Drought and Carbon Cycling of Grassland Ecosystems under Global Change: A Review
Abstract
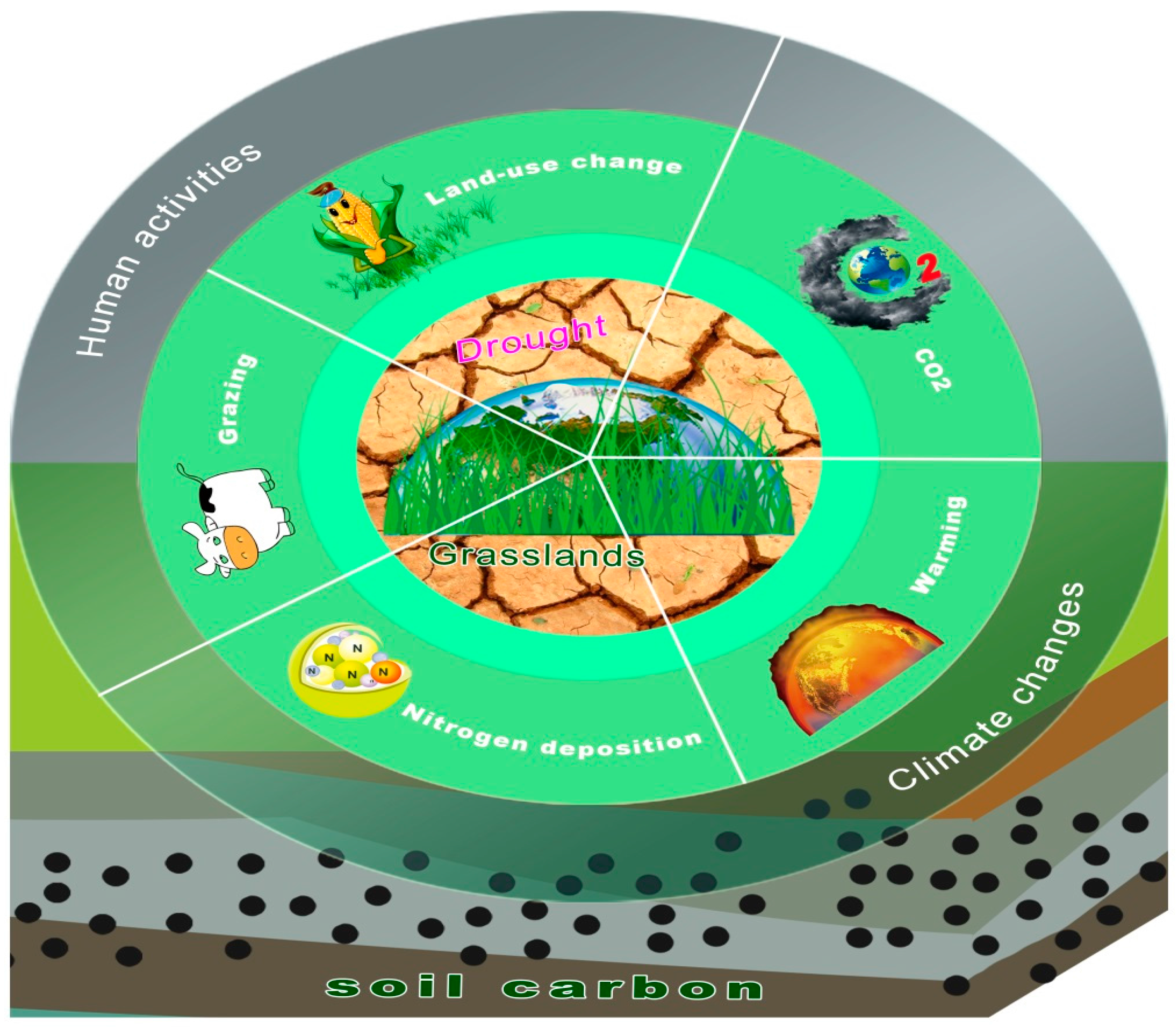
:1. Introduction
2. Drought and Grassland C Cycling under Global Change
2.1. Overview
2.2. Drought and Grassland C Cycling
2.3. Elevated CO2 Concentrations
2.4. Global Warming
2.5. N Deposition
2.6. Grazing
2.7. Land-Use Change
2.8. Grassland Ecosystem Properties
3. Syntheses and Perspectives
3.1. Combined Effects of Multiple Factors
- (1)
- Under particular scenarios such as droughts, recognizing the key environmental factors impacting grassland C cycling in the real, multi-factor world is important. It is necessary to carry out multi-factor experiments to determine the contribution of each factor and their interactive effects with other factors on grassland C cycling at the ecosystem scale.
- (2)
- The quantitative impacts between drought and other factors (e.g., elevated CO2 concentrations, global warming, N deposition, grazing, and other land-use change) on grassland C cycling are not clearly proposed at the ecosystem level.
- (3)
- The next challenge is to establish the quantitative relationships between different C fluxes and different global change factors under different drought scenarios as soon as possible.
- (4)
- Multi-factor response models should be developed with better coupling mechanisms to examine the interactive effects of global change multi-factors on the carbon and water processes of ecosystems, especially under different levels of drought stress or other extreme scenarios. The data–model fusion has become essential to assess the interactive effects of multiple factors in global change research.
