Dual-Targeting for the Elimination of Cancer Cells with Increased Selectivity
Abstract
:1. Introduction
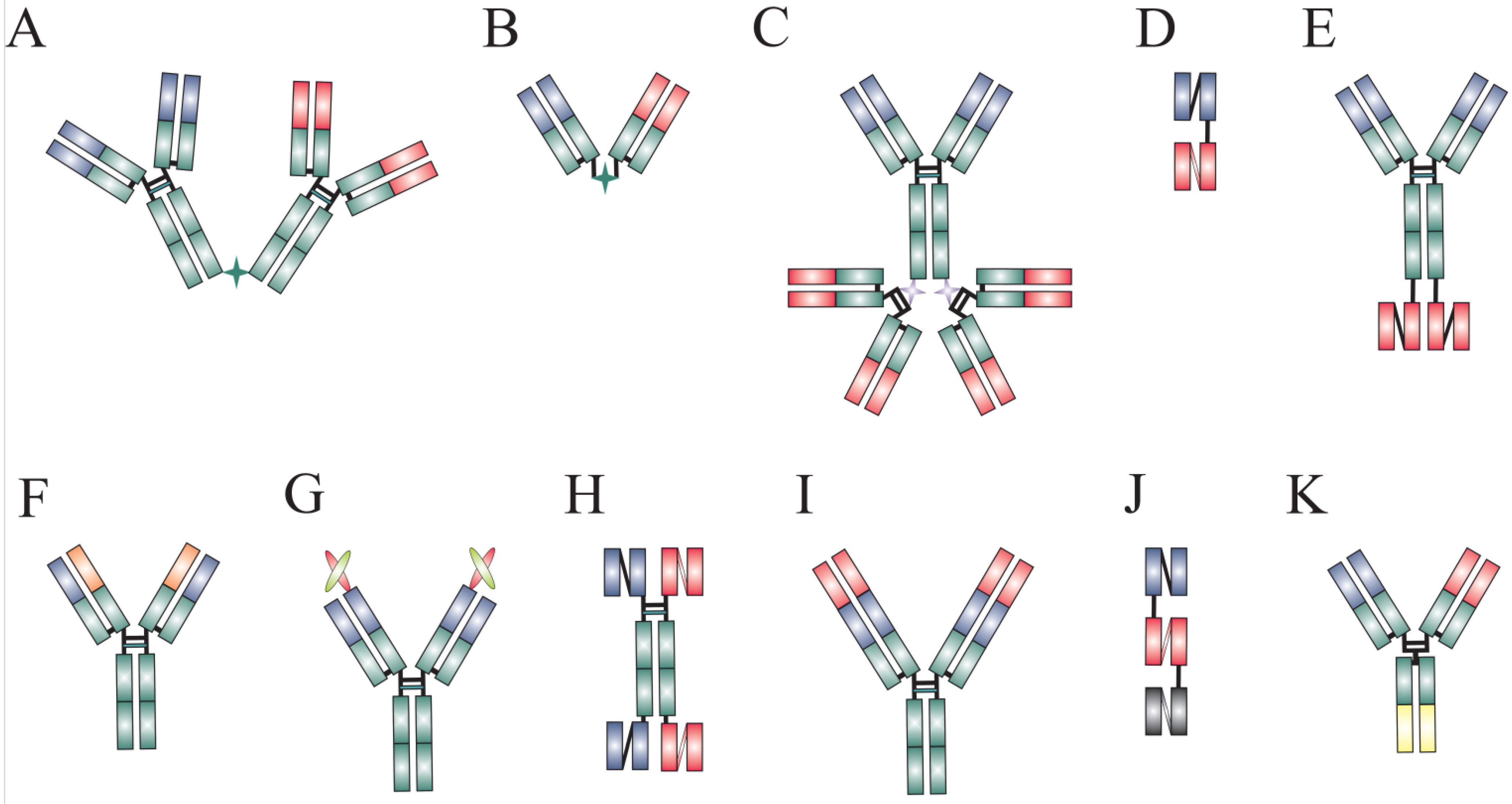
| Format | Specificity | Disease | Function | Company | Ref. |
|---|---|---|---|---|---|
| Bispecific agents without intrinsic effector domain | |||||
| F(ab’)2 | CD20/CD22 | B-ALL | Signaling | - | [25] |
| Bs-scFv | ErbB2/ErbB3 | Breast cancer | Signaling | - | [26] |
| HexAb | CD20/CD74 | Lymphoma | Signaling | Immunomedics IBC Pharmaceuticals | [27] |
| Two-in-one antibody | HER3/EGFR | Several carcinomas | Signal inhibition | Genentech | [28] |
| Tetravalent bs-ab | EGFR/IGF-1R | Several carcinomas | Signal inhibition | Biogen Idec | [29] |
| Bs-scFv | ErbB2/ErbB3 | Breast cancer | Signal inhibition | Merrimack Pharmaceuticals | [30] |
| Bispecific diabody | VEGFR2 | Several carcinomas | Signal inhibition | - | [31] |
| Bispecific diabody | VEGFR2/VEGFR3 | Several carcinomas | Signal inhibition | ImClone Systems Incorporated | [32] |
| Bispecific adnectin | EGFR/IGF-1R | Several carcinomas | Signal inhibition | Bristol-Myers Squibb/ Adnexus | [33] |
| Bispecific CovX-Body (CVX241) | VEGF/Ang2 | Epithelial Ovarian cancer or primary peritoneal cancer | Signal inhibition | Pfizer | [34] |
| Bispecific scFv | PDGFRß/VEGF-A | Several carcinomas | Signal inhibition | - | [35] |
| DVD-IgG | IL-1α/ IL-1β | Inflammatory diseases | Signal inhibition | Abbott | [36] |
| Gold-nanoparticle | EGFR/FRα | ovarian cancer | Signal inhibition | - | [37] |
| Retargeting agents | |||||
| F(ab’)3 | EGFR/Her2/CD64 | Several carcinomas | Retargeting | Medarex | [38] |
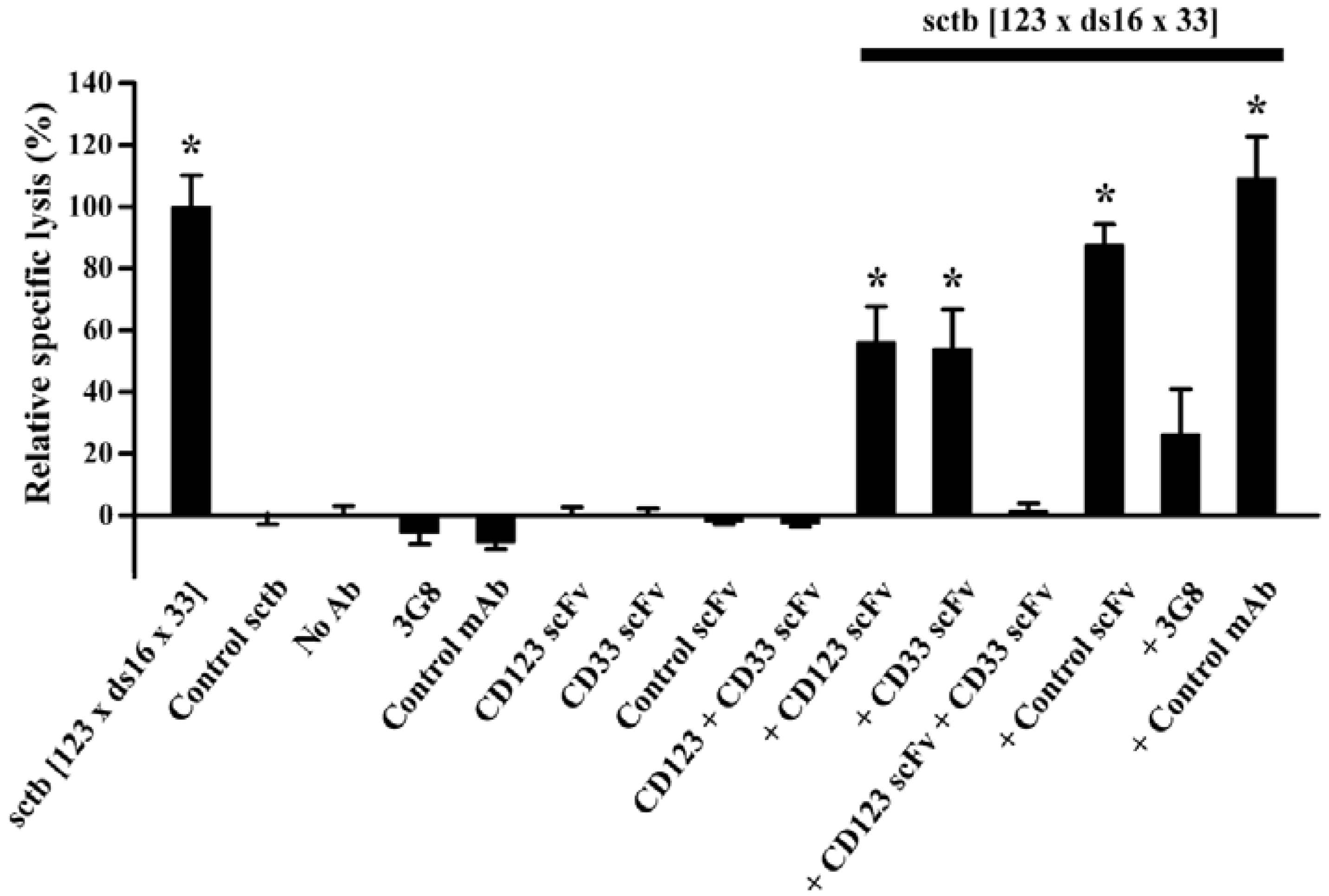
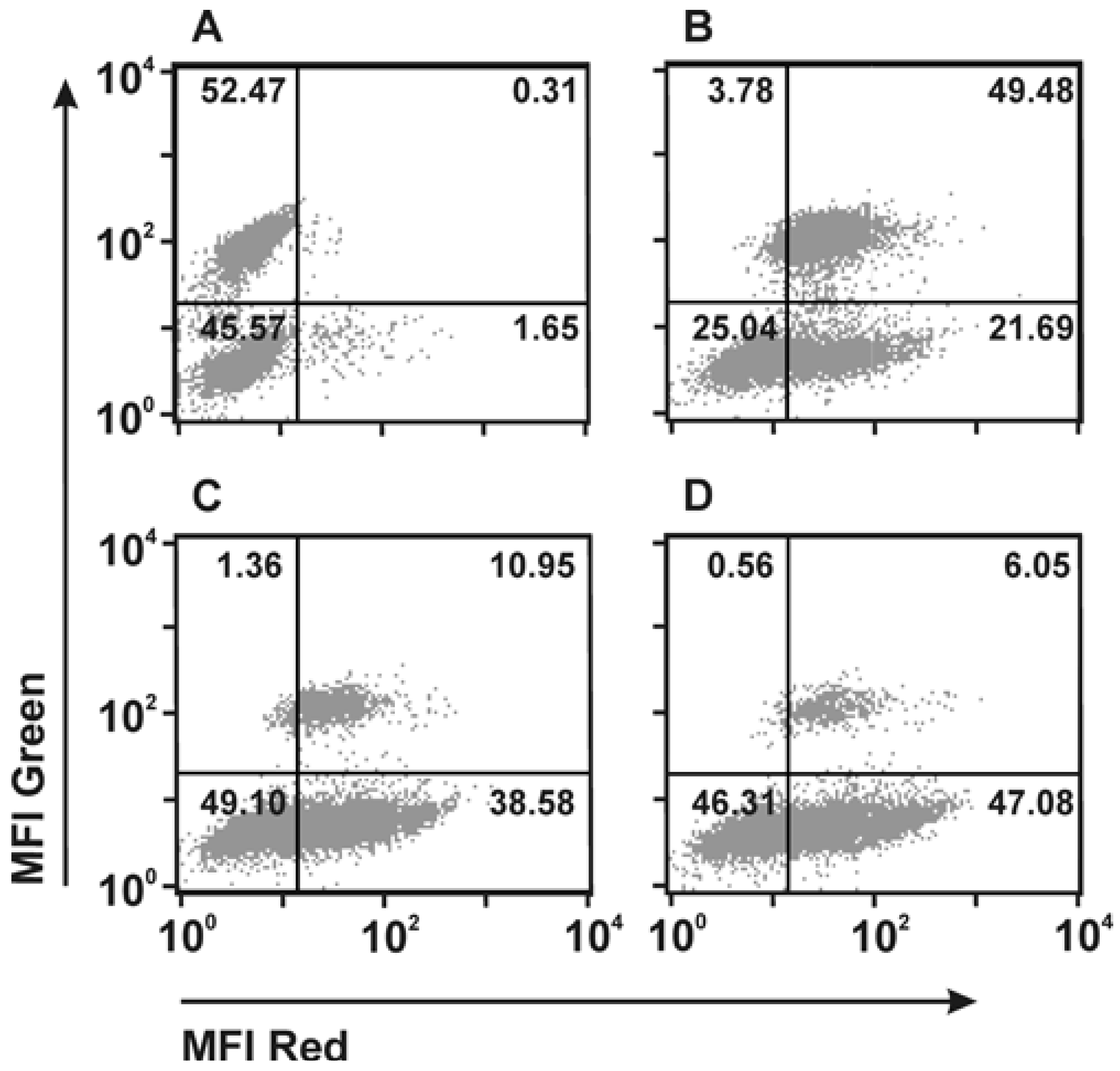
| Triplebody | CD19/CD33/CD16 | MLL | Retargeting | - | [13] |
| Triplebody | CD19/HLAII/CD16 | B-ALL | Retargeting | - | [12] |
| Triplebody | CD123/CD33/CD16 | AML | Retargeting | SpectraMab | [11] |
| Toxins | |||||
| Ligand-directed toxin | EGFR/IL-4R | Pancreatic cancer | Toxic | - | [39,40] |
| Immunotoxin | CD19/CD22 | B-ALL | Toxic | - | [41] |
| Ligand-directed toxin | EGFR/IL-13R | Pancreatic cancer | Toxic | - | [40] |
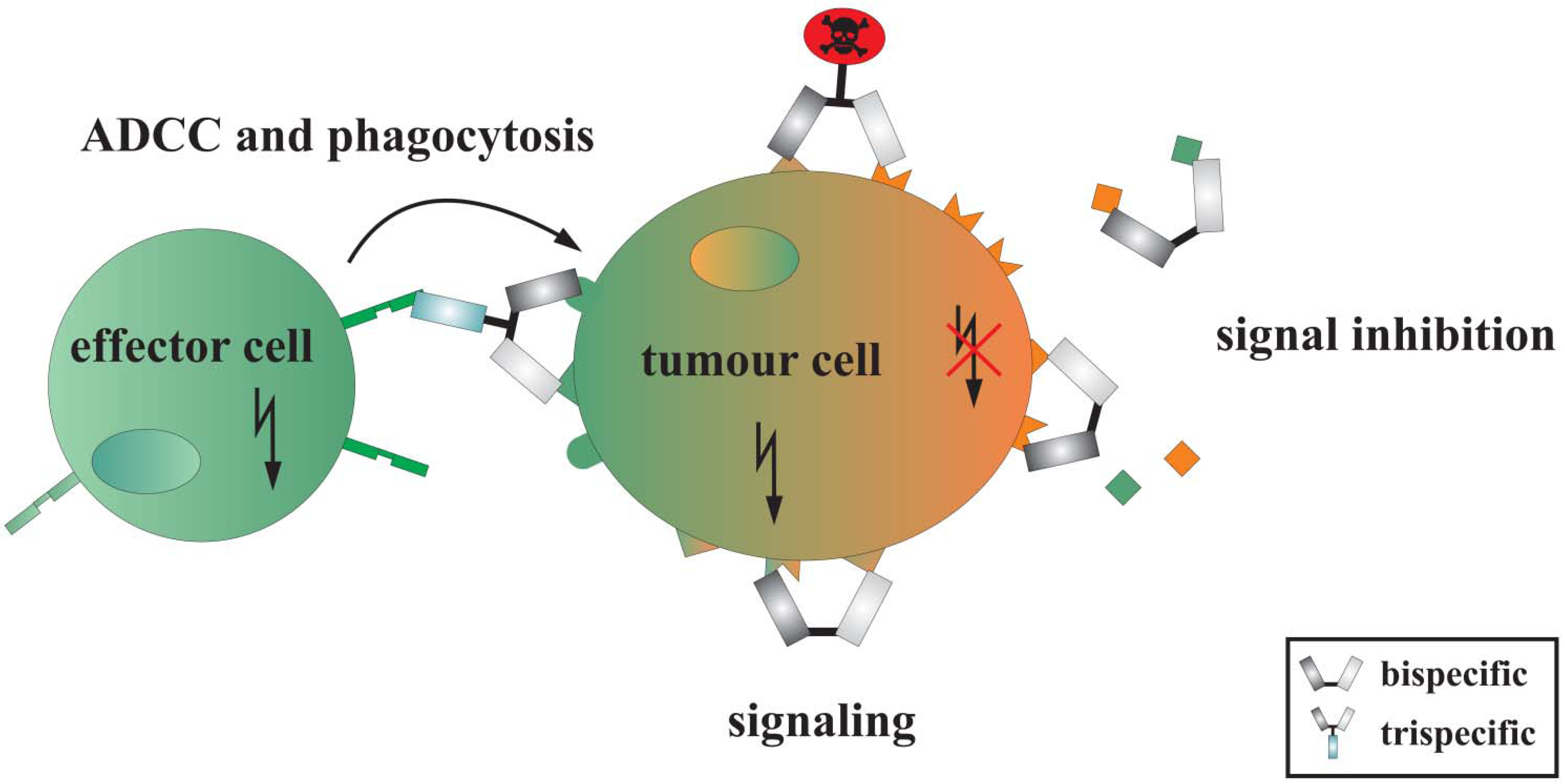
