Statistical Analysis of the Spatial Distribution of Multi-Elements in an Island Arc Region: Complicating Factors and Transfer by Water Currents
Abstract
:1. Introduction
2. Geological and Marine Settings
2.1. Riverine System
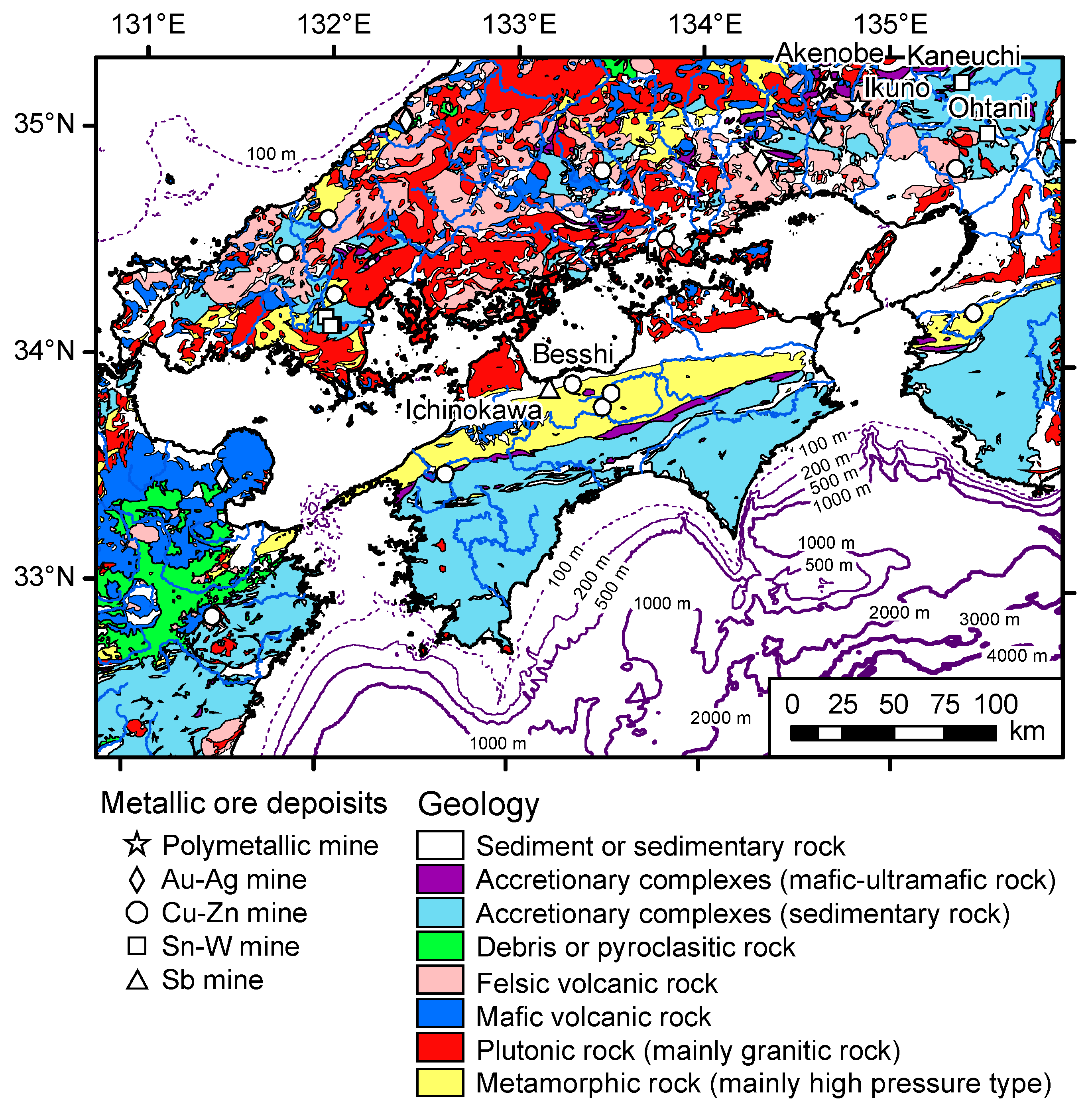
2.2. Geology and Terrestrial Metalliferous Deposits
2.3. Marine Topography, Hydrographic Condition, and Geology
3. Materials and Methods
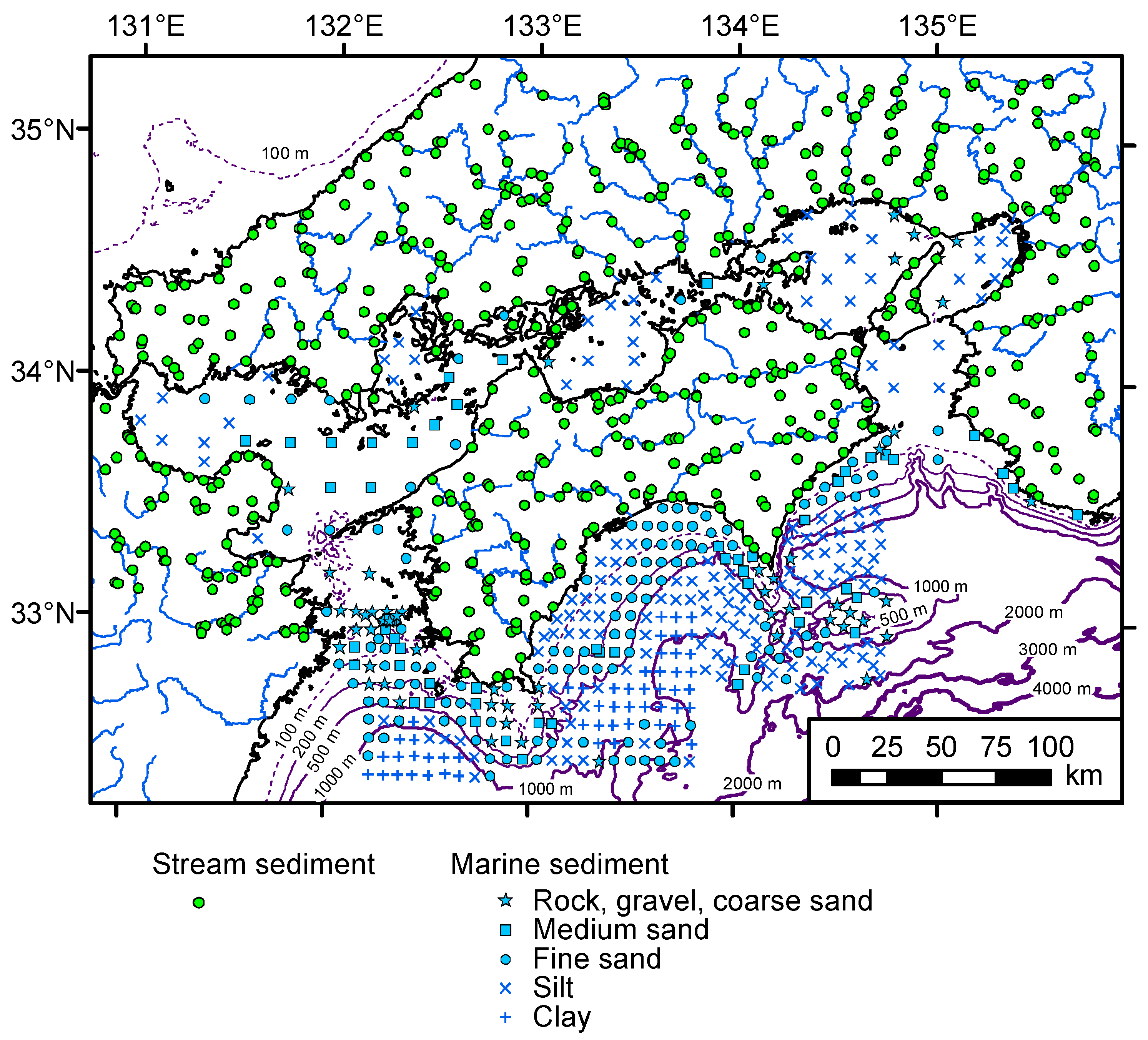
3.1. Samples, Sampling Methods, and Processing
3.2. Geochemical and Spatial Analyses
4. Results
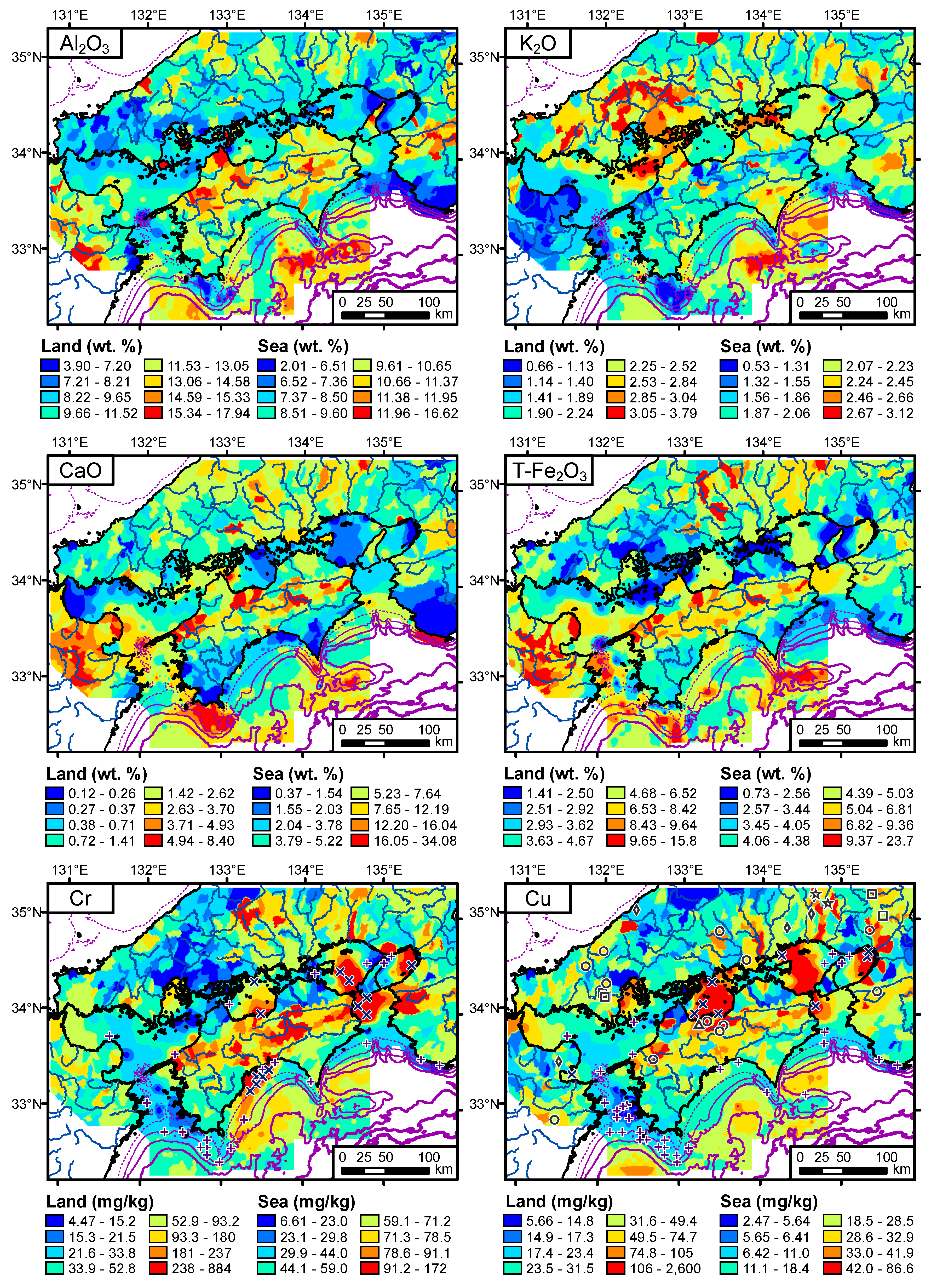
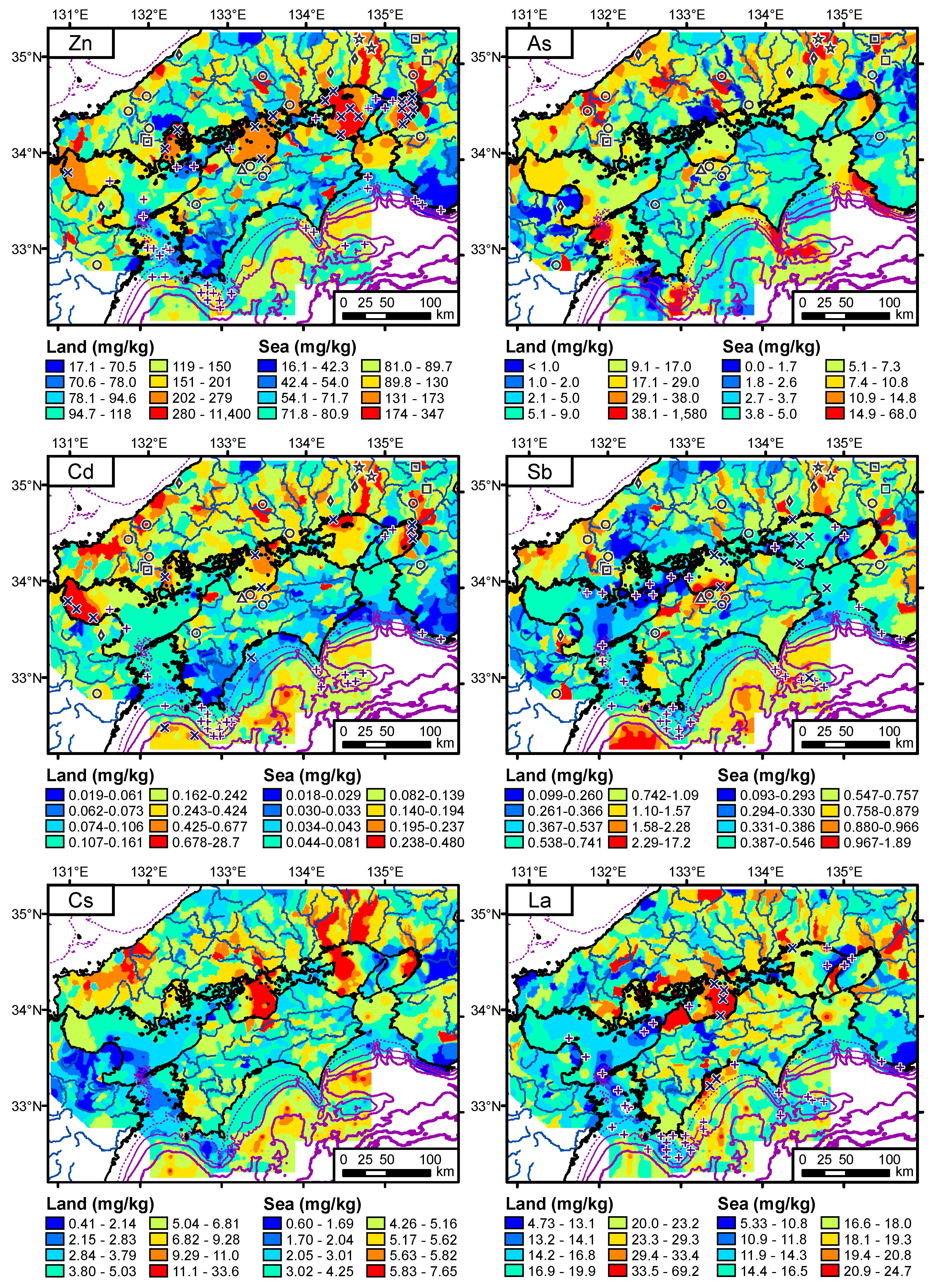
4.1. Spatial Distribution of Terrestrial Elemental Concentrations
4.2. Spatial Distribution of Elemental Concentrations in Coastal Sea Sediments
5. Discussion
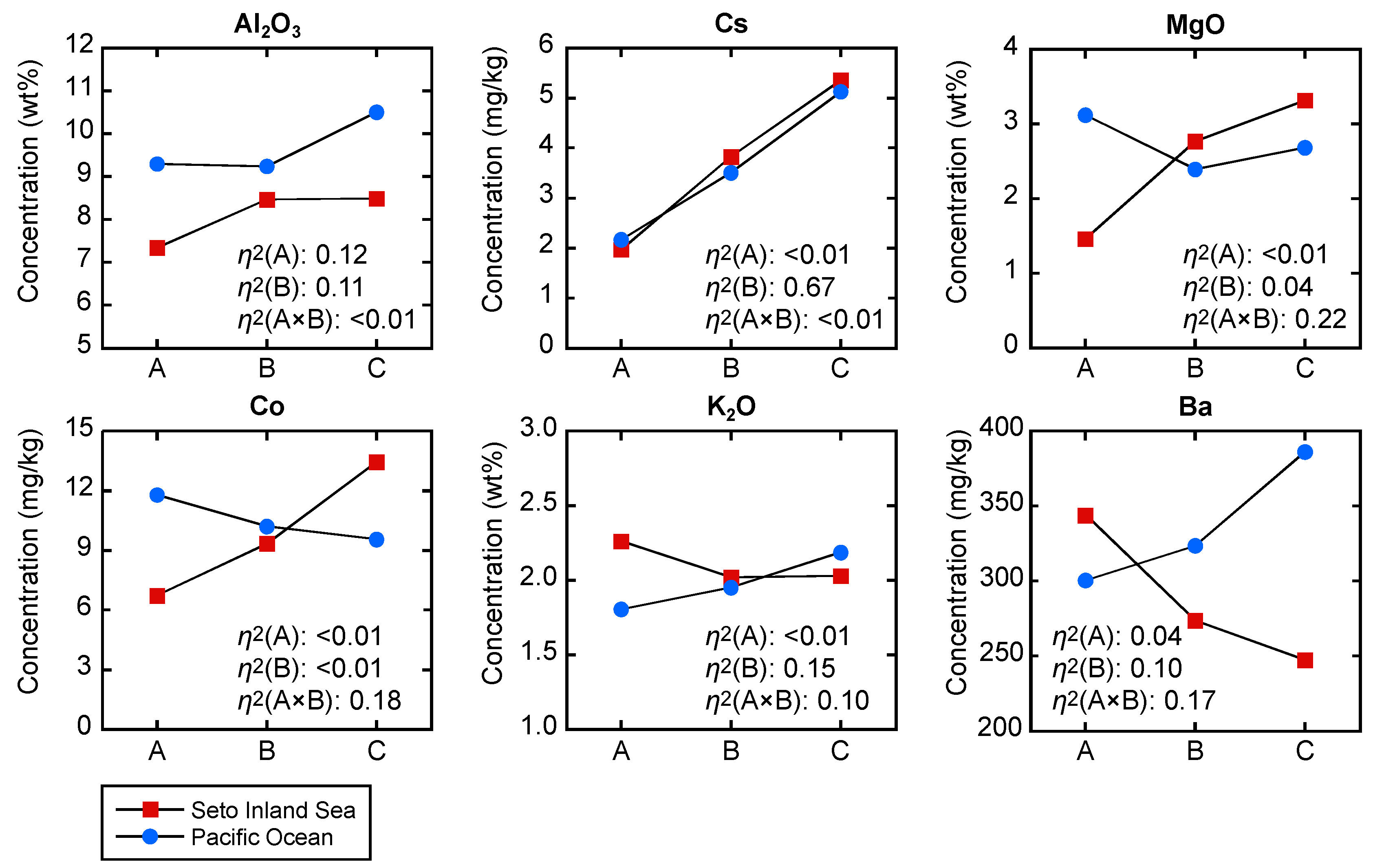
5.1. Analysis of Variance (ANOVA) to Reveal the Effects of Particle Size and Regional Difference
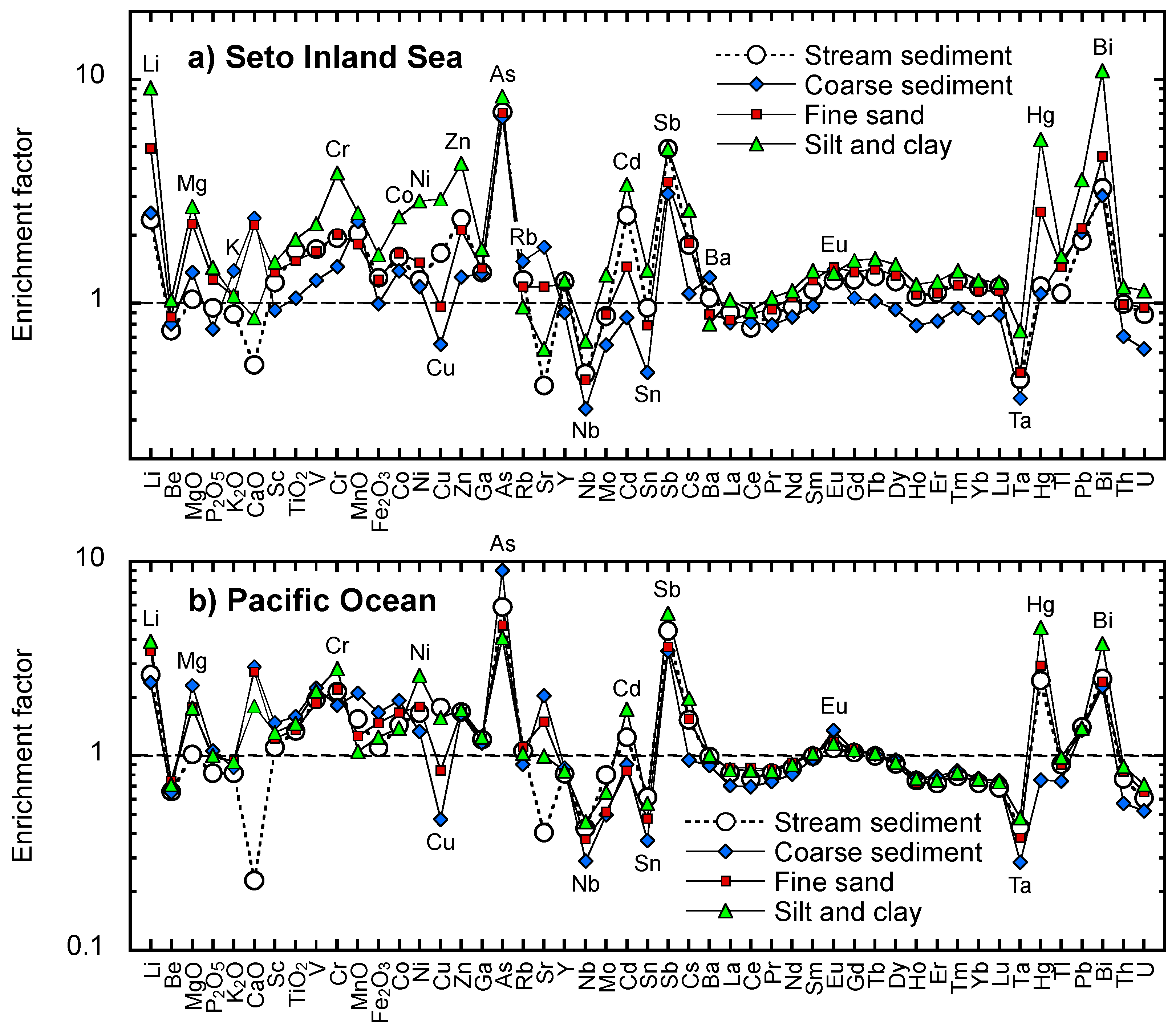
5.2. Comparison of Elemental Abundances in Coastal Sea and Stream Sediments between the Seto Inland Sea and the Pacific Ocean
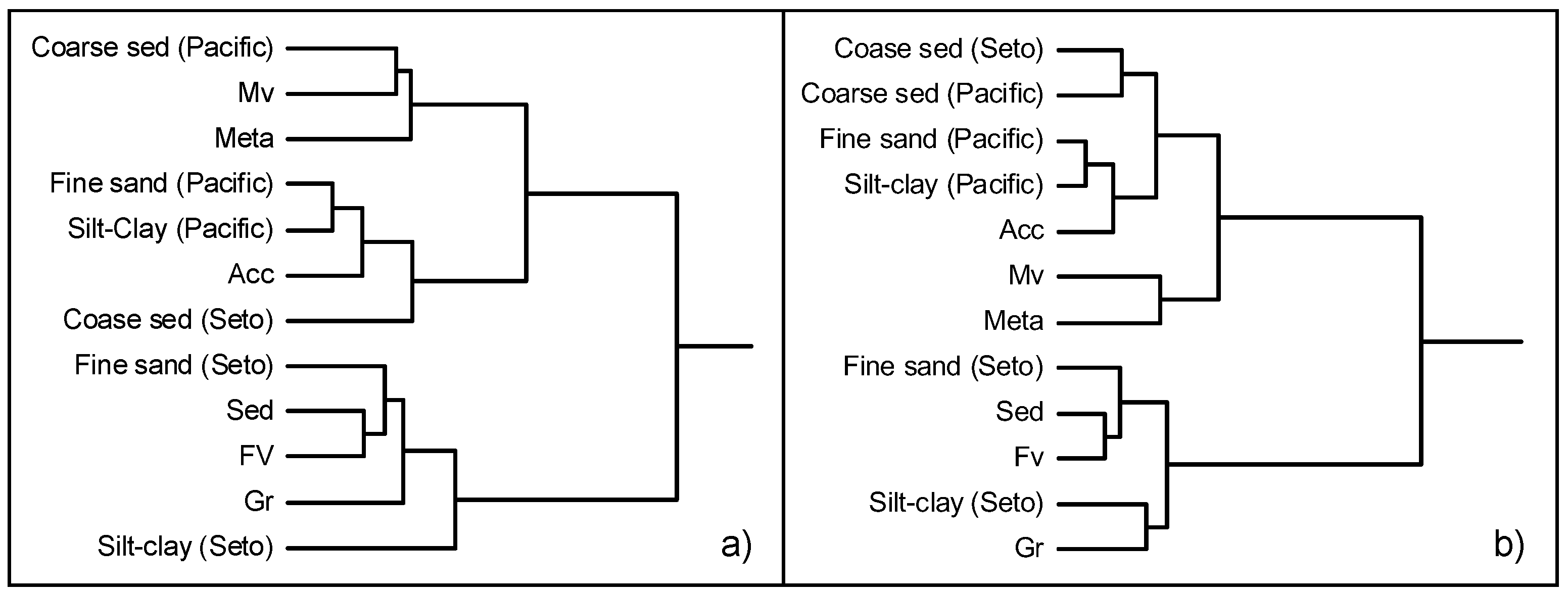
5.3. Discrimination of Coastal Sea Sediments Using Cluster Analysis
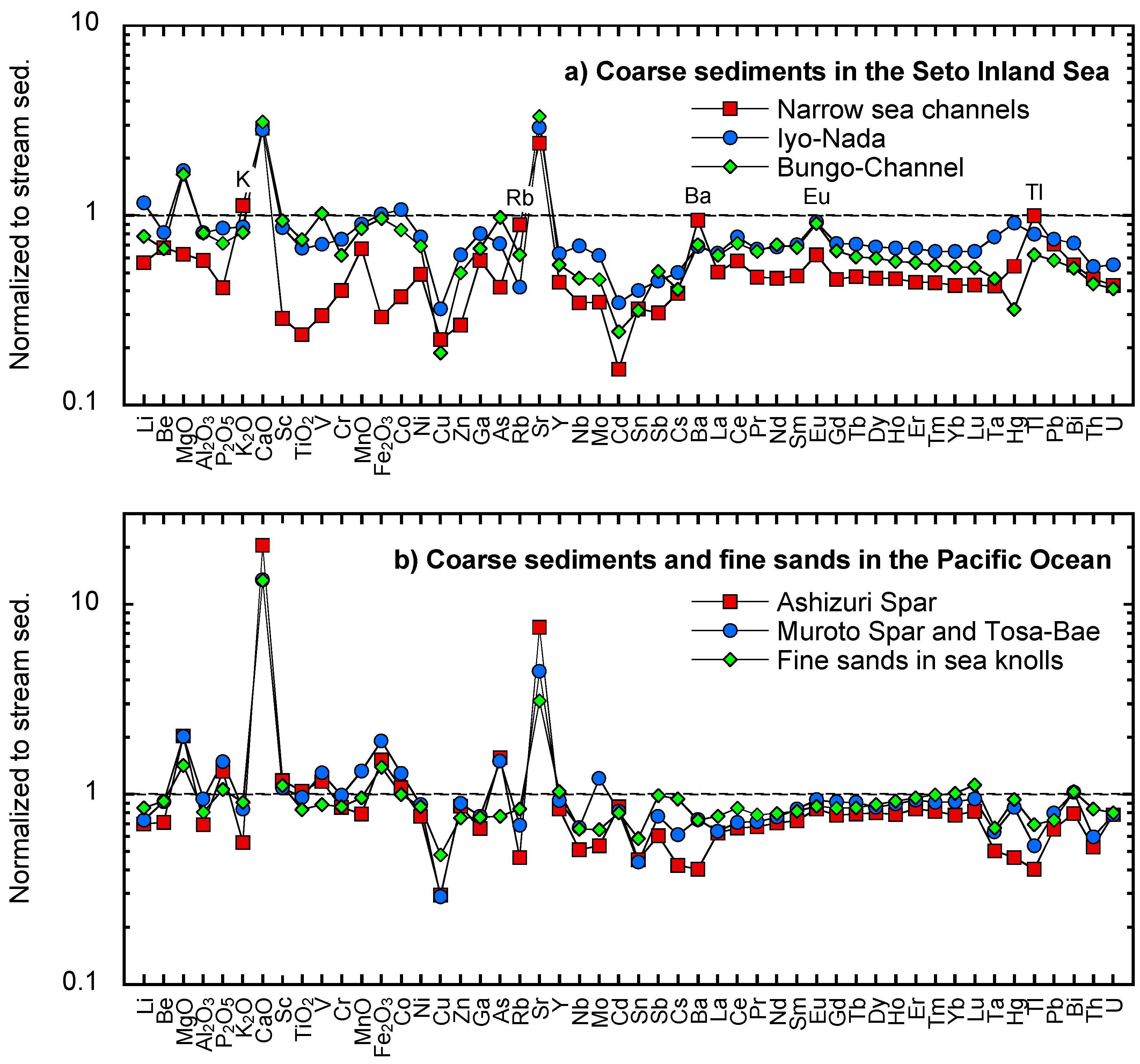
5.4. Discrepancies in the Geochemistry of Coarse Marine Sediments and Stream Sediments
5.4.1. Fractionation of Mineralogical Compositions in Coarse Sediments by a Strong Tidal Current
5.4.2. Denudation of the Basement Rocks of the Topographic Highs
5.5. The Transfer of Silty and Clayey Sediments Influenced by Periodic Currents, Tidal Currents, and a Water Mass Boundary
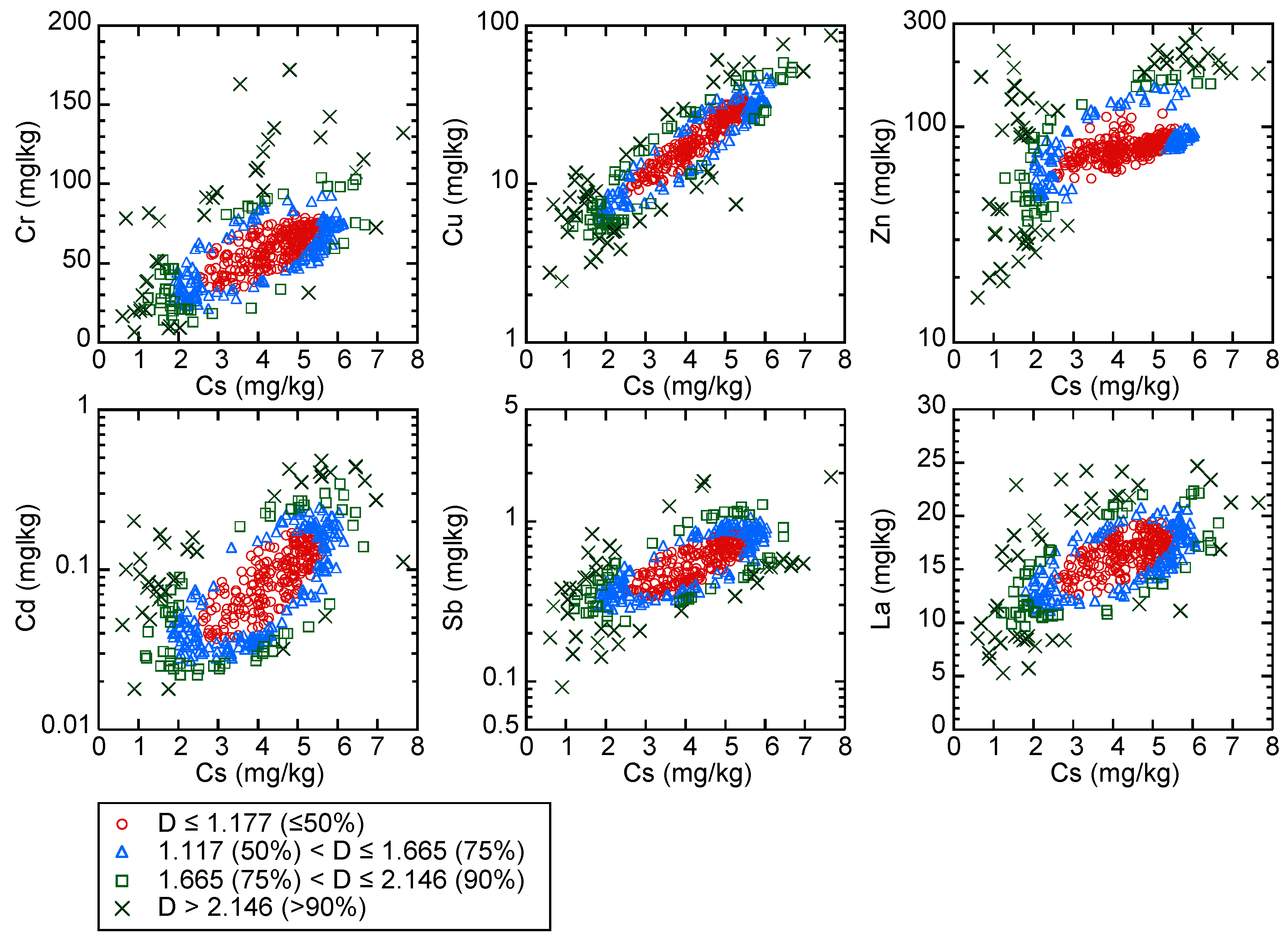
5.5.1. Mahalanobis’ Generalized Distances for Finding Outliers
5.5.2. Transport of Elements in Silt Derived from the Parent Lithology
5.5.3. Transfer of Materials Related to Mineral Deposits and Anthropogenic Activity
5.5.4. Geochemical Features of Silt and Clay in the Deep Sea Basins of the Pacific Ocean
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| River System | Sediment Yield (×103 m3/Year) | River Water Discharge 1 (×106 m3/Year) | Discharged Area | Relative Rate to Total Sediment Yield |
|---|---|---|---|---|
| Shikoku region | ||||
| Yoshino (Ys) | 870 | 3202 | Kii Channel | 11% |
| Naka (Na) | 510 | 1628 | Kii Channel | 7% |
| Toki (To) | 83 | (34) 2 | Ibi-Seto-Nada | 1% |
| Shigenobu (Sg) | 97 | 46 | Iyo-Nada | 1% |
| Hiji (H) | 215 | 1111 | Iyo-Nada | 3% |
| Mononobe (M) | 458 | 777 | Tosa Bay | 6% |
| Niyodo (Ni) | 534 | 3101 | Tosa Bay | 7% |
| Shimanto (Sm) | 699 | 4712 | Tosa Bay | 9% |
| Chugoku region | ||||
| Yoshii (Yi) | 290 | (1125) 2 | Ibi-Seto-Nada | 4% |
| Asahi (Ah) | 238 | 1492 | Ibi-Seto-Nada | 3% |
| Takahari (Ta) | 358 | 1605 | Ibi-Seto-Nada | 5% |
