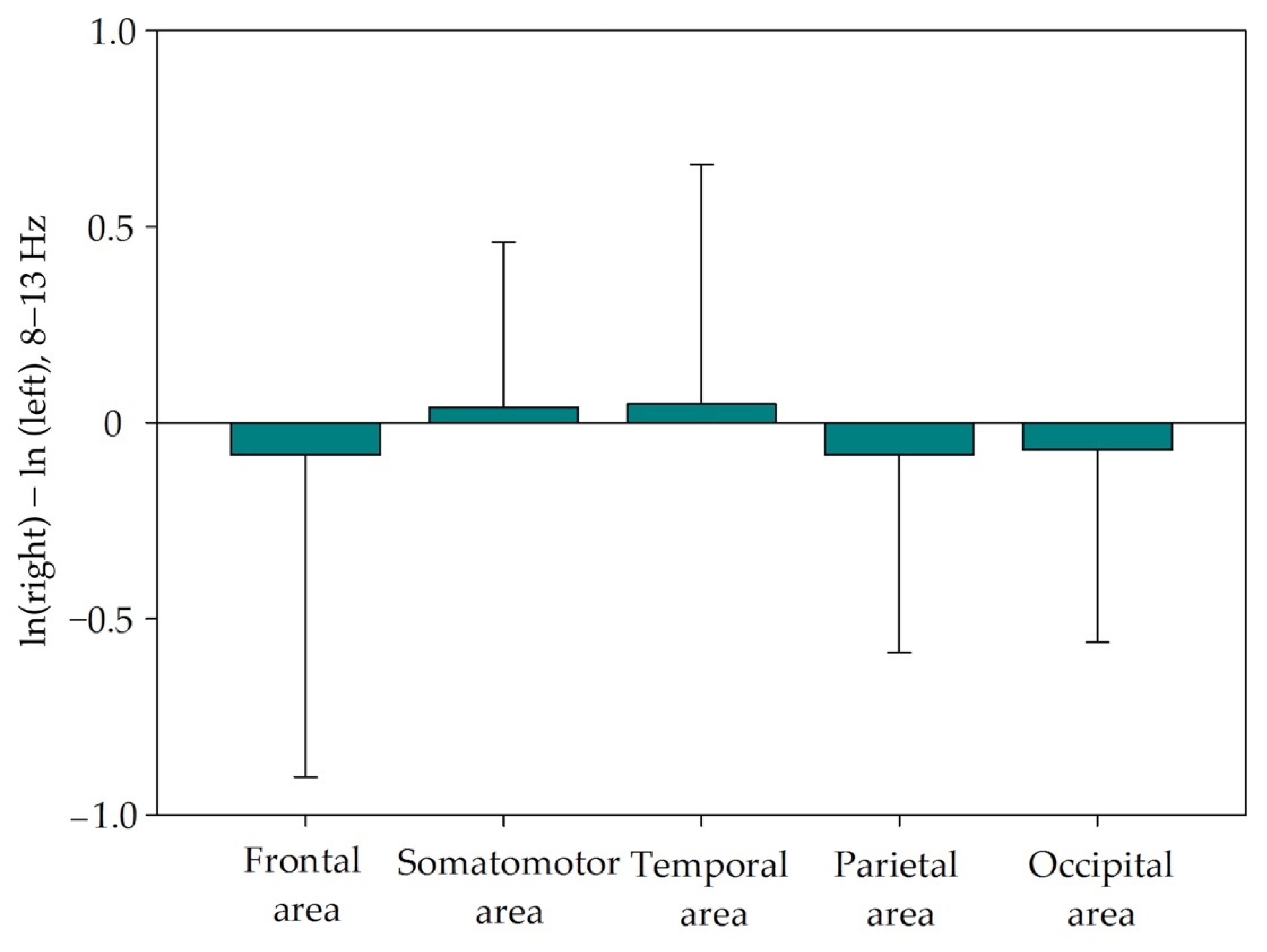
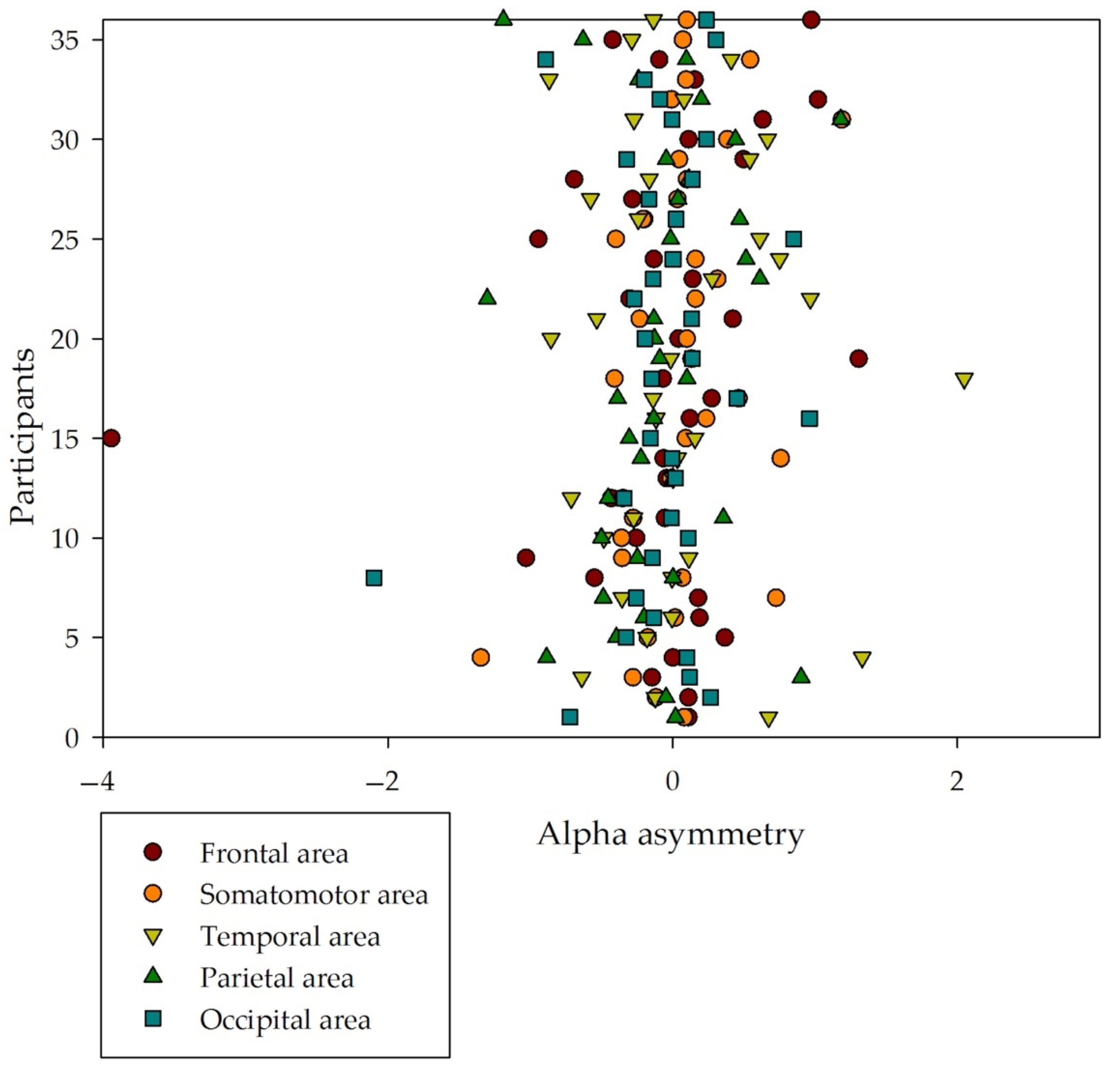
3.2. Alpha Asymmetry
3.3. Alpha Asymmetry and Media Expertise
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martinez-Conde, S.; Macknik, S.L.; Hubel, D.H. The role of fixational eye movements in visual perception. Nat. Rev. Neurosci. 2004, 5, 229–240. [Google Scholar] [CrossRef]
- Seriès, P.; Seitz, A.R. Learning what to expect (in visual perception). Front. Hum. Neurosci. 2013, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Pylyshyn, Z. Is vision continuous with cognition? The case for cognitive impenetrability of visual perception Zenon. Behav. Brain Sci. 1999, 22, 341–423. [Google Scholar] [CrossRef] [PubMed]
- Yamins, D.L.K.; Di Carlo, J.J. Using goal-driven deep learning models to understand sensory cortex. Nat. Neurosci. 2016, 19, 356–365. [Google Scholar] [CrossRef]
- Groen, I.I.A.; Silson, E.H.; Baker, C.I. Contributions of low- and high-level properties to neural processing of visual scenes in the human brain. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160102. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, S.; Fize, D.; Marlot, C. Speed of processing in the human visual system. Nature 1996, 381, 520–522. [Google Scholar] [CrossRef]
- Buzsáki, G. How the Brain ‘Constructs’ the Outside World. Sci. Am. 2022, 326, 36–43. [Google Scholar] [CrossRef]
- Hawkins, J.; Blakeslee, S. On Intelligence; Henry Holt and Company: New York, NY, USA, 2004. [Google Scholar]
- Zeki, S.; Bartels, A. Toward a theory of visual consciousness. Conscious. Cogn. 1999, 8, 225–259. [Google Scholar] [CrossRef] [PubMed]
- Pearlman, K. Cutting Rhythms: Shaping the Film Edit; Focal Press: Oxford, UK; Elsevier: Oxford, UK, 2009; ISBN 978-0-240-81014-0. [Google Scholar]
- Zacks, J.M.; Speer, N.K.; Reynolds, J.R. Segmentation in reading and film comprehension. J. Exp. Psychol. Gen. 2009, 138, 307–327. [Google Scholar] [CrossRef]
- Andreu-Sánchez, C.; Martín-Pascual, M.Á.; Gruart, A.; Delgado-García, J.M. Chaotic and Fast Audiovisuals Increase Attentional Scope but Decrease Conscious Processing. Neuroscience 2018, 394, 83–97. [Google Scholar] [CrossRef]
- Andreu-Sánchez, C.; Martín-Pascual, M.Á.; Gruart, A.; Delgado-García, J.M. Eyeblink rate watching classical Hollywood and post-classical MTV editing styles, in media and non-media professionals. Sci. Rep. 2017, 7, 43267. [Google Scholar] [CrossRef]
