Urban Avian Conservation Planning Using Species Functional Traits and Habitat Suitability Mapping
Abstract
1. Introduction
2. Materials and Methods
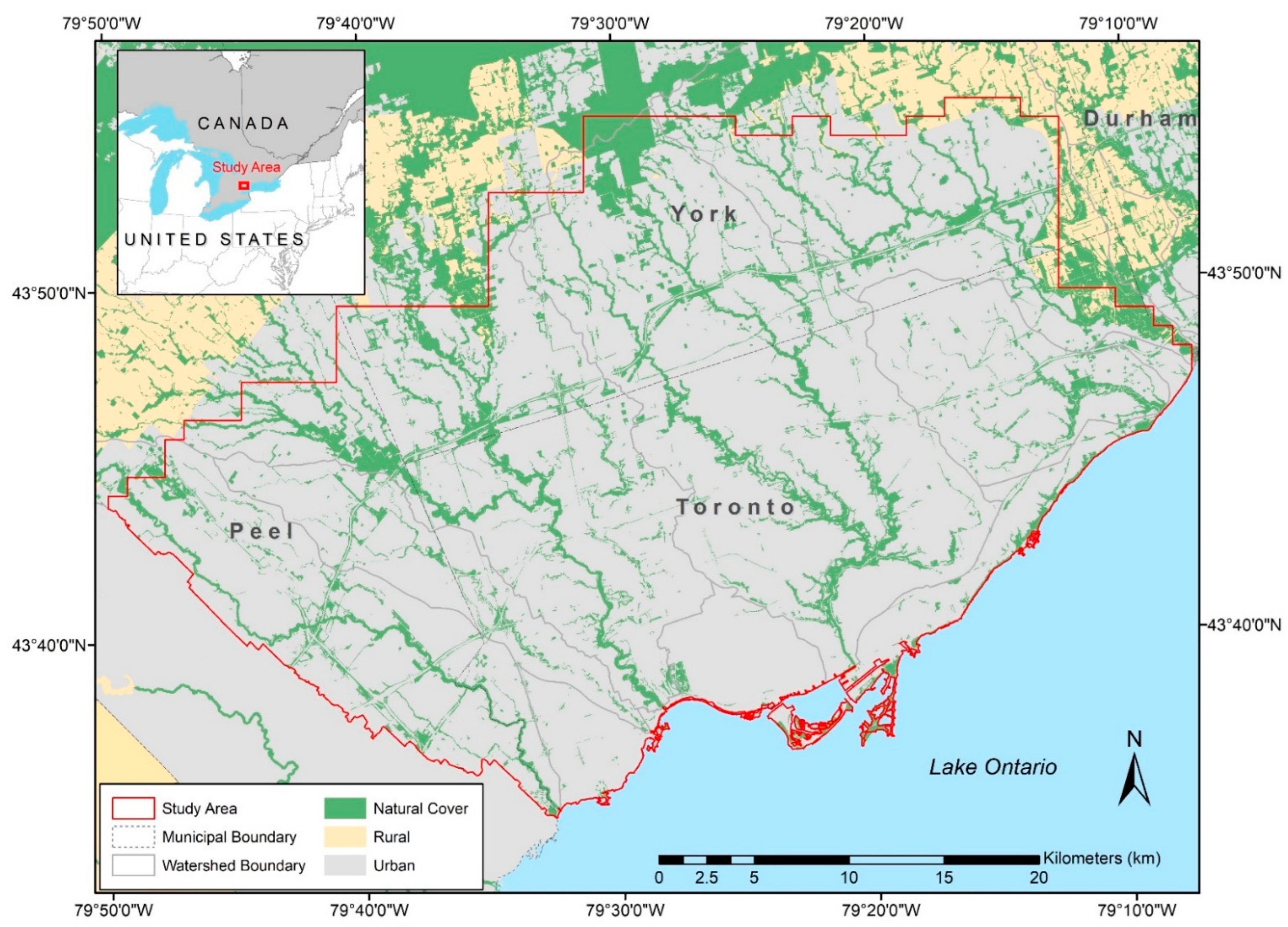
2.1. Study Area
2.2. Avian Species Data
2.3. Landscape Characteristics Data
2.3.1. Patch Quality
2.3.2. Habitat Connectivity
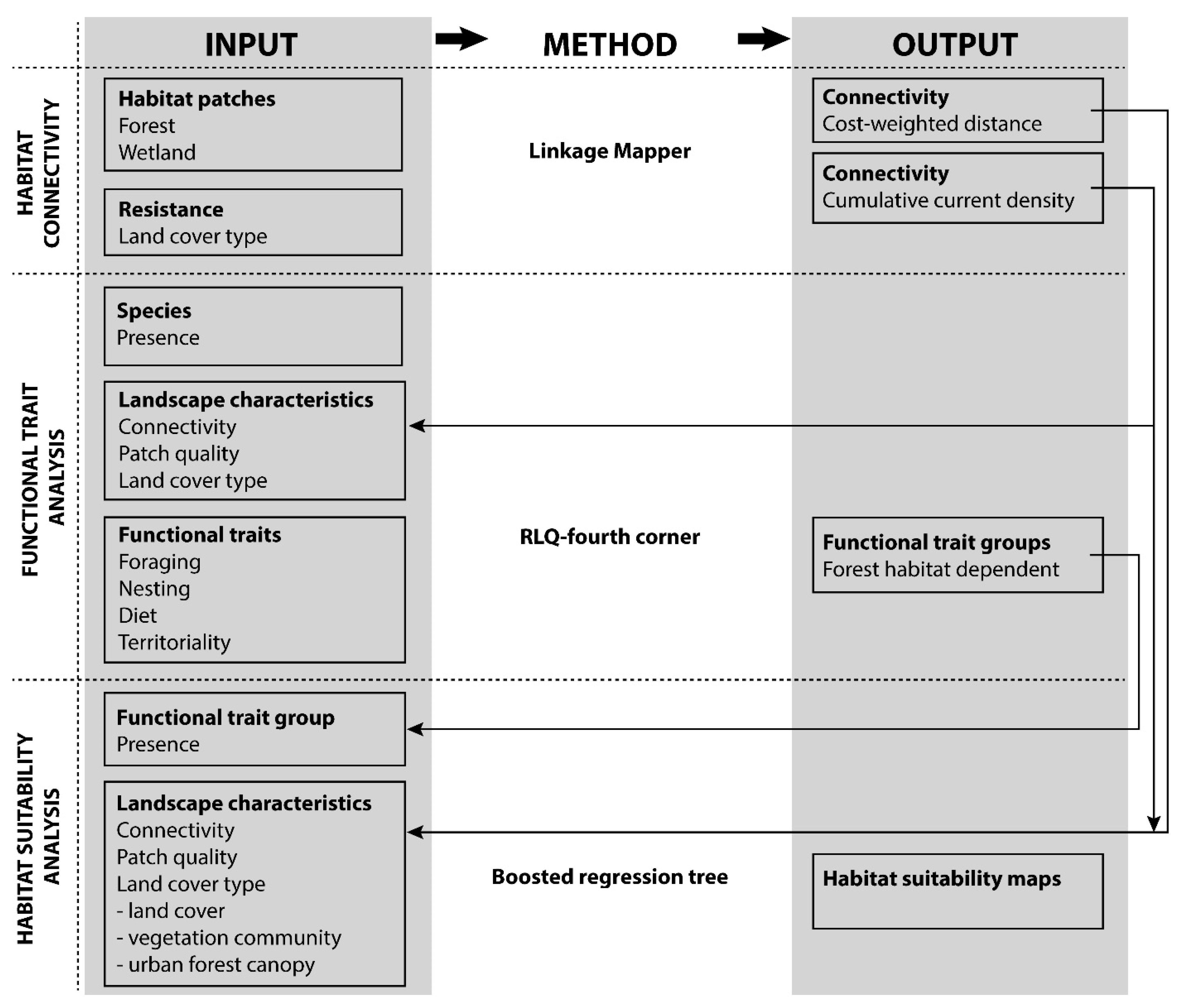
2.4. Methodological Approach
2.4.1. Functional Trait Analysis/RLQ-Fourth Corner
2.4.2. Habitat Suitability Analysis (HSA)
2.4.3. Combining Trait-Based and Habitat Suitability Analysis
3. Results
3.1. Functional Trait Analysis/RLQ-Fourth Corner
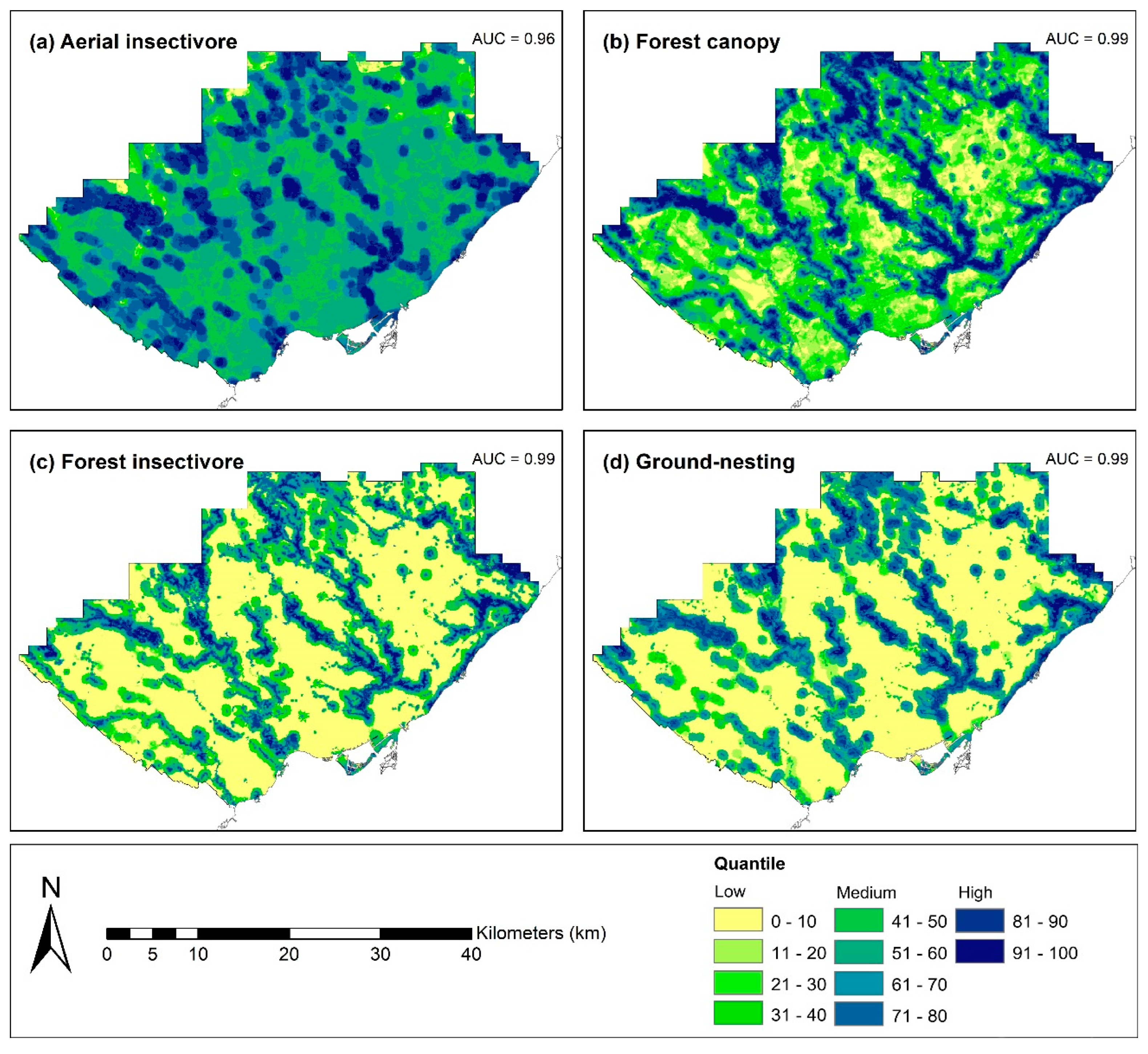
3.2. Habitat Suitability Analysis
4. Discussion
5. Limitations & Future Steps
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Z.; He, C.; Wu, J. The Relationship between Habitat Loss and Fragmentation during Urbanization: An Empirical Evaluation from 16 World Cities. PLoS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef]
- Scolozzi, R.; Geneletti, D. A Multi-Scale Qualitative Approach to Assess the Impact of Urbanization on Natural Habitats and Their Connectivity. Environ. Impact Assess. Rev. 2012, 36, 9–22. [Google Scholar] [CrossRef]
- Johnson, M.T.J.; Munshi-South, J. Evolution of Life in Urban Environments. Science 2017, 358, eaam8327. [Google Scholar] [CrossRef]
