New Insights on the Simultaneous Removal by Adsorption on Organoclays of Humic Acid and Phenol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods and Instrumentation
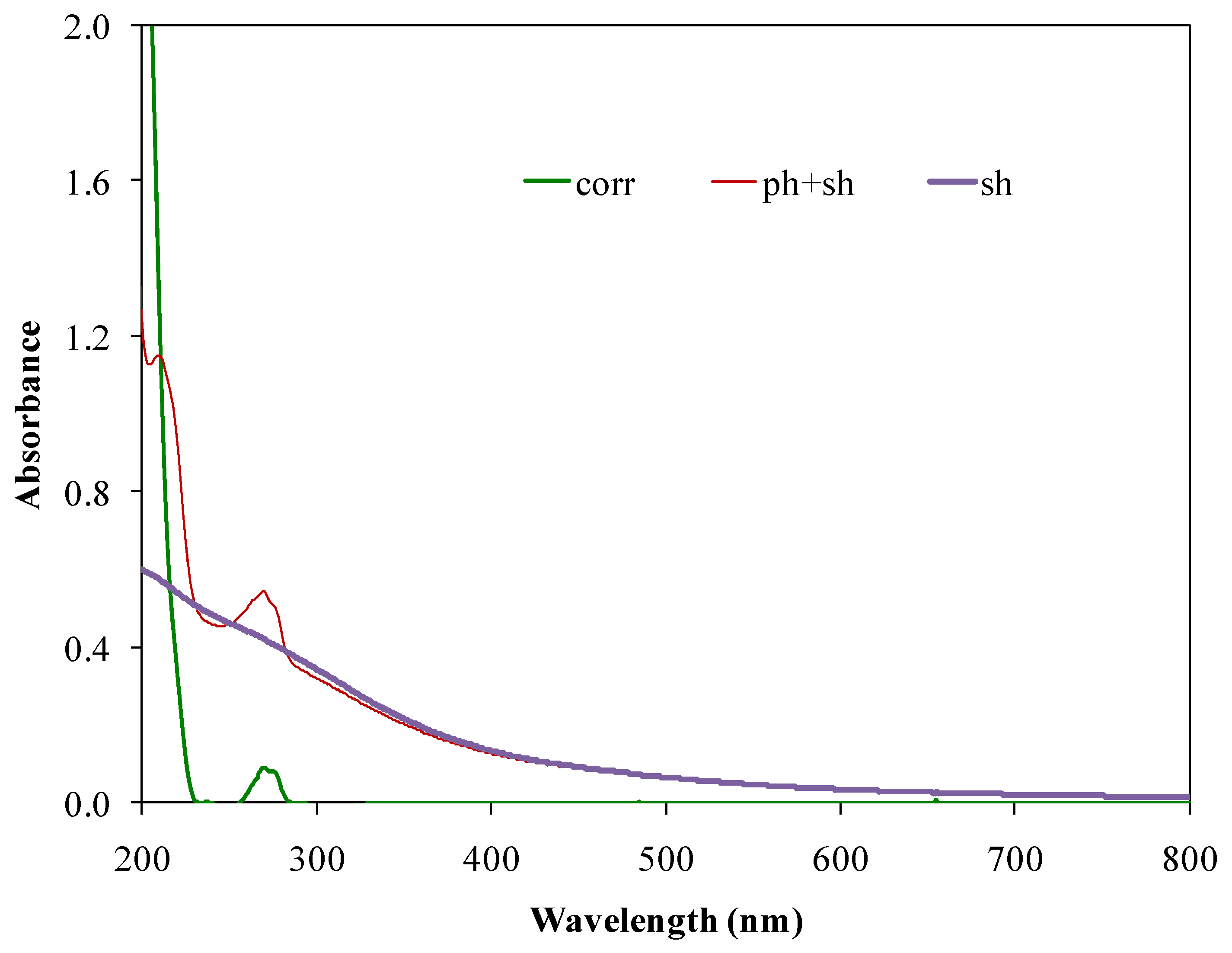
3. Results and Discussion
3.1. Selection of the Clay Minerals
3.2. Selecting the Proper Organophilization Methodology