| Ashido (Ad) | 142 | 187 | Bingo-Nada | 2% |
| Ohta (Ot) | (238) 3 | - | Hiroshima Bay | 3% |
| Oze (Oz) | 48 | 255 | Hiroshima Bay | 1% |
| Sanami (Sn) | 95 | 261 | Suo-Nada | 1% |
| Kinki region | ||||
| Ibo (I) | 174 | 676 | Harima-Nada | 2% |
| Kako (Ka) | 247 | 918 | Harima-Nada | 3% |
| Yodo (Yd) | 1310 | 5376 | Osaka Bay | 17% |
| Yamato (Ym) | 123 | (426) 2 | Osaka Bay | 2% |
| Kino (Ki) | 377 | 1184 | Kii Channel | 5% |
| Kyushu region | ||||
| Yamakuni (Yk) | 99 | 418 | Suo-Nada | 1% |
| Ohita (Oi) | 150 | 498 | Beppu Bay | 2% |
| Ohno (On) | 240 | 1616 | Beppu Bay | 3% |
| Banjo (B) | 89 | 392 | Bungo Channel | 1% |
Appendix B
| Element | Sed | Sed_u | Acc | Gr | Mv | Fv | Phy | Meta | Other |
|---|---|---|---|---|---|---|---|---|---|
| n = 70 | n = 11 | n = 157 | n = 71 | n = 41 | n = 61 | n = 13 | n = 47 | n = 105 | |
| wt % | |||||||||
| Na2O | 2.06 | 2.21 | 1.74 | 2.89 | 2.44 | 1.82 | 2.34 | 2.26 | 2.08 |
| MgO | 1.34 | 1.63 | 1.54 | 1.03 | 3.17 | 1.12 | 3.80 | 3.57 | 1.98 |
| Al2O3 | 11.54 | 9.89 | 11.47 | 11.43 | 12.14 | 10.35 | 13.43 | 13.35 | 11.05 |
| P2O5 | 0.109 | 0.157 | 0.093 | 0.098 | 0.156 | 0.085 | 0.181 | 0.148 | 0.103 |
| K2O | 2.21 | 2.22 | 2.28 | 2.57 | 1.13 | 2.65 | 1.34 | 1.74 | 2.18 |
| CaO | 1.21 | 1.41 | 0.52 | 1.58 | 3.60 | 1.07 | 4.33 | 2.87 | 1.80 |
| TiO2 | 0.56 | 0.65 | 0.49 | 0.50 | 1.10 | 0.48 | 1.17 | 0.73 | 0.68 |
| MnO | 0.088 | 0.083 | 0.091 | 0.116 | 0.152 | 0.117 | 0.187 | 0.168 | 0.126 |
| T-Fe2O3 | 3.94 | 4.59 | 4.13 | 3.82 | 7.00 | 4.10 | 9.42 | 7.21 | 5.19 |
| mg/kg | |||||||||
| Li | 35.2 | 32.5 | 42.7 | 30.4 | 25.5 | 38.4 | 24.2 | 37.3 | 37.6 |
| Be | 1.54 | 1.70 | 1.66 | 2.34 | 1.20 | 1.95 | 1.32 | 1.38 | 1.64 |
| Sc | 8.33 | 8.23 | 8.55 | 7.41 | 14.1 | 7.80 | 20.3 | 18.3 | 11.0 |
| V | 69 | 59 | 82 | 52 | 155 | 62 | 237 | 155 | 97 |
| Cr | 47.1 | 110 | 55.6 | 24.1 | 53.7 | 32.8 | 38.6 | 169 | 61.1 |
| Co | 9.05 | 11.9 | 10.4 | 7.1 | 18.5 | 8.6 | 22.7 | 23.8 | 13.1 |
| Ni | 18.7 | 38.4 | 26.2 | 9.3 | 17.2 | 13.0 | 12.6 | 59.5 | 25.3 |
| Cu | 29.5 | 67.7 | 35.9 | 25.2 | 27.0 | 27.7 | 31.9 | 62.3 | 35.2 |
| Zn | 108 | 194 | 101 | 114 | 130 | 137 | 131 | 122 | 130 |
| Ga | 16.3 | 16.8 | 16.6 | 19.2 | 17.9 | 17.8 | 18.8 | 17.9 | 17.1 |
| As | 6 | 7 | 8 | 7 | 4 | 22 | 8 | 8 | 13 |
| Rb | 102 | 109 | 102 | 121 | 39 | 153 | 49.8 | 73 | 104 |
| Sr | 110 | 97 | 90 | 100 | 254 | 91 | 301 | 144 | 122 |
| Y | 18.0 | 20.3 | 11.9 | 24.7 | 17.2 | 20.5 | 23.2 | 24.8 | 19.5 |
| Nb | 8.77 | 10.05 | 8.12 | 10.8 | 11.4 | 8.82 | 8.82 | 8.29 | 8.63 |
| Mo | 0.77 | 1.79 | 1.00 | 0.91 | 1.03 | 0.98 | 1.46 | 0.94 | 1.07 |
| Cd | 0.13 | 0.20 | 0.11 | 0.17 | 0.15 | 0.26 | 0.16 | 0.18 | 0.20 |
| Sn | 3.62 | 7.72 | 2.95 | 4.60 | 2.36 | 4.30 | 2.17 | 2.81 | 4.04 |
| Sb | 0.80 | 1.21 | 0.75 | 0.38 | 0.52 | 0.92 | 0.48 | 0.85 | 0.92 |