2. Dual-Targeting Agents
2.1. Induction and Inhibition of Intracellular Signals by Dual-Targeting Agents
2.2. Retargeting Agents

2.3. Dual-Targeting Ligand-Directed-Toxins and Antibody-Drug Conjugates
3. Conclusions
Acknowledgements
References and Notes
- Adams, G.P.; Weiner, L.M. Monoclonal antibody therapy of cancer. Nat. Biotechnol. 2005, 23, 1147–1157. [Google Scholar] [CrossRef]
- Duebel, S. Handbook of Therapeutic Antibodies; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2007. [Google Scholar]
- Weiner, L.M.; Dhodapkar, M.V.; Ferrone, S. Monoclonal antibodies for cancer immunotherapy. Lancet 2009, 373, 1033–1040. [Google Scholar]
- Bargou, R.; Leo, E.; Zugmaier, G.; Klinger, M.; Goebeler, M.; Knop, S.; Noppeney, R.; Viardot, A.; Hess, G.; Schuler, M.; et al. Tumor regression in cancer patients by very low doses of a T cell-engaging antibody. Science 2008, 321, 974–977. [Google Scholar]
- Clynes, R. Antitumor antibodies in the treatment of cancer: Fc receptors link opsonic antibody with cellular immunity. Hematol. Oncol. Clin. North Am. 2006, 20, 585–612. [Google Scholar] [CrossRef]
- de Haij, S.; Jansen, J.H.; Boross, P.; Beurskens, F.J.; Bakema, J.E.; Bos, D.L.; Martens, A.; Verbeek, J.S.; Parren, P.W.; van de Winkel, J.G.; et al. In vivo cytotoxicity of type I CD20 antibodies critically depends on Fc receptor ITAM signaling. Cancer Res. 2010, 70, 3209–3217. [Google Scholar]
- Heiss, M.M.; Murawa, P.; Koralewski, P.; Kutarska, E.; Kolesnik, O.O.; Ivanchenko, V.V.; Dudnichenko, A.S.; Aleknaviciene, B.; Razbadauskas, A.; Gore, M.; et al. The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: Results of a prospective randomized phase II/III trial. Int. J. Cancer 2010, 127, 2209–2221. [Google Scholar] [CrossRef]
- Musolino, A.; Naldi, N.; Bortesi, B.; Pezzuolo, D.; Capelletti, M.; Missale, G.; Laccabue, D.; Zerbini, A.; Camisa, R.; Bisagni, G.; et al. Immunoglobulin G fragment C receptor polymorphisms and clinical efficacy of trastuzumab-based therapy in patients with HER-2/neu-positive metastatic breast cancer. J. Clin. Oncol. 2008, 26, 1789–1796. [Google Scholar]
