Temporal Resource Continuity Increases Predator Abundance in a Metapopulation Model: Insights for Conservation and Biocontrol
Abstract
1. Introduction
2. Materials and Methods
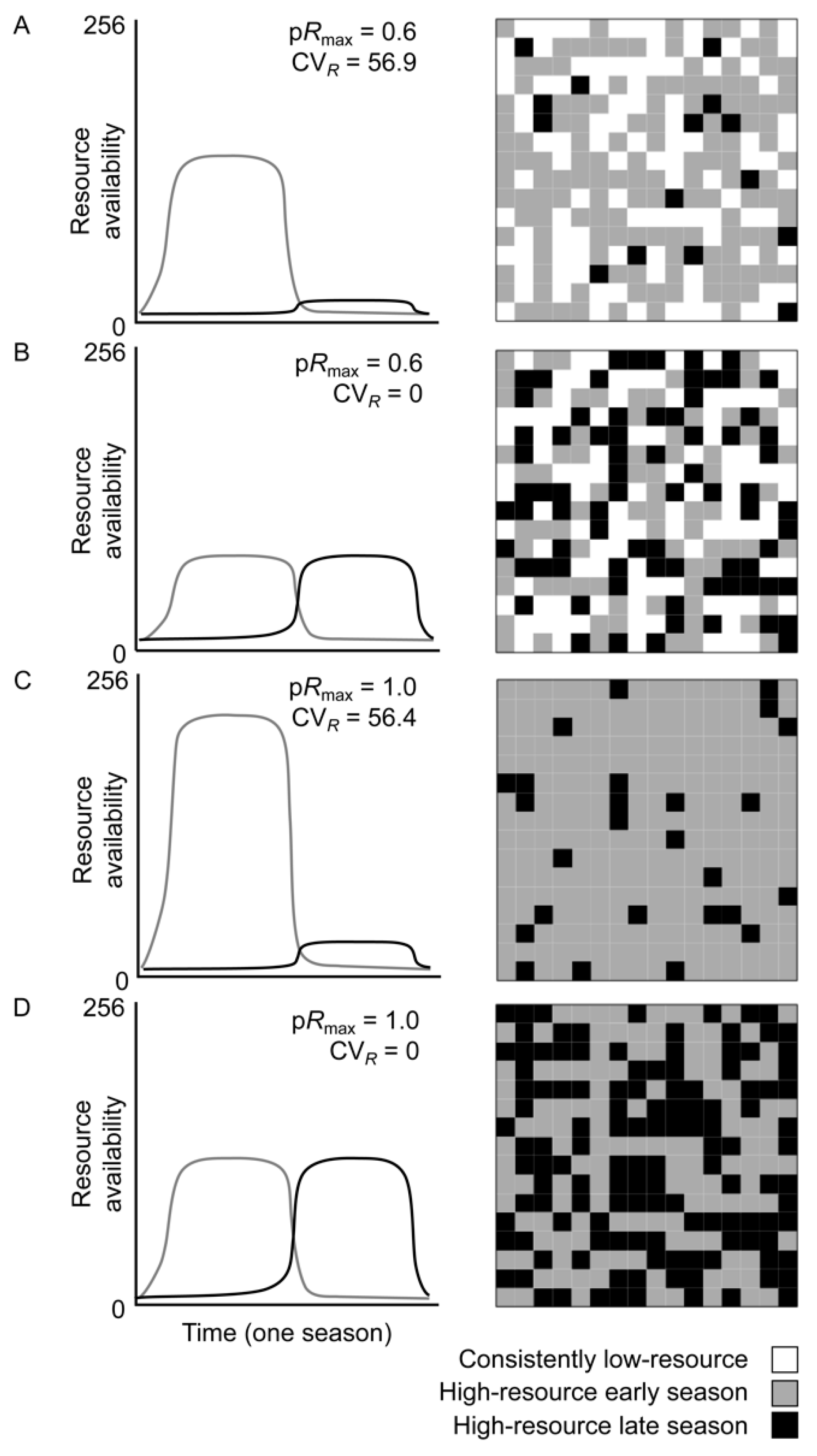
2.1. Model Landscapes
2.2. Predator–Prey Metapopulation Model
2.3. Model Analysis
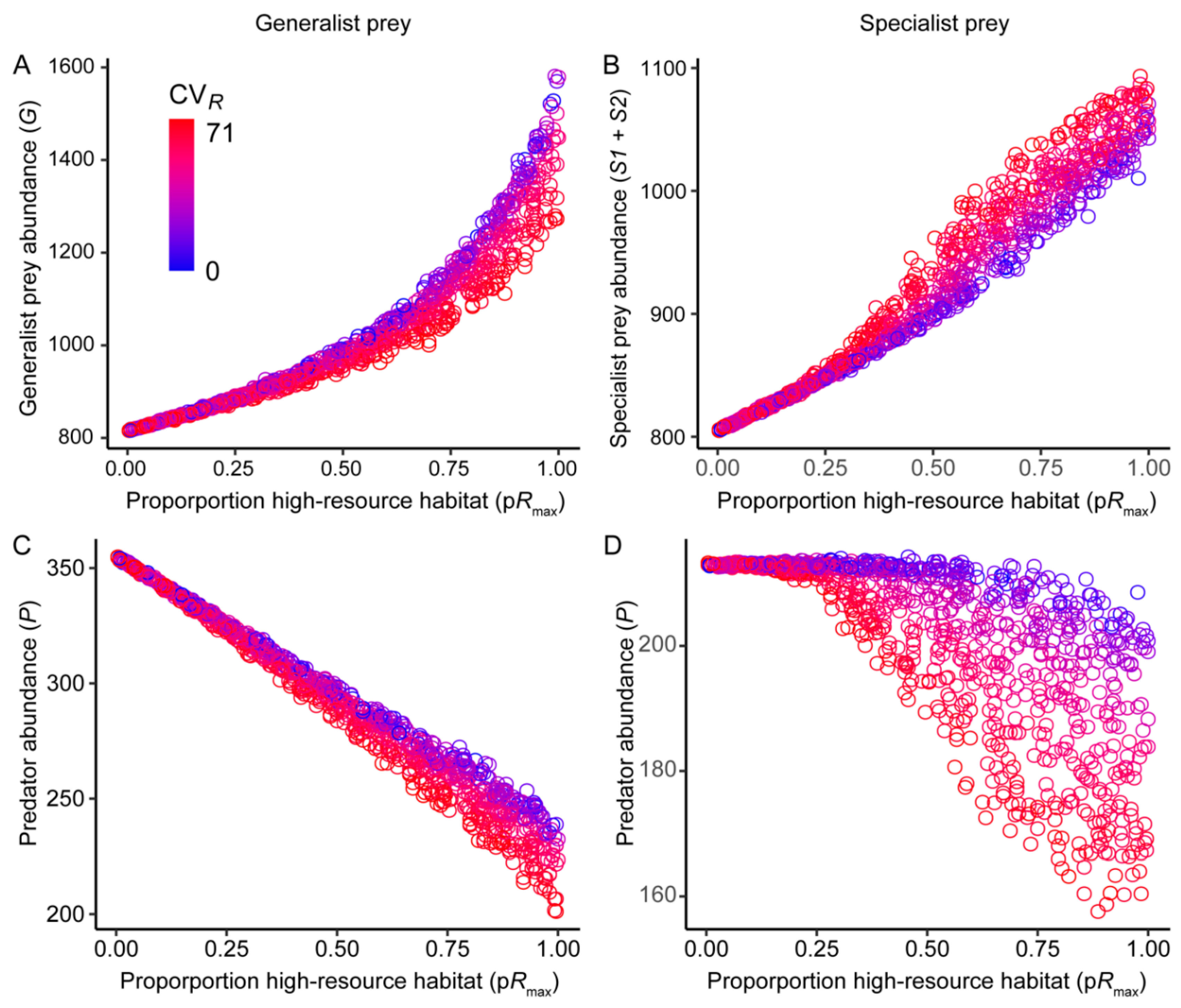
3. Results
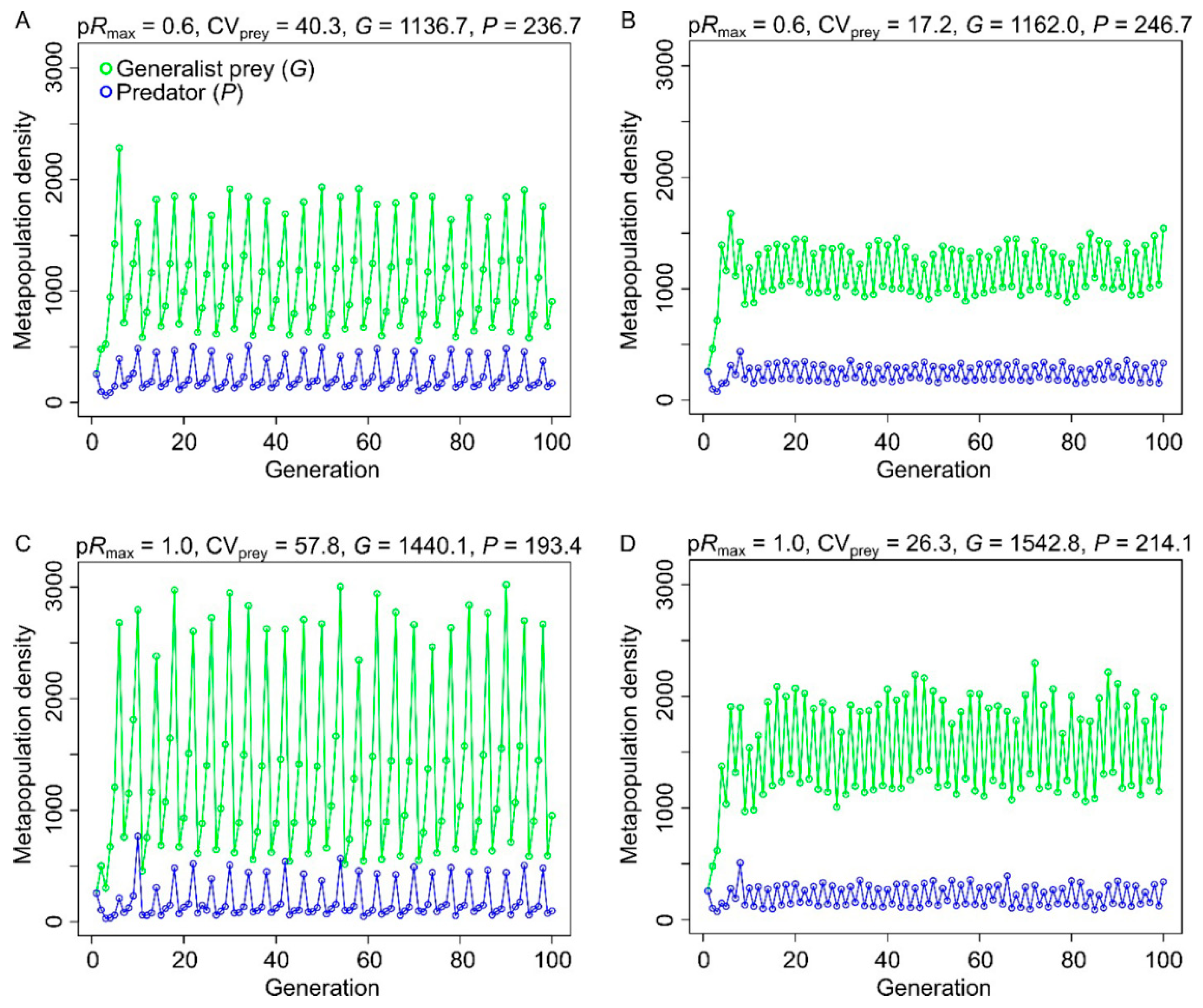
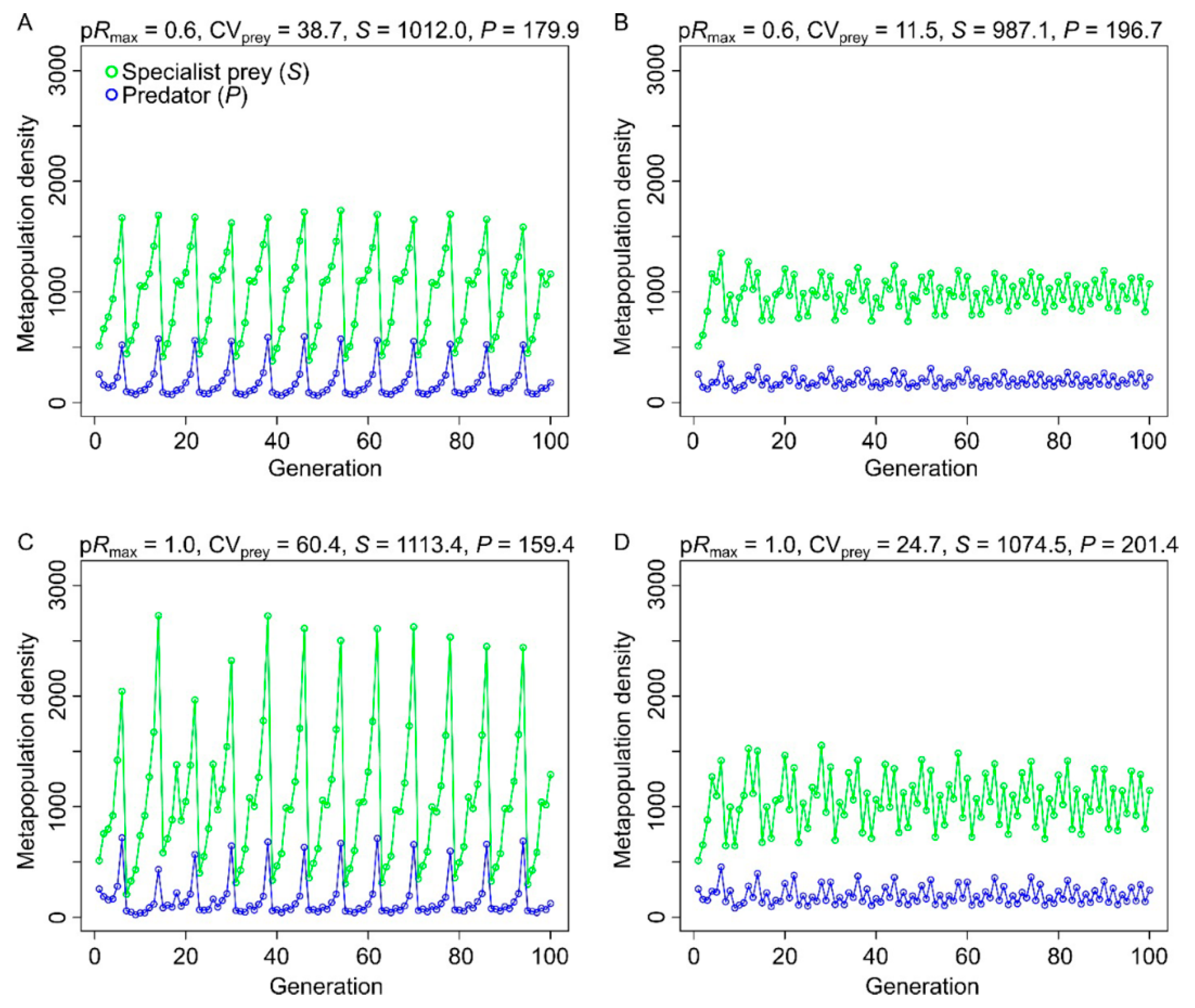
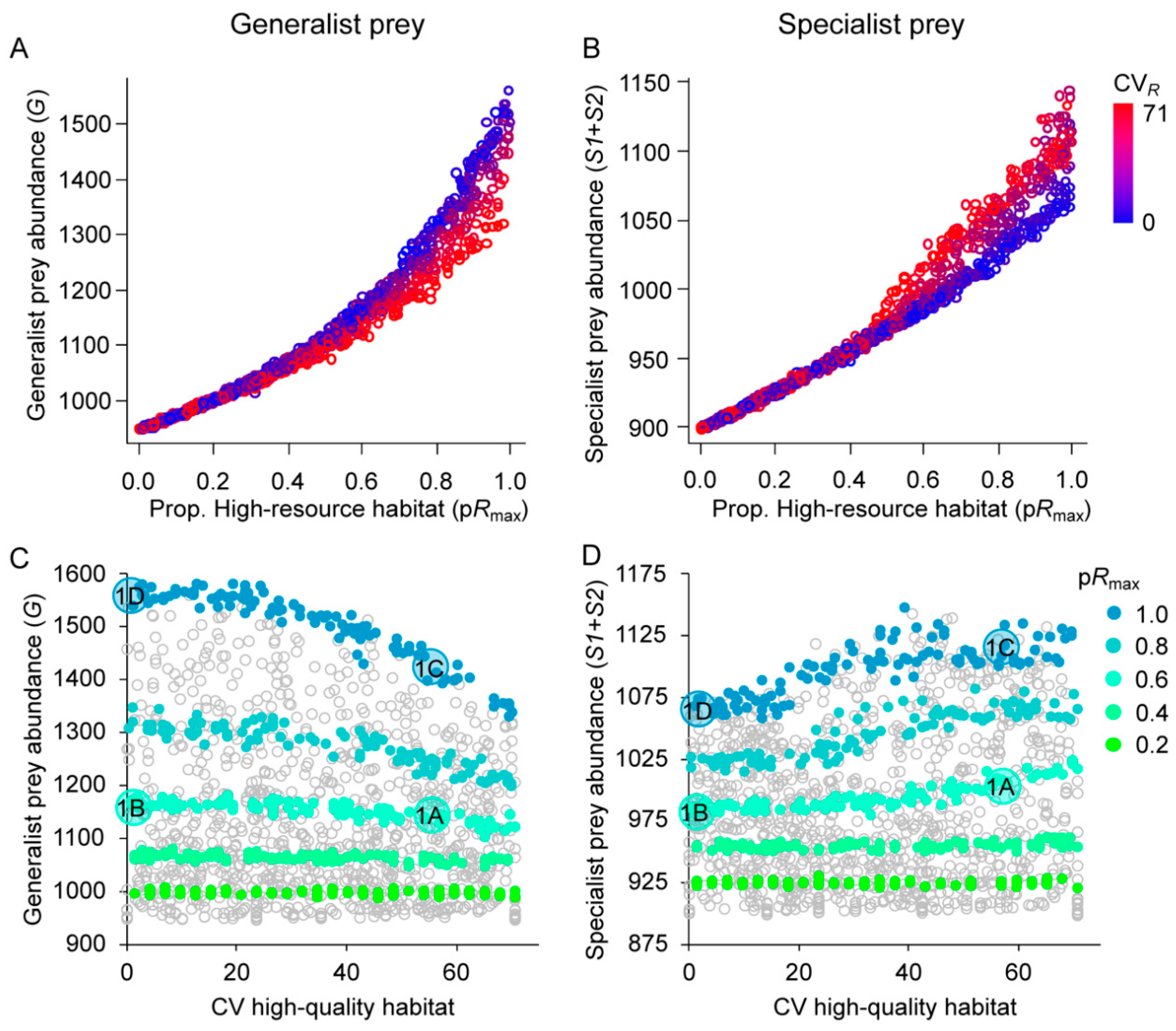
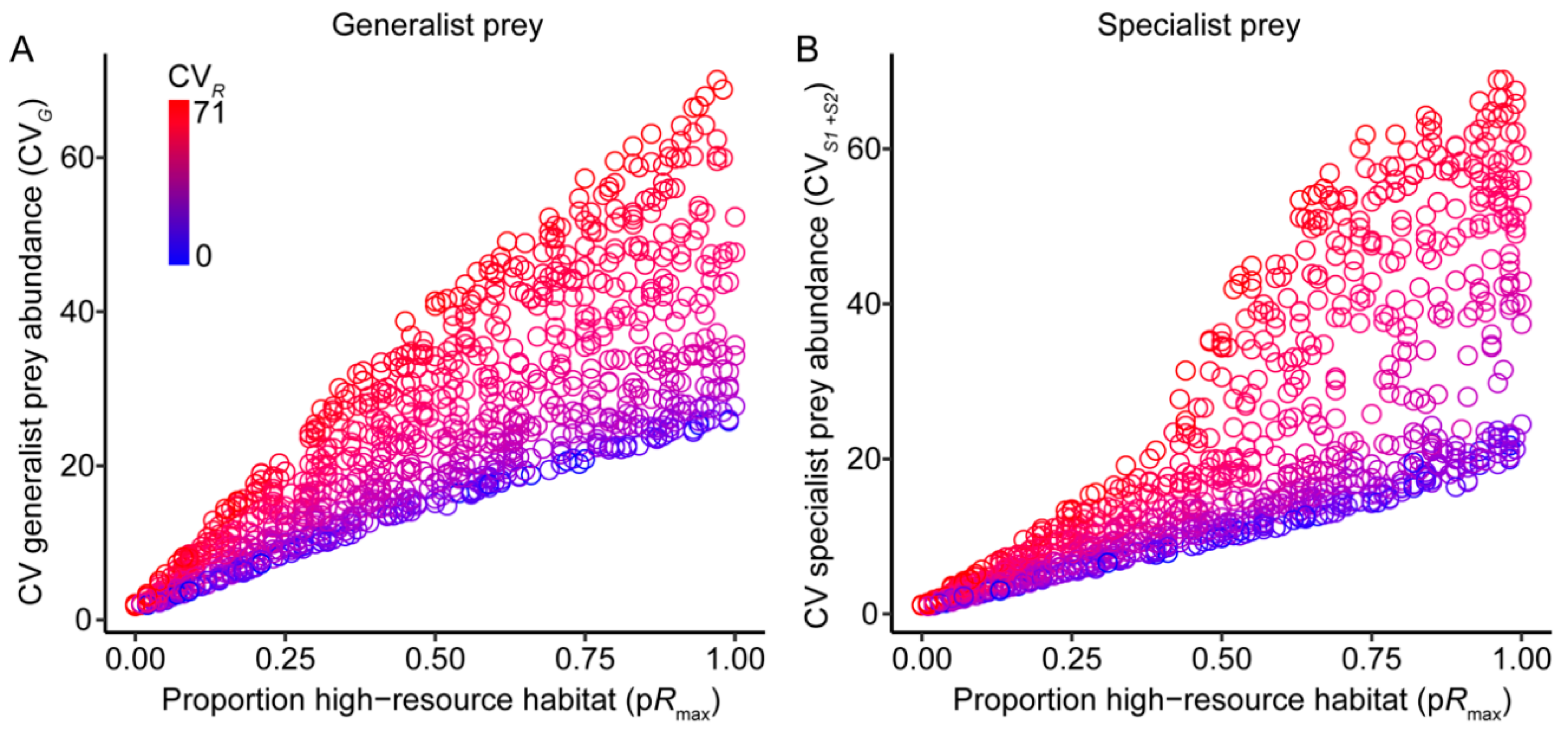
3.1. Effects of Resource Amount and Continuity on Prey
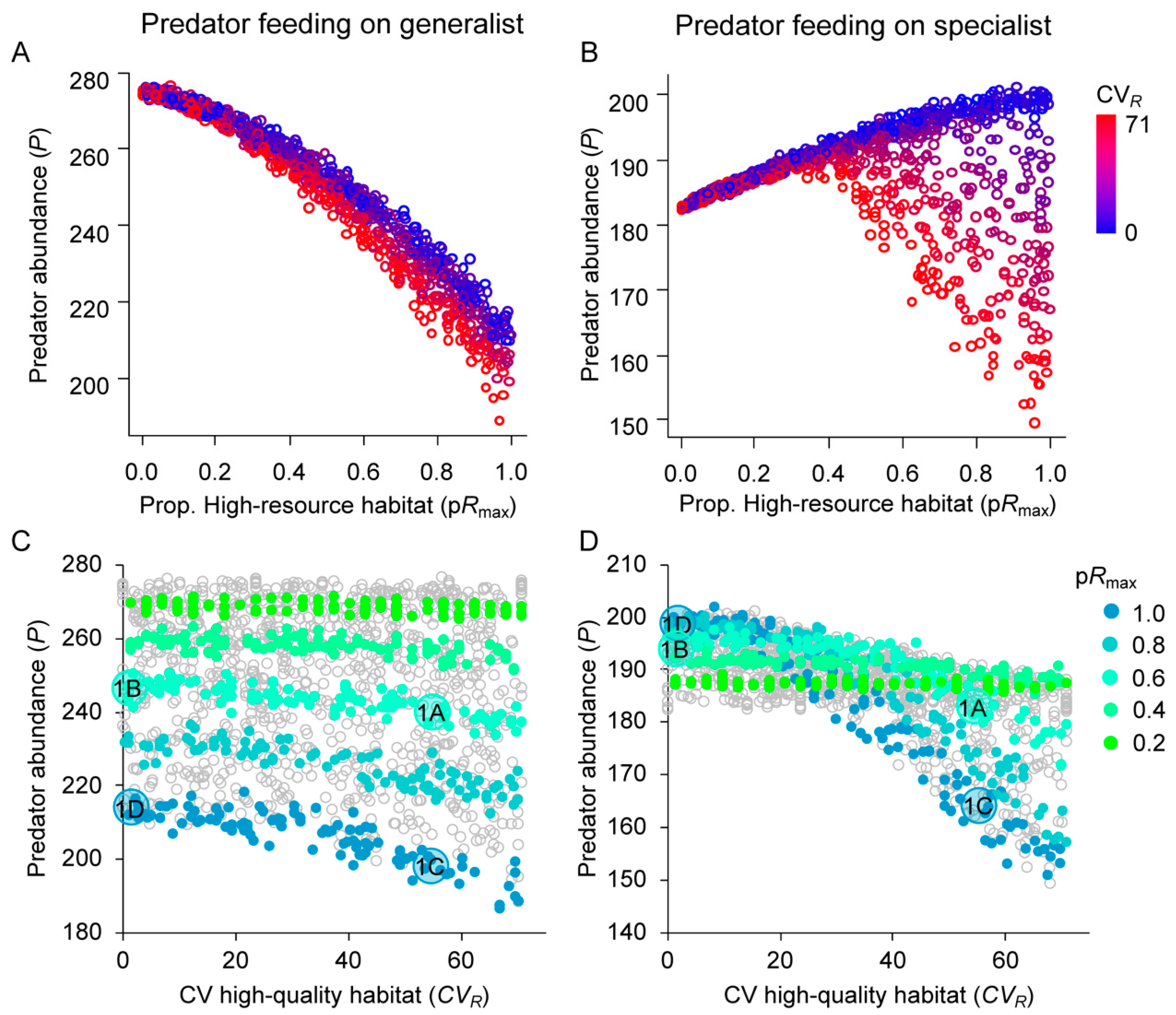
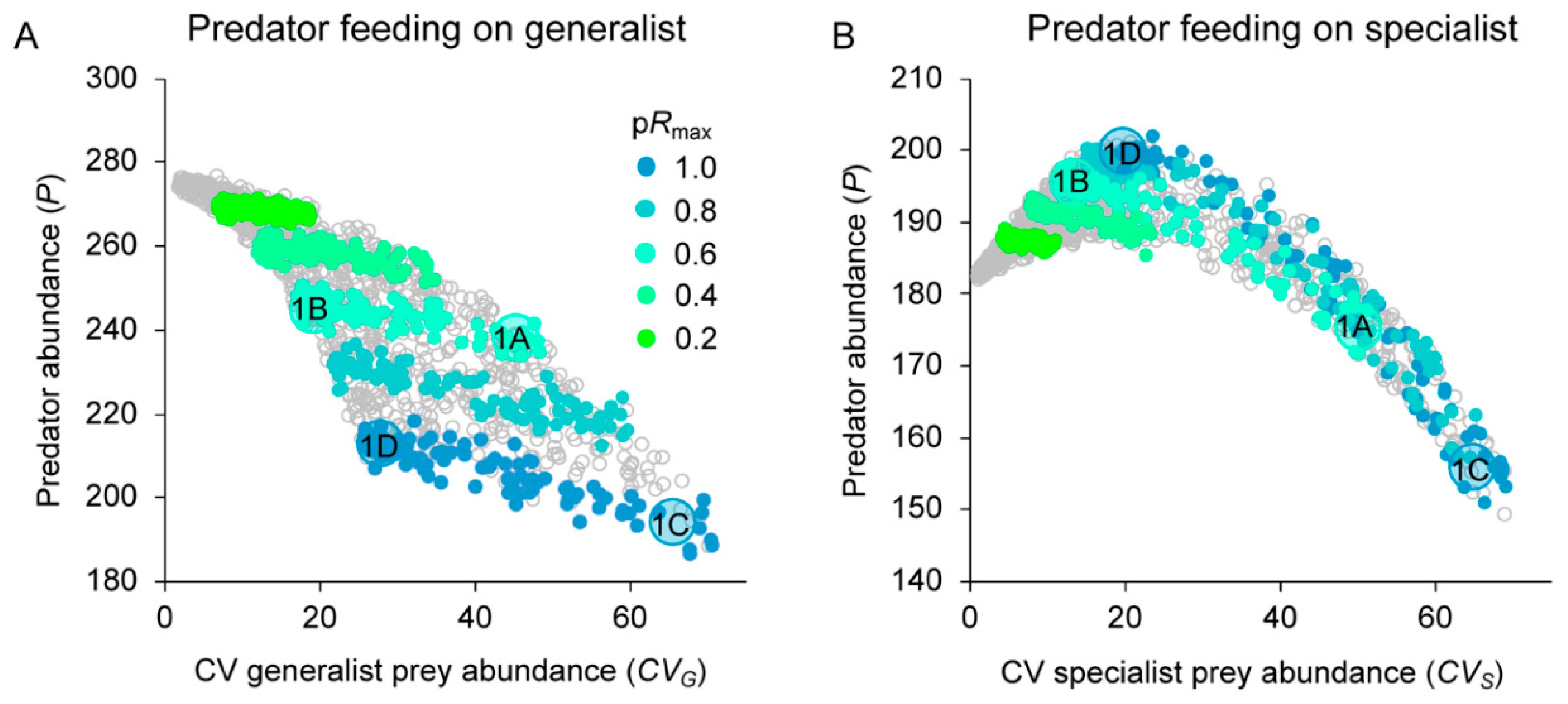
3.2. Effects of Resource Amount and Continuity on Predators
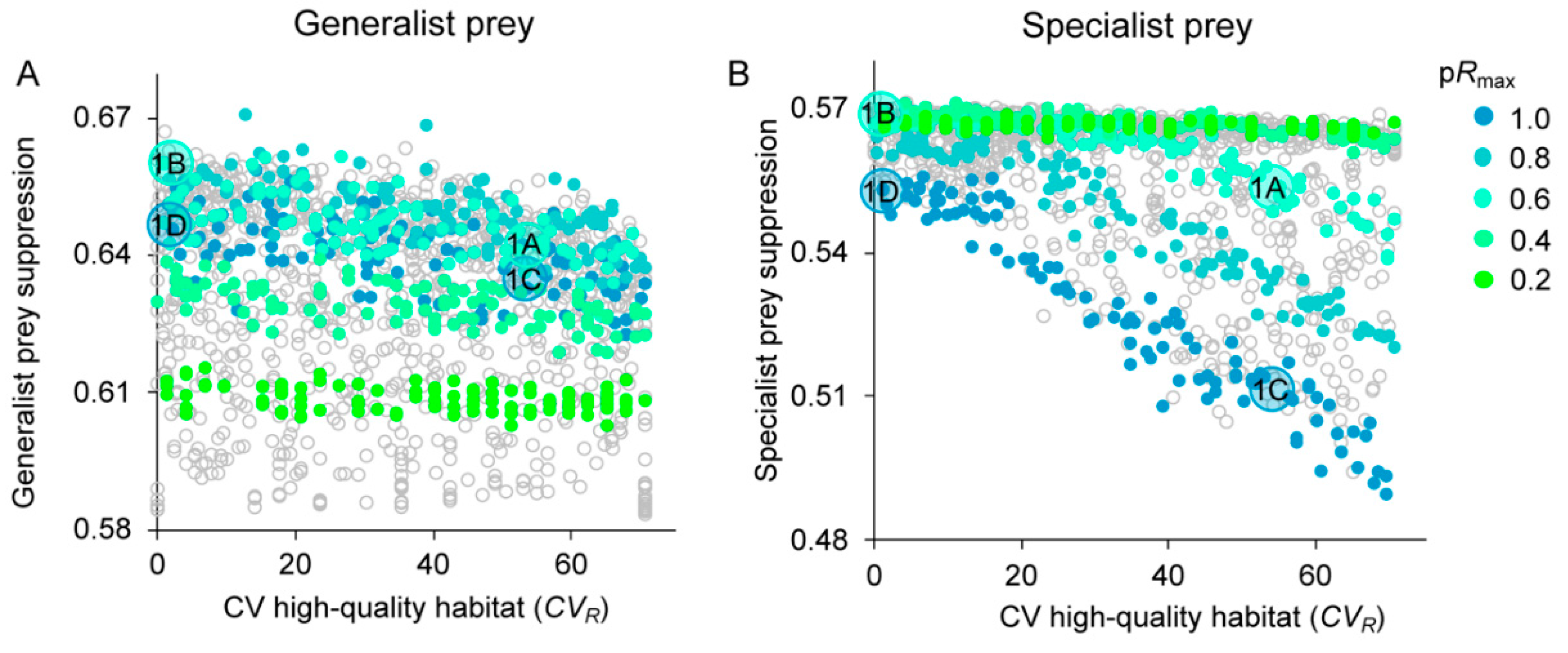
3.3. Prey Suppression
4. Discussion
4.1. Generalist Prey
4.2. Specialist Prey
4.3. Applications to Biological Control and Conservation
4.4. Limitations and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | Definition | Value | |
|---|---|---|---|
| Landscape | R | Number of high-resource patches in the landscape | 0 to 256 |
| pRmax | Proportion of maximum R | 0 to 1 | |
| CVR | Coefficient of variation of R between seasons | Equation (3) | |
| Prey | Git | Generalist prey population density in patch i at time t | Equation (1) |