3.2. A Framework for Assessment and Application of Combined Effects
- (1)
- Defines the intensity and duration of drought at which ecological thresholds are triggered;
- (2)
- Represents ecological mechanisms of response to drought under current and future global change scenarios at different spatial–temporal scales;
- (3)
- Assesses the hierarchical responses of an ecosystem to drought and global change, including individual, species, and ecosystem responses;
- (4)
- Quantifies the contribution of other global change drivers that prevent ecological thresholds triggered by droughts;
- (5)
- Provides a baseline to assess the impact of drought under global change;
- (6)
- Assesses the contribution of ecological thresholds to the fate of grassland C cycling;
- (7)
- Gives suggestions to managers can use to enhance C sink of grasslands.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Joos, O.; Hagedorn, F.; Heim, A.; Gilgen, A.; Schmidt, M.; Siegwolf, R.; Buchmann, N. Summer drought reduces total and litter-derived soil CO2 effluxes intemperate grassland–clues from a 13C litter addition experiment. Biogeosciences 2010, 7, 1031–1041. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Scurlock, J.; Hall, D. The global carbon sink: A grassland perspective. Glob. Chang. Biol. 1998, 4, 229–233. [Google Scholar] [CrossRef]
- Imer, D.; Merbold, L.; Eugster, W.; Buchmann, N. Temporal and spatial variations of soil CO2, CH4 and N2O fluxes at three differently managed grasslands. Biogeosciences 2013, 10, 5931–5945. [Google Scholar] [CrossRef]
- Abberton, M.; Conant, R.; Batello, C. Grassland Carbon Sequestration: Management, Policy and Economics; FAO: Rome, Italy, 2010; pp. 1–53. [Google Scholar]
- Cherwin, K.; Knapp, A. Unexpected patterns of sensitivity to drought in three semi-arid grasslands. Oecologia 2012, 169, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Stocker, T.F.; Dahe, Q.; Plattner, G.-K. Climate Change 2013: The Physical Science Basis. Working Group I Contribution to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Summary for Policymakers (IPCC, 2013); IPCC: Bern, Switzerland, 2013; pp. 1–33. [Google Scholar]
- Vogel, A.; Scherer-Lorenzen, M.; Weigelt, A. Grassland resistance and resilience after drought depends on management intensity and species richness. PLoS ONE 2012, 7, e36992. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Chen, J.; Dong, G.; Jiang, S.; Li, L.; Guo, J.; Shao, C. Heat waves reduce ecosystem carbon sink strength in a Eurasian meadow steppe. Environ. Res. 2015, 144, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Knapp, A.K.; Fay, P.A.; Blair, J.M.; Collins, S.L.; Smith, M.D.; Carlisle, J.D.; Harper, C.W.; Danner, B.T.; Lett, M.S.; McCarron, J.K. Rainfall variability, carbon cycling, and plant species diversity in a mesic grassland. Science 2002, 298, 2202–2205. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.D. The ecological role of climate extremes: Current understanding and future prospects. J. Ecol. 2011, 99, 651–655. [Google Scholar] [CrossRef]
- Smith, M.D.; Knapp, A.K.; Collins, S.L. A framework for assessing ecosystem dynamics in response to chronic resource alterations induced by global change. Ecology 2009, 90, 3279–3289. [Google Scholar] [CrossRef] [PubMed]
- Brookshire, E.; Weaver, T. Long-term decline in grassland productivity driven by increasing dryness. Nat. Commun. 2015, 6, 7148–7152. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.; Reichstein, M.; Bahn, M.; Thonicke, K.; Frank, D.; Mahecha, M.D.; Smith, P.; Velde, M.; Vicca, S.; Babst, F. Effects of climate extremes on the terrestrial carbon cycle: Concepts, processes and potential future impacts. Glob. Chang. Biol. 2015, 21, 2861–2880. [Google Scholar] [CrossRef] [PubMed]
- Bahn, M.; Reichstein, M.; Guan, K.; Moreno, J.; Williams, C. Preface: Climate extremes and biogeochemical cycles in the terrestrial biosphere: Impacts and feedbacks across scales. Biogeosciences 2015, 12, 4827–4830. [Google Scholar] [CrossRef]
- Yuan, W.; Cai, W.; Chen, Y.; Liu, S.; Dong, W.; Zhang, H.; Yu, G.; Chen, Z.; He, H.; Guo, W. Severe summer heatwave and drought strongly reduced carbon uptake in Southern China. Sci. Rep. 2016, 6, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Heimann, M.; Reichstein, M. Terrestrial ecosystem carbon dynamics and climate feedbacks. Nature 2008, 451, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Dieleman, W.I.; Vicca, S.; Dijkstra, F.A.; Hagedorn, F.; Hovenden, M.J.; Larsen, K.S.; Morgan, J.A.; Volder, A.; Beier, C.; Dukes, J.S. Simple additive effects are rare: A quantitative review of plant biomass and soil process responses to combined manipulations of CO2 and temperature. Glob. Chang. Biol. 2012, 18, 2681–2693. [Google Scholar] [CrossRef] [PubMed]
- Allaby, M. Grasslands; Infobase Publishing: New York, NY, USA, 2009; pp. 1–126. [Google Scholar]
- De Boeck, H.J.; Dreesen, F.E.; Janssens, I.A.; Nijs, I. Whole-system responses of experimental plant communities to climate extremes imposed in different seasons. New Phytol. 2011, 189, 806–817. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.J. Grasses and Grassland Ecology; Oxford University Press: Oxford, UK, 2009; pp. 1–300. [Google Scholar]
- Tardieu, F.; Granier, C.; Muller, B. Water deficit and growth. Co-ordinating processes without an orchestrator? Curr. Opin. Plant Boil. 2011, 14, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Ciais, P.; Viovy, N.; Vuichard, N.; Herrero, M.; Havlík, P.; Wang, X.; Sultan, B.; Soussana, J.F. Effect of climate change, CO2 trends, nitrogen addition, and land-cover and management intensity changes on the carbon balance of European grasslands. Glob. Chang. Biol. 2016, 22, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.D.; Knapp, A.K. Physiological and morphological traits of exotic, invasive exotic, and native plant species in tallgrass prairie. Int. J. Plant Sci. 2001, 162, 785–792. [Google Scholar] [CrossRef]
