Assessment of Streamside Management Zones for Conserving Benthic Macroinvertebrate Communities Following Timber Harvest in Eastern Kentucky Headwater Catchments
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Treatments
2.3. Invertebrate Sampling
2.4. Data Treamtent
2.5. Analysis
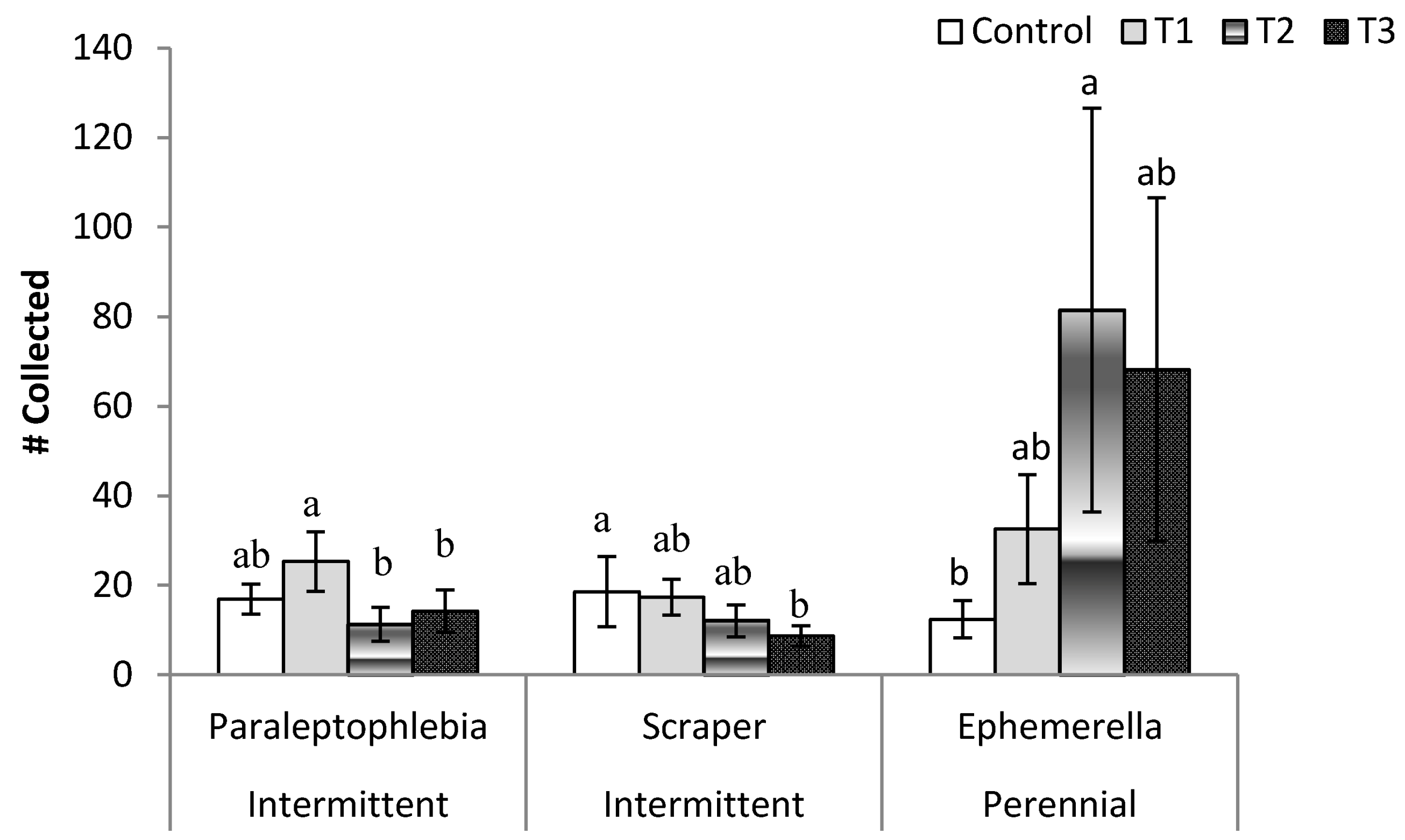
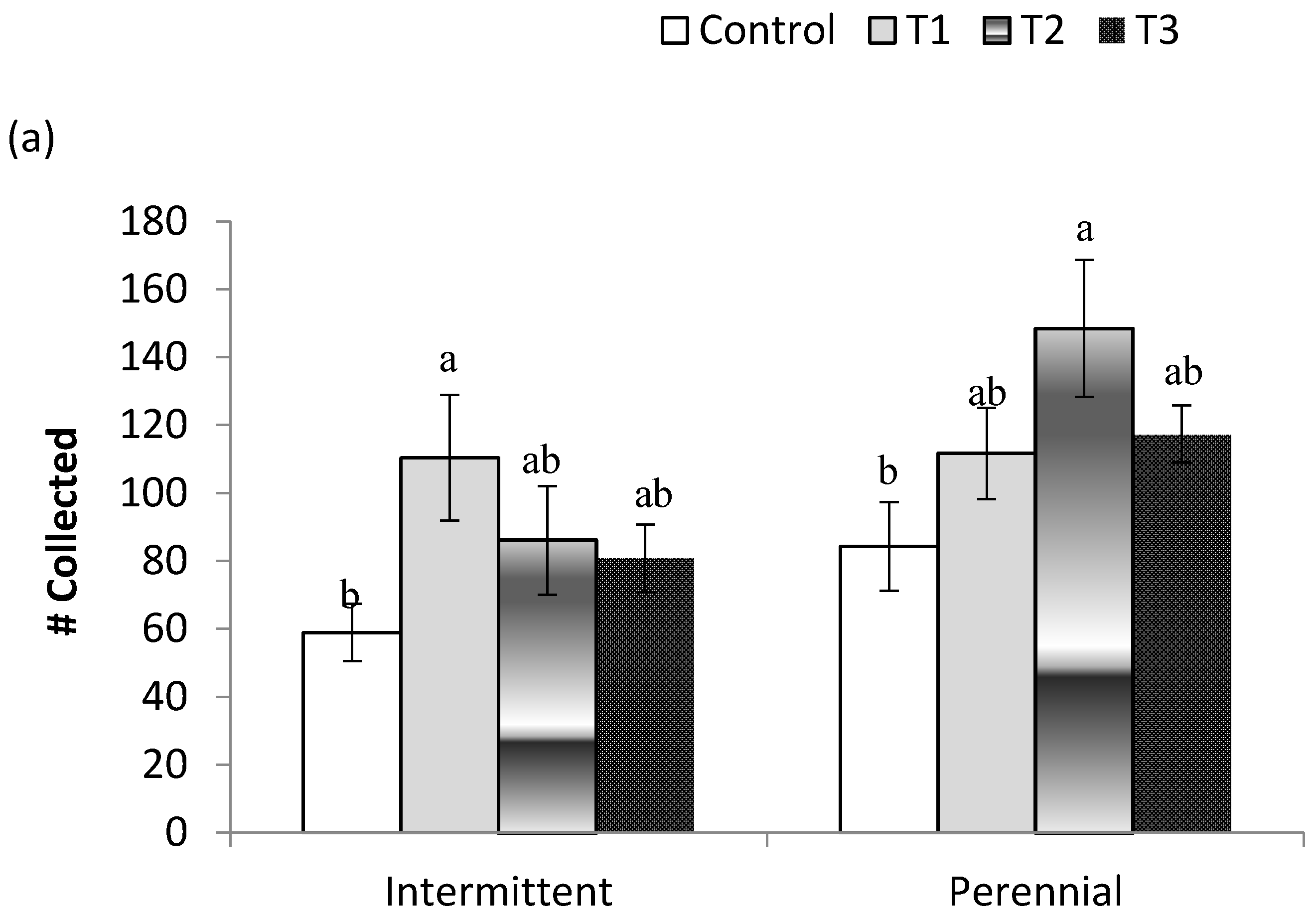
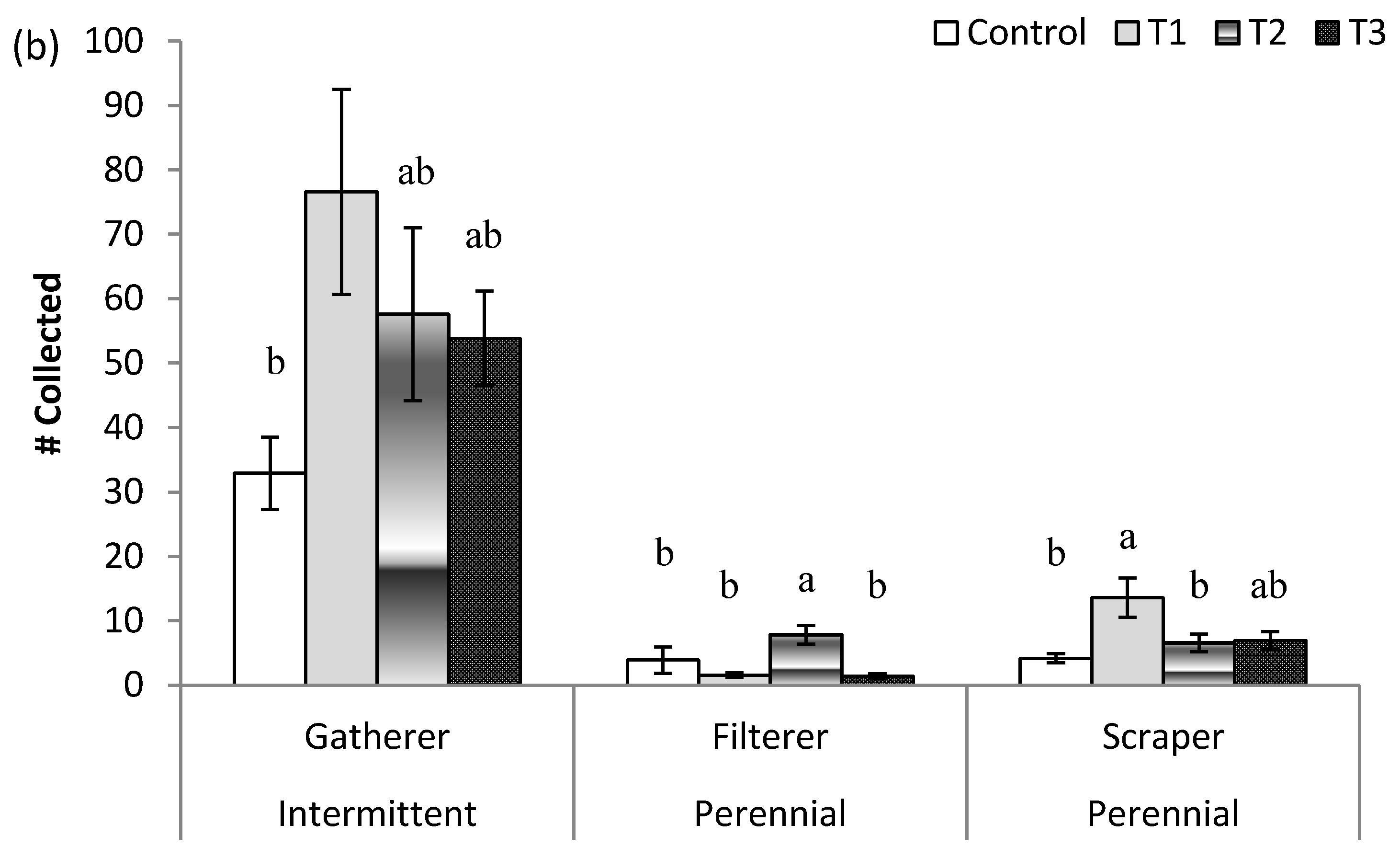
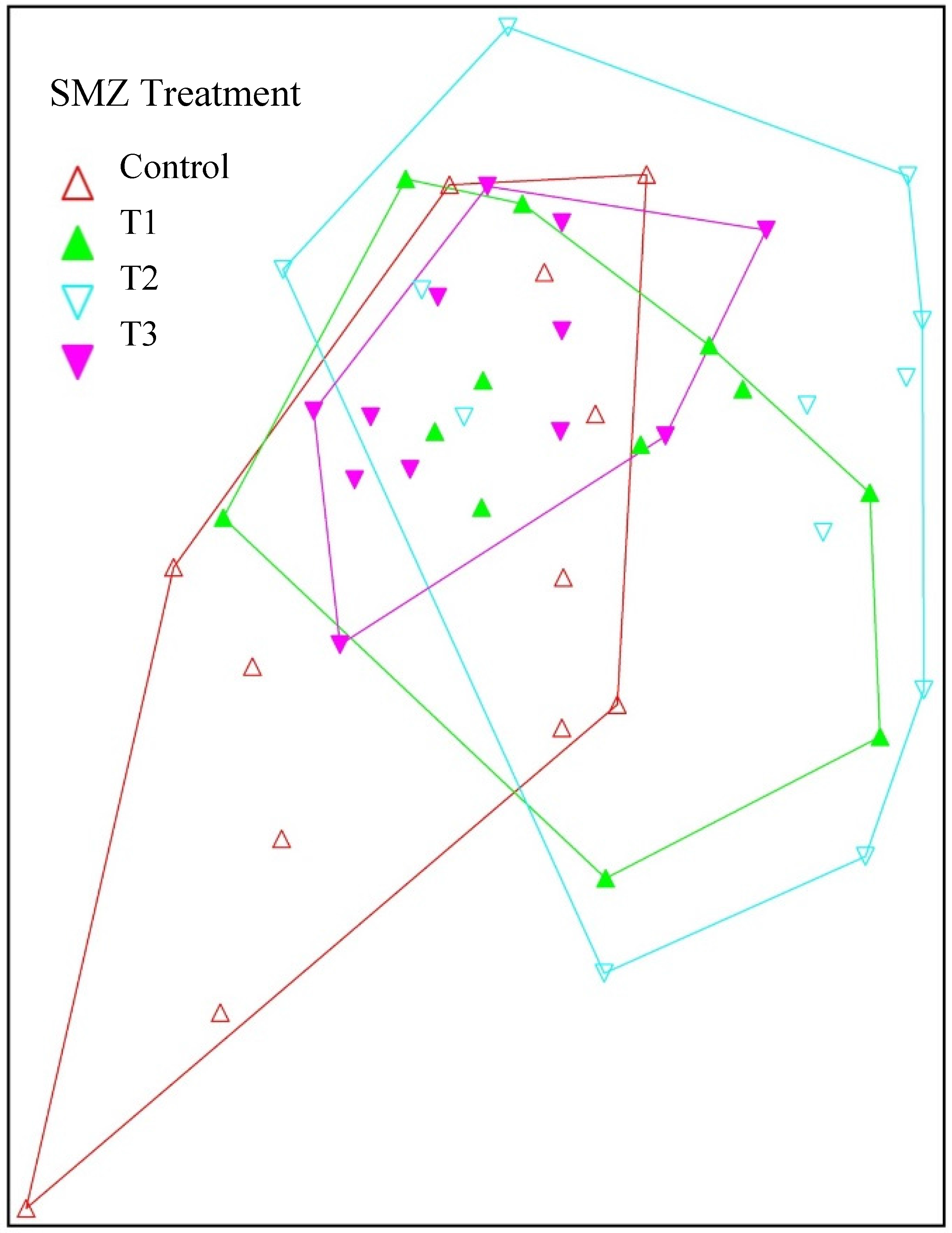
3. Results
3.1. Pre-Treatment Aquatic Communities
3.2. Post-Treatment Aquatic Communities
3.3. Stream Community Metrics
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BMP | Best management practice |
| DBH | Diameter at breast height |
| NMDS | Non-metric multidimensional scaling |
| SMZ | Streamside management zone |
References
- Gomi, T.; Sidle, R.C.; Richardson, J.S. Understanding processes and downstream linkages of headwater systems. Bioscience 2002, 52, 905–916. [Google Scholar] [CrossRef]
- MacDonald, L.H.; Coe, D. Influence of headwater streams on downstream reaches in forested Areas. For. Sci. 2007, 53, 148–168. [Google Scholar]
- Alexander, L.C.; Hawthorne, D.J.; Palmer, M.A.; Lamp, W.O. Loss of genetic diversity in the North American mayfly Ephemerella invaria associated with deforestation of headwater streams. Freshw. Biol. 2011, 56, 1456–1467. [Google Scholar] [CrossRef]
- Lowe, W.H.; Likens, G.E. Moving headwater streams to the head of the class. Bioscience 2005, 55, 196–197. [Google Scholar] [CrossRef]
- Meyer, J.L.; Strayer, D.L.; Wallace, J.B.; Eggert, S.L.; Helfman, G.S.; Leonard, N.E. The Contribution of Headwater Streams to Biodiversity in River Networks. J. Am. Water Resour. Assoc. 2007, 43, 86–103. [Google Scholar] [CrossRef]
- Karr, J.R.; Schlosser, I.J. Water resources and the land-water interface. Science 1978, 201, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Gregory, S.V.; Swanson, F.J.; McKee, W.A.; Cummins, K.W. An ecosystem perspective of riparian zones. Bioscience 1991, 41, 540–551. [Google Scholar] [CrossRef]
