Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode
Abstract
:1. Introduction
2. Materials and Methods
2.1. Manufacture of Pyrrhotite-Coated Graphite Cathode
2.2. Experimental System and Pretreatment of Materials
2.3. MFCs Start-Up and Operation
2.4. Degradation Characterization of Cr (VI) and Cathode Products Analysis
3. Results and Discussion
3.1. The Electricity Production Performance of MFCs
3.2. Enhanced Cr (VI) Removal Efficiency by Pyrrhotite-Coated Cathode
3.3. Effect of Initial Cr (VI) Concentration for Cr (VI) Removal
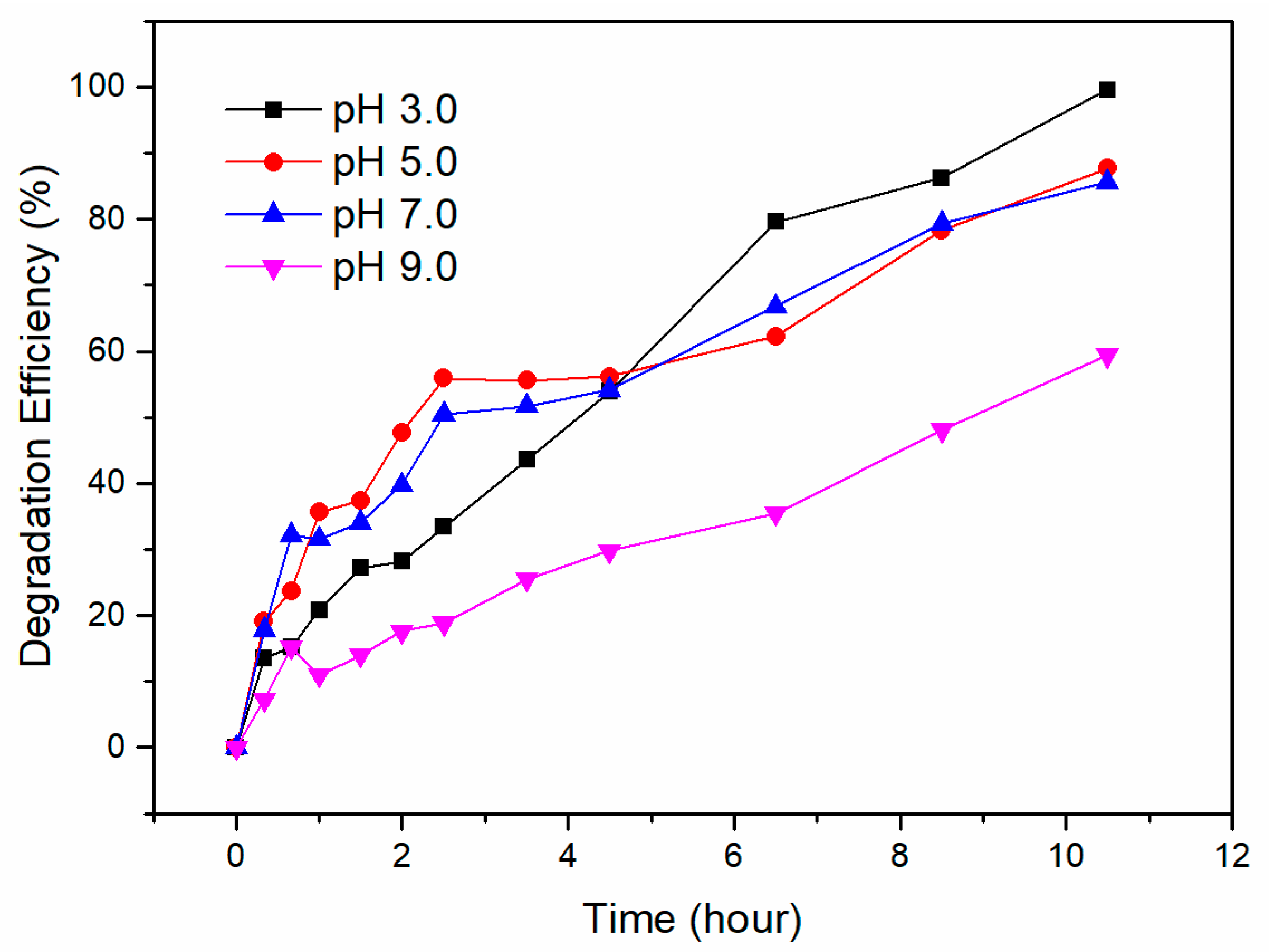
3.4. Removal Efficiencies of Cr (VI) under Different pH Values
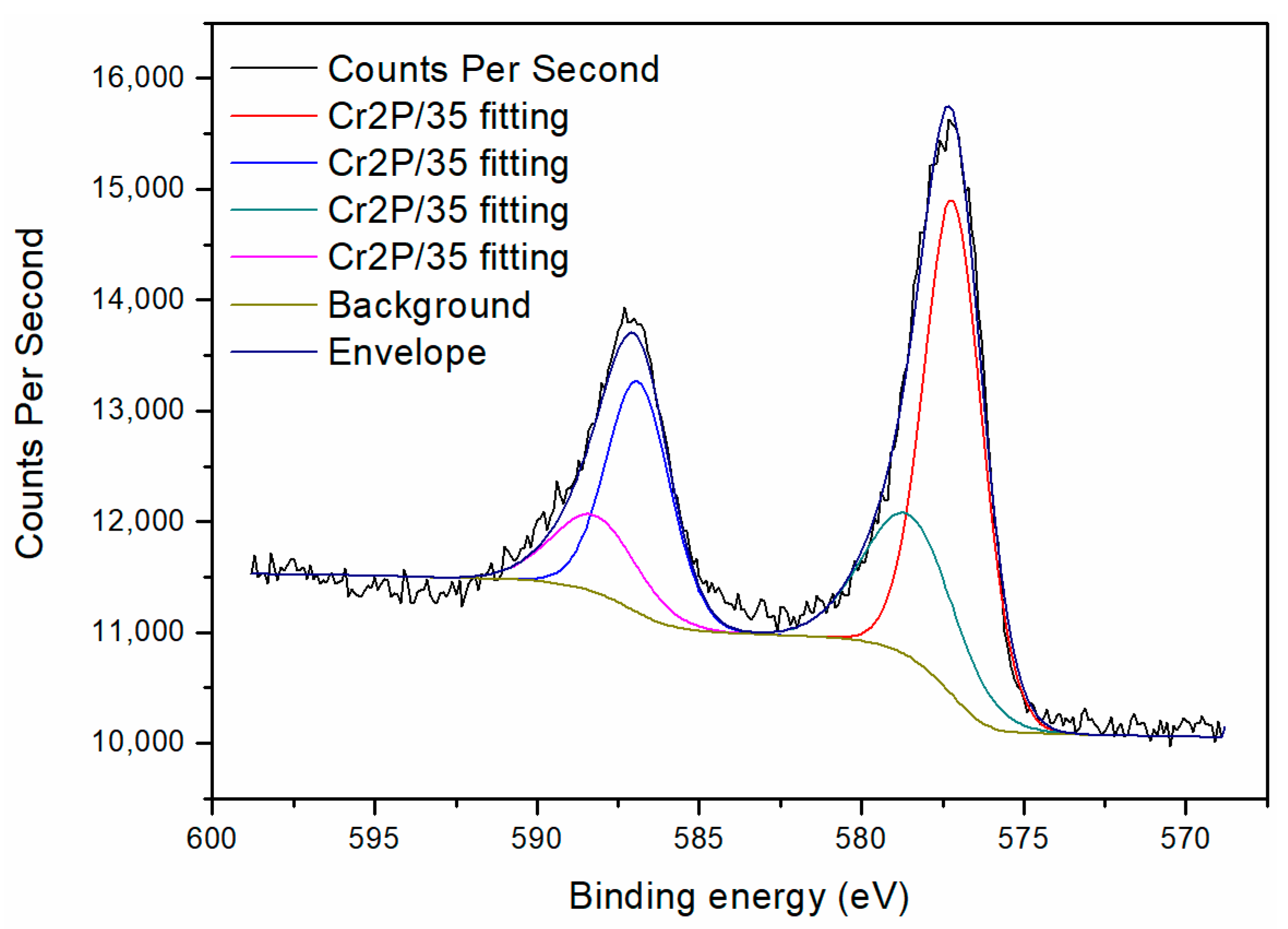
3.5. XPS Analysis of Reduced Production of Cr (VI) on Cathode
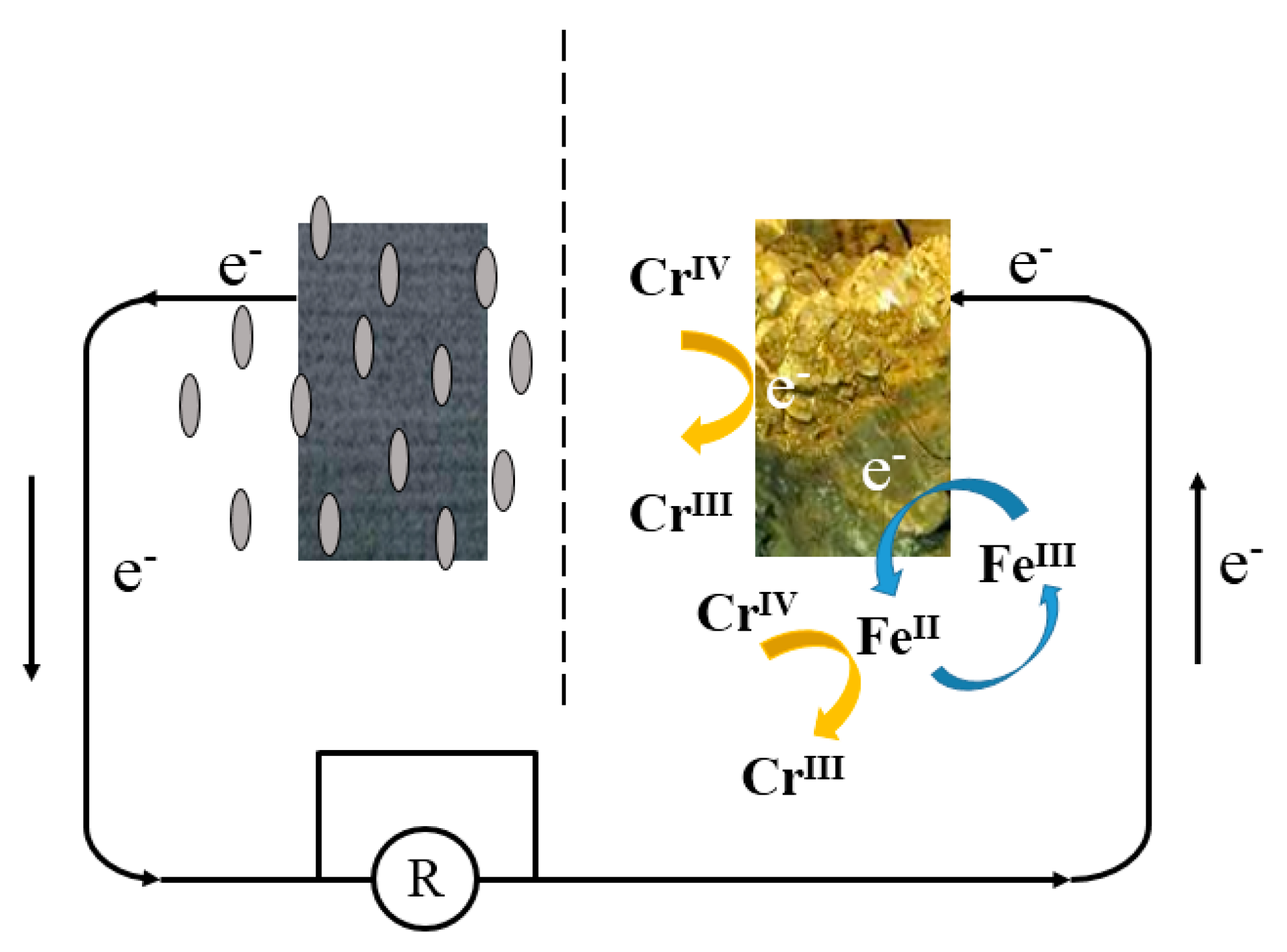
3.6. Mechanisms of Enhanced Cr (VI) Removal by Pyrrhotite-Cathode MFC
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Qiu, B.; Xu, C.; Sun, D.; Wei, H.; Zhang, X.; Guo, J.; Wang, Q.; Rutman, D.; Guo, Z.; Wei, S. Polyaniline coating on carbon fiber fabrics for improved hexavalent chromium removal. RSC Adv. 2014, 4, 29855–29865. [Google Scholar] [CrossRef]