- Zhang, W.; Gordon, M.; Schultheis, A.M.; Yang, D.Y.; Nagashima, F.; Azuma, M.; Chang, H.M.; Borucka, E.; Lurje, G.; Sherrod, A.E.; et al. FCGR2A and FCGR3A polymorphisms associated with clinical outcome of epidermal growth factor receptor expressing metastatic colorectal cancer patients treated with single-agent cetuximab. J. Clin. Oncol. 2007, 25, 3712–3718. [Google Scholar]
- Kellner, C.; Bruenke, J.; Stieglmaier, J.; Schwemmlein, M.; Schwenkert, M.; Singer, H.; Mentz, K.; Peipp, M.; Lang, P.; Oduncu, F.; et al. A novel CD19-directed recombinant bispecific antibody derivative with enhanced immune effector functions for human leukemic cells. J. Immunother. 2008, 31, 871–884. [Google Scholar] [CrossRef]
- Kugler, M.; Stein, C.; Kellner, C.; Mentz, K.; Saul, D.; Schwenkert, M.; Schubert, I.; Singer, H.; Oduncu, F.; Stockmeyer, B.; et al. A recombinant trispecific single-chain Fv derivative directed against CD123 and CD33 mediates effective elimination of acute myeloid leukaemia cells by dual targeting. Br. J. Haematol. 2010, 150, 574–586. [Google Scholar] [CrossRef]
- Schubert, I.; Kellner, C.; Stein, C.; Kugler, M.; Schwenkert, M.; Saul, D.; Berens, C.; Oduncu, F.; Stockmeyer, B.; Mackensen, A.; et al. A recombinant triplebody with specificity for CD19 and HLA-DR mediates preferential binding to antigen double-positive cells by dual-targeting. mAbs 2012, 4, 45–56. [Google Scholar] [CrossRef]
- Schubert, I.; Kellner, C.; Stein, C.; Kugler, M.; Schwenkert, M.; Saul, D.; Mentz, K.; Singer, H.; Stockmeyer, B.; Hillen, W.; et al. A single-chain triplebody with specificity for CD19 and CD33 mediates effective lysis of mixed lineage leukemia cells by dual targeting. MAbs 2011, 3, 21–30. [Google Scholar] [CrossRef]
- Topp, M.S.; Kufer, P.; Gokbuget, N.; Goebeler, M.; Klinger, M.; Neumann, S.; Horst, H.A.; Raff, T.; Viardot, A.; Schmid, M.; et al. Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J. Clin. Oncol. 2011, 29, 2493–2498. [Google Scholar]
- Topp, M.G.N.; Kufer, P.; Zugmaier, G.; Degenhard, E.; Neumann, S.; Horst, H.A.; Viardot, A.; Schmid, M.; Ottmann, O.G.; Schmidt, M.; et al. Treatment with anti-CD19 BiTE antibody bli¬natumomab (MT103/MEDI-538) is able to eliminate minimal residual disease (MRD) in patients with B-precursor acute lmphoblastic leukemia (ALL): First results of an ongoing phase II study (ASH Annual Meeting Abstract). Blood 2008, 112, 1926. [Google Scholar]