| S1it | Specialist 1 prey population density in patch i at time t | Equation (1) | |
| S2it | Specialist 2 prey population density in patch i at time t | Equation (1) | |
| Sit | Combined specialist prey density in patch i at time t | S2it + S2it | |
| r | Prey intrinsic growth rate | 1.0 | |
| Kit | Prey carrying capacity in patch i at time t | 100 (xi = 1) or 10 (xi = 0) | |
| mG, S1, or S2 | Prey mortality rate | 0.1 or 0.8 | |
| dG, S1, or S2 | Proportion of G, S1, or S2 dispersing from each patch | 0.2 | |
| Iit | Sum of prey immigrating to patch i at time t | - | |
| Eit | Prey emigrating from patch i at time t | - | |
| CVprey | Prey (G or S) within-season temporal variance (coefficient of variation) | Equation (10) | |
| Predator | Pit | Predator population density in patch i at time t | Equations (5) and (6) |
| c | Predator conversion efficiency of prey | 0.6 | |
| a | Predator attack rate | 0.6 | |
| mP | Predator mortality rate | 0.1 | |
| dP | Proportion of P dispersing to new patch | Equations (7) and (8) | |
| IPit | Sum of predators immigrating to patch i at time t | - | |
| EPit | Predators emigrating from patch i at time t | - |
References
- Fahrig, L. Effects of habitat fragmentation on biodiversity. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 487–515. [Google Scholar] [CrossRef]