- Naiman, R.J.; Décamps, H.; Henri, D. The ecology of interfaces: Riparian zones. Annu. Rev. Ecol. Syst. 1997, 28, 621–658. [Google Scholar] [CrossRef]
- Laeser, S.R.; Baxter, C.V.; Fausch, K.D. Riparian vegetation loss, stream channelization, and web-weaving spiders in northern Japan. Ecol. Res. 2005, 20, 646–651. [Google Scholar] [CrossRef]
- Richardson, J.S.; Danehy, R.J. A synthesis of the ecology of headwater streams and their riparian zones in temperate forests. For. Sci. 2007, 53, 131–147. [Google Scholar]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Knight, A.W.; Bottorff, R.L. The importance of riparian vegetation to stream ecosystems. In California Riparian Systems: Ecology, Conservation, and Productive Management; University of California Press: Berkely, CA, USA, 1984; pp. 160–167. [Google Scholar]
- Wallace, J.B.; Eggert, S.L.; Meyer, J.L.; Webster, J.R. Effects of resource limitation on a detrital-based ecosystem. Ecol. Monogr. 1999, 69, 409–442. [Google Scholar] [CrossRef]
- Hilsenhoff, W. An improved biotic index of organic stream pollution. Gt. Lakes Entomol. 1987, 20, 31–40. [Google Scholar]
- Hilsenhoff, W.L. Rapid field assessment of organic pollution with a family-level biotic index. J. N. Am. Benthol. Soc. 1988, 7, 65–68. [Google Scholar] [CrossRef]
- Karr, J.R. Defining and measuring river health. Freshw. Biol. 1999, 41, 221–234. [Google Scholar] [CrossRef]
- Rios, S.L.; Bailey, R.C. Relationship between Riparian Vegetation and Stream Benthic Communities at Three Spatial Scales. Hydrobiologia 2006, 553, 153–160. [Google Scholar] [CrossRef]
- Bracken, L.J.; Croke, J. The concept of hydrological connectivity and its contribution to understanding runoff-dominated geomorphic systems. Hydrol. Process. 2007, 21, 1749–1763. [Google Scholar] [CrossRef]