- Bluemel, C.; Hausmann, S.; Fluhr, P.; Sriskandarajah, M.; Stallcup, W.B.; Baeuerle, P.A.; Kufer, P. Epitope distance to the target cell membrane and antigen size determine the potency of T cell-mediated lysis by BiTE antibodies specific for a large melanoma surface antigen. Cancer Immunol. Immunother. 2010, 59, 1197–1209. [Google Scholar]
- Huhalov, A.; Chester, K.A. Engineered single chain antibody fragments for radioimmunotherapy. Q. J. Nucl. Med. Mol. Imaging. 2004, 48, 279–288. [Google Scholar]
- Kipriyanov, S.M.; Moldenhauer, G.; Schuhmacher, J.; Cochlovius, B.; Von der Lieth, C.W.; Matys, E.R.; Little, M. Bispecific tandem diabody for tumor therapy with improved antigen binding and pharmacokinetics. J. Mol. Biol. 1999, 293, 41–56. [Google Scholar] [CrossRef]
- Raghavan, M.; Chen, M.Y.; Gastinel, L.N.; Bjorkman, P.J. Investigation of the interaction between the class I MHC-related Fc receptor and its immunoglobulin G ligand. Immunity 1994, 1, 303–315. [Google Scholar] [CrossRef]
- Kubetzko, S.; Balic, E.; Waibel, R.; Zangemeister-Wittke, U.; Pluckthun, A. PEGylation and multimerization of the anti-p185HER-2 single chain Fv fragment 4D5: Effects on tumor targeting. J. Biol. Chem. 2006, 281, 35186–35201. [Google Scholar]
- Schoonjans, R.; Willems, A.; Schoonooghe, S.; Fiers, W.; Grooten, J.; Mertens, N. Fab chains as an efficient heterodimerization scaffold for the production of recombinant bispecific and trispecific antibody derivatives. J. Immunol. 2000, 165, 7050–7057. [Google Scholar]
- Muller, D.; Karle, A.; Meissburger, B.; Hofig, I.; Stork, R.; Kontermann, R.E. Improved pharmacokinetics of recombinant bispecific antibody molecules by fusion to human serum albumin. J. Biol. Chem. 2007, 282, 12650–12660. [Google Scholar]
- Kontermann, R.E. Strategies for extended serum half-life of protein therapeutics. Curr. Opin. Biotechnol. 2011, 22, 868–876. [Google Scholar] [CrossRef]
