Exposure of Mediterranean Countries to Ocean Acidification
Abstract
:1. Introduction
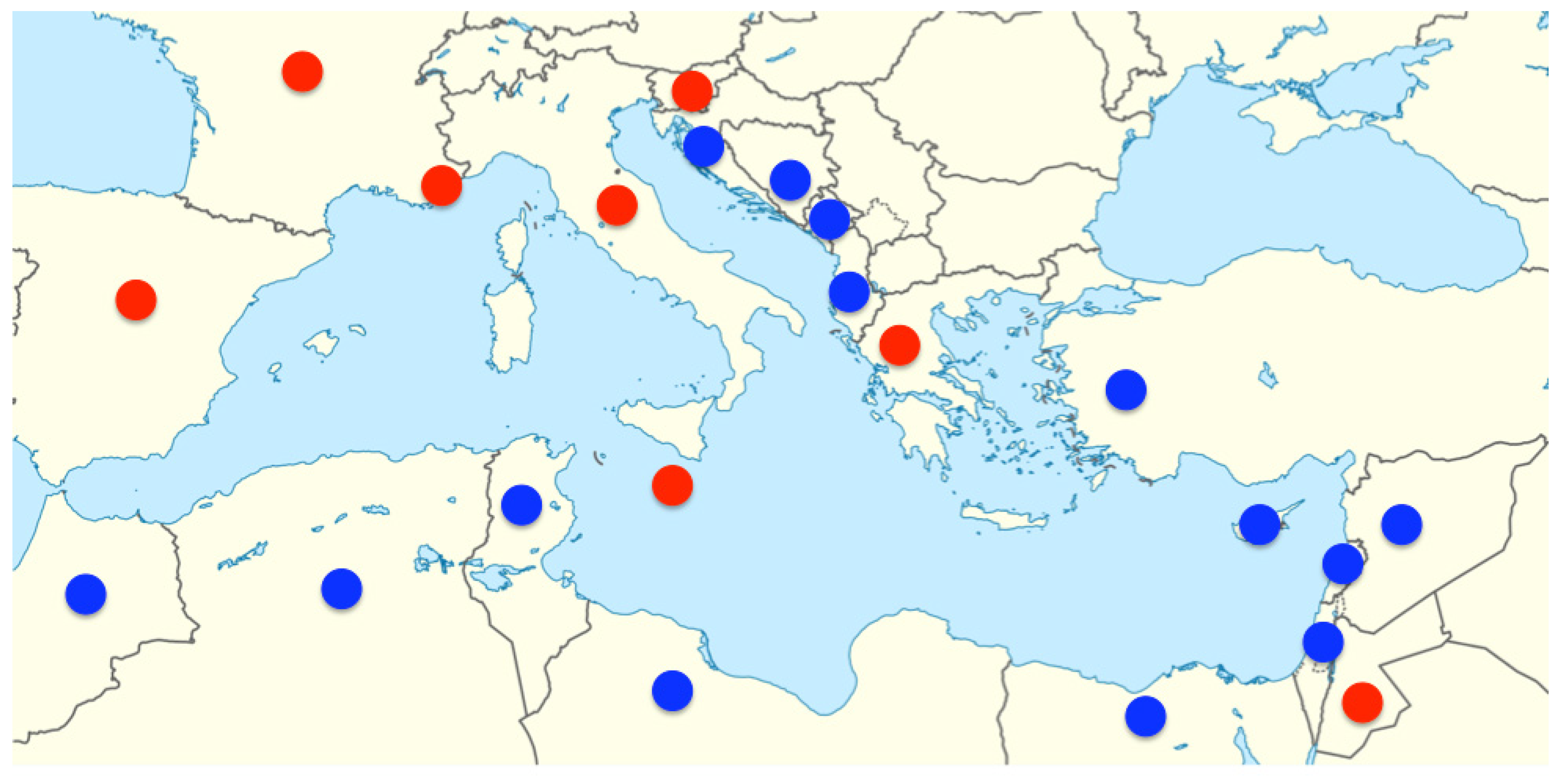
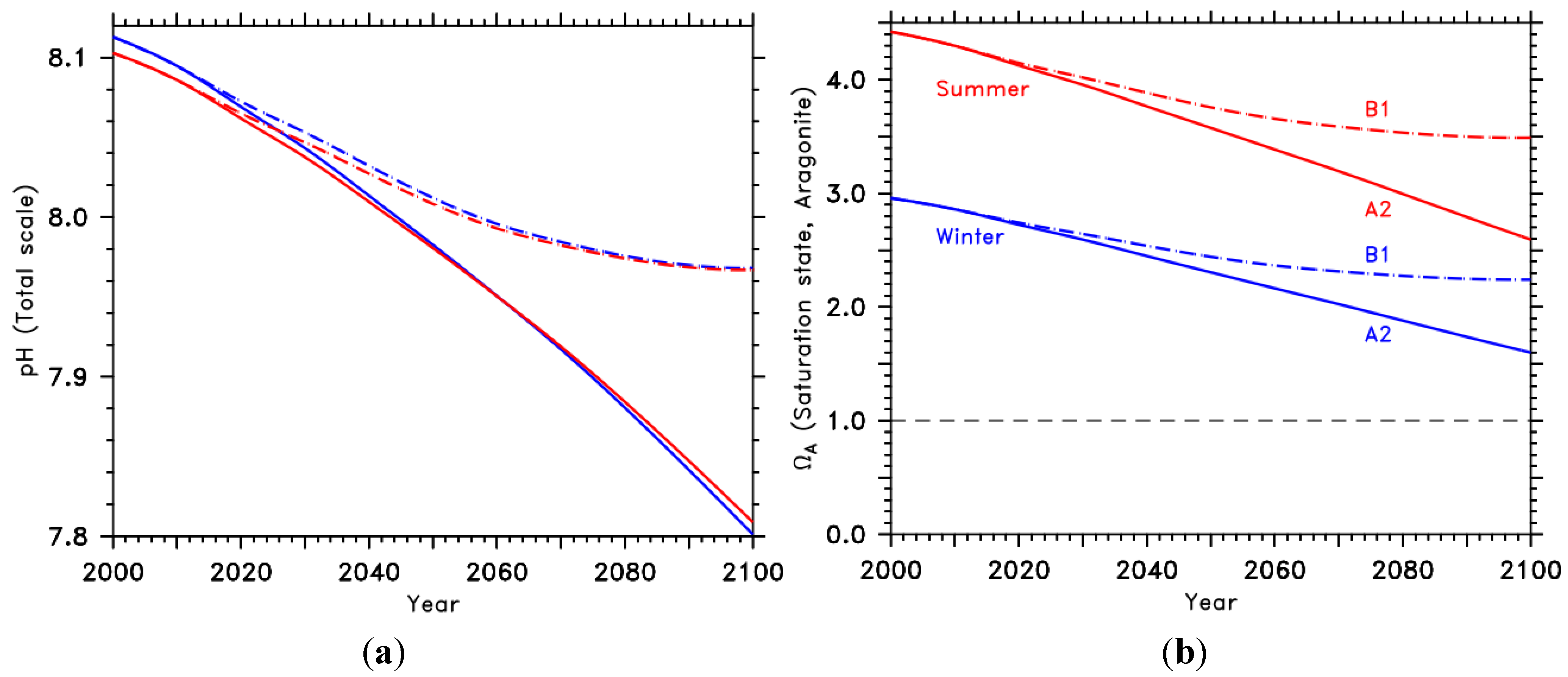
2. Ocean Acidification’s Development in the Mediterranean
3. Known Ocean Acidification Responses of Mediterranean Species
| Harvested Mediterranean species | Fishery type | # Articles on this species | # Articles on this genus | Other studied species of the same genus (# articles) |
|---|---|---|---|---|
| CRUSTACEANS | ||||
| Carcinus aestuarii | WC | 0 | 6 | C. maenas (6) |
| Hommarus gammarus | WC | 3 | 1 | H. americanus (1) |
| Nephrops norvegicus | WC | 2 | 0 | |
| Palaemon serratus | WC | 1 | 2 | P. californicus (1), P. elegans (1) |
| Penaeus indicus | A | 0 | 1 | P. plebejus (1) |
| Penaeus vannamei | A | 0 | 1 | P. plebejus (1) |
| ECHINODERMS | ||||
| Paracentrotus lividus | WC | 8 | 0 | |
| MOLLUSCS | ||||
| Crassostrea gigas | A | 13 | 13 | C. hongkongensis (1), C. virginica (12) |
| Haliotis tuberculata | WC | 0 | 6 | H. coccoradiata (2), H. discus (2), H. Kamtschatkana (1), H. rufescens (1) |
| Loligo vulgaris | WC | 1 | 0 | |
| Mytilus edulis | A | 14 | 14 | M. californianus (2), M. chilensis (2), M. galloprovincialis (9), M. trossulus (1) |
| Mytilus galloprovincialis | A | 9 | 19 | M. californianus (2), M. chilensis (2), M. edulis (14), M. trossulus (1) |
| Ostrea edulis | A, WC | 3 | 0 | |
| Patella caerula | WC | 0 | 1 | P. vulgata (1) |
| Pecten jacobaeus | WC | 0 | 3 | P. maximus (3) |
| Ruditapes decussatus | A, WC | 3 | 1 | R. philippinarum (1) |
| Ruditapes philippinarum | A | 1 | 3 | R. decussatus (3) |
| Sepia elegans | WC | 0 | 10 | S. officinalis (10) |
| Sepia officinalis | WC | 10 | 0 |
4. Dependence of Mediterranean Countries on Fisheries Harvests
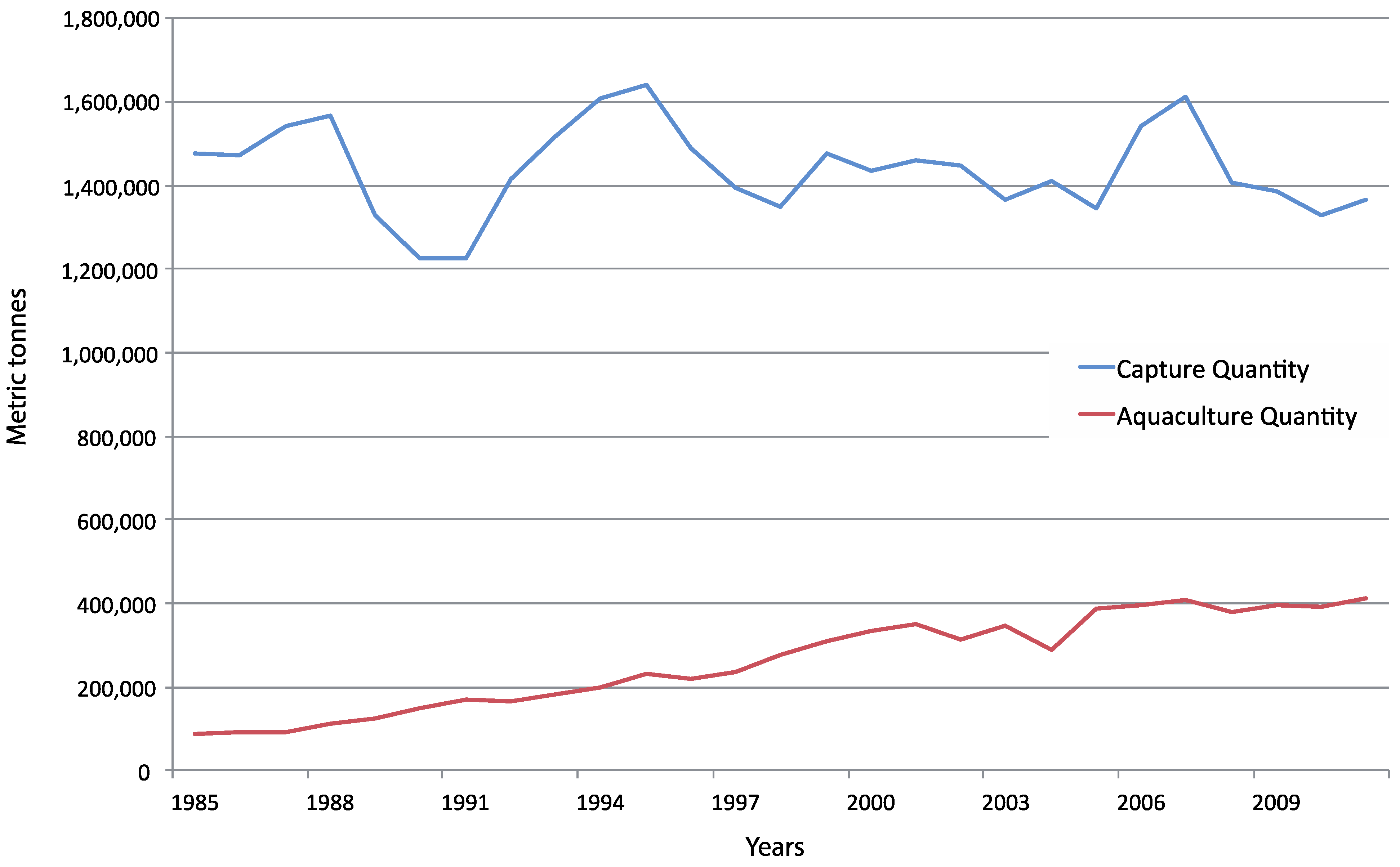
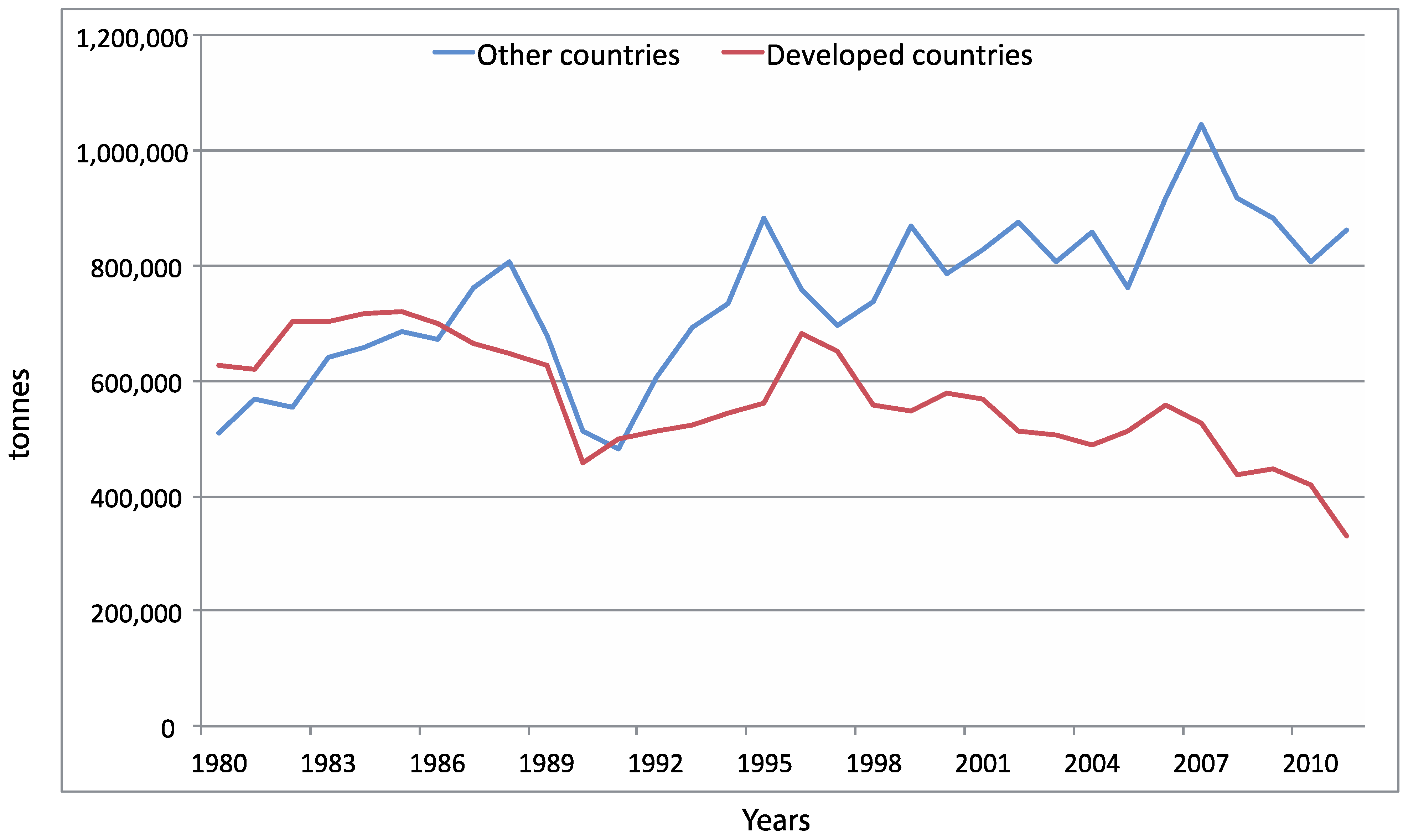