- Legrand, L.; El Figuigui, A.; Mercier, F.; Chausse, A. Reduction of aqueous chromate by Fe (II)/Fe (III) carbonate green rust: Kinetic and mechanistic studies. Environ. Sci. Technol. 2004, 38, 4587–4595. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.J.; Wang, S.L.; Huang, P.M.; Tzou, Y.M.; Liu, J.C.; Chen, C.C.; Chen, J.H.; Lin, C. Chromate reduction by zero-valent Al metal as catalyzed by polyoxometalate. Water Res. 2009, 43, 5015–5022. [Google Scholar] [CrossRef] [PubMed]
- Kimbrough, D.E.; Cohen, Y.; Winer, A.M.; Creelman, L.; Mabuni, C. A critical assessment of chromium in the environment. Crit. Rev. Environ. Sci. Technol. 1999, 29, 1–46. [Google Scholar] [CrossRef]
- Wang, C.C.; Du, X.D.; Li, J.; Guo, X.X.; Wang, P.; Zhang, J. Photocatalytic Cr(VI) reduction in metal-organic frameworks: A mini-review. Appl. Catal. B 2016, 193, 198–216. [Google Scholar] [CrossRef]
- Kongsricharoern, N.; Polprasert, C. Chromium removal by a bipolar electro-chemical precipitation process. Water Sci. Technol. 1996, 34, 109–116. [Google Scholar]
- Rengaraj, S.; Joo, C.K.; Kim, Y.; Yi, J. Kinetics of removal of chromium from water and electronic process wastewater by ion exchange resins: 1200H, 1500H and IRN97H. J. Hazard. Mater. 2003, 102, 257–275. [Google Scholar] [CrossRef]
- Aravindhan, R.; Madhan, B.; Rao, J.R.; Nair, B.U.; Ramasami, T. Bioaccumulation of chromium from tannery wastewater: An approach for chrome recovery and reuse. Environ. Sci. Technol. 2003, 38, 300–306. [Google Scholar] [CrossRef]