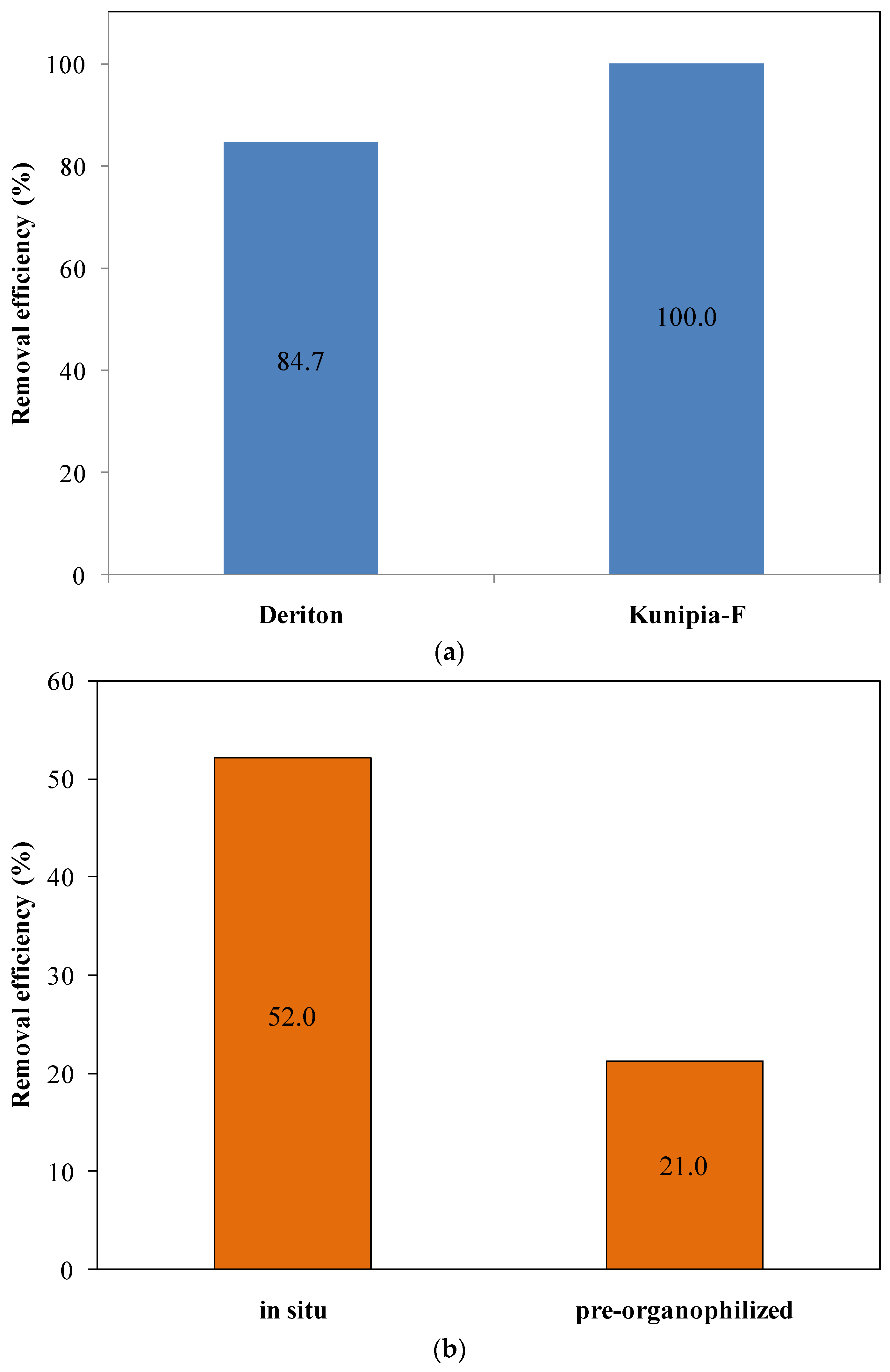
- the competitive adsorption of HTAB
- the pH change during the adsorption process
- the input sequence of the pollutants, HTAB and the adsorbent
3.3. The Separate Removal of Model Pollutants
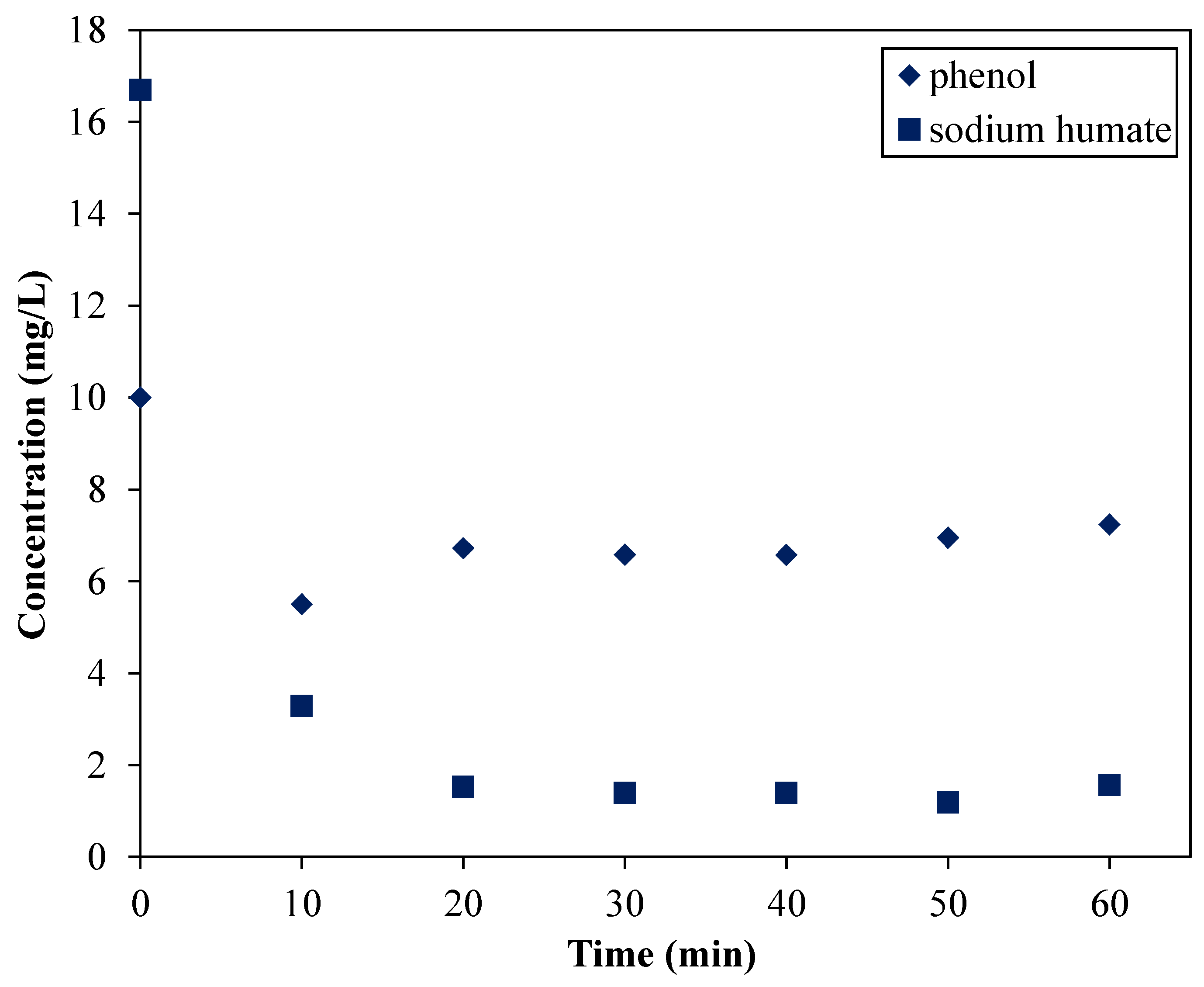
