Anti-Drug Antibodies: Emerging Approaches to Predict, Reduce or Reverse Biotherapeutic Immunogenicity
Abstract
1. Introduction
2. Most ADAs Require CD4+ T-Effector Help
3. Potential of Tolerogenic DC Presentation to Prevent ADAs
4. Identifying and Modifying HLA-Restricted T-Cell Epitopes in Biotherapeutics
5. Identifying and Modifying B-Cell Epitopes in Biotherapeutics
6. Prediction of Immunogenicity and Patient Outcomes
7. Summary
8. Patents
Funding
Conflicts of Interest
Disclaimer
References
- World Health Organization. Guidelines on the Quality, Safety and Efficacy of Biotherapeutic Protein Products Prepared by Recombinant DNA Technology; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Partridge, M.A.; Purushothama, S.; Elango, C.; Lu, Y. Emerging technologies and generic assays for the detection of anti-drug antibodies. J. Immunol. Res. 2016, 2016, 6262383. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.S.; Sauna, Z.E. Immunogenicity assessment during the development of protein therapeutics. J. Pharm. Pharmacol. 2018, 70, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Lollar, P.; Healey, J.F.; Barrow, R.T.; Parker, E.T. Factor VIII inhibitors. Adv. Exp. Med. Biol. 2001, 489, 65–73. [Google Scholar] [PubMed]
- Franchini, M.; Santoro, C.; Coppola, A. Inhibitor incidence in previously untreated patients with severe haemophilia B: A systematic literature review. Thromb. Haemost. 2016, 116, 201–203. [Google Scholar] [PubMed]
- Banugaria, S.G.; Prater, S.N.; Ng, Y.K.; Kobori, J.A.; Finkel, R.S.; Ladda, R.L.; Chen, Y.T.; Rosenberg, A.S.; Kishnani, P.S. The impact of antibodies on clinical outcomes in diseases treated with therapeutic protein: Lessons learned from infantile pompe disease. Genet. Med. Off. J. Am. Coll. Med. Genet. 2011, 13, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Holgate, R.G.; Baker, M.P. Circumventing immunogenicity in the development of therapeutic antibodies. IDrugs Investig. Drugs J. 2009, 12, 233–237. [Google Scholar]
- Liu, M.; Degner, J.; Davis, J.W.; Idler, K.B.; Nader, A.; Mostafa, N.M.; Waring, J.F. Identification of HLA-DRB1 association to adalimumab immunogenicity. PLoS ONE 2018, 13, e0195325. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Tardif, J.C.; Amarenco, P.; Duggan, W.; Glynn, R.J.; Jukema, J.W.; Kastelein, J.J.P.; Kim, A.M.; Koenig, W.; Nissen, S.; et al. Lipid-reduction variability and antidrug-antibody formation with bococizumab. N. Engl. J. Med. 2017, 376, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Bartelds, G.M.; de Groot, E.; Nurmohamed, M.T.; Hart, M.H.; van Eede, P.H.; Wijbrandts, C.A.; Crusius, J.B.; Dijkmans, B.A.; Tak, P.P.; Aarden, L.; et al. Surprising negative association between IgG1 allotype disparity and anti-adalimumab formation: A cohort study. Arthrit. Res. Ther. 2010, 12, R221. [Google Scholar] [CrossRef] [PubMed]
- Vermeire, S.; Gils, A.; Accossato, P.; Lula, S.; Marren, A. Immunogenicity of biologics in inflammatory bowel disease. Therap. Adv. Gastroenterol. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Danese, S.; Fiorino, G. Anti-TNF biosimilars in inflammatory bowel disease: Searching the proper patient’s profile? Curr. Med. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.H.; Mirakhur, B.; Chan, E.; Le, Q.T.; Berlin, J.; Morse, M.; Murphy, B.A.; Satinover, S.M.; Hosen, J.; Mauro, D.; et al. Cetuximab-induced anaphylaxis and IgE specific for galactose-alpha-1,3-galactose. N. Engl. J. Med. 2008, 358, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Mahlangu, J.N.; Weldingh, K.N.; Lentz, S.R.; Kaicker, S.; Karim, F.A.; Matsushita, T.; Recht, M.; Tomczak, W.; Windyga, J.; Ehrenforth, S.; et al. Changes in the amino acid sequence of the rFVIIa analog, vatreptacog alfa, are associated with clinical immunogenicity. J. Thromb. Haemost. 2015, 13, 1989–1998. [Google Scholar] [CrossRef] [PubMed]
- Abshire, T.; Kenet, G. Recombinant factor VIIa: Review of efficacy, dosing regimens and safety in patients with congenital and acquired factor VIII or IX inhibitors. J. Thromb. Haemost. 2004, 2, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Santagostino, E.; Escobar, M.; Ozelo, M.; Solimeno, L.; Arkhammar, P.; Lee, H.Y.; Rosu, G.; Giangrande, P. Recombinant activated factor VII in the treatment of bleeds and for the prevention of surgery-related bleeding in congenital haemophilia with inhibitors. Blood Rev. 2015, 29 (Suppl. 1), S9–S18. [Google Scholar] [CrossRef]
- Yank, V.; Tuohy, C.V.; Logan, A.C.; Bravata, D.M.; Staudenmayer, K.; Eisenhut, R.; Sundaram, V.; McMahon, D.; Olkin, I.; McDonald, K.M.; et al. Systematic review: Benefits and harms of in-hospital use of recombinant factor VIIa for off-label indications. Ann. Intern. Med. 2011, 154, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Mahlangu, J.; Paz, P.; Hardtke, M.; Aswad, F.; Schroeder, J. Trust trial: Bay 86-6150 use in haemophilia with inhibitors and assessment for immunogenicity. Haemoph. Off. J. World Fed. Hemoph. 2016, 22, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, S.S.; Muhlfelder, T. Antibodies to recombinant human erythropoietin causing pure red cell aplasia. Clin. Nephrol. 1997, 47, 331–335. [Google Scholar] [PubMed]
