Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers
Abstract
:1. Introduction
2. Materials and Methods
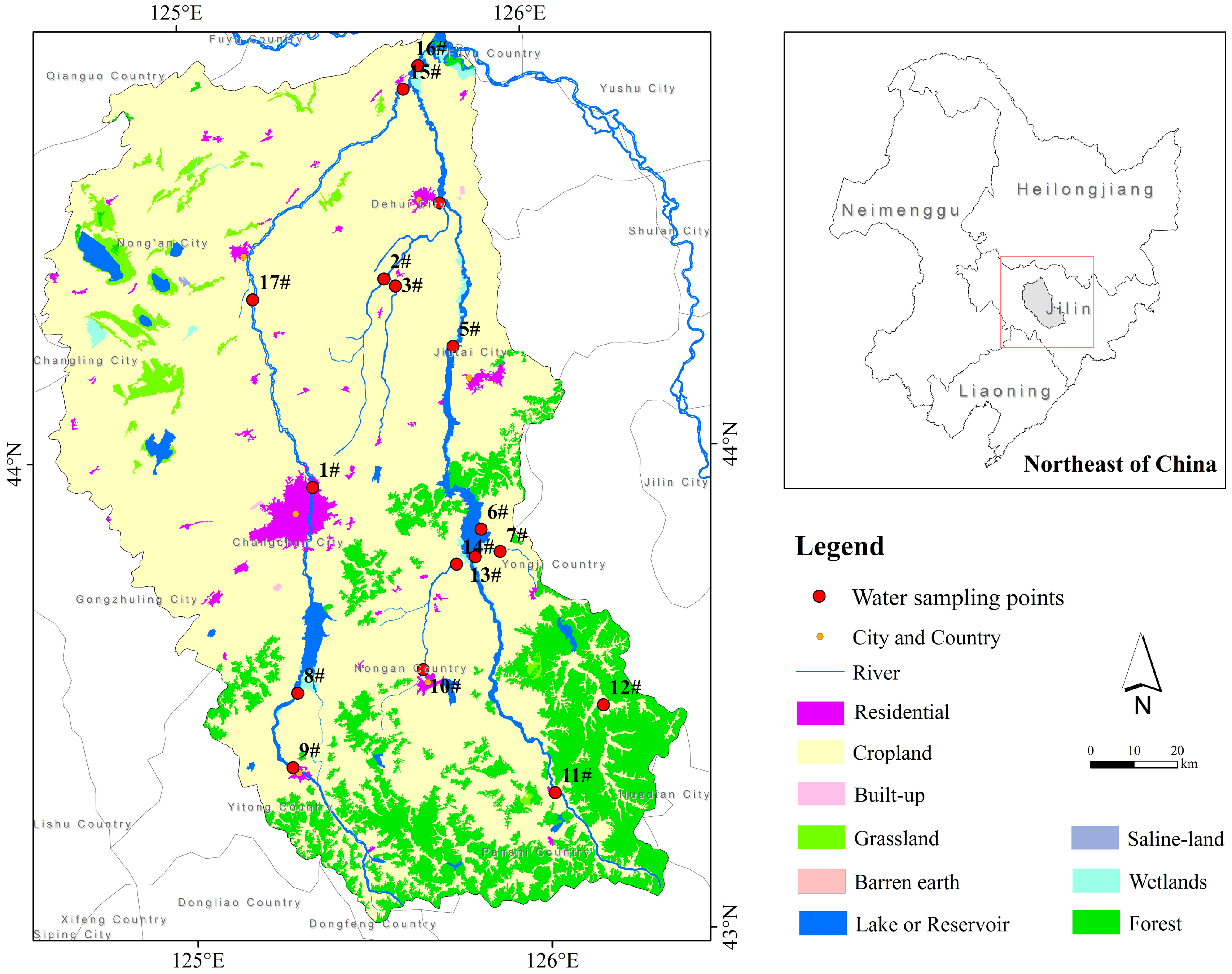
2.1. Study Area
2.2. In Situ Data Collection and Processing
2.3. Measurement of Absorbance and Processing
2.4. Water Quality Measurement and Evaluation
2.5. Paramaterization and Statistical Analysis
3. Results
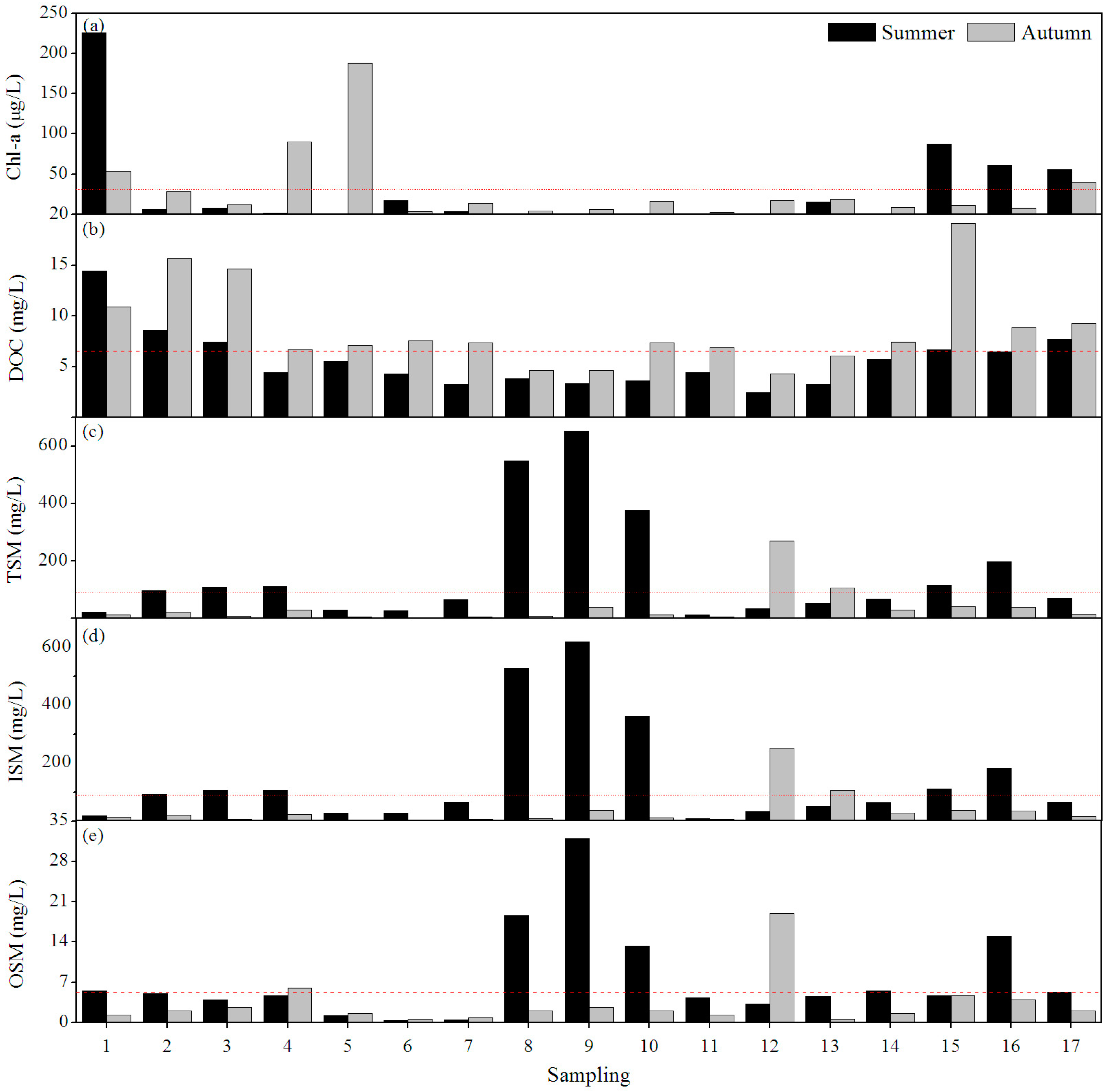
3.1. Pollution Status
3.2. Water Quality
3.2.1. Seasonal Variation
3.2.2. Spatial Variation
3.3. Spectral Characteristics of CDOM Absorption
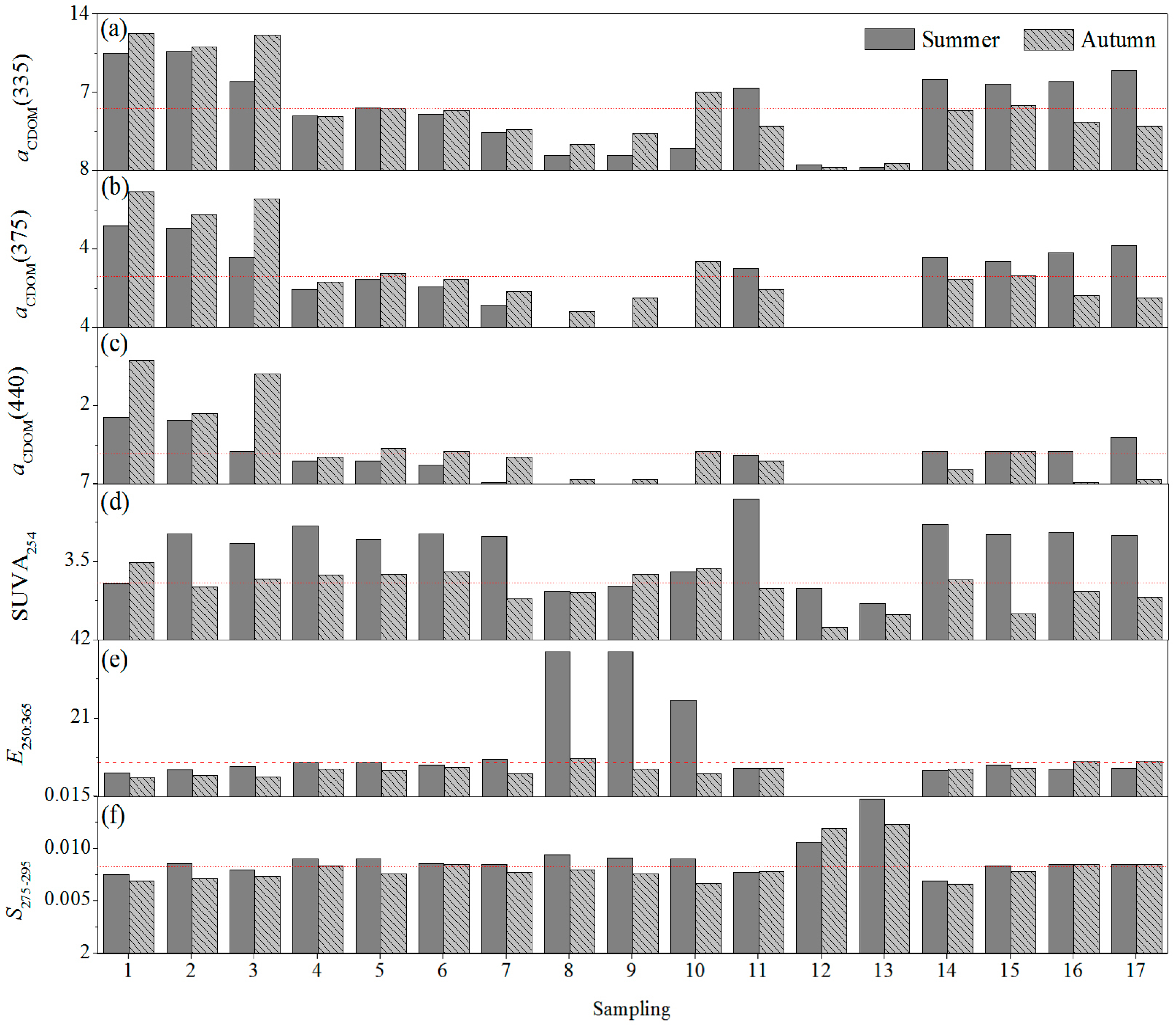
3.3.1. Seasonal Variation
3.3.2. Spatial Variation
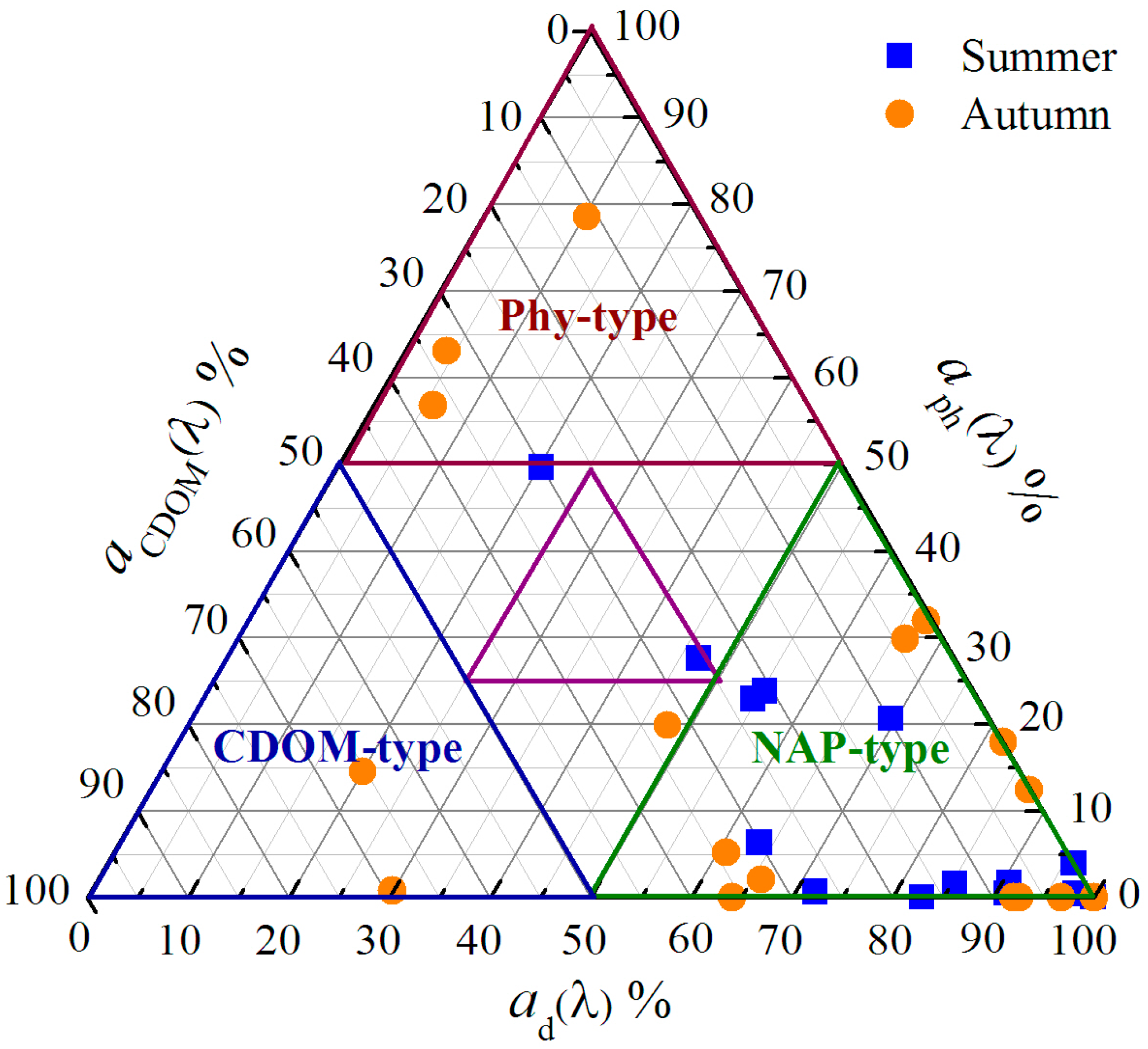
3.3.3. Relative Contributions of CDOM Absorption
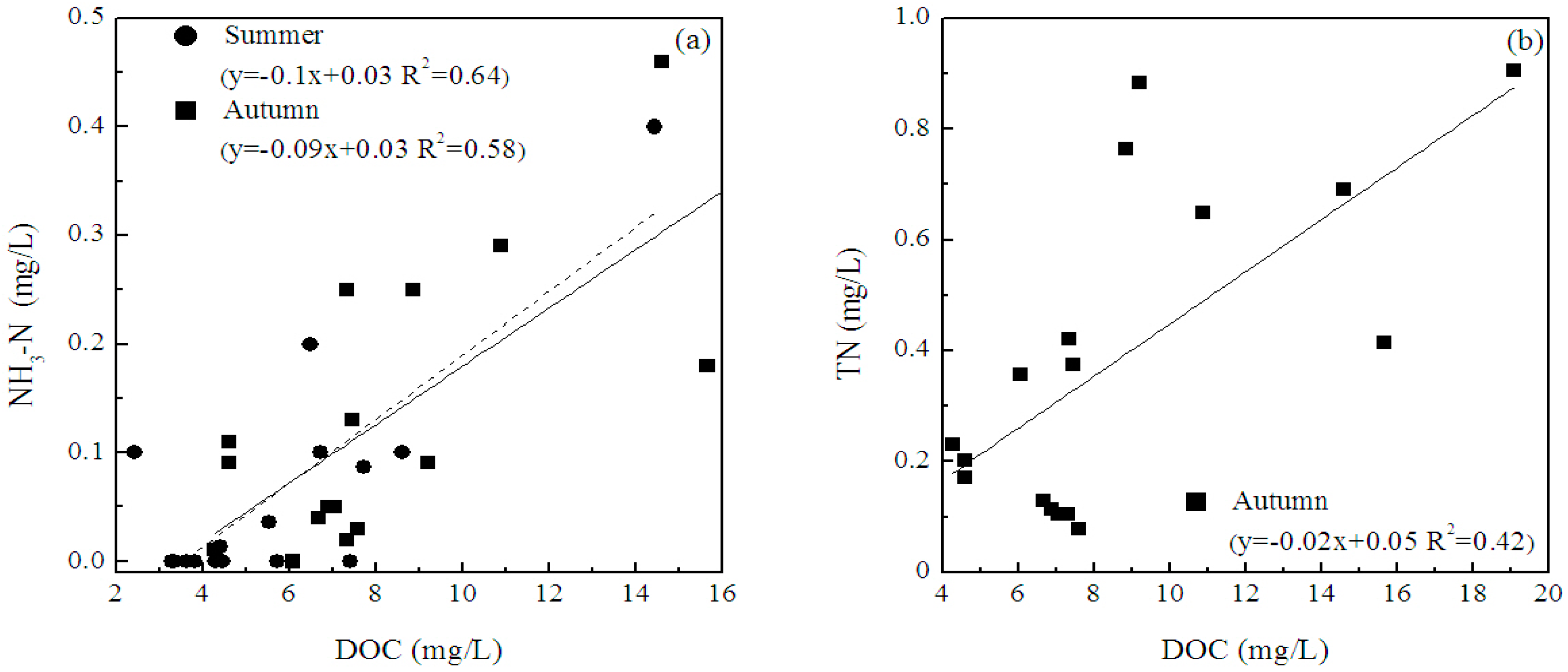
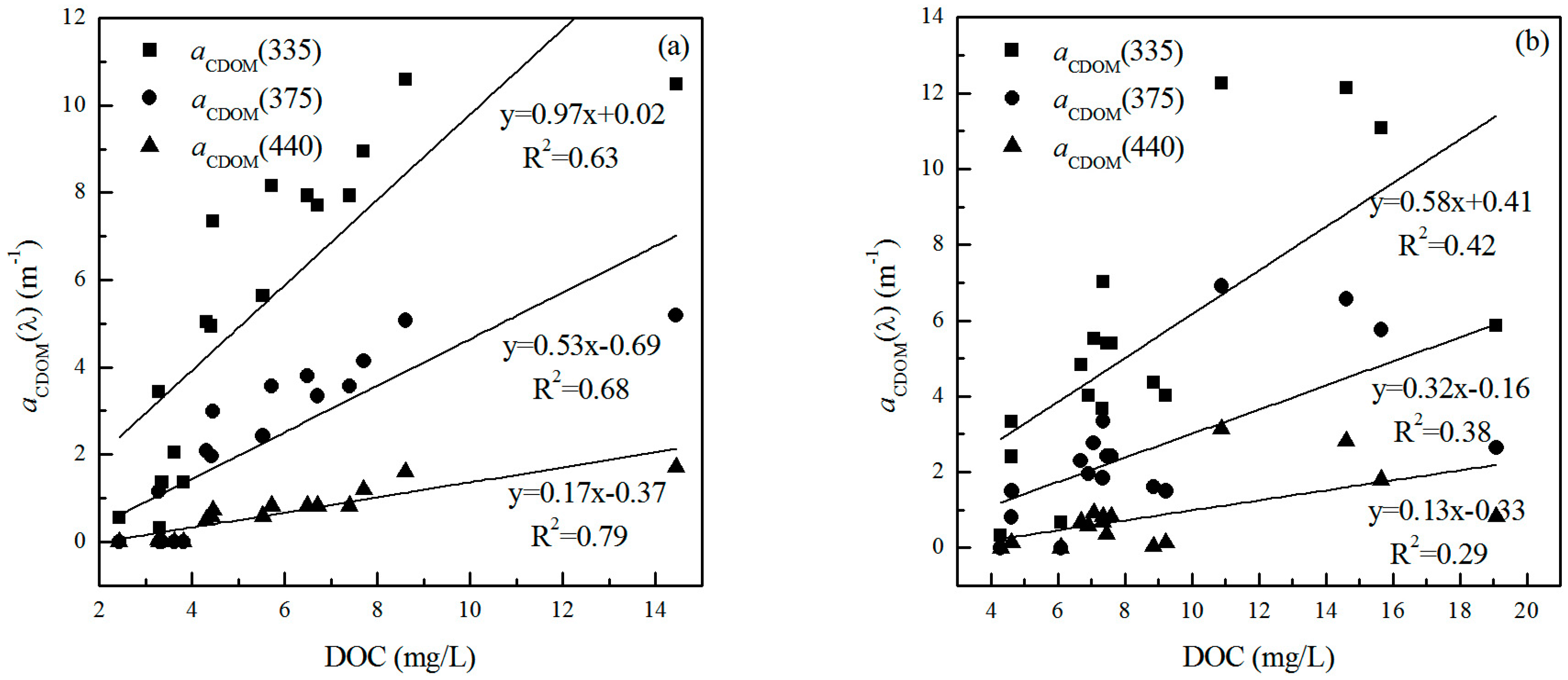
3.4. Relationship between DOC and CDOM
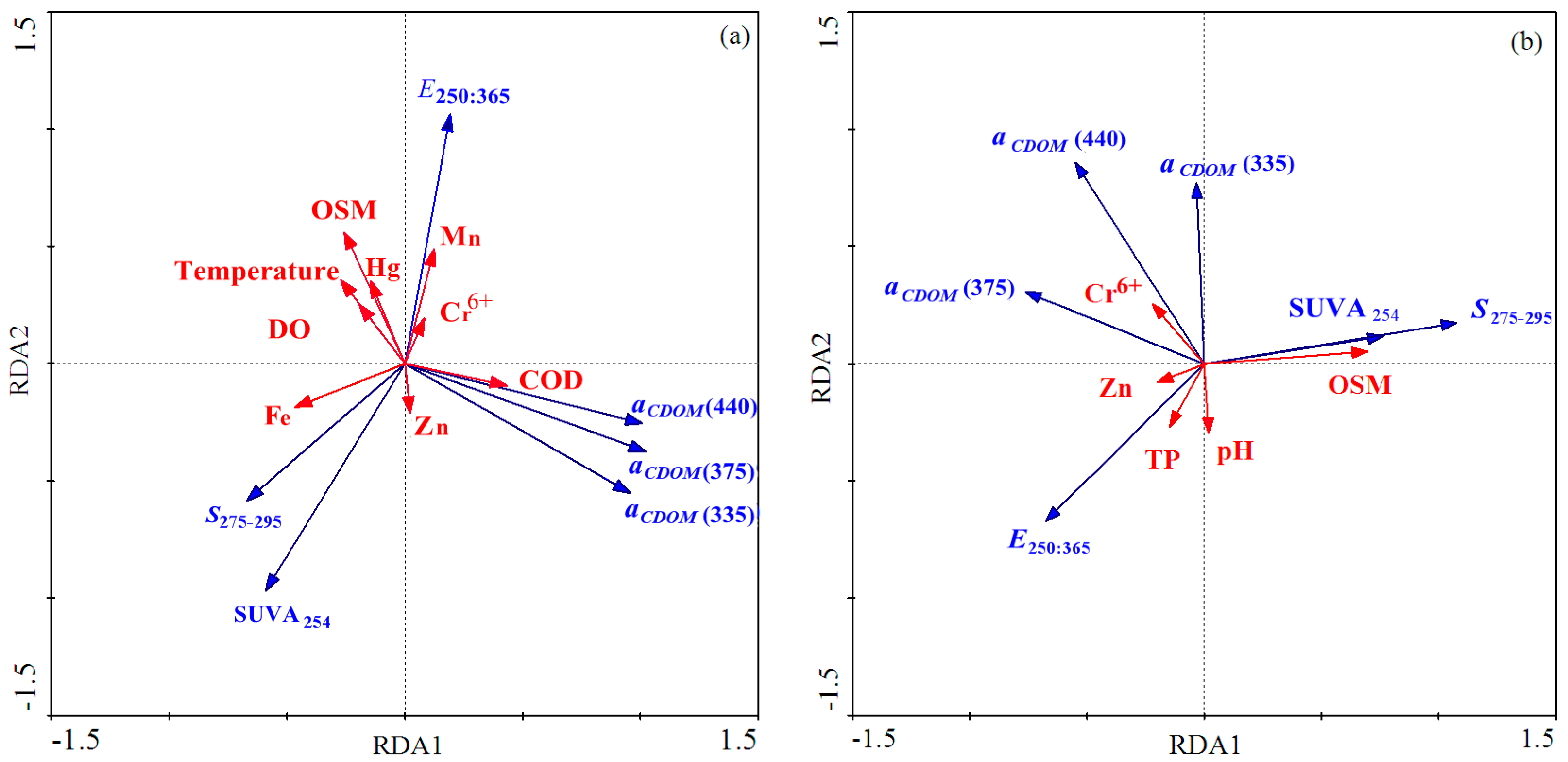
3.5. Correlations between Environmental Factors and CDOM Absorption
4. Discussion
4.1. Dissolved Organic Carbon in Riverine Waters
4.2. Analysis of CDOM Spectral Characteristics
4.3. CDOM-DOC Relationship and Environmental Factors
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Coble, P.G. Characterization of marine and terrestrial DOM in seawater using excitation-emission matrix spectroscopy. Mar. Chem. 1996, 51, 325–346. [Google Scholar] [CrossRef]
- Gao, J.; Yang, H.; Li, B. Investigating the Roles of Dissolved Organic Matter on Arsenic Mobilization and Speciation in Environmental Water. CLEAN Soil Air Water 2016, 44, 818–828. [Google Scholar] [CrossRef]
- Hosen, J.D.; McDonough, O.T.; Febria, C.M.; Palmer, M.A. Dissolved organic matter quality and bioavailability changes across an urbanization gradient in headwater streams. Environ. Sci. Technol. 2014, 48, 7817–7824. [Google Scholar] [CrossRef] [PubMed]
- Heinz, M.; Graeber, D.; Zak, D.; Zwirnmann, E.; Gelbrecht, J.; Pusch, M.T. Comparison of organic matter composition in agricultural versus forest affected headwaters with special emphasis on organic nitrogen. Environ. Sci. Technol. 2015, 49, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Wetzel, R.G. Limnology: Lake and River Ecosystems; Academic Press: Cambridge, MA, USA, 2001. [Google Scholar]
- D’Sa, E.J.; Miller, R.L.; Del Castillo, C. Bio-optical properties and ocean color algorithms for coastal waters influenced by the Mississippi River during a cold front. Appl. Opt. 2006, 45, 7410–7428. [Google Scholar] [CrossRef] [PubMed]
