EEG Resting Asymmetries and Frequency Oscillations in Approach/Avoidance Personality Traits: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Strategies
- Asymmetry and brain activity: “EEG asymmetry” OR left OR right OR lateral* OR front* OR posterior OR prefrontal OR parietal* OR electroenceph* OR oscill* OR rhythms OR coupling OR “frequency oscillations” OR synchronization OR desynchronization OR alpha OR delta OR theta OR beta OR gamma AND rest*;
- Approach/avoidance motivation: “approach motivation” OR motivation* OR approach* OR BAS OR reward* OR “positive affect” OR “avoidance motivation” OR avoid* OR “negative affect” OR BIS OR withdraw* OR inhibit* OR threat* OR fear OR FFS OR FFFS.
2.2. Eligibility Criteria
2.3. Data Collection and Quality Assessment
- (1)
- Adequacy of the criteria adopted for the inclusion of participants in the sample (absence of clinical psychological disorders or other diseases, suspension of drug or psychotropic substances in case used);
- (2)
- Sample and setting characteristics (mean age and standard deviation, gender, education, and handedness);
- (3)
- Methodological criteria used for the electrophysiological measures (open or closed eyes recording, reference, length, counterbalance between open/closed eyes order and participants);
- (4)
- Occurrence of contextual or interactional variables not considered in the study that could constitute confounding factors (time of day and season of EEG recording, measures of mood state before and during EEG recording, menstrual cycle for women, and gender of experimenters);
- (5)
- Reliability of electrophysiological measures (test–retest sessions of EEG recording);
- (6)
- Adequacy of the statistical analysis used (including the strategies to deal with confounding factors considered in the study).
3. Results
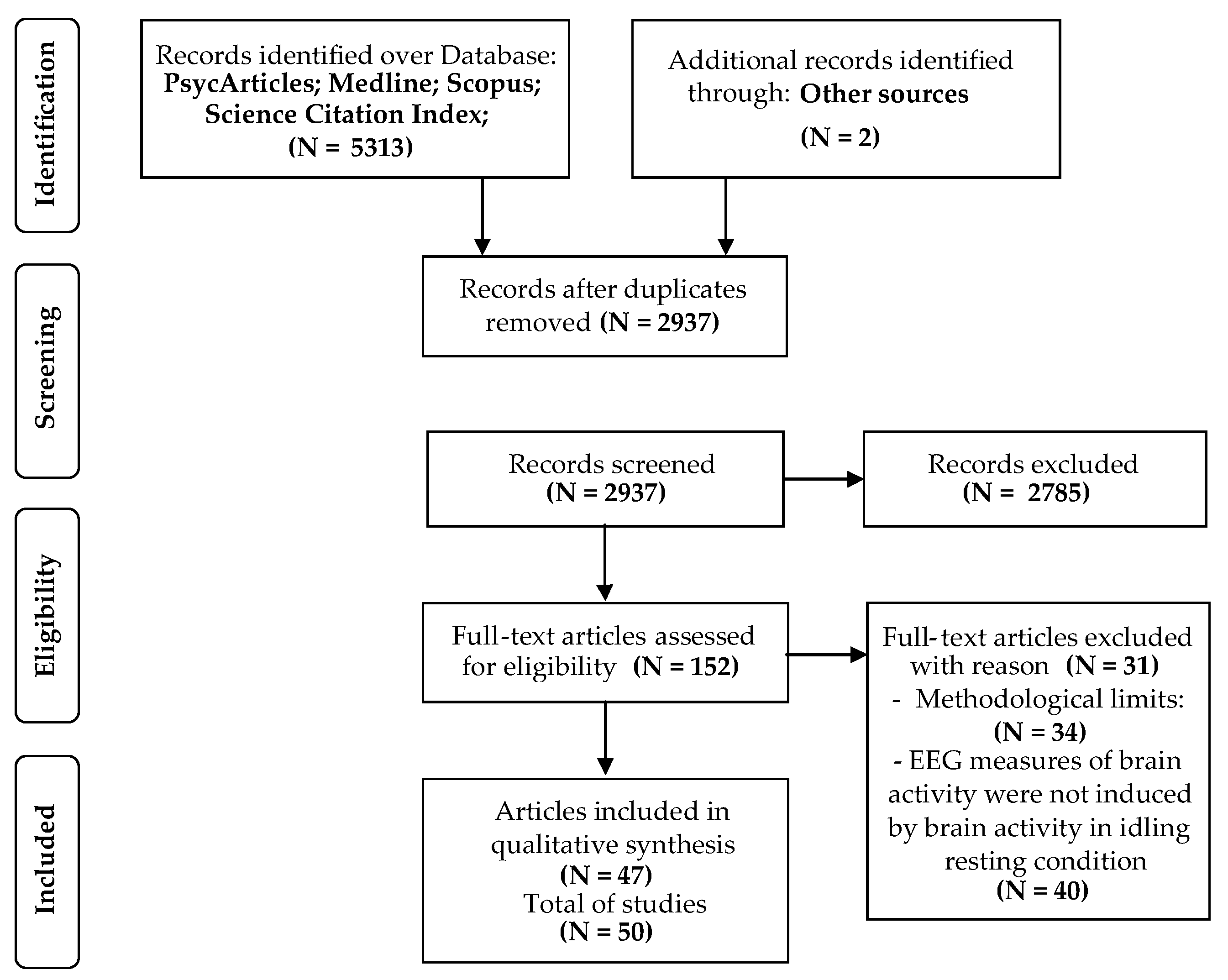
3.1. Studies Selection
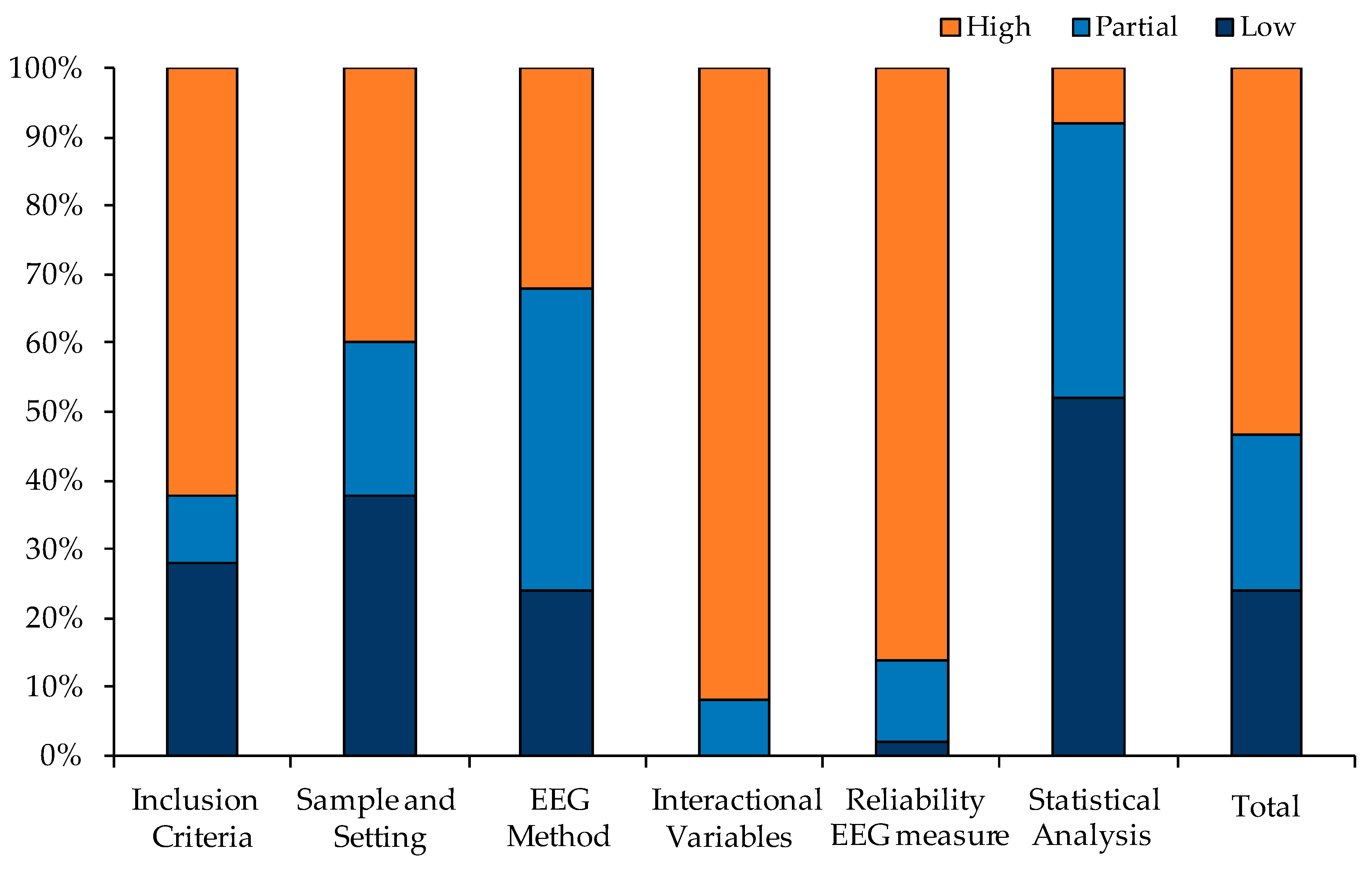
3.2. Quality Assessment
3.3. Demographic Features
3.4. Approach/Avoidance Personality Traits and Electrocortical Measures
3.5. Approach/Avoidance Personality Traits and EEG Gamma Rhythm
3.6. Approach/Avoidance Personality Traits and EEG Beta Rhythm
3.7. Approach/Avoidance Personality Trait, Interactional Variables, and EEG Alpha Rhythm
3.7.1. Approach/Avoidance Personality Traits and EEG Alpha Rhythm
3.7.2. Interactional and Contextual Variables in the Relationship between Personality Traits and EEG Alpha Asymmetry
3.8. Approach/Avoidance Personality Traits and EEG Theta Rhythm
3.9. Approach/Avoidance Personality Traits and EEG Delta Rhythm
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Başar, E.; Schürmann, M.; Demiralp, T.; Başar-Eroglu, C.; Ademoglu, A. Event-related oscillations are ‘real brain responses’—Wavelet analysis and new strategies. Int. J. Psychophysiol. 2001, 39, 91–127. [Google Scholar] [CrossRef]
- Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res. Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Davidson, R.J. Anterior cerebral asymmetry and the nature of emotion. Brain Cogn. 1992, 20, 125–151. [Google Scholar] [CrossRef]
- Harmon-Jones, E. Clarifying the emotive functions of asymmetrical frontal cortical activity. Psychophysiology 2003, 40, 838–848. [Google Scholar] [CrossRef] [PubMed]
- Harmon-Jones, E.; Gable, P.A. On the role of asymmetric frontal cortical activity in approach and withdrawal motivation: An updated review of the evidence. Psychophysiology 2018, 55, e12879. [Google Scholar] [CrossRef] [PubMed]
- Näpflin, M.; Wildi, M.; Sarnthein, J. Test–retest reliability of resting EEG spectra validates a statistical signature of persons. Clin. Neurophysiol. 2007, 118, 2519–2524. [Google Scholar] [CrossRef] [PubMed]
- Kuper, N.; Käckenmester, W.; Wacker, J. Resting frontal EEG asymmetry and personality traits: A meta-analysis. Eur. J. Pers. 2019, 33, 154–175. [Google Scholar] [CrossRef]
- Schutter, D.J.; de Weijer, A.D.; Meuwese, J.D.; Morgan, B.; van Honk, J. Interrelations between motivational stance, cortical excitability, and the frontal electroencephalogram asymmetry of emotion: A transcranial magnetic stimulation study. Hum. Brain Mapp. 2008, 29, 574–580. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 6, e1000100. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Knyazev, G.G.; Savostyanov, A.N.; Levin, E.A. Alpha oscillations as a correlate of trait anxiety. Int. J. Psychophysiol. 2004, 53, 147–160. [Google Scholar] [CrossRef]
- Beaton, E.A.; Schmidt, L.A.; Ashbaugh, A.R.; Santesso, D.L.; Antony, M.M.; McCabe, R.E. Resting and reactive frontal brain electrical activity (EEG) among a non-clinical sample of socially anxious adults: Does concurrent depressive mood matter? Neuropsychiatr. Dis. Treat. 2008, 4, 187. [Google Scholar]
- Hall, E.E.; Ekkekakis, P.; Petruzzello, S.J. Predicting affective responses to exercise using resting EEG frontal asymmetry: Does intensity matter? Biol. Psychol. 2010, 83, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Keune, P.M.; Bostanov, V.; Kotchoubey, B.; Hautzinger, M. Mindfulness versus rumination and behavioral inhibition: A perspective from research on frontal brain asymmetry. Pers. Individ. Differ. 2012, 53, 323–328. [Google Scholar] [CrossRef]
- Pérez-Edgar, K.; Kujawa, A.; Nelson, S.K.; Cole, C.; Zapp, D.J. The relation between electroencephalogram asymmetry and attention biases to threat at baseline and under stress. Brain Cogn. 2013, 82, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, G.A.; Burgess, A.P. Hypnotic induction is followed by state-like changes in the organization of EEG functional connectivity in the theta and beta frequency bands in high-hypnotically susceptible individuals. Front. Hum. Neurosci. 2014, 8, 528. [Google Scholar] [CrossRef]
- Käckenmester, W.; Kroencke, L.; Wacker, J. Frontal asymmetry predicts the incentive value of perceptual information. Int. J. Psychophysiol. 2018, 134, 22–29. [Google Scholar] [CrossRef]
- Adolph, D.; von Glischinski, M.; Wannemüller, A.; Margraf, J. The influence of frontal alpha-asymmetry on the processing of approach-and withdrawal-related stimuli—A multichannel psychophysiology study. Psychophysiology 2017, 54, 1295–1310. [Google Scholar] [CrossRef]
- Massar, S.A.A.; Rossi, V.; Schutter, D.J.L.G.; Kenemans, J.L. Baseline EEG theta/beta ratio and punishment sensitivity as biomarkers for feedback-related negativity (FRN) and risk-taking. Clin. Neurophysiol. 2012, 123, 1958–1965. [Google Scholar] [CrossRef] [PubMed]
- Hagemann, D.; Naumann, E.; Thayer, J.F.; Bartussek, D. Does resting electroencephalograph asymmetry reflect a trait? An application of latent state-trait theory. J. Pers. Soc. Psychol. 2002, 82, 619. [Google Scholar] [CrossRef]
- Harmon-Jones, E.; Allen, J.J. Anger and frontal brain activity: EEG asymmetry consistent with approach motivation despite negative affective valence. J. Pers. Soc. Psychol. 1998, 74, 1310. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Jadad, A.R. Blind assessment of the quality of trial reports. Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Institute, J.B. The Joanna Briggs Institute Critical Appraisal Tools for Use in JBI Systematic Reviews Checklist for Analytical Cross Sectional Studies; The Joanna Briggs Institute: North Adelaide, Australia, 2017. [Google Scholar]
- Hagemann, D. Individual differences in anterior EEG asymmetry: Methodological problems and solutions. Biol. Psychol. 2004, 67, 157–182. [Google Scholar] [CrossRef]
- Tomarken, A.J.; Davidson, R.J.; Wheeler, R.E.; Doss, R.C. Individual differences in anterior brain asymmetry and fundamental dimensions of emotion. J. Pers. Soc. Psychol. 1992, 62, 676. [Google Scholar] [CrossRef]
- Tomarken, A.J.; Davidson, R.J. Frontal brain activation in repressors and nonrepressors. J. Abnorm. Psychol. 1994, 103, 339. [Google Scholar] [CrossRef] [PubMed]
- Sutton, S.K.; Davidson, R.J. Prefrontal brain asymmetry: A biological substrate of the behavioral approach and inhibition systems. Psychol. Sci. 1997, 8, 204–210. [Google Scholar] [CrossRef]
