Appropriate Feature Set and Window Parameters Selection for Efficient Motion Intent Characterization towards Intelligently Smart EMG-PR System
Abstract
1. Introduction
- Towards providing a standard guideline that would aid the development of intelligently intuitive and symmetric prosthesis for upper limb amputees, this study systematically investigated the interrelations of multiple dynamic factors (windowing parameters, signal conditioning, and feature sets) on the movement intent decoding performance of EMG-PR system based on Linear Discriminant Analysis (LDA). It is worth noting that this issue has rarely been investigated to date.
- A framework for optimal feature set and windowing parameter (window length and increment) construction in context of movement intent decoding was established. This enables the identification of features with low computation and high discrimination capability alongside their corresponding windowing parameters for both amputees and healthy subjects.
- Further, the tradeoff between the decoding accuracy of a range of feature sets and their computational complexity across a combination of window parameters was examined with the aim of triggering positive development in the field of smart prosthetic control system and other pattern recognition-based systems that focus on providing smart healthcare services.
2. Materials and Methods
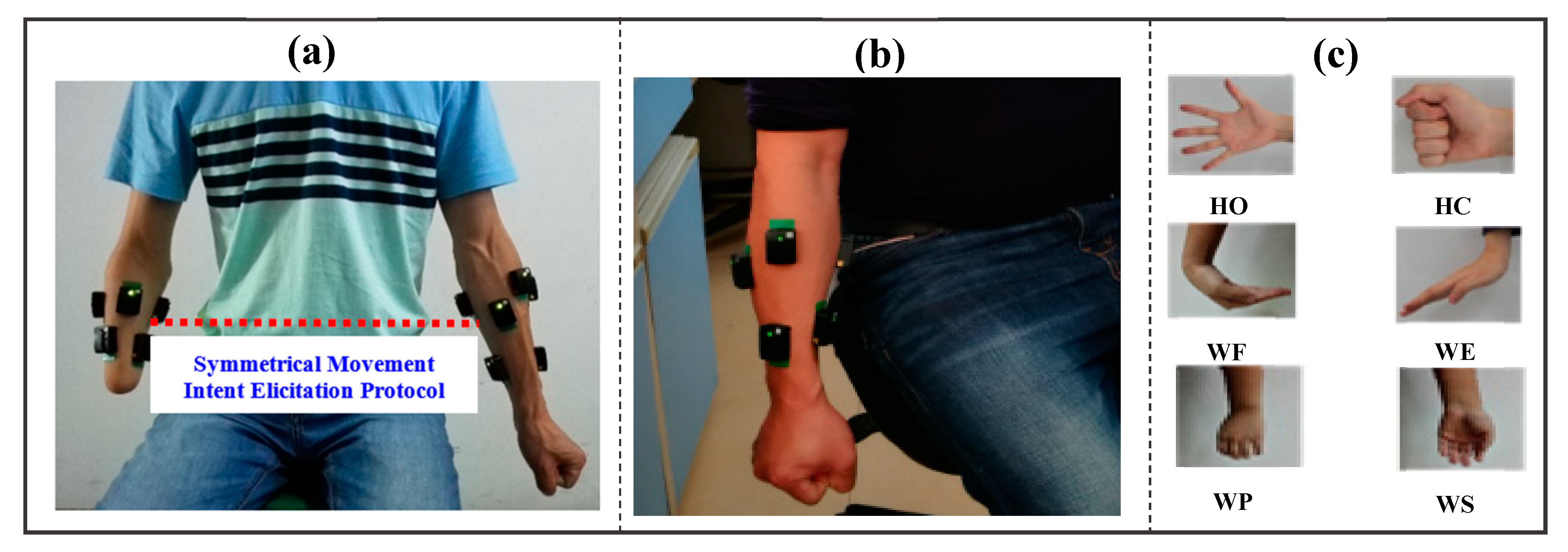
2.1. Participant Information
2.2. EMG Data Measurement
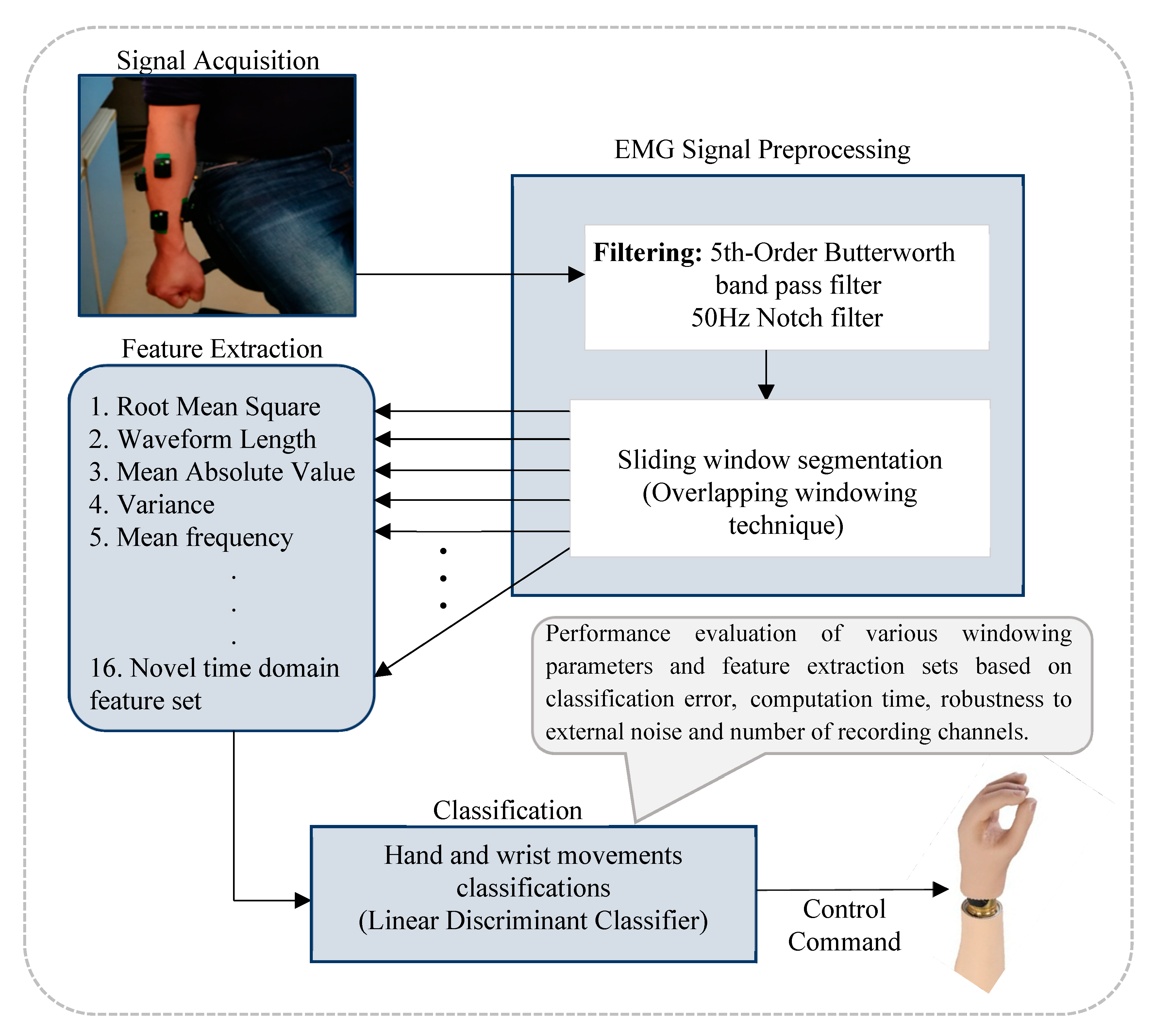
2.3. Preprocessing of the Measure sEMG Data
2.4. Windowing Technique
2.5. Feature Extraction Procedure
2.6. Evaluation Metrics
- The commonly applied metric know as classification error (CE) which represent the number of non-correctly identified samples over the sum of all samples (Equation (1)) was utilized to evaluate the classification accuracy of the feature extracted methods:
- The F1_score was utilized to further validate the performance of the extracted feature sets. This metric was computed as the weighted average of precision and recall (Equations (2) and (3)) [37]. Basically, the F1_score reveals the performance of the classifier in classifying the data points of a particular feature set compared to others,where TP is the count of true positives, TN is the count of true negatives, FP represent number of false positives, and FN is the number of false negatives obtained from a confusion matrix. It is worth noting that F1_score reaches its best value at 1 and worst at 0.