- Turrini, T.; Knop, E. A Landscape Ecology Approach Identifies Important Drivers of Urban Biodiversity. Glob. Chang. Biol. 2015, 21, 1652–1667. [Google Scholar] [CrossRef]
- UN-DESA. World Urbanization Prospects: The 2018 Revision (ST/ESA/SER.A/420); UN-DESA: New York, NY, USA, 2019. [Google Scholar]
- Xu, C.; Haase, D.; Pauleit, S. The Impact of Different Urban Dynamics on Green Space Availability: A Multiple Scenario Modeling Approach for the Region of Munich, Germany. Ecol. Indic. 2018, 93, 1–12. [Google Scholar] [CrossRef]
- Aronson, M.F.J.; Lepczyk, C.A.; Evans, K.L.; Goddard, M.A.; Lerman, S.B.; MacIvor, J.S.; Nilon, C.H.; Vargo, T. Biodiversity in the City: Key Challenges for Urban Green Space Management. Front. Ecol. Environ. 2017, 15, 189–196. [Google Scholar] [CrossRef]
- Kowarik, I. Novel Urban Ecosystems, Biodiversity, and Conservation. Environ. Pollut. 2011, 159, 1974–1983. [Google Scholar] [CrossRef]
- Filazzola, A.; Shrestha, N.; MacIvor, J.S. The Contribution of Constructed Green Infrastructure to Urban Biodiversity: A Synthesis and Meta-Analysis. J. Appl. Ecol. 2019, 56, 2131–2143. [Google Scholar] [CrossRef]