- Hagemann, D.; Naumann, E.; Lürken, A.; Becker, G.; Maier, S.; Bartussek, D. EEG asymmetry, dispositional mood and personality. Pers. Individ. Differ. 1999, 27, 541–568. [Google Scholar] [CrossRef]
- Kline, J.P.; Knapp-Kline, K.; Schwartz, G.E.; Russek, L.G. Anterior asymmetry, defensiveness, and perceptions of parental caring. Pers. Individ. Differ. 2001, 31, 1135–1145. [Google Scholar] [CrossRef]
- Kline, J.P.; Blackhart, G.C.; Joiner, T.E. Sex, lie scales, and electrode caps: An interpersonal context for defensiveness and anterior electroencephalographic asymmetry. Pers. Individ. Differ. 2002, 33, 459–478. [Google Scholar] [CrossRef]
- Blackhart, G.C.; Kline, J.P.; Donohue, K.F.; LaRowe, S.D.; Joiner, T.E. Affective responses to EEG preparation and their link to resting anterior EEG asymmetry. Pers. Individ. Differ. 2002, 32, 167–174. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J. Frontal EEG asymmetry and the behavioral activation and inhibition systems. Psychophysiology 2003, 40, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Knyazev, G.G.; Slobodskaya, H.R. Personality trait of behavioral inhibition is associated with oscillatory systems reciprocal relationships. Int. J. Psychophysiol. 2003, 48, 247–261. [Google Scholar] [CrossRef]
- Razoumnikova, O. Interaction of personality and intelligence factors in cortex activity modulation. Pers. Individ. Differ. 2003, 35, 135–162. [Google Scholar] [CrossRef]
- Minnix, J.A.; Kline, J.P. Neuroticism predicts resting frontal EEG asymmetry variability. Pers. Individ. Differ. 2004, 36, 823–832. [Google Scholar] [CrossRef]
- Hewig, J.; Hagemann, D.; Seifert, J.; Naumann, E.; Bartussek, D. The relation of cortical activity and BIS/BAS on the trait level. Biol. Psychol. 2006, 71, 42–53. [Google Scholar] [CrossRef]
- Tran, Y.; Craig, A.; Boord, P.; Connell, K.; Cooper, N.; Gordon, E. Personality traits and its association with resting regional brain activity. Int. J. Psychophysiol. 2006, 60, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Jaušovec, N.; Jaušovec, K. Personality, gender and brain oscillations. Int. J. Psychophysiol. 2007, 66, 215–224. [Google Scholar] [CrossRef]
- Smit, D.J.A.; Posthuma, D.; Boomsma, D.I.; De Geus, E.J.C. The relation between frontal EEG asymmetry and the risk for anxiety and depression. Biol. Psychol. 2007, 74, 26–33. [Google Scholar] [CrossRef]
- Fleck, J.I.; Green, D.L.; Stevenson, J.L.; Payne, L.; Bowden, E.M.; Jung-Beeman, M.; Kounios, J. The transliminal brain at rest: Baseline EEG, unusual experiences, and access to unconscious mental activity. Cortex 2008, 44, 1353–1363. [Google Scholar] [CrossRef]
- Gatt, J.M.; Kuan, S.A.; Dobson-Stone, C.; Paul, R.H.; Joffe, R.T.; Kemp, A.H.; Williams, L.M. Association between BDNF Val66Met polymorphism and trait depression is mediated via resting EEG alpha band activity. Biol. Psychol. 2008, 79, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Kline, J.P.; Allen, S. The failed repressor: EEG asymmetry as a moderator of the relation between defensiveness and depressive symptoms. Int. J. Psychophysiol. 2008, 68, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Mathersul, D.; Williams, L.M.; Hopkinson, P.J.; Kemp, A.H. Investigating models of affect: Relationships among EEG alpha asymmetry, depression, and anxiety. Emotion 2008, 8, 560. [Google Scholar] [CrossRef]
- Santesso, D.L.; Segalowitz, S.J.; Ashbaugh, A.R.; Antony, M.M.; McCabe, R.E.; Schmidt, L.A. Frontal EEG asymmetry and sensation seeking in young adults. Biol. Psychol. 2008, 78, 164–172. [Google Scholar] [CrossRef]
- Master, S.L.; Amodio, D.M.; Stanton, A.L.; Yee, C.M.; Hilmert, C.J.; Taylor, S.E. Neurobiological correlates of coping through emotional approach. Brain Behav. Immun. 2009, 23, 27–35. [Google Scholar] [CrossRef]
- Pavlenko, V.B.; Chernyi, S.V.; Goubkina, D.G. EEG correlates of anxiety and emotional stability in adult healthy subjects. Neurophysiology 2009, 41, 337–345. [Google Scholar] [CrossRef]
- Peterson, C.K.; Harmon-Jones, E. Circadian and seasonal variability of resting frontal EEG asymmetry. Biol. Psychol. 2009, 80, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Knyazev, G.G.; Slobodskoi-Plyusnin, Y.Y.; Savost’yanov, A.N.; Levin, E.A.; Bocharov, A.V. Reciprocal relationships between the oscillatory systems of the brain. Neurosci. Behav. Physiol. 2010, 40, 29. [Google Scholar] [CrossRef]
- Mikolajczak, M.; Bodarwé, K.; Laloyaux, O.; Hansenne, M.; Nelis, D. Association between frontal EEG asymmetries and emotional intelligence among adults. Pers. Individ. Differ. 2010, 48, 177–181. [Google Scholar] [CrossRef]
- Wacker, J.; Gatt, J.M. Resting posterior versus frontal delta/theta EEG activity is associated with extraversion and the COMT VAL158MET polymorphism. Neurosci. Lett. 2010, 478, 88–92. [Google Scholar] [CrossRef]
- Wacker, J.; Chavanon, M.L.; Stemmler, G. Resting EEG signatures of agentic extraversion: New results and meta-analytic integration. J. Res. Pers. 2010, 44, 167–179. [Google Scholar] [CrossRef]
- Chavanon, M.L.; Wacker, J.; Stemmler, G. Rostral anterior cingulate activity generates posterior versus anterior theta activity linked to agentic extraversion. Cogn. Affect. Behav. Neurosci. 2011, 11, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Koehler, S.; Wacker, J.; Odorfer, T.; Reif, A.; Gallinat, J.; Fallgatter, A.J.; Herrmann, M.J. Resting posterior minus frontal EEG slow oscillations is associated with extraversion and DRD2 genotype. Biol. Psychol. 2011, 87, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Konareva, I.N. Modulation of low-frequency EEG rhythms under conditions of an activation reaction: Dependence on psychological characteristics of personality. Neurophysiology 2011, 42, 434–445. [Google Scholar] [CrossRef]
- Knyazev, G.G.; Bocharov, A.V.; Pylkova, L.V. Extraversion and fronto-posterior EEG spectral power gradient: An independent component analysis. Biol. Psychol. 2012, 89, 515–524. [Google Scholar] [CrossRef] [PubMed]
- De Pascalis, V.; Cozzuto, G.; Caprara, G.V.; Alessandri, G. Relations among EEG-alpha asymmetry, BIS/BAS, and dispositional optimism. Biol. Psychol. 2013, 94, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Alessandri, G.; Caprara, G.V.; De Pascalis, V. Relations among EEG-alpha asymmetry and positivity personality trait. Brain Cogn. 2015, 97, 10–21. [Google Scholar] [CrossRef]
- Gable, P.A.; Mechin, N.C.; Hicks, J.A.; Adams, D.L. Supervisory control system and frontal asymmetry: Neurophysiological traits of emotion-based impulsivity. Soc. Cogn. Affect. Neurosci. 2015, 10, 1310–1315. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, R.; Cui, H.; Wu, M.; Wang, Q.; Zhao, Y.; Liu, Y. Variations in resting frontal alpha asymmetry between high-and low-neuroticism females across the menstrual cycle. Psychophysiology 2015, 52, 182–191. [Google Scholar] [CrossRef]
- Korjus, K.; Uusberg, A.; Uusberg, H.; Kuldkepp, N.; Kreegipuu, K.; Allik, J.; Aru, J. Personality cannot be predicted from the power of resting state EEG. Front. Hum. Neurosci. 2015, 9, 63. [Google Scholar] [CrossRef]
- Tullett, A.M.; Wildschut, T.; Sedikides, C.; Inzlicht, M. Right-frontal cortical asymmetry predicts increased proneness to nostalgia. Psychophysiology 2015, 52, 990–996. [Google Scholar] [CrossRef]
- Adolph, D.; Margraf, J. The differential relationship between trait anxiety, depression, and resting frontal α-asymmetry. J. Neural Transm. 2017, 124, 379–386. [Google Scholar] [CrossRef]
- Neal, L.B.; Gable, P.A. Regulatory control and impulsivity relate to resting frontal activity. Soc. Cogn. Affect. Neurosci. 2017, 12, 1377–1383. [Google Scholar] [CrossRef]
- De Groot, K.; Van Strien, J.W. Spontaneous resting-state gamma oscillations are not predictive of autistic traits in the general population. Eur. J. Neurosci. 2018, 48, 2928–2937. [Google Scholar] [CrossRef]
