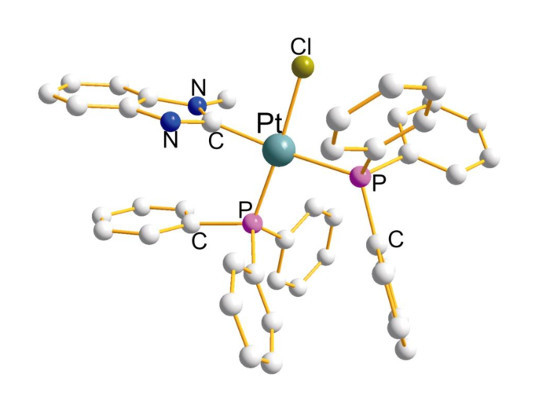
Organophosphines in Cis-PtP2CCl Derivatives Structural Aspects
Abstract
1. Introduction
2. Cis-PtP2CCl Derivatives
2.1. Cis-Pt(η1-PL)2(η1-CL)(η1-Cl)
2.2. Cis-Pt(η2-P2L)(η1-CL)(η1-Cl)
- four-membered: 1.98 Å (C, trans to P) < 2.227 Å (P, trans to Cl) < 2.295 Å (P, trans to C) < 2.365 Å (Cl, trans to P);
- five-membered: 2.11 Å (C, trans to P) < 2.207 Å (P, trans to Cl) < 2.295 Å (P. trans to C) < 2.360 Å (Cl, trans to P);
- six-membered: 2.105 Å (C, trans to P) < 2.232 Å (P, trans to Cl) < 2.318 Å (P, trans to C) < 2.360 Å (Cl, trans to P);
- seven-membered: 2.14 Å (C, trans to P) < 2.230 Å (P, trans to Cl) < 2.326 Å (P, trans to C) < 2.362 Å (Cl, trans to P).
2.3. Cis-Pt(η1-PL)(η2-P,CL)(η1-Cl)
3. Conclusions
- -
- Pt(η1-PL)2(η1-CL)(η1-Cl) (17 examples);
- -
- Pt(η2-P2L)(η1-CL)(η1-Cl) (43 examples);
- -
- Pt(η1-PL)(η2-P,CL)(η1-Cl) (3 examples).
- -
- Pt-P (trans to X): 2.214 Å (O) < 2.220 Å (F) < 2.222 Å (Cl) < 2.240 Å (N) < 2.300 Å (C) < 2.323 Å (H);
- -
- Pt-X (trans to P): 1.65 Å (H) < 2.02 Å (F) < 2.07 Å (N) < 2.075 Å (O) < 2.222 Å (Cl)
Acknowledgments
Conflicts of Interest
Abbreviations
| But2P(η2-C7H6)PBut2 | di-t-butyl(2-di-t-butylphosphino)benzyl)phosphine |
| C2H6ClO2 | chloro(methoxycarbonyl)methyl |
| C3H4PPh2 | 1-(diphenylphosphino)propenyl |
| C4H8O | tetrahydrofuran |
| C5H2Ph2 | 2-(2,3-diphenylcycloprop-2-en-1-yl)vinyl |
| (C5H10N)2P(η2-C5H8)P(NC5H10)2 | 1,2-bis(bis(piperidin-1-yl)phosphorino)cyclopentane |
| (η2-C6H12)P(CH2)2P(η2-C6H12) | 1,1′-ethane-1,2-diylbis(2,5-dimethylphospholane) |
| (η2-C6H12)P(η2-C6H4)P(η2-C6H12) | 1,2-bis((hexa-2,5-diyl)phosphino)benzene |
| C7H13O2 | 1-(tert-butoxycarbinol)ethyl |
| C8H12N2 | 1-methylbenzimidazol-2-ylidene |
| C8H15N2 | 3-((dimethylamino)methylene)-1-methylpyrrolidin-2-ylidene |
| C8H3F6 | 2,5-bis(trifluoromethyl)phenyl |
| C8H5BN(SiMe3)2 | 1-(bis(trimethylsilyl)amino)-3-phenyl-1H-borizen-2-yl |
| (η2-C10H16O3)P(η2-C7H6)P(η2-C10H16O3) | bis(1,3,5,7-tetramethyl-2,4,6-trioxa-8-phosphatricyclo[3.3.1.13,7]dec-8-yl)benzyl |
| (η2-C10H20)P(η2-C6H4)P(η2-C10H20) | 1,1′-(1,2-phenylene)bis(2,5-diisopropylphospholane) |
| C10H11ClN2SSi | 1-(chloro(phenyl)sulfidosilyl)-3-methylimidazolin-2-ylidene |
| C11H19N2 | 2,5-bis((dimethylamino)methylene)cyclopentylidene |
| (C12H4F13)2P(CH2)3P(C12H4F13)2 | propane-1,3-diyl(bis(4-tridecafluorohexyl)phenyl |