- Litschert, S.E.; MacDonald, L.H. Frequency and characteristics of sediment delivery pathways from forest harvest units to streams. For. Ecol. Manag. 2009, 259, 143–150. [Google Scholar] [CrossRef]
- Kreutzweiser, D.; Capell, S.; Good, K.; Holmes, S. Sediment deposition in streams adjacent to upland clearcuts and partially harvested riparian buffers in boreal forest catchments. For. Ecol. Manag. 2009, 258, 1578–1585. [Google Scholar] [CrossRef]
- Lynch, J.A.; Rishel, G.B.; Corbett, E.S. Thermal alteration of streams draining clearcut watersheds: Quantification and biological implications. Hydrobiologia 1984, 111, 161–169. [Google Scholar] [CrossRef]
- Sponseller, R.A.; Benfield, E.F.; Valett, H.M.; Issues, A. Relationships between land use, spatial scale and stream macroinvertebrate communities. Freshw. Biol. 2001, 46, 1409–1424. [Google Scholar] [CrossRef]
- Newbold, J.D.; Erman, D.C.; Roby, K.B.; Ridge, O. Effects of Logging on Macroinvertebrates in Streams With and Without Buffer Strips. Can. J. Fish. Aquat. Sci. 1980, 37, 1076–1085. [Google Scholar] [CrossRef]
- Gurtz, M.E.; Wallace, J.B. Substrate-Mediated Response of Stream Invertebrates to Disturbance. Ecology 1984, 65, 1556–1559. [Google Scholar] [CrossRef]
- Noel, D.S.; Martin, C.W.; Federer, C.A. Effects of forest clearcutting in New England on stream macroinvertebrates and periphyton. Environ. Manag. 1986, 10, 661–670. [Google Scholar] [CrossRef]
- Stone, M.K.; Wallace, J.B. Long-term recovery of a mountain stream from clear-cut logging: The effects of forest succession on benthic invertebrate community structure. Freshw. Biol. 1998, 39, 151–169. [Google Scholar] [CrossRef]
- Lynch, J.A.; Corbett, E.S.; Mussallem, K. Best management practices for controlling nonpoint-source pollution on forested watersheds. J. Soil Water Conserv. 1985, 40, 164–167. [Google Scholar]