- De Pascalis, V.; Sommer, K.; Scacchia, P. Resting frontal asymmetry and reward sensitivity theory motivational traits. Sci. Rep. 2018, 8, 13154. [Google Scholar] [CrossRef]
- Threadgill, A.H.; Gable, P.A. Resting beta activation and trait motivation: Neurophysiological markers of motivated motor-action preparation. Int. J. Psychophysiol. 2018, 127, 46–51. [Google Scholar] [CrossRef]
- Paban, V.; Modolo, J.; Mheich, A.; Hassan, M. Psychological resilience correlates with EEG source-space brain network flexibility. Network Neurosci. 2019, 3, 539–550. [Google Scholar] [CrossRef]
- De Pascalis, V.; Vecchio, A.; Cirillo, G. Resting anxiety increases EEG delta–beta correlation: Relationships with the Reinforcement Sensitivity Theory Personality traits. Pers. Individ. Differ. 2020, 156, 109796. [Google Scholar] [CrossRef]
- Zanesco, A.P.; King, B.G.; Skwara, A.C.; Saron, C.D. Within and between-person correlates of the temporal dynamics of resting EEG microstates. NeuroImage 2020, 211, 116631. [Google Scholar] [CrossRef]
- Zhang, J.; Hua, Y.; Xiu, L.; Oei, T.P.; Hu, P. Resting state frontal alpha asymmetry predicts emotion regulation difficulties in impulse control. Pers. Individ. Differ. 2020, 159, 109870. [Google Scholar] [CrossRef]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Chapman, L.J.; Chapman, J.P. The measurement of handedness. Brain Cogn. 1987, 6, 175–183. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Wheelwright, S.; Skinner, R.; Martin, J.; Clubley, E. The autism-spectrum quotient (AQ): Evidence from asperger syndrome/high-functioning autism, malesand females, scientists and mathematicians. J. Autism Dev. Disord. 2001, 31, 5–17. [Google Scholar] [CrossRef]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 1988, 54, 1063. [Google Scholar] [CrossRef]
- Larsen, R.J.; Diener, E.; Cropanzano, R.S. Cognitive operations associated with individual differences in affect intensity. J. Pers. Soc. Psychol. 1987, 53, 767. [Google Scholar] [CrossRef]
- Crowne, D.P.; Marlowe, D. Social Desirability Scale; John Wiley: Hoboken, NJ, USA, 1964. [Google Scholar]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.E. State-Trait Anxiety Inventory Manual; Mind Garden, Inc.: Palo Alto, CA, USA, 1970. [Google Scholar]
- Bendig, A.W. The development of a short form of the Manifest Anxiety Scale. J. Consult. Clin. Psychol. 1956, 20, 384. [Google Scholar] [CrossRef]
- Beck, A.T.; Ward, C.; Mendelson, M.; Mock, J.; Erbaugh, J. Beck depression inventory (BDI). Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Von Zerssen, D.; Koeller, D.M. Depressivitätsskala: Manual [Depression Scale: Manual]; Weinheim: Göttingen, Germany, 1976. [Google Scholar]
- Carver, C.S.; White, T.L. Behavioral inhibition, behavioral activation, and affective responses to impending reward and punishment: The BIS/BAS scales. J. Pers. Soc. Psychol. 1994, 67, 319. [Google Scholar] [CrossRef]
- Hartig, J.; Moosbrugger, H. Die “ARES-Skalen” zurErfassung der individuellen BIS-und BAS-Sensitivität. ZDDP 2003, 24, 293–310. [Google Scholar]
- Rosenberg, M. Rosenberg self-esteem scale (RSE). Accept. Commit. Ther. Meas. Package 1965, 61, 18. [Google Scholar]
- Eysenck, H.J.; SBG, E. Eysenck Personality Questionnaire-Junior (EPQ-J) & Adult (EPQ-A); Hodder and Stoughton Educational: London, UK, 1975. [Google Scholar]
- Russek, L.G.; Schwartz, G.E. Perceptions of parental caring predict health status in midlife: A 35-year follow-up of the Harvard Mastery of Stress Study. Psychosom. Med. 1997, 59, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Mehrabian, A.; Russell, J.A. An Approach to Environmental Psychology; The MIT Press: Cambridge, MA, USA, 1974. [Google Scholar]
- Wilson, G.D.; Gray, J.A.; Barrett, P.T. A factor analysis of the Gray-Wilson personality questionnaire. Pers. Individ. Differ. 1990, 11, 1037–1044. [Google Scholar] [CrossRef]
- Corr, P.J.; Cooper, A.J. The Reinforcement Sensitivity Theory of Personality Questionnaire (RST-PQ): Development and validation. Psychol. Assess. 2016, 28, 1427. [Google Scholar] [CrossRef]
- Whiteside, S.P.; Lynam, D.R.; Miller, J.D.; Reynolds, S.K. Validation of the UPPS impulsive behaviour scale: A four-factor model of impulsivity. Eur. J. Pers. 2005, 19, 559–574. [Google Scholar] [CrossRef]
- Ovchinnikov, B.V.; Pavlov, K.V.; Vladimirova, I.M. Vashpsikhologicheskiy Tip [Your Psychological Type]; Andreev Isynov’ya Publ.: St. Petersburg, Russia, 1994. [Google Scholar]
- Diener, E.D.; Emmons, R.A.; Larsen, R.J.; Griffin, S. The satisfaction with life scale. J. Pers. Assess. 1985, 49, 71–75. [Google Scholar] [CrossRef]
- Amthauer, R. Intelligenz-Struktur-Test.: IST 70; Verlag für Psychologie Hogrefe: Göttingen, Germany, 1970. [Google Scholar]
- Costa, P.T.; McCrae, R.R. Normal personality assessment in clinical practice: The NEO Personality Inventory. J. Pers. Assess. 1992, 4, 5. [Google Scholar] [CrossRef]
- Costa, P.T.; McCrae, R.R. Revised NEO Personality Inventory (NEO-PI-R) and Neo Five-Factor Inventory (NEO-FFI); Psychological Assessment Resources: Odessa, FL, USA, 1992. [Google Scholar]
- John, O.P.; Naumann, L.P.; Soto, C.J. Handbook of Personality: Theory and Research, 3rd ed.; Guilford Press: New York, NY, USA, 2008; pp. 114–158. [Google Scholar]
- Batcho, K.I. Nostalgia: A psychological perspective. Percept. Mot. Ski. 1995, 80, 131–143. [Google Scholar] [CrossRef]
- Barrett, F.S.; Grimm, K.J.; Robins, R.W.; Wildschut, T.; Sedikides, C.; Janata, P. Music-evoked nostalgia: Affect, memory, and personality. Emotion 2010, 10, 390–403. [Google Scholar] [CrossRef]
- Mayer, J.D.; Salovey, P.; Caruso, D.R. Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT) Item Booklet; University of New Hampshire: Durham, NC, USA, 2002. [Google Scholar]
- Achenbach, T.M. Conceptualization of developmental psychopathology. In Handbook of Developmental Psychopathology; Springer: Boston, MA, USA, 1990; pp. 3–14. [Google Scholar]
- De Wilde, G.J.S. Nenrotische Labiliteit, Gemeten Volgens de Vragenlijst Methode [Neuroticism, Measured by Means of a Self Report Inventory]; Van Rossen: Amsterdam, The Netherlands, 1970. [Google Scholar]
- Lange, R.; Thalbourne, M.A.; Houran, J.; Storm, L. The Revised Transliminality Scale: Reliability and validity data from a Rasch top-down purification procedure. Conscious. Cogn. 2000, 9, 591–617. [Google Scholar] [CrossRef]
- Connor, K.M.; Davidson, J.R. Development of a new resilience scale: The Connor-Davidson resilience scale (CD-RISC). Depress. Anxiety 2003, 18, 76–82. [Google Scholar] [CrossRef]
- Lovibond, P.F.; Lovibond, S.H. The structure of negative emotional states: Comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behav. Res. Ther. 1995, 33, 335–343. [Google Scholar] [CrossRef]
- Gratz, K.L.; Roemer, L. Multidimensional assessment of emotion regulation and dysregulation: Development, factor structure, and initial validation of the difficulties in emotion regulation scale. J. Psychopathol. Behav. Assess. 2004, 26, 41–54. [Google Scholar] [CrossRef]
- Scheier, M.F.; Carver, C.S.; Bridges, M.W. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): A reevaluation of the Life Orientation Test. J. Pers. Soc. Psychol. 1994, 67, 1063. [Google Scholar] [CrossRef]
- Caprara, G.V.; Alessandri, G.; Eisenberg, N.; Kupfer, A.; Steca, P.; Caprara, M.G.; Abela, J. The positivity scale. Psychol. Assess. 2012, 24, 701. [Google Scholar] [CrossRef] [PubMed]
- Cyders, M.A.; Smith, G.T.; Spillane, N.S.; Fischer, S.; Annus, A.M.; Peterson, C. Integration of impulsivity and positive mood to predict risky behavior: Development and validation of a measure of positive urgency. Psychol. Assess. 2007, 19, 107. [Google Scholar] [CrossRef]
- Zuckerman, M. Behavioral Expressions and Biosocial Bases of Sensation Seeking; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Fromme, K.; Katz, E.C.; Rivet, K. Outcome expectancies and risk-taking behavior. Cogn. Ther. Res. 1997, 21, 421–442. [Google Scholar] [CrossRef]