- Dai, A. Drought under global warming: A review. WIREs Clim. Chang. 2011, 2, 45–65. [Google Scholar] [CrossRef]
- Ciais, P.; Reichstein, M.; Viovy, N.; Granier, A.; Ogée, J.; Allard, V.; Aubinet, M.; Buchmann, N.; Bernhofer, C.; Carrara, A. Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature 2005, 437, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.; Mateus, J.; Aires, L.; Pita, G.; Pio, C.; David, J.; Andrade, V.; Banza, J.; David, T.; Paço, T. Net ecosystem carbon exchange in three contrasting Mediterranean ecosystems—The effect of drought. Biogeosciences 2007, 4, 791–802. [Google Scholar] [CrossRef]
- Van Eekeren, N.; Bommelé, L.; Bloem, J.; Schouten, T.; Rutgers, M.; de Goede, R.; Reheul, D.; Brussaard, L. Soil biological quality after 36 years of ley-arable cropping, permanent grassland and permanent arable cropping. Appl. Soil Ecol. 2008, 40, 432–446. [Google Scholar] [CrossRef]
- Jentsch, A.; Kreyling, J.; Beierkuhnlein, C. A new generation of climate-change experiments: Events, not trends. Front. Ecol. Environ. 2007, 5, 365–374. [Google Scholar] [CrossRef]
- White, P.S.; Jentsch, A. The search for generality in studies of disturbance and ecosystem dynamics. In Progress in Botany; Springer: Berlin, Germany, 2001; pp. 399–450. [Google Scholar]
- Xiao, J.; Zhuang, Q.; Liang, E.; McGuire, A.D.; Moody, A.; Kicklighter, D.W.; Shao, X.; Melillo, J.M. Twentieth-Century Droughts and Their Impacts on Terrestrial Carbon Cycling in China. Earth Interact. 2009, 13, 1–31. [Google Scholar] [CrossRef]
- Thiessen, S.; Gleixner, G.; Wutzler, T.; Reichstein, M. Both priming and temperature sensitivity of soil organic matter decomposition depend on microbial biomass—An incubation study. Soil Biol. Biochem. 2013, 57, 739–748. [Google Scholar] [CrossRef]
- Hufkens, K.; Keenan, T.F.; Flanagan, L.B.; Scott, R.L.; Bernacchi, C.J.; Joo, E.; Brunsell, N.A.; Verfaillie, J.; Richardson, A.D. Productivity of North American grasslands is increased under future climate scenarios despite rising aridity. Nat. Clim. Chang. 2016. [Google Scholar] [CrossRef]
- Leitinger, G.; Ruggenthaler, R.; Hammerle, A.; Lavorel, S.; Schirpke, U.; Clement, J.C.; Lamarque, P.; Obojes, N.; Tappeiner, U. Impact of droughts on water provision in managed alpine grasslands in two climatically different regions of the Alps. Ecohydrology 2015, 8, 1600–1613. [Google Scholar] [CrossRef] [PubMed]
- Hasibeder, R.; Fuchslueger, L.; Richter, A.; Bahn, M. Summer drought alters carbon allocation to roots and root respiration in mountain grassland. New Phytol. 2015, 205, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Jentsch, A.; Kreyling, J.; Elmer, M.; Gellesch, E.; Glaser, B.; Grant, K.; Hein, R.; Lara, M.; Mirzae, H.; Nadler, S.E. Climate extremes initiate ecosystem-regulating functions while maintaining productivity. J. Ecol. 2011, 99, 689–702. [Google Scholar] [CrossRef]
- Nemani, R.R.; Keeling, C.D.; Hashimoto, H.; Jolly, W.M.; Piper, S.C.; Tucker, C.J.; Myneni, R.B.; Running, S.W. Climate-driven increases in global terrestrial net primary production from 1982 to 1999. Science 2003, 300, 1560–1563. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, G. Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J. Exp. Bot. 2008, 59, 3317–3325. [Google Scholar] [CrossRef] [PubMed]
- Hoover, D.L.; Rogers, B.M. Not all droughts are created equal: The impacts of interannual drought pattern and magnitude on grassland carbon cycling. Glob. Chang. Biol. 2015, 22, 1809–1820. [Google Scholar] [CrossRef] [PubMed]
- Canarini, A.; Dijkstra, F.A. Dry-rewetting cycles regulate wheat carbon rhizodeposition, stabilization and nitrogen cycling. Soil Biol. Biochem. 2015, 81, 195–203. [Google Scholar] [CrossRef]
- Xu, Z.-Z.; Zhou, G.-S. Effects of water stress and high nocturnal temperature on photosynthesis and nitrogen level of a perennial grass Leymus chinensis. Plant Soil 2005, 269, 131–139. [Google Scholar] [CrossRef]
- Harper, C.W.; Blair, J.M.; Fay, P.A.; Knapp, A.K.; Carlisle, J.D. Increased rainfall variability and reduced rainfall amount decreases soil CO2 flux in a grassland ecosystem. Glob. Chang. Biol. 2005, 11, 322–334. [Google Scholar] [CrossRef]
- Fay, P.A.; Carlisle, J.D.; Knapp, A.K.; Blair, J.M.; Collins, S.L. Productivity responses to altered rainfall patterns in a C4-dominated grassland. Oecologia 2003, 137, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Potts, D.L.; Huxman, T.E.; Cable, J.M.; English, N.B.; Ignace, D.D.; Eilts, J.A.; Mason, M.J.; Weltzin, J.F.; Williams, D.G. Antecedent moisture and seasonal precipitation influence the response of canopy-scale carbon and water exchange to rainfall pulses in a semi-arid grassland. New Phytol. 2006, 170, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, A.; Fort, H. Catastrophic phase transitions and early warnings in a spatial ecological model. J. Stat. Mech. Theory Exp. 2009, 5, 1255–1274. [Google Scholar] [CrossRef]
- Heschel, M.S.; Riginos, C. Mechanisms of selection for drought stress tolerance and avoidance in Impatiens capensis (Balsaminaceae). Am. J. Bot. 2005, 92, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Blum, A. Plant Water Relations, Plant Stress and Plant Production; Springer: Berlin, Germany, 2011; pp. 1–305. [Google Scholar]
- Heitschmidt, R.; Haferkamp, M. Ecological consequences of drought and grazing on grasslands of the Northern Great Plains. In Changing Precipitation Regimes and Terrestrial Ecosystems: A North American Perspective; University of Arizona Press: Tucson, AZ, USA, 2003; pp. 207–226. [Google Scholar]
- Bloor, J.M.; Bardgett, R.D. Stability of above-ground and below-ground processes to extreme drought in model grassland ecosystems: Interactions with plant species diversity and soil nitrogen availability. Perspect. Plant Ecol. Evol. Syst. 2012, 14, 193–204. [Google Scholar] [CrossRef]
- Gilgen, A.; Buchmann, N. Response of temperate grasslands at different altitudes to simulated summer drought differed but scaled with annual precipitation. Biogeosci. Discuss. 2009, 6, 2525–2539. [Google Scholar] [CrossRef]