| Cs | 4.84 | 3.89 | 4.97 | 4.44 | 2.39 | 7.65 | 3.24 | 5.06 | 5.59 |
| Ba | 469 | 491 | 448 | 401 | 352 | 456 | 402 | 351 | 414 |
| La | 20.7 | 30.9 | 19.4 | 24.1 | 16.4 | 20.9 | 18.2 | 20.0 | 19.4 |
| Ce | 38.9 | 57.1 | 37.3 | 41.2 | 29.2 | 35.3 | 37.7 | 39.4 | 34.0 |
| Pr | 4.63 | 6.74 | 4.42 | 5.42 | 3.87 | 4.66 | 4.94 | 4.80 | 4.55 |
| Nd | 17.8 | 26.2 | 16.9 | 22.0 | 15.9 | 17.8 | 20.6 | 19.6 | 17.9 |
| Sm | 3.72 | 5.44 | 3.21 | 4.59 | 3.27 | 3.62 | 4.46 | 4.27 | 3.73 |
| Eu | 0.78 | 0.88 | 0.65 | 0.73 | 0.94 | 0.71 | 1.24 | 1.07 | 0.79 |
| Gd | 3.40 | 4.37 | 2.78 | 4.31 | 3.09 | 3.33 | 4.40 | 4.28 | 3.48 |
| Tb | 0.59 | 0.72 | 0.45 | 0.75 | 0.54 | 0.59 | 0.75 | 0.77 | 0.59 |
| Dy | 3.10 | 3.61 | 2.14 | 4.00 | 2.70 | 3.05 | 3.87 | 4.05 | 3.10 |
| Ho | 0.60 | 0.67 | 0.41 | 0.79 | 0.52 | 0.61 | 0.75 | 0.81 | 0.60 |
| Er | 1.77 | 1.89 | 1.14 | 2.34 | 1.56 | 1.81 | 2.24 | 2.29 | 1.80 |
| Tm | 0.28 | 0.29 | 0.18 | 0.40 | 0.25 | 0.30 | 0.36 | 0.36 | 0.29 |
| Yb | 1.75 | 1.74 | 1.11 | 2.58 | 1.53 | 1.89 | 2.23 | 1.97 | 1.82 |
| Lu | 0.26 | 0.27 | 0.16 | 0.39 | 0.24 | 0.28 | 0.33 | 0.27 | 0.26 |
| Ta | 0.77 | 0.87 | 0.73 | 1.11 | 0.80 | 0.70 | 0.72 | 0.71 | 0.68 |
| Hg | 0.06 | 0.12 | 0.12 | 0.03 | 0.06 | 0.04 | 0.06 | 0.07 | 0.04 |
| Tl | 0.63 | 0.62 | 0.58 | 0.69 | 0.33 | 0.85 | 0.47 | 0.44 | 0.64 |
| Pb | 26.0 | 37.1 | 24.2 | 32.4 | 22.6 | 44.1 | 19.0 | 20.6 | 29.6 |
| Bi | 0.23 | 0.32 | 0.28 | 0.40 | 0.17 | 0.52 | 0.25 | 0.25 | 0.39 |
| Th | 8.21 | 12.8 | 6.62 | 14.4 | 4.37 | 8.46 | 5.78 | 6.74 | 7.33 |
| U | 1.83 | 2.27 | 1.51 | 3.31 | 1.03 | 2.41 | 1.61 | 1.21 | 1.79 |
Appendix C
| Element | W-Value of Shapiro–Wilk Statistics | Skewness | Data Transformation | ||
|---|---|---|---|---|---|
| Unchanged | Log-Transformed | Unchanged | Log-Transformed | ||
| MgO | 0.884 | 0.872 | 1.3 | −1.3 | Unchanged |
| Al2O3 | 0.971 | 0.869 | −0.48 | −2.0 | Unchanged |
| P2O5 | 0.168 | 0.870 | 19 | 0.96 | Log-transformed |
| K2O | 0.949 | 0.820 | −0.78 | −2.3 | Unchanged |
| CaO | 0.777 | 0.980 | 2.5 | −0.34 | Log-transformed |
| TiO2 | 0.722 | 0.873 | 3.3 | −0.80 | Log-transformed |
| MnO | 0.731 | 0.946 | 2.9 | 0.72 | Log-transformed |
| T-Fe2O3 | 0.667 | 0.839 | 3.4 | −0.32 | Log-transformed |
| Li | 0.924 | 0.975 | 1.1 | −0.46 | Log-transformed |
| Be | 0.957 | 0.854 | −0.60 | −2.0 | Unchanged |
| Sc | 0.769 | 0.874 | 2.6 | −0.84 | Log-transformed |
| V | 0.580 | 0.914 | 6.1 | −0.19 | Log-transformed |
| Cr | 0.952 | 0.923 | 0.78 | −1.2 | Unchanged |
| Co | 0.804 | 0.905 | 2.5 | −0.55 | Log-transformed |
| Ni | 0.883 | 0.930 | 1.9 | −1.0 | Log-transformed |
| Cu | 0.922 | 0.965 | 1.1 | −0.44 | Log-transformed |
| Zn | 0.734 | 0.896 | 2.7 | −0.02 | Log-transformed |
| Ga | 0.919 | 0.781 | −1.2 | −2.6 | Unchanged |