- Stanton, A.L.; Danoff-Burg, S.; Cameron, C.L.; Ellis, A.P. Coping through emotional approach: Problems of conceptualizaton and confounding. J. Pers. Soc. Psychol. 1994, 66, 350. [Google Scholar] [CrossRef]
- Steyer, R.; Schwenkmezger, P.; Notz, P.; Eid, M. Multidimensional Mood State Questionnaire (MDBF); Hogrefe: Göttingen, Germany, 1997. [Google Scholar]
- Cattell, H.E.; Mead, A.D. The Sixteen Personality Factor Questionnaire (16PF). In The SAGE Handbook of Personality Theory and Assessment; Boyle, G.J., Matthews, G., Saklofske, D.H., Eds.; Personality Measurement and Testing; Sage Publications, Inc.: Thousand Oaks, CA, USA, 2008; Volume 2, pp. 135–159. [Google Scholar] [CrossRef]
- Ruch, W.; Angleitner, A.; Strelau, J. The Strelau Temperament Inventory—Revised (STI-R): Validity studies. Eur. J. Pers. 1991, 5, 287–308. [Google Scholar] [CrossRef]
- Rusalov, V.M. Object-related and communicative aspects of human temperament: A new questionnaire of the structure of temperament. Pers. Individ. Differ. 1989, 10, 817–827. [Google Scholar] [CrossRef]
- Wiggins, J.S.; Trapnell, P.; Phillips, N. Psychometric and geometric characteristics of the Revised Interpersonal Adjective Scales (IAS-R). Multivar. Behav. Res. 1988, 23, 517–530. [Google Scholar] [CrossRef]
- Petrides, K.V.; Pérez, J.C.; Furnham, A. The Trait Emotional Intelligence Questionnaire (TEIQue). A measure of emotional self-efficacy. In Proceedings of the XI Biennial Meeting of the International Society for the Study of the Individual Differences, Graz, Austria, 13 July 2003. [Google Scholar]
- McNaughton, N.; DeYoung, C.G.; Corr, P.J. Neuroimaging Personality, Social Cognition, and Character; Academic Press: Cambridge, MA, USA, 2016; pp. 25–49. [Google Scholar]
- Gray, J.A. A Model for Personality; Springer: Berlin/Heidelberg, Germany, 1981; pp. 246–276. [Google Scholar]
- Davidson, R.J. Anterior electrophysiological asymmetries, emotion, and depression: Conceptual and methodological conundrums. Psychophysiology 1998, 35, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.A. Explorations in Temperament; Springer: Boston, MA, USA, 1991; pp. 105–128. [Google Scholar]
- Davidson, R.J. Parsing affective space: Perspectives from neuropsychology and psychophysiology. Neuropsychology 1993, 7, 464. [Google Scholar] [CrossRef]
- Gray, J.A.; Nebylitsyn, V.D. Biological Bases of Individual Behavior; Academic Press: Cambridge, MA, USA, 1972; pp. 182–205. [Google Scholar]
- Gray, J.A. Brain systems that mediate both emotion and cognition. Cogn. Emot. 1990, 4, 269–288. [Google Scholar] [CrossRef]
- Corr, P.J. Reinforcement Sensitivity Theory (RST): Introduction. In The Reinforcement Sensitivity Theory of Personality; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar]
- Corr, P.J. Reinforcement sensitivity theory of personality questionnaires: Structural survey with recommendations. Pers. Individ. Differ. 2016, 89, 60–64. [Google Scholar] [CrossRef]
- Costa, P.T.; McCrae, R.R. The NEO Personalityinventory; Psychological Assessment Resources: Odessa, FL, USA, 1985. [Google Scholar]
- Smits, D.J.; Boeck, P.D. From BIS/BAS to the big five. EPJ 2006, 20, 255–270. [Google Scholar] [CrossRef]
- Matyjek, M.; Bayer, M.; Dziobek, I. Autistic Traits Affect Reward Anticipation but not Reception. Sci. Rep. 2020, 10, 8396. [Google Scholar] [CrossRef]
- Bacon, A.M.; Corr, P.J. Motivating emotional intelligence: A reinforcement sensitivity theory (RST) perspective. Motiv. Emot. 2017, 41, 254–264. [Google Scholar] [CrossRef][Green Version]
- Taubitz, L.E.; Pedersen, W.S.; Larson, C.L. BAS Reward Responsiveness: A unique predictor of positive psychological functioning. Pers. Individ. Differ. 2015, 80, 107–112. [Google Scholar] [CrossRef]
- Jasper, H.H.; Andrews, H.L. Electro-encephalography: III. Normal differentiation of occipital and precentral regions in man. Arch. Neurol. Psychiatry 1938, 39, 96–115. [Google Scholar] [CrossRef]
- Buzsáki, G.; Wang, X.J. Mechanisms of gamma oscillations. Annu. Rev. Neurosci. 2012, 35, 203–225. [Google Scholar] [CrossRef]
- Murty, D.V.; Shirhatti, V.; Ravishankar, P.; Ray, S. Large visual stimuli induce two distinct gamma oscillations in primate visual cortex. J. Neurosci. 2018, 38, 2730–2744. [Google Scholar] [CrossRef] [PubMed]
- Veit, J.; Hakim, R.; Jadi, M.P.; Sejnowski, T.J.; Adesnik, H. Cortical gamma band synchronization through somatostatin interneurons. Nat. Neurosci. 2017, 20, 951. [Google Scholar] [CrossRef] [PubMed]
- Kinreich, S.; Djalovski, A.; Kraus, L.; Louzoun, Y.; Feldman, R. Brain-to-brain synchrony during naturalistic social interactions. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Colgin, L.L.; Denninger, T.; Fyhn, M.; Hafting, T.; Bonnevie, T.; Jensen, O.; Moser, E.I. Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 2009, 462, 353–357. [Google Scholar] [CrossRef]
- Fries, P. Rhythms for cognition: Communication through coherence. Neuron 2015, 88, 220–235. [Google Scholar] [CrossRef]
- Thalbourne, M.A. Transliminality: A review. IJP 2000, 11, 1–34. [Google Scholar]
- Thalbourne, M.A.; Bartemucci, L.; Delin, P.S.; Fox, B.; Nofi, O. Transliminality: Its nature and correlates. J. Am. Soc. Psych. Res. 1997, 91, 305–331. [Google Scholar]
- Das, N.; Gastaut, H. Variations in the electrical activity of the brain, heart, and skeletal muscles during yogic meditation and trance. Clin. Neurophysiol. 1955, 6, 211–219. [Google Scholar]
- Gray, C.M.; König, P.; Engel, A.K.; Singer, W. Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature 1989, 338, 334–337. [Google Scholar] [CrossRef]
- Gregoriou, G.G.; Gotts, S.J.; Zhou, H.; Desimone, R. High-frequency, long-range coupling between prefrontal and visual cortex during attention. Science 2009, 324, 1207–1210. [Google Scholar] [CrossRef]
- Pesaran, B.; Pezaris, J.S.; Sahani, M.; Mitra, P.P.; Andersen, R.A. Temporal structure in neuronal activity during working memory in macaque parietal cortex. Nat. Neurosci. 2002, 5, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hagoort, P.; Jensen, O. Language prediction is reflected by coupling between frontal gamma and posterior alpha oscillations. J. Cogn. Neurosci. 2018, 30, 432–447. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.X.; David, N.; Vogeley, K.; Elger, C.E. Gamma-band activity in the human superior temporal sulcus during mentalizing from nonverbal social cues. Psychophysiology 2009, 46, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Van Diessen, E.; Senders, J.; Jansen, F.E.; Boersma, M.; Bruining, H. Increased power of resting-state gamma oscillations in autism spectrum disorder detected by routine electroencephalography. Eur. Arch. Psychiatry Clin. Neurosci. 2015, 265, 537–540. [Google Scholar] [CrossRef]
- Roopun, A.K.; Middleton, S.J.; Cunningham, M.O.; LeBeau, F.E.N.; Bibbig, A.; Whittington, M.A.; Traub, R.D. A beta2-frequency (20–30 Hz) oscillation in nonsynaptic networks of somatosensory cortex. Proc. Natl. Acad. Sci. USA 2006, 103, 15646–15650. [Google Scholar] [CrossRef]
- Murthy, V.N.; Fetz, E.E. Coherent 25-to 35-Hz oscillations in the sensorimotor cortex of awake behaving monkeys. Proc. Natl. Acad. Sci. USA 1992, 89, 5670–5674. [Google Scholar] [CrossRef]
- Canolty, R.T.; Cadieu, C.F.; Koepsell, K.; Ganguly, K.; Knight, R.T.; Carmena, J.M. Detecting event-related changes of multivariate phase coupling in dynamic brain networks. J. Neurophys. 2012, 107, 2020–2031. [Google Scholar] [CrossRef]
- Engel, A.K.; Senkowski, D.; Schneider, T.R. The Neural Bases of Multisensory Processes; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2012. [Google Scholar]
- Pfurtscheller, G.; Stancak, A., Jr.; Edlinger, G. On the existence of different types of central beta rhythms below 30 Hz. Clin. Neurophysiol. 1997, 102, 316–325. [Google Scholar] [CrossRef]
- Kamiński, J.; Brzezicka, A.; Gola, M.; Wróbel, A. Beta band oscillations engagement in human alertness process. Int. J. Psychophysiol. 2012, 85, 125–128. [Google Scholar] [CrossRef]