| (η2-C16H16)P(CH2)2(η2-C16H16) | 1,1′-ethane-1,2-diylbis(2,5-diphenylphospholane) |
| η2-C19H16P | 2-(diphenylphosphino)-5-methylphenyl |
| η2-C24H32PSi2 | 1-methyl-2,6-bis(trimethylsilyl)-3,5-diphenylphosphinine |
| cppc | 1-[chloro(piperidin-1-yl)phosphino]-2-[bis(piperidin-1-yl) phosphino]cyclopentane |
| diop | Ph2PCH2CHOC(H2)COCHCH2PPh2 |
| P(NC5H4OS)Ph2 | 2-(diphenylphosphino)-2-thienylcarboxamide |
| P(η2-C7H5NO)Ph | 3-phenyl-1,3-dihydro-2H-1,3-benzaphosphol-2-one |
| Ph2P(η2-C10H14N2)PPh2 | (3-(adenin-9-yl)propyl)bi(diphenylphosphinomethyl) amine |
| Ph2P(η2-C21H21N3)PPh2 | N,N′-dibenzyl-N,N′-bis((diphenylphosphino)methyl)pyridine-2,6-diamine |
| Ph2P(η2-C5H8)PPh2 | 1,2-bis(diphenylphosphino)cyclopentane |
| Ph2PCH2(Me)CH2(Me)PPh2 | 2,3-bis(diphenylphosphino)butane |
| POP | N,N′,N″,N‴-tetra-tert-butyl-N,N″-bis(trimethylsilyl) tetraamidodiphosphit |
| tbtpb | 2,4,6-Tri-t-butyl-2,4-dihydro-5-hydroxy-1,3-di-oxo-1,3,5-triphosphabenzene-P,C |
References
- Melník, M.; Mikuš, P. Organophosphines in organoplatinum complexes structural aspects of PtPC3 and PtP2CX (X = H, F or OL) derivatives. J. Organomet. Chem. 2016, 811, 1–7. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Organophosphines in organoplatinum complexes: Structural aspects of PtP2CX (X = NL or BL) derivatives. J. Organomet. Chem. 2016, 813, 78–83. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Organophosphines in organoplatinum complexes—Structural aspects of trans-PtP2CCl derivatives. J. Organomet. Chem. 2016, 819, 46–52. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Organophosphines in organoplatinum complexes—Structural aspects OF PtP2CX (X = Br or I) derivatives. J. Organomet. Chem. 2016, 823, 97–102. [Google Scholar] [CrossRef]
- Braunschweig, H.; Ye, Q.; Radacki, K. Borylene-based functionalization of Pt–alkynyl complexes by photochemical borylene transfer from [(OC)5Cr=BN(SiMe3)2]. Chem. Commun. 2009, 6979–6981. [Google Scholar] [CrossRef] [PubMed]
- Jacob, V.; Weakley, T.J.R.; Haley, M.M. Rearrangement of a σ-2-(Cycloprop-2-enyl)vinyl- to an η3-Cyclopentadienylplatinum(II) Complex. Selective Protonolysis of the Platinum−Methyl Bond. Organometallics 2002, 21, 5394–5401. [Google Scholar] [CrossRef]
- Bardi, R.; Del Pra, A.; Piazzesi, A.M.; Minniti, D.; Romeo, R. Crystal and Molecular Structure of di(triethylphosphino)(ethyl)chloride platinum(II) complex. Cryst. Struct. Commun. 1981, 10, 333–335. [Google Scholar]