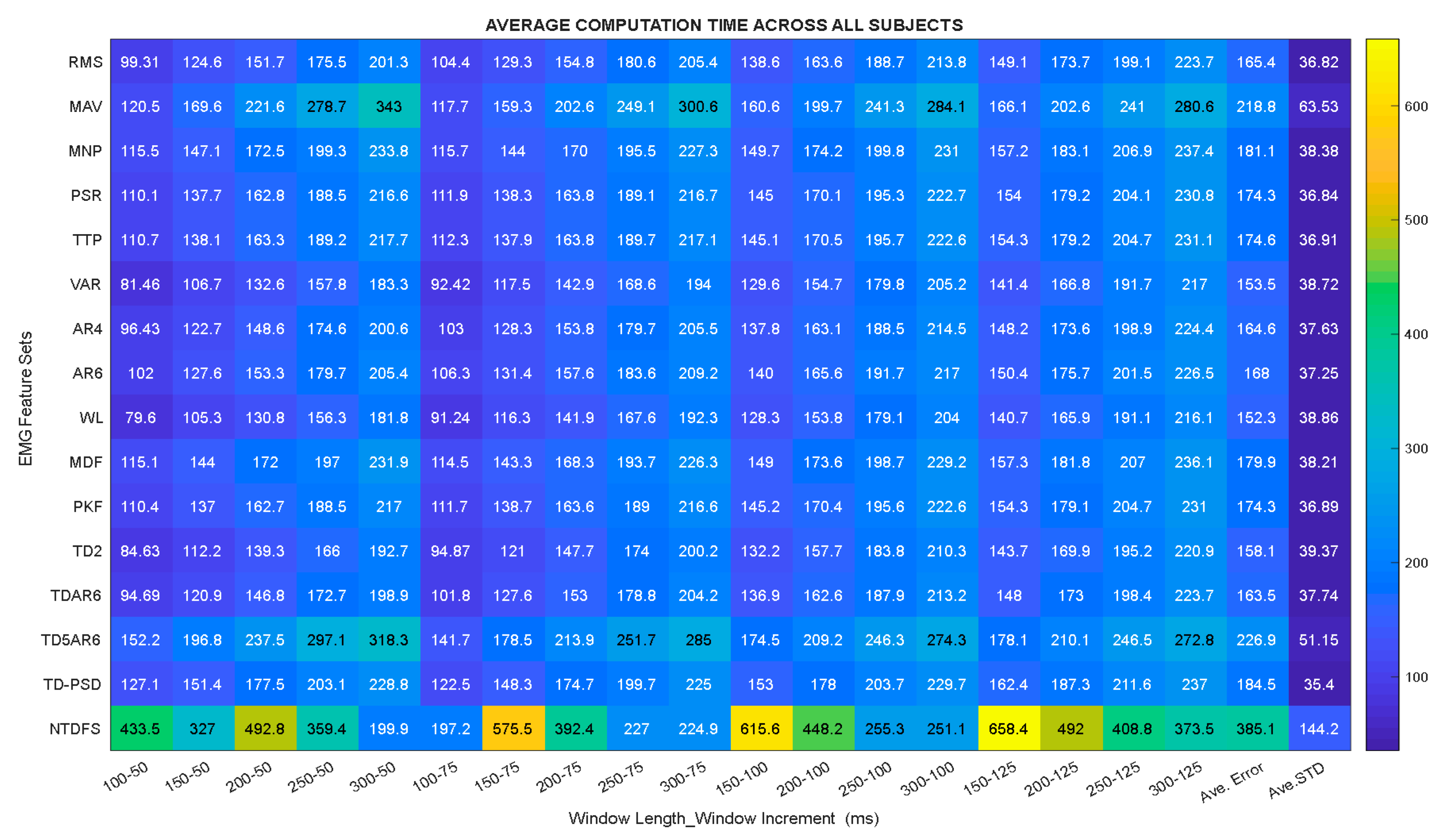
- In principle, the computation time of a feature set would generally influence the response time of the microprocessor-based controller embedded in the prosthesis socket [15]. In this regard, the computation time of each extracted feature set presented in Table 2 was investigated by adopting the formulae in Equation (4) that was proposed by Weir and Farell [15],where D is the delay, is the window length, is the window increment and is the signal processing time. It should be noted that the configuration of the system utilized for this study is Microsoft window 7 professional with 64-bit operating system, Intel(R) Core(TM) i7 processor with processing speed of 3.6 GHz and 8 GB random access memory.