- Ring, D.B.; Hsieh-Ma, S.T.; Shi, T.; Reeder, J. Antigen forks: bispecific reagents that inhibit cell growth by binding selected pairs of tumor antigens. Cancer Immunol. Immunother. 1994, 39, 41–48. [Google Scholar] [CrossRef]
- Tuscano, J.M.; Ma, Y.; Martin, S.M.; Kato, J.; O'Donnell, R.T. The Bs20x22 anti-CD20-CD22 bispecific antibody has more lymphomacidal activity than do the parent antibodies alone. Cancer Immunol. Immunother. 2011, 60, 771–780. [Google Scholar] [CrossRef]
- Robinson, M.K.; Hodge, K.M.; Horak, E.; Sundberg, A.L.; Russeva, M.; Shaller, C.C.; von Mehren, M.; Shchaveleva, I.; Simmons, H.H.; Marks, J.D.; et al. Targeting ErbB2 and ErbB3 with a bispecific single-chain Fv enhances targeting selectivity and induces a therapeutic effect in vitro. Br. J. Cancer 2008, 99, 1415–1425. [Google Scholar] [CrossRef]
- Gupta, P.; Goldenberg, D.M.; Rossi, E.A.; Cardillo, T.M.; Byrd, J.C.; Muthusamy, N.; Furman, R.R.; Chang, C.H. Dual-targeting immunotherapy of lymphoma: potent cytotoxicity of anti-CD20/CD74 bispecific antibodies in mantle cell and other lymphomas. Blood 2012. Epub ahead of print. [Google Scholar]
- Schaefer, G.; Haber, L.; Crocker, L.M.; Shia, S.; Shao, L.; Dowbenko, D.; Totpal, K.; Wong, A.; Lee, C.V.; Stawicki, S.; et al. A two-in-one antibody against HER3 and EGFR has superior inhibitory activity compared with monospecific antibodies. Cancer Cell 2011, 20, 472–486. [Google Scholar] [CrossRef]
- Dong, J.; Sereno, A.; Aivazian, D.; Langley, E.; Miller, B.R.; Snyder, W.B.; Chan, E.; Cantele, M.; Morena, R.; Joseph, I.B.; et al. A stable IgG-like bispecific antibody targeting the epidermal growth factor receptor and the type I insulin-like growth factor receptor demonstrates superior anti-tumor activity. MAbs 2011, 3, 273–288. [Google Scholar] [CrossRef]
- McDonagh, C.F.; Huhalov, A.; Harms, B.D.; Adams, S.; Paragas, V.; Oyama, S.; Zhang, B.; Luus, L.; Overland, R.; Nguyen, S.; et al. Antitumor activity of a novel bispecific antibody that targets the ErbB2/ErbB3 oncogenic unit and inhibits heregulin-induced activation of ErbB3. Mol. Cancer Ther. 2012, 11, 582–593. [Google Scholar] [CrossRef]