- Lim, L.C. Acquired red cell aplasia in association with the use of recombinant erythropoietin in chronic renal failure. Hematology 2005, 10, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Neutralizing antibodies during treatment of multiple sclerosis with interferon beta-1b: Experience during the first three years. The IFNb multiple sclerosis study group and the University of British Columbia MS/MRI analysis group. Neurology 1996, 47, 889–894. [Google Scholar]
- Deisenhammer, F. Neutralizing antibodies to interferon-beta and other immunological treatments for multiple sclerosis: Prevalence and impact on outcomes. CNS Drugs 2009, 23, 379–396. [Google Scholar] [CrossRef] [PubMed]
- Rossert, J.; Casadevall, N.; Eckardt, K.U. Anti-erythropoietin antibodies and pure red cell aplasia. J. Am. Soc. Nephrol. 2004, 15, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Macdougall, I.C.; Casadevall, N.; Locatelli, F.; Combe, C.; London, G.M.; Di Paolo, S.; Kribben, A.; Fliser, D.; Messner, H.; McNeil, J.; et al. Incidence of erythropoietin antibody-mediated pure red cell aplasia: The prospective immunogenicity surveillance registry (prims). Nephrol. Dial. Transplant. 2015, 30, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Rudensky, A.; Preston-Hurlburt, P.; Hong, S.C.; Barlow, A.; Janeway, C.A., Jr. Sequence analysis of peptides bound to MHC class II molecules. Nature 1991, 353, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Hunt, D.F.; Michel, H.; Dickinson, T.A.; Shabanowitz, J.; Cox, A.L.; Sakaguchi, K.; Appella, E.; Grey, H.M.; Sette, A. Peptides presented to the immune system by the murine class II major histocompatibility complex molecule I-Ad. Science 1992, 256, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Sunshine, G.H.; Katz, D.R.; Feldmann, M. Dendritic cells induce T cell proliferation to synthetic antigens under Ir gene control. J. Exp. Med. 1980, 152, 1817–1822. [Google Scholar] [CrossRef] [PubMed]
- Katsnelson, A. Kicking off adaptive immunity: The discovery of dendritic cells. J. Exp. Med. 2006, 203, 1622. [Google Scholar] [CrossRef] [PubMed]
- Hemmi, H.; Akira, S. TLR signalling and the function of dendritic cells. Chem. Immunol. Allergy 2005, 86, 120–135. [Google Scholar] [PubMed]
- Horton, C.; Shanmugarajah, K.; Fairchild, P.J. Harnessing the properties of dendritic cells in the pursuit of immunological tolerance. Biomed. J. 2017, 40, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Kuang, R.; Perruche, S.; Chen, W. Apoptotic cell-linked immunoregulation: Implications for promoting immune tolerance in transplantation. Cell Biosci. 2015, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Archer, G.E.; Tedder, T.F. Isolation and generation of human dendritic cells. Curr. Protocols Immunol. 2012. [Google Scholar] [CrossRef]
- Tangri, S.; Mothe, B.R.; Eisenbraun, J.; Sidney, J.; Southwood, S.; Briggs, K.; Zinckgraf, J.; Bilsel, P.; Newman, M.; Chesnut, R.; et al. Rationally engineered therapeutic proteins with reduced immunogenicity. J. Immunol. 2005, 174, 3187–3196. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.L.; Okita, D.K.; Conti-Fine, B.M. T cell recognition of the A2 domain of coagulation factor VIII in hemophilia patients and healthy subjects. J. Thromb. Haemost. 2004, 2, 1908–1917. [Google Scholar] [CrossRef] [PubMed]
- Meunier, S.; Menier, C.; Marcon, E.; Lacroix-Desmazes, S.; Maillere, B. CD4 T cells specific for factor VIII are present at high frequency in healthy donors and comprise naive and memory cells. Blood Adv. 2017, 1, 1842–1847. [Google Scholar] [CrossRef] [PubMed]
- Kamate, C.; Lenting, P.J.; van den Berg, H.M.; Mutis, T. Depletion of CD4+/CD25high regulatory T cells may enhance or uncover factor VIII-specific T-cell responses in healthy individuals. J. Thromb. Haemost. 2007, 5, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, S.K.; Grifoni, A.; Pham, J.; Vaughan, K.; Sidney, J.; Peters, B.; Sette, A. Development of a strategy and computational application to select candidate protein analogues with reduced HLA binding and immunogenicity. Immunology 2018, 153, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Raghava, G.P. Propred: Prediction of HLA-DR binding sites. Bioinformatics 2001, 17, 1236–1237. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.; Lund, O.; Buus, S.; Lundegaard, C. MHC class II epitope predictive algorithms. Immunology 2010, 130, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.; Lundegaard, C.; Blicher, T.; Peters, B.; Sette, A.; Justesen, S.; Buus, S.; Lund, O. Quantitative predictions of peptide binding to any HLA-DR molecule of known sequence: NetMHCIIpan. PLoS Comput. Biol. 2008, 4, e1000107. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Garza, E.N.; Mazor, R.; Linehan, J.L.; Pastan, I.; Pepper, M.; Baker, D. Removing T-cell epitopes with computational protein design. Proc. Natl. Acad. Sci. USA 2014, 111, 8577–8582. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Trincado, J.L.; Gomez-Perosanz, M.; Reche, P.A. Fundamentals and methods for T- and B-cell epitope prediction. J. Immunol. Res. 2017, 2017, 2680160. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.S. Do tregitopes have the potential to impact the current treatment landscape of autoimmune diseases? Exp. Rev. Clin. Immunol. 2013, 9, 1155–1157. [Google Scholar] [CrossRef] [PubMed]