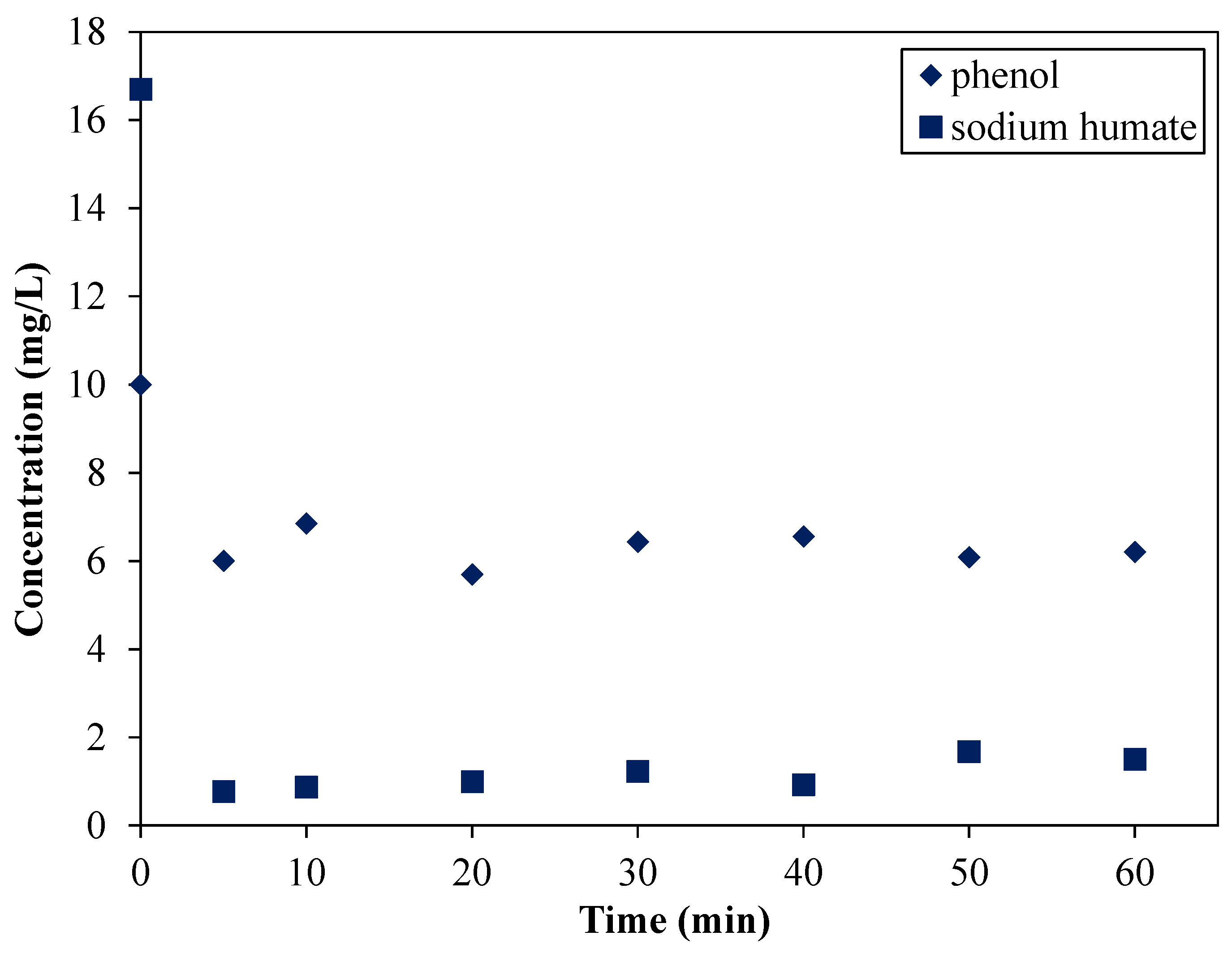
3.3.1. The Impact of Instantaneous Intake of the Adsorbent—the Possibility of Competitive Adsorption of HTAB
- (1) aqueous solution of the model pollutant;
- (2) swollen clay mineral;
- (3) HTAB.
- (1) solution with model pollutant;
- (2) swollen clay mineral containing HTAB.