- Colzato, L.S.; Slagter, H.A.; Spapé, M.M.A.; Hommel, B. Blinks of the eye predict blinks of the mind. Neuropsychologia 2008, 46, 3179–3183. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Jiang, X.; Tien, G.; Meneghetti, A.; Panton, O.N.M.; Atkins, M.S. Workload assessment of surgeons: Correlation between NASA TLX and blinks. Surg. Endosc. Other Interv. Tech. 2012, 26, 2746–2750. [Google Scholar] [CrossRef]
- Andreu-Sánchez, C.; Martín-Pascual, M.Á.; Gruart, A.; Delgado-García, J.M. The Effect of Media Professionalization on Cognitive Neurodynamics During Audiovisual Cuts. Front. Syst. Neurosci. 2021, 1, 598383. [Google Scholar] [CrossRef] [PubMed]
- Bertamini, M.; Makin, A.D.J. Brain activity in response to visual symmetry. Symmetry 2014, 6, 975–996. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J.B. Frontal EEG asymmetry and the behavioral activation and inhibition systems. Psychophysiology 2003, 40, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Coan, J.A.; Allen, J.J.B. Frontal EEG asymmetry as a moderator and mediator of emotion. Biol. Psychol. 2004, 67, 7–50. [Google Scholar] [CrossRef] [PubMed]
- Fischer, N.L.; Peres, R.; Fiorani, M. Frontal alpha asymmetry and theta oscillations associated with information sharing intention. Front. Behav. Neurosci. 2018, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.A.; Fox, N.A. Electroencephalogram asymmetry during emotionally evocative films and its relation to positive and negative affectivity. Brain Cogn. 1992, 20, 280–299. [Google Scholar] [CrossRef]
- Tomarken, A.J.; Davidson, R.J.; Henriques, J.B. Resting Frontal Brain Asymmetry Predicts Affective Responses to Films. J. Pers. Soc. Psychol. 1990, 59, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Ohme, R.; Reykowska, D.; Wiener, D.; Choromanska, A. Application of frontal EEG asymmetry to advertising research. J. Econ. Psychol. 2010, 31, 785–793. [Google Scholar] [CrossRef]
- Germeys, F.; D’Ydewalle, G. The psychology of film: Perceiving beyond the cut. Psychol. Res. 2007, 71, 458–466. [Google Scholar] [CrossRef]
- Magliano, J.P.; Zacks, J.M. The impact of continuity editing in narrative film on event segmentation. Cogn. Sci. 2011, 35, 1489–1517. [Google Scholar] [CrossRef] [PubMed]
- Francuz, P.; Zabielska-Mendyk, E. Does the Brain Differentiate Between Related and Unrelated Cuts When Processing Audiovisual Messages? An ERP Study. Media Psychol. 2013, 16, 461–475. [Google Scholar] [CrossRef]
