1. Introduction
Biomanipulation by removing zooplanktivorous and benthivorous fish has been used as a method to restore and improve lake water quality for many years [
1,
2,
3,
4,
5]. By decreasing the top-down control from fish on particularly large-sized zooplankton, the aim is to increase the filtration of zooplankton capacity on phytoplankton and thereby create clearer water. Removal of benthivorous fish species such as carp (
Cyprinus carpio), gizzard shad (
Dorosoma cepedianum) and bream (
Abramis brama) may also improve lake water quality in shallow lakes by reducing the sediment resuspension and nutrient recycling caused by their feeding activity [
6,
7] and possibly also by reducing the amount of loose sediment, which otherwise would be more easily resuspended by wind [
8].
Numerous examples of biomanipulation have been given in the literature during the past 30 years, including more general approaches integrating the results from a number of case studies [
6,
9,
10,
11]. Overall, clear effects of biomanipulation have often been recorded, among these a trophic cascade impacting most trophic levels; however, the effects have sometimes been weak [
12,
13,
14]. In some biomanipulation projects, for example those undertaken in Danish lakes, a clear tendency to a return to the previous turbid conditions after 5 to 10 years has been observed [
10]. The failure to establish a long-lasting clear water state and inability to create permanent effects have been ascribed to a number of factors. First, adequate reduction of the external nutrient loading is crucial to obtain permanent effects [
2,
3,
15,
16]. In shallow lakes, TP concentrations below 0.05 mg·L
−1 have been suggested as a prerequisite of this [
17]. Furthermore, a sufficient number of fish need to be removed, depending also on TP concentrations [
18]. In shallow lakes, extensive cover of submerged macrophytes is important for stabilising and maintaining clear water conditions [
