Small Boreal Lake Ecosystem Evolution under the Influence of Natural and Anthropogenic Factors: Results of Multidisciplinary Long-Term Study
Abstract
:1. Introduction
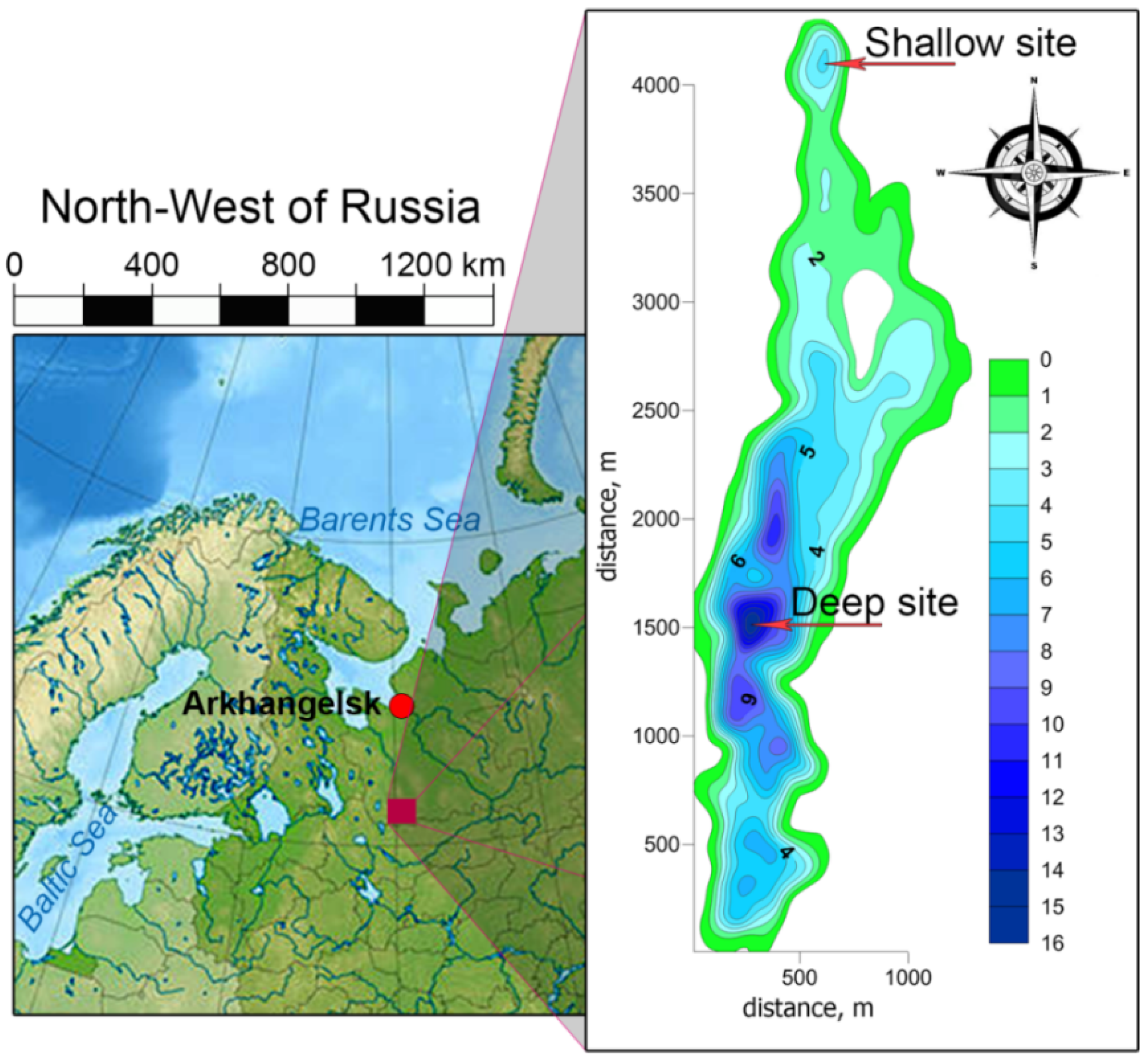
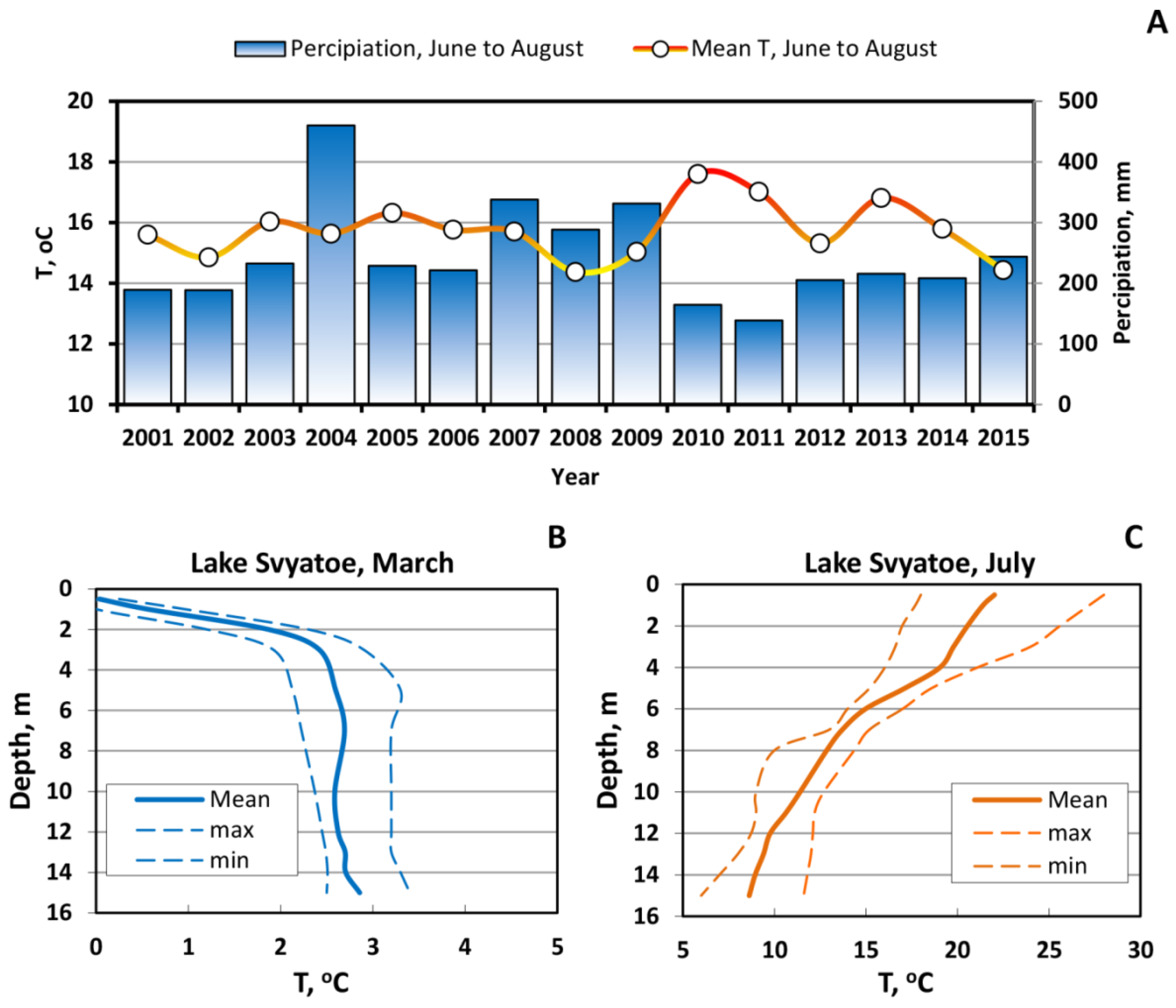
2. Study Site Description, Sampling, Methods, and Analysis
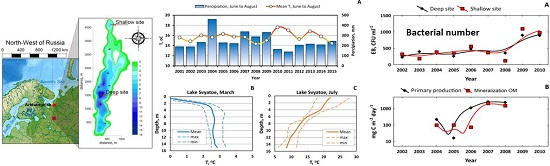
3. Results and Discussion
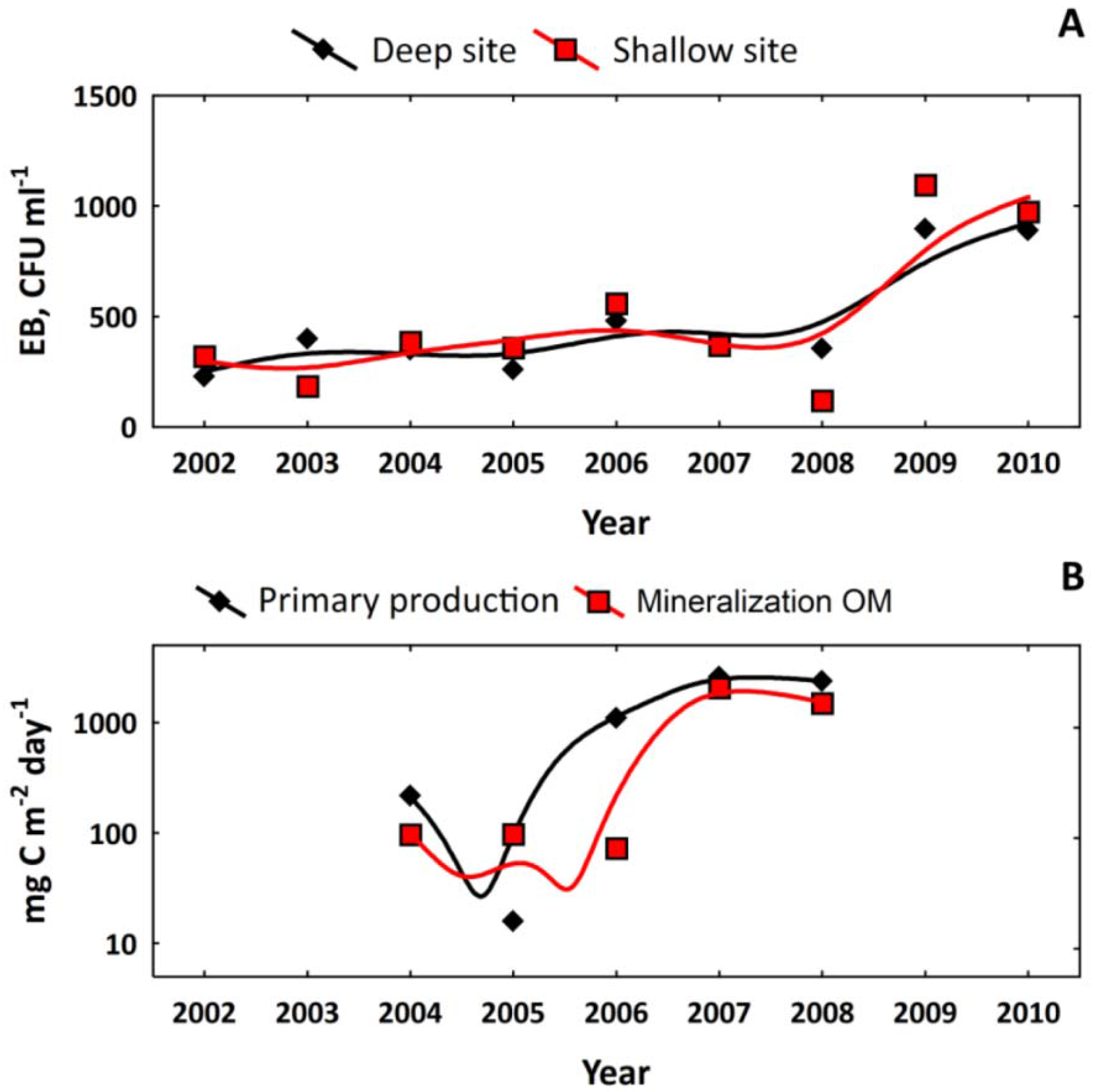
3.1. Long-Term Evolution of Lake Ecosystem
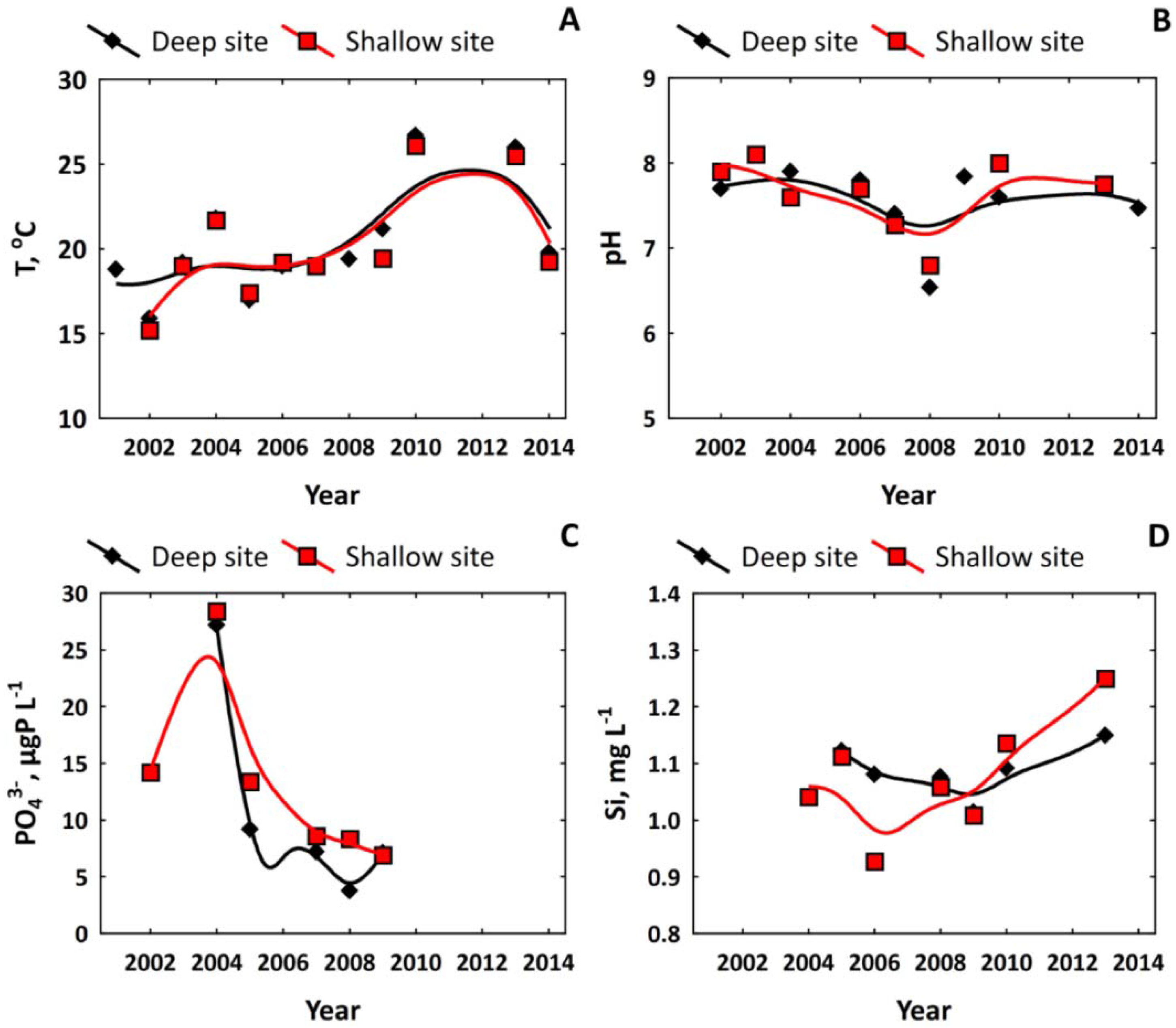
3.2. Nutrient Regime in Pristine and Anthropogenically-Affected Zones of the Lake
3.3. Biota of Lake Svyatoe
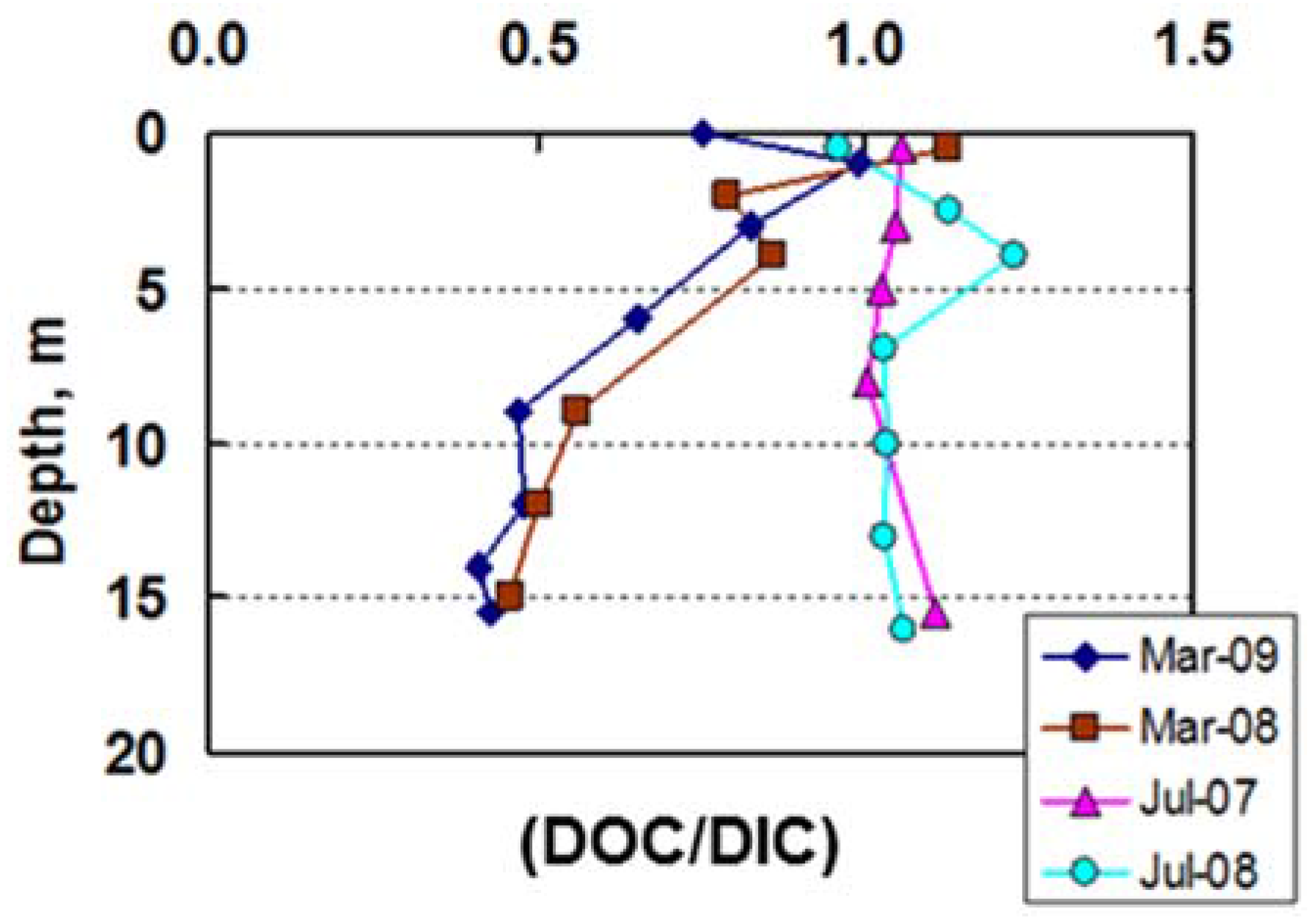
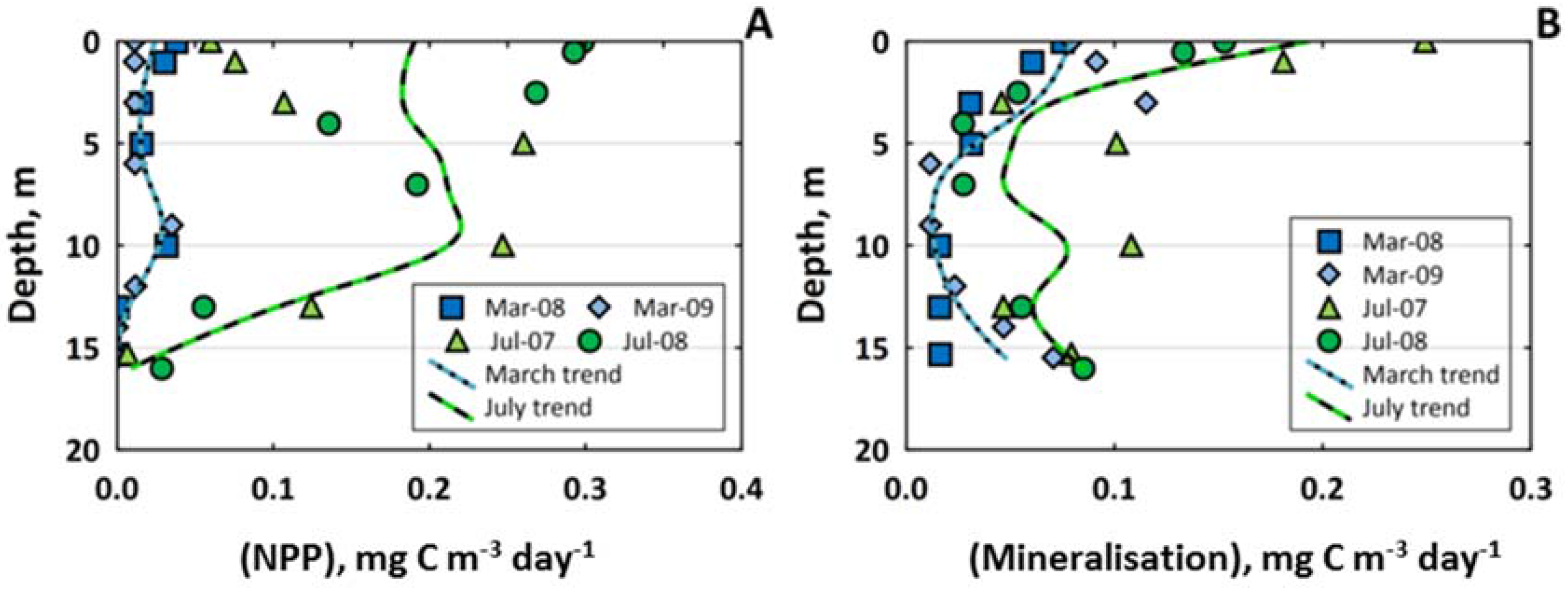
3.4. Carbon Biogeochemistry
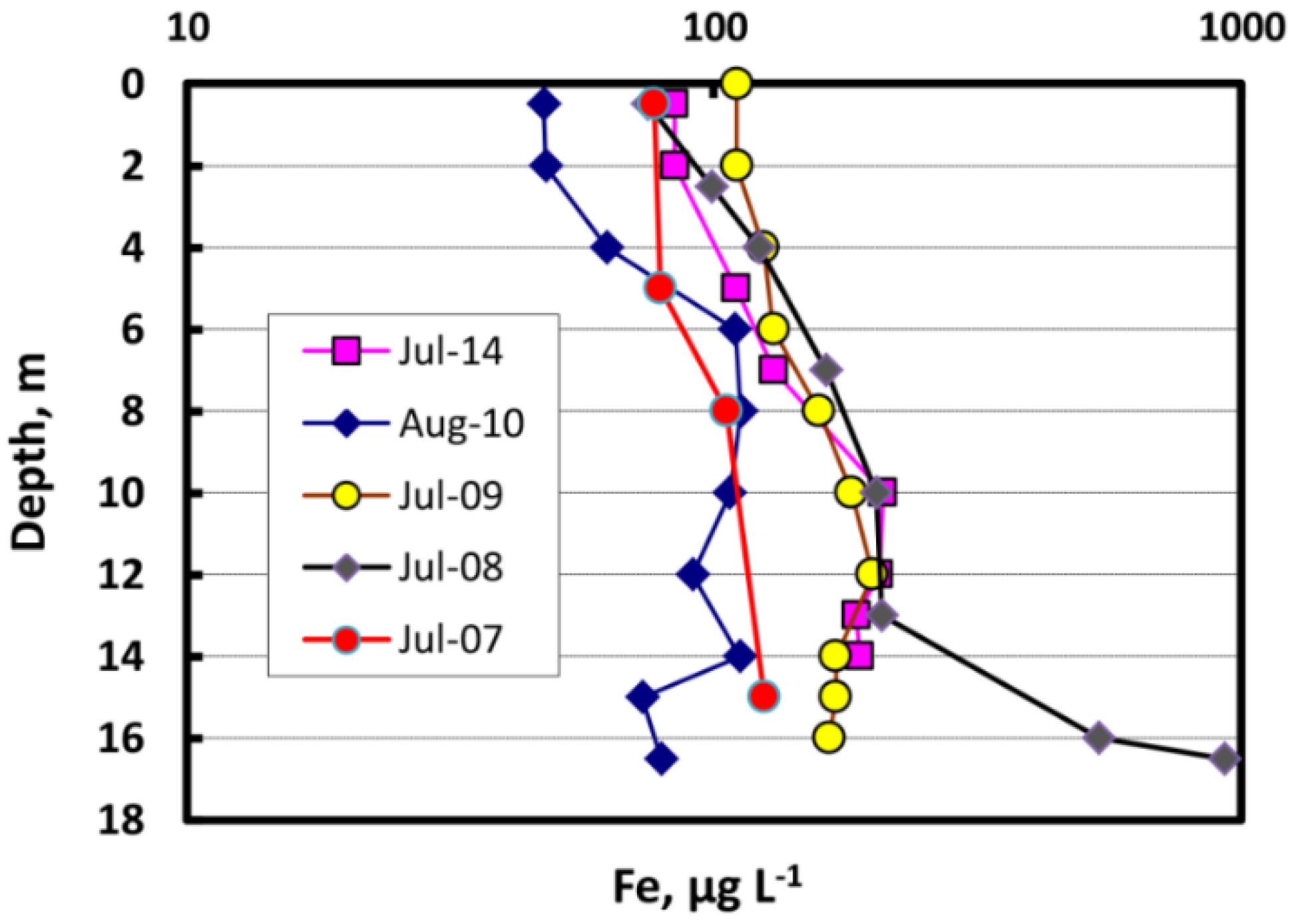
3.5. Trace Metal Speciation and Cycling in the Water Column
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Algesten, G.; Sobek, S.; Bergstro, A.K.; Agren, A.; Tranvik, L.J.; Jansson, M. Role of lakes for organic carbon cycling in the boreal zone. Glob. Chang. Biol. 2003, 10, 141–147. [Google Scholar] [CrossRef]
- Worrall, F.; Harriman, R.; Evans, C.D.; Watts, C.D.; Adamson, J.; Neal, C.; Tipping, E.; Burt, T.; Grieve, I.; Monteith, D.; et al. Trends in dissolved organic carbon in UK rivers and lakes. Biogeochemistry 2004, 70, 369–402. [Google Scholar] [CrossRef]
- Kothawala, D.N.; Stedmon, C.A.; Müller, R.A.; Weyhenmeyer, G.A.; Köhler, S.J.; Tranvik, L.J. Controls of dissolwed organic matter quality: Evidence from a large-scale boreal lake suvery. Glob. Chang. Biol. 2014, 20, 1101–1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagano, T.; Bida, M.; Kenny, J.F. Trends in levels of allochthonous dissolved organic carbon in natural water: A review of potential mechanisms under a changing climate. Water 2014, 6, 2862–2897. [Google Scholar] [CrossRef]
- Grönlund, E. The recovery of two polluted subarctic lakes-towards nutrient management or a pristine state? Water 2012, 4, 793–814. [Google Scholar] [CrossRef]
- Filella, M.; Rodrigues-Murillo, J.C. Long-term trends of organic carbon concentrations in freshwaters: Strengths and weaknesses of existing evidence. Water 2014, 6, 1360–1418. [Google Scholar] [CrossRef]