- D’Sa, E.J.; Goes, J.I.; Gomes, H.; Mouw, C. Absorption and fluorescence properties of chromophoric dissolved organic matter of the eastern Bering Sea in the summer with special reference to the influence of a cold pool. Biogeosciences 2014, 11, 3225–3244. [Google Scholar] [CrossRef]
- Liang, Y.; Xiao, X.; Du, E.; Song, C.; Song, C.; Zhao, Y.; Liu, X. Chromophoric Dissolved Organic Matter Fluctuation Assessment in an Urban River. CLEAN Soil Air Water 2014, 43, 1128–1135. [Google Scholar] [CrossRef]
- Coble, P.G. Marine optical biogeochemistry: The chemistry of ocean color. Chem. Rev. 2007, 107, 402–418. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.A.; Zepp, R.G. Invited Review Role of photoreactions in the formation of biologically labile compounds from dissolved organic matter. Oceanography 1997, 42, 523–524. [Google Scholar]
- Mopper, K.; Zhou, X.; Kieber, R.J.; Kieber, D.J.; Sikorski, R.J.; Jones, R.D. Photochemical degradation of dissolved organic carbon and its impact on the oceanic carbon cycle. Nature 1991, 353, 60–62. [Google Scholar] [CrossRef]
- Lapierre, J.F.; Giorgio, P.A. Geographical and environmental drivers of regional differences in the lake pCO2 versus DOC relationship across northern landscapes. J. Geophys. Res. Biogeosci. 2012, 117, 6841–6847. [Google Scholar] [CrossRef]
- Köhler, S.J.; Kothawala, D.; Futter, M.N.; Liungman, O.; Tranvik, L. In-lake processes offset increased terrestrial inputs of dissolved organic carbon and color to lakes. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.; Bolton, L.; Newson, M.; Spencer, R.G. Spectrophotometric properties of surface water dissolved organic matter in an afforested upland peat catchment. Hydrol. Process. 2008, 22, 2325–2336. [Google Scholar] [CrossRef]
- Stedmon, C.A.; Thomas, D.N.; Papadimitriou, S.; Granskog, M.A.; Dieckmann, G.S. Using fluorescence to characterize dissolved organic matter in Antarctic sea ice brines. J. Geophys. Res. Biogeosci. 2011, 116, 752–767. [Google Scholar] [CrossRef]
- Siegel, D.A.; Maritorena, S.; Nelson, N.B.; Hansell, D.A.; Lorenzi-Kayser, M. Global distribution and dynamics of colored dissolved and detrital organic materials. J. Geophys. Res. Oceans 2002, 107. [Google Scholar] [CrossRef]