- Bresciani-Pahor, N.; Plazzotta, M.; Randaccio, L.; Bruno, G.; Ricevato, V.; Romeo, R.; Belluco, U. Crystal and molecular structure of cis-chloro-p-tolyl-bis(triethylphosphine)platinum(II) and of cis-chloro-perfluorophenyl-bis(triethylphosphine)platinum(II). Inorg. Chim. Acta 1978, 31, 171–175. [Google Scholar] [CrossRef]
- Stienborn, D.; Becke, S.; Bruhn, C.; Heinemann, F.W. Platin(II)-Komplexe mit schwefelfunktionalisierten Alkenylliganden. J. Oragnomet. Chem. 1998, 556, 189–196. [Google Scholar] [CrossRef]
- Russell, D.R.; Tucker, P.A. Crystal and molecular structure of cis-chloro(3-chloro-1,1,3,3-tetrafluoropropan-2-one)bis(triphenylphosphine)platinum(II). J. Chem. Soc. Dalton Trans. 1975, 2222–2225. [Google Scholar] [CrossRef]
- Engelter, C.; Moss, J.R.; Niven, M.L.; Nassimbeni, L.R.; Reid, G. A cationic ylide complex of platinum(ii): Its structure and formation from a chloromethyl-platinum complex. J. Organomet. Chem. 1982, 232, C78–C80. [Google Scholar] [CrossRef]
- Engelter, C.; Moss, J.R.; Nassimbeni, L.R.; Niven, M.L.; Reidaud, G.; Spiers, J.C. Haloalkyl complexes of the transition metals: IV. The formation of a cationic ylide complex of platinum(II) from a chloromethylplatinum(II) complex and the crystal structures of cis-[Pt(CH2PPh3)X(PPh3)2]I (X = Cl or I). J. Organomet. Chem. 1986, 315, 255–268. [Google Scholar] [CrossRef]
- Henderson, W.; Fawcett, J.; Kemmitt, R.D.W.; McKenna, P.; Russell, D.R. Decomposition pathways of platinum(II) complexes containing alkyl and halide ligands investigated by electrospray mass spectrometry. The X-ray crystal structure of cis-[PtCl{CH2C(O)CH2Cl}(PPh3)2]. Polyhedron 1997, 16, 2455–2463. [Google Scholar] [CrossRef]
- Baukova, T.V.; Oleinikova, N.A.; Kuzmina, L.G. Synthesis, structure, and properties of organoplatinum(ii) derivatives of diphenyl ether. Izv. Akad. Nauk SSSR Ser. Khimia 1994, 43, 1719–1725. [Google Scholar] [CrossRef]
- Alcarazo, M.; Radkowski, K.; Goddard, R.; Furstner, A. Metal complexes with carbene ligands stabilized by lateral enamines. Chem. Commun. 2011, 47, 776–778. [Google Scholar] [CrossRef] [PubMed]
- Brendler, E.; Hill, A.F.; Wagler, J. A Donor-Stabilized Silanethione or a Si-Substituted N-Heterocyclic Platinum. Chem. Eur. J. 2008, 14, 11300–11304. [Google Scholar] [CrossRef] [PubMed]
- Kosterke, T.; Pape, T.; Hahn, F.E. Synthesis of NHC Complexes by Oxidative Addition of 2-Chloro-N-methylbenzimidazole. J. Am. Chem. Soc. 2011, 133, 2112–2115. [Google Scholar] [CrossRef] [PubMed]