- Xiao, D.; Dai, K.; Qu, Y.; Yin, Y.; Chen, H. Hydrothermal synthesis of α-Fe2O3/g-C3N4 composite and its efficient photocatalytic reduction of Cr(VI) under visible light. Appl. Surf. Sci. 2015, 358, 181–187. [Google Scholar] [CrossRef]
- Tao, H.C.; Liang, M.; Li, W.; Zhang, L.J.; Ni, J.R.; Wu, W.M. Removal of copper from aqueous solution by electrodeposition in cathode chamber of microbial fuel cell. J. Hazard. Mater. 2011, 189, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.; Cui, Y.F. Recovery of silver from wastewater coupled with power generation using a microbial fuel cell. Bioresour. Technol. 2012, 107, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.J.; Lim, B.; Choi, C. Removal of Hg2+ as an electron acceptor coupled with power generation using a microbial fuel cell. Bioresour. Technol. 2011, 102, 6304–6307. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, X.; Lei, L. Electricity production during the treatment of real electroplating wastewater containing Cr6+ using microbial fuel cell. Process Biochem. 2008, 43, 1352–1358. [Google Scholar] [CrossRef]
- Wang, G.; Huang, L.; Zhang, Y. Cathodic reduction of hexavalent chromium [Cr(VI)] coupled with electricity generation in microbial fuel cells. Biotechnol. Lett. 2008, 30, 1959–1966. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yuan, Y.; Li, F.B.; Feng, C.H. In-situ Cr(VI) reduction with electrogenerated hydrogen peroxide driven by iron-reducing bacteria. Bioresour. Technol. 2011, 102, 2468–2473. [Google Scholar] [CrossRef] [PubMed]