- Miller, E.K.; Lundqvist, M.; Bastos, A.M. Working Memory 2.0. Neuron 2018, 100, 463–475. [Google Scholar] [CrossRef]
- Siegel, M.; Engel, A.K.; Donner, T.H. Cortical network dynamics of perceptual decision-making in the human brain. Front. Hum. Neurosci. 2011, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- McFarland, D.J.; Miner, L.A.; Vaughan, T.M.; Wolpaw, J.R. Mu and beta rhythm topographies during motor imagery and actual movements. Brain Topogr. 2000, 12, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Von Stein, A.; Rappelsberger, P.; Sarnthein, J.; Petsche, H. Synchronization between temporal and parietal cortex during multimodal object processing in man. Cereb. Cortex 1999, 9, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Engel, A.K.; Fries, P. Beta-band oscillations—Signalling the status quo? Curr. Opin. Neurobiol. 2010, 20, 156–165. [Google Scholar] [CrossRef]
- Kononowicz, T.W.; Roger, C.; VanWassenhove, V. Temporal metacognition as the decoding of self-generated brain dynamics. Cereb. Cortex 2019, 29, 4366–4380. [Google Scholar] [CrossRef]
- Hanslmayr, S.; Spitzer, B.; Bäuml, K.H. Brain oscillations dissociate between semantic and nonsemantic encoding of episodic memories. Cereb. Cortex 2009, 19, 1631–1640. [Google Scholar] [CrossRef]
- Jensen, O.; Goel, P.; Kopell, N.; Pohja, M.; Hari, R.; Ermentrout, B. On the human sensorimotor-cortex beta rhythm: Sources and modeling. Neuroimage 2005, 26, 347–355. [Google Scholar] [CrossRef]
- Berger, H. Über das elektroenkephalogramm des menschen. Arch. Psychiatr. Nervenkrankh. 1929, 87, 527–570. [Google Scholar] [CrossRef]
- Katznelson, R.D. Chapter 6 in Nunez, P.L. Electric Fields of the Brain: The Neurophysics of EEG; Oxford University Press: New York, NY, USA, 1981. [Google Scholar]
- Knyazev, G.G. Motivation, emotion, and their inhibitory control mirrored in brain oscillations. Neurosci. Biobehav. Rev. 2007, 31, 377–395. [Google Scholar] [CrossRef]
- Pfurtscheller, G. Induced oscillations in the alpha band: Functional meaning. Epilepsia 2003, 44, 2–8. [Google Scholar] [CrossRef]
- Clark, J.M. Contributions of inhibitory mechanisms to unified theory in neuroscience and psychology. Brain Cogn. 1996, 30, 127–152. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J. EEG measures of cerebral asymmetry: Conceptual and methodological issues. Int. J. Neurosci. 1988, 39, 71–89. [Google Scholar] [CrossRef] [PubMed]
- Eysenck, H.J.; Eysenck, M.W. Personality and Individual Differences; Plenum: New York, NY, USA, 1987. [Google Scholar]
- Kline, J.P.; Blackhart, G.C.; Woodward, K.M.; Williams, S.R.; Schwartz, G.E. Anterior electroencephalographic asymmetry changes in elderly women in response to a pleasant and an unpleasant odour. Biol. Psychol. 2000, 52, 241–250. [Google Scholar] [CrossRef]
- Myers, L.B.; Brewin, C.R.; Winter, D.A. Repressive coping and self-reports of parenting. Br. J. Clin. Psychol. 1999, 38, 73–82. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J.; Harmon-Jones, E. Voluntary facial expression and hemispheric asymmetry over the frontal cortex. Psychophysiology 2001, 38, 912–925. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J.; Ekman, P.; Saron, C.D.; Senulis, J.A.; Friesen, W.V. Approach-withdrawal and cerebral asymmetry: Emotional expression and brain physiology: I. J. Pers. Soc. Psychol. 1990, 58, 330. [Google Scholar] [CrossRef]
- Gray, J.A. The Nature of Emotion: Fundamental Questions; Oxford University Press: Oxford, UK, 1994; pp. 329–331. [Google Scholar]
- Hewig, J.; Hagemann, D.; Seifert, J.; Naumann, E.; Bartussek, D. On the selective relation of frontal cortical asymmetry and anger-out versus anger-control. J. Pers. Soc. Psychol. 2004, 87, 926. [Google Scholar] [CrossRef]
- Harmon-Jones, E.; Allen, J.J. Behavioral activation sensitivity and resting frontal EEG asymmetry: Covariation of putative indicators related to risk for mood disorders. J. Abnorm. Psychol. 1997, 106, 159. [Google Scholar] [CrossRef]
- Heller, W. Neuropsychological mechanisms of individual differences in emotion, personality, and arousal. Neuropsychology 1993, 7, 476. [Google Scholar] [CrossRef]
- Zuckerman, M.; Kuhlman, D.M. Personality and risk-taking: Common biosocial factors. J. Pers. 2000, 68, 999–1029. [Google Scholar] [CrossRef]
- Mayer, J.D.; Salovey, P. The Intelligence of Emotional Intelligence; Elsevier: Amsterdam, The Netherlands, 1993. [Google Scholar]
- Mikolajczak, M.; Nelis, D.; Hansenne, M.; Quoidbach, J. If you can regulate sadness, you can probably regulate shame: Associations between trait emotional intelligence, emotion regulation and coping efficiency across discrete emotions. Pers. Individ. Differ. 2008, 44, 1356–1368. [Google Scholar] [CrossRef]
- Kemp, A.H.; Cooper, N.J.; Hermens, G.; Gordon, E.; Bryant, R.; Williams, L.M. Toward an integrated profile of emotional intelligence: Introducing a brief measure. J. Integr. Neurosci. 2005, 4, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.A.; Duke, L.M.; Allen, J.J. Resting frontal electroencephalographic asymmetry in depression: Inconsistencies suggest the need to identify mediating factors. Psychophysiology 1998, 35, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Buzsáki, G.; Logothetis, N.; Singer, W. Scaling brain size, keeping timing: Evolutionary preservation of brain rhythms. Neuron 2013, 80, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Kramis, R.; Vanderwolf, C.H.; Bland, B.H. Two types of hippocampal rhythmical slow activity in both the rabbit and the rat: Relations to behavior and effects of atropine, diethyl ether, urethane, and pentobarbital. Exp. Neurol. 1975, 49, 58–85. [Google Scholar] [CrossRef]
- Lang, M.; Lang, W.; Diekmann, V.; Kornhuber, H.H. The frontal theta rhythm indicating motor and cognitive learning. Clin. Neurophysiol. Supplement. 1987, 40, 322–327. [Google Scholar]
- Buzsáki, G. Theta oscillations in the hippocampus. Neuron 2002, 33, 325–340. [Google Scholar] [CrossRef]
- Berry, S.D.; Thompson, R.F. Prediction of learning rate from the hippocampal electroencephalogram. Science 1978, 200, 1298–1300. [Google Scholar] [CrossRef]
- Buzsáki, G. Theta rhythm of navigation: Link between path integration and landmark navigation, episodic and semantic memory. Hippocampus 2005, 15, 827–840. [Google Scholar] [CrossRef]
- Knyazev, G.G.; Slobodskoy-Plusnin, J.Y. Substance use underlying behavior: Investigation of theta and high frequency oscillations in emotionally relevant situations. Clin. EEG Neurosci. 2009, 40, 1–4. [Google Scholar] [CrossRef]
- Krishnan, M.; Mariappan, M. EEG-based brain-machine interface (BMI) for controlling mobile robots: The trend of prior studies. J. Electron. 2015, 3, 159–165. [Google Scholar]
- Steriade, M.; Contreras, D.; Dossi, R.C.; Nunez, A. The slow (<1 Hz) oscillation in reticular thalamic and thalamocortical neurons: Scenario of sleep rhythm generation in interacting thalamic and neocortical networks. J. Neurosci. 1993, 13, 3284–3299. [Google Scholar] [PubMed]
- Michel, C.M.; Henggeler, B.; Brandeis, D.; Lehmann, D. Localization of sources of brain alpha/theta/delta activity and the influence of the mode of spontaneous mentation. Physiol. Measur. 1993, 14, A21. [Google Scholar] [CrossRef] [PubMed]
- Lambertz, M.; Langhorst, P. Simultaneous changes of rhythmic organization in brainstem neurons, respiration, cardiovascular system and EEG between 0.05 Hz and 0.5 Hz. J. Auton. Nerv. Syst. 1998, 68, 58–77. [Google Scholar] [CrossRef]
- Leung, L.S.; Yim, C.C. Rhythmic delta-frequency activities in the nucleus accumbens of anesthetized and freely moving rats. Can. J. Phisiol. Pharmacol. 1993, 71, 311–320. [Google Scholar] [CrossRef]
- Grace, A.A. The tonic/phasic model of dopamine system regulation: Its relevance for understanding how stimulant abuse can alter basal ganglia function. Drug Alcohol Depend. 1995, 37, 111–129. [Google Scholar] [CrossRef]