4.1. Wild vs. Aquaculture Harvests

| Species | 2010 | 2011 | ||
|---|---|---|---|---|
| Volume (tons) | Value (Thousands of USD) | Volume (tons) | Value (Thousands of USD) | |
| Molluscs | 451,909 | 861,779 | 474,062 | 922,807 |
| Fish | 249,016 | 1,526,191 | 266,559 | 1,707,428 |
| Other | 8,138 | 32,715 | 8,662 | 37,440 |
4.2. Wild Harvests
4.2.1. Industrial and Artisanal Fleets
| Species | Croatia | Bosnia and Her. | Italy | Greece | Mor. | Slov. | Tunisia | Turkey | Albania | Algeria | Spain | Egypt | France |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sea urchins | 3 | ||||||||||||
| Oysters | 555 | 20 | 10 | 141 | 10 | 31 | <0.5 | 8,800 | |||||
| Mussels | 3,013 | 50 | 58,479 | 22,653 | 301 | 696 | 2,666 | 1360 | 44 | <0.5 | 16,060 |
4.2.2. Recreational Fisheries
4.2.3. Employment
| Country | Employment—Capture Fisheries | ||||
|---|---|---|---|---|---|
| Primary (No. Fishers) | Secondary (No. Employed in Processing, Distribution, etc.) | Year of Data Collection | |||
| Albania 1 | 2,400 | ND | ND | ||
| Algeria 1 | 28,225 | 84,675 | 2000 | ||
| Bosnia | ND | ND | ND | ||
| Croatia 1 | 20,000 | ND | 2006 | ||
| Cyprus 2 | 1,350 | ND | 2005 | ||
| Egypt *1 | 600,400 | ND | 2000 | ||
| France *2 | 19,624 | ND | 2008 | ||
| Greece 3 | 29,313 | ND | 2008 | ||
| Israel 1 | 1,503 | 2,300 | 2005 | ||
| Italy 3 | 29,562 | ND | 2008 | ||
| Lebanon 4 | 4,000 | ND | Unclear | ||
| Libya 1 | 11,500 | 3,500 | 2003 | ||
| Malta 2 | 2,100 | ND | 2005 | ||
| Monaco | ND | ND | ND | ||
| Montenegro | ND | ND | ND | ||
| Morocco *1 | 70,000 | 40,000 | 2005 | ||
| Palestine | ND | ND | ND | ||
| Slovenia 2 | 343 | ND | 2005 | ||
| Spain *3 | 30,958 | ND | 2008 | ||
| Syria 1 | 13,252 | 7550 | 2005 | ||
| Tunisia 1 | 53,000 | 47,000 | 2003 | ||
| Turkey *3 | 53,893 | ND | 2008 | ||