- Binkley, D.; Brown, T. Effects of Forest and Range Management on Water Quality; U.S. Government Publishing Office: Washington, DC, USA, 1993.
- Stringer, J.W.; Perkins, C. FOR-67: Kentucky Forest Practice Guidelines for Water Quality Management; University of Kentuck Cooperative Extension Service: Lexington, KY, USA, 2001. [Google Scholar]
- Witt, E. Evaluating Streamside Management Zone Effectiveness in Forested Watersheds of the Cumberland Plateau. Ph.D. Thesis, University of Kentucky, Lexington, KY, USA, 2012. [Google Scholar]
- Arthur, M.; Coltharp, G.B.; Brown, D.L. Effects of best management practices on forest stream water quality in Eastern Kentucky. J. Am. Water Resour. Assoc. 1998, 34, 481–495. [Google Scholar] [CrossRef]
- Clinton, B.D. Stream water responses to timber harvest: Riparian buffer width effectiveness. For. Ecol. Manag. 2011, 261, 979–988. [Google Scholar] [CrossRef]
- Rudolph, D.; Dickson, J. Streamside zone width and amphibian and reptile abundance. Southwest. Nat. 1990, 35, 472–476. [Google Scholar] [CrossRef]
- Thurmond, D. Effect of streamside management zone width on avifauna communities. South. J. Appl. For. 1995, 19, 166–169. [Google Scholar]
- Vowell, J.L. Using stream bioassessment to monitor best management practice effectiveness. For. Ecol. Manag. 2001, 143, 237–244. [Google Scholar] [CrossRef]
- Stringer, J.; Lowe, L.; Smidt, M.; Perkins, C. Field Guide to Best Management Practices for Timber Harvesting in Kentucky; University of Kentucky Cooperative Extension Service: Lexington, KY, USA, 1997. [Google Scholar]
- Stringer, J.; Thompson, A. Comparison of forestry best management practices, Part I: Streamside management zones. For. Landowner 2000, 59, 22–27. [Google Scholar]
- McDowell, R.C. The Geology of Kentucky—A Text to Accompany the Geologic Map of Kentucky; U.S.G.P.O.: Washington, DC, USA, 1986.