- Cohen, T.; Moise, L.; Ardito, M.; Martin, W.; De Groot, A.S. A method for individualizing the prediction of immunogenicity of protein vaccines and biologic therapeutics: Individualized T cell epitope measure (item). J. Biomed. Biotechnol. 2010, 2010, 961752. [Google Scholar] [CrossRef] [PubMed]
- Griswold, K.E.; Bailey-Kellogg, C. Design and engineering of deimmunized biotherapeutics. Curr. Opin. Struct. Biol. 2016, 39, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Schubert, B.; Scharfe, C.; Donnes, P.; Hopf, T.; Marks, D.; Kohlbacher, O. Population-specific design of de-immunized protein biotherapeutics. PLoS Comput. Biol. 2018, 14, e1005983. [Google Scholar] [CrossRef] [PubMed]
- Jawa, V.; Joubert, M.K.; Zhang, Q.; Deshpande, M.; Hapuarachchi, S.; Hall, M.P.; Flynn, G.C. Evaluating immunogenicity risk due to host cell protein impurities in antibody-based biotherapeutics. AAPS J. 2016, 18, 1439–1452. [Google Scholar] [CrossRef] [PubMed]
- Joubert, M.K.; Deshpande, M.; Yang, J.; Reynolds, H.; Bryson, C.; Fogg, M.; Baker, M.P.; Herskovitz, J.; Goletz, T.J.; Zhou, L.; et al. Use of in vitro assays to assess immunogenicity risk of antibody-based biotherapeutics. PLoS ONE 2016, 11, e0159328. [Google Scholar] [CrossRef] [PubMed]
- Castaman, G.; Eckhardt, C.; van Velzen, A.; Linari, S.; Fijnvandraat, K. Emerging issues in diagnosis, biology, and inhibitor risk in mild hemophilia A. Semin. Thromb. Hemost. 2016, 42, 507–512. [Google Scholar] [PubMed]
- Jacquemin, M.; Vantomme, V.; Buhot, C.; Lavend’homme, R.; Burny, W.; Demotte, N.; Chaux, P.; Peerlinck, K.; Vermylen, J.; Maillere, B.; et al. CD4+ T-cell clones specific for wild-type factor VIII: A molecular mechanism responsible for a higher incidence of inhibitor formation in mild/moderate hemophilia a. Blood 2003, 101, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- James, E.A.; Kwok, W.W.; Ettinger, R.A.; Thompson, A.R.; Pratt, K.P. T-cell responses over time in a mild hemophilia A inhibitor subject: Epitope identification and transient immunogenicity of the corresponding self-peptide. J. Thromb. Haemost. 2007, 5, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, R.A.; James, E.A.; Kwok, W.W.; Thompson, A.R.; Pratt, K.P. Lineages of human T-cell clones, including T helper 17/T helper 1 cells, isolated at different stages of anti-factor VIII immune responses. Blood 2009, 114, 1423–1428. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, R.A.; James, E.A.; Kwok, W.W.; Thompson, A.R.; Pratt, K.P. HLA-DR-restricted T-cell responses to factor VIII epitopes in a mild haemophilia A family with missense substitution A2201P. Haemoph. Off. J. World Fed. Hemoph. 2010, 16, 44–55. [Google Scholar] [CrossRef] [PubMed]
- James, E.A.; van Haren, S.D.; Ettinger, R.A.; Fijnvandraat, K.; Liberman, J.A.; Kwok, W.W.; Voorberg, J.; Pratt, K.P. T-cell responses in two unrelated hemophilia A inhibitor subjects include an epitope at the factor VIII R593C missense site. J. Thromb. Haemost. 2011, 9, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Pratt, K.P.; Qian, J.; Ellaban, E.; Okita, D.K.; Diethelm-Okita, B.M.; Conti-Fine, B.; Scott, D.W. Immunodominant T-cell epitopes in the factor VIII C2 domain are located within an inhibitory antibody binding site. Thromb. Haemost. 2004, 92, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Steinitz, K.N.; van Helden, P.M.; Binder, B.; Wraith, D.C.; Unterthurner, S.; Hermann, C.; Schuster, M.; Ahmad, R.U.; Weiller, M.; Lubich, C.; et al. CD4+ T-cell epitopes associated with antibody responses after intravenously and subcutaneously applied human FVIII in humanized hemophilic E17 HLA-DRB1*1501 mice. Blood 2012, 119, 4073–4082. [Google Scholar] [CrossRef] [PubMed]
- Moise, L.; Song, C.; Martin, W.D.; Tassone, R.; De Groot, A.S.; Scott, D.W. Effect of HLA DR epitope de-immunization of factor VIII in vitro and in vivo. Clin. Immunol. 2012, 142, 320–331. [Google Scholar] [CrossRef] [PubMed]
- van Haren, S.D.; Herczenik, E.; ten Brinke, A.; Mertens, K.; Voorberg, J.; Meijer, A.B. HLA-DR-presented peptide repertoires derived from human monocyte-derived dendritic cells pulsed with blood coagulation factor VIII. Mol. Cell. Proteom. 2011, 10, M110–002246. [Google Scholar] [CrossRef] [PubMed]
