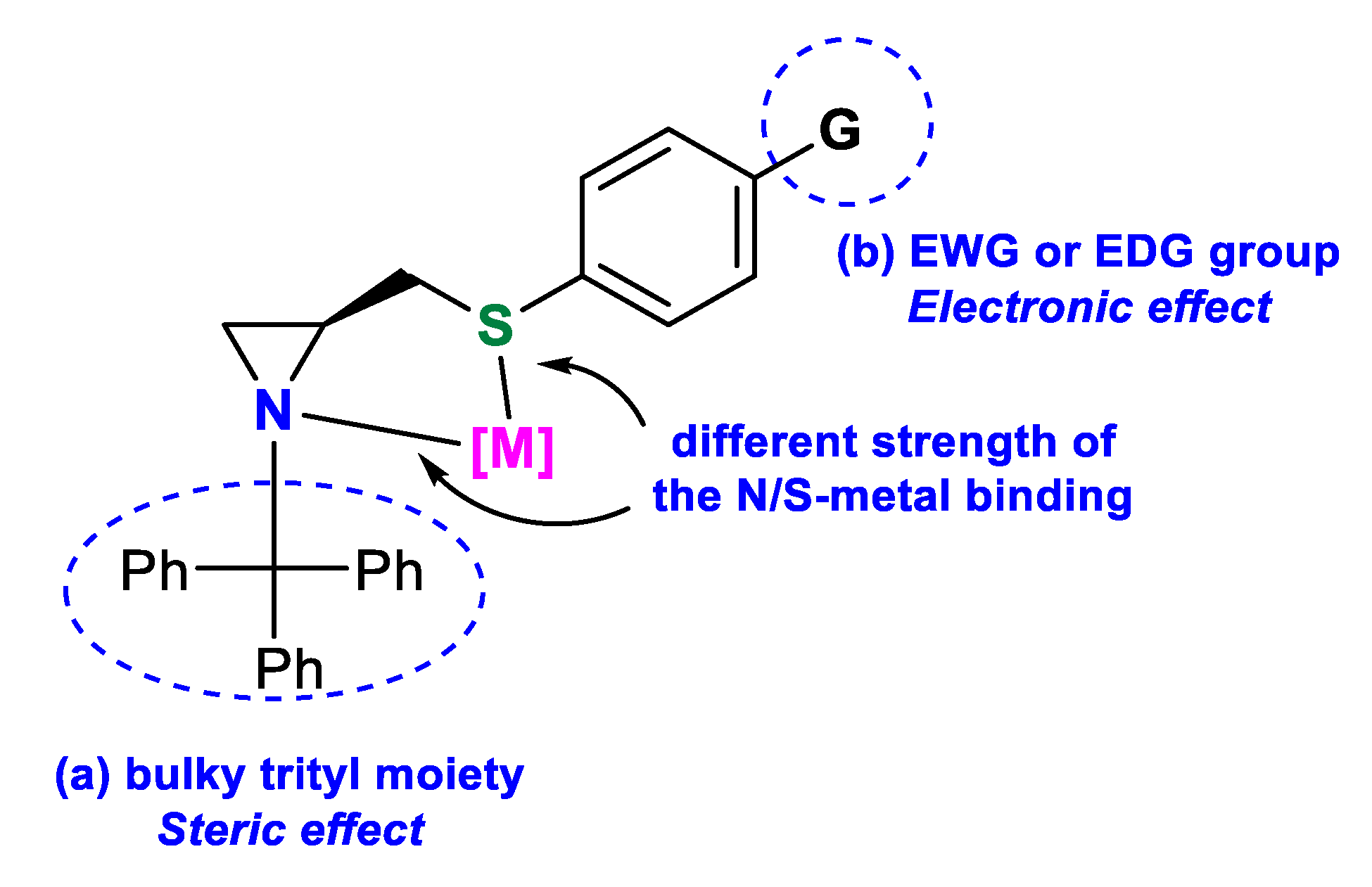
Chiral Aziridine Sulfide N(sp3),S-Ligands for Metal-Catalyzed Asymmetric Reactions
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Procedures and Analysis Data
3.2.1. Synthesis of (S)-Methyl 3-Hydroxy-2- (Tritylamino)Propanoate 10
3.2.2. Synthesis of (S)-Methyl 1-Tritylaziridine-2-Carboxylate 11
3.2.3. Synthesis of f (S)-(1-Tritylaziridin-2-yl)Methanol 12
3.2.4. Synthesis of (S)-(1-Tritylaziridin-2-yl)Methyl 4- Methylbenzenesulfonate 13
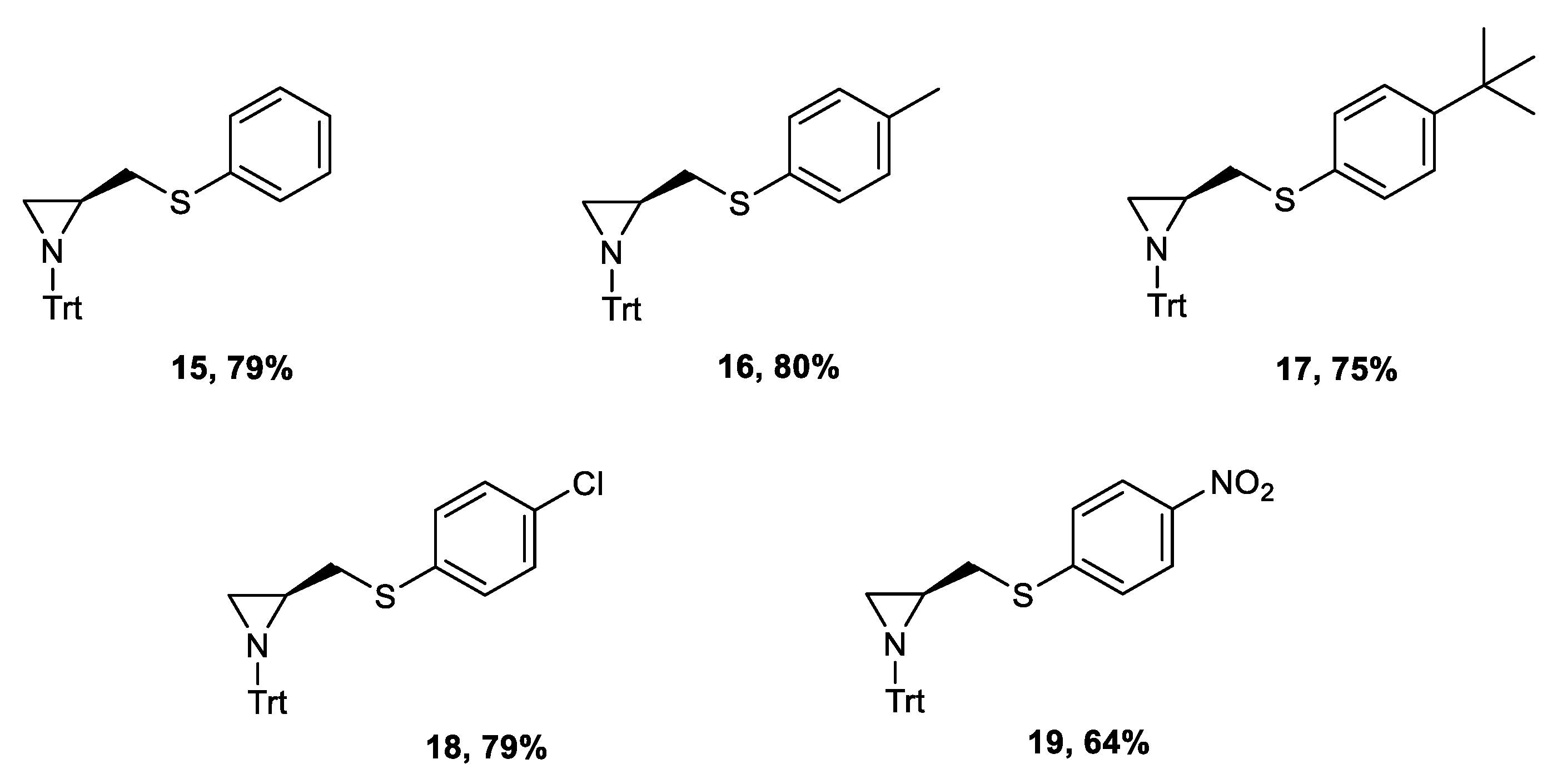
3.2.5. Synthesis of 2-Thiophenylaziridines 15–19
(S)-2-(Phenylsulfanylmethyl)-1-Tritylaziridine 15
(S)-2-(p-Tolylsulfanylmethyl)-1-Tritylaziridine 16
(S)-2-((4-Tert-Butylphenylsulfanyl)Methyl)-1-Tritylaziridine 17
(S)-2-((4-Chlorophenylsulfanyl)Methyl)-1-Tritylaziridine 18
(S)-2-((4-Nitrophenylsulfanyl)Methyl)-1-Tritylaziridine 19
3.3. General Procedure for AAA (Trost–Tsuji) Reaction
3.4. General Procedure for the Asymmetric Addition of Diethylzinc to Aldehydes
3.5. General Procedure for the Asymmetric Addition of Phenylethynylzinc to Benzaldehyde
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Degennaro, L.; Trinchera, P.; Luisi, R. Recent Advances in the Stereoselective Synthesis of Aziridines. Chem. Rev. 2014, 114, 7881–7929. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.S. Advances in the synthesis and chemistry of aziridines. Adv. Heterocycl. Chem. 2019, 129, 245–335. [Google Scholar]
- Botuha, C.; Chemla, F.; Ferreira, F.; Pérez-Luna, A. Aziridines in Natural Product Synthesis. In Heterocycles in Natural Product Synthesis; Majumdar, K.C., Chattopadhyay, S.K., Eds.; Wiley: Hoboken, NJ, USA, 2011; pp. 154–196. [Google Scholar]
- Macha, L.; D’hooghe, M.; Ha, H.-J. Deployment of Aziridines for the Synthesis of Alkaloids and Their Derivatives. Synthesis 2019, 51, 1491–1515. [Google Scholar]
- Tanner, D. Chiral Aziridines—Their Synthesis and Use in Stereoselective Transformations. Angew. Chem. Int. Ed. 1994, 33, 599–619. [Google Scholar] [CrossRef]