- Pelczar, E.M.; Nytko, E.A.; Zhuravel, M.A.; Smith, J.M.; Glueck, D.C.; Sommer, R.; Incarvito, C.D.; Rheingold, A.L. Synthesis and structure of platinum and palladium complexes of dimesitylphosphine. Polyhedron 2002, 21, 2409–2419. [Google Scholar] [CrossRef]
- Johnson, D.K.; Rukaschaisirikul, T.; Sun, Y.; Taylor, N.J.; Canty, A.J.; Carty, A.J. Spectroscopic Properties of Inorganic and Organometallic Compounds. Inorg. Chem. 1993, 32, 5544–5549. [Google Scholar] [CrossRef]
- Milton, H.L.; Wheatley, M.V.; Slawin, A.M.Z.; Woollins, J.D. Synthesis and coordination of 2-diphenylphosphinothiophenocarboxamide and bis (2,5-diphenylphosphinepicolinamide). Polyhedron 2004, 23, 2575–2585. [Google Scholar] [CrossRef]
- Bennett, J.; Doyle, R.J.; Lee, H.Y.; Lu, D.; Salem, G.; Speldewinde, D.J.; Tifan, M.; Willis, A.C. Synthesis of 1,3-azaphosphol-2-ones. Crystal and molecular structures of [SP-4-2]-dichlorobis (3-phenyl-1,3-dihydrobenzo[1,3]azaphosphol-2-one-P)palladium(II) and its chloro(methyl)platinum(II) analogue. Dalton Trans. 2010, 39, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Scherer, O.J.; Konrad, R.; Guggolz, E.; Ziegler, M.L. Elementorganische Amin/Imin-Verbindungen, XXIV. Tris{(tert-butylimino)[tert-butyl(trimethylsilyl)amino]-phosphan}platin(0)– ein wertvoller Synthesebaustein. Chem. Ber. 1983, 116, 2676–2690. [Google Scholar] [CrossRef]
- Deacon, G.B.; Nelson, K.T.; Tiekink, E.R.T. [Bis(diphenylphosphino)methane-P,P’](chloro)(pentafluorophenyl)platinum(II): PtCl(dppm)C6F5. Acta Crystallogr. Sect. C 1991, 47, 955–957. [Google Scholar] [CrossRef]
- Iverson, C.N.; Lachicotte, R.J.; Muller, C.; Jones, W.D. η2-Coordination and C−H Activation of Electron-Poor Arenes. Organometallics 2002, 21, 5320–5333. [Google Scholar] [CrossRef]
- Del Pra, A.; Zanotti, G.; Prazzesi, A.; Belluco, U.; Ros, R. The crystal and molecular structure of PtCl(CF3)(cis-Ph2PCH=CHPPh2). Trans. Met. Chem. 1979, 4, 381–384. [Google Scholar] [CrossRef]
- Calligarlies, M.; Carturan, G.; Nardin, G.; Scrivanti, A.; Wojcicki, A. Metal complexes with carbene ligands stabilized by lateral enamines. Organometallics 1983, 2, 865–874. [Google Scholar]
- Hughes, R.P.; Ward, A.J.; Golen, J.A.; Incarvito, C.O.; Rheingold, A.L.; Zakharov, L.N. Synthesis, molecular structures, and chemistry of some new palladium(II) and platinum(II) complexes with pentafluorophenyl ligands. Dalton Trans. 2004, 2720–2727. [Google Scholar] [CrossRef] [PubMed]