- Song, K.S.; Zang, S.Y.; Zhao, Y.; Li, L.; Du, J.; Zhang, N.N.; Liu, L. Spatiotemporal characterization of dissolved carbon for inland waters in semi-humid/semi-arid region, China. Hydrol. Earth Syst. Sci. 2013, 17, 4269–4281. [Google Scholar] [CrossRef]
- Zhu, W.; Yu, Q.; Tian, Y.Q.; Becker, B.L.; Zheng, T.; Carrick, H.J. An assessment of remote sensing algorithms for colored dissolved organic matter in complex freshwater environments. Remote Sens. Environ. 2014, 140, 766–778. [Google Scholar] [CrossRef]
- Griffin, C.G.; Frey, K.E.; Rogan, J.; Holmes, R.M. Spatial and inter-annual variability of dissolved organic matter in the Kolyma River, East Siberia, observed using satellite imagery. J. Geophys. Res. Biogeosci. 2011, 116, 165–176. [Google Scholar] [CrossRef]
- Bieroza, M.; Baker, A.; Bridgeman, J. Relating freshwater organic matter fluorescence to organic carbon removal efficiency in drinking water treatment. Sci. Total Environ. 2009, 407, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Raymond, P.A.; McClelland, J.W.; Holmes, R.M.; Zhulidov, A.V.; Mull, K.; Peterson, B.J.; Striegl, R.G.; Aiken, G.R.; Gurtovaya, T.Y. Flux and age of dissolved organic carbon exported to the Arctic Ocean: A carbon isotopic study of the five largest arctic rivers. Glob. Biogeochem. Cycles 2007, 21, 89. [Google Scholar] [CrossRef]
- Holmes, R.M.; Bromiley, P.; Devers, C.E.; Holcomb, T.R.; McGuire, J.B. Management theory applications of prospect theory: Accomplishments, challenges, and opportunities. J. Manag. 2011, 37, 1069–1107. [Google Scholar] [CrossRef]
- Holmes, R.M.; McClelland, J.W.; Peterson, B.J.; Tank, S.E.; Bulygina, E.; Eglinton, T.I.; Gordeev, V.V.; Gurtovaya, T.Y.; Raymond, P.A.; Repeta, D.J.; et al. Seasonal and annual fluxes of nutrients and organic matter from large rivers to the Arctic Ocean and surrounding seas. Estuaries Coasts 2012, 35, 369–382. [Google Scholar] [CrossRef]
- Spencer, R.G.; Hernes, P.J.; Ruf, R.; Baker, A.; Dyda, R.Y.; Stubbins, A.; Six, J. Temporal controls on dissolved organic matter and lignin biogeochemistry in a pristine tropical river, Democratic Republic of Congo. J. Geophys. Res. Biogeosci. 2010, 115. [Google Scholar] [CrossRef]