- McCoull, W.; Davies, F.A. Recent Synthetic Applications of Chiral Aziridines. Synthesis 2000, 10, 1347–1365. [Google Scholar] [CrossRef]
- Braga, A.L.; Paixao, M.W.; Westermann, B.; Schneider, P.H.; Wessjohann, L.A. Aziridine-Modified Amino Alcohols as Efficient Modular Catalysts for Highly Enantioselective Alkenylzinc Additions to Aldehydes. Synlett 2007, 6, 917–920. [Google Scholar] [CrossRef]
- Jarzyński, S.; Utecht, G.; Leśniak, S.; Rachwalski, M. Highly enantioselective asymmetric reactions involving zinc ions promoted by chiral aziridine alcohols. Tetrahedron Asymmetry 2017, 28, 1774–1779. [Google Scholar] [CrossRef]
- Wujkowska, Z.; Jarzyński, S.; Pieczonka, A.M.; Leśniak, S.; Rachwalski, M. Highly enantioselective addition of arylzinc reagents to aldehydes promoted by chiral aziridine alcohols. Tetrahedron Asymmetry 2016, 27, 1238–1244. [Google Scholar] [CrossRef]
- Jarzyński, S.; Leśniak, S.; Pieczonka, A.M.; Rachwalski, M. N-Trityl-aziridinyl alcohols as highly efficient chiral catalysts in asymmetric additions of organozinc species to aldehydes. Tetrahedron Asymmetry 2015, 26, 35–40. [Google Scholar] [CrossRef]
- Braga, A.L.; Milani, P.; Paixao, M.W.; Zeni, G.; Rodrigues, O.E.D.; Alves, E.F. Aziridine sulfides and disulfides as catalysts for the enantioselective addition of diethylzinc to aldehydes. Chem. Comm. 2004, 21, 2488–2489. [Google Scholar] [CrossRef] [PubMed]
- Jarzyński, S.; Rachwalski, M.; Pieczonka, A.M.; Wujkowska, Z.; Leśniak, S. Highly efficient conjugate additions of diethylzinc to enones promoted by chiral aziridine alcohols and aziridine ethers. Tetrahedron Asymmetry 2015, 26, 924–927. [Google Scholar] [CrossRef]
- Buchcic, A.; Zawisza, A.; Leśniak, S.; Rachwalski, M. Asymmetric Friedel-Crafts Alkylation of Indoles Catalyzed by Chiral Aziridine-Phosphines. Catalysts 2020, 10, 971. [Google Scholar] [CrossRef]