- Smith, T. The attentional theory of cinematic continuity. Projections 2012, 6, 1–27. [Google Scholar] [CrossRef]
- Jasper, H.H. The ten twenty electrode system of the International Federation. Report of the committee on methods of clinical examination in electroencephalography. Electroencephalogr. Clin. Neurophysiol. 1958, 10, 371–375. [Google Scholar] [CrossRef]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Mognon, A.; Jovicich, J.; Bruzzone, L.; Buiatti, M. ADJUST: An automatic EEG artifact detector based on the joint use of spatial and temporal features. Psychophysiology 2010, 48, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Mathewson, K.J.; Hashemi, A.; Sheng, B.; Sekuler, A.B.; Bennett, P.J.; Schmidt, L.A. Regional electroencephalogram (EEG) alpha power and asymmetry in older adults: A study of short-term test-retest reliability. Front. Aging Neurosci. 2015, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J. Emotion and Affective Style: Hemispheric Substrates. Psychol. Sci. 1992, 3, 39–43. [Google Scholar] [CrossRef]
- Andreu-Sánchez, C.; Martín-Pascual, M.Á.; Gruart, A.; Delgado-García, J.M. Looking at reality versus watching screens: Media professionalization effects on the spontaneous eyeblink rate. PLoS ONE 2017, 12, e0176030. [Google Scholar] [CrossRef]
- Maguire, E.A.; Woollett, K.; Spiers, H.J. London taxi drivers and bus drivers: A structural MRI and neuropsychological analysis. Hippocampus 2006, 16, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Maguire, E.A.; Gadian, D.G.; Johnsrude, I.S.; Good, C.D.; Ashburner, J.; Frackowiak, R.S.; Frith, C.D. Navigation-related structural change in the hippocampi of taxi drivers. Proc. Natl. Acad. Sci. USA 2000, 97, 4398–4403. [Google Scholar] [CrossRef]
- Lotze, M.; Scheler, G.; Tan, H.-R.; Braun, C.; Birbaumer, N. The musician’s brain: Functional imaging of amateurs and professionals during performance and imagery. Neuroimage 2003, 20, 1817–1829. [Google Scholar] [CrossRef]
- Faubert, J. Professional athletes have extraordinary skills for rapidly learning complex and neutral dynamic visual scenes. Sci. Rep. 2013, 3, 1154. [Google Scholar] [CrossRef] [PubMed]