- Van Haren, S.D.; Wroblewska, A.; Herczenik, E.; Kaijen, P.H.; Ruminska, A.; ten Brinke, A.; Meijer, A.B.; Voorberg, J. Limited promiscuity of HLA-DRB1 presented peptides derived of blood coagulation factor VIII. PLoS ONE 2013, 8, e80239. [Google Scholar] [CrossRef] [PubMed]
- Peyron, I.; Hartholt, R.B.; Pedro-Cos, L.; van Alphen, F.; Brinke, A.T.; Lardy, N.; Meijer, A.B.; Voorberg, J. Comparative profiling of HLA-DR and HLA-DQ associated factor VIII peptides presented by monocyte-derived dendritic cells. Haematologica 2018, 103, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Hrdinova, J.; Verbij, F.C.; Kaijen, P.H.P.; Hartholt, R.B.; van Alphen, F.; Lardy, N.; Ten Brinke, A.; Vanhoorelbeke, K.; Hindocha, P.J.; De Groot, A.S.; et al. Mass spectrometry-assisted identification of ADAMTS13-derived peptides presented on HLA-DR and HLA-DQ. Haematologica 2018. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, R.A.; Paz, P.; James, E.A.; Gunasekera, D.; Aswad, F.; Thompson, A.R.; Matthews, D.C.; Pratt, K.P. T cells from hemophilia A subjects recognize the same HLA-restricted FVIII epitope with a narrow TCR repertoire. Blood 2016, 128, 2043–2054. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, R.A.; Liberman, J.A.; Gunasekera, D.; Puranik, K.; James, E.A.; Thompson, A.R.; Pratt, K.P. FVIII proteins with a modified immunodominant T-cell epitope exhibit reduced immunogenicity and normal FVIII activity. Blood Adv. 2018, 2, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Pautard, B.; D’Oiron, R.; Li Thiao Te, V.; Lavend’homme, R.; Saint-Remy, J.M.; Peerlinck, K.; Jacquemin, M. Successful immune tolerance induction by FVIII in hemophilia A patients with inhibitor may occur without deletion of FVIII-specific T cells. J. Thromb. Haemost. 2011, 9, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Vassall, A.N.; Eberle, J.A.; Beers, R.; Weldon, J.E.; Venzon, D.J.; Tsang, K.Y.; Benhar, I.; Pastan, I. Identification and elimination of an immunodominant T-cell epitope in recombinant immunotoxins based on pseudomonas exotoxin A. Proc. Natl. Acad. Sci. USA 2012, 109, E3597–E3603. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Eberle, J.A.; Hu, X.; Vassall, A.N.; Onda, M.; Beers, R.; Lee, E.C.; Kreitman, R.J.; Lee, B.; Baker, D.; et al. Recombinant immunotoxin for cancer treatment with low immunogenicity by identification and silencing of human T-cell epitopes. Proc. Natl. Acad. Sci. USA 2014, 111, 8571–8576. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Zhang, J.; Xiang, L.; Addissie, S.; Awuah, P.; Beers, R.; Hassan, R.; Pastan, I. Recombinant immunotoxin with T-cell epitope mutations that greatly reduce immunogenicity for treatment of mesothelin-expressing tumors. Mol. Cancer Ther. 2015, 14, 2789–2796. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Onda, M.; Park, D.; Addissie, S.; Xiang, L.; Zhang, J.; Hassan, R.; Pastan, I. Dual B- and T-cell de-immunization of recombinant immunotoxin targeting mesothelin with high cytotoxic activity. Oncotarget 2016, 7, 29916–29926. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Onda, M.; Pastan, I. Immunogenicity of therapeutic recombinant immunotoxins. Immunol. Rev. 2016, 270, 152–164. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Kaplan, G.; Park, D.; Jang, Y.; Lee, F.; Kreitman, R.; Pastan, I. Rational design of low immunogenic anti CD25 recombinant immunotoxin for T cell malignancies by elimination of T cell epitopes in PE38. Cell. Immunol. 2017, 313, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Michalska, M.; Schultze-Seemann, S.; Kuckuck, I.; Wolf, P. In vitro evaluation of humanized/de-immunized anti-PSMA immunotoxins for the treatment of prostate cancer. Anticancer Res. 2018, 38, 61–69. [Google Scholar] [PubMed]
- Mufarrege, E.F.; Giorgetti, S.; Etcheverrigaray, M.; Terry, F.; Martin, W.; De Groot, A.S. De-immunized and functional therapeutic (deft) versions of a long lasting recombinant alpha interferon for antiviral therapy. Clin. Immunol. 2017, 176, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Yeung, V.P.; Chang, J.; Miller, J.; Barnett, C.; Stickler, M.; Harding, F.A. Elimination of an immunodominant CD4+ T cell epitope in human IFN-beta does not result in an in vivo response directed at the subdominant epitope. J. Immunol. 2004, 172, 6658–6665. [Google Scholar] [CrossRef] [PubMed]
- Warmerdam, P.A.; Plaisance, S.; Vanderlick, K.; Vandervoort, P.; Brepoels, K.; Collen, D.; De Maeyer, M. Elimination of a human T-cell region in staphylokinase by T-cell screening and computer modeling. Thromb. Haemost. 2002, 87, 666–673. [Google Scholar] [PubMed]