- Tandukar, M.; Huber, S.J.; Onodera, T.; Pavlostathis, S.G. Pavlostathis, Biological chromium(VI) reduction in the cathode of amicrobial fuel cell. Environ. Sci. Technol. 2009, 43, 8159–8165. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, J.; Quan, X.; Yang, F. Enhancement of hexavalent chromium reduction and electricity production from a biocathode microbial fuel cell. Bioprocess Biosyst. Eng. 2010, 33, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Xafenias, N.; Zhang, Y.; Banks, C.J. Enhanced performance of hexavalent chromium reducing cathodes in the presence of Shewanella oneidensis MR-1 and lactate. Environ. Sci. Technol. 2013, 47, 4512–4520. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chai, X.; Cheng, S.; Chen, G. Evaluation of carbonbased materials in tubular biocathode microbial fuel cells in terms of hexavalent chromium reduction and electricity generation. Chem. Eng. J. 2011, 166, 652–661. [Google Scholar] [CrossRef]
- Huang, L.; Chai, X.; Chen, G.; Logan, B.E. Effect of set potential on hexavalent chromium reduction and electricity generation from biocathode microbial fuel cells. Environ. Sci. Technol. 2011, 45, 5025–5031. [Google Scholar] [CrossRef] [PubMed]
- Roche, I.; Katuri, K.; Scott, K. A microbial fuel cell using manganese oxide oxygen reduction catalysts. J. Appl. Electrochem. 2010, 40, 13–21. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, C.; Zhuang, L.; Li, W.; Zhou, S.; Zhang, J. Manganese dioxide as an alternative cathodic catalyst to platinum in microbial fuel cells. Biosens. Bioelectron. 2009, 24, 2825–2829. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hu, B.; Suib, S.; Lei, Y.; Li, B. Manganese dioxide as a new cathode catalyst in microbial fuel cells. J. Power Sources 2010, 195, 2586–2591. [Google Scholar] [CrossRef]
- Morris, J.M.; Jin, S.; Wang, J.; Zhu, C.; Urynowicz, M.A. Lead dioxide as an alternative catalyst to platinum in microbial fuel cells. Electrochem. Commun. 2007, 9, 1730–1734. [Google Scholar] [CrossRef]
- Ren, G.; Ding, H.; Li, Y.; Lu, A. Natural Hematite as a Low-Cost and Earth-Abundant Cathode Material for Performance Improvement of Microbial Fuel Cells. Catalysts 2016, 6, 157. [Google Scholar] [CrossRef]
- Lu, A.; Li, Y.; Jin, S.; Ding, H.; Zeng, C.; Wang, X.; Wang, C. Microbial fuel cell equipped with a photocatalytic rutile-coated cathode. Energy Fuels 2009, 24, 1184–1190. [Google Scholar] [CrossRef]