- Dainese, M.; Martin, E.A.; Aizen, M.A.; Albrecht, M.; Bartomeus, I.; Bommarco, R.; Carvalheiro, L.G.; Chaplin-Kramer, R.; Gagic, V.; Garibaldi, L.A.; et al. A global synthesis reveals biodiversity-mediated benefits for crop production. Sci. Adv. 2019, 5, eaax0121. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.M.; Lonsdorf, E.; Neel, M.C.; Williams, N.M.; Ricketts, T.H.; Winfree, R.; Bommarco, R.; Brittain, C.; Burley, A.L.; Cariveau, D.; et al. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol. Lett. 2013, 16, 584–599. [Google Scholar] [CrossRef]
- Chaplin-Kramer, R.; O’Rourke, M.E.; Blitzer, E.J.; Kremen, C. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecol. Lett. 2011, 14, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Ricketts, T.H.; Regetz, J.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Bogdanski, A.; Gemmill-Herren, B.; Greenleaf, S.S.; Klein, A.M.; Mayfield, M.M.; et al. Landscape effects on crop pollination services: Are there general patterns? Ecol. Lett. 2018, 499–515. [Google Scholar] [CrossRef]
- Vasseur, C.; Joannon, A.; Aviron, S.; Burel, F.; Meynard, J.-M.; Baudry, J. The cropping systems mosaic: How does the hidden heterogeneity of agricultural landscapes drive arthropod populations? Agric. Ecosyst. Environ. 2013, 166, 3–14. [Google Scholar] [CrossRef]