- Lu, D.; Kotanides, H.; Jimenez, X.; Zhou, Q.; Persaud, K.; Bohlen, P.; Witte, L.; Zhu, Z. Acquired antagonistic activity of a bispecific diabody directed against two different epitopes on vascular endothelial growth factor receptor 2. J. Immunol. Methods 1999, 230, 159–171. [Google Scholar] [CrossRef]
- Jimenez, X.; Lu, D.; Brennan, L.; Persaud, K.; Liu, M.; Miao, H.; Witte, L.; Zhu, Z. A recombinant, fully human, bispecific antibody neutralizes the biological activities mediated by both vascular endothelial growth factor receptors 2 and 3. Mol. Cancer Ther. 2005, 4, 427–434. [Google Scholar]
- Emanuel, S.L.; Engle, L.J.; Chao, G.; Zhu, R.R.; Cao, C.; Lin, Z.; Yamniuk, A.P.; Hosbach, J.; Brown, J.; Fitzpatrick, E.; et al. A fibronectin scaffold approach to bispecific inhibitors of epidermal growth factor receptor and insulin-like growth factor-I receptor. MAbs 2011, 3, 38–48. [Google Scholar] [CrossRef]
- Doppalapudi, V.R.; Huang, J.; Liu, D.; Jin, P.; Liu, B.; Li, L.; Desharnais, J.; Hagen, C.; Levin, N.J.; Shields, M.J.; et al. Chemical generation of bispecific antibodies. Proc. Natl. Acad. Sci. USA 2010, 107, 22611–22616. [Google Scholar]
- Mabry, R.; Gilbertson, D.G.; Frank, A.; Vu, T.; Ardourel, D.; Ostrander, C.; Stevens, B.; Julien, S.; Franke, S.; Meengs, B.; et al. A dual-targeting PDGFRbeta/VEGF-A molecule assembled from stable antibody fragments demonstrates anti-angiogenic activity in vitro and in vivo. MAbs 2010, 2, 20–34. [Google Scholar] [CrossRef]
- Wu, C.; Ying, H.; Bose, S.; Miller, R.; Medina, L.; Santora, L.; Ghayur, T. Molecular construction and optimization of anti-human IL-1alpha/beta dual variable domain immunoglobulin (DVD-Ig) molecules. MAbs 2009, 1, 339–347. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Khan, J.A.; Curran, G.L.; Robertson, J.D.; Bhattacharya, R.; Mukherjee, P. Efficient delivery of gold nanoparticles by dual receptor targeting. Adv. Mater. 2011, 23, 5034–5038. [Google Scholar]
- Somasundaram, C.; Sundarapandiyan, K.; Keler, T.; Deo, Y.M.; Graziano, R.F. Development of a trispecific antibody conjugate that directs two distinct tumor-associated antigens to CD64 on myeloid effector cells. Hum. Antibodies 1999, 9, 47–54. [Google Scholar]