- Schwatz, B.D.; Brennessel, W.W.; Jones, W.D. C−CN vs. C−H Cleavage of Benzonitrile Using [(dippe)PtH]2. Organometallics 2011, 30, 1523–1529. [Google Scholar]
- Deacon, G.B.; Elliott, P.W.; Erven, A.P.; Meyer, G. The Synthesis by Decarboxylation Reactions and Crystal Structures of 1, n–Bis(diphenylphosphino)alkane(pentafluorophenyl)-platinum(II) Complexes. Z. Anorg. Allg. Chem. 2005, 631, 843–850. [Google Scholar] [CrossRef]
- Wicht, D.K.; Glueck, D.S.; Liable-Sands, L.M.; Rheingold, A.L. Terminal Platinum(II) Perfluoroacyl Phosphido Complexes: Synthesis and Dynamic Processes. Organometallics 1999, 18, 5130–5140. [Google Scholar] [CrossRef]
- You, Y.J.; Chen, J.T.; Cheng, M.C.; Wang, Y. Solvent-controlled equilibria of substitution and isomerization of.alpha.-ketoacyl complexes of platinum(II). X-ray single-crystal structure of cis-Pt(COCOPh)(Cl)(PPh2CH2CH2PPh2). Inorg. Chem. 1991, 30, 3621–3625. [Google Scholar] [CrossRef]
- Scriban, C.; Glueck, D.S.; Zakharov, L.N.; Kassel, W.S.; DiPasquale, A.G.; Golen, J.A.; Rheingold, A.L. P−C and C−C Bond Formation by Michael Addition in Platinum-Catalyzed Hydrophosphination and in the Stoichiometric Reactions of Platinum Phosphido Complexes with Activated Alkenes. Organometallics 2006, 25, 5757–5767. [Google Scholar] [CrossRef]
- Bennett, M.A.; Kwan, L.; Rae, A.D.; Wenger, E.; Willis, A.C. Preparation and reactivity of mononuclear platinum(0) complexes containing a η2-coordinated alkynylphosphine. J. Chem. Soc. Dalton Trans. 2002, 226–233. [Google Scholar] [CrossRef]
- Johansson, M.H.; Malmstrom, T.; Wendt, O.F. Chiral platinum(II) complexes. Crystal and molecular structures of cis-[PtPhCl((R,R)-CHIRAPHOS)] and cis-[PtCl2((R,R)-CHIRAPHOS)]. Inorg. Chim. Acta. 2001, 316, 149–152. [Google Scholar] [CrossRef]
- Ferguson, G.; Gallagher, J.F.; McAlees, A.J.; McCrindle, P. Mechanism and Stereochemistry of the Reaction of Dichloroplatinum(II) Complexes with Diazo Compounds. X-ray Structures of Four Key Products: [(2R,3R)-Bis(diphenylphosphino)butane]chloro-[(S)-chloro(methoxycarbonyl)methyl]platinum(II), (η4-1,5-Cyclooctadiene)[3-chloro-5-(dimethoxyphosphonyl)-2-methoxy-4,1,2-platinaoxaphospholane P-oxide],(R,R)-[Chloro(dimethoxyphosphonyl)methyl][chloro-(trimethylsilyl)methyl] (1,5-cyclooctadiene) platinum(II), and Chloro[chloro(dimethoxyphosphonyl)methyl](η4-1,5-cyclooctadiene)platinum(II). Organometallics 1997, 16, 1053–1062. [Google Scholar]