- Fernández-Juricic, E. Bird Community Composition Patterns in Urban Parks of Madrid: The Role of Age, Size and Isolation. Ecol. Res. 2000, 15, 373–383. [Google Scholar] [CrossRef]
- Almas, A.D.; Conway, T.M. The Role of Native Species in Urban Forest Planning and Practice: A Case Study of Carolinian Canada. Urban For. Urban Green. 2016, 17, 54–62. [Google Scholar] [CrossRef]
- Shoffner, A.; Wilson, A.M.; Tang, W.; Gagné, S.A. The Relative Effects of Forest Amount, Forest Configuration, and Urban Matrix Quality on Forest Breeding Birds. Sci. Rep. 2018, 8, 17140. [Google Scholar] [CrossRef] [PubMed]
- Mouillot, D.; Graham, N.A.J.; Villéger, S.; Mason, N.W.H.; Bellwood, D.R. A Functional Approach Reveals Community Responses to Disturbances. Trends Ecol. Evol. 2013, 28, 167–177. [Google Scholar] [CrossRef]
- Volaire, F.; Gleason, S.M.; Delzon, S. What Do You Mean “Functional” in Ecology? Patterns versus Processes. Ecol. Evol. 2020, 10, 11875–11885. [Google Scholar] [CrossRef]
- Croci, S.; Butet, A.; Clergeau, P. Does Urbanization Filter Birds on the Basis of Their Biological Traits? Condor 2008, 110, 223–240. [Google Scholar] [CrossRef]
- Lerman, S.B.; Nislow, K.H.; Nowak, D.J.; DeStefano, S.; King, D.I.; Jones-Farrand, D.T. Using Urban Forest Assessment Tools to Model Bird Habitat Potential. Landsc. Urban Plan. 2014, 122, 29–40. [Google Scholar] [CrossRef]