19], and the overall biomanipulation success may depend on the internal nutrient loading and the interacting effects of nutrient concentrations, fish recolonization and macrophyte abundance [
11,
14,
20].
Post-restoration data sets rarely cover more than a few years after restoration, which renders it difficult to draw any firm conclusions on how often long-term or permanent effects are achieved. Most lake restorations are conducted as a single intervention and repeated lake restorations via fish removal are rare. If a first biomanipulation is not successful in the long term, it has been suggested that a second biomanipulation demands less effort due to the slow recovery of some large benthivores, higher abundance of potential piscivores (e.g., perch,
Perca fluviatilis) and reduced risk of high internal phosphorus loading [
6].
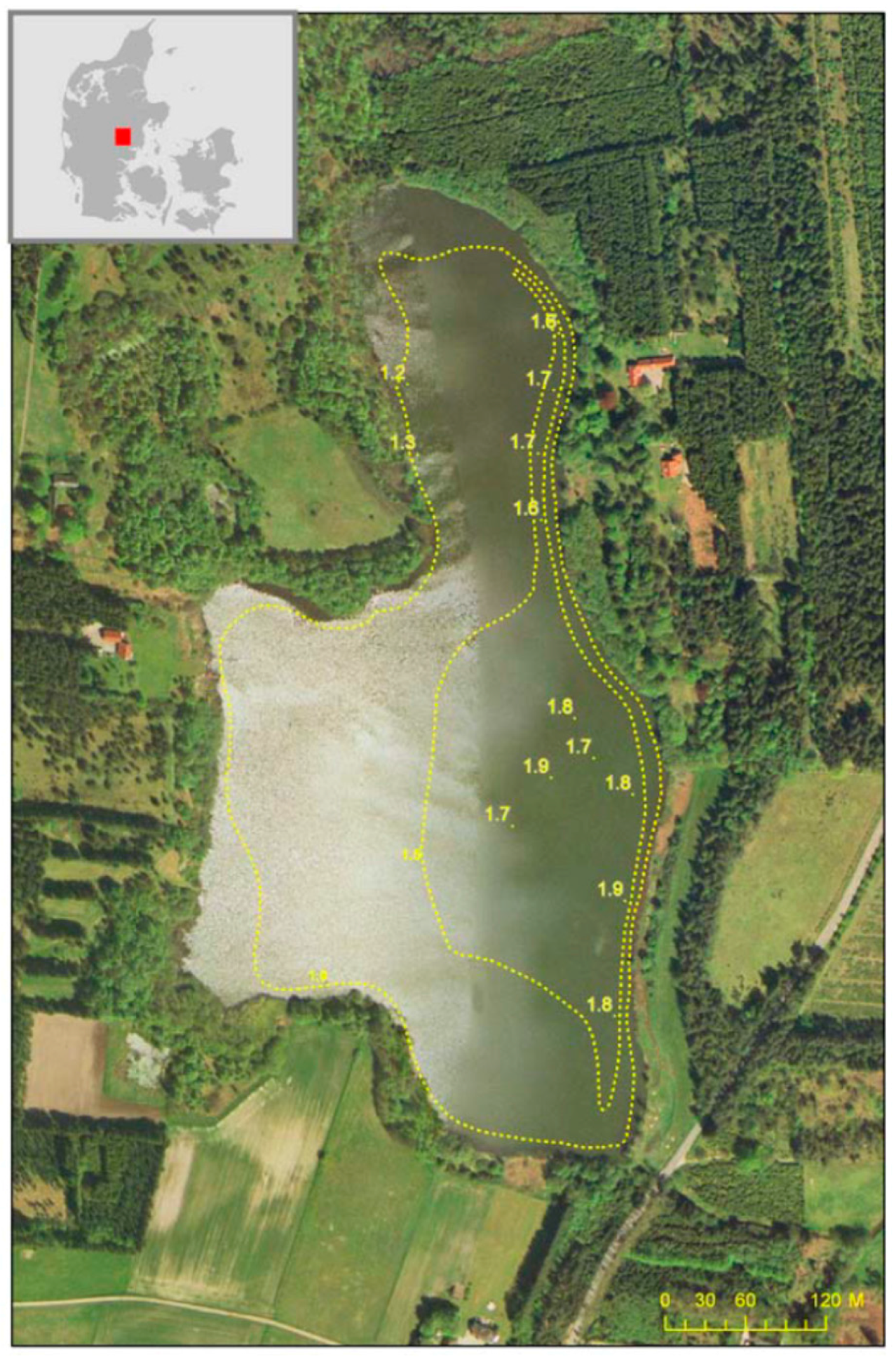
In this paper, we describe the results obtained from two biomanipulation cases conducted with a 20-year interval in Lake Væng, Denmark. The lake has been monitored yearly since 1986 for a number of chemical and biological parameters. Our aim was to study the long-term effects of fish biomanipulation on different trophic levels and nutrient dynamics and to investigate the impacts of repeated fish removal as a lake restoration tool. Our hypothesis was that repeated fish removal would imitate the effects achieved by the first biomanipulation, but with less fishing effort.
4. Discussion
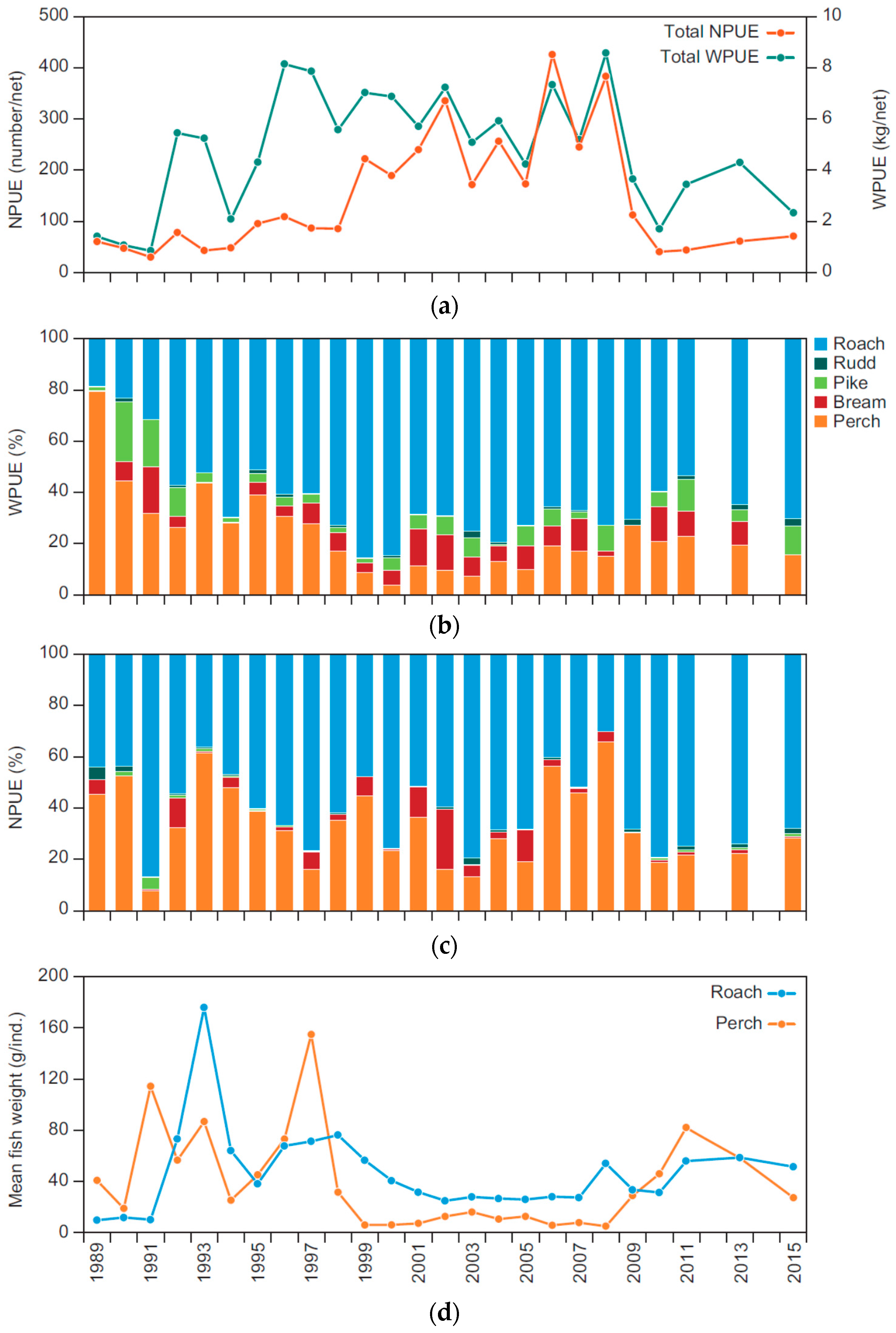
We would like to emphasise three main findings from our long-term study of Lake Væng: (1) biomanipulation by fish removal can greatly improve lake water quality for a number of years and repeated fish removal can help maintain the positive effects; (2) the effects of the fish removal cascade all the way down to the level of nutrients, altering the nitrogen and phosphorus cycling markedly on both the yearly and seasonal scale; and (3) less fishing effort seems needed at the second biomanipulation to achieve similar effects as during the first manipulation.