- Cohen, A.L.; Crowder, D.W. The impacts of spatial and temporal complexity across landscapes on biological control: A review. Curr. Opin. Insect Sci. 2017, 20, 13–18. [Google Scholar] [CrossRef]
- Schellhorn, N.A.; Gagic, V.; Bommarco, R. Time will tell: Resource continuity bolsters ecosystem services. Trends Ecol. Evol. 2015, 30, 524–530. [Google Scholar] [CrossRef]
- Iuliano, B.; Gratton, C. Temporal resource (dis)continuity for conservation biological control: From field to landscape scales. Front. Sustain. Food Syst. 2020, 4. [Google Scholar] [CrossRef]
- Dunning, J.B.; Danielson, B.J.; Pulliam, H.R. Ecological processes that affect populations in complex landscapes. Oikos 1992, 65, 169–175. [Google Scholar] [CrossRef]
- Mallinger, R.E.; Gibbs, J.; Gratton, C. Diverse landscapes have a higher abundance and species richness of spring wild bees by providing complementary floral resources over bees’ foraging periods. Landsc. Ecol. 2016, 31, 1523–1535. [Google Scholar] [CrossRef]
- Rundlöf, M.; Persson, A.S.; Smith, H.G.; Bommarco, R. Late-season mass-flowering red clover increases bumble bee queen and male densities. Biol. Conserv. 2014, 172, 138–145. [Google Scholar] [CrossRef]