- Oliveira, J.M.; Segurado, P.; Santos, J.M.; Teixeira, A.; Ferreira, M.T.; Cortes, R.V. Modelling Stream-Fish Functional Traits in Reference Conditions: Regional and Local Environmental Correlates. PLoS ONE 2012, 7, e45787. [Google Scholar] [CrossRef] [PubMed]
- Devictor, V.; Julliard, R.; Clavel, J.; Jiguet, F.; Lee, A.; Couvet, D. Functional Biotic Homogenization of Bird Communities in Disturbed Landscapes. Glob. Ecol. Biogeogr. 2008, 17, 252–261. [Google Scholar] [CrossRef]
- Dray, S.; Choler, P.; Dolédec, S.; Peres-Neto, P.R.; Thuiller, W.; Pavoine, S.; ter Braak, C.J.F. Combining the Fourth-Corner and the RLQ Methods for Assessing Trait Responses to Environmental Variation. Ecology 2014, 95, 14–21. [Google Scholar] [CrossRef]
- Cartwright, L.A.; Hayes, S.; Tozer, D.C.; Clayton, D.; Burns, M.A.; Lewis, D.; Gaetz, N.; Shrestha, N. Assessing Terrestrial Wildlife Populations in the Toronto and Region Area of Concern. J. Great Lakes Res. 2021, 47, 273–282. [Google Scholar] [CrossRef]
- Statistics Canada. Canada’s Large Urban Centres Continue to Grow and Spread. 2022. Available online: https://www150.statcan.gc.ca/n1/daily-quotidien/220209/dq220209b-eng.htm (accessed on 1 September 2022).