The effects of both biomanipulation attempts in Lake Væng were comparable with those obtained in other shallow Danish lakes [
10], and in many other parts of the world as well [
9,
11,
29]. We are not able to ascertain whether the fish biomass was lower before the second than before the first biomanipulation in Lake Væng, but the chemical and biological effects of the second biomanipulation appear to be just as strong as after the first biomanipulation, even though 30% less fish was removed. This indicates that a follow-up fish removal requires less effort, in our case probably due to the presence of a relatively large population of small perch after the first manipulation. These small perch have the potential to reach an older, larger and piscivorous stage after fish removal and thereby contribute to controlling the roach population [
6]. Perch mean weight increased after both biomanipulations, reflecting the potential of perch to reach the piscivorous stage when mass removal of planktivorous and benthivorous fish produces a dietary change in the remaining species towards increased utilisation of benthic resources [
30].
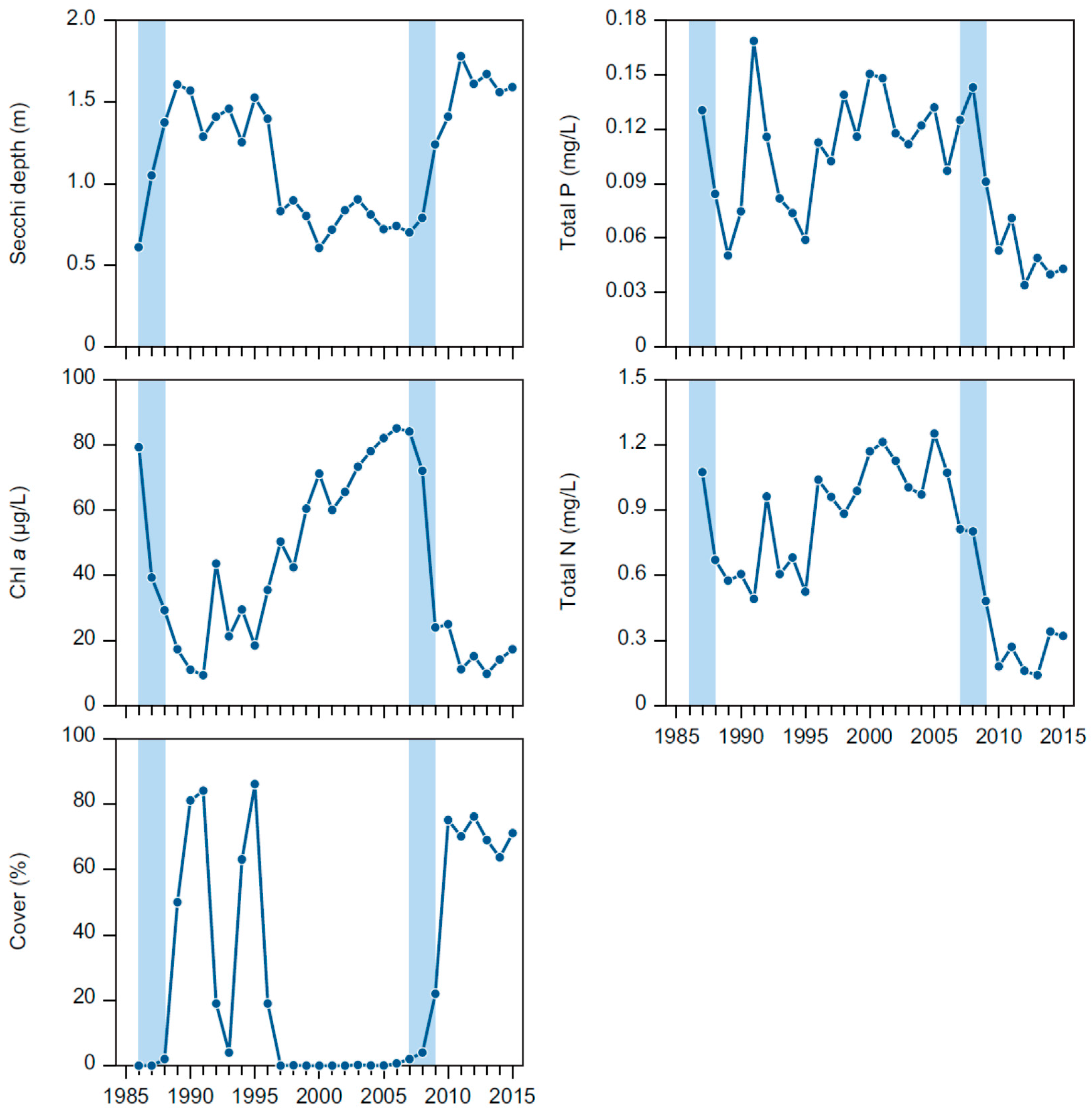
The effects obtained from the first and second biomanipulation were remarkably similar: within 1–2 years after the fish removal, the lake had changed from a turbid to clear water state with a strongly reduced phytoplankton biomass, within 2–3 years the submerged macrophyte community (dominated by