| As | 0.615 | 0.899 | 4.7 | −0.98 | Log-transformed |
| Rb | 0.985 | 0.897 | −0.44 | −1.5 | Unchanged |
| Sr | 0.647 | 0.964 | 3.7 | 0.69 | Log-transformed |
| Y | 0.977 | 0.991 | 0.66 | −0.29 | Log-transformed |
| Nb | 0.990 | 0.945 | 0.01 | −0.93 | Unchanged |
| Mo | 0.378 | 0.961 | 11.22 | 0.85 | Log-transformed |
| Cd | 0.835 | 0.978 | 1.8 | 0.14 | Log-transformed |
| Sn | 0.519 | 0.936 | 6.8 | 0.53 | Log-transformed |
| Sb | 0.931 | 0.980 | 1.1 | −0.27 | Log-transformed |
| Cs | 0.961 | 0.885 | −0.42 | −1.3 | Unchanged |
| Ba | 0.771 | 0.886 | 4.4 | −1.2 | Log-transformed |
| La | 0.981 | 0.917 | −0.45 | −1.3 | Unchanged |
| Ce | 0.981 | 0.924 | −0.38 | −1.2 | Unchanged |
| Pr | 0.973 | 0.897 | −0.61 | −1.5 | Unchanged |
| Nd | 0.969 | 0.888 | −0.64 | −1.6 | Unchanged |
| Sm | 0.975 | 0.905 | −0.44 | −1.4 | Unchanged |
| Eu | 0.987 | 0.971 | 0.20 | −0.64 | Unchanged |
| Gd | 0.983 | 0.942 | 0.01 | −1.0 | Unchanged |
| Tb | 0.982 | 0.971 | 0.36 | −0.62 | Unchanged |
| Dy | 0.981 | 0.982 | 0.48 | −0.46 | Log-transformed |
| Ho | 0.980 | 0.989 | 0.56 | −0.34 | Log-transformed |
| Er | 0.982 | 0.988 | 0.53 | −0.36 | Log-transformed |
| Tm | 0.983 | 0.989 | 0.50 | −0.36 | Log-transformed |
| Yb | 0.985 | 0.986 | 0.45 | −0.42 | Log-transformed |
| Lu | 0.987 | 0.983 | 0.40 | −0.50 | Unchanged |
| Ta | 0.986 | 0.948 | 0.10 | −0.87 | Unchanged |
| Hg | 0.187 | 0.819 | 19 | −1.6 | Log-transformed |
| Tl | 0.964 | 0.876 | 0.20 | −1.6 | Unchanged |
| Pb | 0.637 | 0.893 | 4.3 | 1.2 | Log-transformed |
| Bi | 0.753 | 0.975 | 2.5 | 0.31 | Log-transformed |
| Th | 0.804 | 0.890 | 3.2 | −1.0 | Log-transformed |
| U | 0.886 | 0.988 | 1.8 | 0.10 | Log-transformed |
| Coarse Sediment | Fine Sand | Silt and Clay | |
|---|---|---|---|
| Seto Inland Sea | n = 26 | n = 15 | n = 47 |
| Pacific Ocean | n = 75 | n = 107 | n = 193 |
References
- Imai, N.; Terashima, S.; Ohta, A.; Mikoshiba, M.; Okai, T.; Tachibana, Y.; Togashi, S.; Matsuhisa, Y.; Kanai, Y.; Kamioka, H. Geochemical Map of Sea and Land of Japan; Geological Survey of Japan, AIST: Tsukuba, Japan, 2010. [Google Scholar]
- Webb, J.S.; Thornton, I.; Thompson, M.; Howarth, R.J.; Lowenstein, P.L. The Wolfson Geochemical Atlas of England and Wales; Clarendon Press: Oxford, UK, 1978. [Google Scholar]
- Salminen, R.; Batista, M.J.; Bidovec, M.; Demetriades, A.; De Vivo, B.; De Vos, W.; Duris, M.; Gilucis, A.; Gregorauskiene, V.; Halamic, J.; et al. Geochemical Atlas of Europe. Part 1—Background Information, Methodology and Maps; Geological Survey of Finland: Espoo, Finland, 2005. [Google Scholar]
- Caritat, P.D.; Cooper, M. National Geochemical Survey of Australia: Data Quality Assessment; Geoscience Australia Record 2011/21; p. 478. Available online: http://www.ga.gov.au/corporate_data/71971/Rec2011_021_Vol1.pdf (accessed on 17 November 2014).