- Ontario Ministry of Municipal Affairs and Housing (OMMAH) Greenbelt Plan (2017). 2017. Available online: https://files.ontario.ca/greenbelt-plan-2017-en.pdf (accessed on 1 September 2022).
- Toronto and Region Conservation Authority. Toronto and Region Watersheds: Report Card 2018; Toronto and Region Conservation Authority: Downsview, ON, Canada, 2018. [Google Scholar]
- Toronto and Region Conservation Authority. Scoring and Ranking TRCA’s Vegetation Communities, Flora, and Fauna Species; Toronto and Region Conservation Authority: Downsview, ON, Canada, 2017. [Google Scholar]
- Cadman, M.D.; Sutherland, D.A.; Beck, G.G.; Lepage, D.; Couturier, A.R. Atlas of the Breeding Birds of Ontario, 2001–2005; Bird Studies Canada, Environment Canada, Ontario Field Ornithologists, Ontario Ministry of Natural Resources, and Ontario Nature: Toronto, ON, Canada, 2007. [Google Scholar]
- Lee, H.T.; Bakowsky, W.D.; Riley, J.; Bowles, J.; Puddister, M.; Uhlig, P.; McMurray, S. Ecological Land Classification for Southern Ontario: First Approximation and Its Application; Ontario Ministry of Natural Resources, Southcentral Science Section: North Bay, ON, Canada, 1998. [Google Scholar]
- City of Toronto Forest and Land Cover-City of Toronto Open Data Portal. 2018. Available online: https://open.toronto.ca/dataset/forest-and-land-cover/ (accessed on 14 September 2018).
- Timmins, T.; Sawka, M. 2021 York Region Canopy Cover Assessment Technical Report; Toronto and Region Conservation Authority: Vaughan, ON, Canada, 2022. [Google Scholar]
- Soverel, N.; Dranga, S.; Blackwell, B.A. An Assessment of Urban Tree Canopy Cover in Peel Region 2015; B.A. Blackwell & Associates Ltd.: North Vancouver, BC, Canada, 2017. [Google Scholar]
- ESRI. ArcGIS, Version 10.4.1; Environmental Systems Research Institute: Redlands, CA, USA, 2015.
- Tremblay, M.A.; St. Clair, C.C. Permeability of a Heterogeneous Urban Landscape to the Movements of Forest Songbirds. J. Appl. Ecol. 2011, 48, 679–688. [Google Scholar] [CrossRef]
- Grafius, D.R.; Corstanje, R.; Siriwardena, G.M.; Plummer, K.E.; Harris, J.A. A Bird’s Eye View: Using Circuit Theory to Study Urban Landscape Connectivity for Birds. Landsc. Ecol. 2017, 32, 1771–1787. [Google Scholar] [CrossRef]
- Marzluff, J.M.; DeLap, J.H.; Oleyar, M.D.; Whittaker, K.A.; Gardner, B. Breeding Dispersal by Birds in a Dynamic Urban Ecosystem. PLoS ONE 2016, 11, 1–20. [Google Scholar] [CrossRef]
- McRae, B.; Kavanagh, D. Linkage Mapper Connectivity Analysis Software; The Nature Conservancy: Seattle, WA, USA, 2011. [Google Scholar]
- DeGraaf, R.M.; Tilghman, N.G.; Anderson, S.H. Foraging Guilds of North American Birds. Environ. Manage. 1985, 9, 493–536. [Google Scholar] [CrossRef]
- Ferenc, M.; Sedláček, O.; Fuchs, R. How to Improve Urban Greenspace for Woodland Birds: Site and Local-Scale Determinants of Bird Species Richness. Urban Ecosyst. 2014, 17, 625–640. [Google Scholar] [CrossRef]
- Murtagh, F.; Legendre, P. Ward’s Hierarchical Agglomerative Clustering Method: Which Algorithms Implement Ward’s Criterion? J. Classif. 2014, 31, 274–295. [Google Scholar] [CrossRef]
- Dray, S.; Dufour, A.-B. The Ade4 Package: Implementing the Duality Diagram for Ecologists. J. Stat. Softw. 2007, 22, 1–20. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography. 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Hastie, T. A Working Guide to Boosted Regression Trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef]
- Greenwell, B.; Boehmke, B.; Cunningham, J.; GBM Developers. Gbm: Generalized Boosted Regression Models; R Package Version 2.1.8.1. 2020. Available online: https://CRAN.R-project.org/package=gbm (accessed on 1 September 2022).