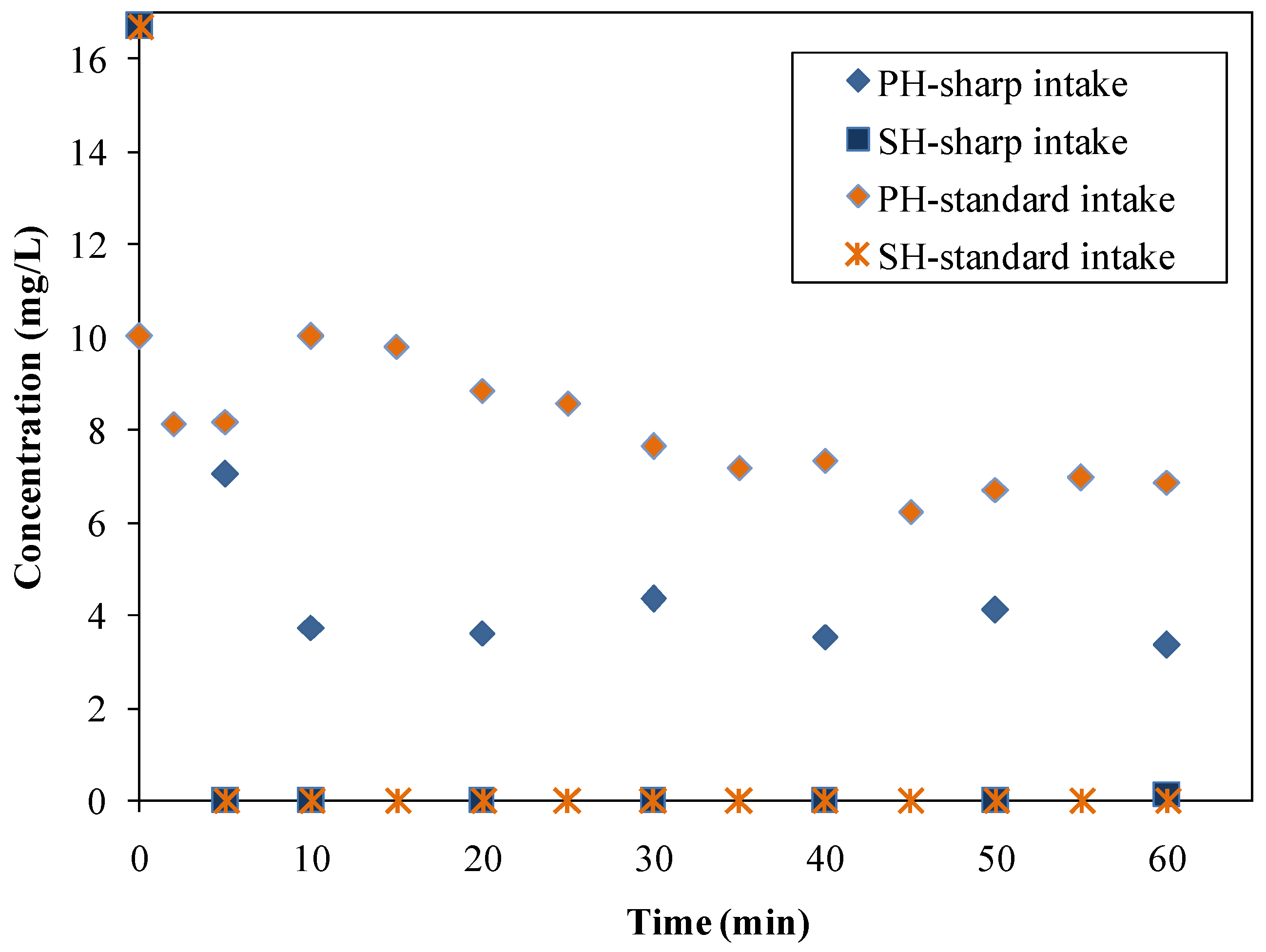
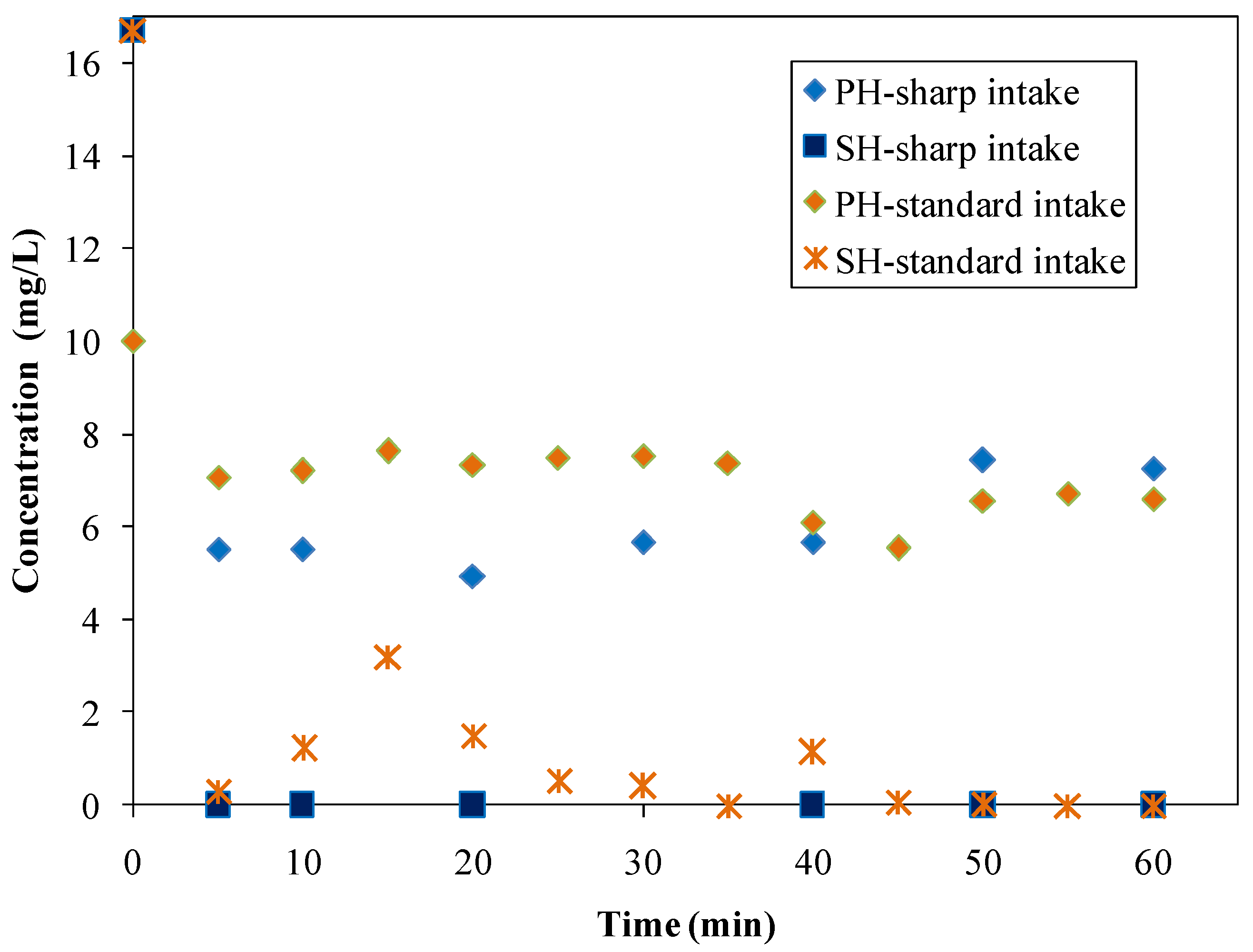
- standard intake investigations: after 40 min in the case of phenol (although a slight concentration decrease was observed starting from the 10th min) and immediately in the case of SH;
- sharp intake investigations: after approximately 10 min in the case of phenol and immediately in the case of SH.
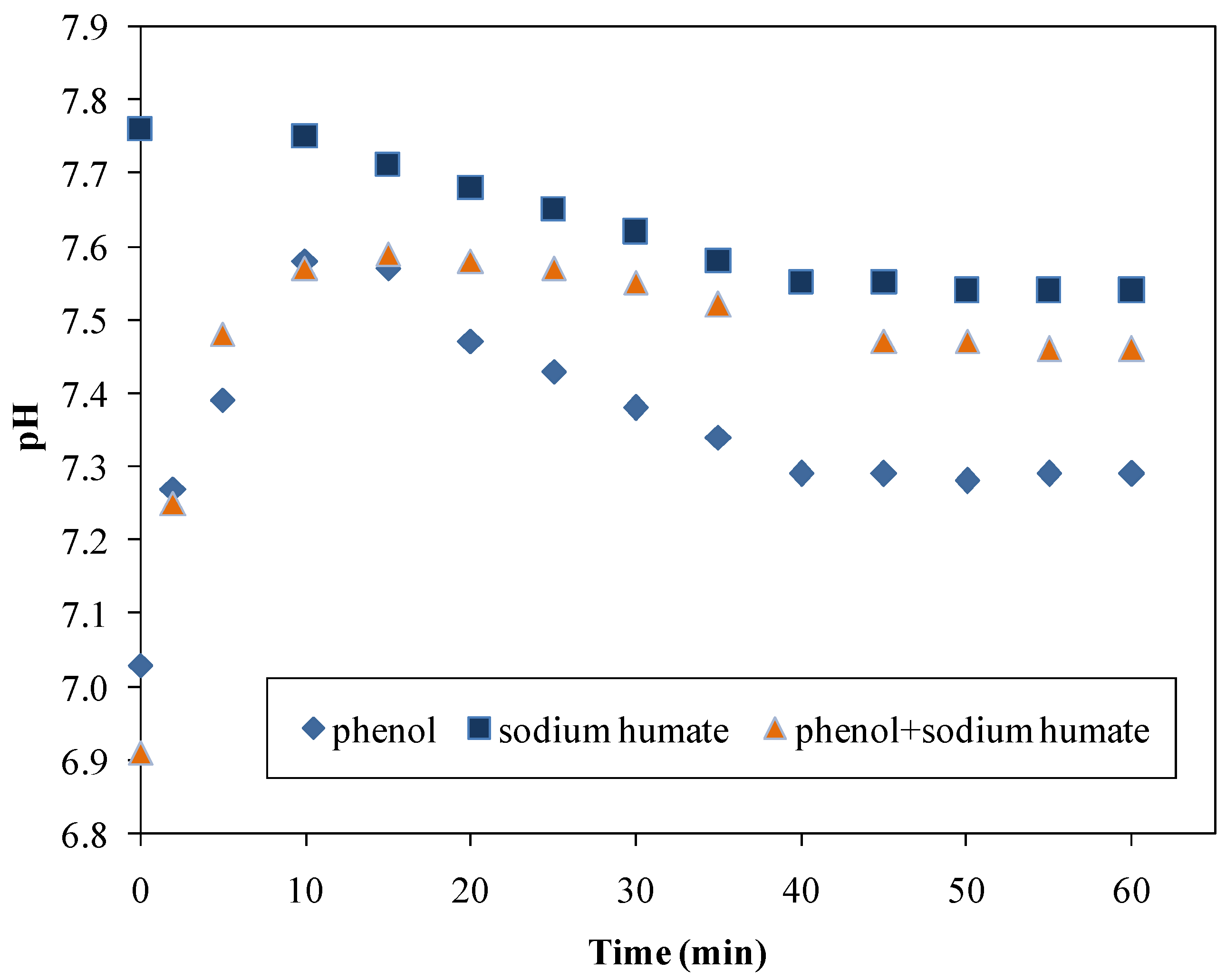
3.3.2. The Change of pH During Adsorption Processes
| Sample | pH |
|---|---|
| Milli-Q water | 6.9 |
| Clay Mineral, Kunipia (500 mg∙L−1) | 7.9 |
| Cationic surfactant, HTAB—5.75∙10−4 mol∙dm−3 | 5.9 |
| In situ adsorbent | 7.5 |
3.4. Experiments with Multiple Model Pollutants
3.4.1. The Impact of Instantaneous Intake of Adsorbent
3.4.2. The pH Changes during the Adsorption
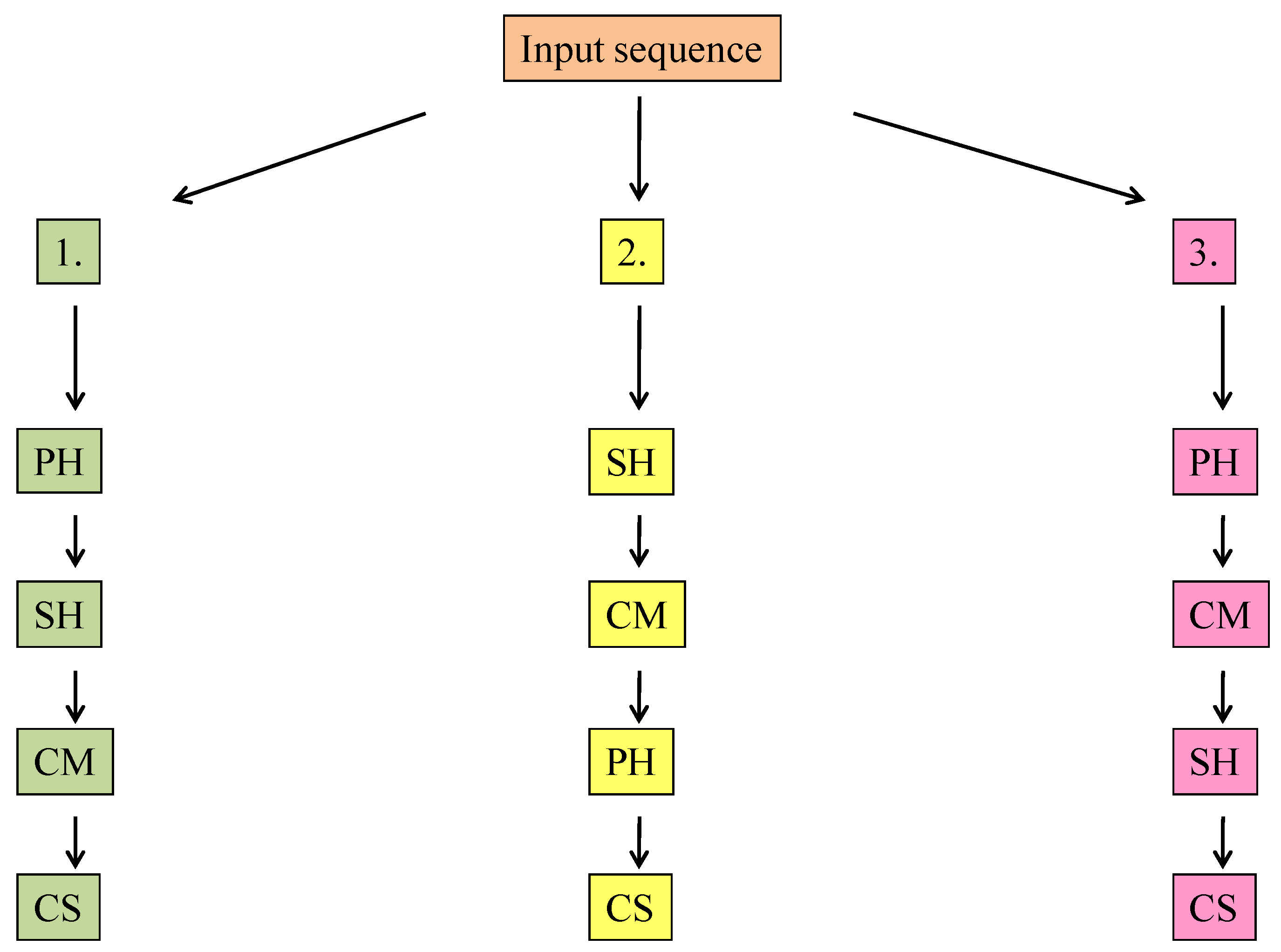
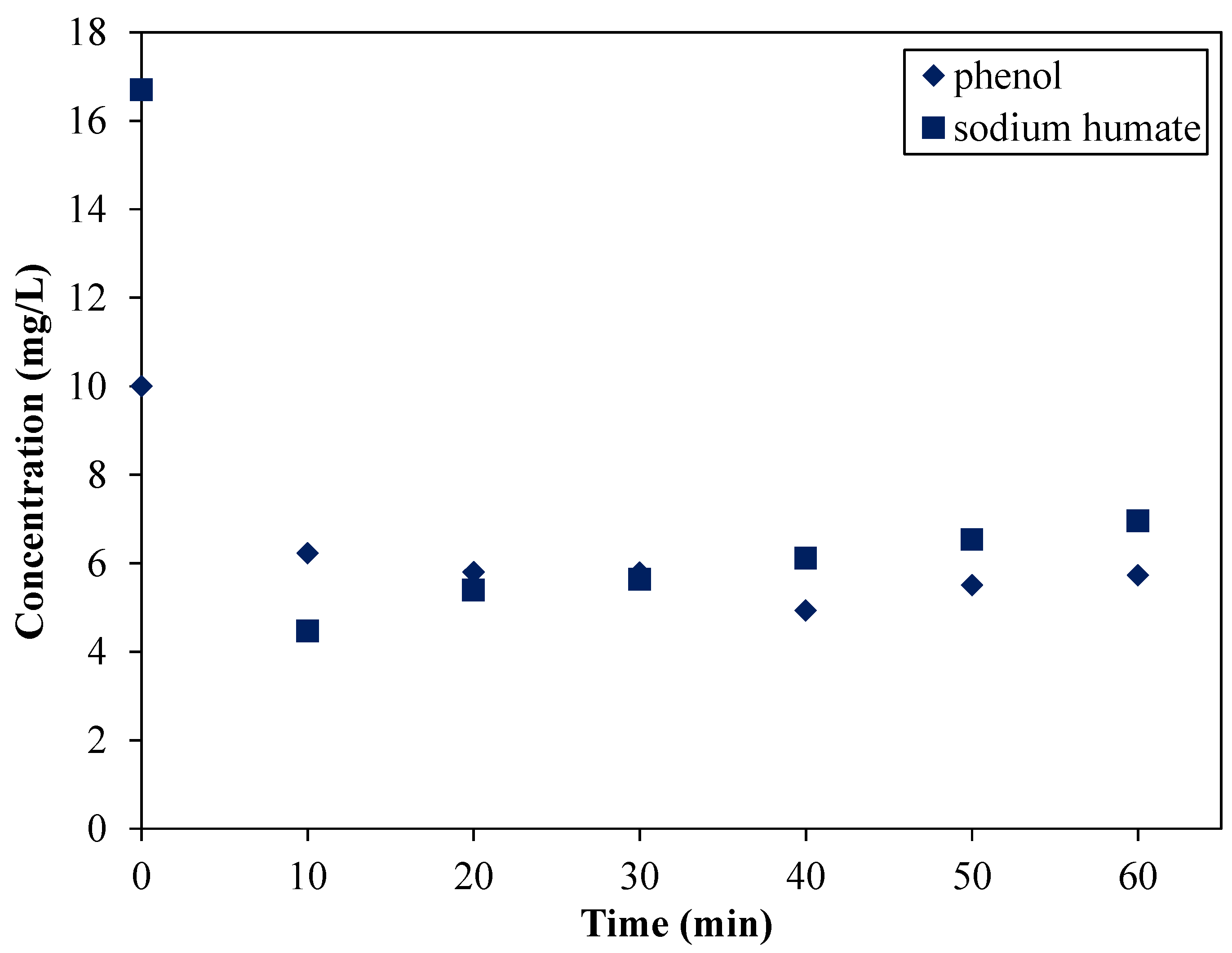
3.5. Changing the Input Sequence
- (i)
- The half amount of the introduced SH was adsorbed in 5 min.
- (ii)
- Between the 10th and 20th min, a smaller fraction of the adsorbed phenol desorbed from the organoclay’s surface.
- (iii)
- After 20 min, the phenol’s concentration was stationary (≈7 mg∙L−1), and, surprisingly, sodium humate was not completely adsorbed during this experiment.
- (iv)
- The equilibrium concentration of SH was 1–2 mg∙L−1, but between 5 and 10 min a fluctuation was observed in the concentration values. Typically, the increase of the SH concentration showed an opposing behavior compared to the concentration change of phenol.