4.3. Consumption and Trade of Seafood
| Developed Countries | kg/cap/y | Developing Countries | kg/cap/y |
|---|---|---|---|
| Albania | 3.4 | Algeria | 4.5 |
| Bosnia Herzegovina | 4.2 | Cyprus | 10.1 |
| Croatia | 10.3 | Egypt | 5.1 |
| France | 20 | Lebanon | 8 |
| Greece | 8.6 | Libya | 5.9 |
| Israel | 4.8 | Morocco | 9.4 |
| Italy | 12.5 | Syria | 2.2 |
| Malta | 24.9 | Tunisia | 8.9 |
| Montenegro | 2.5 | Turkey | 5.5 |
| Slovenia | 5.1 | ||
| Spain | 20.2 |

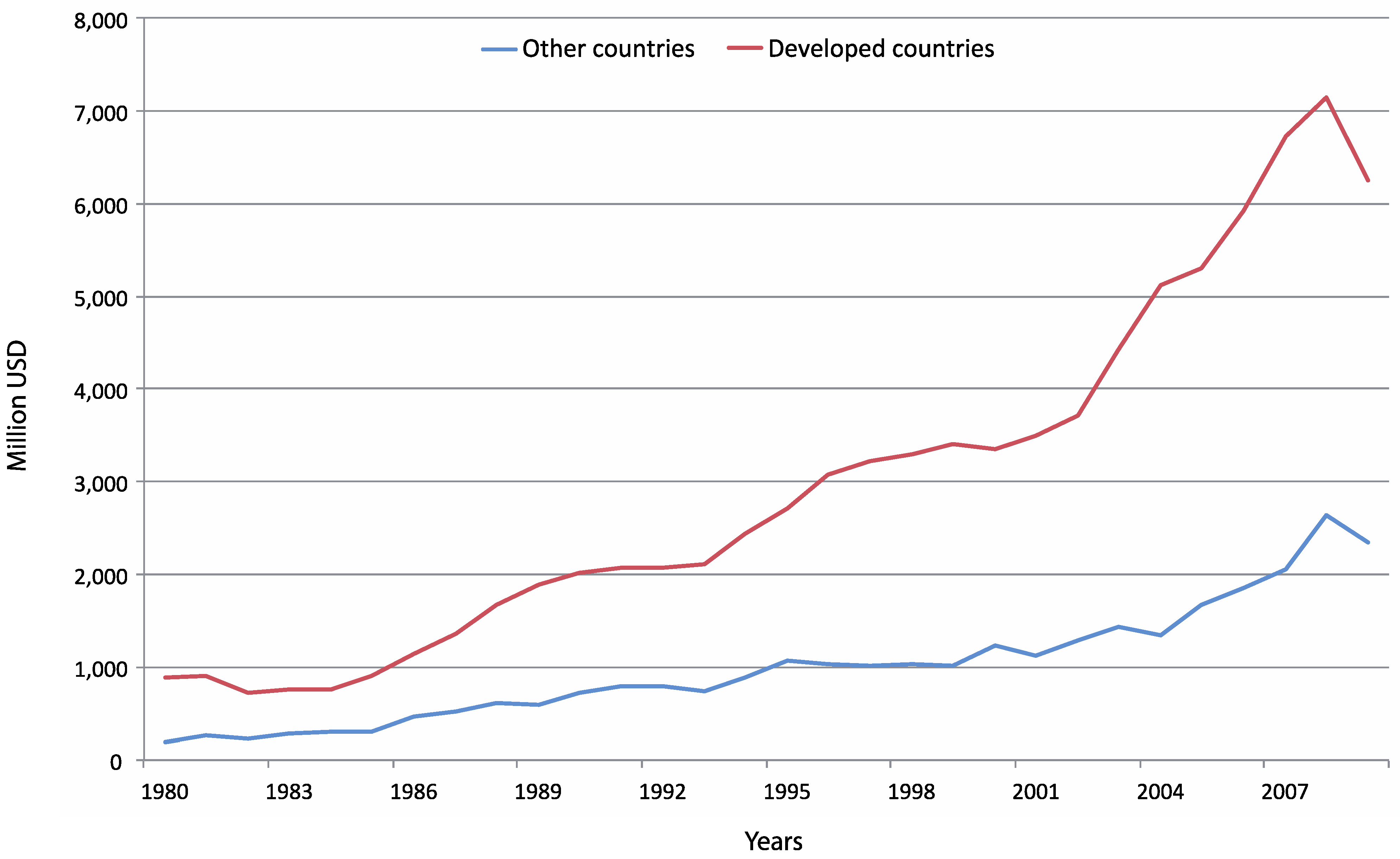
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mangos, A.; Bassino, J.-P.; Sauzade, D. The Economic Value of Sustainable Benefits Rendered by the Mediterranean. Blue Plan Papers 8; United Nations Environment Program/Mediterranean Action Plan Regional Activity Centre: Valbonne, France, 2010. [Google Scholar]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Ben Rais Lasram, F.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediteranean sea: Estimates, patterns, and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef]
- Sabine, C.L.; Feely, R.A.; Gruber, N.; Key, R.M.; Lee, K.; Bullister, J.L.; Wanninkhof, R.; Wong, C.S.; Wallace, W.R.; Tilbrook, B.; et al. The oceanic sink for anthropogenic CO2. Science 2004, 305, 367–371. [Google Scholar] [CrossRef]
- Hofmann, M.; Schellnhuber, H.J. Ocean acidification: A millennial challenge. Energy Environ. Sci. 2010, 3, 1883–1896. [Google Scholar] [CrossRef]
- Dupont, S.; Pörtner, H. Marine science: Get ready for ocean acidification. Nature 2013, 498. [Google Scholar] [CrossRef]
- Ries, J.B.; Cohen, A.L.; McCorkle, D.C. Marine calcifiers exhibit mixed responses to CO2-induced ocean acidification. Geology 2009, 37, 1131–1134. [Google Scholar] [CrossRef]