- Kvasov, D.D.; Martinson, G.G.; Raykas, A.V. History of Lake Ladoga, Lake Onega, Pskovsko-Chudskoe Lakes, Lake Baikal and Lake Hankey; Nauka: Leningrad, Russia, 1990. (In Russian) [Google Scholar]
- Sayad, L.; Kherici-Bousnoubra, H.; Drouiche, N.; Houhamdi, M.; Kherici, N. Calculation and interpretation of effluent discharge objectives of dairy industry: Case Edough’s dairy—Annaba (Algeria). Ecol. Eng. 2014, 73, 421–424. [Google Scholar] [CrossRef]
- Bahroun, S.; Bousnoubra, H.; Drouiche, N.; Kherici, N. Analysis of wastewater discharges to the Wadi Kebir East River by the environmental discharge objectives (EDO) method. Desal. Water Treat. 2016. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Pokrovsky, O.S.; Moreva, O.Y.; Chupakov, A.V.; Zabelina, S.A.; Klimov, S.I.; Shorina, N.V.; Vorobieva, T.Y. Decrease of concentration and colloidal fraction of organic carbon and trace elements in response to the anomalously hot summer 2010 in a humic boreal lake. Sci. Total Environ. 2013, 463/464, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Pokrovsky, O.S.; Shirokova, L.S. Diurnal variations of dissolved and colloidal organic carbon and trace metals in a boreal lake during summer bloom. Water Res. 2013, 47, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Pokrovsky, O.S.; Shirokova, L.S.; Zabelina, S.A.; Vorobieva, T.Y.; Moreva, O.Y.; Klimov, S.I.; Chupakov, A.V.; Shorina, N.V.; Kokryatskaya, N.M.; Audry, S.; et al. Size fractionation of trace elements in a seasonally stratified boreal lakes: Control of organic matter and iron colloids. Aquat. Geochem. 2012, 18, 115–139. [Google Scholar] [CrossRef]
- Koroleff, F. Total and organic nitrogen. In Methods for Seawater Analysis; Grasshoff, K., Ehrhardt, M., Kremling, K., Eds.; Verlag Chemie: Weinheim, Germany, 1983; pp. 162–168. [Google Scholar]
- Koroleff, F. Determination of phosphorus. In Methods for Seawater Analysis; Grasshoff, K., Ehrhardt, M., Kremling, K., Eds.; Verlag Chemie: Weinheim, Germany, 1983; pp. 125–136. [Google Scholar]