- Woods, A.; Omernik, J.; Martin, W.; Pond, G.; Andrews, W.; Call, S.; Comstock, J.; Taylor, D. Ecoregions of Kentucky (Color Poster with Map, Descriptive Text, Summary Tables, and Photographs); U.S. Geological Survey: Reston, VA, USA, 2002.
- Smith, C.H.; Lamson, N.I.; Miller, G.W. An esthetic alternative to clearcutting? Deferment cutting in easter hardwoods. J. For. 1989, 87, 14–18. [Google Scholar]
- Miller, G.W.; Kochenderfer, J.N.; Fekedulegn, D.B. Influence of individual reserve trees on nearby reproduction in two-aged Appalachian hardwood stands. For. Ecol. Manag. 2006, 224, 241–251. [Google Scholar] [CrossRef]
- Dillaway, D.N.; Stringer, J.W.; Rieske, L.K. Light availability influences root carbohydrates, and potentially vigor, in white oak advance regeneration. For. Ecol. Manag. 2007, 250, 227–233. [Google Scholar] [CrossRef]
- Merritt, R.W.; Cummins, K.W. An Introduction to the Aquatic Insects of North America, 3rd ed.; Kendall Hunt Pub Co.: Dubuque, IA, USA, 1996. [Google Scholar]
- Kerans, B.; Karr, J.R. A benthic index of biotic integrity (B-IBI) for rivers of the Tennessee Valley. Ecol. Appl. 1994, 4, 768–785. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish; EPA 841-B-99-002; U.S. Environmental Protection Agency, Office of Water: Washington, DC, USA, 1999.
- Hilsenhoff, W.L. Using a Biotic Index to Evaluate Water Quality in Streams; Wisconsin Department of Natural Resources: Madison, WI, USA, 1982.
- Lenat, D.R. A biotic index for the southeastern United States: Derivation and list of tolerance values, with criteria for assigning water-quality ratings. J. N. Am. Benthol. Soc. 1993, 12, 279–290. [Google Scholar] [CrossRef]
- Pond, G.J.; Call, S.M.; Brumley, J.F.; Compton, M.C. The Kentucky Macroinvertebrate Bioassessment Index; Kentucky Department for Environmental Protection, Division of Water: Frankfort, KY, USA, 2003.
- Magurran, A.E. Ecological Diversity and Its Measure; Princeton University Press: Princeton, NJ, USA, 1988. [Google Scholar]
- McCune, B.; Grace, J.B. Analysis of Ecological Communities; MjM Software Design: Gleneden Beach, OR, USA, 2002. [Google Scholar]
- McCune, B.; Medford, M.J. PC-ORD: Multivariate Analysis of Ecological Data; MjM Software Design: Gleneden Beach, OR, USA, 1999. [Google Scholar]
- Haggerty, S.M.; Batzer, D.P.; Jackson, C.R. Macroinvertebrate response to logging in coastal headwater streams of Washington, USA. Can. J. Fish. Aquat. Sci. 2004, 61, 529–537. [Google Scholar] [CrossRef]
- Moldenke, A.R.; Ver, L.C. Effects of clearcutting and riparian buffers on the yield of adult aquatic macroinvertebrates from headwater streams. For. Sci. 2007, 53, 308–319. [Google Scholar]
- Earson, S.C.F.P.; Pearson, S.F.; Manuwal, D.A. Breeding Bird Response To Riparian Buffer Width In Managed Pacific Northwest Douglas-Fir Forests. Ecol. Appl. 2001, 11, 840–853. [Google Scholar] [CrossRef]
- Miller, D.A.; Thill, R.E.; Melchiors, M.A.; Wigley, T.B.; Tappe, P.A. Small mammal communities of streamside management zones in intensively managed pine forests of Arkansas. For. Ecol. Manag. 2004, 203, 381–393. [Google Scholar] [CrossRef]