- Berridge, K.C.; Robinson, T.E.; Aldridge, J.W. Dissecting components of reward: ‘liking’, ‘wanting’, and learning. Curr. Opin. Pharmacol. 2009, 9, 65–73. [Google Scholar] [CrossRef]
- Gray, J.A. The Neuropsychology of Anxiety; Clarendon Press/Oxford University Press: Oxford, UK, 1982. [Google Scholar]
- Eysenck, H.J. Biological Dimensions of Personality; The Guilford Press: New York, NY, USA, 1990. [Google Scholar]
- Spielberger, C.D.; Sydeman, S.J.; Owen, A.E.; Marsh, B.J. Measuring Anxiety and Anger with the State-Trait. Anxiety Inventory (STAI) and the State-Trait. Anger Expression Inventory (STAXI); Lawrence Erlbaum Associates Publishers: Mahwah, NJ, 1999. [Google Scholar]
- Robinson, D.L. Properties of the diffuse thalamocortical system and human personality: A direct test of Pavlovian/Eysenckian theory. Pers. Individ. Differ. 1982, 3, 1–16. [Google Scholar] [CrossRef]
- Depue, R.A.; Collins, P.F. Neurobiology of the structure of personality: Dopamine, facilitation of incentive motivation, and extraversion. Behav. Brain Sci. 1999, 22, 491–517. [Google Scholar] [CrossRef]
- Sawyer, D.A.; Julia, H.L.; Turin, A.C. Caffeine and human behavior: Arousal, anxiety, and performance effects. J. Behav. Med. 1982, 5, 415–439. [Google Scholar] [CrossRef]
- Gibbs, F.A.; Gibbs, E.L.; Lennox, W.G. Effect on the electro-encephalogram of certain drugs which influence nervous activity. Arch. Intern. Med. 1937, 60, 154–166. [Google Scholar] [CrossRef]
- Buzsáki, G.; Watson, B.O. Brain rhythms and neural syntax: Implications for efficient coding of cognitive content and neuropsychiatric disease. Dialogues Clin. Neurosci. 2012, 14, 345. [Google Scholar] [PubMed]
- Gasser, T.; Bächer, P.; Steinberg, H. Test-retest reliability of spectral parameters of the EEG. Clin. Neurophysiol. 1985, 60, 312–319. [Google Scholar] [CrossRef]
- Hagemann, D.; Naumann, E.; Becker, G.; Maier, S.; Bartussek, D. Frontal brain asymmetry and affective style: A conceptual replication. Psychophysiology 1998, 35, 372–388. [Google Scholar] [CrossRef]
- Brodal, P. The Central Nervous System: Structure and Function; Oxford University Press: Oxford, UK, 1992; pp. 246–261. [Google Scholar]
- Verleger, R. The instruction to refrain from blinking affects auditory P3 and N1 amplitudes. Clin. Neurophysiol. 1991, 78, 240–251. [Google Scholar] [CrossRef]
- Hagemann, D.; Naumann, E.; Thayer, J.F. The quest for the EEG reference revisited: A glance from brain asymmetry research. Psychophysiology 2001, 38, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Melrose, S. Seasonal affective disorder: An overview of assessment and treatment approaches. Depress. Res. Treat. 2015. [Google Scholar] [CrossRef]
- King, J.A.; Rosal, M.C.; Ma, Y.; Reed, G.; Kelly, T.A.; Ockene, I.S. Sequence and seasonal effects of salivary cortisol. J. Behav. Med. 2000, 26, 67–73. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J.; McKnight, P.E. A capability model of individual differences in frontal EEG asymmetry. Biol. Psychol. 2006, 72, 198–207. [Google Scholar] [CrossRef]
- Wallace, J. An abilities conception of personality: Some implications for personality measurement. Am. Psychol. 1966, 21, 132. [Google Scholar] [CrossRef]
- Cloninger, C.R.; Przybeck, T.R.; Svrakic, D.M. The tridimensional personality questionnaire: US normative data. Psychol. Rep. 1991, 69, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
| Participants | Personality Domain | Electrophysiological Domain | Bias | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Authors | N | Age Mean (SD) | Sex Men % | Self-Report | Trait/s Considered | Resting State Recording Condition (EEG Reference, Length) | Results and Indexes of Brain Activation | EEG Test–Retest | Risk % | |
| Tomarken et al., 1992 [26] | 90 | 19 (-) | 0 | EHI a PANAS b AIM c | Handedness Positive Affect (PA), Negative Affect (NA) Intensity of individual experiences emotion | Eight of 1-min baseline, balanced for four opened (O) and four closed (C)-eyes order, (O-C-C-O-C-O-O-C and C-O-O-C-O-C-C-O), participants, and sessions. Online ref. linked CZ site. Offline re-ref. average earlobes. | Left anterior α-asymmetry is positively related to PA, and negatively related to NA. This pattern of EEG activation is stable over time. | Two measures 2-after 3 weeks | 58.4 | |
| Tomarken and Davidson, 1994 [27] | 90 | 19 (-) | 0 | EHI a MCSD d STAI e BDI f | Handedness Defensiveness Trait Anxiety (TA) Depression Trait (TD) | 8 of 1-min baseline, balanced for 4 O and 4 C eyes order, (O-C-C-O-C-O-O-C and C-O-O-C-O-C-C-O), participants, and sessions. Online ref. linked CZ site. Offline re-ref. average earlobes. | The α activity in mid-frontal and left lateral frontal sites is related to a higher level of defensiveness than a lower level. | Two measures 2-after 3 weeks | 58.4 | |
| Sutton and Davidson, 1997 [28] | 46 | 20 (-) | 50 | EHI a PANAS b BIS/BAS Scale g | Handedness PA, NA Behavioral Inhibition System (BIS), Behavioral Approach System (BAS) | Eight of 1-min baseline, balanced for four O and four C eyes order, participants, and sessions. Online ref. linked earlobes. Offline re-ref. average earlobes. | Resting prefrontal activation is an index of EEG α-asymmetry related to Gray’s BAS and BIS rather than PA and NA. | Two measures two after 3 months | 66.7 | |
| Hagemann et al., 1999 [29] | 36 | 23.5 (4.0) | 36 | EHI a PANAS b EPQ h | Handedness PA, NA E, N | Eight of 1-min baseline, balanced for four O and four C eyes order (O-C-C-O-C-O-O or C-O-O-CO-C-CO), and participants. Online ref. linked mastoids. Offline re-ref. CZ site. | Lateral α-asymmetry in resting EEG is related to NA but is not significantly related to PA. | One Measure | 50.0 | |
| Kline et al., 2001 [30] | 72 | 24.4 (4.6) | 42 | EHI a EPQ h HPCS i MCSD d | Handedness Neuroticism (N), Psychoticism (P), Extraversion (E), Lie (L) Maternal and Paternal caring Defensiveness | Six of 1-min baseline, balanced for three O and three C eyes order, and participants. Online ref. linked earlobes. | Defensiveness and perceived maternal (but not paternal) caring, separately predict left lateral frontal activation of the EEG α-asymmetry. | One Measure | 66.7 | |
| Kline et al., 2002 [31] | 235 | 20.4 (4.1) | 67 | EHI a EPQ h | Handedness N, E, P, L | Six of 1-min baseline, three O eyes, three C eyes. Online ref. linked earlobes. | Defensiveness is related with right frontal α-activity, in the presence of opposite-sex experimenters but not same-sex. | One Measure | 58.4 | |
| Blackhart et al., 2002 [32] | 77 | 20.5 (3.7) | 47 | EHI a SAM j | Handedness Pleasure–Displeasure, Arousal–Non arousal, Dominance–Submissive. | Six of 1-min baseline, three O eyes, three C eyes. Online ref. linked earlobes. | Negative mood post-preparation, predicts left frontal α-asymmetry during resting EEG, in women. | One Measure | 66.7 | |
| Coan and Allen, 2003 [33] | 32 | 20.5 (-) | 28 | BIS/BAS Scale g | BIS, BAS | Eight of 1-min baseline balanced for four O and four C eyes order. Online ref. Cz site. Offline re-ref. linked mastoids. Offline re-ref. average brain activity. | Resting EEG left frontal -asymmetry is related to greater BAS scores; higher BIS scores are related to right frontal activity in the mid-frontal region, only. | One Measure | 83.4 | |
| Knyazev and Slobodskaya, 2003 [34] | 47 | 23.9 (5.3) | 13 | GWPQ k EPQ h STAI e | BIS, BAS E, N, P TA | Six of 60-s baseline, balanced for a pseudo-random O/C-eyes order, and participants. Online ref. linked earlobes. | Higher δ–α EEG frequency oscillations coupling, are related to increased behavioral inhibition. | One Measure | 75.0 | |
| Razoumnikova, 2003 [35] | 46 | 18 (-) | 100 | EPQ h AIS m KTI l | E, N, P Emotional Intelligence: Verbal, Numerical, Figural Sensation–Intuition (S), Thinking–Feeling (T), Judging–Perceiving (J) | 3 min resting period with C eyes. Online ref. linked earlobes. | EEG rhythms are relating to IQ, S, E, and N. Cognitive abilities in higher IQ subjects are related with the cortex connectivity, and θ1–θ2–α1 EEG rhythms. The β2 rhythm is predominant in right hemisphere. | One Measure | 83.4 | |