- In the context of EMG-based pattern recognition system, an ideal feature extraction method would normally be influenced/affected by unwanted disturbances that may degrade the decoding of the user’s intended limb movement. Therefore, it is important to quantify the robustness of a feature in other to guarantee that the features would be consistently stable when applied in real-life applications. In this regard, the stability index metrics adopted in a previous study [38], which is defined by Equation (5) was applied to examine the robustness of the feature extraction methods in the presence of noise,where the numerator is the average classification performance, the denominator is the scaled standard deviation, ∝ is the scaled value and is set to 0.1 and N is the sample size. The value of ∝ was chosen after many trials.
2.7. Machine Learning Classification Technique
3. Results
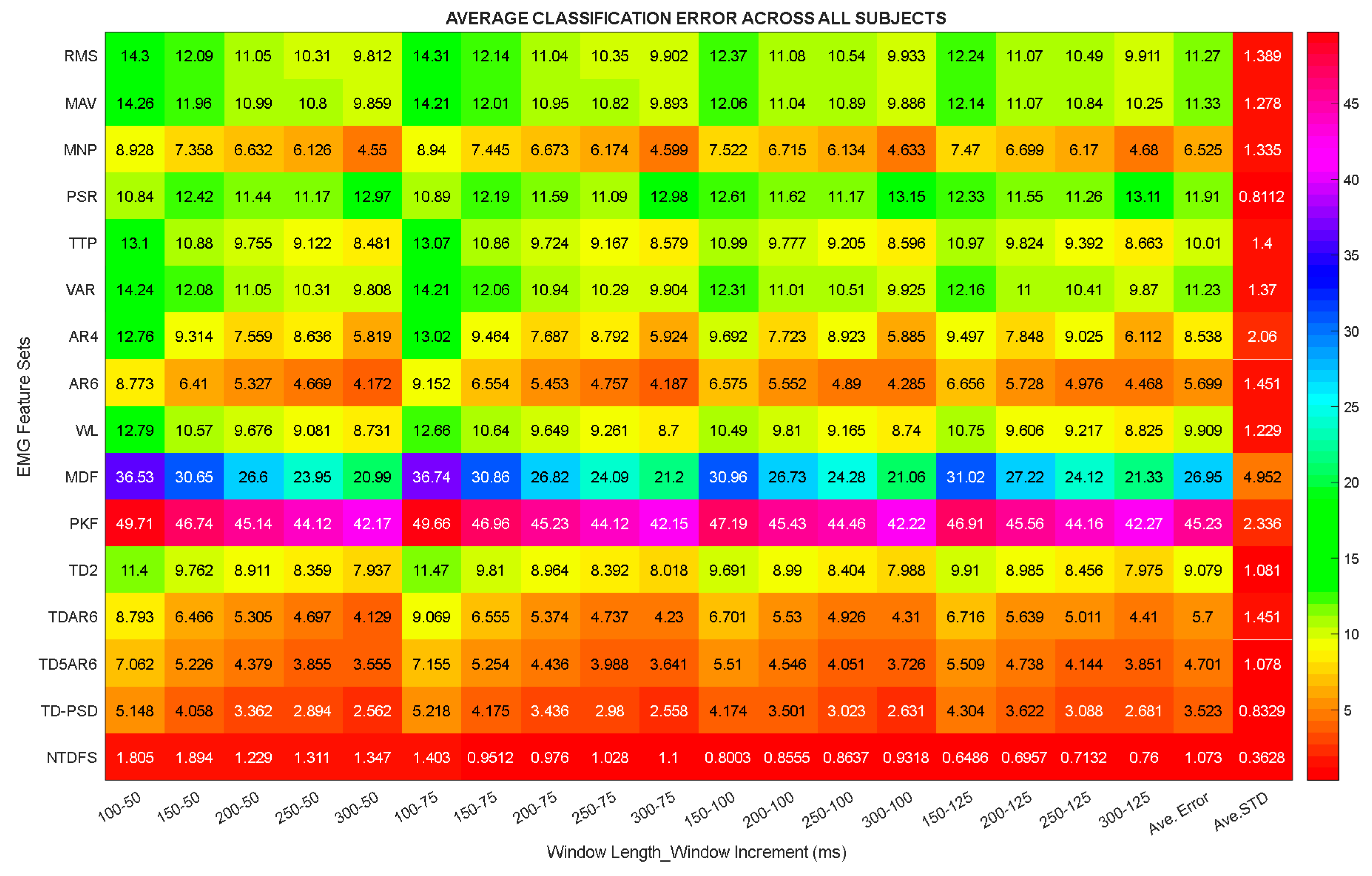
3.1. Analysis of the Feature Sets Based on Classification Error across Windowing Parameters
3.2. Analysis of the Feature Sets Based on Computation Time across Windowing Parameters
3.3. Analysis of the Feature Sets’ Data Point Characterization Using F1-Score Metric
3.4. Effect of Disturbance on the Feature Set Performance
3.5. The Effect of Number of Channels on the Feature Sets across Windowing Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Samuel, O.W.; Asogbon, M.G.; Geng, Y.; Al-Timemy, A.H.; Pirbhulal, S.; Ji, N.; Chen, S.; Fang, P.; Li, G. Intelligent EMG pattern recognition control method for upper-limb multifunctional prostheses: Advances, current challenges, and future prospects. IEEE Access 2019, 7, 10150–10165. [Google Scholar] [CrossRef]
- Geng, Y.; Ouyang, Y.; Samuel, O.W.; Chen, S.; Lu, X.; Lin, C.; Li, G. A robust sparse representation based pattern recognition approach for myoelectric control. IEEE Access 2018, 6, 38326–38335. [Google Scholar] [CrossRef]
- Khokhlova, M.; Migniot, C.; Morozov, A.; Sushkova, O.; Dipanda, A. Normal and pathological gait classification LSTM model. Artif. Intell. Med. 2019, 94, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Amsüss, S.; Goebel, P.M.; Jiang, N.; Graimann, B.; Paredes, L.; Farina, D. Self-correcting pattern recognition system of surface EMG signals for upper limb prosthesis control. IEEE Trans. Biomed. Eng. 2014, 61, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Phinyomark, A.; Khushaba, R.N.; Scheme, E. Feature extraction and selection for myoelectric control based on wearable EMG sensors. Sensors 2018, 18, 1615. [Google Scholar] [CrossRef] [PubMed]
- Samuel, O.W.; Zhou, H.; Li, X.; Wang, H.; Zhang, H.; Sangaiah, A.K.; Li, G. Pattern recognition of electromyography signals based on novel time domain features for amputees’ limb motion classification. Comput. Electr. Eng. 2018, 67, 646–655. [Google Scholar] [CrossRef]
- Bi, M. Control of Robot Arm Motion Using Trapezoid Fuzzy Two-Degree-of-Freedom PID Algorithm. Symmetry 2020, 12, 665. [Google Scholar] [CrossRef]
- Samuel, O.W.; Fang, P.; Chen, S.; Geng, Y.; Li, G. Activity recognition based on pattern recognition of myoelectric signals for rehabilitation. In Handbook of Large-Scale Distributed Computing in Smart Healthcare; Springer: Cham, Switzerland, 2017; pp. 427–442. [Google Scholar]
- Rohm, M.; Schneiders, M.; Müller, C.; Kreilinger, A.; Kaiser, V.; Müller-Putz, G.R.; Rupp, R. Hybrid brain–computer interfaces and hybrid neuroprostheses for restoration of upper limb functions in individuals with high-level spinal cord injury. Artif. Intell. Med. 2013, 59, 133–142. [Google Scholar] [CrossRef]
- Moloudi, M.; Mazinan, A.H. Controlling disturbances of islanding in a gas power plant via fuzzy-based neural network approach with a focus on load-shedding system. Complex Intell. Syst. 2019, 5, 79–89. [Google Scholar] [CrossRef]
- Samuel, O.W.; Li, X.; Geng, Y.; Asogbon, M.G.; Fang, P.; Huang, Z.; Li, G. Resolving the adverse impact of mobility on myoelectric pattern recognition in upper-limb multifunctional prostheses. Comput. Biol. Med. 2017, 90, 76–87. [Google Scholar] [CrossRef]
- Zardoshti-Kermani, M.; Wheeler, B.C.; Badie, K.; Hashemi, R.M. EMG feature evaluation for movement control of upper extremity prostheses. IEEE Trans. Rehabil. Eng. 1995, 3, 324–333. [Google Scholar] [CrossRef]