- Toming, K.; Kutser, T.; Tuvikene, L.; Viik, M.; Nõges, T. Dissolved organic carbon and its potential predictors in eutrophic lakes. Water Res. 2016, 102, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Hestir, E.L.; Brando, V.; Campbell, G.; Dekker, A.; Malthus, T. The relationship between dissolved organic matter absorption and dissolved organic carbon in reservoirs along a temperate to tropical gradient. Remote Sens. Environ. 2015, 156, 395–402. [Google Scholar] [CrossRef]
- Stedmon, C.A.; Markager, S. Resolving the variability in dissolved organic matter fluorescence in a temperate estuary and its catchment using PARAFAC analysis. Limnol. Oceanogr. 2005, 50, 686–697. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, E.; Yin, Y.; Van Dijk, M.A.; Feng, L.; Shi, Z.; Qin, B. Characteristics and sources of chromophoric dissolved organic matter in lakes of the Yungui Plateau, China, differing in trophic state and altitude. Limnol. Oceanogr. 2010, 55, 2645–2659. [Google Scholar] [CrossRef]
- D’Sa, E.J.; DiMarco, S.F. Seasonal variability and controls on chromophoric dissolved organic matter in a large river-dominated coastal margin. Limnol. Oceanogr. 2009, 54, 2233–2242. [Google Scholar] [CrossRef]
- Curtis, P.J.; Adams, H.E. Dissolved organic matter quantity and quality from freshwater and saltwater lakes in east-central Alberta. Biogeochemistry 1995, 30, 59–76. [Google Scholar] [CrossRef]
- Kowalczuk, P.; Cooper, W.J.; Durako, M.J.; Kahn, A.E.; Gonsior, M.; Young, H. Characterization of dissolved organic matter fluorescence in the South Atlantic Bight with use of PARAFAC model: Relationships between fluorescence and its components, absorption coefficients and organic carbon concentrations. Mar. Chem. 2010, 118, 22–36. [Google Scholar] [CrossRef]
- Zhou, Y.; Jeppesen, E.; Zhang, Y.; Shi, K.; Liu, X.; Zhu, G. Dissolved organic matter fluorescence at wavelength 275/342 nm as a key indicator for detection of point-source contamination in a large Chinese drinking water lake. Chemosphere 2016, 144, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Shao, T.; Song, K.; Du, J.; Zhao, Y.; Ding, Z.; Guan, Y.; Zhang, B. Seasonal Variations of CDOM Optical Properties in Rivers Across the Liaohe Delta. Wetlands 2015, 36, 181–192. [Google Scholar] [CrossRef]
- Wen, Z.D.; Song, K.S.; Zhao, Y.; Du, J.; Ma, J.H. Influence of environmental factors on spectral characteristic of chromophoric dissolved organic matter (CDOM) in Inner Mongolia Plateau, China. Hydrol. Earth Syst. Sci. 2016, 20, 787–801. [Google Scholar] [CrossRef]