| Minnix and Kline, 2004 [36] | 140 | 20.4 (4.1) | 30 | EHI a EPQ h | Handedness N, E, P, L | Six of 1-min baseline, three O eyes, three C eyes. Online ref. linked earlobes. | EEG Resting Mid-Frontal α–band activity is related with higher N. | One Measure | 75.0 | |
| Hewig et al., 2006 [37] | 59 | 24 (3.3) | 49 | EHI a BIS/BAS Scale g EPQ h | Handedness BIS, BAS E, N | 12 of 1-min baseline, six O and six C eyes, balanced for two orders (OCCO-COOC-OCCO; COOC-OCCO-COOC) and participants. Online ref. linked CZ. | There is no relation between resting frontal α–activity of EEG, and BIS/BAS traits. | Four measures one every 4 weeks | 58.4 | |
| Tran et al., 2006 [38] | 699 | 36 (-) | 49 | NEO-FFI n | E, N, Openness (O), Agreeableness (A), Consciousness (C). | 3 min resting period with C eyes. Offline re-ref. linked mastoids. | N scores are higher in women than men. In both, δ–θ activity is related to E and C, while, α–β activity is related to NEO–FFI traits in males. | One Measure | 75.0 | |
| Jaušovec and Jaušovec, 2007 [39] | 110 | 21 (-) | 50 | NEO-FFI n MSCEIT o | N, E, O, A, C Emotional Intelligence: Verbal, Performance, Experiential–Emotional, Strategic–Emotional | 5 min resting period with C eyes and 5 min resting period with O eyes. Repeating O/C eyes order after each minute of recording. Online ref. linked mastoids. | Women with extreme traits levels differ in the γ band activity than males; vice-versa in the α1 band. Brain activity increased in parietal-occipital areas rather in frontal. | One Measure | 66.7 | |
| Smit et al., 2007 [40] | Young Mid-Aged | 381 380 | 26.2 (4.1) 49.4 (7.2) | - - | No specified STAI e YARS p ABV q | Handedness TA Depression Trait (TD) N, Somatic-Anxiety | 3 min resting period with C eyes. Online ref. linked earlobes. | Frontal α-Asymmetry (FA) in rest-EEG, is heritable in young adults but not in middle-aged. FA is related to the risk of anxiety and depression in young women. | One Measure | 83.4 |
| Fleck et al., 2008 [41] | 34 | 22.4 (4.7) | 47 | RTS r | Transliminality (Tr) | 3.5 min resting period with C eyes and 3.5 min resting period with O eyes. Online ref. linked mastoids. | Higher Tr scores related with lower α1, β1, γ1 activity in left posterior cortex, and lower α2, β1, γ activity in right superior temporal areas, than lower scores. Lower levels of Tr, related to increased γ in the mid-frontal areas, than higher. | One Measure | 83.4 | |
| Gatt et al., 2008 [42] | 117 | 36.9 (12.6) | 38 | DASS-21 s | TD | 2 min resting period with C eyes followed by 2 min O eyes. Offline re-ref. linked earlobes. | BDNF M/M genotype is a predictor of working memory activity. The EEG α-power mediates the effect of this phenotype on TD expression. | One Measure | 66.7 | |
| Kline and Allen, 2008 [43] | 71 | 18.9 (1.5) | 30 | EHI a EPQ h MCSD d BDI f TMAS ee | Handedness N, E, P, L Defensiveness TD TA | Six of 1-min baseline, balanced for 3 O eyes and 3 C eyes orders (O–C–C–O– C–O; C–O–O–C–O–C). Online ref. linked earlobes. Offline re-ref. average brain activity. | In the context of increased α-band activity in the right lateral frontal asymmetry EPQL traits are positively related to BDI. | Two measures two after 3 weeks | 41.7 | |
| Mathersul et al., 2008 [44] | 428 | 34.9 (12.6) | 50 | No specified DASS-21 s | Handedness TD, TA | 2 min resting period with O eyes and 2 min with C eyes. Offline re-ref. average brain activity. | An increased α-asymmetry in the right parietal-temporal areas related to higher anxiety comorbidity and depression trait. Right parietal-temporal α-activity related to anxiety apprehension. | One Measure | 58.4 | |
| Santesso et al., 2008 [45] | Study1 Study2 | 37 44 | 19.5 (1.4) 18.6 (0.7) | 24 100 | EHI a SSS-V u FIS v | Handedness Sensation Seeking Risk-taking behaviors | 1 min resting period with O eyes and 1 min with C eyes. Online ref. linked CZ. Offline re-ref. average brain activity. | Sensation seeking is associated with a greater left frontal -asymmetry in resting EEG. | One Meas. One Meas. | 66.7 66.7 |
| Schutter et al., 2008 [8] | 24 | 22.2 (2.0) | 46 | BIS/BAS Scale g | BIS, BAS | Four of 1-min baseline balanced for two O and two C eyes order (O–C–O–C). Online ref. voltage. Offline re-ref. CZ. | Β-asymmetry in resting EEG reflects approach–avoidance motivational predispositions. | One Measure | 41.7 | |
| Master et al., 2009 [46] | 46 | 20.1 (1.5) | 43 | No specified EAC w BIS/BAS Scale g | Handedness Emotional Approach BIS, BAS | Eight of 1-min baseline balanced for four O and four C eyes order. Online ref. Left-lobe. Offline re-ref. lobes average. | Emotional expression is significantly related to greater left-sided frontal A-asymmetry in the rest-EEG state. | One Measure | 58.4 | |
| Pavlenko et al., 2009 [47] | 111 | 21.5 (-) | 42 | STAI e Cattell 16PFx | State and Trait Anxiety A, B, C, E, F, G, H, I, L, M, N, O, Q1, Q2, Q3, Q4.+ | Two of 1-min baseline, one with O eyes and one with C eyes. Online ref. linked an active electrode n.a. | State anxiety is positively related to β2 spectral power density in the temporal occipital areas of the right hemisphere (O-eyes). Trait anxiety is positively related to β1, β2. | One Measure | 75.0 | |
| Peterson and Harmon-Jones, 2009 [48] | Study1 Study2 | 72 65 | Stud. (-) Stud. (-) | - 58 | IAS-R y Time Variables BIS/BAS Scale g | Nurturance and Dominance Day Time, Year season BIS, BAS | Four of 1-min baseline balanced for two O and two C eyes order (O–C–C–O or C–O–O–C) and participants. Online ref. eft-earlobe. Offline re-ref. earlobes average. | Frontal α-asymmetry in resting EEG reflects circadian and seasonal influences. The right frontal activity is increased during fall mornings. | One Measure One Measure | 66.7 50.0 |
| Knyazev et al., 2010 [49] | 132 | 27.5 (-) | 39 | EPQ h GWPQ k STAI e | N BIS TA | 3-min rest period with C eyes, 3- min with O eyes balanced for O/C-eyes order. Online ref. lobes. | Increased coupling δ–β frequency oscillations is related with state anxiety. | One Measure | 75.0 | |
| Mikolajczak et al., 2010 [50] | 31 | 22.4 (3.8) | 20 | TEI–Que z | Well-Being, Self-Control, Emotionality, Sociability | Eight of 1-min baseline balanced for four O and four C eyes order. Online ref. linked earlobe. | Resting EEG α–frontal asymmetry is related to emotional intelligence. | One Measure | 83.3 | |
| Wacker and Gatt, 2010 [51] | 1093 | 39.3 (18.7) | 46 | NEO–FFI n | N, E, O, A, C | 2-min resting period with O eyes followed by 2-min C eyes. Online ref. linked mastoids. | The δ–θ activity in posterior vs. frontal areas related to E and COMT VAL-158 MET polymorphism. | One Measure | 66.7 | |
| Wacker et al., 2010 [52] | Study3 Study4 | 112 35 | 23.8 (3.4) 22.9 (3.4) | 100 62 | ARES gg BIS/BAS Scale g | BAS, BIS BAS, BIS | Five for 90-s of resting period with C eyes. Online ref. linked CZ site. Offline re-ref. linked mastoids. | Resting posterior vs. frontal δ–θ EEG activity, is related to BAS/AE but BAS/AE, not related to frontal α -asymmetry. | Three meas. two after 1 year, three n.a. One Meas. | 66.7 91.7 |
| Chavanon et al., 2011 [53] | 78 | 23.2 (2.7) | 100 | MPQ–NE EPQ h | Reactivity, Alienation, and Aggression E, N, P | Five for 1-min resting period with C eyes. Online ref. linked CZ site. Offline re-ref. avrage brain activity. | Rostral anterior cingulate activity generates the posterior vs. anterior θ activity, related to AE. | One Measure | 66.7 | |
| Koehler et al., 2011 [54] | 141 | 27.8 (-) | 52 | NEO–PI–R nn | N, E, O, A, C | 5-min rest period with closed eyes. Online ref. linked between Fz-Cz sites. Offline re-ref. average brain activity. | Resting posterior minus frontal EEG δ–θ activity is related with E and DRD2 genotype. | One Measure | 83.4 | |