- Shinoda, M.; Nachinshonhor, G.; Nemoto, M. Impact of drought on vegetation dynamics of the Mongolian steppe: A field experiment. J. Arid Environ. 2010, 74, 63–69. [Google Scholar] [CrossRef]
- Vries, F.T.D.; Liiri, M.E.; Bjørnlund, L.; Setälä, H.M.; Christensen, S.; Bardgett, R.D. Legacy effects of drought on plant growth and the soil food web. Oecologia 2012, 170, 821–833. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.E.; Wallenstein, M.D. Climate change alters ecological strategies of soil bacteria. Ecol. Lett. 2013, 17, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Fuchslueger, L.; Bahn, M.; Fritz, K.; Hasibeder, R.; Richter, A. Experimental drought reduces the transfer of recently fixed plant carbon to soil microbes and alters the bacterial community composition in a mountain meadow. New Phytol. 2014, 201, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.E.; Wallenstein, M.D. Soil microbial community response to drying and rewetting stress: Does historical precipitation regime matter? Biogeochemistry 2012, 109, 101–116. [Google Scholar] [CrossRef]
- Scott, R.L.; Biederman, J.A.; Hamerlynck, E.P.; Barron-Gafford, G.A. The carbon balance pivot point of southwestern US semiarid ecosystems: Insights from the 21st century drought. J. Geophys. Res. Biogeosci. 2015, 120, 2612–2624. [Google Scholar] [CrossRef]
- Liebig, M.; Kronberg, S.; Hendrickson, J.; Gross, J. Grazing management, season, and drought contributions to near-surface soil property dynamics in semiarid rangeland. Rangel. Ecol. Manag. 2014, 67, 266–274. [Google Scholar] [CrossRef]
- Williams, N.; Holland, K. The ecology and invasion history of hawkweeds (Hieracium species) in Australia. Plant Prot. Q. 2007, 22, 76–80. [Google Scholar]
- Reichstein, M.; Bahn, M.; Ciais, P.; Frank, D.; Mahecha, M.D.; Seneviratne, S.I.; Zscheischler, J.; Beer, C.; Buchmann, N.; Frank, D.C. Climate extremes and the carbon cycle. Nature 2013, 500, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Kreyling, J.; Beierkuhnlein, C.; Elmer, M.; Pritsch, K.; Radovski, M.; Schloter, M.; Wöllecke, J.; Jentsch, A. Soil biotic processes remain remarkably stable after 100-year extreme weather events in experimental grassland and heath. Plant Soil 2008, 308, 175–188. [Google Scholar] [CrossRef]
- Craine, J.M.; Towne, E.; Tolleson, D.; Nippert, J.B. Precipitation timing and grazer performance in a tallgrass prairie. Oikos 2013, 122, 191–198. [Google Scholar] [CrossRef]
- Schubert, S.D.; Suarez, M.J.; Pegion, P.J.; Koster, R.D.; Bacmeister, J.T. On the cause of the 1930s Dust Bowl. Science 2004, 303, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Van der Molen, M.; Dolman, A.; Ciais, P.; Eglin, T.; Gobron, N.; Law, B.; Meir, P.; Peters, W.; Phillips, O.; Reichstein, M. Drought and ecosystem carbon cycling. Agric. For. Meteorol. 2011, 151, 765–773. [Google Scholar] [CrossRef]
- Bradley, B.A.; Houghton, R.; Mustard, J.F.; Hamburg, S.P. Invasive grass reduces aboveground carbon stocks in shrublands of the Western US. Glob. Chang. Biol. 2006, 12, 1815–1822. [Google Scholar] [CrossRef]
- Martí-Roura, M.; Casals, P.; Romanyà, J. Temporal changes in soil organic C under Mediterranean shrublands and grasslands: Impact of fire and drought. Plant Soil 2011, 338, 289–300. [Google Scholar] [CrossRef]
- Louhaichi, M.; Tastad, A. The Syrian steppe: Past trends, current status, and future priorities. Rangelands 2010, 32, 2–7. [Google Scholar] [CrossRef]
- Davis, M.A.; Grime, J.P.; Thompson, K. Fluctuating resources in plant communities: A general theory of invasibility. J. Ecol. 2000, 88, 528–534. [Google Scholar] [CrossRef]
- Jentsch, A.; Beierkuhnlein, C. Research frontiers in climate change: Effects of extreme meteorological events on ecosystems. C. R. Geosci. 2008, 340, 621–628. [Google Scholar] [CrossRef]
- Ferlan, M.; Eler, K.; Simončič, P.; Batič, F.; Vodnik, D. Carbon and water flux patterns of a drought-prone mid-succession ecosystem developed on abandoned karst grassland. Agric. Ecosyst. Environ. 2016, 220, 152–163. [Google Scholar] [CrossRef]
- Tilman, D.; Reich, P.B.; Knops, J.; Wedin, D.; Mielke, T.; Lehman, C. Diversity and productivity in a long-term grassland experiment. Science 2001, 294, 843–845. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Wedin, D.; Knops, J. Productivity and sustainability influenced by biodiversity in grassland ecosystems. Nature 1996, 379, 718–720. [Google Scholar] [CrossRef]
- Smith, P.; Fang, C.; Dawson, J.J.; Moncrieff, J.B. Impact of global warming on soil organic carbon. Adv. Agron. 2008, 97, 1–43. [Google Scholar]
- Reich, P.B.; Hobbie, S.E.; Lee, T.D. Plant growth enhancement by elevated CO2 eliminated by joint water and nitrogen limitation. Nat. Geosci. 2014, 7, 920–924. [Google Scholar] [CrossRef]
- Campbell, B.; Smith, D.S.; Pastures, G. A synthesis of recent global change research on pasture and rangeland production: Reduced uncertainties and their management implications. Agric. Ecosyst. Environ. 2000, 82, 39–55. [Google Scholar] [CrossRef]
- Roy, J.; Piconcochard, C.; Augusti, A.; Benot, M.L.; Thiery, L.; Darsonville, O.; Landais, D.; Piel, C.; Defossez, M.; Devidal, S. Elevated CO2 maintains grassland net carbon uptake under a future heat and drought extreme. Proc. Natl. Acad. Sci. USA 2016, 113, 6224–6229. [Google Scholar] [CrossRef] [PubMed]
- Dukes, J.S.; Chiariello, N.R.; Cleland, E.E.; Moore, L.A.; Shaw, M.R.; Thayer, S.; Tobeck, T.; Mooney, H.A.; Field, C.B. Responses of grassland production to single and multiple global environmental changes. PLoS Biol. 2005, 3, e319. [Google Scholar] [CrossRef] [PubMed]
- Niklaus, P.A.; Spinnler, D.; Körner, C. Soil moisture dynamics of calcareous grassland under elevated CO2. Oecologia 1998, 117, 201–208. [Google Scholar] [CrossRef]
- Soussana, J.F.; Lüscher, A. Temperate grasslands and global atmospheric change: A review. Grass Forage Sci. 2007, 62, 127–134. [Google Scholar] [CrossRef]