- Oh, S.; Stish, B.J.; Sachdev, D.; Chen, H.; Dudek, A.Z.; Vallera, D.A. A novel reduced immunogenicity bispecific targeted toxin simultaneously recognizing human epidermal growth factor and interleukin-4 receptors in a mouse model of metastatic breast carcinoma. Clin. Cancer Res. 2009, 15, 6137–6147. [Google Scholar] [CrossRef]
- Oh, S.; Stish, B.J.; Vickers, S.M.; Buchsbaum, D.J.; Saluja, A.K.; Vallera, D.A. A new drug delivery method of bispecific ligand-directed toxins, which reduces toxicity and promotes efficacy in a model of orthotopic pancreatic cancer. Pancreas 2010, 39, 913–922. [Google Scholar] [CrossRef]
- Vallera, D.A.; Chen, H.; Sicheneder, A.R.; Panoskaltsis-Mortari, A.; Taras, E.P. Genetic alteration of a bispecific ligand-directed toxin targeting human CD19 and CD22 receptors resulting in improved efficacy against systemic B cell malignancy. Leuk. Res. 2009, 33, 1233–1242. [Google Scholar] [CrossRef]
- Hurwitz, S.J.; Zhang, H.; Yun, S.; Batuwangala, T.D.; Steward, M.; Holmes, S.D.; Rycroft, D.; Pan, L.; Tighiouart, M.; Shin, H.J.; et al. Pharmacodynamics of DT-IgG, a dual-targeting antibody against VEGF-EGFR, in tumor xenografted mice. Cancer Chemother. Pharmacol. 2011, 69, 577–590. [Google Scholar]
- Kontermann, R.E. Bispecific Antibodies, 1st ed.; Springer-Verlag Berlin: Heidelberg, Germany, 2011; p. 373. [Google Scholar]
- Brinkmann, U.; Reiter, Y.; Jung, S.H.; Lee, B.; Pastan, I. A recombinant immunotoxin containing a disulfide-stabilized Fv fragment. Proc. Natl. Acad. Sci. USA 1993, 90, 7538–7542. [Google Scholar] [CrossRef]
- Jordan, C.T.; Upchurch, D.; Szilvassy, S.J.; Guzman, M.L.; Howard, D.S.; Pettigrew, A.L.; Meyerrose, T.; Rossi, R.; Grimes, B.; Rizzieri, D.A.; et al. The interleukin-3 receptor alpha chain is a unique marker for human acute myelogenous leukemia stem cells. Leukemia 2000, 14, 1777–1784. [Google Scholar]
- Taussig, D.C.; Pearce, D.J.; Simpson, C.; Rohatiner, A.Z.; Lister, T.A.; Kelly, G.; Luongo, J.L.; Danet-Desnoyers, G.A.; Bonnet, D. Hematopoietic stem cells express multiple myeloid markers: implications for the origin and targeted therapy of acute myeloid leukemia. Blood 2005, 106, 4086–4092. [Google Scholar]