- Byrne, M.; Gonzalez-Bernat, M.; Doo, S.; Foo, S.; Soars, N.; Lamare, M. Effects of ocean warming and acidification on embryos and non-calcifying larvae of the invasive sea star Patiriella regularis. Mar. Ecol. Prog. Ser. 2013, 473, 235–246. [Google Scholar] [CrossRef]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.; Hendriks, I.E.; Ramajo, L.; Singh, G.S.; Duarte, C.M.; Gattuso, J.-P. Impacts of ocean acidification on marine organisms: Quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 2013, 19, 1884–1896. [Google Scholar] [CrossRef]
- Hendriks, I.E.; Duarte, C.M.; Alvarez, M. Vulnerability of marine biodiversity to ocean acidification: A meta-analysis. Estuar. Coast. Shelf Sci. 2010, 86, 157–164. [Google Scholar] [CrossRef]
- Munday, P.L.; Dixson, D.L.; Donelson, J.M.; Jones, G.P.; Pratchett, M.S.; Devitsina, G.V.; Doving, K.B. Ocean acidification impairs olfactory discrimination and homing ability of a marine fish. Proc. Nat. Acad. Sci. USA 2009, 106, 1848–1852. [Google Scholar] [CrossRef]
- Munday, P.L.; Dixson, D.L.; McCormick, M.I.; Meekan, M.; Ferrari, M.C.; Chivers, D.P. Replenishment of fish populations is threatened by ocean acidification. Proc. Natl. Acad. Sci. USA 2010, 107, 12930–12934. [Google Scholar]
- Hamilton, T-J.; Holcombe, A.; Tresguerres, M. CO2-induced ocean acidification increases anxiety in Rockfish via alteration of GABAA receptor functioning. Proc. R. Soc. B 2014, 281. [Google Scholar] [CrossRef]
- Munday, P.L.; Warner, R.R.; Monro, K.; Pandolfi, J.M.; Marshall, D.J. Predicting evolutionary responses to climate change in the sea. Ecol. Lett. 2013, 16, 1488–1500. [Google Scholar] [CrossRef]
- Creative Commons. CC-BY-SA-3.0. Available online: http://creativecommons.org/licenses/by-sa/3.0 (accessed on 15 February 2014).
- Mathis, J-T.; Cooley, S-R.; Lucey, N.; Colt, S.; Ekstrom, J.; Hurst, T.; Hauri, C.; Evans, W.; Cross, J.N.; Feely, R-A. Ocean Acidification Risk Assessment for Alaska’s Fishery Sector. Prog. Oceanogr. in press.