- Shirokova, L.S.; Pokrovsky, O.S.; Kirpotin, S.N.; Dupré, B. Heterotrophic bacterio-plankton in thawed lakes of northern part of Western Siberia controls the CO2 flux to the atmosphere. Int. J. Environ. Stud. 2009, 66, 433–445. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Pokrovsky, O.S.; Kirpotin, S.N.; Desmukh, C.; Pokrovsky, B.G.; Audry, S.; Viers, J. Biogeochemistry of organic carbon, CO2, CH4, and trace elements in thermokarst water bodies in discontinuous permafrost zones of Western Siberia. Biogeochemistry 1983, 113, 573–593. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Pokrovsky, O.S.; Viers, J.; Klimov, S.I.; Moreva, O.Y.; Zabelina, S.A.; Vorobieva, T.Y.; Dupré, B. Diurnal variations of trace elements and heterotrophic bacterioplankton concentration in a small boreal lake of the White Sea basin. Ann. Limnol. 2010, 46, 67–75. [Google Scholar] [CrossRef]
- Guseva, K.A. Methods of phytoplankton counting. Trudy Inst. Biol. Vodochr. 1959, 2, 44–51. (In Russian) [Google Scholar]
- Trifonova, I.S. Phytoplankton composition and biomass structure in relation to trophic gradient in some temperate and subarctic lakes of northwestern Russia and the Prebaltic. Hydrobiologia 1998, 369/370, 99–108. [Google Scholar] [CrossRef]
- Makarova, I.V.; Pichkily, L.O. On some questions of the method of biomass of phytoplankton calculation. Bot. J. (Botanicheskii Zhurnal) 1970, 55, 1488–1494. [Google Scholar]
- Odum, E.P. Fundamentals of Ecology; Saunders: Philadelphia, PA, USA, 1971. [Google Scholar]
- Sladecek, V. System of water quality from the biological point of view. Arch. Hydrobiol. 1973, 7, 1–218. [Google Scholar]
- Barinova, S.S.; Medvedeva, L.A.; Anisimova, O.V. Biodiversity of Algae—Indicators of the Environment; Tell-Aviv Publ. House: Tel-Aviv, Israel, 2006. [Google Scholar]
- Zhakov, L.A. Formation and Stricture of Fish Population of Lakes of NW of the USSR; Nauka: Moscow, Russia, 1984. (In Russian) [Google Scholar]
- Sobek, S.; Algesten, G.; Bergstrom, A.K.; Jansson, M.; Tranvik, L.J. The catchment and climate regulation of pCO2 in boreal lakes. Glob. Chang. Biol. 2003, 9, 630–641. [Google Scholar] [CrossRef]