- Li, Y.; Lu, A.; Ding, H.; Wang, X.; Wang, C.; Zeng, C.; Yan, Y. Microbial fuel cells using natural pyrrhotite as the cathodic heterogeneous Fentoncatalyst towards the degradation of biorefractory organics in landfill leachate. Electrochem. Commun. 2010, 12, 944–947. [Google Scholar] [CrossRef]
- Li, Y.; Lu, A.; Ding, H.; Jin, S.; Yan, Y.; Wang, C.; Zeng, C.; Wang, X. Cr(VI) reduction at rutile-catalyzed cathode in microbial fuel cells. Electrochem. Commun. 2009, 11, 1496–1499. [Google Scholar] [CrossRef]
- Demoisson, F.; Mullet, M.; Humbert, B. Pyrite Oxidation by Hexavalent Chromium: Investigation of the Chemical Processes by Monitoring of Aqueous Metal Species. Environ. Sci. Technol. 2005, 39, 8747–8752. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jung, P.K.; Moon, H.S.; Chon, C.M. Reduction of hexavalentchromiumby pyrite-rich andesite indifferent anionic solutions. Environ. Geol. 2002, 42, 642–648. [Google Scholar] [CrossRef]
- Lu, A.; Zhong, S.; Chen, J.; Shi, J.; Tang, J.; Lu, X. Removal of Cr(VI) and Cr(III) from aqueous solutions and industrial wastewaters by natural clino-pyrrhotite. Environ. Sci. Technol. 2006, 40, 3064–3069. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.L.; Han, H.L.; Shen, J.Q. Effects of cathodic electron acceptors and potassium ferricyanide concentrations on the performance of microbial fuel cell. Int. J. Hydrog. Energy 2012, 37, 12980–12986. [Google Scholar] [CrossRef]