- Braga, A.L.; Paixao, M.W.; Milani, P.; Silveira, C.C.; Rodrigues, O.E.D.; Alves, E.F. New Aziridine Sulfide Ligands for Palladium-Catalyzed Asymmetric Allylic Alkylation. Synlett 2004, 7, 1297–1299. [Google Scholar] [CrossRef]
- Niu, J.-L.; Wang, M.-C.; Kong, P.-P.; Chen, Q.-T.; Zhu, Y.; Song, M.-P. Origin of enantioselectivity with heterobidentate sulfide-tertiary amine (sp3) ligands in palladium-catalyzed allylic substitution. Tetrahedron 2009, 65, 8869–8878. [Google Scholar] [CrossRef]
- Borges, R.; Andrade, F.C.D.; Schwab, R.S.; Sousa, F.S.S.; de Souza, M.N.; Savegnago, L.; Schneider, P.H. Straightforward synthesis and antioxidant studies of chalcogenoaziridines. Benzeneperoxyseleninic Acids—Synthesis and Properties. Tetrahedron 2016, 57, 3501–3504. [Google Scholar] [CrossRef]
- Zielińska-Błajet, M.; Rewucki, P.; Walenczak, S. Sulfur-containing derivatives from (1R)-(-)-myrtenal designed as chiral ligands. Tetrahedron 2016, 72, 3851–3857. [Google Scholar] [CrossRef]
- Zielińska-Błajet, M.; Siedlecka, R.; Skarżewski, J. Chiral phenylselenyl derivatives of pyrrolidine and Cinchona alkaloids: Nitrogen-selenium donating ligands in palladium-catalyzed asymmetric allylic alkylation. Tetrahedron Asymmetry 2007, 18, 131–136. [Google Scholar] [CrossRef]
- Wojaczyńska, E.; Zielińska-Błajet, M.; Turowska-Tyrk, I.; Skarżewski, J. Sulfoxides derived from Cinchona alkaloids—Chiral ligands in palladium-catalyzed asymmetric allylic alkylation. Tetrahedron Asymmetry 2010, 21, 853–858. [Google Scholar] [CrossRef]
- Huang, Z.; Lai, H.; Qin, Y. Syntheses of Novel Chiral Sulfinamido Ligands and Their Applications in Diethylzinc Additions to Aldehydes. J. Org. Chem. 2007, 72, 1373–1378. [Google Scholar] [CrossRef] [PubMed]
- Prause, F.; Wagner, S.; Breuning, M. Enantioselective addition of diethylzinc to aldehydes catalyzed by 5-cis-substituted proline derivatives. Tetrahedron 2019, 75, 94–101. [Google Scholar] [CrossRef]