- Intergovernmental Panel on Climate Change. Workshop Report of the Intergovernmental Panel on Climate Change Workshop on Impacts of Ocean Acidification on Marine Biology and Ecosystems; Field, C.B., Barros, V., Stocker, T.F., Qin, D., Mach, K.J., Plattner, G.-K., Mastrandrea, M.D., Tignor, M., Ebi, K.L., Eds.; IPCC Working Group II Technical Support Unit, Carnegie Institution: Stanford, CA, USA, 2011; p. 164. [Google Scholar]
- Hilmi, N.; Allemand, D.; Dupont, S.; Safa, A.; Haraldsson, G.; Nunes, P.L.D.; Moore, C.; Hattam, C.; Reynaud, S.; Hall-Spencer, J.M.; et al. Towards improved socio-economic assessments of ocean acidification’s impacts. Mar. Biol. 2013, 160, 1773–1787. [Google Scholar] [CrossRef]
- Touratier, F.; Goyet, C. Decadal evolution of anthropogenic CO2 in the northwestern Mediterranean Sea from the mid-1990s to the mid-2000s. DeepSea Res. Pt I 2009, 56, 1708–1716. [Google Scholar] [CrossRef]
- Touratier, F.; Goyet, C. Impact of the Eastern Mediterranean Transient on the distribution of anthropogenic CO2 and first estimate of acidification for the Mediterranean Sea. Deep Sea Res. Pt I 2011, 58, 1–15. [Google Scholar] [CrossRef]
- Palmiéri, J.; Orr, J.C.; Dutay, J.-C.; Béranger, K.; Schneider, A.; Beuvier, J.; Somot, S. Simulated anthropogenic CO2 uptake and acidification of the Mediterranean Sea. Biogeosci. Discuss. 2014, 11, 6461–6517. [Google Scholar] [CrossRef]
- Orr, J.C. Recent and future changes in ocean carbonate chemistry. In Ocean Acidification; Gattuso, J.-P., Hansson, L., Eds.; Oxford University Press: Oxford, UK, 2011; pp. 41–63. [Google Scholar]
- Lavigne, H.; Gattuso, J.-P. Seacarb: Seawater Carbonate Chemistry with R. R Package Version 2.4.1. Available online: http://CRAN.Rproject.org/package=seacarb (accessed on 9 October 2011).