- Argazzi, R.; Bergamini, P.; Costa, E.; Gee, V.; Hogg, J.K.; Martin, A.; Orpen, A.G.; Pringle, P.G. Anchimeric Assistance by Platinum(II) in the Epimerizations of [PtX(CHXSiMe3)(R,R-chiraphos)]. Organometallics 1996, 15, 5591–5597. [Google Scholar] [CrossRef]
- Dahlenburg, L.; Mertel, S. Chiral bisphosphanes. V. Chloro{rac,trans-1-[chloro(piperidin-1-yl) phosphino]-2-[bis(piperidin-1-yl)phosphino]cyclopentane-P,P’}neopentylplatinum(II) acetone solvate. Acta. Crystallogr. Sect. C 1999, 55, 347–349. [Google Scholar] [CrossRef]
- Squires, M.E.; Sardella, D.J.; Kool, L.B. C-H Activation by [Bis(dialkoxyphosphino)ethane]platinum(0) and [Bis(diaminophosphino)ethane]platinum(0) Complexes: Platinum-Platinum Dimer Formation Limits Yields. Organometallics 1994, 13, 2970–2978. [Google Scholar] [CrossRef]
- Dahlenburg, L.; Mertel, S. Chiral chelate phosphanes: XI. Application of cyclopentane-based C2 chiral bis(phosphane) ligands C5H8(PR2)2 to Pt Sn-catalyzed styrene hydroformylation. J. Organomet. Chem. 2001, 630, 221–243. [Google Scholar] [CrossRef]
- Quino-o, M.A.; Zureick, A.H.; Blank, N.F.; Anderson, B.J.; Chapp, T.W.; Kim, Y.; Glueck, D.S.; Rheingold, A.L. Synthesis and Structure of Platinum Bis(phospholane) Complexes Pt(diphos*)(R)(X), Catalyst Precursors for Asymmetric Phosphine Alkylation. Organometallics 2012, 31, 6900–6910. [Google Scholar] [CrossRef]
- Scriban, C.; Wicht, D.K.; Glueck, D.S.; Zakharov, L.N.; Golen, J.A.; Rheingold, A.L. Platinum(II) Phosphido Complexes as Metalloligands. Structural and Spectroscopic Consequences of Conversion from Terminal to Bridging Coordination. Organometallics 2006, 25, 3370–3378. [Google Scholar] [CrossRef]
- Brunker, T.J.; Blank, N.F.; Moncarz, J.R.; Scriban, C.; Anderson, B.J.; Glueck, D.S.; Zakharov, L.N.; Golen, J.A.; Sommer, R.D.; Incarvito, C.D. Chiral Palladium(0) trans-Stilbene Complexes: Synthesis, Structure, and Oxidative Addition of Phenyl Iodide. Organometallics 2005, 24, 2730–2746. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Sostero, S.; Ganter, C.; Hogg, J.; Orpen, A.G.; Pringle, P.G. The reactions of Me3SiCHN2 with [PtX2(S,S-skewphos)]: Highly diastereoselective carbene insertions into Pt Cl bonds, and Me3SiCHN2 as a CH2N2 equivalent. J. Organomet. Chem. 1993, 455, C13–C16. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Orpen, A.G.; Ganter, C.; Pringle, P.G. Highly stereospecific carbene insertions into platinum–halide bonds. Crystal structure of [PtCl(S-CHClSiMe3)(S,S-Ph2PCHMeCH2CHMePPh2)]. J. Chem. Soc. Dalton Trans. 1994, 651–655. [Google Scholar] [CrossRef]