- Mandelik, Y.; Winfree, R.; Neeson, T.; Kremen, C. Complementary habitat use by wild bees in agro-natural landscapes. Ecol. Appl. 2012, 22, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Levins, R. Some Demographic and Genetic Consequences of Environmental Heterogeneity for Biological Control. Bull. Entomol. Soc. Am. 1969, 15, 237–240. [Google Scholar] [CrossRef]
- Ives, A.R.; Settle, W.H. Metapopulation dynamics and pest control in agricultural systems. Am. Nat. 1997, 149, 220–246. [Google Scholar] [CrossRef]
- Bianchi, F.J.J.A.; Van der Werf, W. Model evaluation of the function of prey in non-crop habitats for biological control by ladybeetles in agricultural landscapes. Ecol. Model. 2004, 171, 177–193. [Google Scholar] [CrossRef]
- Le Gal, A.; Robert, C.; Accatino, F.; Claessen, D.; Lecomte, J. Modelling the interactions between landscape structure and spatio-temporal dynamics of pest natural enemies: Implications for conservation biological control. Ecol. Model. 2020, 420, 108912. [Google Scholar] [CrossRef]
- Taylor, A.D. Metapopulations, Dispersal, and Predator-Prey Dynamics: An Overview. Ecology 1990, 71, 429–433. [Google Scholar] [CrossRef]