- Englehart, K.; Hudgins, B. A robust, real-time control scheme for multifunction myoelectric control. IEEE Trans. Biomed. Eng. 2003, 50, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.H.; Hargrove, L.J.; Lock, B.A.; Kuiken, T.A. Determining the optimal window length for pattern recognition-based myoelectric control: Balancing the competing effects of classification error and controller delay. IEEE Trans. Neural Syst. Rehabil. Eng. 2011, 19, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Schultz, A.E.; Kuiken, T.A. Quantifying pattern recognition based myoelectric control of multifunctional transradial prostheses. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 185–192. [Google Scholar] [PubMed]
- Menon, R.; Di, C.G.; Lakany, H.; Petropoulakis, L.; Conway, B.A.; Soraghan, J.J. Study on interaction between temporal and spatial information in classification of EMG signals for myoelectric prostheses. IEEE Trans. Neural Syst. Rehabil. Eng. 2011, 25, 1832–1842. [Google Scholar] [CrossRef] [PubMed]
- Farrell, T.R. Analysis window induced controller delay for multifunctional prostheses. Invol. Myoelectric Controls Symp. 2008, 2008, 225–228. [Google Scholar]
- Farrell, T.R.; Weir, R.F. The optimal controller delay for myoelectric prostheses. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 111–118. [Google Scholar] [CrossRef]
- Graupe, D.; Salahi, J.; Kohn, K.H. Multifunctional prosthesis and orthosis control via microcomputer identification of temporal pattern differences in single-site myoelectric signals. J. Biomed. Eng. 1982, 4, 17–22. [Google Scholar] [CrossRef]
- Graupe, D.; Salahi, J.; Zhang, D. Stochastic analysis of myoelectric temporal signatures for multifunctional single-site activation of prostheses and orthoses. J. Biomed. Eng. 1985, 7, 18–29. [Google Scholar] [CrossRef]
- Hefftner, G.; Zucchini, W.; Jaros, G.G. The electromyogram (EMG) as a control signal for functional neuromuscular stimulation. I. Autoregressive modeling as a means of EMG signature discrimination. IEEE Trans. Biomed. Eng. 1988, 35, 230–237. [Google Scholar] [CrossRef]
- Li, X.; Zhuo, Q.; Zhang, X.; Samuel, O.W.; Xia, Z.; Zhang, X.; Fang, P.; Li, G. FMG-based body motion registration using piezoelectret sensors. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Orlando, FL, USA, 16–20 August 2016; pp. 4626–4629. [Google Scholar]
- Samuel, O.W.; Asogbon, G.M.; Sangaiah, A.K.; Fang, P.; Li, G. An integrated decision support system based on ANN and Fuzzy_AHP for heart failure risk prediction. Expert Syst. Appl. 2017, 68, 163–172. [Google Scholar] [CrossRef]
- Naderpour, H.; Mirrashid, M. Moment capacity estimation of spirally reinforced concrete columns using ANFIS. Complex Intell. Syst. 2020, 6, 97–107. [Google Scholar] [CrossRef]
- Rainoldi, A.; Melchiorri, G.; Caruso, I. A method for positioning electrodes during surface EMG recordings in lower limb muscles. J. Neurosci. Methods 2004, 134, 37–43. [Google Scholar] [CrossRef]
- Kim, K.S.; Choi, H.H.; Moon, C.S.; Mun, C.W. Comparison of k-nearest neighbor, quadratic discriminant and linear discriminant analysis in classification of electromyogram signals based on the wrist-motion directions. Curr. Appl. Phys. 2011, 11, 740–745. [Google Scholar] [CrossRef]
- Oskoei, M.A.; Hu, H. Support vector machine-based classification scheme for myoelectric control applied to upper limb. IEEE Trans. Biomed. Eng. 2008, 55, 1956–1965. [Google Scholar] [CrossRef] [PubMed]
- Hudgins, B.; Parker, P.; Scott, R.N. A new strategy for multifunction myoelectric control. IEEE Trans. Biomed. Eng. 2003, 40, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Lee, S.P. EMG pattern recognition based on artificial intelligence techniques. IEEE Trans. Rehabil. Eng. 1998, 6, 400–405. [Google Scholar] [CrossRef]
- Du, S.; Vuskovic, M. Temporal vs. spectral approach to feature extraction from prehensile EMG signals. In Proceedings of the 2004 IEEE International Conference on Information Reuse and Integration, Las Vegas, NV, USA, 8–10 November 2004; pp. 344–350. [Google Scholar]
- Qingju, Z.; Zhizeng, L. Wavelet de-noising of electromyography. In Proceedings of the 2006 International Conference on Mechatronics and Automation 2006, Luoyang, Henan, China, 25–28 June 2006; pp. 1553–1558. [Google Scholar]
- Biopac Systems, Inc. Application Note 118: EMG Frequency Signal Analysis. Available online: http://www.biopac.com/Manuals/app_pdf/app118.pdf (accessed on 23 April 2020).