- Harding, F.A.; Liu, A.D.; Stickler, M.; Razo, O.J.; Chin, R.; Faravashi, N.; Viola, W.; Graycar, T.; Yeung, V.P.; Aehle, W.; et al. A beta-lactamase with reduced immunogenicity for the targeted delivery of chemotherapeutics using antibody-directed enzyme prodrug therapy. Mol. Cancer ther. 2005, 4, 1791–1800. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Mathieu, M.; Lee, S.; Wang, X.; Kee, Y.S.; Bevers, J.J., 3rd; Ciferri, C.; Estavez, A.; Wong, M.; Chiang, N.Y.; et al. Molecular characterization of human anti-hinge antibodies derived from single-cell cloning of normal human B cells. J. Biol. Chem. 2018, 293, 906–919. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.W. From IgG fusion proteins to engineered-specific human regulatory T cells: A life of tolerance. Front. Immunol. 2017, 8, 1576. [Google Scholar] [CrossRef] [PubMed]
- Wraith, D.C. The future of immunotherapy: A 20-year perspective. Front. Immunol. 2017, 8, 1668. [Google Scholar] [CrossRef] [PubMed]
- Sicard, A.; Levings, M.K.; Scott, D.W. Engineering therapeutic T cells to suppress alloimmune responses using TCRs, CARs or BARs. Am. J. Transplant. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Singh, S.K.; Wang, X.; Rup, B.; Gill, D. Coupling of aggregation and immunogenicity in biotherapeutics: T- and B-cell immune epitopes may contain aggregation-prone regions. Pharm. Res. 2011, 28, 949–961. [Google Scholar] [CrossRef] [PubMed]
- Attanavanich, K.; Kearney, J.F. Marginal zone, but not follicular B cells, are potent activators of naive CD4 T cells. J. Immunol. 2004, 172, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Van Schooten, C.J.; Shahbazi, S.; Groot, E.; Oortwijn, B.D.; van den Berg, H.M.; Denis, C.V.; Lenting, P.J. Macrophages contribute to the cellular uptake of von Willebrand factor and factor VIII in vivo. Blood 2008, 112, 1704–1712. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, A.; Dasgupta, S.; Delignat, S.; Caligiuri, G.; Christophe, O.D.; Bayry, J.; Nicoletti, A.; Kaveri, S.V.; Lacroix-Desmazes, S. Splenic marginal zone antigen-presenting cells are critical for the primary allo-immune response to therapeutic factor VIII in hemophilia A. J. Thromb. Haemost. 2009, 7, 1816–1823. [Google Scholar] [CrossRef] [PubMed]
- Zerra, P.E.; Cox, C.; Baldwin, W.H.; Patel, S.R.; Arthur, C.M.; Lollar, P.; Meeks, S.L.; Stowell, S.R. Marginal zone B cells are critical to factor VIII inhibitor formation in mice with hemophilia A. Blood 2017, 130, 2559–2568. [Google Scholar] [CrossRef] [PubMed]
- Suan, D.; Sundling, C.; Brink, R. Plasma cell and memory B cell differentiation from the germinal center. Curr. Opin. Immunol. 2017, 45, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Sevy, A.M.; Healey, J.F.; Deng, W.; Spiegel, P.C.; Meeks, S.L.; Li, R. Epitope mapping of inhibitory antibodies targeting the C2 domain of coagulation factor VIII by hydrogen-deuterium exchange mass spectrometry. J. Thromb. Haemost. 2013, 11, 2128–2136. [Google Scholar] [CrossRef] [PubMed]
- Brier, S.; Le Mignon, M.; Jain, K.; Lebrun, C.; Peurois, F.; Kellenberger, C.; Bordas-Le Floch, V.; Mascarell, L.; Nony, E.; Moingeon, P. Characterization of epitope specificities of reference antibodies used for the quantification of the birch pollen allergen bet v 1. Allergy 2017, 73, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Meeks, S.L.; Healey, J.F.; Parker, E.T.; Barrow, R.T.; Lollar, P. Antihuman factor VIII C2 domain antibodies in hemophilia a mice recognize a functionally complex continuous spectrum of epitopes dominated by inhibitors of factor VIII activation. Blood 2007, 110, 4234–4242. [Google Scholar] [CrossRef] [PubMed]
- Meeks, S.L.; Healey, J.F.; Parker, E.T.; Barrow, R.T.; Lollar, P. Nonclassical anti-C2 domain antibodies are present in patients with factor VIII inhibitors. Blood 2008, 112, 1151–1153. [Google Scholar] [CrossRef] [PubMed]
- Markovitz, R.C.; Healey, J.F.; Parker, E.T.; Meeks, S.L.; Lollar, P. The diversity of the immune response to the A2 domain of human factor VIII. Blood 2013, 121, 2785–2795. [Google Scholar] [CrossRef] [PubMed]
- Kahle, J.; Orlowski, A.; Stichel, D.; Healey, J.F.; Parker, E.T.; Jacquemin, M.; Krause, M.; Tiede, A.; Schwabe, D.; Lollar, P.; et al. Frequency and epitope specificity of anti-factor VIII C1 domain antibodies in acquired and congenital hemophilia A. Blood 2017, 130, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Lollar, P. Pathogenic antibodies to coagulation factors. Part one: Factor VIII and factor IX. J. Thromb. Haemost. 2004, 2, 1082–1095. [Google Scholar] [CrossRef] [PubMed]
- Barrow, R.T.; Lollar, P. Neutralization of antifactor VIII inhibitors by recombinant porcine factor VIII. J. Thromb. Haemost. 2006, 4, 2223–2229. [Google Scholar] [CrossRef] [PubMed]
- Lollar, P.; Parker, E.T.; Fay, P.J. Coagulant properties of hybrid human/porcine factor VIII molecules. J. Biol. Chem. 1992, 267, 23652–23657. [Google Scholar] [PubMed]
- Lollar, P. Analysis of factor VIII inhibitors using hybrid human/porcine factor VIII. Thromb. Haemost. 1997, 78, 647–651. [Google Scholar] [PubMed]
- Lin, J.C.; Ettinger, R.A.; Schuman, J.T.; Zhang, A.H.; Wamiq-Adhami, M.; Nguyen, P.C.; Nakaya-Fletcher, S.M.; Puranik, K.; Thompson, A.R.; Pratt, K.P. Six amino acid residues in a 1200 Å2 interface mediate binding of factor VIII to an IgG4-kappa inhibitory antibody. PLoS ONE 2015, 10, e0116577. [Google Scholar]