- Lorion, C.M.; Kennedy, B.P. Riparian forest buffers mitigate the effects of deforestation on fish assemblages in tropical headwater streams. Ecol. Appl. 2009, 19, 468–79. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.V.; Aguila, Y.; Brown, K.B.; Fowler, W.P. Responses of benthic macroinvertebrates in small intermittent streams to silvicultural practices. Hydrobiologia 1997, 347, 119–125. [Google Scholar] [CrossRef]
- Carroll, G.D.; Schoenholtz, S.H.; Young, B.W.; Dibble, E.D. Effectiveness of forestry streamside management zones in the sand-clay hills of Mississippi: Early indications. Water Air Soil Pollut. Focus 2004, 4, 275–296. [Google Scholar] [CrossRef]
- Duncan, W.F.A.; Brusven, M.A. Benthic macroinvertebrates in logged and unlogged low-order southeast Alaskan streams. Freshw. Invertebr. Biol. 1985, 4, 125–132. [Google Scholar]
- Grubbs, S. Influence of flow permanence on headwater macroinvertebrate communities in a Cumberland Plateau watershed, USA. Aquat. Ecol. 2011, 45, 185–195. [Google Scholar] [CrossRef]
- Wallace, J.B.; Webster, J.R. The role of macroinvertebrates in stream ecosystem function. Annu. Rev. Entomol. 1996, 41, 115–139. [Google Scholar] [CrossRef] [PubMed]
- Bowker, D.W. Forest Harvest Equipment Movement and Sediment Delivery to Streams. Master’s Thesis, University of Kentucky, Lexington, KY, USA, 2013. [Google Scholar]
- Carlson, J.Y.; Andrus, C.W.; Froehlich, H.A. Woody debris, channel features, and macroinvertebrates of streams with logged and undisturbed riparian timber in Northeastern Oregon, U.S.A. Can. J. Fish. Aquat. Sci. 1990, 47, 1103–1111. [Google Scholar] [CrossRef]
- Flecker, A.S.; Feifarek, B. Disturbance and the temporal variability of invertebrate assemblages in two Andean streams. Freshw. Biol. 1994, 31, 131–142. [Google Scholar] [CrossRef]
- Death, R.G. The effect of patch disturbance on stream invertebrate community structure: The influence of disturbance history. Oecologia 1996, 108, 567–576. [Google Scholar] [CrossRef]
- Richards, C.; Minshall, G. The influence of periphyton abundance on Baetis bicaudatus distribution and colonization in a small stream. J. N. Am. Benthol. Soc. 1988, 7, 77–86. [Google Scholar] [CrossRef]
- Boulton, A.J.; Peterson, C.G.; Grimm, N.B.; Fisher, S.G. Stability of an aquatic macroinvertebrate community in a multiyear hydrologic disturbance regime. Ecology 1992, 73, 2192–2207. [Google Scholar] [CrossRef]
- Ruse, L.P. Chironomid emergence from an English chalk stream during a 3 year study. Arch. Hydrobiol. 1995, 133, 223–244. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 1–26. [Google Scholar] [CrossRef]
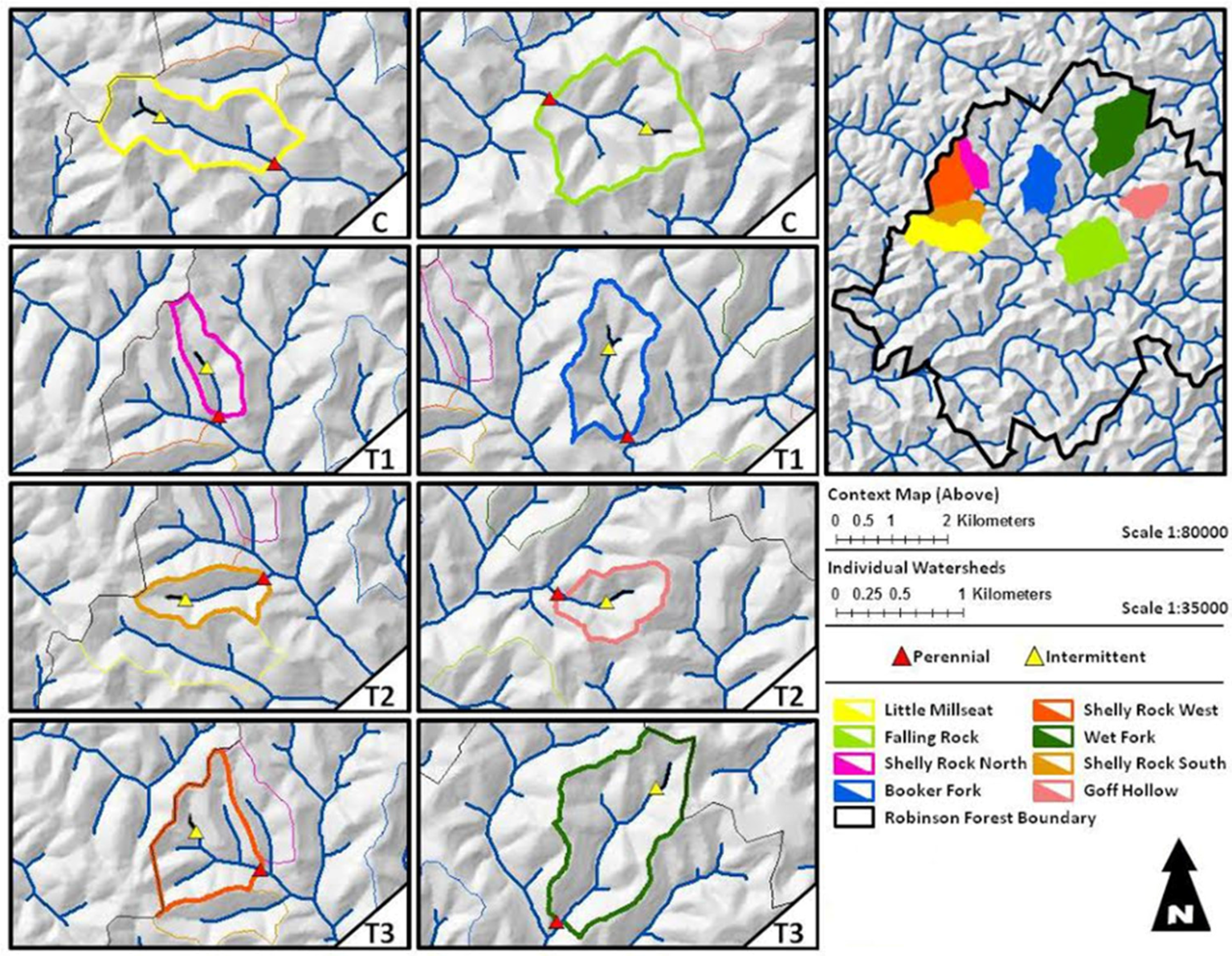
| Watershed Name | Flow Permanence Class | Treatment | Area (ha) | Bank Full Width (m) | Drainage Density (m/m2) | Aspect |
|---|---|---|---|---|---|---|
| Little Millseat | Intermittent | Control | 27 | 1.38 | 0.0050 | Southeast |
| Perennial | 79 | 2.65 | 0.0048 | |||
| Falling Rock | Intermittent | Control | 25 | 1.30 | 0.0071 | Northeast |
| Perennial | 97 | 1.99 | 0.0038 | |||
| Booker Hollow | Intermittent | T1 | 27 | 3.14 | 0.0036 | Northeast |
| Perennial | 59 | 3.19 | 0.0047 | |||
| North Shelly Rock | Intermittent | T1 | 16 | 2.44 | 0.0061 | South |
| Perennial | 27 | 3.08 | 0.0051 | |||