- Al-Timemy, A.H.; Khushaba, R.N.; Bugmann, G.; Escudero, J. Improving the performance against force variation of EMG controlled multifunctional upper-limb prostheses for transradial amputees. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 24, 650–661. [Google Scholar] [CrossRef]
- Khushaba, R.N.; Takruri, M.; Miro, J.V.; Kodagoda, S. Towards limb position invariant myoelectric pattern recognition using time-dependent spectral features. Neural Netw. 2014, 55, 42–58. [Google Scholar] [CrossRef] [PubMed]
- Philipson, L. The Electromyographic Signal Used for Control of Upper Extremity Prostheses and for Quantification of Motor Blockade during Epidural Anaesthesia. Ph.D. Dissertation, Linköping University, Linköping, Sweden, 1987. [Google Scholar]
- Samuel, O.W.; Asogbon, M.G.; Geng, Y.; Chen, S.; Feng, P.; Chuang, L.; Wang, L.; Li, G. A novel time-domain descriptor for improved prediction of upper limb movement intent in EMG-PR system. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Honolulu, HI, USA, 18–21 July 2018; pp. 3513–3516. [Google Scholar]
- Samuel, O.W.; Yang, B.; Geng, Y.; Asogbon, M.G.; Pirbhulal, S.; Mzurikwao, D.; Idowu, O.P.; Ogundele, T.J.; Li, X.; Chen, S.; et al. A new technique for the prediction of heart failure risk driven by hierarchical neighborhood component-based learning and adaptive multi-layer networks. Future Gener. Comput. Syst. 2020, 110, 781–794. [Google Scholar] [CrossRef]
- Tkach, D.; Huang, H.; Kuiken, T.A. Study of stability of time-domain features for electromyographic pattern recognition. J. Neuroeng. Rehabil. 2010, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Samuel, O.W.; Wei, Y.; Li, G. Improving the robustness of real-time myoelectric pattern recognition against arm position changes in transradial amputees. Biomed Res. Int. 2017, 2017, 5090454. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Samuel, O.W.; Huang, Z.; Fang, P.; Li, G. Improving the robustness of electromyogram-pattern recognition for prosthetic control by a postprocessing strategy. Front. Neurorobotics 2017, 11, 51. [Google Scholar] [CrossRef]
- Samuel, O.W.; Li, X.; Fang, P.; Li, G. Examining the effect of subjects’ mobility on upper-limb motion identification based on EMG-pattern recognition. In Proceedings of the 2016 Asia-Pacific Conference on Intelligent Robot Systems, Tokyo, Japan, 20–22 July 2016; pp. 137–141. [Google Scholar]
- Zhang, J.; Williams, S.O.; Wang, H. Intelligent computing system based on pattern recognition and data mining algorithms. Sustain. Comput. Inform. Syst. 2018, 20, 192–202. [Google Scholar] [CrossRef]
- Li, J.; Fong, S.; Wong, R.K.; Millham, R.; Wong, K.K. Elitist binary wolf search algorithm for heuristic feature selection in high-dimensional bioinformatics datasets. Sci. Rep. 2018, 7, 1–4. [Google Scholar] [CrossRef]