- Spencer, R.G.; Butler, K.D.; Aiken, G.R. Dissolved organic carbon and chromophoric dissolved organic matter properties of rivers in the USA. J. Geophys. Res. Biogeosci. 2012, 117, 34–47. [Google Scholar] [CrossRef]
- Jiao, K.W.; Li, F.X.; Zhou, Q.X. Spatial distribution and pollution level evaluation of nutrients in the Songhua River Basin. J. Agro-Environ. Sci. 2015, 34, 769–775. [Google Scholar]
- Williamson, C.E.; Brentrup, J.A.; Zhang, J.; Renwick, W.H.; Hargreaves, B.R.; Knoll, L.B.; Overholt, E.P.; Rose, K.C. Lakes as sensors in the landscape: Optical metrics as scalable sentinel responses to climate change. Limnol. Oceanogr. 2014, 59, 840–850. [Google Scholar] [CrossRef]
- Bricaud, A.; Morel, A.; Prieur, L. Absorption by dissolved organic matter of the sea (yellow substance) in the UV and visible domains. Limnol. Oceanogr. 1981, 26, 43–53. [Google Scholar] [CrossRef]
- Song, K.; Li, L.; Wang, Z.; Liu, D.; Zhang, B.; Xu, J.; Wang, Y. Retrieval of total suspended matter (TSM) and chlorophyll-a (Chl-a) concentration from remote-sensing data for drinking water resources. Environ. Monit. Assess. 2012, 184, 1449–1470. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.C.; Baker, K.S. Optical properties of the clearest natural waters (200–800 nm). Appl. Opt. 1981, 20, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Carder, K.L.; Steward, R.G.; Harvey, G.R.; Ortner, P.B. Marine humic and fulvic acids: Their effects on remote sensing of ocean chlorophyll. Limnol. Oceanogr. 1989, 34, 68–81. [Google Scholar] [CrossRef]
- Helms, J.R.; Stubbins, A.; Ritchie, J.D.; Minor, E.C.; Kieber, D.J.; Mopper, K. Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnol. Oceanogr. 2008, 53, 955–969. [Google Scholar] [CrossRef]
- Kowalczuk, P.; Cooper, W.J.; Whitehead, R.F.; Durako, M.J.; Sheldon, W. Characterization of CDOM in an organic-rich river and surrounding coastal ocean in the South Atlantic Bight. Aquat. Sci. 2003, 65, 384–401. [Google Scholar] [CrossRef]
- Singh, S.; D’Sa, E.J.; Swenson, E.M. Chromophoric dissolved organic matter (CDOM) variability in Barataria Basin using excitation-emission matrix (EEM) fluorescence and parallel factor analysis (PARAFAC). Sci. Total Environ. 2010, 408, 3211–3222. [Google Scholar] [CrossRef] [PubMed]