Elodea after both biomanipulations) had developed from almost absence to almost complete cover all over the lake from bottom to surface and, finally, the shifts between turbid and clear water changed the internal cycling of nutrients markedly and in the same manner. After the second biomanipulation, lower nitrogen concentrations and a more diverse macrophyte community have been observed, but overall the effects of the two biomanipulations have been very similar and they suggest a strong top-down impact of fish followed by similar cascading effects. Although nutrients and phytoplankton are not independent variables, many of these effects most likely reflect the reduced phytoplankton biomass created through by less nutrients and higher zooplankton grazing [
6] and improved light conditions, enabling a shift from mainly pelagic to mainly benthic primary producers. The major decline in CHL after biomanipulation has previously been shown to produce relatively minor effects on gross production and respiration but a higher seasonal amplitude in net production [
6], reflecting the shift in dominant primary producers and higher variability in the biomass of macrophytes. Not many case studies on repeated fish removals are available, but in Dutch Lake Zwemlust a second fish removal was observed to create similar effects as the first removal [
31]. The absence of long-term effects was ascribed to the high nutrient loading, which only allows submerged macrophytes to dominate in the “window of opportunity” occurring in the first years after the fish removal.
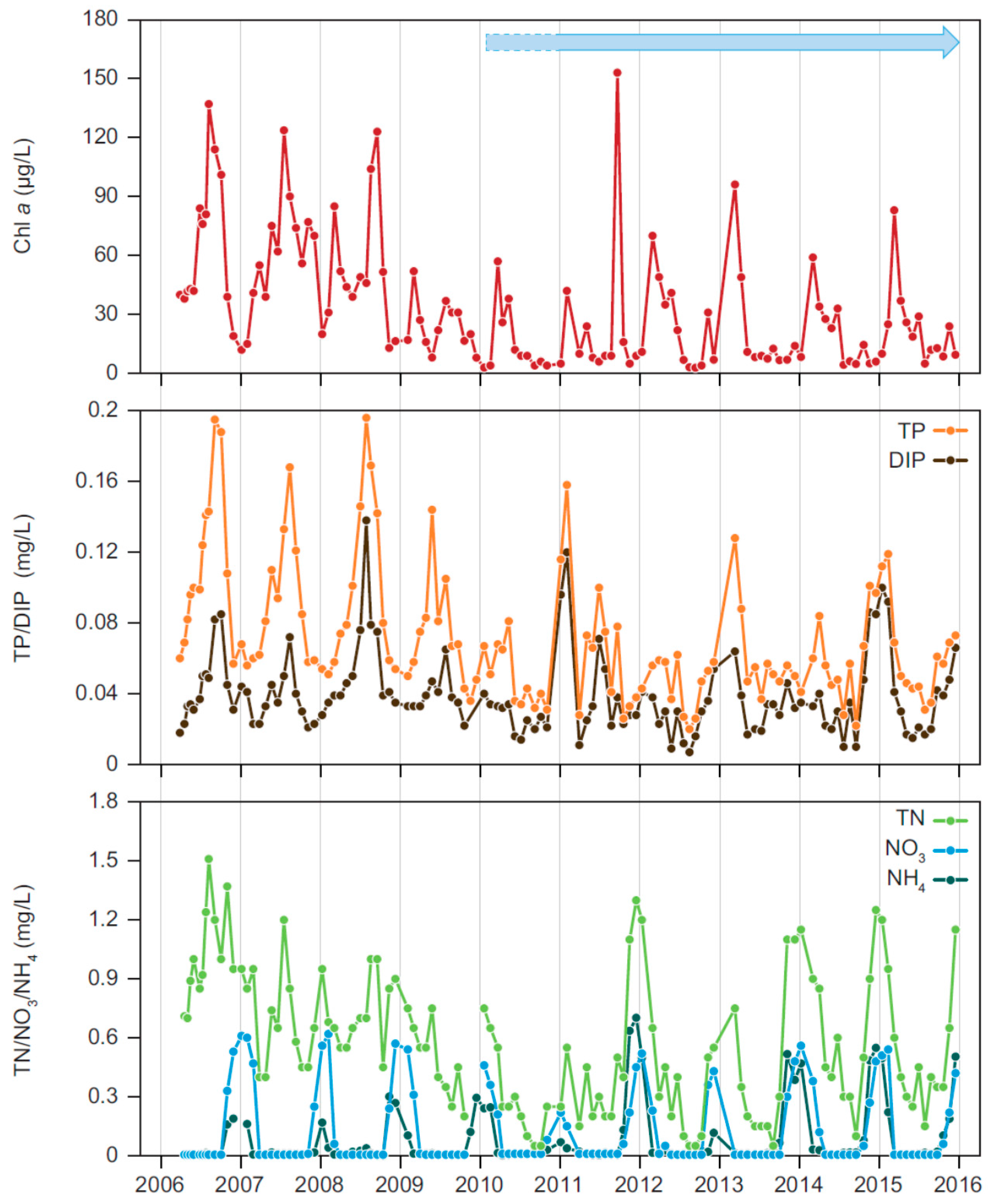
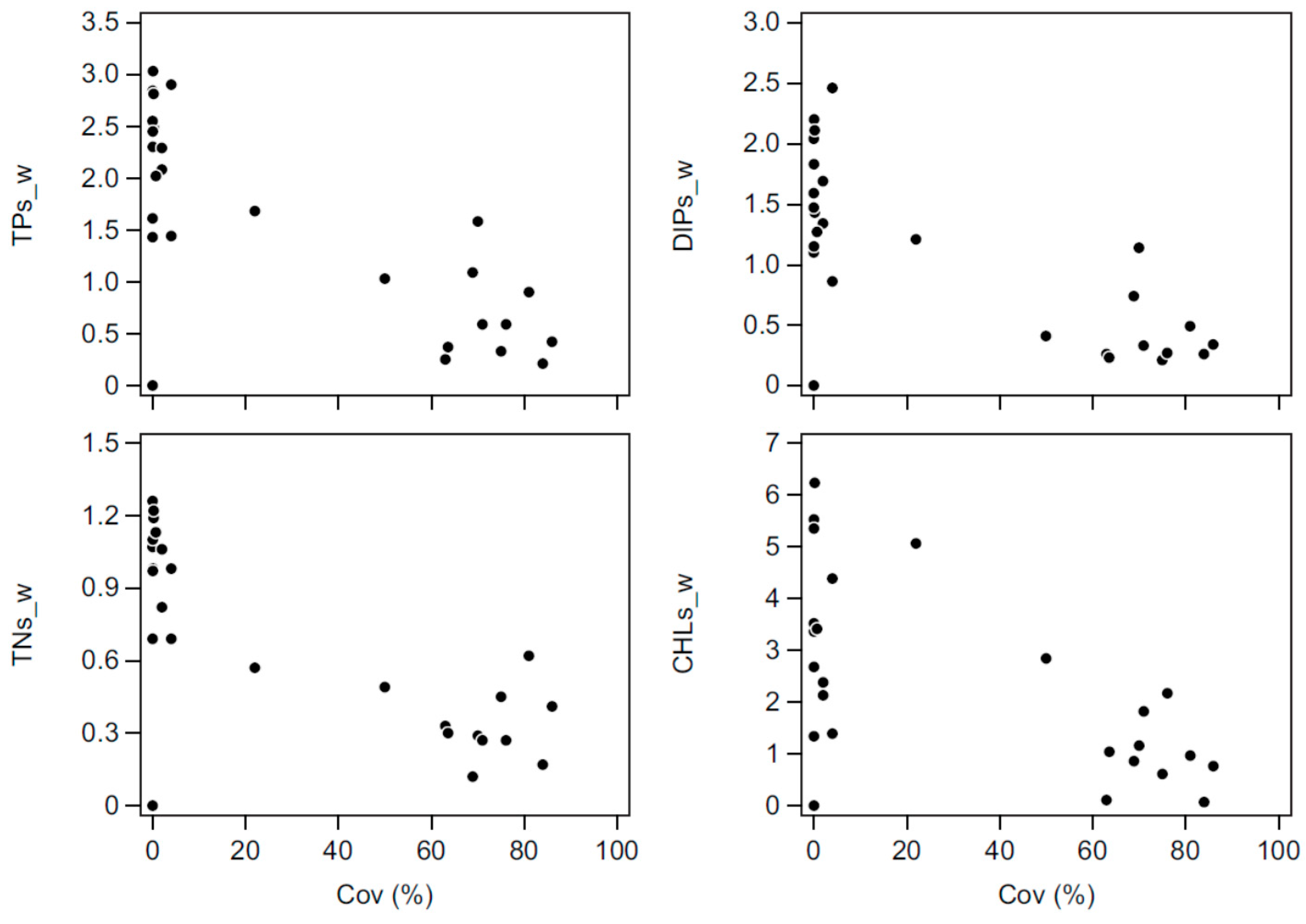