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kárpáti, Z.; Sajgó, C.; Vető, I.; Klopp, G.; Horváth, I. Organic matter in thermal waters of the Pannonian Basin—A preliminary report on aromatic compounds. Org. Geochem. 1999, 30, 701–712. [Google Scholar]
- Pauwels, H.; Fouillac, C.; Goff, F.; Vuataz, F.D. The isotopic and chemical composition of CO2-rich thermal waters in the Mont-Dore region (Massif-Central, France). Appl. Geochem. 1997, 12, 411–427. [Google Scholar] [CrossRef]
- Saha, N.C.; Bhunia, F.; Kaviraj, A. Toxicity of Phenol to Fish and Aquatic Ecosystem. Environ. Contam. Toxicol. 1999, 63, 195–202. [Google Scholar] [CrossRef]
- Megharaj, M.; Pearson, H.W.; Venkateswarlu, K. Toxicity of phenol and three nitrophenols towards growth and metabolic activities of Nostoc linckia, isolated from soil. Environ. Contam. Toxicol. 1991, 21, 578–584. [Google Scholar] [CrossRef]
- Abdollahi, M.; Hassani, S.; Derakhshani, M. Phenol. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Elsevier Academic Press: Amsterdam, The Netherland, 2014; pp. 871–873. [Google Scholar]
- Stanisavljevic, M.; Nedic, L. Removal of phenol from industrial wastewaters by horseradish (Cochlearia armoracia L) peroxidase. Facta Univ. Ser. Work. Living Environ. Prot. 2004, 2, 345–349. [Google Scholar]
- Hu, C.; Wang, Y.; Tang, H. Destruction of phenol aqueous solution by photocatalysis or direct photolysis. Chemosphere 2000, 41, 1205–1209. [Google Scholar]
- Ilisz, I.; Dombi, A.; Mogyorósi, K.; Farkas, A.; Dékány, I. Removalof 2-chlorophenol from water by adsorption combined with TiO2 photocatalysis. Appl. Catal. B Environ. 2002, 39, 247–256. [Google Scholar] [CrossRef]