- Weishaar, J.L.; Aiken, G.R.; Bergamaschi, B.A.; Fram, M.S.; Fujii, R.; Mopper, K. Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ. Sci. Technol. 2003, 37, 4702–4708. [Google Scholar] [CrossRef] [PubMed]
- Cory, R.M.; McKnight, D.M.; Chin, Y.P.; Miller, P.; Jaros, C.L. Chemical characteristics of fulvic acids from Arctic surface waters: Microbial contributions and photochemical transformations. J. Geophys. Res. Biogeosci. 2007, 112, 315–331. [Google Scholar] [CrossRef]
- Peuravuori, J.; Pihlaja, K. Molecular size distribution and spectroscopic properties of aquatic humic substances. Anal. Chim. Acta 1997, 337, 133–149. [Google Scholar] [CrossRef]
- Zhang, Y.; Qin, B.; Zhu, G.; Zhang, L.; Yang, L. Chromophoric dissolved organic matter (CDOM) absorption characteristics in relation to fluorescence in Lake Taihu, China, a large shallow subtropical lake. Hydrobiologia 2007, 581, 43–52. [Google Scholar] [CrossRef]
- Yu, H.; Qu, F.; Sun, L.; Liang, H.; Han, Z.; Chang, H.; Shao, S.; Li, G. Relationship between soluble microbial products (SMP) and effluent organic matter (EfOM): Characterized by fluorescence excitation emission matrix coupled with parallel factor analysis. Chemosphere 2015, 121, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.J.; Prairie, Y.T.; Caraco, N.F.; McDowell, W.H.; Tranvik, L.J.; Striegl, R.G.; Melack, J. Plumbing the global carbon cycle: Integrating inland waters into the terrestrial carbon budget. Ecosystems 2007, 10, 172–185. [Google Scholar] [CrossRef]
- Jiang, R.; Hatano, R.; Zhao, Y.; Kuramochi, K.; Hayakawa, A.; Woli, K.P.; Shimizu, M. Factors controlling nitrogen and dissolved organic carbon exports across timescales in two watersheds with different land uses. Hydrol. Process. 2014, 28, 5105–5121. [Google Scholar] [CrossRef]
- Baker, A.; Lamont-BIack, J. Fluorescence of dissolved organic matter as a natural tracer of ground water. Ground Water 2001, 39, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Asmala, E.; Autio, R.; Kaartokallio, H.; Pitkänen, L.; Stedmon, C.; Thomas, D.N. Bioavailability of riverine dissolved organic matter in three Baltic Sea estuaries and the effect of catchment land use. Biogeosciences 2013, 10, 6969–6986. [Google Scholar] [CrossRef]