- Holling, C.S. Some characteristics of simple types of predation and parasitism. Can. Entomol. 1959, 91, 385–398. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Paradox of Enrichment: Destabilization of Exploitation Ecosystems in Ecological Time. Science 1971, 171, 385–387. [Google Scholar] [CrossRef]
- Jensen, C.X.J.; Ginzburg, L.R. Paradoxes or theoretical failures? The jury is still out. Ecol. Model. 2005, 188, 3–14. [Google Scholar] [CrossRef]
- Settle, W.H.; Ariawan, H.; Astuti, E.T.; Cahyana, W.; Hakim, A.L.; Hindayana, D.; Lestari, A.S. Managing tropical rice pests through conservation of generalist natural enemies and alternative prey. Ecology 1996, 77, 1975–1988. [Google Scholar] [CrossRef]
- Holt, R.D. Population dynamics in two-patch environments: Some anomalous consequences of an optimal habitat distribution. Theor. Popul. Biol. 1985, 28, 181–208. [Google Scholar] [CrossRef]
- Harwood, J.D.; Obrycki, J.J. The role of alternative prey in sustaining predator populations. In Proceedings of the Second International Symposium on Biological Control of Arthropods, Davos, Switzerland, 12–16 September 2005; pp. 453–462. [Google Scholar]
- Harvey, J.A.; Heinen, R.; Armbrecht, I.; Basset, Y.; Baxter-Gilbert, J.H.; Bezemer, T.M.; Böhm, M.; Bommarco, R.; Borges, P.A.V.; Cardoso, P.; et al. International scientists formulate a roadmap for insect conservation and recovery. Nat. Ecol. Evol. 2020, 4, 174–176. [Google Scholar] [CrossRef]