- Smith, D.B.; Cannon, W.F.; Woodruff, L.G.; Solano, F.; Ellefsen, K.J. Geochemical and Mineralogical Maps for Soils of the Conterminous United States; U.S. Geological Survey Open-File Report 2014-1082. Available online: http://pubs.usgs.gov/of/2014/1082/pdf/ofr2014–1082.pdf (accessed on 19 May 2014).
- Xie, X.J.; Chen, H.X. Global geochemical mapping and its implementation in the Asia-Pacific region. Appl. Geochem. 2001, 16, 1309–1321. [Google Scholar]
- Ohta, A.; Imai, N. Comprehensive survey of multi-elements in coastal sea and stream sediments in the island arc region of Japan: Mass transfer from terrestrial to marine environments. In Advanced Topics in Mass Transfer; El-Amin, M., Ed.; InTech: Rijeka, Croatia, 2011; pp. 373–398. [Google Scholar]
- Ohta, A.; Imai, N.; Terashima, S.; Tachibana, Y.; Ikehara, K.; Okai, T.; Ujiie-Mikoshiba, M.; Kubota, R. Elemental distribution of coastal sea and stream sediments in the island-arc region of Japan and mass transfer processes from terrestrial to marine environments. Appl. Geochem. 2007, 22, 2872–2891. [Google Scholar] [CrossRef]
- Kuwashiro, I. Submarine topography of Japanese Inlandsea Setonaikai. Geogr. Rev. Jpn. 1959, 32, 24–35. [Google Scholar] [CrossRef]
- Yanagi, T. Currents and sediment transport in the Seto Inland Sea, Japan. In Residual Currents and Long-Term Transport; Cheng, R.T., Ed.; Springer: Beilin, Germany, 1990; Volume 38, pp. 348–355. [Google Scholar]
- Sano, S.; Inouchi, Y.; Kanai, Y.; Maruoka, N. Geochemical properties of surface sediments in Seto Inland Sea part 1: Aki-nada surface sediments. Mem. Fac. Educ. Ehime Univ. Ser. III Nat. Sci. 2000, 20, 1–9. [Google Scholar]
- Inouchi, Y. Distribution of bottom sediments in the Seto Inland Sea—The influence of tidal currents on the distribution of bottom sediments. J. Geol. Soc. Jpn. 1982, 88, 665–681. [Google Scholar] [CrossRef]
- Yanagi, T.; Hagita, T.; Saino, T. Episodic outflow of suspended sediments from the Kii channel to the Pacific Ocean in winter. J. Oceanogr. 1994, 50, 99–108. [Google Scholar] [CrossRef]
- Kawana, K.; Tanimoto, T. Turbid bottom water layer and bottom sediment in the Seto Inland Sea. J. Oceanogr. Soc. Jpn. 1984, 40, 175–183. [Google Scholar] [CrossRef]
- Hoshika, A.; Shiozawa, T.; Kawana, K.; Tanimoto, T. Heavy metal pollution in sediment from the Seto Inland Sea, Japan. Mar. Pollut. Bull. 1991, 23, 101–105. [Google Scholar] [CrossRef]
- Akimoto, T.; Kawagoe, S.; Kazama, S. Estimation of sediment yield in Japan by using climate projection model. Proc. Hydraul. Eng. 2009, 53, 655–660. [Google Scholar]
- Geological Map of Japan, 1:1,000,000, 3rd ed.; Geological Survey of Japan, AIST: Tsukuba, Japan, 1992.