- Hijmans, R.J.; Steven, P.; Leathwick, J.; Elith, J. Dismo: Species Distribution Modeling; R Package Version 1.3-9. 2017. Available online: https://CRAN.R-project.org/package=dismo (accessed on 1 September 2022).
- Hijmans, R.J. Raster: Geographic Data Analysis and Modeling; R Package Version 3.6-3. 2020. Available online: https://CRAN.R-project.org/package=raster (accessed on 1 September 2022).
- Møller, A.P.; Diaz, M.; Flensted-Jensen, E.; Grim, T.; Ibáñez-Álamo, J.D.; Jokimäki, J.; Mänd, R.; Markó, G.; Tryjanowski, P. High Urban Population Density of Birds Reflects Their Timing of Urbanization. Oecologia 2012, 170, 867–875. [Google Scholar] [CrossRef]
- Evens, R.; Beenaerts, N.; Neyens, T.; Witters, N.; Smeets, K.; Artois, T. Proximity of Breeding and Foraging Areas Affects Foraging Effort of a Crepuscular, Insectivorous Bird. Sci. Rep. 2018, 8, 3008. [Google Scholar] [CrossRef]
- Razeng, E.; Watson, D.M. Nutritional Composition of the Preferred Prey of Insectivorous Birds: Popularity Reflects Quality. J. Avian Biol. 2015, 46, 89–96. [Google Scholar] [CrossRef]
- Ares, A.; Neill, A.R.; Puettmann, K.J. Understory Abundance, Species Diversity and Functional Attribute Response to Thinning in Coniferous Stands. For. Ecol. Manage. 2010, 260, 1104–1113. [Google Scholar] [CrossRef]
- Whitaker, D.M.; Carroll, A.L.; Montevecchi, W.A. Elevated Numbers of Flying Insects and Insectivorous Birds in Riparian Buffer Strips. Can. J. Zool. 2000, 78, 740–747. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Foppen, R.P.B.; Van Turnhout, C.A.M.; De Kroon, H.; Jongejans, E. Declines in Insectivorous Birds Are Associated with High Neonicotinoid Concentrations. Nature 2014, 511, 341–343. [Google Scholar] [CrossRef] [PubMed]
- Environment Canada. How Much Habitat Is Enough? 3rd ed.; Environment Canada: Toronto, ON, Canada, 2013.