- Copin-Montégut, C.; Bégovic, M. Distributions of carbonate properties and oxygen along the water column (0–2000 m) in the central part of the NW Mediterranean Sea (Dyfamed site): Influence of winter vertical mixing on air–sea CO2 and O2 exchanges. DeepSea Res. Pt II 2002, 49, 2049–2066. [Google Scholar] [CrossRef]
- Ricke, K.L.; Orr, J.C.; Schneider, K.; Caldeira, K. Risks to coral reefs from ocean carbonate chemistry changes in recent earth system model projections. Environ. Res. Lett. 2013, 8. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Fisheries and Aquaculture. Available online: http://www.fao.org/fishery/naso/search/en (accessed on 15 February 2012).
- Food and Agriculture Organization. Fisheries and Aquaculture. Available online: http://www.fao.org/fishery/culturedspecies/search/en (accessed on 15 February 2012).
- Dissanayake, A. Ocean acidification and warming effects on crustacea: Possible future scenarios. In The Mediterranean Sea: Its History and Present Challenges; Goffredo, S., Dubinsky, Z., Eds.; Springer: Dordrecht, the Netherlands, 2013; pp. 363–372. [Google Scholar]
- Dupont, S.T.; Thorndyke, M.S. Direct impacts of near-future ocean acidification on sea urchins. In Climate Change Perspective from the Atlantic: Past, Present and Future; Fernández-Palacios, J.M., de Nascimento, L., Hernández, J.C., Clemente, S., González, A., Díaz-González, J.P., Eds.; Servicio de Publicaciones, Universidad de La Laguna: Santa Cruz de Tenerife, Spain, 2013; pp. 461–485. [Google Scholar]
- Gazeau, F.; Parker, L.M.; Comeau, S.; Gattuso, J.-P.; O’Connor, W.A.; Martin, S.; Pörtner, H.O.; Ross, P.M. Impacts of ocean acidification on marine shelled molluscs. Mar. Biol. 2013, 8, 2207–2245. [Google Scholar]
- Sunday, J.M.; Calosi, P.; Dupont, S.; Munday, P.L.; Stillman, J.H.; Reusch, T.B.H. Evolution in an acidifying ocean. Trends Ecol. Evol. 2014, 29, 117–125. [Google Scholar] [CrossRef]
- Dupont, S.; Dorey, N.; Thorndyke, M. What meta-analysis can tell us on vulnerability of marine biodiversity to ocean acidification? Estuar. Coast. Shelf Sci. 2010, 89, 182–185. [Google Scholar] [CrossRef]
- Wittmann, A.; Pörtner, H.O. Sensitivities of extant animal taxa to ocean acidification. Nat.Clim. Chang. 2013, 3, 995–1001. [Google Scholar] [CrossRef]
- Hall-Spencer, J.M.; Rodolfo-Metalpa, R.; Martin, S.; Ransome, E.; Fine, M.; Turner, S.M.; Rowley, S.J.; Tedesco, D.; Buia, M.-C. Volcanic carbon dioxide vents reveal ecosystem effects of ocean acidification. Nature 2008, 454, 96–99. [Google Scholar] [CrossRef]
- Kerrison, P.; Hall-Spencer, J.M.; Suggett, D.; Hepburn, L.J.; Steinke, M. Assessment of pH variability at coastal CO2 vents for ocean acidification studies. Estuar. Coast. Shelf Sci. 2011, 94, 129–137. [Google Scholar]
- Barry, J.-P.; Hall-Spencer, J.M.; Tyrell, T. In situ perturbation experiments: Natural venting sites, spatial/temporal gradients in ocean pH, manipulative in situ pCO2 perturbations. In Guide to Best Practices in Ocean Acidification Research and Data Reporting; Riebesell, U., Fabry, V.J., Hansson, L., Gattuso, J.-P., Eds.; Publications Office of the European Union: Luxembourg, Luxembourg, 2010; pp. 123–136. [Google Scholar]
- Cigliano, M.; Gambi, M.C.; Rodolfo-Metalpa, R.; Patti, F.P.; Hall-Spencer, J.M. Effects of ocean acidification on invertebrate settlement. Mar. Biol. 2010, 157, 2489–2502. [Google Scholar] [CrossRef]
- Hall-Spencer, J.M. No reason for complacency. Nat. Clim. Chang. 2011, 1, 174. [Google Scholar] [CrossRef]
- Riebesell, U. Climate change: Acid test for marine biodiversity. Nature 2008, 454, 46–47. [Google Scholar] [CrossRef]
- Wernberg, T.; Smale, D.A.; Thomsen, M.S. A decade of climate change experiments on marine organisms: Procedures, patterns and problems. Glob. Chang. Biol. 2012, 18, 1491–1498. [Google Scholar] [CrossRef]
- Suggett, D.J.; Hall-Spencer, J.M.; Rodolfo-Metalpa, R.M.; Boatman, T.G.; Payton, R.; Pettay, D.T.; Johnson, V.R.; Warner, M.E.; Lawson, T. Sea anemones may thrive in a high CO2 world. Glob. Chang. Biol. 2012, 18, 3015–3025. [Google Scholar] [CrossRef]
- Calosi, P.; Rastrick, S.P.S.; Graziano, M.; Thomas, S.C.; Baggini, C.; Carter, H.A.; Hall-Spencer, J.M.; Milazzo, M.; Spicer, J.I. Acid-base and ion-regulation capacity-dependent distribution of sea urchins living near shallow water CO2 vents. Mar. Pollut. Bull. 2013, 73, 470–484. [Google Scholar] [CrossRef]
- Rodolfo-Metalpa, R.; Houlbrèque, F.; Tambutté, É.; Boisson, F.; Baggini, C.; Patti, F.P.; Jeffree, R.; Fine, M.; Foggo, A.; Gattuso, J.-P.; et al. Coral and mollusc resistance to ocean acidification adversely affected by warming. Nat. Clim. Chang. 2011, 1, 308–312. [Google Scholar] [CrossRef]
- Dupont, S.; Ortega-Martinez, O.; Thorndyke, M. Impact of near-future ocean acidification on echinoderms. Ecotoxicology 2010, 19, 449–462. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. The State of World Fisheries and Aquaculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; p. 176. [Google Scholar]
- Franquesa, R.; Oliver, P.; Basurco, B. The Mediterranean fisheries sector: A review of facts and figures. In The Mediterranean Fisheries Sector. A Reference Publication for the VII Meeting of Ministers of Agriculture and Fisheries of CIHEAM Member Countries; Basurco, B., Ed.; Options Mediterraneéenes: Séries B; Estudes et Recherches: Zaragoza, Spain, 2008; Volume 62, pp. 9–41. [Google Scholar]
- FAO Country Profiles. Available online: http://www.fao.org/fishery/countryprofiles/search/en (accessed on10 December 2013).