- Sobek, S.; Söderbäck, B.; Karsson, S.; Andersson, E.; Brunbergn, A.-K. A carbon budget of a small humic lake: An example of the importance of lakes for organic matter cycling in boreal catchments. Ambio 2006, 35, 469–475. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Viers, J.; Dupré, B.; Chabaux, F.; Gaillardet, J.; Audry, S.; Prokushkin, A.S.; Shirokova, L.S.; Kirpotin, S.N.; Lapitsky, S.A.; et al. Biogeochemistry of carbon, major and trace elements in watersheds of Northern Eurasia drained to the Arctic Ocean: The change of fluxes, sources and mechanisms under the climate warming prospective. C. R. Geosci. 2012, 344, 663–677. [Google Scholar] [CrossRef]
- Hamilton-Taylor, J.; Smith, E.J.; Davison, W.; Sugiyama, M. Resolving and modeling the effects of Fe and Mn redox cycling on trace metal behavior in a seasonally anoxic lake. Geochim. Cosmochim. Acta 2005, 69, 1947–1960. [Google Scholar] [CrossRef] [Green Version]
- Amon, R.M.W.; Benner, R. Photochemical and microbial consumption of dissolved organic carbon and dissolved oxygen in the Amazon River System. Geochim. Cosmochim. Acta 1996, 60, 1783–1792. [Google Scholar] [CrossRef]
- Jonsson, A.; Aberg, J.; Lindroth, A.; Jansson, M. Gas transfer rate and CO2 flux between an unproductive lake and the atmosphere in northern Sweden. J. Geophys. Res. 2008, 113, G04006. [Google Scholar] [CrossRef]
- Jonsson, A.; Meili, M.; Bergstrom, A.K.; Jansson, M. Whole-lake mineralization of allochthonous organic carbon in a large humic lake (Ortrasket, N. Sweden). Limnol. Oceanogr. 2001, 46, 1691–1700. [Google Scholar] [CrossRef]
- Pers, C.; Rahm, L.; Jonsson, A.; Bergström, A.K.; Jansson, M. Modelling dissolved organic carbon turnover in humic Lake Örträsket, Sweden. Environ. Model. Assess. 2001, 6, 159–172. [Google Scholar] [CrossRef]