- Raji, M.; Le, T.M.; Fülöp, F.; Szakonyi, Z. Synthesis and Investigation of Pinane-Based Chiral Tridentate Ligands in the Asymmetric Addition of Diethylzinc to Aldehydes. Catalysts 2020, 10, 474. [Google Scholar] [CrossRef]
- Obieziurska, M.; Pacuła, A.J.; Laskowska, A.; Długosz-Pokorska, A.; Janecka, A.; Ścianowski, J. Seleninic Acid Potassium Salts as Water-Soluble Biocatalysts with Enhanced Bioavailability. Materials 2020, 13, 661. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Van Vrankel, D.L. Asymmetric Transition Metal-Catalyzed Allylic Alkylations. Chem. Rev. 1996, 96, 395–422. [Google Scholar] [CrossRef]
- Wang, M.-C.; Zhang, Q.-J.; Zhao, W.-X.; Wang, X.-D.; Ding, X.; Jing, T.-T.; Song, M.-P. Evaluation of Enantiopure N-(Ferrocenylmethyl)azetidin-2-yl(diphenyl)methanol for Catalytic Asymmetric Addition of Organozinc Reagents to Aldehydes. J. Org. Chem. 2008, 73, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.-X.; Wu, J.; Au-Yeung, T.T.-L.; Yip, C.-W.; Haynes, R.K.; Chan, A.S.C. Highly Enantioselective Phenyl Transfer to Aryl Aldehydes Catalyzed by Easily Accessible Chiral Tertiary Aminonaphthol. J. Org. Chem. 2005, 70, 1093–1095. [Google Scholar] [CrossRef] [PubMed]


| Base | Solvent | T (PhSM Formation) | T (Substitution Reaction) | Time | Product |
|---|---|---|---|---|---|
| LiOH | THF/EtOH (3:1) | r.t. | 120 °C | 24 h | 14 |
| LiOH | THF/EtOH (3:1) | r.t. | 100 °C | 24 h | 14 |
| LiOH | THF/EtOH (3:1) | r.t. | 100 °C | 1 h | 14 |
| LiOH | THF/EtOH (3:1) | r.t. | 70 °C | 24 h | 15 (25%) |
| LiOH | THF/EtOH (3:1) | 70 °C | 70 °C | 24 h | 15 (28%) |
| LiOH | THF | r.t. | 70 °C | 24 h | 15 (5%) |
| NaOH | THF/EtOH (3:1) | r.t. | 70 °C | 24 h | 15 (8%) |
| NaH | THF/EtOH (3:1) | r.t. | 70 °C | 24 h | - |
| NaH | THF | r.t. | 70 °C | 24 h | - |
| NaH | DMSO | r.t. | 70 °C | 24 h | - |
| LiOH | THF/EtOH (3:1), 5% H2O | 70 °C | 70 °C | 24 h | 15 (40%) |
| LiOH | THF/EtOH (3:1), 10% H2O | 70 °C | 70 °C | 24 h | 15 (79%) |
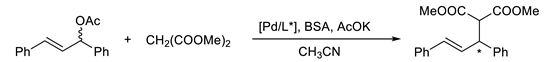