- Aborokbah, M.M.; Al-Mutairi, S.; Sangaiah, A.K.; Samuel, O.W. Adaptive context aware decision computing paradigm for intensive health care delivery in smart cities—A case analysis. Sustain. Cities Soc. 2018, 41, 919–924. [Google Scholar] [CrossRef]
- Wei, L.; Wan, S.; Guo, J.; Wong, K.K. A novel hierarchical selective ensemble classifier with bioinformatics application. Artif. Intell. Med. 2017, 83, 82–90. [Google Scholar] [CrossRef]
- Ferreri, F.; Ponz, D.; Vollero, L.; Guerra, A.; Di, P.G.; Petrichella, S.; Benvenuto, A.; Tombini, M.; Rossini, L.; Denaro, L.; et al. Does an intraneural interface short-term implant for robotic hand control modulate sensorimotor cortical integration? An EEG-TMS co-registration study on a human amputee. Restor. Neurol. Neurosci. 2014, 32, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Asogbon, M.G.; Samuel, O.W.; Geng, Y.; Oluwagbemi, O.; Ning, J.; Chen, S.; Ganesh, N.; Feng, P.; Li, G. Towards resolving the co-existing impacts of multiple dynamic factors on the performance of EMG-pattern recognition based prostheses. Comput. Methods Programs Biomed. 2020, 184, 105278. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Lin, C.; Samuel, O.W.; Xu, L.; Li, G. Continuous estimation of upper limb joint angle from sEMG signals based on SCA-LSTM deep learning approach. Biomed. Signal Process. Control 2020, 61, 102024. [Google Scholar] [CrossRef]
- Samuel, O.W.; Asogbon, M.G.; Geng, Y.; Rusydi, M.I.; Mzurikwao, Z.B.; Chen, S.; Feng, P.; Li, G. Characterizing Multiple Patterns of Motor Intent Using Spatial-Temporal Information for Intuitively Active Motor Training in Stroke Survivors. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 3831–3834. [Google Scholar]
- Asogbon, M.G.; Samuel, O.W.; Geng, Y.; Chen, S.; Mzurikwao, D.; Fang, P.; Li, G. Effect of window conditioning parameters on the classification performance and stability of EMG-based feature extraction methods. In Proceedings of the 2018 IEEE International Conference on Cyborg and Bionic Systems (CBS), Shenzhen, China, 25–27 October 2018; pp. 576–580. [Google Scholar]

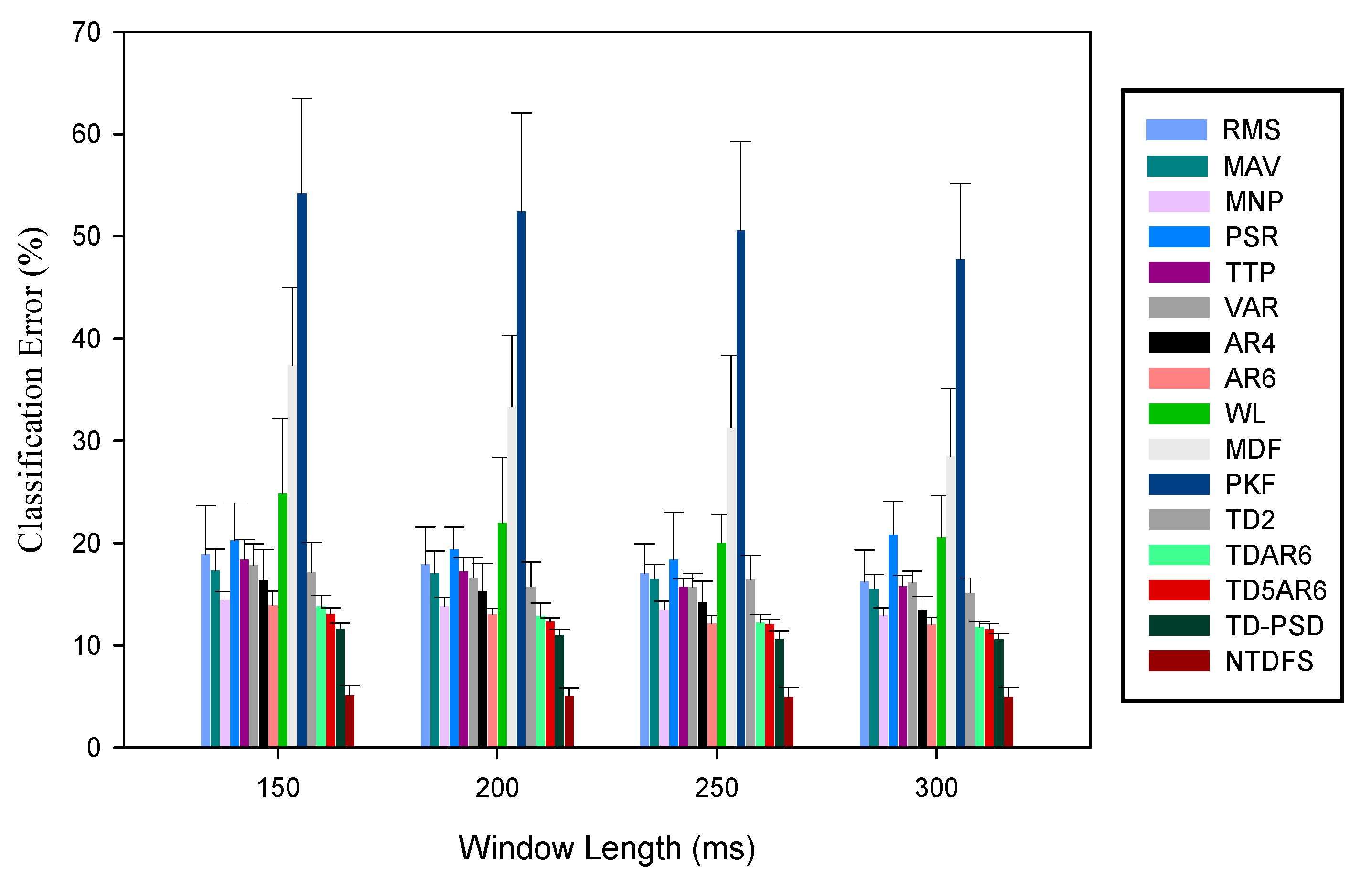
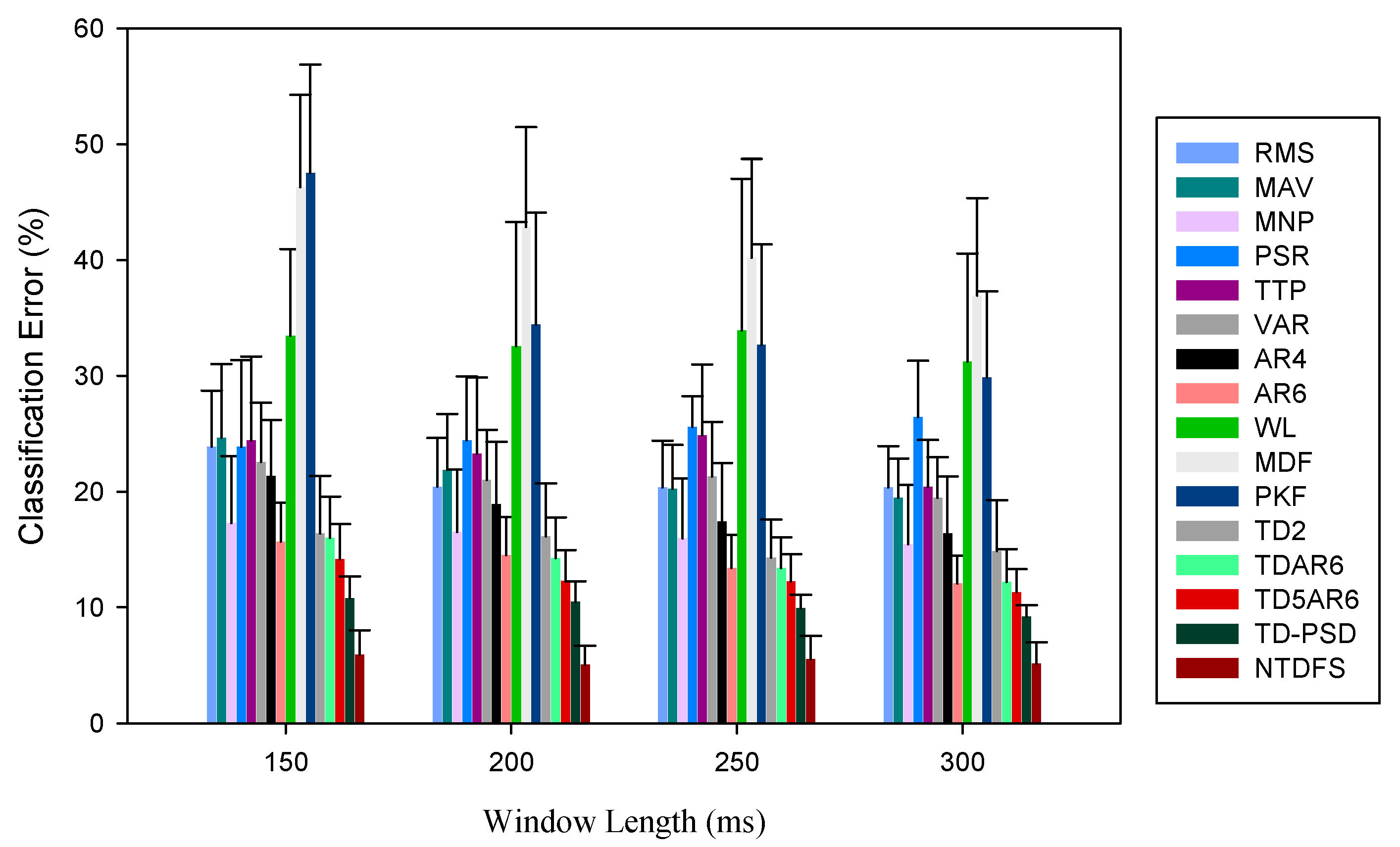
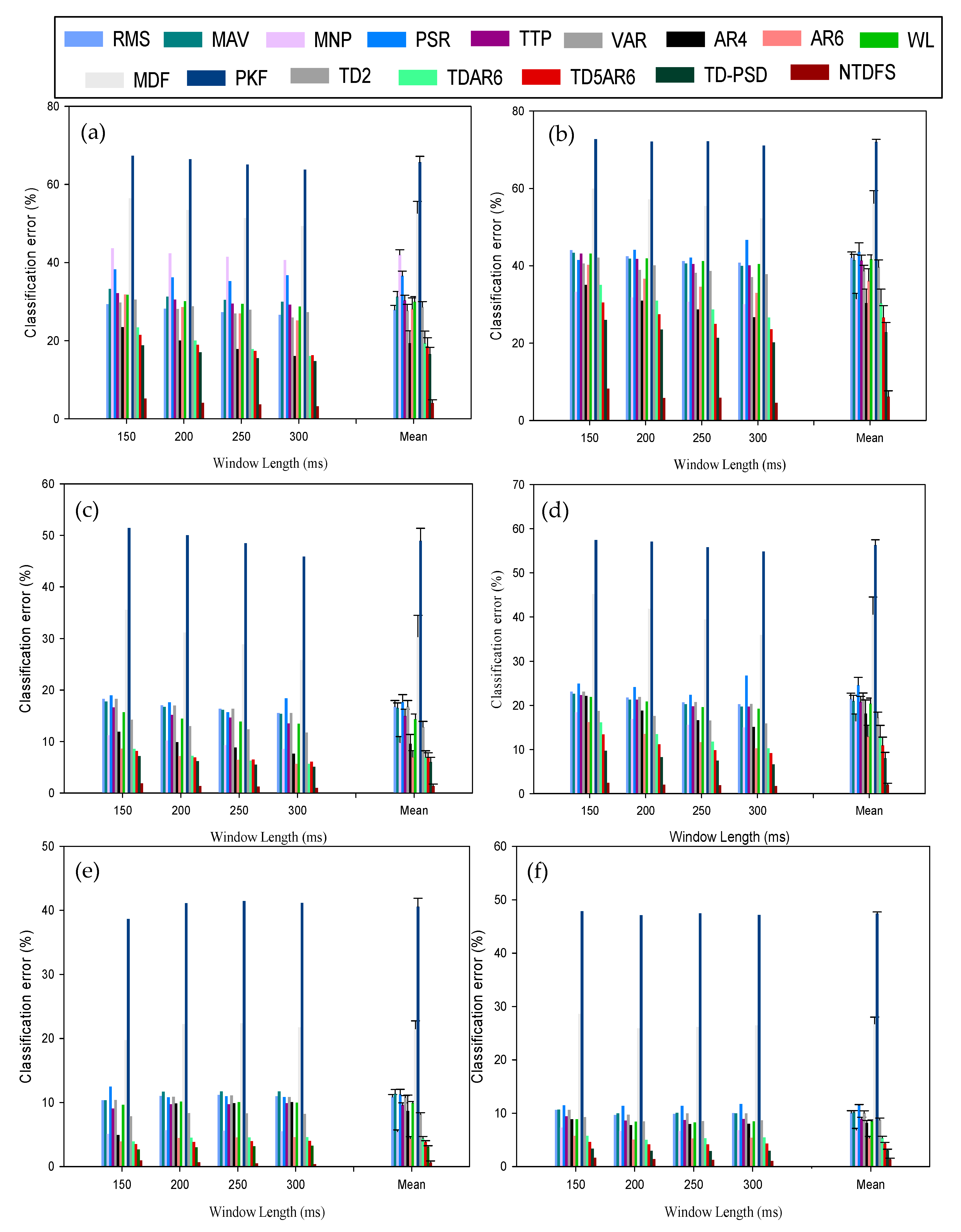
| S/No. | Window Lengths | Window Increments | |||
|---|---|---|---|---|---|
| 1 | 100 | 50 | 75 | - | - |
| 2 | 150 | 50 | 75 | 100 | 125 |
| 3 | 200 | 50 | 75 | 100 | 125 |
| 4 | 250 | 50 | 75 | 100 | 125 |
| 5 | 300 | 50 | 75 | 100 | 125 |
| Feature Extraction Methods | ||
|---|---|---|
| S/N | Features Description | Mathematical Expression |
| 1 | Root Mean Square (RMS): It is modeled as amplitude modulated Gaussian random process whose relates to constant force and non-fatiguing contraction [11,26]. | |
| 2 | Waveform Length (WL): This is the aggregate length of the EMG waveform in an analysis window [27,28]. | |