- Manderscheid, R.; Weigel, H.-J. Drought stress effects on wheat are mitigated by atmospheric CO2 enrichment. Agron. Sustain. Dev. 2007, 27, 79–87. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, Y.; Cui, X.Y.; Zhao, H.; Xu, C.; Zhou, X.; Xu, Z. Responses of soil respiration and its components to drought stress. J. Soils Sediments 2014, 14, 99–109. [Google Scholar] [CrossRef]
- Soussana, J.; Casella, E.; Loiseau, P. Long-term effects of CO2 enrichment and temperature increase on a temperate grass sward. Plant Soil 1996, 182, 101–114. [Google Scholar] [CrossRef]
- Tubiello, F.N.; Soussana, J.-F.; Howden, S.M. Crop and pasture response to climate change. Proc. Natl. Acad. Sci. USA 2007, 104, 19686–19690. [Google Scholar] [CrossRef] [PubMed]
- Lehner, B.; Döll, P.; Alcamo, J.; Henrichs, T.; Kaspar, F. Estimating the impact of global change on flood and drought risks in Europe: A continental, integrated analysis. Clim. Chang. 2006, 75, 273–299. [Google Scholar] [CrossRef]
- Zhou, G.; Wang, Y.; Wang, S. Responses of grassland ecosystems to precipitation and land use along the Northeast China Transect. J. Veg. Sci. 2002, 13, 361–368. [Google Scholar] [CrossRef]
- Mcdowell, N.G. Mechanisms linking drought, hydraulics, carbon metabolism, and vegetation mortality. Plant Physiol. 2011, 155, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- McDowell, N.G.; Sevanto, S. The mechanisms of carbon starvation: How, when, or does it even occur at all? New Phytol. 2010, 186, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Bork, E.; Cahill, J.; Chang, S.; Deutsch, E.; Attaeian, B.; Wang, P.; Shore, B.; White, S. Rangeland Responses to Climate Change; Department of Agricultural, Food & Nutritional Science, Agriculture/Forestry Centre, University of Alberta Edmonton: Edmonton, AB, Canada, 2009; pp. 1–115. [Google Scholar]
- Wan, S.; Norby, R.J.; Ledford, J.; Weltzin, J.F. Responses of soil respiration to elevated CO2, air warming, and changing soil water availability in a model old-field grassland. Glob. Chang. Biol. 2007, 13, 2411–2424. [Google Scholar] [CrossRef]
- Lindberg, N.; Bengtsson, J. Recovery of forest soil fauna diversity and composition after repeated summer droughts. Oikos 2006, 114, 494–506. [Google Scholar] [CrossRef]
- De Boeck, H.D.; Lemmens, C.; Zavalloni, C.; Gielen, B.; Malchair, S.; Carnol, M.; Merckx, R.; Van den Berge, J.; Ceulemans, R.; Nijs, I. Biomass production in experimental grasslands of different species richness during three years of climate warming. Biogeosciences 2008, 5, 585–594. [Google Scholar] [CrossRef]
- Zavalloni, C.; Gielen, B.; Lemmens, C.; De Boeck, H.; Blasi, S.; Van den Bergh, S.; Nijs, I.; Ceulemans, R. Does a warmer climate with frequent mild water shortages protect grassland communities against a prolonged drought? Plant Soil 2008, 308, 119–130. [Google Scholar] [CrossRef]
- Lombardozzi, D.L.; Bonan, G.B.; Smith, N.G.; Dukes, J.S.; Fisher, R.A. Temperature acclimation of photosynthesis and respiration: A key uncertainty in the carbon cycle-climate feedback. Geophys. Res. Lett. 2015, 42, 8624–8631. [Google Scholar] [CrossRef]
- Melillo, J.; Steudler, P.; Aber, J.; Newkirk, K.; Lux, H.; Bowles, F.; Catricala, C.; Magill, A.; Ahrens, T.; Morrisseau, S. Soil warming and carbon-cycle feedbacks to the climate system. Science 2002, 298, 2173–2176. [Google Scholar] [CrossRef] [PubMed]
- Pendall, E.; Bridgham, S.; Hanson, P.J.; Hungate, B.; Kicklighter, D.W.; Johnson, D.W.; Law, B.E.; Luo, Y.; Megonigal, J.P.; Olsrud, M. Below-ground process responses to elevated CO2 and temperature: A discussion of observations, measurement methods, and models. New Phytol. 2004, 162, 311–322. [Google Scholar] [CrossRef]
- Sanaullah, M.; Chabbi, A.; Rumpel, C.; Kuzyakov, Y. Carbon allocation in grassland communities under drought stress followed by 14C pulse labeling. Soil Biol. Biochem. 2012, 55, 132–139. [Google Scholar] [CrossRef]
- Hoover, D.L.; Knapp, A.K.; Smith, M.D. The immediate and prolonged effects of climate extremes on soil respiration in a mesic grassland. J. Geophys. Res. Biogeosci. 2016, 121, 1–11. [Google Scholar] [CrossRef]
- Emmett, B.A.; Beier, C.; Estiarte, M.; Tietema, A.; Kristensen, H.L.; Williams, D.; Penuelas, J.; Schmidt, I.; Sowerby, A. The response of soil processes to climate change: Results from manipulation studies of shrublands across an environmental gradient. Ecosystems 2004, 7, 625–637. [Google Scholar] [CrossRef]
- Knapp, A.K.; Beier, C.; Briske, D.D.; Classen, A.T.; Luo, Y.; Reichstein, M.; Smith, M.D.; Smith, S.D.; Bell, J.E.; Fay, P.A. Consequences of more extreme precipitation regimes for terrestrial ecosystems. Bioscience 2008, 58, 811–821. [Google Scholar] [CrossRef]
- Davidson, E.A.; Janssens, I.A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 2006, 440, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Conant, R.T.; Drijber, R.A.; Haddix, M.L.; Parton, W.J.; Paul, E.A.; Plante, A.F.; Six, J.; Steinweg, J.M. Sensitivity of organic matter decomposition to warming varies with its quality. Glob. Chang. Biol. 2008, 14, 868–877. [Google Scholar] [CrossRef]
- Sanaullah, M.; Chabbi, A.; Girardin, C.; Durand, J.-L.; Poirier, M.; Rumpel, C. Effects of drought and elevated temperature on biochemical composition of forage plants and their impact on carbon storage in grassland soil. Plant Soil 2014, 374, 767–778. [Google Scholar] [CrossRef]
- Mowll, W.; Blumenthal, D.M.; Cherwin, K.; Smith, A.; Symstad, A.J.; Vermeire, L.T.; Collins, S.L.; Smith, M.D.; Knapp, A.K. Climatic controls of aboveground net primary production in semi-arid grasslands along a latitudinal gradient portend low sensitivity to warming. Oecologia 2015, 177, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Stursova, M.; Crenshaw, C.L.; Sinsabaugh, R.L. Microbial responses to long-term N deposition in a semiarid grassland. Microb. Ecol. 2006, 51, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Naeem, S.; Thompson, L.J.; Lawler, S.P.; Lawton, J.H.; Woodfin, R.M. Declining biodiversity can alter the performance of ecosystems. Nature 1994, 368, 734–737. [Google Scholar] [CrossRef]