- Kelton, N.; Molot, L.A.; Dillon, P.J. Effect of ultraviolet and visible radiation on iron lability in boreal and artificial waters. Aquat. Sci. 2007, 69, 86–95. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Murray, J.W.; Paul, B. The geochemical cycling of trace elements in a biogenic meromictic lake. Geochim. Cosmochim. Acta 1994, 58, 3993–4008. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Murray, J.W.; Paul, B. The geochemical cycling of stable Pb, 210Pb, and 210Po in seasonally anoxic Lake Sammamish, Washington, USA. Geochim. Cosmochim. Acta 1995, 59, 4845–4861. [Google Scholar] [CrossRef]
- Kokryatskaya, N.M.; Zabelina, S.A.; Savvichev, A.S.; Moreva, O.Y.; Vorovieva, T.Y. Seasonal Biogeochemical and Microbiological Studies of Small Lakes in Taiga Zone of Northwestern Russian (Arkhangelsk Province). Water Resour. 2012, 39, 105–117. [Google Scholar] [CrossRef]
- Tranvik, L.J.; Downing, J.A.; Cotner, J.B.; Loiselle, S.A.; Striegl, R.G.; Ballatore, T.J.; Dillon, P.; Finlay, K.; Fortino, K.; Knoll, L.B.; et al. Lakes and reservoirs as regulators of carbon cycling and climate. Limnol. Oceanogr. 2009, 54, 2298–2314. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Schott, J. Iron colloids/organic matter associated transport of major and trace elements in small boreal rivers and their estuaries (NW Russia). Chem. Geol. 2002, 190, 141–179. [Google Scholar] [CrossRef]
- Vasyukova, E.V.; Pokrovsky, O.S.; Viers, J.; Oliva, P.; Dupré, B.; Martin, F.; Candaudaup, F. Trace elements in organic- and iron-rich surficial fluids of the Boreal zone Assessing colloidal forms via dialysis and ultrafiltration. Geochim. Cosmochim. Acta 2010, 74, 449–468. [Google Scholar] [CrossRef]
- Audry, S.; Pokrovsky, O.S.; Shirokova, L.S.; Kirpotin, S.N.; Dupré, B. Organic matter mineralization and trace element post-depositional redistribution in Western Siberia thermokarst lake sediments. Biogeosciences 2011, 8, 565–583. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Bredoire, R.; Rols, J.-L.; Pokrovsky, O.S. Moss and peat leachate degradability by heterotrophic bacteria: Fate of organic carbon and trace metals. Geomicrobiol. J. 2015. [Google Scholar] [CrossRef]