Both biomanipulations had pronounced effects on the nutrient concentrations in Lake Væng, and TP and TN were reduced to about 50% of pre-biomanipulation levels with increasing water clarity. As no changes in external loading have occurred, the lower concentrations must be produced by changes in the internal nutrient dynamics. Particularly, the internal summer release of phosphorus, which is often seen in shallow eutrophic lakes [
32,
33,
34], most likely decreased notably, which suggests a radical change in the way that the sediment acts as a sink or source of phosphorus depending on the biological structure. Similar effects have been observed in other biomanipulated lakes and in comparisons of inside and outside submerged macrophyte stands [
10,
35]. This may in part be attributed to improved light conditions at the sediment surface. This leads to higher benthic primary production [
36,
37], facilitating the uptake of phosphorus from the lake water and a subsequent reduction of the sediment phosphorus release due to the improved redox conditions. More details on the role of benthic algae in the phosphorus exchange between sediment and water are available in [
38]. In a Dutch shallow lake ecosystem [
39], phosphorus concentrations fluctuated strongly relative to variations in the growth of aquatic plants despite the fact that external phosphorus loading was stable. Years with low Cov had higher P concentrations than years with high Cov, the patterns thus being identical with those recorded in Lake Væng. In addition, lower sedimentation of phytoplankton may increase redox conditions in the sediment and reduced fish-induced resuspension may contribute to lower internal P loading. For nitrogen, decreased density of cyprinids (mainly roach and bream) was observed to lead to decreased N concentrations and increased N retention in four Danish lakes [