| 3 | Mean Absolute Value (MAV): is an average of absolute value of the EMG signal in an analysis time window [12,27]. | |
| 4 | Variance (VAR): measures the power of the EMG signal [12,29]. | |
| 5 | Mean Frequency (MNF): is this feature is calculated as the sum of product of the EMG power spectrum and frequency divided by the total sum of spectrum intensity [27,30]. | |
| 6 | PSR: Power Spectral Ratio(PSR)as ratio between the energy P0 which is nearby the maximum value of the EMG power spectrum and the energy P which is the whole energy of the EMG power spectrum: [31] | |
| 7 | TTP: Total Power (TTP): computes the total EMG signal power spectrum: [30,32] | O |
| 8 | PKF: Peak Frequency (PKF): this is a frequency at which the maximal power take place [32] | |
| 9 | Median Frequency (MDF):is a frequency at which the spectrum is divided into two regions with equal amplitude [27] | |
| 10 | 4th order Autoregressive Coefficient (AR4): The feature model the signal by previous data point of the EMG signal and as well gives information about the state of muscle contraction [24,27,28]. | , = 4 |
| 11 | 6th order Autoregressive Coefficient (AR6): [11,12,29]. | , = 6 |
| 12 | TD2: Summation of Square root (ASS) and absolute value of Summation of exponent root and Mean (ASM) of the data in a given analysis window [6] | ’ |
| 13 | Time Domain Power Spectral Descriptor (TD-PSD): this feature sets estimate a set of power spectrum characteristics directly from the time-domain: [33,34] | , , , |
| 14 | Four time Domain and AR6 (TDAR6): Combination of RMS and AR6 [23,24,26,27] | MS and AR6 |
| 15 | Five time Domain and AR6 (TD5AR6): combination of MAV, RMS, WL, ZC, SSC and AR6(ZC: Zero Crossing, SSC: Slope Sign Change) [11,12,26,27,28,29,35] | , ZC = |
| 16 | Novel Time Domain Feature Set (NTDFS), that combined neuromuscular information for adequate characterization of EMG signal patterns even in the presence of co-founding factors [36] | , , |
| S/No. | Window Length Feature Sets  | 150 | 200 | 250 | 300 | Mean ± SD |
|---|---|---|---|---|---|---|
| 1 | RMS | 0.8829 | 0.8978 | 0.9046 | 0.9096 | 0.8987 ± 0.0116 |
| 2 | MAV | 0.8862 | 0.8978 | 0.9039 | 0.9103 | 0.8995 ± 0.0103 |
| 3 | MNP | 0.9167 | 0.9269 | 0.9343 | 0.9392 | 0.9293 ± 0.0098 |
| 4 | PSR | 0.8750 | 0.8843 | 0.8877 | 0.8647 | 0.8779 ± 0.0103 |
| 5 | TTP | 0.8955 | 0.9089 | 0.9157 | 0.9228 | 0.9107 ± 0.0116 |
| 6 | VAR | 0.8837 | 0.8985 | 0.9039 | 0.9099 | 0.8990 ± 0.0112 |
| 7 | AR4 | 0.8813 | 0.9086 | 0.9204 | 0.9324 | 0.9107 ± 0.0219 |
| 8 | AR6 | 0.9253 | 0.9387 | 0.9477 | 0.9550 | 0.9417 ± 0.0128 |
| 9 | WL | 0.9053 | 0.9131 | 0.9204 | 0.9242 | 0.9157 ± 0.0084 |
| 10 | MDF | 0.6654 | 0.7083 | 0.7384 | 0.7759 | 0.7220 ± 0.0468 |
| 11 | PKF | 0.4987 | 0.5109 | 0.5223 | 0.5376 | 0.5174 ± 0.0165 |
| 12 | TD2 | 0.9014 | 0.9093 | 0.9171 | 0.9220 | 0.9124 ± 0.0090 |