- Bercovitch, F.B. Conservation conundrum: Endangered predators eating endangered prey. Afr. J. Ecol. 2018, 56, 434–435. [Google Scholar] [CrossRef]
- Roemer, G.W.; Wayne, R.K. Conservation in Conflict: The Tale of Two Endangered Species. Conserv. Biol. 2003, 17, 1251–1260. [Google Scholar] [CrossRef]
- Vandermeer, J.; Armbrecht, I.; de la Mora, A.; Ennis, K.K.; Fitch, G.; Gonthier, D.J.; Hajian-Forooshani, Z.; Hsieh, H.-Y.; Iverson, A.; Jackson, D.; et al. The Community Ecology of Herbivore Regulation in an Agroecosystem: Lessons from Complex Systems. BioScience 2019, 69, 974–996. [Google Scholar] [CrossRef]
- Snyder, W.E. Give predators a complement: Conserving natural enemy biodiversity to improve biocontrol. Biol. Control 2019, 135, 73–82. [Google Scholar] [CrossRef]
- Redlich, S.; Martin, E.A.; Steffan-Dewenter, I. Landscape-level crop diversity benefits biological pest control. J. Appl. Ecol. 2018, 55, 2419–2428. [Google Scholar] [CrossRef]
- Bosem-Baillod, A.; Tscharntke, T.; Clough, Y.; Batáry, P. Landscape-scale interactions of spatial and temporal cropland heterogeneity drive biological control of cereal aphids. J. Appl. Ecol. 2017, 54, 1804–1813. [Google Scholar] [CrossRef]