40]. In Lake Væng, summer N-ret% increased from 22%–39% before to 60%–72% after the first biomanipulation, probably due to a decrease in organic N in the lake, reduced resuspension by fish and higher denitrification in the sediment [
40].
The importance of biological structure for the internal cycling of nutrients is also seen by the shifts in the seasonal concentrations of phosphorus, reflecting the high fluctuations in the abundance of submerged macrophytes. A likely explanation for the peaks in high winter concentrations of TP and DIP in periods with high Cov could be phosphorus release from decomposing macrophytes and redox sensitive release of phosphorus accumulated in the sediment. A dense and senescent macrophyte biomass at the sediment surface can result in reduced redox conditions and a reduced P sorption capacity [
41].
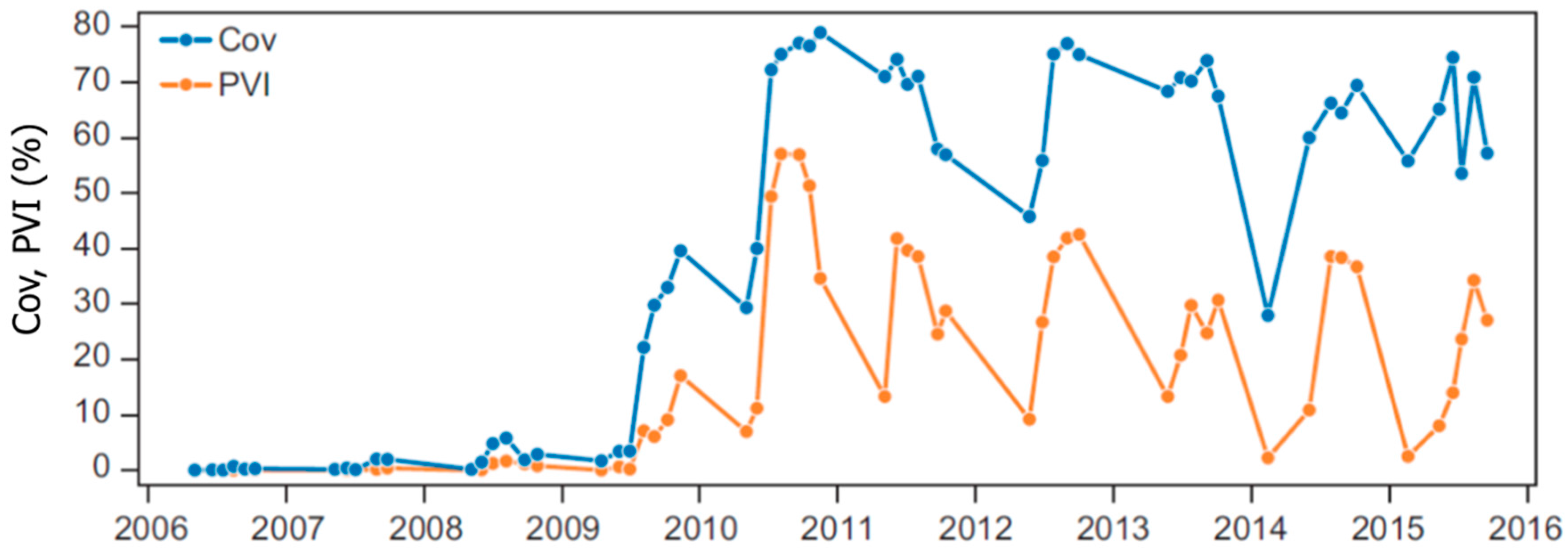
Highly dynamic and cyclical growth of submerged macrophytes and regime shifts between turbid and clear water conditions as those seen in Lake Væng have been recorded in several other lakes [
39,
42,
43]. In Lake Væng, P retention has been high, particularly after the second biomanipulation as demonstrated by low P concentrations in the lake water. Consequently, a mobile pool of phosphorus has accumulated in the sediment, a pool that may eventually be released, particularly if the lake returns to turbid conditions. High macrophyte Cov may then in the long term increase the risk of a return to turbid conditions, a negative feedback that might be supported by the build-up of decaying plant material in the sediment [