- Fichot, C.G.; Benner, R. The spectral slope coefficient of chromophoric dissolved organic matter (S275–295) as a tracer of terrigenous dissolved organic carbon in river-influenced ocean margins. Limnol. Oceanogr. 2012, 57, 1453–1466. [Google Scholar] [CrossRef]
- Del Castillo, C.E.; Gilbes, F.; Coble, P.G.; Müller-Karger, F.E. On the dispersal of riverine colored dissolved organic matter over the West Florida Shelf. Limnol. Oceanogr. 2000, 45, 1425–1432. [Google Scholar] [CrossRef]
- Gonnelli, M.; Vestri, S.; Santinelli, C. Chromophoric dissolved organic matter and microbial enzymatic activity. A biophysical approach to understand the marine carbon cycle. Biophys. Chem. 2013, 182, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, Y.; Pan, J. Distributions of colored dissolved organic matter and dissolved organic carbon in the Pearl River Estuary, China. Cont. Shelf Res. 2004, 24, 1845–1856. [Google Scholar] [CrossRef]
- Brezonik, P.L.; Olmanson, L.G.; Finlay, J.C.; Bauer, M.E. Factors affecting the measurement of CDOM by remote sensing of optically complex inland waters. Remote Sens. Environ. 2015, 157, 199–215. [Google Scholar] [CrossRef]
- Prieur, L.; Sathyendranath, S. An optical classification of coastal and oceanic waters based on the specific spectral absorption curves of phytoplankton pigments, dissolved organic matter, and other particulate materials. Limnol. Oceanogr. 1981, 26, 671–689. [Google Scholar] [CrossRef]
- Organelli, E.; Bricaud, A.; Antoine, D.; Matsuoka, A. Seasonal dynamics of light absorption by chromophoric dissolved organic matter (CDOM) in the NW Mediterranean Sea (BOUSSOLE site). Deep Sea Res. I Oceanogr. Res. Pap. 2014, 91, 72–85. [Google Scholar] [CrossRef]
- Müller, R.A.; Futter, M.N.; Sobek, S.; Nisell, J.; Bishop, K.; Weyhenmeyer, G.A. Water renewal along the aquatic continuum offsets cumulative retention by lakes: Implications for the character of organic carbon in boreal lakes. Aquat. Sci. 2013, 75, 535–545. [Google Scholar] [CrossRef]
- Borisover, M.; Bukhanovsky, N.; Lapides, I.; Yariv, S. Thermal treatment of organoclays: Effect on the aqueous sorption of nitrobenzene on n-hexadecyltrimethyl ammonium montmorillonite. Appl. Surf. Sci. 2010, 256, 5539–5544. [Google Scholar] [CrossRef]
- Stanley, E.H.; Powers, S.M.; Lottig, N.R.; Buffam, I.; Crawford, J.T. Contemporary changes in dissolved organic carbon (DOC) in human-dominated rivers: Is there a role for DOC management? Freshw. Biol. 2012, 57, 26–42. [Google Scholar] [CrossRef]