- Sirami, C.; Gross, N.; Baillod, A.B.; Bertrand, C.; Carrié, R.; Hass, A.; Henckel, L.; Miguet, P.; Vuillot, C.; Alignier, A.; et al. Increasing crop heterogeneity enhances multitrophic diversity across agricultural regions. Proc. Natl. Acad. Sci. USA 2019, 116, 16442–16447. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spiesman, B.; Iuliano, B.; Gratton, C. Temporal Resource Continuity Increases Predator Abundance in a Metapopulation Model: Insights for Conservation and Biocontrol. Land 2020, 9, 479. https://doi.org/10.3390/land9120479
Spiesman B, Iuliano B, Gratton C. Temporal Resource Continuity Increases Predator Abundance in a Metapopulation Model: Insights for Conservation and Biocontrol. Land. 2020; 9(12):479. https://doi.org/10.3390/land9120479
Chicago/Turabian StyleSpiesman, Brian, Benjamin Iuliano, and Claudio Gratton. 2020. "Temporal Resource Continuity Increases Predator Abundance in a Metapopulation Model: Insights for Conservation and Biocontrol" Land 9, no. 12: 479. https://doi.org/10.3390/land9120479
APA StyleSpiesman, B., Iuliano, B., & Gratton, C. (2020). Temporal Resource Continuity Increases Predator Abundance in a Metapopulation Model: Insights for Conservation and Biocontrol. Land, 9(12), 479. https://doi.org/10.3390/land9120479