- General Fisheries Commission for the Mediterranean. General Fisheries Commission for the Mediterranean Scientific Advisory Committee (SAC). GFCM, Rome. 2011. Available online: http://www.pescaricreativa.org/docs/fao/gfcm_sac102010.pdf (accessed on 15 February 2012).
- SEC. Communication from the Commission to the European Parliament and the Council—Building a Sustainable Future for Aquaculture—A New Impetus for the Strategy for the Sustainable Development of European Aquaculture {SEC(2009) 453} {SEC(2009) 454}; SEC: Bruxelles, Belgium, 2009. [Google Scholar]
- García-Flórez, L.; Morales, J.; Gaspar, M.B.; Castilla, D.; Mugerza, E.; Berthou, P.; García de la Fuente, L.; Oliveira, M.; Moreno, O.; García del Hoyo, J.J.; et al. A novel and simple approach to define artisanal fisheries in Europe. Mar. Policy 2014, 44, 152–159. [Google Scholar] [CrossRef]
- Lleonart, J.; Maynou, F. Fish stock assessments in the Mediterranean: State of the art. Sci. Mar. 2003, 61, 37–49. [Google Scholar]
- Macfadyen, G.; Salz, P.; Cappell, R. Characteristics of Small-Scale Coastal Fisheries in Europe; European Parliament: Brussels, Belgium, 2011. [Google Scholar]
- Guyader, O.; Berthou, P.; Koutsikopoulos, C.; Alban, F.; Demanèche, S.; Gaspar, M.B.; Eschbaum, R.; Fahy, E.; Tully, O.; Reynal, L.; et al. Small scale fisheries in Europe: A comparative analysis based on a selection of case studies. Fish. Res. 2013, 140, 1–13. [Google Scholar] [CrossRef]
- Joint Research Centre. Summary of the Annual Economic Report on the EU Fishing Fleet; Anderson, J., Carvalho, N., Eds.; Publications Office of the European Union: Luxembourg, Luxembourg, 2012. [Google Scholar]
- Marttin, F.; Barone, M.; Bizsel, C.; Fayed, S.; Hadjistephanou, N.; Krouma, I.; Majdalani, S.; Özdemire, A.; Salem, A.; Vassiliades, L. Brief Introduction to the Eastern Mediterranean Fisheries Sector; Food and Agriculture Organization of the United Nations: Rome, Italy, 2006. [Google Scholar]
- Turkish Statistical Institute. Available online: http://www.turkstat.gov.tr/PreTabloArama.do?metod=search&araType=vt (accessed on 20 May 2012).
- Turkstat, Fisheries Statistics. Turkiye Istatistik Kurumu. Available online: http://www.tuik.gov.tr (accessed on 20 May 2012).
- Andersson, A.J.; Mackenzie, F.T. Revisiting four scientific debates in ocean acidification research. Biogeosciences 2012, 9, 893–905. [Google Scholar] [CrossRef]
- Franquesa, R.; Gordoa, A.; Mona, T.; Nuss, S.; Borrego, J.R. The Recreational Fishing in the Central and Western European Mediterranean frame. GEM UB.. Available online: www.gemub.com/pdf/recreofao.pdf (accessed on 15 January 2013).
- Herfaut, J.; Levrel, H.; Thébaud, O.; Veron, G. The nationwide assessment of marine recreational fishing: A French example. Ocean Coast. Manag. 2013, 78, 121–131. [Google Scholar] [CrossRef]
- FAO Fisheries and Aquaculture Department. Statistics and Information Service FishStat: Universal Software for Fishery Statistical Time Series. Available online: http://www.fao.org/fishery/statistics/software/fishstat/en (accessed on 17 November 2011).
- Lloret, J.; Zaragoza, N.; Caballero, D.; Riera, V. Biological and socio-economic implications of recreational boat fishing for the management of fisheries resources in the marine reserve of Cap de Creus (NW Mediterranean). Fish. Res. 2008, 91, 252–259. [Google Scholar] [CrossRef]
- Morales-Nin, B.; Moranta, J.; García, C.; Tugores, M.P.; Grau, A.M.; Riera, F.; Cerdà, M. The recreational fishery off Majorca Island (western Mediterranean): Some implications for coastal resource management. ICES J. Mar. Sci. 2005, 62, 727–739. [Google Scholar] [CrossRef]
- Gaudin, C.; de Young, C. Recreational Fisheries in the Mediterranean Countries: A Review of Existing Legal Frameworks; Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. [Google Scholar]
- Organisation for Economic Co-operation and Development. OECD Review of Fisheries 2011; OECD Publishing: Paris, France, 2012. [Google Scholar]
- World Bank Development Indicators. Available online: http://data.worldbank.org/ (accessed on 20 May 2013).