- Pavlova, O.A.; Pokrovsky, O.S.; Manasypov, R.M.; Shirokova, L.S.; Vorobyev, S.N. Seasonal dynamics of phytoplankton in acidic and humic environment in shallow thaw ponds of western Siberia, discontinuous permafrost zone. Ann. Limnol. 2016, 52, 47–60. [Google Scholar] [CrossRef]

| Parameter | Year | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2013 | 2014 | |
| T | + | + | + | + | + | + | + | + | + | + | + | + |
| pH | – | + | + | + | + | + | + | + | + | + | + | + |
| TDS | – | + | + | + | + | + | + | + | + | + | + | + |
| BOD5 | – | – | + | + | + | + | – | – | – | – | – | – |
| O2 | – | – | + | + | + | + | + | + | + | + | + | + |
| P total | – | – | – | – | + | + | + | + | + | + | – | – |
| P organic | – | – | – | – | – | – | + | + | + | + | – | – |
| PO43− | – | – | + | + | + | + | + | + | + | + | – | – |
| N-NO3 | – | – | – | – | – | – | + | + | + | + | – | – |
| N-NO2 | – | – | + | + | + | + | + | + | + | + | – | – |
| N total | – | – | – | – | – | – | – | + | + | + | – | – |
| N organic | – | – | – | – | – | – | + | + | + | + | – | – |
| N-NH3 | – | – | – | – | – | – | + | + | + | + | – | – |
| SO42− | – | – | – | – | – | – | + | + | + | + | – | + |
| H2S, S2− | – | – | – | – | + | + | + | + | + | + | + | + |
| Si | – | – | + | + | + | + | + | + | + | + | – | – |
| Fe, Mn, TE | – | – | – | – | – | – | + | + | + | + | – | – |
| Cl− | – | – | – | – | – | – | + | + | + | + | – | + |
| DOC | – | – | – | – | – | – | + | + | + | + | – | + |
| DIC | – | – | – | – | – | – | + | + | + | + | – | + |
| EB | + | + | + | + | + | + | + | + | + | + | – | – |
| FOB | – | + | + | + | + | + | + | + | + | + | – | – |
| OB | – | – | – | – | + | + | + | + | + | + | – | – |
| TB | + | + | + | + | + | – | – | – | – | – | – | – |
| Chl a | – | – | – | – | – | – | – | + | + | – | – | – |
| Phytoplankton | – | – | – | – | + | – | + | – | – | – | – | – |
| Zooplankton | – | – | + | + | + | – | + | – | + | + | – | – |
| PP and BD | + | + | + | + | + | + | + | + | + | + | – | – |
| Fish | – | – | – | + | + | + | – | – | – | – | – | – |
| Date | U-Test | PO43− | Porg | NO3− | NH4+ | Norg | Si | Co | Ni | Cu | Zn | As | Cd | Pb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| March 2008 | p-value | 1.00 | 0.49 | 0.82 | 0.85 | 0.82 | 0.6 | 0.07 | 0.23 | 0.04 | 0.04 | 0.04 | 0.37 | 1.00 |