| South Shelly Rock | Intermittent | T2 | 19 | 2.56 | 0.0040 | East |
| Perennial | 33 | 3.27 | 0.0045 | |||
| Goff Hollow | Intermittent | T2 | 31 | 2.44 | 0.0023 | Northeast |
| Perennial | 38 | 3.08 | 0.0058 | |||
| Wet Fork | Intermittent | T3 | 32 | 2.30 | 0.0061 | Southwest |
| Perennial | 112 | 4.74 | 0.0046 | |||
| West Shelly Rock | Intermittent | T3 | 18 | 3.14 | 0.0106 | Southeast |
| Perennial | 72 | 3.56 | 0.0057 |
| Response Variable | Global F/p | 2004 Intermittent | ||
|---|---|---|---|---|
| SMZ | Season | SMZ × Season | ||
| Chironomidae | 5.1/<0.01 | 1.0/0.41 | 31.6/<0.01 | 0.2/0.90 |
| Leuctra | 5.6/<0.01 | 1.3/0.30 | 35.7/<0.01 | 0.2/0.93 |
| Ephemerella | 0.8/0.53 | 1.1/0.38 | 0.61/0.44 | 0.7/0.56 |
| Paraleptophlebia | 11.1/<0.01 | 3.6/0.02 | 60.6/<0.01 | 1.1/0.38 |
| Baetis | 8.7/<0.01 | 2.6/0.07 | 45.4/<0.01 | 2.2/0.10 |
| Collector–Filterer | 2.8/0.02 | 1.5/0.22 | 4.8/0.03 | 4.0/0.10 |
| Collector–Gatherer | 6.8/<0.01 | 2.3/0.10 | 39.1/<0.01 | 0.2/0.93 |
| Scraper | 5.9/<0.01 | 3.6/0.02 | 27.1/<0.01 | 1.5/0.25 |
| Shredder | 4.7/<0.01 | 0.4/0.77 | 30.1/<0.01 | 0.5/0.68 |
| Predator | 3.8/<0.01 | 1.2/0.34 | 21.2/<0.01 | 0.3/0.80 |
| %EPT | 0.8/0.59 | 1.0/0.42 | 1.1/0.31 | 0.6/0.61 |
| Hilsenhoff | 1.8/0.12 | 2.9/0.05 | 2.7/0.11 | 0.4/0.73 |
| Simpson Diversity | 0.8/0.59 | 1.4/0.27 | 0.2/0.63 | 0.5/0.69 |
| Total Abundance | 4.1/<0.01 | 0.8/0.52 | 24.8/<0.01 | 0.2/0.93 |
| Genus Richness | 1.5/0.20 | 0.4/0.73 | 6.5/0.02 | 0.8/0.48 |
| 2004 Perennial | ||||
| Response Variable | Global F/p | SMZ | Season | SMZ × Season |
| Chironomidae | 3.1/<0.01 | 1.0/0.39 | 11.2/<0.01 | 2.5/0.08 |
| Leuctra | 1.4/0.25 | 0.1/0.96 | 9.5/<0.01 | 0.1/0.99 |
| Ephemerella | 8.2/ <0.01 | 3.6/0.02 | 41.7/<0.01 | 1.0/0.39 |
| Paraleptophlebia | 2.3/0.05 | 0.7/0.54 | 13.6/<0.01 | 0.5/0.98 |
| Baetis | 18.5/<0.01 | 1.2/0.34 | 121.3/<0.01 | 1.2/0.34 |
| Collector–Filterer | 0.7/0.65 | 1.2/0.33 | 0.3/0.57 | 0.6/0.65 |
| Collector–Gatherer | 0.7/0.67 | 0.1/0.50 | 0.1/0.78 | 0.8/0.51 |
| Scraper | 3.3/<0.01 | 2.1/0.13 | 9.3/<0.01 | 2.4/0.08 |
| Shredder | 0.8/0.61 | 1.9/0.18 | 1.9/0.18 | 0.4/0.76 |
| Predator | 0.7/0.70 | 1.5/0.23 | 1.5/0.23 | 0.1/0.94 |
| %EPT | 1.3/0.28 | 1.5/0.24 | 1.5/0.22 | 1.4/0.27 |
| Hilsenhoff | 13.7/<0.01 | 5.5/<0.01 | 70.3/<0.01 | 1.0/0.42 |
| Simpson Diversity | 1.2/0.31 | 1.1/0.36 | 0.7/0.40 | 1.8/0.16 |
| Total Abundance | 1.3/0.26 | 0.8/0.48 | 4.7/0.04 | 0.7/0.56 |
| Genus Richness | 2.5/0.03 | 2.5/0.07 | 9.3/<0.01 | 0.6/0.61 |
| Response Variable | 2013 Intermittent | |||
|---|---|---|---|---|
| Global F | SMZ | Season | SMZ × Season | |
| Paraleptophlebia | 3.3/<0.01 | 3.0/0.04 | 3.9/0.05 | 3.3/0.03 |
| Chironomidae | 2.6/0.03 | 0.4/0.79 | 14.6/<0.01 | 0.7/0.54 |
| Ephemerella | 1.9/0.09 | 1.1/0.35 | 2.1/0.15 | 2.6/0.06 |
| Ameletus | 0.7/0.65 | 0.7/0.54 | 2.7/0.11 | 0.1/0.98 |
| Hexatoma | 0.5/0.81 | 0.3/0.82 | 0.6/0.43 | 0.7/0.55 |
| Collector–Filterer | 1.7/0.13 | 2.9/0.05 | 2.5/0.12 | 0.2/0.90 |
| Collector–Gatherer | 3.8/<0.01 | 3.1/0.04 | 8.6/<0.01 | 2.9/0.05 |
| Scraper | 1.2/0.34 | 2.0/0.13 | 0.01/0.92 | 0.7/0.52 |
| Shredder | 4.4/<0.01 | 1.0/0.40 | 25.6/<0.01 | 0.7/0.59 |