- Shon, H.K.; Vigneswaran, S.; Kim, I.S.; Cho, J.; Ngo, H.H. Fouling of ultrafiltration membrane by effluent organic matter: A detailed characterization using different organic fractions in wastewater. J. Membr. Sci. 2008, 278, 232–238. [Google Scholar] [CrossRef]
- Joshi, I.; D’Sa, E.J. Seasonal Variation of Colored Dissolved Organic Matter in Barataria Bay, Louisiana, Using Combined Landsat and Field Data. Remote Sens. 2015, 7, 12478–12502. [Google Scholar] [CrossRef]


| P | Level |
|---|---|
| P < 0.8 | Qualification |
| 0.8 ≤ P ≤ 1.0 | Basic qualification |
| 1.0 < P ≤ 2 | Contamination |
| P ≥ 2 | Serious contamination |
| Average | pH | DO (mg/L) | NH3-N (mg/L) | COD (mg/L) | Fe (mg/L) | Mn (mg/L) | Zn (mg/L) | Hg (mg/L) | Cr6+ (mg/L) | p |
|---|---|---|---|---|---|---|---|---|---|---|
| Standards | 6–9 | 2 | 2.0 | 40 | 0.3 | 0.1 | 2.0 | 0.001 | 0.1 | - |
| Summer | 6.79 | 2.93 | 0.07 | 392.16 | 0.62 | 0.28 | 0.06 | 0.34 | 0.012 | 8.11 |
| Autumn | 6.08 | 9.47 | 0.15 | 49.37 | 2.83 | 0.54 | 0.13 | 0 | 0.002 | 9.03 |
| Summer | Autumn | |||
|---|---|---|---|---|
| Avg ± SD. | Min–Max | Avg ± SD. | Min–Max | |
| aCDOM(335) | 5.5 ± 3.5 | 0.3–10.6 | 5.4 ± 3.5 | 0.3–12.3 |
| aCDOM(375) | 2.3 ± 1.8 | 0–5.2 | 2.6 ± 2.0 | 0–6.9 |
| aCDOM(440) | 0.6 ± 0.6 | 0–1.7 | 0.8 ± 0.9 | 0–3.1 |
| SUVA254 | 4.0 ± 1.4 | 1.6–6.3 | 2.3 ± 0.8 | 0.6–3.5 |
| E250:365 | 11.7 ± 11.5 | 0–38.7 | 6.4 ± 2.8 | 0–10.1 |
| S275–295 | 0.0089 ± 0.0020 | 0.0069–0.0147 | 0.0082 ± 0.0016 | 0.0066–0.0123 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Zhang, J.; Mu, G.; Ju, H.; Wang, R.; Li, D.; Shabbir, A.H. Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers. Water 2016, 8, 399. https://doi.org/10.3390/w8090399
Li S, Zhang J, Mu G, Ju H, Wang R, Li D, Shabbir AH. Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers. Water. 2016; 8(9):399. https://doi.org/10.3390/w8090399
Chicago/Turabian StyleLi, Sijia, Jiquan Zhang, Guangyi Mu, Hanyu Ju, Rui Wang, Danjun Li, and Ali Hassan Shabbir. 2016. "Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers" Water 8, no. 9: 399. https://doi.org/10.3390/w8090399
APA StyleLi, S., Zhang, J., Mu, G., Ju, H., Wang, R., Li, D., & Shabbir, A. H. (2016). Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers. Water, 8(9), 399. https://doi.org/10.3390/w8090399