- Mcgee, R.A.; Clark, S.E.; Symons, D.K. Does the Conners’ Continuous Performance Test aid in ADHD diagnosis? J. Abnorm. Child Psychol. 2000, 28, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Memória, C.M.; Muela, H.C.S.; Moraes, N.C.; Costa-Hong, V.A.; Machado, M.F.; Nitrini, R.; Bortolotto, L.A.; Yassuda, M.S. Applicability of the test of variables of attention—T.O.V.A in Brazilian adults. Dement. Neuropsychol. 2018, 12, 394–401. [Google Scholar] [CrossRef]
| Brain Area | Left Hemisphere | Right Hemisphere |
|---|---|---|
| Frontal | Fp1 | Fp2 |
| F3 | F4 | |
| F7 | F8 | |
| Somatomotor | C3 | C4 |
| Temporal | T7 | T8 |
| Parietal | P3 P7 | P4 P8 |
| Occipital | O1 | O2 |
| Pairwise Comparison | Rank Sum Diff. | Significant? | p-Value |
|---|---|---|---|
| Frontal left vs. frontal right | 3.000 | No | >0.9999 |
| Frontal left vs. somatomotor left | 213.500 | Yes | <0.0001 |
| Frontal left vs. somatomotor right | 204.500 | Yes | <0.0001 |
| Frontal left vs. temporal left | 165.000 | Yes | <0.0001 |
| Frontal left vs. temporal right | 180.000 | Yes | <0.0001 |
| Frontal left vs. parietal left | 95.000 | Yes | 0.0098 |
| Frontal left vs. parietal right | 115.000 | Yes | 0.0003 |
| Frontal left vs. occipital left | 146.000 | Yes | <0.0001 |
| Frontal left vs. occipital right | 148.000 | Yes | <0.0001 |
| Frontal right vs. somatomotor left | 210.500 | Yes | <0.0001 |
| Frontal right vs. somatomotor right | 201.500 | Yes | <0.0001 |
| Frontal right vs. temporal left | 162.000 | Yes | <0.0001 |
| Frontal right vs. temporal right | 177.000 | Yes | <0.0001 |
| Frontal right vs. parietal left | 92.000 | Yes | 0.0154 |
| Frontal right vs. parietal right | 112.000 | Yes | 0.0006 |
| Frontal right vs. occipital left | 143.000 | Yes | <0.0001 |
| Frontal right vs. occipital right | 145.000 | Yes | <0.0001 |
| Somatomotor left vs. somatomotor right | −9.000 | No | >0.9999 |
| Somatomotor left vs. temporal left | −48.500 | No | >0.9999 |
| Somatomotor left vs. temporal right | −33.500 | No | >0.9999 |
| Somatomotor left vs. parietal left | −118.500 | Yes | 0.0002 |
| Somatomotor left vs. parietal right | −98.500 | Yes | 0.0057 |
| Somatomotor left vs. occipital left | −67.500 | No | 0.3871 |
| Somatomotor left vs. occipital right | −65.500 | No | 0.4853 |
| Somatomotor right vs. temporal left | −39.500 | No | >0.9999 |
| Somatomotor right vs. temporal right | −24.500 | No | >0.9999 |
| Somatomotor right vs. parietal left | −109.500 | Yes | 0.0009 |
| Somatomotor right vs. parietal right | −89.500 | Yes | 0.0222 |