- Malvarosa, L.; de Young, C. Fish Trade among Mediterranean Countries: Intraregional Trade and Import-Export with the European Union; Studies and Reviews: General Fisheries Commission for the Mediterranean. No. 86; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010. [Google Scholar]
- Duarte, C.M.; Hendriks, I.E.; More, T.S.; Olsen, Y.S.; Steckbauer, A.; Ramajo, L.; Carstensen, J.; Trotter, J.A.; McCulloch, M. Is ocean acidification an open-ocean syndrome? Understanding anthropogenic impacts on seawater pH. Estuaries Coasts 2013, 36, 221–236. [Google Scholar] [CrossRef]
- Barton, A.; Hales, B.; Waldbusser, G-G.; Langdon, C.; Feely, R-A. The Pacific oyster, Crassostrea gigas, shows negative correlation to naturally elevated carbon dioxide levels: Implications for near-term ocean acidification effects. Limnol. Oceanogr. 2012, 57, 698–710. [Google Scholar] [CrossRef]
- Cooley, S.R.; Lucey, N.; Kite-Powell, H.; Doney, S.C. Nutrition and income from mollusks today imply vulnerability to ocean acidification tomorrow. Fish Fish. 2012, 13, 182–215. [Google Scholar] [CrossRef]
- Rodrigues, L.C.; van den Bergh, J.C.; Ghermandi, A. Socio-economic impacts of ocean acidification in the Mediterranean Sea. Mar. Policy 2013, 38, 447–456. [Google Scholar] [CrossRef]
- Allison, E.-H.; Perry, A.-L.; Badjeck, M.-C.; Adger, W.-N.; Brown, K.; Conway, D.; Halls, A.-S.; Pilling, G.-M.; Dulvy, N.-K. Vulnerability of national economies to the impacts of climate change on fisheries. Fish Fish. 2009, 10, 173–196. [Google Scholar] [CrossRef]
Appendix
| Phylum | Species Name | Aquaculture | Fishery |
|---|---|---|---|
| Cnidarians | Anemonia sulcata (=A. viridis) | x | |
| Corallium rubrum | x | ||
| Crustaceans | Aristeus antennatus | x | |
| Carcinus aestuarii | x | ||
| Dromia personata | x | ||
| Eriphia verrucosa | x | ||
| Hommarus gammarus | x | ||
| Liocarcinus corrugatus | x | ||
| Maja crispata | x | ||
| Maja squinado | x | ||
| Nephrops norvegicus | x | ||
| Palaemon serratus | x | ||
| Palinurus elephas | x | ||
| Palinurus mauritanicus | x | ||
| Parapenaeus longirostris | x | ||
| Paromola cuvieri | x | ||
| Penaeus indicus | x | ||
| Penaeus vannamei | x | ||
| Scyllarides lattus | x | ||
| Scyllarus arctus | x | ||
| Squilla mantis | x | ||
| Echinoderms | Paracentrotus lividus | x | |
| Fish | Acipenser baerii | x | |
| Argyrosomus regius | x | ||
| Boops boops | x | ||
| Conger conger | x | ||
| Dentex dentex | x | ||
| Dicentrarchus labrax | x | ||
| Engraulis encrasicolus | x | ||
| Lophius piscatorius | x | ||
| Merluccius merluccius | x | ||
| Mricomesistius poutassou | x | ||
| Mugil cephalus | x | ||
| Mugilidae sp. | x | ||
| Mullus barbatus | x | ||
| Mullus surmuletus | x | ||
| Oncorhyncus mykiss | x | ||
| Pagellus bogaraveo | x | ||
| Pagellus erythrinus | x | ||
| Pagrus major | x | ||
| Pagrus pagrus | x | ||
| Phycis blennioides | x | ||
| Pristirius melanostomus | x | ||
| Psetta maxima | x | ||
| Puntazzo puntazzo | x | ||
| Sardina pilchardus | x | ||
| Sardinella aurita | x | ||
| Sciaenops ocellatus | x | ||
| Scomber scombrus | x | ||
| Scophthalmus maeoticus | x | ||
| Siganus rivulatus | x | ||
| Sparus aurata | x | ||
| Spicara maena | x | ||
| Spicara smaris | x | ||
| Trachurus trachurus | x | ||
| Trachyrhynchus trachyrhynchus | x | ||
| Trisopterus minutus | x | ||
| Umbrina cirrosa | x | ||
| Molluscs | Aequipecten opercularis | x | |
| Alloteuthis media | x | ||
| Arca noae | x | ||
| Astrae rugosa | x | ||
| Bolinus brandaris | x | ||
| Buccinulum corneum | x | ||
| Cardium edule | x | ||
| Ceratoderma glaucum | x | ||
| Cerithium vulgatum | x | ||
| Crassostrea gigas | x | ||
| Donax semistriatus | x | ||
| Eledone cirrhosa | x | ||
| Eledone moschata | x | ||
| Ensis siliqua | x | ||
| Glycimeris bimaculata | x | ||
| Haliotis tuberculata | x | ||
| Hexaplex trunculus | x | ||
| Loligo vulgaris | x | ||
| Monodonta articulata | x | ||
| Mytilus edulis | x | ||
| Mytilus galloprovincialis | x | ||
| Naverita josephina | x | ||
| Octopus vulgaris | x | ||
| Ostrea edulis | x | x | |
| Patella caerulea | x | ||
| Pecten jacobaeus | x | ||
| Rossia macrosoma | x | ||
| Ruditapes decussatus | x | x | |
| Ruditapes philippinarum | x | ||
| Sepia elegans | x | ||
| Sepia officinalis | x | ||
| Tadarodes sagittatus | x | ||
| Venerupis aurea | x | ||
| Venus verrucosa | x | ||
| Tunicates | Microcosmus sulcatus | x | |
| Microcosmus sabatieri | x |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hilmi, N.; Allemand, D.; Cinar, M.; Cooley, S.; Hall-Spencer, J.M.; Haraldsson, G.; Hattam, C.; Jeffree, R.A.; Orr, J.C.; Rehdanz, K.; et al. Exposure of Mediterranean Countries to Ocean Acidification. Water 2014, 6, 1719-1744. https://doi.org/10.3390/w6061719
Hilmi N, Allemand D, Cinar M, Cooley S, Hall-Spencer JM, Haraldsson G, Hattam C, Jeffree RA, Orr JC, Rehdanz K, et al. Exposure of Mediterranean Countries to Ocean Acidification. Water. 2014; 6(6):1719-1744. https://doi.org/10.3390/w6061719
Chicago/Turabian StyleHilmi, Nathalie, Denis Allemand, Mine Cinar, Sarah Cooley, Jason M Hall-Spencer, Gunnar Haraldsson, Caroline Hattam, Ross A. Jeffree, James C. Orr, Katrin Rehdanz, and et al. 2014. "Exposure of Mediterranean Countries to Ocean Acidification" Water 6, no. 6: 1719-1744. https://doi.org/10.3390/w6061719
APA StyleHilmi, N., Allemand, D., Cinar, M., Cooley, S., Hall-Spencer, J. M., Haraldsson, G., Hattam, C., Jeffree, R. A., Orr, J. C., Rehdanz, K., Reynaud, S., Safa, A., & Dupont, S. (2014). Exposure of Mediterranean Countries to Ocean Acidification. Water, 6(6), 1719-1744. https://doi.org/10.3390/w6061719