| (S)/(D) | 2.1 | 0.9 | 0.9 | 1.2 | 1.0 | 0.9 | 3.6 | 0.8 | 0.3 | 1.3 | 1.1 | 1.8 | 1.1 | |
| July 2008 | p-value | 1.00 | 0.01 | 0.01 | 1.00 | 0.04 | 0.46 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| (S)/(D) | 0.5 | 1.9 | 0.2 | 1.0 | 1.2 | 0.6 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | |
| March 2009 | p-value | 0.01 | 0.04 | 0.01 | 0.52 | 0.92 | 0.52 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| (S)/(D) | 2.0 | 0.7 | 1.2 | 0.9 | 1.0 | 1.0 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | |
| July 2009 | p-value | 0.24 | 0.08 | 0.24 | 0.07 | 0.24 | 0.24 | 1.00 | 0.06 | 0.06 | 0.11 | 0.46 | 0.06 | 0.06 |
| (S)/(D) | 0.5 | 6.6 | 0.8 | 0.5 | 1.3 | 0.7 | 1.0 | 0.8 | 1.1 | 0.3 | 1.0 | 0.4 | 2.8 | |
| March 2010 | p-value | 1.00 | 0.62 | 0.87 | 0.87 | 0.13 | 0.24 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| (S)/(D) | 1.4 | 1.1 | 1.0 | 1.3 | 0.8 | 0.8 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | |
| Auguest 2010 | p-value | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | 0.29 | 0.13 | 0.56 | 0.05 | 0.41 | 0.05 |
| (S)/(D) | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | 0.9 | 1.1 | 1.8 | 1.5 | 1.3 | 12.8 |
| Component of Ecosystem | R2 | ||||||
|---|---|---|---|---|---|---|---|
| T, °C | pH | TDS | O2 | NO2− | PO43− | Si | |
| Eutrophic bacterioplankton | 0.76 * | 0.66 * | −0.43 | 0.75 * | −0.30 | 0.31 | −0.57 |
| Facultative-oligotrophic bacterioplankton | 0.15 | 0.11 | 0.41 | 0.74 * | −0.13 | −0.10 | 0.07 |
| Total number of bacterioplankton | 0.78 * | 0.82 ** | −0.24 | 0.77 * | 0.32 | 0.60 * | −0.40 |
| Biomass of bacterioplankton | 0.59 * | 0.66 * | −0.01 | 0.80 * | −0.38 | 0.26 | −0.24 |
| Temperature | pH | Oxygen and PO43− |
|---|---|---|
| EB = 591 × T − 11177 | EB = 1070 × pH − 7056 | EB = 1492 × (O2) − 9731 |
| – | – | FOB = 1487 × (O2) − 9393 |
| TB = 1.35 × T − 25.4 | TB = 2.97 × pH − 19.9 | TB = 0.149 × (PO43−) − 0.544 |
| BB = 0.586 × T − 11.3 | BB = 1.36 × pH − 9.45 | BB = 2.06 × (O2) − 14.0 |