| Somatomotor right vs. occipital left | −58.500 | No | >0.9999 |
| Somatomotor right vs. occipital right | −56.500 | No | >0.9999 |
| Temporal left vs. temporal right | 15.000 | No | >0.9999 |
| Temporal left vs. parietal left | −70.000 | No | 0.2896 |
| Temporal left vs. parietal right | −50.000 | No | >0.9999 |
| Temporal left vs. occipital left | −19.000 | No | >0.9999 |
| Temporal left vs. occipital right | −17.000 | No | >0.9999 |
| Temporal right vs. parietal left | −85.000 | Yes | 0.0422 |
| Temporal right vs. parietal right | −65.000 | No | 0.5131 |
| Temporal right vs. occipital left | −34.000 | No | >0.9999 |
| Temporal right vs. occipital right | −32.000 | No | >0.9999 |
| Parietal left vs. parietal right | 20.000 | No | >0.9999 |
| Parietal left vs. occipital left | 51.000 | No | >0.9999 |
| Parietal left vs. occipital right | 53.000 | No | >0.9999 |
| Parietal right vs. occipital left | 31.000 | No | >0.9999 |
| Parietal right vs. occipital right | 33.000 | No | >0.9999 |
| Occipital left vs. occipital right | 2.000 | No | >0.9999 |
| Brain Area | Left Hemisphere | Right Hemisphere | Asymmetry (SD) |
|---|---|---|---|
| Frontal | 3.006 (3.936) | 2.557 (2.004) | −0.0822 (0.822) |
| Somatomotor | 0.545 (0.462) | 0.577 (0.534) | 0.0383 (0.422) |
| Temporal | 0.696 (0.603) | 0.658 (0.527) | 0.0474 (0.610) |
| Parietal | 1.029 (0.944) | 0.988 (0.865) | −0.0826 (0.504) |
| Occipital | 0.742 (0.688) | 0.712 (0.691) | −0.0826 (0.504) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andreu-Sánchez, C.; Martín-Pascual, M.Á.; Gruart, A.; Delgado-García, J.M. Brain Symmetry in Alpha Band When Watching Cuts in Movies. Symmetry 2022, 14, 1980. https://doi.org/10.3390/sym14101980
Andreu-Sánchez C, Martín-Pascual MÁ, Gruart A, Delgado-García JM. Brain Symmetry in Alpha Band When Watching Cuts in Movies. Symmetry. 2022; 14(10):1980. https://doi.org/10.3390/sym14101980
Chicago/Turabian StyleAndreu-Sánchez, Celia, Miguel Ángel Martín-Pascual, Agnès Gruart, and José María Delgado-García. 2022. "Brain Symmetry in Alpha Band When Watching Cuts in Movies" Symmetry 14, no. 10: 1980. https://doi.org/10.3390/sym14101980
APA StyleAndreu-Sánchez, C., Martín-Pascual, M. Á., Gruart, A., & Delgado-García, J. M. (2022). Brain Symmetry in Alpha Band When Watching Cuts in Movies. Symmetry, 14(10), 1980. https://doi.org/10.3390/sym14101980