37]. It has been suggested that also the climate may contribute to the regime shifts through lowered macrophyte production, and in Lake Tåkern, Sweden, an eight-year cyclic periodicity of organic nitrogen has been observed to depend on the seasonal macrophyte production [
44]. Nitrogen may also be important, particularly for creating favourable growth conditions for submerged macrophytes, and high N concentrations have negative impacts on macrophytes [
45,
46,
47].
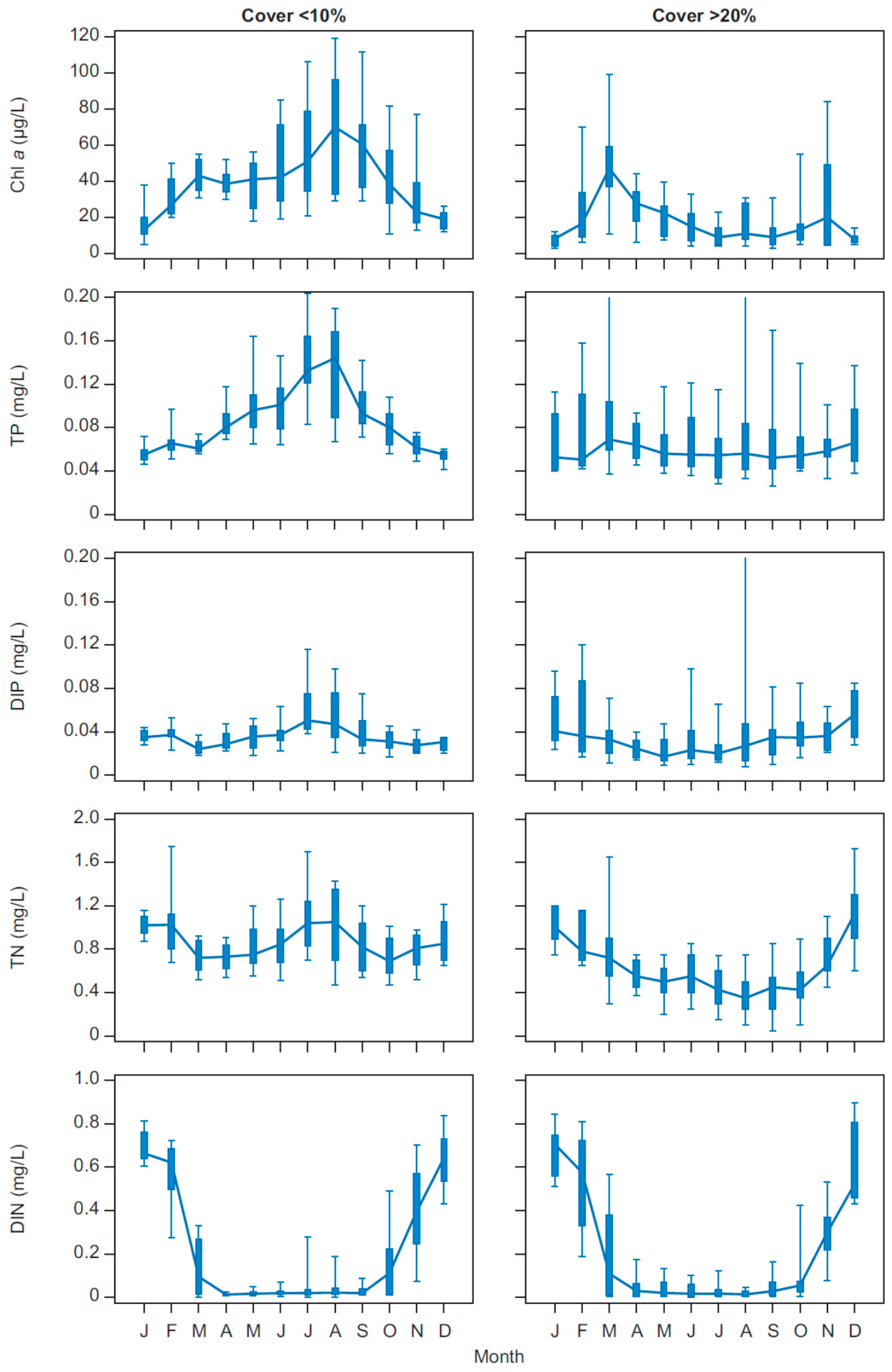
In Lake Væng, the seasonality in phytoplankton biomass changed markedly after the two biomanipulations, with a shift from maximum CHL concentrations during summer when the lake was turbid and without macrophytes to a maximum in winter when the lake was clear and rich in macrophytes. During summer in the presence of macrophytes, phytoplankton may be limited by grazing from zooplankton and other filtrators, by nutrients and the low light availability caused by the high macrophyte density. The significantly low DIN levels throughout summer under both clear and turbid conditions indicate that phytoplankton may be limited by nitrogen during the major part of their growing season, as also seen in other Danish shallow lakes and in other parts of the world [
48,
49]. During winter, increased nutrient availability, lower macrophyte abundance and less tall macrophytes may increase the phytoplankton biomass.