- Nguyen, P.C.; Lewis, K.B.; Ettinger, R.A.; Schuman, J.T.; Lin, J.C.; Healey, J.F.; Meeks, S.L.; Lollar, P.; Pratt, K.P. High-resolution mapping of epitopes on the C2 domain of factor VIII by analysis of point mutants using surface plasmon resonance. Blood 2014, 123, 2732–2739. [Google Scholar] [CrossRef] [PubMed]
- Healey, J.F.; Barrow, R.T.; Tamim, H.M.; Lubin, I.M.; Shima, M.; Scandella, D.; Lollar, P. Residues Glu2181-Val2243 contain a major determinant of the inhibitory epitope in the C2 domain of human factor VIII. Blood 1998, 92, 3701–3709. [Google Scholar] [PubMed]
- Scandella, D.; Gilbert, G.E.; Shima, M.; Nakai, H.; Eagleson, C.; Felch, M.; Prescott, R.; Rajalakshmi, K.J.; Hoyer, L.W.; Saenko, E. Some factor VIII inhibitor antibodies recognize a common epitope corresponding to C2 domain amino acids 2248 through 2312, which overlap a phospholipid-binding site. Blood 1995, 86, 1811–1819. [Google Scholar] [PubMed]
- Pratt, K.P.; Shen, B.W.; Takeshima, K.; Davie, E.W.; Fujikawa, K.; Stoddard, B.L. Structure of the C2 domain of human factor VIII at 1.5 Å resolution. Nature 1999, 402, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Clackson, T.; Wells, J.A. A hot spot of binding energy in a hormone-receptor interface. Science 1995, 267, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, P.C., Jr.; Jacquemin, M.; Saint-Remy, J.M.; Stoddard, B.L.; Pratt, K.P. Structure of a factor VIII C2 domain-immunoglobulin IgG4kappa Fab complex: Identification of an inhibitory antibody epitope on the surface of factor VIII. Blood 2001, 98, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.D.; Werther, R.A.; Brison, C.M.; Cragerud, R.K.; Healey, J.F.; Meeks, S.L.; Lollar, P.; Spiegel, P.C., Jr. Structure of the factor VIII C2 domain in a ternary complex with 2 inhibitor antibodies reveals classical and nonclassical epitopes. Blood 2013, 122, 4270–4278. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.D.; Werther, R.A.; Polozova, M.S.; Pohlman, J.; Healey, J.F.; Meeks, S.L.; Lollar, P.; Spiegel, P.C., Jr. Characterization and solution structure of the factor VIII C2 domain in a ternary complex with classical and non-classical inhibitor antibodies. J. Biol. Chem. 2013, 288, 9905–9914. [Google Scholar] [CrossRef] [PubMed]
- Onda, M.; Beers, R.; Xiang, L.; Lee, B.; Weldon, J.E.; Kreitman, R.J.; Pastan, I. Recombinant immunotoxin against B-cell malignancies with no immunogenicity in mice by removal of B-cell epitopes. Proc. Natl. Acad. Sci. USA 2011, 108, 5742–5747. [Google Scholar] [CrossRef] [PubMed]
- Weldon, J.E.; Xiang, L.; Zhang, J.; Beers, R.; Walker, D.A.; Onda, M.; Hassan, R.; Pastan, I. A recombinant immunotoxin against the tumor-associated antigen mesothelin reengineered for high activity, low off-target toxicity, and reduced antigenicity. Mol. Cancer Ther. 2013, 12, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Nagata, S.; Pastan, I. Removal of B cell epitopes as a practical approach for reducing the immunogenicity of foreign protein-based therapeutics. Adv. Drug Deliv. Rev. 2009, 61, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.J.; Chae, C.S.; So, J.S.; Tzartos, S.J.; Souroujon, M.C.; Fuchs, S.; Im, S.H. Suppression of experimental myasthenia gravis by a B-cell epitope-free recombinant acetylcholine receptor. Mol. Immunol. 2008, 46, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Brettschneider, K.; Kahle, J.; Orlowski, A.; Becker-Peters, K.; Stichel, D.; Schulze, J.; Braner, M.; Tampe, R.; Schwabe, D.; et al. Neutralisation of factor VIII inhibitors by anti-idiotypes isolated from phage-displayed libraries. Thromb. Haemost. 2016, 116, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, C.J.; Whelan, S.F.; Hirschler, M.; Allacher, P.; Horling, F.M.; Lawo, J.P.; Oldenburg, J.; Tiede, A.; Male, C.; Windyga, J.; et al. Affinity of FVIII-specific antibodies reveals major differences between neutralizing and nonneutralizing antibodies in humans. Blood 2015, 125, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Reipert, B.M. B-cell memory against factor VIII. Cell. Immunol. 2016, 301, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.H.; Rice, A.S.; Boylan, B.; Payne, A.B.; Kelly, F.M.; Escobar, M.A.; Gill, J.; Leissinger, C.; Soucie, J.M.; Hemophilia Inhibitor Research Study, I. Characteristics of hemophilia patients with factor VIII inhibitors detected by prospective screening. Am. J. Hematol. 2015, 90, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Van Helden, P.M.; van den Berg, H.M.; Gouw, S.C.; Kaijen, P.H.; Zuurveld, M.G.; Mauser-Bunschoten, E.P.; Aalberse, R.C.; Vidarsson, G.; Voorberg, J. IgG subclasses of anti-FVIII antibodies during immune tolerance induction in patients with hemophilia A. Br. J. Haematol. 2008, 142, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Drescher, D.G.; Selvakumar, D.; Drescher, M.J. Analysis of protein interactions by surface plasmon resonance. Adv. Protein Chem. Struct. Biol. 2018, 110, 1–30. [Google Scholar] [PubMed]