| 13 | TDAR6 | 0.9229 | 0.9394 | 0.9456 | 0.9546 | 0.9406 ± 0.0133 |
| 14 | TD5AR6 | 0.9390 | 0.9517 | 0.9586 | 0.9619 | 0.9528 ± 0.0101 |
| 15 | TD-PSD | 0.9559 | 0.9657 | 0.9714 | 0.9753 | 0.9671 ± 0.0084 |
| 16 | NTDFS | 0.9904 | 0.9934 | 0.9952 | 0.9964 | 0.9938 ± 0.0026 |
| S/No. | Window Length Feature Sets  | 150 | 200 | 250 | 300 | Mean ± SD |
|---|---|---|---|---|---|---|
| 1 | RMS | 0.8816 | 0.8912 | 0.8948 | 0.9014 | 0.8922 ± 0.0083 |
| 2 | MAV | 0.8852 | 0.8931 | 0.8979 | 0.9034 | 0.8949 ± 0.0077 |
| 3 | MNP | 0.9366 | 0.9422 | 0.9461 | 0.9497 | 0.9436 ± 0.0056 |
| 4 | PSR | 0.8761 | 0.8870 | 0.8919 | 0.8755 | 0.8826 ± 0.0081 |
| 5 | TTP | 0.8958 | 0.9057 | 0.9097 | 0.9143 | 0.9064 ± 0.0079 |
| 6 | VAR | 0.8819 | 0.8919 | 0.8960 | 0.9013 | 0.8928 ± 0.0082 |
| 7 | AR4 | 0.9262 | 0.9384 | 0.9458 | 0.9514 | 0.9405 ± 0.0109 |
| 8 | AR6 | 0.9065 | 0.9198 | 0.9287 | 0.9346 | 0.9224 ± 0.0122 |
| 9 | WL | 0.9100 | 0.9100 | 0.9200 | 0.9200 | 0.9150 ± 0.0058 |
| 10 | MDF | 0.6703 | 0.7199 | 0.7415 | 0.7671 | 0.7247 ± 0.0411 |
| 11 | PKF | 0.5495 | 0.5743 | 0.5835 | 0.6141 | 0.5804 ± 0.0267 |
| 12 | TD2 | 0.9123 | 0.9184 | 0.9221 | 0.9256 | 0.9196 ± 0.0057 |
| 13 | TDAR6 | 0.9449 | 0.9517 | 0.9559 | 0.9604 | 0.9532 ± 0.0066 |
| 14 | TD5AR6 | 0.9516 | 0.9583 | 0.9612 | 0.9643 | 0.9589 ± 0.0054 |
| 15 | TD-PSD | 0.9622 | 0.9656 | 0.9694 | 0.9730 | 0.9676 ± 0.0047 |
| 16 | NTDFS | 0.9830 | 0.9863 | 0.9877 | 0.9895 | 0.9866 ± 0.0028 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asogbon, M.G.; Samuel, O.W.; Jiang, Y.; Wang, L.; Geng, Y.; Sangaiah, A.K.; Chen, S.; Fang, P.; Li, G. Appropriate Feature Set and Window Parameters Selection for Efficient Motion Intent Characterization towards Intelligently Smart EMG-PR System. Symmetry 2020, 12, 1710. https://doi.org/10.3390/sym12101710
Asogbon MG, Samuel OW, Jiang Y, Wang L, Geng Y, Sangaiah AK, Chen S, Fang P, Li G. Appropriate Feature Set and Window Parameters Selection for Efficient Motion Intent Characterization towards Intelligently Smart EMG-PR System. Symmetry. 2020; 12(10):1710. https://doi.org/10.3390/sym12101710
Chicago/Turabian StyleAsogbon, Mojisola Grace, Oluwarotimi Williams Samuel, Yanbing Jiang, Lin Wang, Yanjuan Geng, Arun Kumar Sangaiah, Shixiong Chen, Peng Fang, and Guanglin Li. 2020. "Appropriate Feature Set and Window Parameters Selection for Efficient Motion Intent Characterization towards Intelligently Smart EMG-PR System" Symmetry 12, no. 10: 1710. https://doi.org/10.3390/sym12101710
APA StyleAsogbon, M. G., Samuel, O. W., Jiang, Y., Wang, L., Geng, Y., Sangaiah, A. K., Chen, S., Fang, P., & Li, G. (2020). Appropriate Feature Set and Window Parameters Selection for Efficient Motion Intent Characterization towards Intelligently Smart EMG-PR System. Symmetry, 12(10), 1710. https://doi.org/10.3390/sym12101710