| Ligand | Yield (%) | R/Sb |
|---|---|---|
| 15 | 67 | 69:31 |
| 16 | 92 | 64:36 |
| 17 | 87 | 63:37 |
| 18 | 72 | 57:43 |
| 19 | 70 | 52:48 |
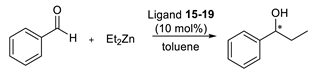
| Ligand | Enantiomer Ratio (a) | Conversion [%] (b) |
|---|---|---|
| 15 | 48.5:51.5 | 98 |
| 16 | 48.6:51.4 | 52 |
| 17 | 52.0:48.0 | 14 |
| 18 | 49.7:50.3 | 26 |
| 19 | 94.2:5.8 | 90 |
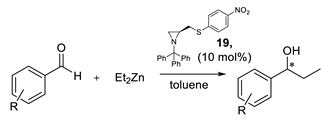
| R | Enantiomer Ratio (a) | Conversion [%] (b) |
|---|---|---|
| H | 94.2:5.8 | 90 |
| 4-OCH3 | 93.1:6.9 | 63 |
| 2-OCH3 | 88.0:12.0 | 93 |
| 2-CH3 | 73.0:27.0 | 63 |
| 2-F | 94.0:6.0 | 93 |
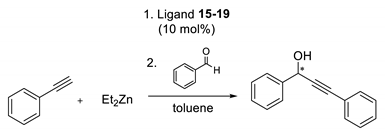
| Ligand | Enantiomer Ratio (b) | Yield (%) (a) |
|---|---|---|
| 15 | 51.0:49.0 | 98 |
| 16 | 51.0:49.0 | 84 |
| 17 | 50.0:50.0 | 44 |
| 18 | 52.0:48.0 | 96 |
| 19 | 57.0:43.0 | 86 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacuła-Miszewska, A.J.; Laskowska, A.; Kmieciak, A.; Zielińska-Błajet, M.; Krzemiński, M.P.; Ścianowski, J. Chiral Aziridine Sulfide N(sp3),S-Ligands for Metal-Catalyzed Asymmetric Reactions. Symmetry 2021, 13, 502. https://doi.org/10.3390/sym13030502
Pacuła-Miszewska AJ, Laskowska A, Kmieciak A, Zielińska-Błajet M, Krzemiński MP, Ścianowski J. Chiral Aziridine Sulfide N(sp3),S-Ligands for Metal-Catalyzed Asymmetric Reactions. Symmetry. 2021; 13(3):502. https://doi.org/10.3390/sym13030502
Chicago/Turabian StylePacuła-Miszewska, Agata J., Anna Laskowska, Anna Kmieciak, Mariola Zielińska-Błajet, Marek P. Krzemiński, and Jacek Ścianowski. 2021. "Chiral Aziridine Sulfide N(sp3),S-Ligands for Metal-Catalyzed Asymmetric Reactions" Symmetry 13, no. 3: 502. https://doi.org/10.3390/sym13030502
APA StylePacuła-Miszewska, A. J., Laskowska, A., Kmieciak, A., Zielińska-Błajet, M., Krzemiński, M. P., & Ścianowski, J. (2021). Chiral Aziridine Sulfide N(sp3),S-Ligands for Metal-Catalyzed Asymmetric Reactions. Symmetry, 13(3), 502. https://doi.org/10.3390/sym13030502