- Shen, Y. Removal of phenol from water by adsorption–flocculationusing organobentonite. Water Res. 2002, 36, 1107–1114. [Google Scholar] [CrossRef]
- Ma, J.; Zhu, L. Removal of phenols from water accompanied withsynthesis of organobentonite in one-step process. Chemosphere 2007, 68, 1883–1888. [Google Scholar] [CrossRef] [PubMed]
- Szabo, E.; Vajda, K.; Vereb, G.; Dombi, A.; Mogyorosi, K.; Abraham, I.; Majer, M. Removal of organic pollutants in model water and thermal wastewater using clay minerals. J. Environ. Sci. Health Part A 2011, 46, 1346–1356. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Martin, M.J.; Dorado, M.C.; Hoyo, C.; Rodriguez-Cruz, M.S. Influence of clay mineral structure and surfactant nature on the adsorption capacity of surfactants by clays. J. Hazard. Mater. 2008, 150, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Mallavarapu, M.; Naidu, R. Preparation, characterization of surfactants modified clay minerals and nitrate adsorption. Appl. Clay Sci. 2010, 48, 92–96. [Google Scholar] [CrossRef]
- Wiles, M.C.; Huebner, H.J.; McDonald, T.J.; Donnelly, K.C.; Phillips, T.D. Matrix-immobilized organoclay for the sorption of polycyclic aromatic hydrocarbons and pentachlorophenol from groundwater. Chemosphere 2005, 59, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Senturk, H.B.; Ozdes, D.; Gundogdu, A.; Duran, C.; Soylak, M. Removal of phenol from aqueous solutions by adsorption onto organomodified Tirebolu bentonite: Equilibrium, kinetic and thermodynamic study. J. Hazard. Mater. 