| Parameter | N % | B % | N % | B % | N % | B % |
|---|---|---|---|---|---|---|
| Depth | 0.5 m | 5 m | 8 m | |||
| Cyanophyta | 87.2 | 26.5 | 89.8 | 56.0 | 75.6 | 4.3 |
| Bacillariophyta | 2.8 | 14.2 | 2.5 | 15.5 | 12.4 | 51.4 |
| Euglenophyta | – | – | 0.0 | 0.3 | – | – |
| Cryptophyta | 2.6 | 3.4 | 3.4 | 4.9 | 2.8 | 2.1 |
| Dinophyta | 0.2 | 49.7 | 0.1 | 21.0 | 0.3 | 39.4 |
| Chrysophyta | – | – | 0.1 | 0.6 | – | – |
| Chlorophyta | 6.4 | 3.9 | 2.9 | 1.3 | 4.4 | 1.4 |
| Xanthophyta | – | – | – | – | – | – |
| Flagellata | 0.7 | 0.3 | 1.1 | 0.4 | 4.6 | 1.4 |
| Raphidophyta | – | – | – | – | – | – |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shirokova, L.; Vorobieva, T.; Zabelina, S.; Klimov, S.; Moreva, O.; Chupakov, A.; Makhnovich, N.; Gogolitsyn, V.; Sobko, E.; Shorina, N.; et al. Small Boreal Lake Ecosystem Evolution under the Influence of Natural and Anthropogenic Factors: Results of Multidisciplinary Long-Term Study. Water 2016, 8, 316. https://doi.org/10.3390/w8080316
Shirokova L, Vorobieva T, Zabelina S, Klimov S, Moreva O, Chupakov A, Makhnovich N, Gogolitsyn V, Sobko E, Shorina N, et al. Small Boreal Lake Ecosystem Evolution under the Influence of Natural and Anthropogenic Factors: Results of Multidisciplinary Long-Term Study. Water. 2016; 8(8):316. https://doi.org/10.3390/w8080316
Chicago/Turabian StyleShirokova, Liudmila, Taissia Vorobieva, Svetlana Zabelina, Sergey Klimov, Olga Moreva, Artem Chupakov, Natalia Makhnovich, Vladimir Gogolitsyn, Elena Sobko, Natalia Shorina, and et al. 2016. "Small Boreal Lake Ecosystem Evolution under the Influence of Natural and Anthropogenic Factors: Results of Multidisciplinary Long-Term Study" Water 8, no. 8: 316. https://doi.org/10.3390/w8080316
APA StyleShirokova, L., Vorobieva, T., Zabelina, S., Klimov, S., Moreva, O., Chupakov, A., Makhnovich, N., Gogolitsyn, V., Sobko, E., Shorina, N., Kokryatskaya, N., Ershova, A., & Pokrovsky, O. (2016). Small Boreal Lake Ecosystem Evolution under the Influence of Natural and Anthropogenic Factors: Results of Multidisciplinary Long-Term Study. Water, 8(8), 316. https://doi.org/10.3390/w8080316