- Lewis, K.B.; Hughes, R.J.; Epstein, M.S.; Josephson, N.C.; Kempton, C.L.; Kessler, C.M.; Key, N.S.; Howard, T.E.; Kruse-Jarres, R.; Lusher, J.M.; et al. Phenotypes of allo- and autoimmune antibody responses to FVIII characterized by surface plasmon resonance. PLoS ONE 2013, 8, e61120. [Google Scholar] [CrossRef] [PubMed]
- Ngo, J.C.; Huang, M.; Roth, D.A.; Furie, B.C.; Furie, B. Crystal structure of human factor VIII: Implications for the formation of the factor IXa-factor VIIIA complex. Structure 2008, 16, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Grotzinger, C. Peptide microarrays for medical applications in autoimmunity, infection, and cancer. Methods Mol. Biol. 2016, 1352, 213–221. [Google Scholar] [PubMed]
- Stephenson, K.E.; Neubauer, G.H.; Reimer, U.; Pawlowski, N.; Knaute, T.; Zerweck, J.; Korber, B.T.; Barouch, D.H. Quantification of the epitope diversity of HIV-1-specific binding antibodies by peptide microarrays for global HIV-1 vaccine development. J. Immunol. Methods 2015, 416, 105–123. [Google Scholar] [CrossRef] [PubMed]
- Ambati, A.; Valentini, D.; Montomoli, E.; Lapini, G.; Biuso, F.; Wenschuh, H.; Magalhaes, I.; Maeurer, M. H1N1 viral proteome peptide microarray predicts individuals at risk for H1N1 infection and segregates infection versus pandemrix((r)) vaccination. Immunology 2015, 145, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Homann, A.; Rockendorf, N.; Kromminga, A.; Frey, A.; Platts-Mills, T.A.; Jappe, U. Glycan and peptide IgE epitopes of the TNF-alpha blockers infliximab and adalimumab—Precision diagnostics by cross-reactivity immune profiling of patient sera. Theranostics 2017, 7, 4699–4709. [Google Scholar] [CrossRef] [PubMed]
- Havenith, H.; Kern, K.; Rautenberger, P.; Spiegel, H.; Szardenings, M.; Ueberham, E.; Lehmann, J.; Buntru, M.; Vogel, S.; Treudler, R.; et al. Combination of two epitope identification techniques enables the rational design of soy allergen Gly m 4 mutants. Biotechnol. J. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Luo, H.; Yan, M.; Zuo, X.; Li, Q.Z. Autoantigen microarray for high-throughput autoantibody profiling in systemic lupus erythematosus. Genom. Proteom. Bioinform. 2015, 13, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Atak, A.; Mukherjee, S.; Jain, R.; Gupta, S.; Singh, V.A.; Gahoi, N.; K, P.M.; Srivastava, S. Protein microarray applications: Autoantibody detection and posttranslational modification. Proteomics 2016, 16, 2557–2569. [Google Scholar] [CrossRef] [PubMed]
- Sauer, U. Analytical protein microarrays: Advancements towards clinical applications. Sensors 2017, 17, 256. [Google Scholar] [CrossRef] [PubMed]
- Ewaisha, R.; Anderson, K.S. Proteomic monitoring of B cell immunity. Methods Mol. Biol. 2016, 1403, 131–152. [Google Scholar] [PubMed]
- Moreira, G.; Fuhner, V.; Hust, M. Epitope mapping by phage display. Methods Mol. Biol. 2018, 1701, 497–518. [Google Scholar] [PubMed]
- Christiansen, A.; Kringelum, J.V.; Hansen, C.S.; Bogh, K.L.; Sullivan, E.; Patel, J.; Rigby, N.M.; Eiwegger, T.; Szepfalusi, Z.; de Masi, F.; et al. High-throughput sequencing enhanced phage display enables the identification of patient-specific epitope motifs in serum. Sci. Rep. 2015, 5, 12913. [Google Scholar] [CrossRef] [PubMed]
- Bazan, J.; Calkosinski, I.; Gamian, A. Phage display—A powerful technique for immunotherapy: 2. Vaccine delivery. Hum. Vaccines Immunother. 2012, 8, 1829–1835. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wei, Y.; Yu, W.; Wang, L.; Zhai, L.; Li, X.; Wang, X.; Zhang, H.; Feng, Z.; Yu, L.; et al. Identification of a conserved linear B-cell epitope in the staphylococcus aureus gapc protein. Microb. Pathog. 2018, 118, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Frietze, K.M.; Pascale, J.M.; Moreno, B.; Chackerian, B.; Peabody, D.S. Pathogen-specific deep sequence-coupled biopanning: A method for surveying human antibody responses. PLoS ONE 2017, 12, e0171511. [Google Scholar] [CrossRef] [PubMed]
- Bazan, J.; Calkosinski, I.; Gamian, A. Phage display—A powerful technique for immunotherapy: 1. Introduction and potential of therapeutic applications. Hum. Vaccines Immunother. 2012, 8, 1817–1828. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Dreskin, S.C. Application of phage peptide display technology for the study of food allergen epitopes. Mol. Nutr. Food Res. 2017, 61, 1600568. [Google Scholar] [CrossRef] [PubMed]
- Casina, V.C.; Hu, W.; Mao, J.H.; Lu, R.N.; Hanby, H.A.; Pickens, B.; Kan, Z.Y.; Lim, W.K.; Mayne, L.; Ostertag, E.M.; et al. High-resolution epitope mapping by HX MS reveals the pathogenic mechanism and a possible therapy for autoimmune TTP syndrome. Proc. Natl. Acad. Sci. USA 2015, 112, 9620–9625. [Google Scholar] [CrossRef] [PubMed]