2009, 172, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Szabó, E.; Pap, Z.; Dombi, A.; Hernádi, K.; Baia, L.; Simon, G. Important aspects on the removal of humic acid and phenolic compounds with clay minerals. “Synergism provided by the pollutants, efficiency given by the media”. Water Air Soil Pollut. 2014, 225, 1888–1897. [Google Scholar]
- Sanchez-Martin, M.J.; Rodriguez-Cruz, M.S.; Andrades, M.S.; Sanchez-Camazano, M. Efficiency of different clay minerals modified with a cationic surfactant in the adsorption of pesticides: Influence of clay type and pesticide hydrophobicity. Appl. Clay Sci. 2006, 31, 216–228. [Google Scholar] [CrossRef]
- Liu, R.; Frost, R.L.; Martens, W.N.; Yuan, Y. Synthesis, characterization of mono, di and tri alkyl surfactant intercalated Wyoming montmorillonite for the removal of phenol from aqueous systems. J. Colloid Interface Sci. 2008, 327, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Tombácz, E.; Szekeres, M. Colloidal behavior of aqueous montmorillonite suspensions: The specific role of pH in the presence of indifferent electrolytes. Appl. Clay Sci. 2004, 27, 75–94. [Google Scholar]
- Banat, F.A.; Al-Bashir, B.; Al-Asheh, S.; Hayajneh, O. Adsorption of phenol by bentonite. Environ. Pollut. 2000, 107, 391–398. [Google Scholar] [CrossRef]
- Ocampo-Perez, R.; Leyva-Ramos, R.; Mendoza-Barron, J.; Guerrero-Coronado, R.M. Adsorption rate of phenol from aqueous solution onto organobentonite: Surface diffusion and kinetic models. J. Colloid Interface Sci. 2011, 364, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Stanković, N.; Logar, M.; Luković, J.; Pantić, J.; Miljević, M.; Babić, B.; Radosavljević-Mihajlović, A. Characterization of bentonite clay from “Greda” deposit. Process. Appl. Ceram. 2011, 5, 97–101. [Google Scholar] [CrossRef]