- Kato, M. Recent foramininifera in the surface sediments in the inland sea of Japan. Ann. Sci. Coll. Lib. Arts Kanazawa Univ. 1995, 19, 63–73. [Google Scholar]
- Okamura, Y. Geologic structure of the upper continental slope off Shikoku and Quaternary tectonic movement of the outer zone of southwest Japan. J. Geol. Soc. Jpn. 1990, 96, 223–237. [Google Scholar] [CrossRef]
- Fujimoto, M. On the flow types and current stability in Tosa bay and adjacent seas. Sea Sky 1987, 62, 127–140. [Google Scholar]
- Miyata, K.; Sakamoto, H.; Momota, M. Oceanographycal structure in the Tosa bay-1. On the tidal current. Bull. Nansei Natl. Fish. Res. Inst. 1980, 12, 115–124. [Google Scholar]
- Imai, N.; Terashima, S.; Ohta, A.; Mikoshiba, M.; Okai, T.; Tachibana, Y.; Togashi, S.; Matsuhisa, Y.; Kanai, Y.; Kamioka, H.; et al. Geochemical Map of Japan, 1st ed.; Geological Survey of Japan, AIST: Tsukuba, Japan, 2004. [Google Scholar]
- Mikoshiba, M.U.; Imai, N.; Tachibana, Y. Geochemical mapping in Shikoku, southwest Japan. Appl. Geochem. 2011, 26, 1549–1568. [Google Scholar] [CrossRef]
- Arita, M.; Kinoshita, Y. Sedimentological map of cape Muroto. In 1:200,000 Marine Geology Map Series 37; Geological Survey of Japan: Tsukuba, Japan, 1990. [Google Scholar]
- Ikehara, K. Sedimentological map of Tosa Wan. In 1:200,000 Marine Geology Map Series 34; Geological Survey of Japan: Tsukuba, Japan, 1988. [Google Scholar]
- Ikehara, K. Sedimentological map south of Bungo channel. In 1:200,000 Marine Geology Map Series 51; Geological Survey of Japan: Tsukuba, Japan, 1999. [Google Scholar]
- Hoshika, A.; Shiozawa, T. Heavy metals and accumulation rates of sediments in Osaka bay, the Seto Inland Sea, Japan. J. Oceanogr. Soc. Jpn. 1986, 42, 39–52. [Google Scholar] [CrossRef]
- Imai, N. Multielement analysis of rocks with the use of geological certified reference material by inductively coupled plasma mass spectrometry. Anal. Sci. 1990, 6, 389–395. [Google Scholar] [CrossRef]
- Ohta, A.; Imai, N.; Terashima, S.; Tachibana, Y.; Ikehara, K.; Nakajima, T. Geochemical mapping in Hokuriku, Japan: Influence of surface geology, mineral occurrences and mass movement from terrestrial to marine environments. Appl. Geochem. 2004, 19, 1453–1469. [Google Scholar] [CrossRef]
- Reimann, C. Geochemical mapping: Technique or art? Geochem. Explor. Environ. Anal. 2005, 5, 359–370. [Google Scholar] [CrossRef]
- Ohta, A.; Imai, N.; Terashima, S.; Tachibana, Y. Investigation of elemental behaviors in Chugoku region of Japan based on geochemical map utilizing stream sediments. Chikyukagaku (Geochemistry) 2004, 38, 203–222. [Google Scholar]
- Ohta, A.; Imai, N.; Terashima, S.; Tachibana, Y. Application of multi-element statistical analysis for regional geochemical mapping in central Japan. Appl. Geochem. 2005, 20, 1017–1037. [Google Scholar] [CrossRef]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.T. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 2011, 6, 135–147. [Google Scholar] [CrossRef]
- Miller, J.C.; Miller, J.N. Statistics for Analytical Chemistry, 6th ed.; Pearson Education Canada: Don Mills, ON, USA, 2010. [Google Scholar]
- Taylor, S.R.; McLennan, S.M. The geochemical evolution of the continental crust. Rev. Geophys. 1995, 33, 241–265. [Google Scholar] [CrossRef]
- Togashi, S.; Imai, N.; Okuyama-Kusunose, Y.; Tanaka, T.; Okai, T.; Koma, T.; Murata, Y. Young upper crustal chemical composition of the orogenic Japan arc. Geochem. Geophys. Geosyst. 2000, 1, 2000GC000083. [Google Scholar] [CrossRef]
- Aitchison, J. The statistical analysis of compositional data. J. R. Stat. Soc. Ser. B 1982, 44, 139–177. [Google Scholar]
- Pawlowsky-Glahn, V.; Egozcue, J.J. Compositional data and their analysis: An introduction. In Compositional Data Analysis in the Geosciences: From Theory to Practice; Buccianti, A., Mateu-Figueras, G.H., Pawlowsky-Glahn, V., Eds.; Geological Society, Publishing House: Bath, UK, 2006; Volume 264, pp. 1–10. [Google Scholar]
- Ward, J.H., Jr. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 1963, 58, 236–244. [Google Scholar] [CrossRef]
- Goldich, S.S. A study in rock-weathering. J. Geol. 1938, 46, 17–58. [Google Scholar] [CrossRef]
- Ohta, A.; Imai, N.; Terashima, S.; Tachibana, Y.; Ikehara, K.; Katayama, H. Elemental distribution of surface sediments around Oki trough including adjacent terrestrial area: Strong impact of Japan Sea Proper Water on silty and clayey sediments. Bull. Geol. Surv. Jpn. 2015, 66, 81–101. [Google Scholar] [CrossRef]
- Mahalanobis, P.C. On the generalized distance in statistics. Proc. Natl. Inst. Sci. 1936, 2, 49–55. [Google Scholar]
- Minakawa, T.; Funakoshi, N.; Morioka, H. Chemical properties of allanite from the Ryoke and Hiroshima granite pegmatites in Shikoku, Japan. Men. Fac. Sci. Ehime Univ. 2001, 7, 1–13. [Google Scholar]
- Nozaki, T.; Nakamura, K.; Osawa, H.; Fujinaga, K.; Kato, Y. Geochemical features and tectonic setting of greenstones from Kunimiyama, northern Chichibu belt, central Shikoku, Japan. Resour. Geol. 2005, 55, 301–310. [Google Scholar] [CrossRef]
- Hoshino, M. On the muddy sediments of the continental shelf adjacent to Japan. J. Geol. Soc. Jpn. 1952, 58, 41–53. [Google Scholar]
- Hoshika, A.; Shiozawa, T. Sedimentation rates and heavy metal pollution of sediments in the Seto Inland Sea. Part 3. Hiuchi-nada. J. Oceanogr. Soc. Jpn. 1984, 40, 334–342. [Google Scholar] [CrossRef]
- Hoshika, A.; Tanimoto, T.; Mishima, Y. Sedimentation processes of particulate matter in the Osaka bay. Oceanogr. Jpn. 1994, 3, 419–425. [Google Scholar] [CrossRef]
- Manabe, T. Distribution of sediments contamination in the eastern Seto Inland Sea. Sea Sky 1991, 67, 1–9. [Google Scholar]
- Hoshika, A.; Shiozawa, T. Sedimentation rates and heavy metal pollution of sediments in the Seto Inland Sea. Part 4. Suo-nada. J. Oceanogr. Soc. Jpn. 1985, 41, 283–290. [Google Scholar] [CrossRef]
- Hudson-Edwards, K.A.; Macklin, M.G.; Curtis, C.D.; Vaughan, D.J. Processes of formation and distribution of Pb-, Zn-, Cd-, and Cu-bearing minerals in the Tyne basin, northeast England: Implications for metal-contaminated river systems. Environ. Sci. Technol. 1996, 30, 72–80. [Google Scholar] [CrossRef]
- Rosenthal, Y.; Lam, P.; Boyle, E.A.; Thomson, J. Authigenic cadmium enrichments in suboxic sediments: Precipitation and postdepositional mobility. Earth Planet. Sci. Lett. 1995, 132, 99–111. [Google Scholar] [CrossRef]
- Mason, R.P.; Fitzgerald, W.F.; Morel, F.M.M. The biogeochemical cycling of elemental mercury: Anthropogenic influences. Geochim. Cosmochim. Acta 1994, 58, 3191–3198. [Google Scholar] [CrossRef]
- Shaw, T.J.; Gieskes, J.M.; Jahnke, R.A. Early diagenesis in differing depositional environments: The response of transition metals in pore water. Geochim. Cosmochim. Acta 1990, 54, 1233–1246. [Google Scholar] [CrossRef]
- Klinkhammer, G.P. Early diagenesis in sediments from the eastern equatorial pacific. II. Pore water metal results. Earth Planet. Sci. Lett. 1980, 49, 81–101. [Google Scholar] [CrossRef]
- Wilson, T.R.S.; Thomson, J.; Colley, S.; Hydes, D.J.; Higgs, N.C.; Sorensen, J. Early organic diagenesis: The significance of progressive subsurface oxidation fronts in pelagic sediments. Geochim. Cosmochim. Acta 1985, 49, 811–822. [Google Scholar] [CrossRef]
- Ministry of Land, Infrastructure, Transport and Tourism. Available online: http://www.mlit.go.jp/river/toukei_chousa/ (accessed on 19 November 2015).