- Kim, J.R.; Kim, J.Y.; Han, S.B.; Park, K.W.; Saratale, G.D.; Oh, S.E. Application of Co-naphthalocyanine (CoNPc) as alternative cathode catalyst and support structure for microbial fuel cells. Bioresour. Technol. 2011, 102, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Yun, Y.S.; Park, J.M. XAS and XPS studies on chromium-binding groups of biomaterial during Cr(VI) biosorption. J. Colloid Interface Sci. 2008, 317, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.R.; Yanful, E.K.; Pratt, A.R. Chemical states in XPS and Raman analysis during removal of Cr(VI) from contaminated water by mixed maghemite–magnetite nanoparticles. J. Hazard. Mater. 2012, 235, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Moulder, J.F. Handbook of X-ray Photoelectron Spectroscopy: A Reference Book of Standard Spectra for Identification and Interpretation of XPS Data; Perkin-Elmer Corporation, Physical Electronics Division: Eden Prairie, MN, USA, 1992; pp. 218–220. [Google Scholar]
- Vaughan, D.J.; Craig, J.R. Mineral Chemistry of Metal Sulphides; Cambridge University Press: Cambridge, UK, 1978; pp. 102–110. [Google Scholar]
- Nowok, J.; Stenberg, V.I. ESR study of clino-pyrrhotite iron vacancies and adsorption of CO and H2S. Appl. Surf. Sci. 1987, 29, 463–473. [Google Scholar] [CrossRef]