- Neumann, M.G.; Gessner, F.; Schmitt, C.C.; Sartorio, R. Influence of the layer charge and the clay particle size on the interactions between the cationic dye methylene blue and clays in an aqueous suspension. J. Colloid Interface Sci. 2002, 255, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Churchman, G.J. Formation of complexes between bentonite and different cationic polyelectrolytes and their use as sorbents for non-ionic and anionic pollutants. Appl. Clay Sci. 2002, 21, 177–189. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szabó, E.; Pap, Z.; Simon, G.; Dombi, A.; Baia, L.; Hernádi, K. New Insights on the Simultaneous Removal by Adsorption on Organoclays of Humic Acid and Phenol. Water 2016, 8, 21. https://doi.org/10.3390/w8010021
Szabó E, Pap Z, Simon G, Dombi A, Baia L, Hernádi K. New Insights on the Simultaneous Removal by Adsorption on Organoclays of Humic Acid and Phenol. Water. 2016; 8(1):21. https://doi.org/10.3390/w8010021
Chicago/Turabian StyleSzabó, Emese, Zsolt Pap, Gergő Simon, András Dombi, Lucian Baia, and Klára Hernádi. 2016. "New Insights on the Simultaneous Removal by Adsorption on Organoclays of Humic Acid and Phenol" Water 8, no. 1: 21. https://doi.org/10.3390/w8010021
APA StyleSzabó, E., Pap, Z., Simon, G., Dombi, A., Baia, L., & Hernádi, K. (2016). New Insights on the Simultaneous Removal by Adsorption on Organoclays of Humic Acid and Phenol. Water, 8(1), 21. https://doi.org/10.3390/w8010021