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Royston, P. A remark on algorithm as181: The w-test for normality. Appl. Stat. J. R. Stat. Soc. 1995, 44, 547–551. [Google Scholar]
- Shaw, R.G.; Mitchell-Olds, T. ANOVA for unbalanced data: An overview. Ecology 1993, 74, 1638–1645. [Google Scholar] [CrossRef]
- Littell, R.C.; Stroup, W.W.; Freund, R. SAS for Linear Models; SAS Institute: Cary, NC, USA, 2002. [Google Scholar]

| Region | Discharged Area | |||
|---|---|---|---|---|
| Pacific Ocean | Kii or Bungo Channel | Seto Inland Sea | Inner Bay | |
| Chugoku region | 0% | 0% | 15% | 4% to Hiroshima Bay |
| Kinki region | 0% | 5% to Kii Channel | 5% | 19% to Osaka Bay |
| Shikoku region | 22% | 18% to Kii Channel | 5% | 0% |
| Kyushu region | 0% | 1% to Bungo Channel | 1% | 5% to Beppu Bay |
| Element | Marine Sediment | Stream Sediment | ||||||
|---|---|---|---|---|---|---|---|---|
| Min | Mean | Median | Max | Min | Mean | Median | Max | |
| wt % | ||||||||
| Na2O | 0.91 | 2.97 | 2.82 | 6.35 | 0.46 | 2.10 | 2.04 | 4.42 |
| MgO | 0.49 | 2.77 | 2.68 | 6.71 | 0.19 | 2.10 | 1.65 | 9.79 |
| Al2O3 | 2.01 | 9.49 | 9.60 | 16.62 | 3.91 | 11.39 | 11.52 | 17.94 |
| P2O5 | 0.025 | 0.113 | 0.108 | 2.56 | 0.021 | 0.125 | 0.109 | 1.22 |
| K2O | 0.53 | 2.03 | 2.06 | 3.12 | 0.66 | 2.18 | 2.24 | 3.79 |
| CaO | 0.33 | 6.51 | 5.22 | 34.1 | 0.12 | 1.83 | 1.41 | 8.40 |
| TiO2 | 0.087 | 0.501 | 0.492 | 2.15 | 0.14 | 0.71 | 0.60 | 3.28 |
| MnO | 0.012 | 0.082 | 0.066 | 0.451 | 0.014 | 0.128 | 0.116 | 1.10 |
| T-Fe2O3 | 0.73 | 4.91 | 4.38 | 23.7 | 1.41 | 5.24 | 4.67 | 15.8 |
| mg/kg | ||||||||
| Li | 7.58 | 51.9 | 50.0 | 136 | 9.33 | 38.5 | 38.3 | 107 |
| Be | 0.32 | 1.41 | 1.44 | 2.25 | 0.71 | 1.77 | 1.67 | 4.75 |
| Sc | 1.77 | 9.82 | 9.69 | 35.1 | 2.31 | 11.6 | 9.57 | 46.8 |
| V | 11.7 | 84.8 | 80.1 | 680 | 10.9 | 103 | 83.4 | 420 |
| Cr | 6.50 | 58.0 | 59.0 | 172 | 4.47 | 84.9 | 52.8 | 884 |
| Co | 1.76 | 10.9 | 9.90 | 41.2 | 1.79 | 13.8 | 11.5 | 60.7 |
| Ni | 1.35 | 28.2 | 28.8 | 138 | 1.38 | 34.3 | 21.7 | 349 |
| Cu | 2.42 | 20.5 | 18.4 | 86.6 | 5.66 | 49.7 | 31.5 | 2599 |
| Zn | 16.1 | 88.1 | 80.9 | 347 | 17.1 | 165 | 118 | 11,444 |
| Ga | 2.77 | 13.5 | 14.2 | 19.0 | 6.97 | 17.4 | 17.2 | 31.7 |
| As | 0.2 | 6.3 | 5.0 | 68 | 1.0 | 18.2 | 8.1 | 1578 |
| Rb | 17.7 | 73.2 | 74.9 | 114 | 12.3 | 105 | 103 | 265 |
| Sr | 70.2 | 326 | 259 | 2525 | 23.4 | 133 | 111 | 1321 |
| Y | 4.95 | 12.9 | 12.6 | 28.8 | 2.24 | 19.6 | 18.7 | 63.6 |
| Nb | 1.59 | 6.79 | 6.80 | 13.9 | 2.88 | 9.75 | 8.86 | 33.3 |
| Mo | 0.14 | 0.76 | 0.63 | 14.8 | 0.25 | 1.34 | 0.99 | 27.4 |
| Cd | 0.018 | 0.101 | 0.081 | 0.480 | 0.019 | 0.29 | 0.16 | 28.7 |
| Sn | 0.37 | 2.15 | 1.98 | 18.9 | 0.87 | 5.05 | 3.47 | 170 |
| Sb | 0.092 | 0.59 | 0.55 | 1.89 | 0.099 | 0.98 | 0.74 | 17.2 |
| Cs | 0.60 | 4.06 | 4.25 | 7.65 | 0.41 | 5.63 | 5.03 | 33.6 |
| As | 0.2 | 6.3 | 5.0 | 68 | 1.0 | 18.2 | 8.1 | 1578 |
| Ba | 67.4 | 340 | 340 | 1649 | 136 | 429 | 431 | 855 |
| La | 5.31 | 16.0 | 16.5 | 24.7 | 4.73 | 21.2 | 19.9 | 69.2 |
| Ce | 12.8 | 33.8 | 34.6 | 58.7 | 7.20 | 39.2 | 36.9 | 170 |
| Pr | 1.28 | 3.78 | 3.87 | 5.75 | 1.06 | 4.90 | 4.63 | 20.8 |
| Nd | 5.22 | 15.0 | 15.3 | 22.2 | 4.08 | 19.2 | 18.2 | 79.9 |
| Sm | 1.02 | 3.02 | 3.09 | 4.59 | 0.78 | 3.94 | 3.71 | 17.4 |
| Eu | 0.29 | 0.68 | 0.68 | 1.15 | 0.18 | 0.81 | 0.78 | 1.72 |
| Gd | 0.92 | 2.72 | 2.74 | 4.44 | 0.61 | 3.63 | 3.39 | 12.9 |
| Tb | 0.16 | 0.45 | 0.45 | 0.78 | 0.10 | 0.63 | 0.59 | 2.02 |
| Dy | 0.80 | 2.23 | 2.20 | 4.07 | 0.51 | 3.21 | 3.03 | 10.3 |
| Ho | 0.16 | 0.42 | 0.42 | 0.80 | 0.091 | 0.62 | 0.59 | 2.01 |
| Er | 0.47 | 1.23 | 1.19 | 2.34 | 0.26 | 1.83 | 1.76 | 6.29 |
| Tm | 0.069 | 0.19 | 0.19 | 0.36 | 0.036 | 0.29 | 0.28 | 1.05 |
| Yb | 0.41 | 1.18 | 1.15 | 2.20 | 0.21 | 1.82 | 1.74 | 6.88 |
| Lu | 0.051 | 0.17 | 0.16 | 0.31 | 0.032 | 0.26 | 0.25 | 1.03 |
| Ta | 0.13 | 0.61 | 0.60 | 1.23 | 0.28 | 0.85 | 0.75 | 5.23 |
| Hg | 0.0005 | 0.139 | 0.138 | 5.19 | 0.010 | 0.117 | 0.060 | 4.60 |
| Tl | 0.068 | 0.48 | 0.49 | 1.00 | 0.075 | 0.64 | 0.61 | 2.52 |
| Pb | 5.00 | 20.8 | 18.6 | 126 | 7.19 | 43.8 | 27.3 | 4177 |
| Bi | 0.036 | 0.32 | 0.28 | 1.43 | 0.033 | 0.52 | 0.30 | 32.1 |
| Th | 1.50 | 5.81 | 6.04 | 26.6 | 1.52 | 9.53 | 7.30 | 259 |
| U | 0.28 | 1.35 | 1.26 | 4.51 | 0.53 | 2.13 | 1.70 | 31.9 |
| Element | Seto Inland Sea | Pacific Ocean | ||||||
|---|---|---|---|---|---|---|---|---|
| Stream Sed. | Marine Sed. | Stream Sed. | Marine Sed. | |||||
| Coarse Sed. | Fine Sand | Silt and Clay | Coarse Sed. | Fine Sand | Silt and Clay | |||
| n = 321 | n = 26 | n = 15 | n = 47 | n = 159 | n = 75 | n = 107 | n = 193 | |
| wt % | ||||||||
| MgO | 1.72 | 1.45 | 2.76 | 3.31 | 1.80 | 3.12 | 2.39 | 2.68 |
| Al2O3 | 11.43 | 7.34 | 8.47 | 8.48 | 12.17 | 9.29 | 9.23 | 10.50 |
| P2O5 | 0.115 | 0.059 | 0.114 | 0.129 | 0.105 | 0.105 | 0.095 | 0.111 |
| K2O | 2.25 | 2.26 | 2.02 | 2.03 | 2.20 | 1.80 | 1.95 | 2.18 |
| CaO | 1.67 | 4.83 | 5.21 | 2.01 | 0.77 | 7.41 | 6.93 | 5.23 |
| TiO2 | 0.64 | 0.25 | 0.43 | 0.54 | 0.54 | 0.49 | 0.42 | 0.51 |
| MnO | 0.119 | 0.086 | 0.079 | 0.109 | 0.096 | 0.100 | 0.060 | 0.056 |
| T-Fe2O3 | 4.88 | 2.39 | 3.54 | 4.59 | 4.45 | 5.13 | 4.50 | 4.29 |
| mg/kg | ||||||||
| Li | 35 | 24 | 55 | 102 | 42 | 29 | 42 | 53 |
| Be | 1.7 | 1.2 | 1.4 | 1.7 | 1.6 | 1.2 | 1.4 | 1.5 |
| Sc | 10 | 4.9 | 8.4 | 9.3 | 9.8 | 10 | 8.3 | 10 |
| V | 78 | 37 | 57 | 75 | 94 | 82 | 69 | 90 |
| Cr | 51 | 25 | 40 | 74 | 60 | 39 | 47 | 68 |
| Co | 12 | 6.7 | 9.3 | 13 | 12 | 12 | 10 | 9.5 |
| Ni | 19 | 11 | 17 | 32 | 27 | 16 | 22 | 36 |
| Cu | 31 | 7.9 | 13 | 41 | 36 | 7.2 | 13 | 28 |
| Zn | 126 | 45 | 83 | 166 | 96 | 71 | 73 | 85 |
| Ga | 17 | 11 | 14 | 16 | 17 | 12 | 13 | 15 |
| As | 8.0 | 4.9 | 5.9 | 7.0 | 7.1 | 8.3 | 4.3 | 4.2 |
| Rb | 107 | 83 | 74 | 60 | 95 | 62 | 76 | 80 |
| Sr | 113 | 300 | 231 | 121 | 112 | 439 | 319 | 242 |
| Y | 21 | 9.6 | 15 | 15 | 14 | 12 | 11 | 13 |
| Nb | 9.1 | 4.0 | 6.3 | 9.4 | 8.5 | 4.4 | 5.7 | 7.9 |
| Mo | 0.98 | 0.47 | 0.74 | 1.1 | 0.96 | 0.46 | 0.47 | 0.68 |
| Cd | 0.18 | 0.041 | 0.079 | 0.19 | 0.10 | 0.054 | 0.050 | 0.13 |
| mg/kg | ||||||||
| Sn | 3.9 | 1.3 | 2.4 | 4.3 | 2.7 | 1.2 | 1.6 | 2.2 |
| Sb | 0.74 | 0.30 | 0.39 | 0.55 | 0.71 | 0.43 | 0.44 | 0.76 |