The numbers of grazing waterfowl, mute swan (
Cygnus olor) and coot (
Fulica atra) have fluctuated widely in Lake Væng from year to year concurrently with the abundance of submerged macrophytes [
23]. Waterfowl may negatively impact the abundance of submerged macrophytes and thereby the stability of clear water conditions, but even though waterfowl were present in high numbers in some years the decline in macrophytes cannot likely be ascribed to waterfowl grazing. A study from UK concluded that the potential of waterfowl herbivory to shift a macrophyte-dominated state into a phytoplankton-dominated state is low due to the recovery of aquatic plants during the growing season when bird populations decline [
50]. In winter 1991/1992 in Lake Væng, the number of coot and mute swan reached a maximum of, respectively, 800 and 300 individuals. During this winter, Cov decreased from 84% to 44% and PVI from 60% to 10%. Total macrophyte consumption by the waterfowl during this winter was estimated to 440 kg·DW·ha
−1 [
23]. The P content of
Elodea varies relatively widely, but assuming a mean content of 0.7% of the dry weight [
51] this would correspond to a potential release (if all phosphorus in the plant tissue is released to the water) of 3 kg·P/ha or 0.3 mg·L
−1 (with a mean depth of 1 m) during the whole winter. Thus, as the hydraulic retention in Lake Væng is only a few weeks, the increase caused by decomposing or ingested macrophytes can probably only explain a minor part of the increase seen in lake water nutrient concentrations.
The future environmental state of Lake Væng is uncertain and a number of unknown factors behind, for example, the fluctuating macrophyte abundance observed make predictions difficult. Factors in favour of clear water conditions are: (1) the macrophyte community appears more stable after the second biomanipulation with a more constant high abundance and presence of more species than after the first biomanipulation; (2) Nitrogen concentrations are lower now than after the first biomanipulation, augmenting the chances of submerged macrophytes maintaining their high abundance and thus stabilising clear water conditions [
45,
52]. Other factors could, however, indicate a return to turbid conditions: (i) the first biomanipulation did not create permanently clear water conditions; and (ii) the high nutrient concentrations in the groundwater may counteract the maintenance of clear water conditions [
24]. The mean concentration of total phosphorus in the groundwater just below the lake bed is 0.162 mg·L
−1 [
24]. However, as TP concentrations vary considerably between the groundwater wells, it is difficult to establish a precise phosphorus loading of the lake. It has been estimated (GEUS, personal communication) that Lake Væng receives between 200 and 280 kg P yearly with the groundwater, corresponding to 1.3–1.8 g·P·m
−2·year
−1 or a mean inlet TP concentration of 0.12 mg·L
−1. This is more than twice as high as the suggested maximum levels of 0.6–0.8 g·P·m
−2·year
−1 [
53] in order to ensure long-term success of biomanipulation in shallow lakes; (3) Paleolimnological investigations of the upper 0.5 m of the sediment involving calculation of diatom-inferred TP concentrations indicate that Lake Væng has been eutrophic for centuries, with TP concentrations fluctuating between 100 and 160 μg·L
−1 [
54]; (4) The dominance of
Elodea may not favour stable clear water conditions as this submerged macrophyte species is known to show rapid changes in abundance. In a Dutch biomanipulation experiment,
Elodea abundance showed high variability, ranging between high to sudden low abundances [
55]; (5) The present high retention of phosphorus entails accumulation of a phosphorus pool in the surface of the sediment, and this might be released should the lake return to turbid conditions. Consequently, at the present nutrient levels, clear water conditions in Lake Væng are difficult to maintain, and regular repetition of biomanipulation therefore seems required.