- Berven, B.M.; Kousantonis, G.A.; Skelton, B.W.; Trengove, R.D.; White, A.H. Highly fluorous complexes of nickel, palladium and platinum: Solubility and catalysis in high pressure CO2. Dalton Trans. 2011, 4167–4174. [Google Scholar] [CrossRef] [PubMed]
- Fanjul, T.; Eastham, G.; Floure, J.; Forrest, S.J.K.; Haddow, M.F.; Hamilton, A.; Pringle, P.G.; Golen, A.G.; Waugh, M. Interplay of bite angle and cone angle effects. A comparison between o-C6H4(CH2PR2)(PR′2) and o-C6H4(CH2PR2)(CH2PR′2) as ligands for Pd-catalysed ethene hydromethoxycarbonylation Dalton Trans. Dalton Trans. 2013, 42, 100–115. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Hua, G.; Bhattacharyya, P.; Slawin, A.M.Z.; Woollins, J.D. Syntheses and Coordination Chemistry of Aminomethylphosphine Derivatives of Adenine. Eur. J. Inorg. Chem. 2003, 2426–2437. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Sostero, S.; Orpen, A.G.; Pringle, P.G. Convenient synthesis of. alpha.-chiral platinum alkyls. Organometallics 1991, 10, 2989–2990. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Sostero, S.; Orpen, A.G.; Pringle, P.G. Diastereoselective carbene insertions into platinum-halogen bonds. Crystal structure of [PtCl(R-CHClCO2Et)(R,R-diop)]. Organometallics 1992, 11, 3879–3885. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Cramer, D.; Hogg, J.; Orpen, A.G.; Pringle, P.G. Diastereoselective C-C Bond Formation by Carbene Insertions into Pt-CH3 Bonds. Organometallics 1994, 13, 1058–1060. [Google Scholar] [CrossRef]
- Bergamini, P.; Costa, E.; Orpen, A.G.; Pringle, P.G.; Smith, M.B. Reactions of Diazo Carbonyls with [PtX(CH3)(chiral diphosphine)] (X = Cl, Br, I): Chemoselectivity and Diastereoselectivity of Pt-C and Pt-X Carbene Insertion. Organometallics 1995, 14, 3178–3186. [Google Scholar] [CrossRef]
- Wang, Z.J.; Wang, X.X.; Wan, C.Q. Chlorido{N2,N6-dibenzyl-N2,N6-bis[(diphenylphosphanyl)methyl]pyridine-2,6-diamine}methylplatinum(II). Acta Crystallogr. Sect. E Struct. Rep. Online 2010, 66, m1341–m1342. [Google Scholar] [CrossRef] [PubMed]