| Predator | 0.5/0.85 | 0.6/0.65 | 0.1/0.78 | 0.5/0.67 |
| %EPT | 0.9/0.54 | 1.5/0.24 | 1.3/0.27 | 0.1/0.94 |
| Hilsenhoff | 1.2/0.31 | 0.7/0.59 | 5.7/0.02 | 0.3/0.81 |
| Simpson Diversity | 1.8/0.12 | 1.8/0.16 | 2.8/0.10 | 1.4/0.25 |
| Total Abundance | 4.2/<0.01 | 3.0/0.04 | 12.3/<0.01 | 2.6/0.07 |
| Genus Richness | 1.7/0.13 | 1.8/0.17 | 6.5/0.02 | 0.1/0.94 |
| 2013 Perennial | ||||
| Response Variable | Global F | SMZ | Season | SMZ × Season |
| Paraleptophlebia | 0.78/0.61 | 1.3/0.31 | 1.5/0.22 | 0.1/0.98 |
| Chironomidae | 6.9/<0.01 | 8.7/<0.01 | 4.3/0.05 | 6.1/<0.01 |
| Ephemerella | 5.1/<0.01 | 5.8/<0.01 | 9.3/<0.01 | 3.0/0.04 |
| Ameletus | 3.1/0.01 | 5.2/<0.01 | 5.8/0.02 | 0.1/0.94 |
| Hexatoma | 2.4/0.04 | 2.0/0.13 | 7.4/<0.01 | 1.2/0.33 |
| Collector–Filterer | 3.4/<0.01 | 7.5/<0.01 | 0.4/0.54 | 0.4/0.78 |
| Collector–Gatherer | 2.4/0.04 | 3.8/0.18 | 0.5/0.50 | 1.7/0.18 |
| Scraper | 3.5/<0.01 | 5.3/<0.01 | 0.7/0.42 | 2.5/0.07 |
| Shredder | 3.0/0.02 | 1.2/0.31 | 8.7/<0.01 | 2.8/0.05 |
| Predator | 1.9/0.09 | 1.6/0.19 | 1.3/0.26 | 2.4 / 0.08 |
| %EPT | 1.3/0.28 | 1.1/0.38 | 1.8/0.18 | 1.4/0.27 |
| Hilsenhoff | 1.8/0.12 | 2.3/0.09 | 0.3/0.60 | 1.7/0.18 |
| Simpson Diversity | 1.1/0.41 | 0.6/0.64 | 0.7/0.40 | 1.7/0.19 |
| Total Abundance | 3.0/0.02 | 3.8/0.02 | 1.9/0.17 | 2.4/0.08 |
| Genus Richness | 1.7/0.14 | 2.1/0.12 | 1.5/0.24 | 1.4/0.26 |
| 2004 Intermittent | 2013 Intermittent | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | T1 | T2 | T3 | Control | T1 | T2 | T3 | ||
| T1 | 0.73 | – | – | – | T1 | 0.63 | – | – | – |
| T2 | 0.68 | 0.75 | – | – | T2 | 0.73 | 0.62 | – | – |
| T3 | 0.68 | 0.77 | 0.76 | – | T3 | 0.71 | 0.74 | 0.73 | – |
| 2004 Perennial | 2013 Perennial | ||||||||
| Control | T1 | T2 | T3 | Control | T1 | T2 | T3 | ||
| T1 | 0.83 | – | – | – | T1 | 0.65 | – | – | – |
| T2 | 0.67 | 0.75 | – | – | T2 | 0.64 | 0.75 | – | – |
| T3 | 0.81 | 0.84 | 0.73 | – | T3 | 0.66 | 0.77 | 0.69 | – |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adkins, J.K.; Barton, C.D.; Grubbs, S.; Stringer, J.W.; Kolka, R.K. Assessment of Streamside Management Zones for Conserving Benthic Macroinvertebrate Communities Following Timber Harvest in Eastern Kentucky Headwater Catchments. Water 2016, 8, 261. https://doi.org/10.3390/w8060261
Adkins JK, Barton CD, Grubbs S, Stringer JW, Kolka RK. Assessment of Streamside Management Zones for Conserving Benthic Macroinvertebrate Communities Following Timber Harvest in Eastern Kentucky Headwater Catchments. Water. 2016; 8(6):261. https://doi.org/10.3390/w8060261
Chicago/Turabian StyleAdkins, Joshua K., Christopher D. Barton, Scott Grubbs, Jeffrey W. Stringer, and Randall K. Kolka. 2016. "Assessment of Streamside Management Zones for Conserving Benthic Macroinvertebrate Communities Following Timber Harvest in Eastern Kentucky Headwater Catchments" Water 8, no. 6: 261. https://doi.org/10.3390/w8060261
APA StyleAdkins, J. K., Barton, C. D., Grubbs, S., Stringer, J. W., & Kolka, R. K. (2016). Assessment of Streamside Management Zones for Conserving Benthic Macroinvertebrate Communities Following Timber Harvest in Eastern Kentucky Headwater Catchments. Water, 8(6), 261. https://doi.org/10.3390/w8060261