- Borken, W.; Matzner, E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob. Chang. Biol. 2009, 15, 808–824. [Google Scholar] [CrossRef]
- Da Silva, E.C.; de Albuquerque, M.B.; de Azevedo Neto, A.D.; da Silva Junior, C.D. Drought and its consequences to plants—From individual to ecosystem. In Responses of Organisms to Water Stress; InTech: Rijeka, Croatia, 2013; pp. 17–47. [Google Scholar]
- Kopittke, G.; Tietema, A.; Verstraten, J. Soil acidification occurs under ambient conditions but is retarded by repeated drought: Results of a field-scale climate manipulation experiment. Sci. Total Environ. 2012, 439, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, G.; Wang, Y. Effects of drought and rewatering on carbon and nitrogen allocations in Leymus chinensis grass. J. Meteorol. Environ. 2007, 3, 65–71. [Google Scholar]
- Xi, N.; Carrère, P.; Bloor, J.M. Plant community responses to precipitation and spatial pattern of nitrogen supply in an experimental grassland ecosystem. Oecologia 2015, 178, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Orwin, K.H.; Stevenson, B.A.; Smaill, S.J.; Kirschbaum, M.U.; Dickie, I.A.; Clothier, B.E.; Garrett, L.G.; Weerden, T.J.; Beare, M.H.; Curtin, D. Effects of climate change on the delivery of soil-mediated ecosystem services within the primary sector in temperate ecosystems: A review and New Zealand case study. Glob. Chang. Biol. 2015, 21, 2844–2860. [Google Scholar] [CrossRef] [PubMed]
- Harpole, W.S.; Tilman, D. Grassland species loss resulting from reduced niche dimension. Nature 2007, 446, 791–793. [Google Scholar] [CrossRef] [PubMed]
- Weltzin, J.F.; Loik, M.E.; Schwinning, S.; Williams, D.G.; Fay, P.A.; Haddad, B.M.; Harte, J.; Huxman, T.E.; Knapp, A.K.; Lin, G. Assessing the response of terrestrial ecosystems to potential changes in precipitation. Bioscience 2003, 53, 941–952. [Google Scholar] [CrossRef]
- Soussana, J.F.; Loiseau, P.; Vuichard, N.; Ceschia, E.; Balesdent, J.; Chevallier, T.; Arrouays, D. Carbon cycling and sequestration opportunities in temperate grasslands. Soil Use Manag. 2004, 20, 219–230. [Google Scholar] [CrossRef]
- Duivenbooden, N.V. Grazing as a tool for rangeland management in semiarid regions: A case study in the north-western coastal zone of Egypt. Agric. Ecosyst. Environ. 1993, 43, 309–324. [Google Scholar] [CrossRef]
- Conant, R.T.; Paustian, K.; Elliott, E.T. Grassland management and conversion into grassland: Effects on soil carbon. Ecol. Appl. 2001, 11, 343–355. [Google Scholar] [CrossRef]
- Li, S.-G.; Eugster, W.; Asanuma, J.; Kotani, A.; Davaa, G.; Oyunbaatar, D.; Sugita, M. Response of gross ecosystem productivity, light use efficiency, and water use efficiency of Mongolian steppe to seasonal variations in soil moisture. J. Geophys. Res.-Biogeosci. 2008, 113, G01019. [Google Scholar] [CrossRef]
- Shao, C.; Chen, J.; Li, L. Grazing alters the biophysical regulation of carbon fluxes in a desert steppe. Environ. Res. Lett. 2013, 8, 025012. [Google Scholar] [CrossRef]
- Coupland, R.T. The effects of fluctuations in weather upon the grasslands of the Great Plains. Bot. Rev. 1958, 24, 273–317. [Google Scholar] [CrossRef]
- Derner, J.D.; Jin, V.L. Chapter 6: Soil Carbon Dynamics and Rangeland Management. In Managing Agricultural Greenhouse Gases; Liebig, M.A., Franzluebbers, A.J., Follett, R.F., Eds.; Academic Press: San Diego, CA, USA, 2012; pp. 79–92. [Google Scholar]
- Mirzaei, H.; Kreyling, J.; Zaman Hussain, M.; Li, Y.; Tenhunen, J.; Beierkuhnlein, C.; Jentsch, A. A single drought event of 100-year recurrence enhances subsequent carbon uptake and changes carbon allocation in experimental grassland communities. J. Plant Nutr. Soil Sci. 2008, 171, 681–689. [Google Scholar] [CrossRef]
- Koerner, S.E.; Collins, S.L. Interactive effects of grazing, drought, and fire on grassland plant communities in North America and South Africa. Ecology 2014, 95, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Koerner, S. Effects of global change on savanna grassland ecosystems. In Climate Change: Implications and Role of Grasslands; University of Dublin: Dublin, Ireland, 2012; pp. 181–188. [Google Scholar]
- Biondini, M.E.; Patton, B.D.; Nyren, P.E. Grazing intensity and ecosystem processes in a northern mixed-grass prairie, USA. Ecol. Appl. 1998, 8, 469–479. [Google Scholar] [CrossRef]
- Liebig, M.; Kronberg, S.; Hendrickson, J.; Dong, X.; Gross, J. Carbon dioxide efflux from long-term grazing management systems in a semiarid region. Agric. Ecosyst. Environ. 2013, 164, 137–144. [Google Scholar] [CrossRef]
- Wang, S.; Wilkes, A.; Zhang, Z.; Chang, X.; Lang, R.; Wang, Y.; Niu, H. Management and land use change effects on soil carbon in northern China’s grasslands: A synthesis. Agric. Ecosyst. Environ. 2011, 142, 329–340. [Google Scholar] [CrossRef]
- Schulp, C.J.; Nabuurs, G.-J.; Verburg, P.H. Future carbon sequestration in Europe-effects of land use change. Agric. Ecosyst. Environ. 2008, 127, 251–264. [Google Scholar] [CrossRef]
- Parton, W.J.; Ojima, D.S.; Schimel, D.S. Environmental change in grasslands: Assessment using models. In Assessing the Impacts of Climate Change on Natural Resource Systems; Springer: Berlin, Germany, 1994; pp. 111–141. [Google Scholar]
- Verburg, P.H. Simulating feedbacks in land use and land cover change models. Landsc. Ecol. 2006, 21, 1171–1183. [Google Scholar] [CrossRef]
- Poschlod, P.; Bakker, J.; Kahmen, S. Changing land use and its impact on biodiversity. Basic Appl. Ecol. 2005, 6, 93–98. [Google Scholar] [CrossRef]
- Vries, F.D.; Liiri, M.E.; Bjørnlund, L. Land use alters the resistance and resilience of soil food webs to drought. Nat. Clim. Chang. 2012, 2, 276–280. [Google Scholar] [CrossRef]
- Daniau, A.-L.; Goñi, M.F.S.; Martinez, P.; Urrego, D.H.; Bout-Roumazeilles, V.; Desprat, S.; Marlon, J.R. Orbital-scale climate forcing of grassland burning in southern Africa. Proc. Natl. Acad. Sci. USA 2013, 110, 5069–5073. [Google Scholar] [CrossRef] [PubMed]