| Cs | 5.1 | 2.0 | 3.8 | 5.4 | 4.6 | 2.2 | 3.5 | 5.1 |
| Ba | 433 | 344 | 274 | 247 | 438 | 300 | 323 | 386 |
| La | 20 | 12 | 14 | 17 | 20 | 13 | 16 | 18 |
| Ce | 37 | 25 | 32 | 33 | 39 | 27 | 34 | 37 |
| Pr | 4.8 | 2.7 | 3.7 | 4.2 | 4.6 | 3.2 | 3.7 | 4.1 |
| Nd | 19 | 11 | 15 | 16 | 18 | 13 | 15 | 16 |
| Sm | 3.9 | 2.1 | 3.2 | 3.5 | 3.6 | 2.7 | 2.9 | 3.2 |
| Eu | 0.83 | 0.60 | 0.71 | 0.67 | 0.77 | 0.73 | 0.64 | 0.70 |
| Gd | 3.6 | 1.9 | 2.9 | 3.3 | 3.2 | 2.5 | 2.5 | 2.8 |
| Tb | 0.63 | 0.31 | 0.51 | 0.56 | 0.51 | 0.40 | 0.40 | 0.45 |
| Dy | 3.3 | 1.6 | 2.6 | 2.9 | 2.6 | 2.0 | 1.9 | 2.2 |
| Ho | 0.64 | 0.31 | 0.49 | 0.54 | 0.48 | 0.38 | 0.35 | 0.42 |
| Er | 1.9 | 0.9 | 1.4 | 1.6 | 1.3 | 1.1 | 1.0 | 1.2 |
| Tm | 0.31 | 0.15 | 0.22 | 0.26 | 0.21 | 0.17 | 0.16 | 0.19 |
| Yb | 1.9 | 0.9 | 1.4 | 1.5 | 1.3 | 1.0 | 1.0 | 1.2 |
| Lu | 0.28 | 0.14 | 0.20 | 0.22 | 0.18 | 0.15 | 0.14 | 0.16 |
| Ta | 0.75 | 0.40 | 0.60 | 0.92 | 0.76 | 0.38 | 0.51 | 0.73 |
| Hg | 0.050 | 0.030 | 0.079 | 0.167 | 0.110 | 0.026 | 0.100 | 0.179 |
| Tl | 0.62 | 0.56 | 0.60 | 0.68 | 0.55 | 0.34 | 0.41 | 0.51 |
| Pb | 28 | 20 | 24 | 39 | 23 | 17 | 17 | 19 |
| Bi | 0.31 | 0.18 | 0.32 | 0.77 | 0.26 | 0.18 | 0.19 | 0.34 |
| Th | 7.9 | 3.7 | 5.9 | 7.0 | 6.6 | 3.7 | 5.4 | 6.6 |
| U | 1.9 | 0.8 | 1.5 | 1.8 | 1.4 | 0.9 | 1.1 | 1.4 |
| Element | Two-Way ANOVA | MF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| F (A) | F (B) | F (A × B) | p (A) | p (B) | p (A × B) | η2 (A) | η2 (B) | η2 (A × B) | ||
| MgO | 2 | 13 | 67 | 0.20 | <0.01 | <0.01 | <0.01 | 0.04 | 0.22 | A × B |
| Al2O3 | 74 | 34 | 2 | <0.01 | <0.01 | 0.13 | 0.12 | 0.11 | <0.01 | Both |
| P2O5 | 10 | 19 | 28 | <0.01 | <0.01 | <0.01 | 0.02 | 0.07 | 0.10 | A × B |
| K2O | 1 | 46 | 31 | 0.36 | <0.01 | <0.01 | <0.01 | 0.15 | 0.10 | B |
| CaO | 110 | 34 | 6 | <0.01 | <0.01 | <0.01 | 0.17 | 0.11 | 0.02 | A |
| TiO2 | 14 | 26 | 19 | <0.01 | <0.01 | <0.01 | 0.02 | 0.09 | 0.07 | B |
| MnO | 53 | 26 | 17 | <0.01 | <0.01 | <0.01 | 0.09 | 0.09 | 0.06 | Both |
| T-Fe2O3 | 37 | 1 | 38 | <0.01 | 0.60 | <0.01 | 0.06 | <0.01 | 0.13 | A × B |
| Li | 98 | 366 | 59 | <0.01 | <0.01 | <0.01 | 0.07 | 0.53 | 0.09 | B |
| Be | 44 | 95 | 13 | <0.01 | <0.01 | <0.01 | 0.06 | 0.27 | 0.04 | B |
| Sc | 33 | 13 | 26 | <0.01 | <0.01 | <0.01 | 0.06 | 0.05 | 0.09 | A × B |
| V | 77 | 18 | 23 | <0.01 | <0.01 | <0.01 | 0.13 | 0.06 | 0.08 | A |
| Cr | 2 | 149 | 21 | 0.16 | <0.01 | <0.01 | <0.01 | 0.37 | 0.05 | B |
| Co | 0 | 0 | 50 | 0.89 | 0.83 | <0.01 | <0.01 | <0.01 | 0.18 | A × B |
| Ni | 29 | 280 | 15 | <0.01 | <0.01 | <0.01 | 0.03 | 0.52 | 0.03 | B |
| Cu | 30 | 595 | 17 | <0.01 | <0.01 | <0.01 | 0.02 | 0.70 | 0.02 | B |
| Zn | 78 | 104 | 115 | <0.01 | <0.01 | <0.01 | 0.08 | 0.22 | 0.24 | A × B |
| Ga | 21 | 191 | 12 | <0.01 | <0.01 | <0.01 | 0.02 | 0.43 | 0.03 | B |
| As | 11 | 28 | 16 | <0.01 | <0.01 | <0.01 | 0.02 | 0.10 | 0.06 | B |
| Rb | 5 | 22 | 26 | 0.02 | <0.01 | <0.01 | <0.01 | 0.08 | 0.09 | A × B |
| Sr | 120 | 113 | 5 | <0.01 | <0.01 | <0.01 | 0.15 | 0.28 | 0.01 | B |
| Y | 19 | 25 | 20 | <0.01 | <0.01 | <0.01 | 0.03 | 0.09 | 0.07 | B |
| Nb | 22 | 152 | 12 | <0.01 | <0.01 | <0.01 | 0.03 | 0.38 | 0.03 | B |
| Mo | 20 | 16 | 8 | <0.01 | <0.01 | <0.01 | 0.04 | 0.06 | 0.03 | B |
| Cd | 20 | 124 | 16 | <0.01 | <0.01 | <0.01 | 0.03 | 0.33 | 0.04 | B |
| Sn | 180 | 239 | 42 | <0.01 | <0.01 | <0.01 | 0.15 | 0.41 | 0.07 | B |
| Sb | 44 | 106 | 3 | <0.01 | <0.01 | 0.03 | 0.06 | 0.29 | <0.01 | B |
| Cs | 2 | 467 | 5 | 0.14 | <0.01 | <0.01 | <0.01 | 0.67 | <0.01 | B |
| Ba | 29 | 34 | 58 | <0.01 | <0.01 | <0.01 | 0.04 | 0.10 | 0.17 | A × B |
| La | 3 | 116 | 5 | 0.07 | <0.01 | <0.01 | <0.01 | 0.33 | 0.01 | B |
| Ce | 24 | 89 | 0 | <0.01 | <0.01 | 0.86 | 0.04 | 0.27 | <0.01 | B |
| Pr | 2 | 128 | 9 | 0.19 | <0.01 | <0.01 | <0.01 | 0.35 | 0.02 | B |
| Nd | 1 | 105 | 12 | 0.26 | <0.01 | <0.01 | <0.01 | 0.30 | 0.03 | B |
| Sm | 2 | 80 | 21 | 0.13 | <0.01 | <0.01 | <0.01 | 0.24 | 0.06 | B |
| Eu | 4 | 8 | 6 | 0.07 | <0.01 | <0.01 | <0.01 | 0.03 | 0.03 | None |
| Gd | 13 | 50 | 27 | <0.01 | <0.01 | <0.01 | 0.02 | 0.16 | 0.09 | B |
| Tb | 38 | 43 | 32 | <0.01 | <0.01 | <0.01 | 0.06 | 0.13 | 0.10 | B |
| Dy | 25 | 44 | 30 | <0.01 | <0.01 | <0.01 | 0.04 | 0.14 | 0.09 | B |
| Ho | 30 | 39 | 25 | <0.01 | <0.01 | <0.01 | 0.05 | 0.13 | 0.08 | B |
| Er | 38 | 36 | 20 | <0.01 | <0.01 | <0.01 | 0.06 | 0.12 | 0.07 | B |
| Tm | 44 | 37 | 18 | <0.01 | <0.01 | <0.01 | 0.07 | 0.12 | 0.06 | B |
| Yb | 42 | 36 | 16 | <0.01 | <0.01 | <0.01 | 0.07 | 0.12 | 0.05 | B |
| Lu | 58 | 26 | 18 | <0.01 | <0.01 | <0.01 | 0.10 | 0.09 | 0.06 | A |
| Ta | 68 | 163 | 19 | <0.01 | <0.01 | <0.01 | 0.08 | 0.37 | 0.04 | B |
| Hg | 0 | 270 | 2 | 0.85 | <0.01 | 0.14 | <0.01 | 0.54 | <0.01 | B |
| Tl | 241 | 107 | 1 | <0.01 | <0.01 | 0.59 | 0.27 | 0.24 | <0.01 | Both |
| Pb | 359 | 58 | 36 | <0.01 | <0.01 | <0.01 | 0.36 | 0.11 | 0.07 | A |
| Bi | 165 | 214 | 57 | <0.01 | <0.01 | <0.01 | 0.14 | 0.37 | 0.10 | B |
| Th | 5 | 206 | 2 | 0.03 | <0.01 | 0.14 | <0.01 | 0.47 | <0.01 | B |
| U | 9 | 114 | 11 | <0.01 | <0.01 | <0.01 | 0.01 | 0.32 | 0.03 | B |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohta, A.; Imai, N.; Tachibana, Y.; Ikehara, K. Statistical Analysis of the Spatial Distribution of Multi-Elements in an Island Arc Region: Complicating Factors and Transfer by Water Currents. Water 2017, 9, 37. https://doi.org/10.3390/w9010037
Ohta A, Imai N, Tachibana Y, Ikehara K. Statistical Analysis of the Spatial Distribution of Multi-Elements in an Island Arc Region: Complicating Factors and Transfer by Water Currents. Water. 2017; 9(1):37. https://doi.org/10.3390/w9010037
Chicago/Turabian StyleOhta, Atsuyuki, Noboru Imai, Yoshiko Tachibana, and Ken Ikehara. 2017. "Statistical Analysis of the Spatial Distribution of Multi-Elements in an Island Arc Region: Complicating Factors and Transfer by Water Currents" Water 9, no. 1: 37. https://doi.org/10.3390/w9010037
APA StyleOhta, A., Imai, N., Tachibana, Y., & Ikehara, K. (2017). Statistical Analysis of the Spatial Distribution of Multi-Elements in an Island Arc Region: Complicating Factors and Transfer by Water Currents. Water, 9(1), 37. https://doi.org/10.3390/w9010037