- Singer, H.; Kellner, C.; Lanig, H.; Aigner, M.; Stockmeyer, B.; Oduncu, F.; Schwemmlein, M.; Stein, C.; Mentz, K.; Mackensen, A.; et al. Effective elimination of acute myeloid leukemic cells by recombinant bispecific antibody derivatives directed against CD33 and CD16. J. Immunother. 2010, 33, 599–608. [Google Scholar] [CrossRef]
- Kugler, M.; Stein, C.; Schwenkert, M.; Saul, D.; Vockentanz, L.; Huber, T.; Wetzel, S.K.; Scholz, O.; Pluckthun, A.; Honegger, A.; et al. Stabilization and humanization of a single-chain Fv antibody fragment specific for human lymphocyte antigen CD19 by designed point mutations and CDR-grafting onto a human framework. Protein Eng. Des. Sel. 2009, 22, 135–147. [Google Scholar]
- Armstrong, S.A.; Look, A.T. Molecular genetics of acute lymphoblastic leukemia. J. Clin. Oncol. 2005, 23, 6306–6315. [Google Scholar] [CrossRef]
- Hrusak, O.; Porwit-MacDonald, A. Antigen expression patterns reflecting genotype of acute leukemias. Leukemia 2002, 16, 1233–1258. [Google Scholar] [CrossRef]
- Schubert, I. University of Erlangen-Nuremberg: Erlangen, Germany, 2012; Unpublished work.
- Oh, S.; Todhunter, D.A.; Panoskaltsis-Mortari, A.; Buchsbaum, D.J.; Toma, S.; Vallera, D.A. A deimmunized bispecific ligand-directed Toxin that shows an impressive anti-pancreatic cancer effect in a systemic nude mouse orthotopic model. Pancreas 2012. Epub ahead of print. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schubert, I.; Stein, C.; Fey, G.H. Dual-Targeting for the Elimination of Cancer Cells with Increased Selectivity. Antibodies 2012, 1, 2-18. https://doi.org/10.3390/antib1010002
Schubert I, Stein C, Fey GH. Dual-Targeting for the Elimination of Cancer Cells with Increased Selectivity. Antibodies. 2012; 1(1):2-18. https://doi.org/10.3390/antib1010002
Chicago/Turabian StyleSchubert, Ingo, Christoph Stein, and Georg H. Fey. 2012. "Dual-Targeting for the Elimination of Cancer Cells with Increased Selectivity" Antibodies 1, no. 1: 2-18. https://doi.org/10.3390/antib1010002
APA StyleSchubert, I., Stein, C., & Fey, G. H. (2012). Dual-Targeting for the Elimination of Cancer Cells with Increased Selectivity. Antibodies, 1(1), 2-18. https://doi.org/10.3390/antib1010002