- Shave, S.; Mann, S.; Koszela, J.; Kerr, A.; Auer, M. Pulse: Quality control and quantification of peptide sequences explored by phage display libraries. PLoS ONE 2018, 13, e0193332. [Google Scholar] [CrossRef] [PubMed]
- Vaisman-Mentesh, A.; Wine, Y. Monitoring phage biopanning by next-generation sequencing. Methods Mol. Biol. 2018, 1701, 463–473. [Google Scholar] [PubMed]
- Turner, K.B.; Naciri, J.; Liu, J.L.; Anderson, G.P.; Goldman, E.R.; Zabetakis, D. Next-generation sequencing of a single domain antibody repertoire reveals quality of phage display selected candidates. PLoS ONE 2016, 11, e0149393. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Nezafat, N.; Rahbar, M.R.; Negahdaripour, M.; Sabetian, S.; Morowvat, M.H.; Ghasemi, Y. Decreasing the immunogenicity of arginine deiminase enzyme via structure-based computational analysis. J. Biomol. Struct. Dyn. 2018, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Jardim, L.L.; Chaves, D.G.; Silveira-Cassette, A.C.O.; Simoes, E.S.A.C.; Santana, M.P.; Cerqueira, M.H.; Prezotti, A.; Lorenzato, C.; Franco, V.; van der Bom, J.G.; et al. Immune status of patients with haemophilia A before exposure to factor VIII: First results from the Hemfil study. Br. J. Haematol. 2017, 178, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Guo, D.; Key, N.S.; Conti-Fine, B.M. Cytokine production by CD4+ T cells specific for coagulation factor VIII in healthy subjects and haemophilia A patients. Thromb. Haemost. 2007, 97, 788–794. [Google Scholar] [PubMed]
- Chaves, D.; Belisario, A.; Castro, G.; Santoro, M.; Rodrigues, C. Analysis of cytokine genes polymorphism as markers for inhibitor development in haemophilia A. Int. J. Immunogenet. 2010, 37, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Chaves, D.G.; Velloso-Rodrigues, C.; Oliveira, C.A.; Teixeira-Carvalho, A.; Santoro, M.M.; Martins-Filho, O.A. A shift towards a T cell cytokine deficiency along with an anti-inflammatory/regulatory microenvironment may enable the synthesis of anti-FVIII inhibitors in haemophilia A patients. Clin. Exp. Immunol. 2010, 162, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.A.; Velloso-Rodrigues, C.; Machado, F.C.; Carvalho, B.N.; Gentz, S.H.; Martins-Filho, O.A.; Chaves, D.G. Cytokine profile and FVIII inhibitors development in haemophilia A. Haemophilia 2013, 19, e139–e142. [Google Scholar] [CrossRef] [PubMed]
- Silveira, A.C.; Santana, M.A.; Ribeiro, I.G.; Chaves, D.G.; Martins-Filho, O.A. The IL-10 polarized cytokine pattern in innate and adaptive immunity cells contribute to the development of FVIII inhibitors. BMC Hematol. 2015, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Boehncke, W.H.; Brembilla, N.C. Immunogenicity of biologic therapies: Causes and consequences. Exp. Rev. Clin. Immunol. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nepom, G.T.; St Clair, E.W.; Turka, L.A. Challenges in the pursuit of immune tolerance. Immunol. Rev. 2011, 241, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.H.; Rossi, R.J.; Yoon, J.; Wang, H.; Scott, D.W. Tolerogenic nanoparticles to induce immunologic tolerance: Prevention and reversal of FVIII inhibitor formation. Cell. Immunol. 2016, 301, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Batsuli, G.; Meeks, S.L.; Herzog, R.W.; Lacroix-Desmazes, S. Innovating immune tolerance induction for haemophilia. Haemoph. Off. J. World Fed. Hemoph. 2016, 22 (Suppl. 5), 31–35. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, R.S.; Lillicrap, D. Tolerogenic properties of Fc portion of IgG and its relevance to the treatment and management of hemophilia. Blood 2018. [Google Scholar] [CrossRef] [PubMed]
- Dawson, N.A.J.; Vent-Schmidt, J.; Levings, M.K. Engineered tolerance: Tailoring development, function, and antigen-specificity of regulatory T cells. Front. Immunol. 2017, 8, 1460. [Google Scholar] [CrossRef] [PubMed]
- Esensten, J.H.; Bluestone, J.A.; Lim, W.A. Engineering therapeutic T cells: From synthetic biology to clinical trials. Annu. Rev. Pathol. 2017, 12, 305–330. [Google Scholar] [CrossRef] [PubMed]
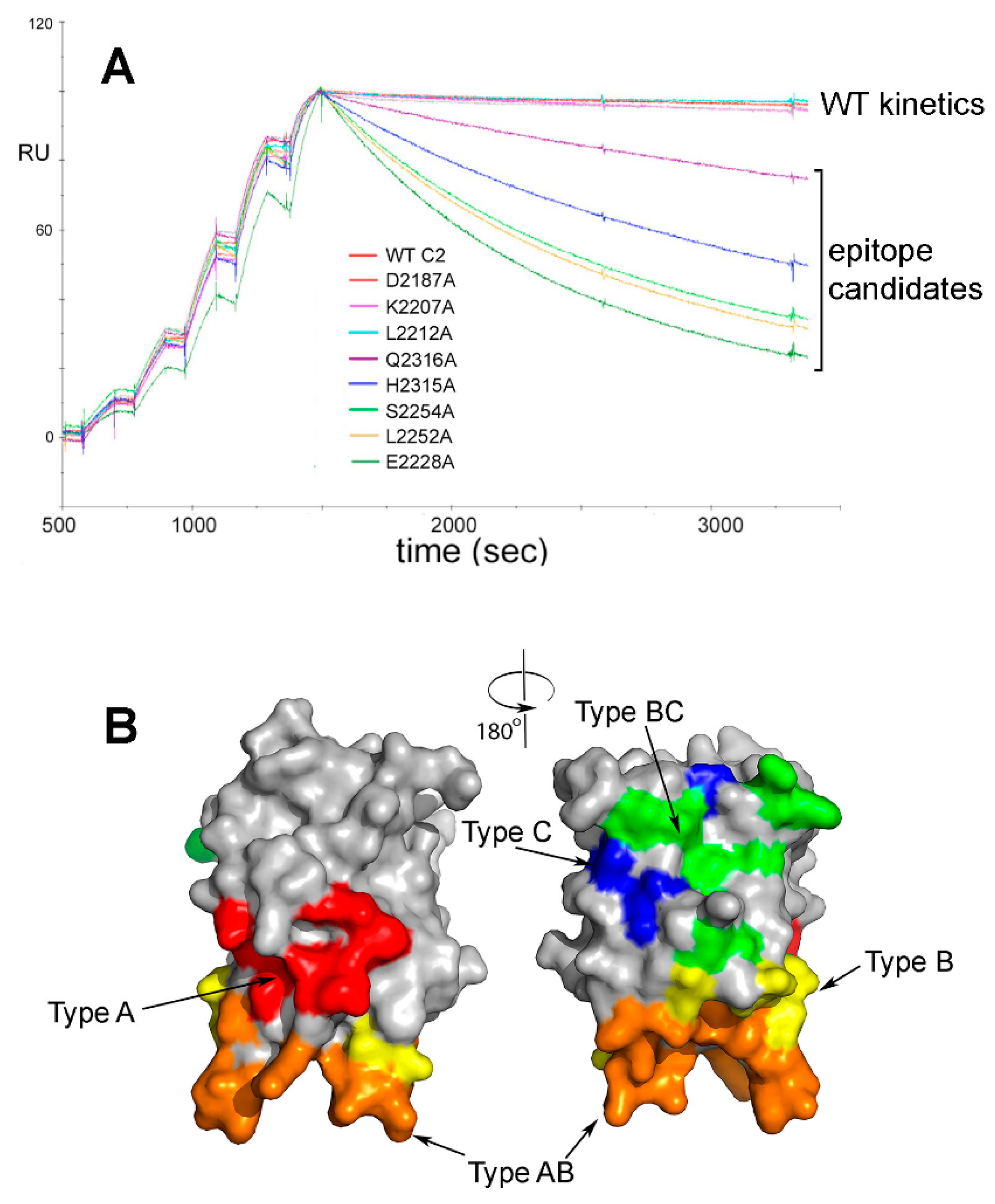
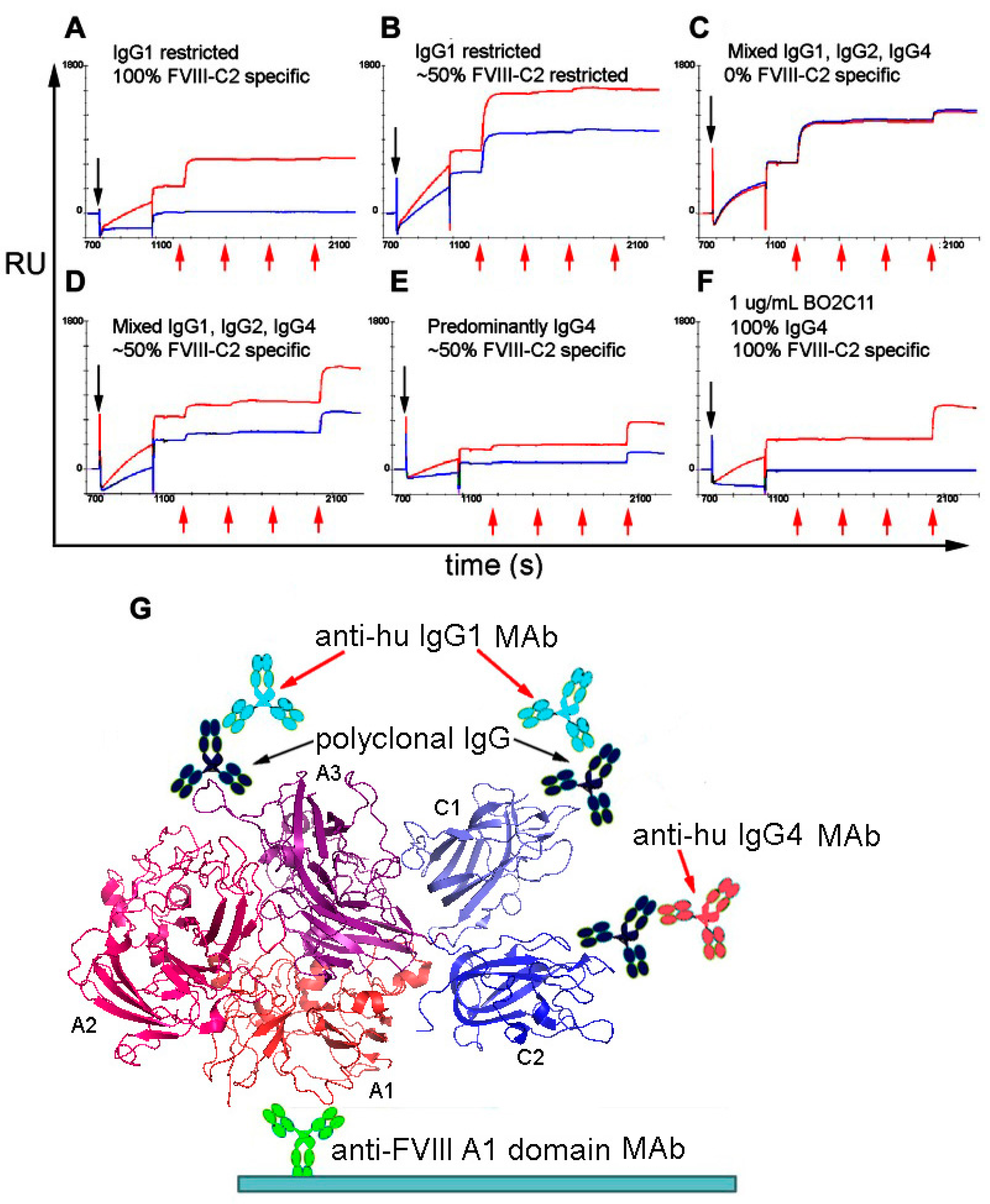
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pratt, K.P. Anti-Drug Antibodies: Emerging Approaches to Predict, Reduce or Reverse Biotherapeutic Immunogenicity. Antibodies 2018, 7, 19. https://doi.org/10.3390/antib7020019
Pratt KP. Anti-Drug Antibodies: Emerging Approaches to Predict, Reduce or Reverse Biotherapeutic Immunogenicity. Antibodies. 2018; 7(2):19. https://doi.org/10.3390/antib7020019
Chicago/Turabian StylePratt, Kathleen P. 2018. "Anti-Drug Antibodies: Emerging Approaches to Predict, Reduce or Reverse Biotherapeutic Immunogenicity" Antibodies 7, no. 2: 19. https://doi.org/10.3390/antib7020019
APA StylePratt, K. P. (2018). Anti-Drug Antibodies: Emerging Approaches to Predict, Reduce or Reverse Biotherapeutic Immunogenicity. Antibodies, 7(2), 19. https://doi.org/10.3390/antib7020019