- Verburg, P.H.; Tabeau, A.; Hatna, E. Assessing spatial uncertainties of land allocation using a scenario approach and sensitivity analysis: A study for land use in Europe. J. Environ. Manag. 2013, 127, S132–S144. [Google Scholar] [CrossRef] [PubMed]
- Pielke, R.A.; Marland, G.; Betts, R.A.; Chase, T.N.; Eastman, J.L.; Niles, J.O.; Running, S.W. The influence of land-use change and landscape dynamics on the climate system: Relevance to climate-change policy beyond the radiative effect of greenhouse gases. Philos. Trans. R. Soc. Lond. A Math. Phys. Eng. Sci. 2002, 360, 1705–1719. [Google Scholar] [CrossRef] [PubMed]
- Lal, R. Global soil erosion by water and carbon dynamics. In Soils and Global Change; Lal, R., Kimble, J., Levine, E., Stewart, B.A., Eds.; CRC Press, Inc.: Boca Raton, FL, USA, 1995; pp. 131–142. [Google Scholar]
- Wright, C.K.; Wimberly, M.C. Recent land use change in the Western Corn Belt threatens grasslands and wetlands. Proc. Natl. Acad. Sci. USA 2013, 110, 4134–4139. [Google Scholar] [CrossRef] [PubMed]
- Garnier, E.; Lavorel, S.; Ansquer, P.; Castro, H.; Cruz, P.; Dolezal, J.; Eriksson, O.; Fortunel, C.; Freitas, H.; Golodets, C. Assessing the effects of land-use change on plant traits, communities and ecosystem functioning in grasslands: A standardized methodology and lessons from an application to 11 European sites. Ann. Bot. 2007, 99, 967–985. [Google Scholar] [CrossRef] [PubMed]
- Eviner, V.T. Plant traits that influence ecosystem processes vary independently among species. Ecology 2004, 85, 2215–2229. [Google Scholar] [CrossRef]
- Lal, R. Soil erosion and carbon dynamics. Soil Tillage Res. 2005, 81, 137–142. [Google Scholar] [CrossRef]
- Knapp, A.K.; Carroll, C.J.; Denton, E.M.; La Pierre, K.J.; Collins, S.L.; Smith, M.D. Differential sensitivity to regional-scale drought in six central US grasslands. Oecologia 2015, 177, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Hoeppner, S.S.; Dukes, J.S. Interactive responses of old-field plant growth and composition to warming and precipitation. Glob. Chang. Biol. 2012, 18, 1754–1768. [Google Scholar] [CrossRef]
- Tilman, D.; Hill, J.; Lehman, C. Carbon-negative biofuels from low-input high-diversity grassland biomass. Science 2006, 314, 1598–1600. [Google Scholar] [CrossRef] [PubMed]
- Steinauer, K.; Tilman, D.; Wragg, P.D.; Cesarz, S.; Cowles, J.M.; Pritsch, K.; Reich, P.B.; Weisser, W.W.; Eisenhauer, N. Plant diversity effects on soil microbial functions and enzymes are stronger than warming in a grassland experiment. Ecology 2015, 96, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.P.; Milcu, A.; Manning, P.; Niklaus, P.A.; Roscher, C.; Power, S.; Reich, P.B.; Scheu, S.; Tilman, D.; Ai, F. Plant diversity drives soil microbial biomass carbon in grasslands irrespective of global environmental change factors. Glob. Chang. Biol. 2015, 21, 4076–4085. [Google Scholar] [CrossRef] [PubMed]
- Loreau, M.; Naeem, S.; Inchausti, P. Biodiversity and Ecosystem Functioning: Synthesis and Perspectives; Oxford University Press: Oxford, UK, 2002; pp. 30–305. [Google Scholar]
- Craine, J.M.; Wedin, D.A.; Reich, P.B. The response of soil CO2 flux to changes in atmospheric CO2, nitrogen supply and plant diversity. Glob. Chang. Biol. 2001, 7, 947–953. [Google Scholar] [CrossRef]
- Fridley, J.D. The influence of species diversity on ecosystem productivity: How, where, and why? Oikos 2001, 93, 514–526. [Google Scholar] [CrossRef]
- Weißhuhn, K.; Auge, H.; Prati, D. Geographic variation in the response to drought in nine grassland species. Basic Appl. Ecol. 2011, 12, 21–28. [Google Scholar] [CrossRef]
- Tilman, D.; El Haddi, A. Drought and biodiversity in grasslands. Oecologia 1992, 89, 257–264. [Google Scholar] [CrossRef]
- Tilman, D.; Downing, J.A. Biodiversity and stability in grasslands. In Ecosystem Management; Springer: Berlin, Germany, 1996; pp. 3–7. [Google Scholar]
- Van Peer, L.; Nijs, I.; Reheul, D.; De Cauwer, B. Species richness and susceptibility to heat and drought extremes in synthesized grassland ecosystems: Compositional vs. physiological effects. Funct. Ecol. 2004, 18, 769–778. [Google Scholar] [CrossRef]
- Amézquita, M.C.; Murgueitio, E.; Ibrahim, M.; Ramírez, B. Carbon sequestration in pasture and silvopastoral systems compared with native forests in ecosystems of tropical America. In Grassland Carbon Sequestration: Management, Policy and Economics; FAO: Rome, Italy, 2010; Volume 11. [Google Scholar]
- Wang, Z.; Silva, L.C.; Sun, G.; Luo, P.; Mou, C.; Horwath, W.R. Quantifying the impact of drought on soil-plant interactions: A seasonal analysis of biotic and abiotic controls of carbon and nutrient dynamics in high-altitudinal grasslands. Plant Soil 2015, 389, 59–71. [Google Scholar] [CrossRef]
- Neely, C.; Bunning, S.; Wilkes, A. Review of evidence on drylands pastoral systems and climate change: Implications and opportunities for mitigation and adaptation. In Land Tenure and Management Unit (NRLA); FAO: Rome, Italy, 2009; pp. 1–50. [Google Scholar]
- Fynn, A.; Alvarez, P.; Brown, J.; George, M.; Kustin, C.; Laca, E.; Oldfield, J.; Schohr, T.; Neely, C.; Wong, C. Soil carbon sequestration in United States rangelands. In Grassland Carbon Sequestration: Management, Policy and Economics; FAO: Rome, Italy, 2010; Volume 11. [Google Scholar]
- Milne, E.; Sessay, M.; Paustian, K.; Easter, M.; Batjes, N.; Cerri, C.; Kamoni, P.; Gicheru, P.; Oladipo, E.; Minxia, M. Towards a standardized system for the reporting of carbon benefits in sustainable land management projects. In Grassland Carbon Sequestration: Management, Policy and Economics; FAO: Rome, Italy, 2010; Volume 11. [Google Scholar]
- Burri, S.; Niklaus, P.; Buchmann, N.; Kahmen, A. Response of grassland soil respiration to drought: Results from an ecosystem manipulation experiment including 19 sites differing in productivity and diversity. In Proceedings of the EGU General Assembly 2015, Vienna, Austria, 12–17 April 2015; p. 9059.