- Schueler, T.R. The Importance of Imperviousness. Watershed Prot. Tech. 1994, 1, 100–111. [Google Scholar]
- Miller, S.G.; Knight, R.L.; Miller, C.K. Influence of Recreational Trails on Breeding Bird Communities. Ecol. Appl. 1998, 8, 162–169. [Google Scholar] [CrossRef]
- Keyser, A.J.; Hill, G.E.; Soehren, E.C. Effects of Forest Fragment Size, Nest Density, and Proximity to Edge on the Risk of Predation to Ground-Nesting Passerine Birds. Conserv. Biol. 1998, 12, 986–994. [Google Scholar] [CrossRef]
- Lepczyk, C.A.; Aronson, M.F.J.; Evans, K.L.; Goddard, M.A.; Lerman, S.B.; Macivor, J.S. Biodiversity in the City: Fundamental Questions for Understanding the Ecology of Urban Green Spaces for Biodiversity Conservation. Bioscience 2017, 67, 799–807. [Google Scholar] [CrossRef]
- Thompson, B. Recreational Trails Reduce the Density of Ground-Dwelling Birds in Protected Areas. Environ. Manage. 2015, 55, 1181–1190. [Google Scholar] [CrossRef]
- Hazen, E.L.; Abrahms, B.; Brodie, S.; Carroll, G.; Welch, H.; Bograd, S.J. Where Did They Not Go? Considerations for Generating Pseudo-Absences for Telemetry-Based Habitat Models. Mov. Ecol. 2021, 9, 5. [Google Scholar] [CrossRef]
- Blair, R.R.B. Land Use and Avian Species Diversity along an Urban Gradient. Ecol. Appl. 1996, 6, 506–519. [Google Scholar] [CrossRef]
- Zurita, G.; Pe’er, G.; Bellocq, M.I.; Hansbauer, M.M. Edge Effects and Their Influence on Habitat Suitability Calculations: A Continuous Approach Applied to Birds of the Atlantic Forest. J. Appl. Ecol. 2012, 49, 503–512. [Google Scholar] [CrossRef]
- Possingham, H.; Ball, I.; Andelman, S. Mathematical Methods for Identifying Representative Reserve Network. In Quantitative Methods for Conservation Biology; Springer: New York, NY, USA, 2000; pp. 291–306. [Google Scholar]
- Dray, S.; Legendre, P. Testing the Species Traits Environment Relationships: The Fourth-Corner Problem Revisited. Ecology 2008, 89, 3400–3412. [Google Scholar] [CrossRef] [PubMed]
| Functional Trait Group | Variable | Relative Influence |
|---|---|---|
| Aerial insectivore | LC_marsh | 39.7 |
| LC_deciduous | 17.4 | |
| C_local_forest | 5.5 | |
| LC_swamp | 5.2 | |
| LC_beach | 4.7 | |
| LC_residential_lowmed | 4.4 | |
| LC_woodland | 3.1 | |
| C_regional_forest | 2.3 | |
| LC_savannah | 2.1 | |
| E_meadow | 2.0 | |
| LC_thicket | 1.7 | |
| C_local_wetland | 1.6 | |
| P_wetland | 1.2 | |
| LC_commercial | 1.1 | |
| P_forest | 1.0 | |
| Forest canopy | C_local_forest | 44.1 |
| LC_deciduous | 41.6 | |
| LC_marsh | 2.6 | |
| LC_residential_lowmed | 1.9 | |
| P_forest | 1.3 | |
| LC_urbanforest | 1.0 | |
| Forest insectivore | C_ local_forest | 59.6 |
| LC_deciduous | 18.6 | |
| LC_swamp | 4.7 | |
| C_regional_forest | 3.7 | |
| LC_marsh | 3.0 | |
| LC_residential_lowmed | 2.3 | |
| P_forest | 2.3 | |
| LC_mixedforest | 1.9 | |
| Ground-nesting | LC_mixedforest | 35.9 |
| P_forest | 26.9 | |
| C_local_forest | 22.5 | |
| LC_urbanforest | 4.9 | |
| C_regional_forest | 3.5 | |
| LC_deciduous | 2.5 | |
| LC_swamp | 1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chin, A.T.M.; Ruppert, J.L.W.; Shrestha, N.; Fortin, M.-J. Urban Avian Conservation Planning Using Species Functional Traits and Habitat Suitability Mapping. Land 2022, 11, 1831. https://doi.org/10.3390/land11101831
Chin ATM, Ruppert JLW, Shrestha N, Fortin M-J. Urban Avian Conservation Planning Using Species Functional Traits and Habitat Suitability Mapping. Land. 2022; 11(10):1831. https://doi.org/10.3390/land11101831
Chicago/Turabian StyleChin, Andrew Tim Man, Jonathan Leo William Ruppert, Namrata Shrestha, and Marie-Josée Fortin. 2022. "Urban Avian Conservation Planning Using Species Functional Traits and Habitat Suitability Mapping" Land 11, no. 10: 1831. https://doi.org/10.3390/land11101831
APA StyleChin, A. T. M., Ruppert, J. L. W., Shrestha, N., & Fortin, M.-J. (2022). Urban Avian Conservation Planning Using Species Functional Traits and Habitat Suitability Mapping. Land, 11(10), 1831. https://doi.org/10.3390/land11101831