| Konareva, 2011 [55] | 118 | 27 (-) | - | Cattell 16 PF x EPQ h STI xx OST xxx | A, B, C, E, F, G, H, I, L, M, N, O, Q1, Q2, Q3, Q4.+ E, N, P Objective plasticity, Social ergoicity Ergonicity, Plasticity, Tempo, Emotionality. | n.a. resting period with O eyes followed by n.a. resting period with C eyes. Online ref. n.a. Offline ref n.a. | Psychological traits of personality modulated the activity of δ and θ EEG rhythms. | One Measure | 91.7 | |
| Knyazev et al., 2012 [56] | 88 | 24.5 (-) | 51 | Self-report ad hoc EPP hh | Positive emotions, mood state E, N, P | 3-min rest period with C eyes and 3-min with O eyes balanced for O/C-eyes order. Online ref. linked lobes. | AE related to higher θ activity in the posterior default mode network and lower θ in the orbitofrontal cortex. | One Measure | 66.7 | |
| De Pascalis et al., 2013 [57] | 51 | 24.1 (3.7) | 0 | EHI a BIS/BAS Scale g LOT–R t PANAS b | Handedness BIS, BAS Optimism PA, NA | Four of 1-min baseline balanced for two O and two C eyes order and participants. Online ref. linked CZ site. Offline re-ref linked earlobes. | Higher BAS scores are related to greater left-sided α-activity in the BA11 area. Optimism is related to higher activity in the BA10 and BA31 areas. | One Measure | 42.8 | |
| Alessandri et al., 2015 [58] | 51 | 24.1 (3.7) | 0 | EHI a GSS gs LOT–R t LSS ls P–OR tt | Handedness Self-esteem Optimism Life Satisfaction Positive orientation | Four of 1-min baseline balanced for two O and two C eyes order and participants. Online ref. linked CZ site. Offline re-ref linked earlobes. | The α-asymmetry in the posterior cingulate cortex is uniquely associated with both positivity scores. | One Measure | 42.8 | |
| Gable et al., 2015 [59] | 126 | Stud. (-) | 46 | Self-report ad hoc PUM ttt BIS/BAS Scale g | Handedness Positive Urgency BIS, BAS | Eight of 1-min baseline, balanced among four O and four C eyes order (C-O-O-C-O-C-C-O or O-C-C-O-C-O-O-C) and participants. Online ref. Left-lobe. Offline re-ref. average brain activity. | BAS trait related to greater left-frontal α-asymmetry, while BIS trait is related to greater right-frontal α-asymmetry. | One Measure | 75.0 | |
| Huang et al., 2015 [60] | 94 | 26.5 (-) | 0 | CC-scale aa EPQ h | Handedness E, N, P, L | 16 of 15-s baseline, eight O and eight C eyes balanced for two groups (O–C–O–C–O–C–O–C and C–O–C–O–C–O–C–O). Online ref. linked mastoids. Offline re-ref. CZ. | Women with higher N scores, exhibit lower-left prefrontal α1– αTotal– asymmetry, than women with lower N during the mid-late luteal phase. | Three measures two after 11–13 days three after 22–24 days | 41.7 | |
| Korjus et al., 2015 [61] | 289 | 22.0 (3.6) | 35 | NEO-PI-R nn | N, E, O, A, C | 1-min; two of 1-min and three of 1-min baseline one O eyes and one C eyes balanced for O/C order. Online ref. linked lobes. Offline re-ref. lobes. | Personality traits cannot be predicted from the power of resting-state EEG. | One Measure | 75.0 | |
| Tullett et al., 2015 [62] | 56 | 19.8 (3.8) | 46 | NI ni SNS ns BFQ nnn | Degree of Nostalgia Frequency of Nostalgia N, E, O, A, C | Eight of 30-s baseline, balanced for four O and four C eyes order. Online ref. digital average. Offline re-ref. lobes. | The right-frontal α-asymmetry predicts an increased proneness to nostalgia scores. | One Measure | 75.0 | |
| Adolph and Margraf, 2017 [63] | 37 | 24.2 (3.8) | 51 | No specified STAI e D–S ff | Handedness TA TD | 16 of 1-min baseline, balanced for eight O and eight C eyes order, and participants (O–C–C–O–C–O–O–C or C–O–O–C–O–C–C–O). Online ref. left mastoid. Offline re-ref. mastoids. | Depression and anxiety are both significantly related to larger right frontal α-asymmetry. | One Measure | 50.0 | |
| Neal and Gable, 2017 [64] | 182 | Stud. (-) | 40 | Checklist ad hoc UPPS-P imp BIS/BAS Scale g | Handedness Impulsivity (Imp) BIS, BAS | Eight of 1-min baseline, balanced for four O and four C eyes order. Online ref. linked left earlobe. Offline re-ref. to earlobes. | Regulatory control and impulsivity are related to BIS. Withdrawal is related to FFFS, and right frontal A-asymmetry. | One Measure | 75.0 | |
| De Groot and Van-Strien, 2018 [65] | 93 | 21.03 (2.4) | 39 | AQ aq | Autistic Traits | 3 min rest period with C eyes, 3 min with O eyes balanced for O/C order. Online ref. linked mastoids. Offline re-ref. to mastoids average. | Spontaneous γ1–γ2 oscillations during resting-state are not predictive of autistic traits in the general population. | One Measure | 58.3 | |
| De Pascalis et al., 2018 [66] | 162 | 23.6 (2.4) | 0 | EHI a RST–PQ kk | Handedness BIS, BAS, FFFS | 4-min rest period with C eyes, 4-min with O eyes balanced among O/C order and participants. Online ref. linked left earlobe. Offline re-ref. to earlobes. | In the subgroup with a young same-sex experimenter, BAS-impulsivity is related to a greater left-frontal α-activity, and FFFS. BIS trait, not related to the greater right-frontal-central activity. | One Measure | 33.4 | |
| Threadgill and Gable, 2018 [67] | 125 | Stud. (-) | - | EHI a BIS/BAS Scale g UPPS-P imp | Handedness BAS, FFFS Impulsivity (Impu) | 4 min rest period with C eyes, 4 min with O eyes balanced between participants. Online ref. linked left earlobe. Offline re-ref. average brain activity. | BAS trait is negatively related to resting β activity. Impu. is related to greater resting β activity. Lower levels of β activity in the motor cortex related to motivational traits. | One Measure | 75.0 | |
| Paban et al., 2019 [68] | 45 | 34.7 (9.1) | 51 | CD–RISC rt | Resilience Trait | 10 min rest period with C eyes. Online ref. individual vertex site. | Resilience is related to resting EEG source-space brain network flexibility. | One Measure | 75.0 | |
| De Pascalis et al., 2020 [69] | 125 | 22.8 (2.8) | 39 | No specified RST–PQkk STAI–State e | Handedness BIS, BAS, Fight–Flight–Freeze System (FFFS) State Anxiety | 3 min rest period with C eyes, 3 min with O eyes balanced among O/C eyes order and participants. Online ref. linked earlobes. Offline re-ref. average brain activity. | BIS trait related to higher δ–β coupling in Anxiety group, and high δ–θ coupling in Relaxation group. In both groups, BAS-GDP is positively related to higher δ–γ coupling. | One Measure | 41.7 | |
| Zanesco et al., 2020 [70] | 187 | 36.1 (20.1) | 64 | BIS/BAS Scale g NEO–FFI p MDMQ ww | BIS, BAS N, E, O, A, C Current Mood state | 16 of 60-s baseline, balanced for eight O, and eight C eyes order and participants. Online ref. linked FCz site. Offline re-ref. average brain activity. | Personality traits can be reflected in microstate dynamics present during periods of quiet rest. | One Measure | 58.4 | |
| Zhang et al., 2020 [71] | 80 | 21.49 (2.5) | 55 | DERS ss STAI–State e STAI–Trait e BDI f | Emotional-Dysregulation State Anxiety TA TD | 12 of 15-s baseline, six O eyes, six C eyes balanced for O/C eyes order and participants. Online ref. linked left mastoid. Offline re-ref. average of mastoids. | The resting-state frontal α-asymmetry predicts the regulation of emotion, mainly in impulse control. | One Measure | 58.4 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vecchio, A.; De Pascalis, V. EEG Resting Asymmetries and Frequency Oscillations in Approach/Avoidance Personality Traits: A Systematic Review. Symmetry 2020, 12, 1712. https://doi.org/10.3390/sym12101712
Vecchio A, De Pascalis V. EEG Resting Asymmetries and Frequency Oscillations in Approach/Avoidance Personality Traits: A Systematic Review. Symmetry. 2020; 12(10):1712. https://doi.org/10.3390/sym12101712
Chicago/Turabian StyleVecchio, Arianna, and Vilfredo De Pascalis. 2020. "EEG Resting Asymmetries and Frequency Oscillations in Approach/Avoidance Personality Traits: A Systematic Review" Symmetry 12, no. 10: 1712. https://doi.org/10.3390/sym12101712
APA StyleVecchio, A., & De Pascalis, V. (2020). EEG Resting Asymmetries and Frequency Oscillations in Approach/Avoidance Personality Traits: A Systematic Review. Symmetry, 12(10), 1712. https://doi.org/10.3390/sym12101712