- Knipe, S.W.; Mycroft, J.R.; Pratt, A.R.; Nesbitt, H.W.; Bancroft, G.M. X-ray Photoelectron spectroscopic study of water adsorptiononiron sulphide minerals. Geochim. Cosmochim. Acta 1995, 59, 1079–1090. [Google Scholar] [CrossRef]
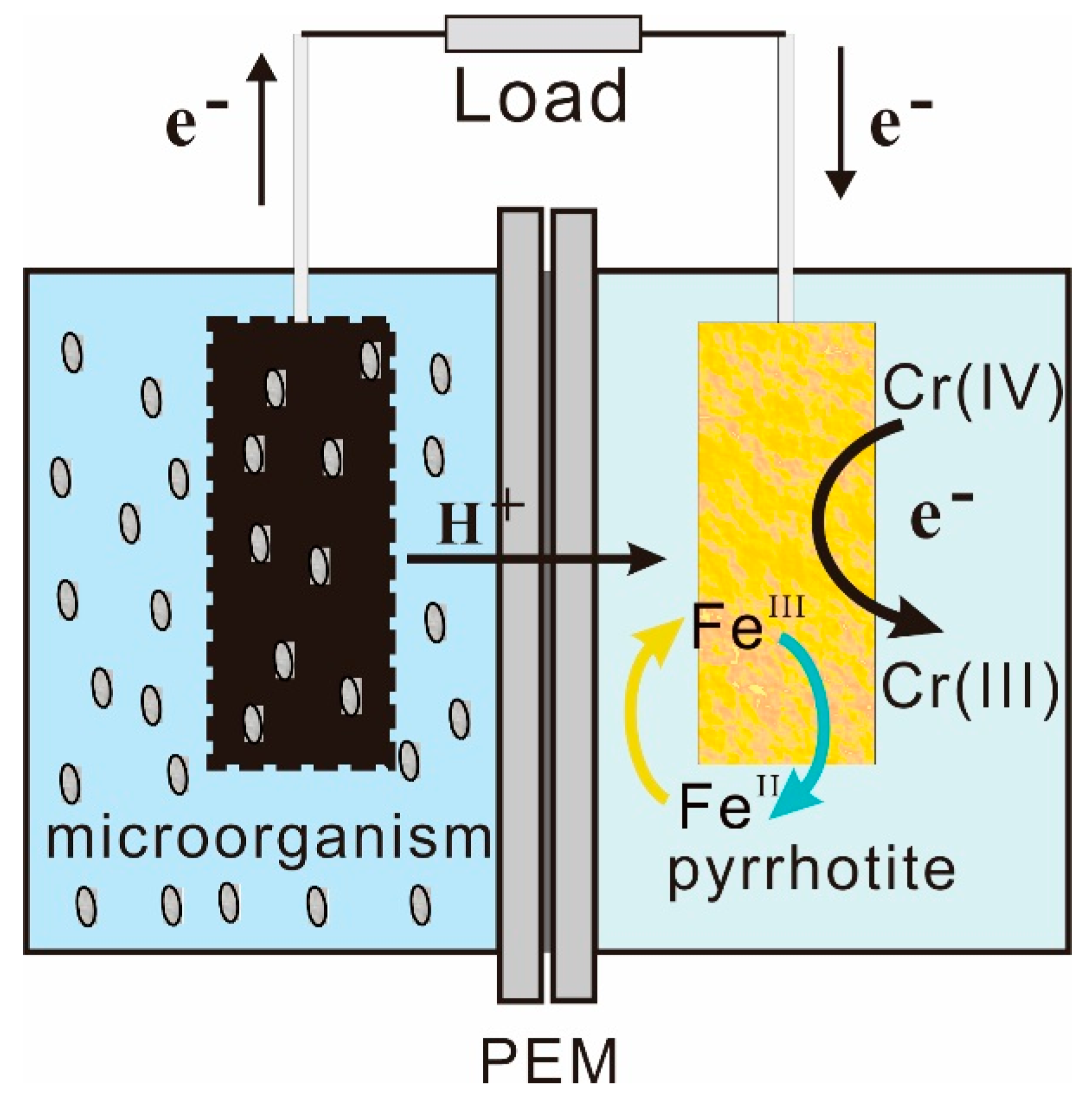
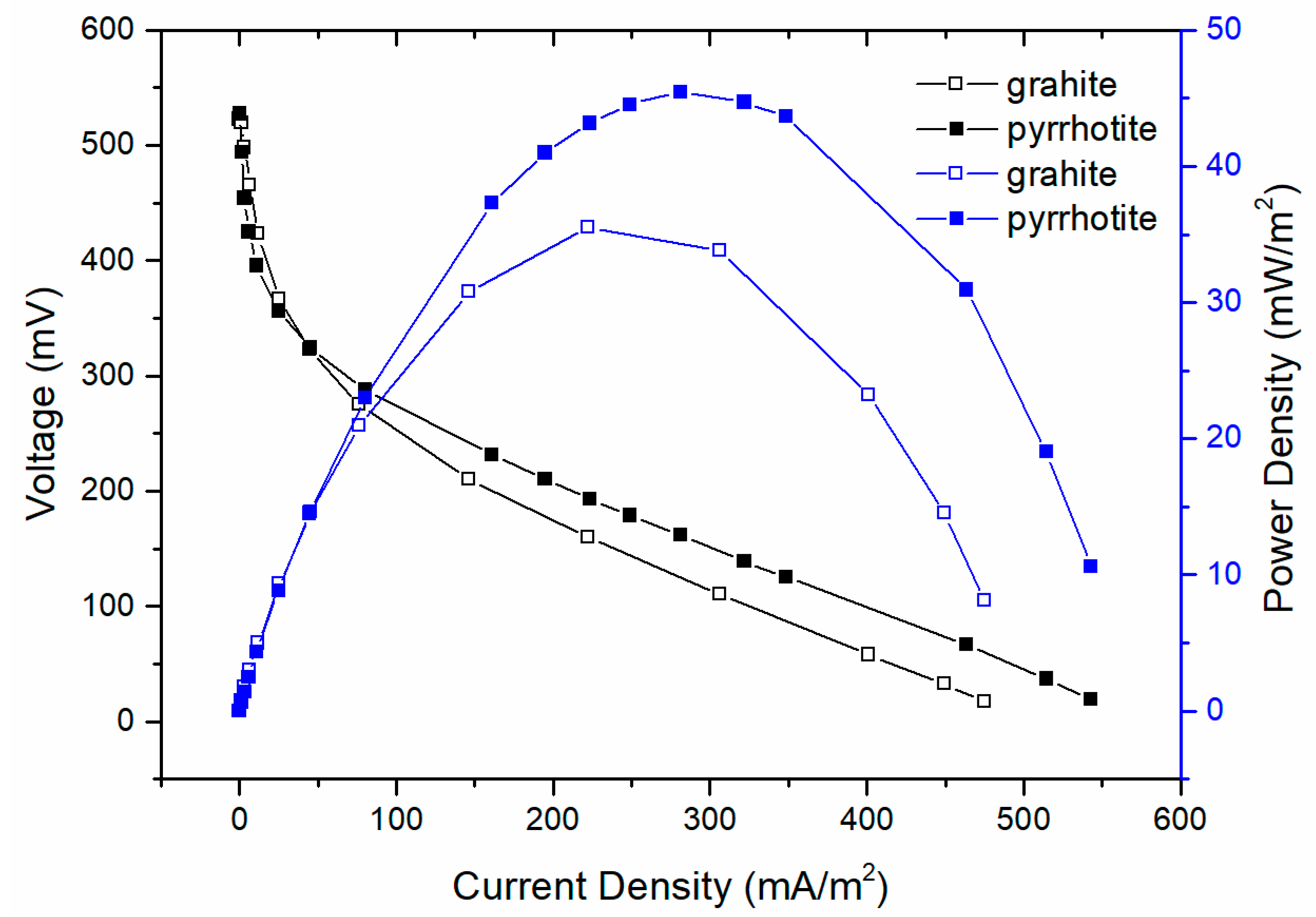
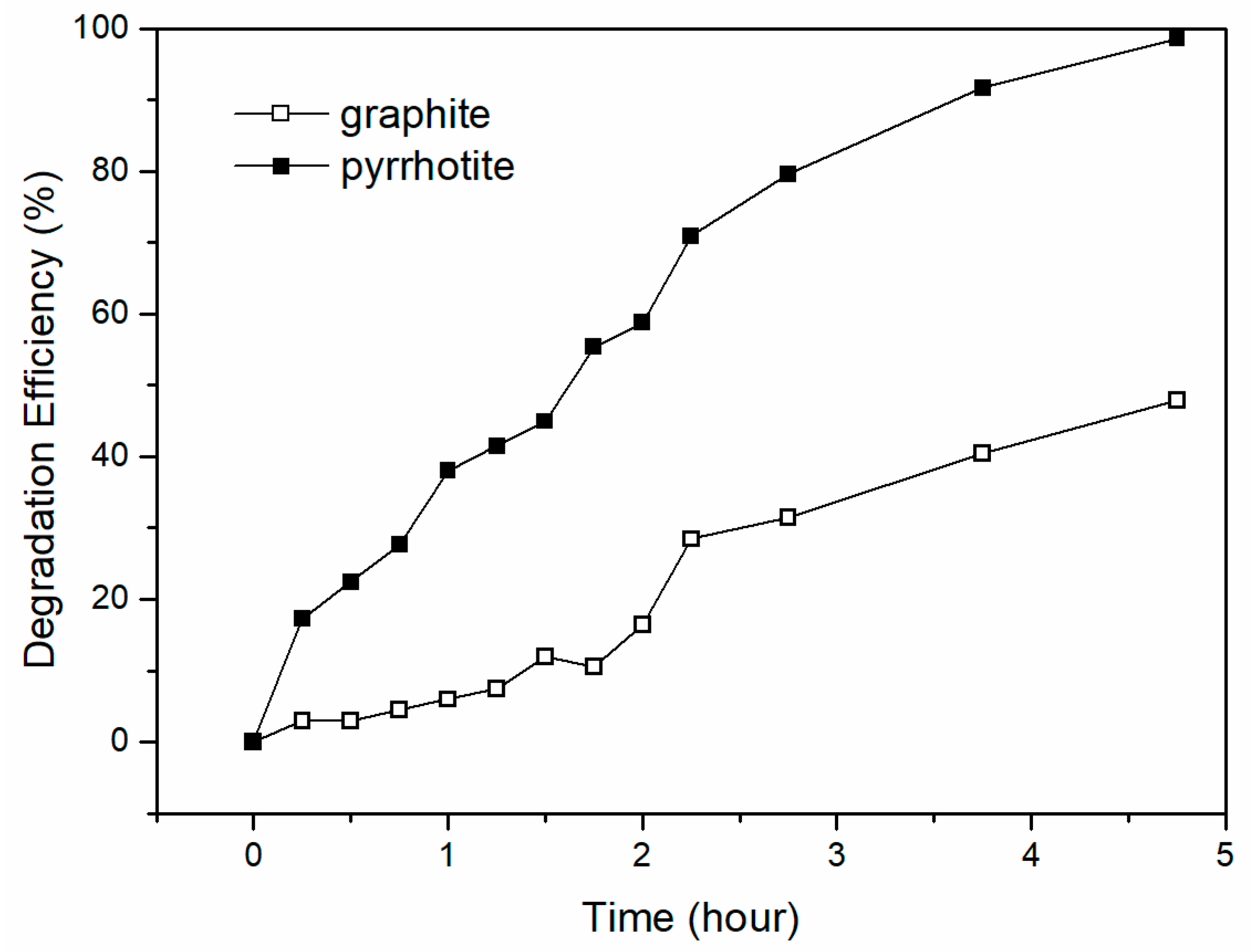
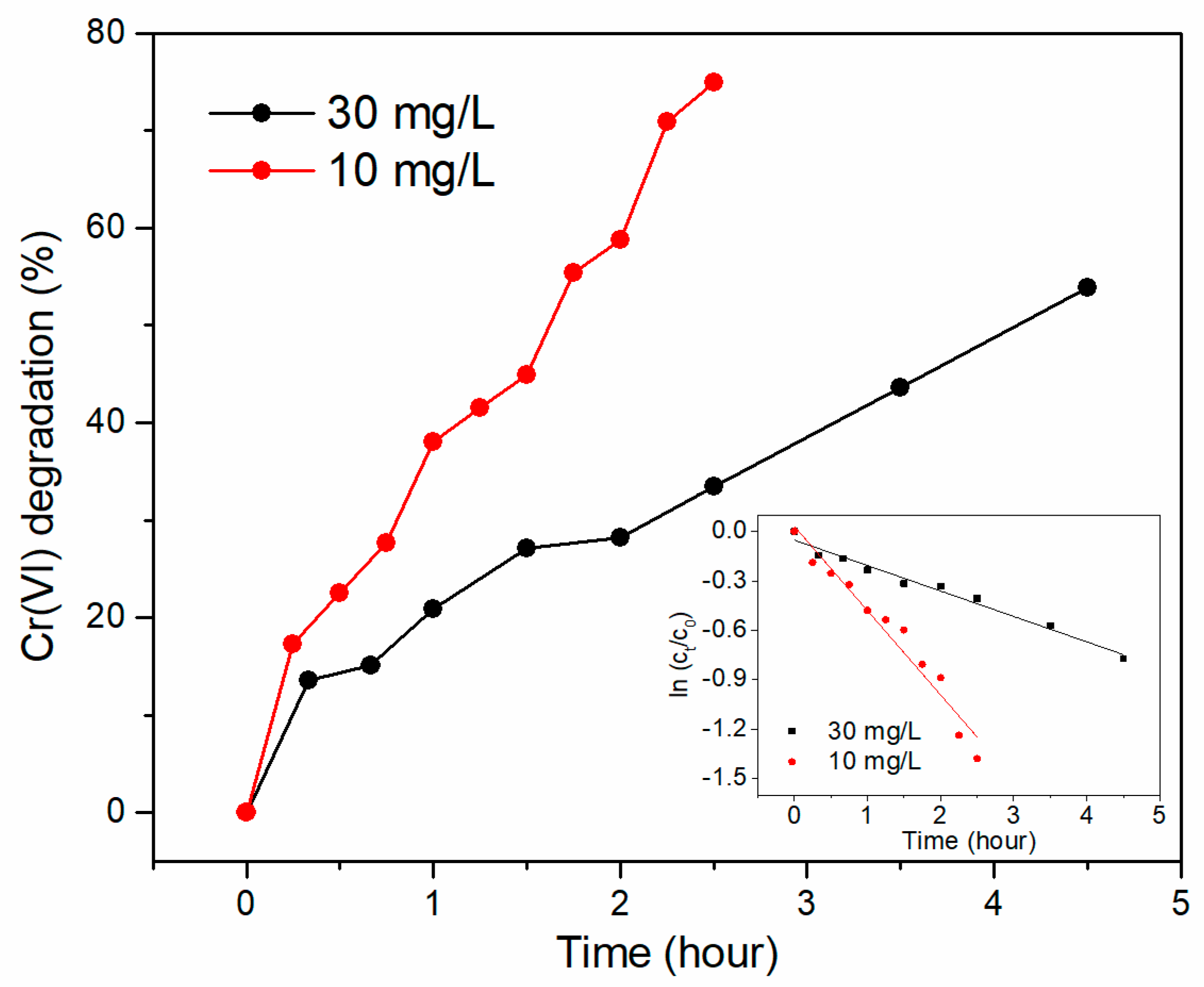
| Cathode | OCV (mV) | Max. Power Density (mW/m2) | System Resistance (Ω) |
|---|---|---|---|
| Graphite | 520 | 35.5 | 24.7 |
| Pyrrhotite-Coated | 528 | 45.4 | 18.1 |
| Growth Percentage | 1.54% | 27.89% | −26.72% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Zhao, W.; Liu, C.; Jiang, T.; Ding, H. Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode. Water 2017, 9, 979. https://doi.org/10.3390/w9120979
Shi J, Zhao W, Liu C, Jiang T, Ding H. Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode. Water. 2017; 9(12):979. https://doi.org/10.3390/w9120979
Chicago/Turabian StyleShi, Junxian, Wenyan Zhao, Chang Liu, Tao Jiang, and Hongrui Ding. 2017. "Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode" Water 9, no. 12: 979. https://doi.org/10.3390/w9120979
APA StyleShi, J., Zhao, W., Liu, C., Jiang, T., & Ding, H. (2017). Enhanced Performance for Treatment of Cr (VI)-Containing Wastewater by Microbial Fuel Cells with Natural Pyrrhotite-Coated Cathode. Water, 9(12), 979. https://doi.org/10.3390/w9120979