- Magdoff, F.; Weil, R.R. Soil Organic Matter in Sustainable Agriculture; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–200. [Google Scholar]
- Bot, A.; Benites, J. The Importance of Soil Organic Matter: Key to Drought-Resistant Soil and Sustained Food Production; Food & Agriculture Organization: Rome, Italy, 2005; pp. 1–303. [Google Scholar]
- Walther, G.-R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.; Fromentin, J.-M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- De Vries, F.T.; Shade, A. Controls on soil microbial community stability under climate change. Front. Microbiol. 2013, 4, 293–293. [Google Scholar] [CrossRef] [PubMed]
- Henry, H.A.; Cleland, E.E.; Field, C.B.; Vitousek, P.M. Interactive effects of elevated CO2, N deposition and climate change on plant litter quality in a California annual grassland. Oecologia 2005, 142, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Langley, J.A.; Megonigal, J.P. Ecosystem response to elevated CO2 levels limited by nitrogen-induced plant species shift. Nature 2010, 466, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Ives, A.R.; Carpenter, S.R. Stability and diversity of ecosystems. Science 2007, 317, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Lau, J.A.; Lennon, J.T. Rapid responses of soil microorganisms improve plant fitness in novel environments. Proc. Natl. Acad. Sci. USA 2012, 109, 14058–14062. [Google Scholar] [CrossRef] [PubMed]
- Pendall, E.; Heisler-White, J.L.; Williams, D.G.; Dijkstra, F.A.; Carrillo, Y.; Morgan, J.A.; LeCain, D.R. Warming reduces carbon losses from grassland exposed to elevated atmospheric carbon dioxide. PLoS ONE 2013, 8, e71921. [Google Scholar] [CrossRef] [PubMed]
- Blair, R.D.; Kaserman, D.L. Law and Economics of Vertical Integration and Control; Academic Press: Cambridge, MA, USA, 2014; pp. 1–203. [Google Scholar]
- Morgan, J.A.; LeCain, D.R.; Pendall, E.; Blumenthal, D.M.; Kimball, B.A.; Carrillo, Y.; Williams, D.G.; Heisler-White, J.; Dijkstra, F.A.; West, M. C4 grasses prosper as carbon dioxide eliminates desiccation in warmed semi-arid grassland. Nature 2011, 476, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Hungate, B.A.; Holland, E.A.; Jackson, R.B.; Chapin, F.S.; Mooney, H.A.; Field, C.B. The fate of carbon in grasslands under carbon dioxide enrichment. Nature 1997, 388, 576–579. [Google Scholar] [CrossRef]
- Norby, R.J.; Luo, Y. Evaluating ecosystem responses to rising atmospheric CO2 and global warming in a multi-factor world. New Phytol. 2004, 162, 281–293. [Google Scholar] [CrossRef]
- Cantarel, A.A.; Bloor, J.M.; Soussana, J.F. Four years of simulated climate change reduces above-ground productivity and alters functional diversity in a grassland ecosystem. J. Veg. Sci. 2013, 24, 113–126. [Google Scholar] [CrossRef]
- Luo, Y.; Gerten, D.; Le Maire, G.; Parton, W.J.; Weng, E.; Zhou, X.; Keough, C.; Beier, C.; Ciais, P.; Cramer, W. Modeled interactive effects of precipitation, temperature, and CO2 on ecosystem carbon and water dynamics in different climatic zones. Glob. Chang. Biol. 2008, 14, 1986–1999. [Google Scholar] [CrossRef]
- Shaw, M.R.; Zavaleta, E.S.; Chiariello, N.R.; Cleland, E.E.; Mooney, H.A.; Field, C.B. Grassland responses to global environmental changes suppressed by elevated CO2. Science 2002, 298, 1987–1990. [Google Scholar] [CrossRef] [PubMed]
- Weaver, J.E. Effects of different intensities of grazing on depth and quantity of roots of grasses. J. Range Manag. 1950, 3, 100–113. [Google Scholar] [CrossRef]
- Guo, L.B.; Gifford, R. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- Bestelmeyer, B.T.; Herrick, J.E.; Brown, J.R.; Trujillo, D.A.; Havstad, K.M. Land management in the American Southwest: A state-and-transition approach to ecosystem complexity. Environ. Manag. 2004, 34, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Bardgett, R.D.; Manning, P.; Morriën, E.; Vries, F.T. Hierarchical responses of plant–soil interactions to climate change: Consequences for the global carbon cycle. J. Ecol. 2013, 101, 334–343. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Grimm, N.B.; Chapin, F.S., III; Bierwagen, B.; Gonzalez, P.; Groffman, P.M.; Luo, Y.; Melton, F.; Nadelhoffer, K.; Pairis, A.; Raymond, P.A. The impacts of climate change on ecosystem structure and function. Front. Ecol. Environ. 2013, 11, 474–482. [Google Scholar] [CrossRef]
- Smith, M.D. An ecological perspective on extreme climatic events: A synthetic definition and framework to guide future research. J. Ecol. 2011, 99, 656–663. [Google Scholar] [CrossRef]
- Shi, X.; Zhao, D.; Wu, S.; Shi, W.; Dai, E.; Wang, W. Climate change risks for net primary production of ecosystems in China. Hum. Ecol. Risk Assess. 2016, 22, 1091–1105. [Google Scholar] [CrossRef]
- Collins, S.L.; Carpenter, S.R.; Swinton, S.M.; Orenstein, D.E.; Childers, D.L.; Gragson, T.L.; Grimm, N.B.; Grove, J.M.; Harlan, S.L.; Kaye, J.P. An integrated conceptual framework for long-term social-ecological research. Front. Ecol. Environ. 2010, 9, 351–357. [Google Scholar] [CrossRef]
- Lei, T.; Wu, J.; Li, X.; Geng, G.; Shao, C.; Zhou, H.; Wang, Q.; Liu, L. A new framework for evaluating the impacts of drought on net primary productivity of grassland. Sci. Total Environ. 2015, 536, 161–172. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lei, T.; Pang, Z.; Wang, X.; Li, L.; Fu, J.; Kan, G.; Zhang, X.; Ding, L.; Li, J.; Huang, S.; et al. Drought and Carbon Cycling of Grassland Ecosystems under Global Change: A Review. Water 2016, 8, 460. https://doi.org/10.3390/w8100460
Lei T, Pang Z, Wang X, Li L, Fu J, Kan G, Zhang X, Ding L, Li J, Huang S, et al. Drought and Carbon Cycling of Grassland Ecosystems under Global Change: A Review. Water. 2016; 8(10):460. https://doi.org/10.3390/w8100460
Chicago/Turabian StyleLei, Tianjie, Zhiguo Pang, Xingyong Wang, Lin Li, June Fu, Guangyuan Kan, Xiaolei Zhang, Liuqian Ding, Jiren Li, Shifeng Huang, and et al. 2016. "Drought and Carbon Cycling of Grassland Ecosystems under Global Change: A Review" Water 8, no. 10: 460. https://doi.org/10.3390/w8100460
APA StyleLei, T., Pang, Z., Wang, X., Li, L., Fu, J., Kan, G., Zhang, X., Ding, L., Li, J., Huang, S., & Shao, C. (2016). Drought and Carbon Cycling of Grassland Ecosystems under Global Change: A Review. Water, 8(10), 460. https://doi.org/10.3390/w8100460