- Moores, A.; Mezailles, N.; Ricard, L.; Jean, Y.; LeFloch, P. η2-Palladium and Platinum(II) Complexes of a λ4-Phosphinine Anion: Syntheses, X-ray Crystal Structures, and DFT Calculations. Organometallics 2004, 23, 2870–2875. [Google Scholar] [CrossRef]
- Bennett, M.A.; Bhargava, S.K.; Priver, S.H.; Willis, A.C. Selective Cleavage by Acids of One Metal–Carbon σ-Bond of a Bis(ortho-platinated) Triarylphosphane: A 31P NMR trans-Influence Series Based on the Unit Pt(κ2-C6H3-5-Me-2-PPh2)(PPh2-4-tol). Eur. J. Inorg. Chem. 2008, 3467–3483. [Google Scholar] [CrossRef]
- Clendenning, S.B.; Hitchcock, P.B.; Nixon, J.F. First η1-ligated 2,4,6-tri-tert-butyl-1,3,5-triphosphabenzene complexes and the remarkable trihydration reaction of trans-[PtCl2(PMe3)(P3C3But3)] to cis-[PtCl(PMe3)(P3O3C3H5But3)], containing the novel CH(But)PH(O)C(But)PH(O)CH(But)P(O) ring system. Chem. Commun. 1999, 1377–1381. [Google Scholar] [CrossRef]
- Melník, M. Structural isomerism of copper(II) compounds. Coord. Chem. Rev. 1982, 47, 239–261. [Google Scholar] [CrossRef]
- Melník, M.; Holloway, C.E. Stereochemistry of platinum coordination compounds. Coord. Chem. Rev. 2006, 250, 2261–2270. [Google Scholar] [CrossRef]
- Melnik, M.; Mikus, P. Stereoisomers of organoplatinum complexes. J. Organomet. Chem. 2015, 239, 779–780. [Google Scholar] [CrossRef]
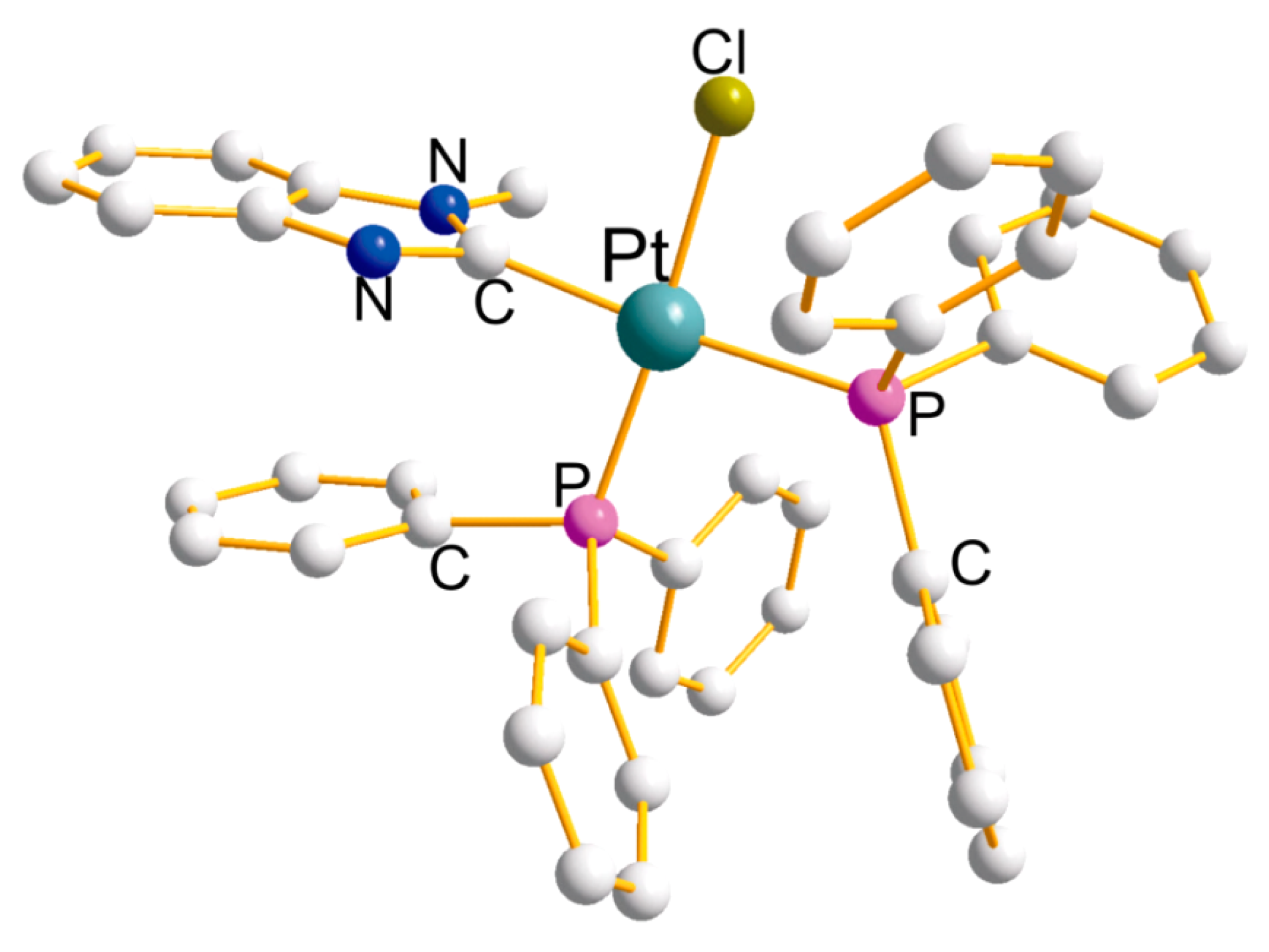
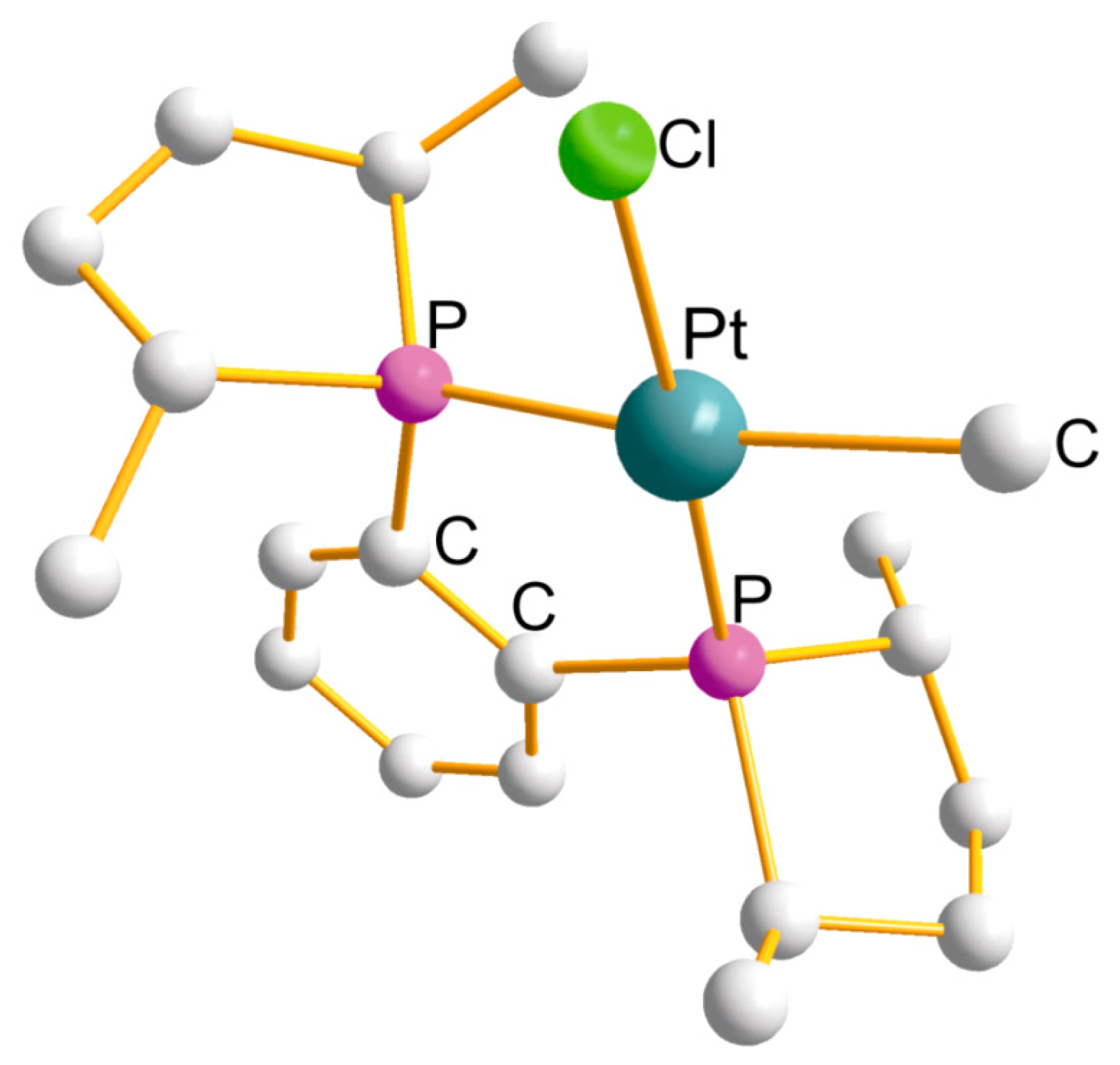
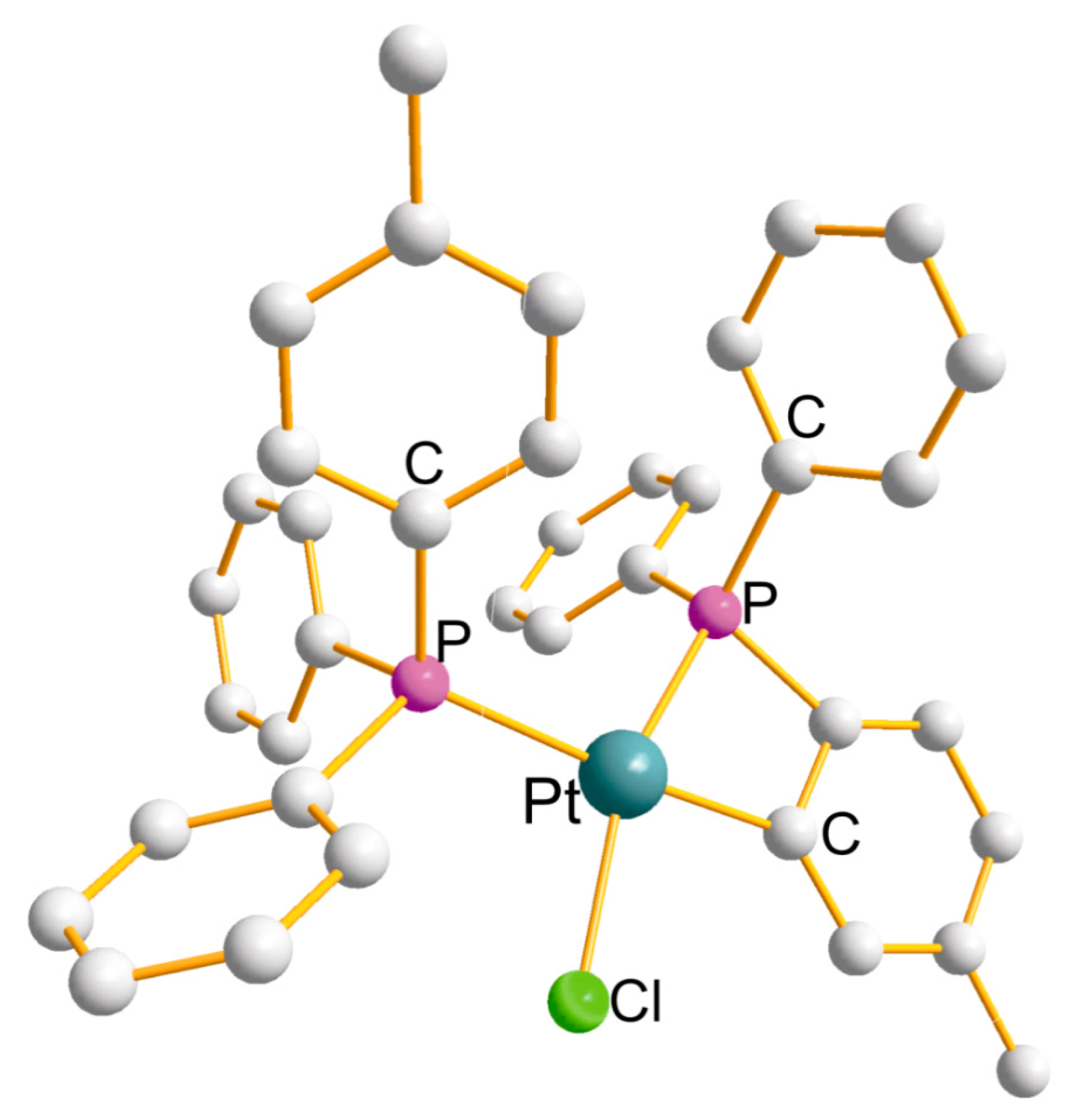
| Ring (Size) | Pt-L(x4) (Å) | P-Pt-P (°) | Cis-L-Pt-L (°) | Trans-L-Pt-L (°) |
|---|---|---|---|---|
| four | 8.87 | 72.2 | 95.8 | 178 |
| five | 8.98 | 86.1 | 91.3 | 175.8 |
| six | 9.01 | 94.0 | 88.4 | 173.5 |
| seven | 9.03 | 98.2 | 87.3 | 171.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melnik, M.; Mikus, P.; Holloway, C.E. Organophosphines in Cis-PtP2CCl Derivatives Structural Aspects. Symmetry 2018, 10, 56. https://doi.org/10.3390/sym10030056
Melnik M, Mikus P, Holloway CE. Organophosphines in Cis-PtP2CCl Derivatives Structural Aspects. Symmetry. 2018; 10(3):56. https://doi.org/10.3390/sym10030056
Chicago/Turabian StyleMelnik, Milan, Peter Mikus, and Clive Edward Holloway. 2018. "Organophosphines in Cis-PtP2CCl Derivatives Structural Aspects" Symmetry 10, no. 3: 56. https://doi.org/10.3390/sym10030056
APA StyleMelnik, M., Mikus, P., & Holloway, C. E. (2018). Organophosphines in Cis-PtP2CCl Derivatives Structural Aspects. Symmetry, 10(3), 56. https://doi.org/10.3390/sym10030056