The Impact of Ocean Acidification on Reproduction, Early Development and Settlement of Marine Organisms
Abstract
1. Introduction
2. Fertilisation
3. Larvae
3.1. Echinoderms
3.2. Molluscs
3.3. Mechanisms in Echinoderms and Molluscs
3.4. Crustaceans
3.5. Corals
3.6. Fish
4. Summary
5. Conclusions
Supplementary Materials
Acknowledgments
References
- Caldeira, K.; Wickett, M.E. Anthropogenic carbon and ocean pH. Nature 2003, 425, 365–365. [Google Scholar] [CrossRef]
- Guinotte, J.M.; Fabry, V.J. Ocean acidification and its potential effects on marine ecosystems. Ann. N. Y. Acad. Sci. 2008, 1134, 320–342. [Google Scholar] [CrossRef] [PubMed]
- Turley, C.; Blackford, J.C.; Widdicombe, S.; Lowe, D.; Nightingale, P.D.; Rees, A.P. Reviewing the impact of increased atmospheric CO2 on oceanic pH and the marine ecosystem. In Avoiding Dangerous Climate Change; Schellnhuber, H.J., Cramer, W., Nakicenovic, N., Wigley, T., Yohe, G., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 65–70. [Google Scholar]
- Royal Society. Ocean Acidification due to Increasing Atmospheric Carbon Dioxide. Policy Document 12/05; The Royal Society: London, UK, 2005; pp. 1–58. [Google Scholar]
- Pörtner, H.O.; Farrell, A.P. Ecology: Physiology and climate change. Science 2008, 322, 690–692. [Google Scholar] [CrossRef] [PubMed]
- Thorson, G. Reproduction and larval ecology of marine bottom invertebrates. Biol. Rev. 1950, 25, 1–45. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, H. Effects of CO2-driven ocean acidification on the early developmental stages of invertebrates. Mar. Ecol. Prog. Ser. 2008, 373, 275–284. [Google Scholar] [CrossRef]
- Dupont, S.; Thorndyke, M.C. Impact of CO2-driven ocean acidification on invertebrates early life-history—What we know, what we need to know and what we can do. Biogeosci. Discuss. 2009, 6, 3109–3131. [Google Scholar]
- Pörtner, H.O.; Langenbuch, M.; Reipschläger, A. Biological impact of elevated ocean CO2 concentrations: Lessons from animal physiology and earth history. J. Oceanogr. 2004, 60, 705–718. [Google Scholar] [CrossRef]
- Przeslawski, R.; Ahyong, S.; Byrne, M.; Wörheide, G.; Hutchings, P. Beyond corals and fish: The effects of climate change on noncoral benthic invertebrates of tropical reefs. Glob. Change Biol. 2008, 14, 2773–2795. [Google Scholar] [CrossRef]
- Kleypas, J.A.; Buddemeier, R.W.; Archer, D.; Gattuso, J.-P.; Langdon, C.; Opdyke, B.N. Geochemical consequences of increased atmospheric carbon dioxide on coral reefs. Science 1999, 284, 118–120. [Google Scholar] [CrossRef]
- Gooding, R.A.; Harley, C.D.G.; Tang, E. Elevated water temperature and carbon dioxide concentration increase the growth of a keystone echinoderm. Proc. Natl. Acad. Sci. USA 2009, 106, 9316–9321. [Google Scholar] [CrossRef]
- Hendriks, I.E.; Duarte, C.M.; Álvarez, M. Vulnerability of marine biodiversity to ocean acidification: A meta-analysis. Estuar. Coast. Shelf Sci. 2010, 86, 157–164. [Google Scholar] [CrossRef]
- Dupont, S.; Dorey, N.; Thorndyke, M. What meta-analysis can tell us about vulnerability of marine biodiversity to ocean acidification? Estuar. Coast. Shelf Sci. 2010, 89, 182–185. [Google Scholar] [CrossRef]
- Hendriks, I.E.; Duarte, C.M. Ocean acidification: Separating evidence from judgment—A reply to Dupont et al. Estuar. Coast. Shelf Sci. 2010, 89, 186–190. [Google Scholar] [CrossRef]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.N.; Singh, G.G. Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol. Lett. 2010, 13, 1419–1434. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, A.; Davis, H.C. The pH tolerance of embryos and larvae of Mercenaria mercenaria and Crassostrea virginica. Biol. Bull. 1966, 131, 427–436. [Google Scholar] [CrossRef]
- Wilson, S.P.; Hyne, R.V. Toxicity of acid-sulfate soil leachate and aluminum to embryos of the Sydney Rock oyster. Ecotoxicol. Environ. Saf. 1997, 37, 30–36. [Google Scholar] [CrossRef]
- Pagano, G.; Cipollaro, M.; Corsale, G. pH-Induced changes in mitotic and developmental patterns in sea urchin embryogenesis. I. Exposure of embryos. Teratog. Carcinog. Mutag. 1985, 5, 101–112. [Google Scholar] [CrossRef]
- Pagano, G.; Cipollaro, M.; Corsale, G. pH-Induced changes in mitotic and developmental patterns in sea urchin embryogenesis. II. Exposure of sperm. Teratog. Carcinog. Mutag. 1985, 5, 113–121. [Google Scholar] [CrossRef]
- Cipollaro, M.; Corsale, G.; Esposito, A. Sublethal pH decrease may cause genetic damage to eukaryotic cell: A study on sea urchins and Salmonella typhimurium. Teratog. Carcinog. Mutag. 1986, 6, 275–287. [Google Scholar] [CrossRef]
- Alvarado-Alvarez, R.; Gould, M.C.; Stephano, J.L. Spawning, in vitro maturation, and changes in oocyte electrophysiology induced by serotonin in Tivela stultorum. Biol. Bull. 1996, 190, 322–328. [Google Scholar] [CrossRef]
- Desrosiers, R.R.; Désilets, J.; Dubé, F. Early developmental events following fertilization in the giant scallop Placopecten magellanicus. Can. J. Fish. Aquat. Sci. 1996, 53, 1382–1392. [Google Scholar] [CrossRef]
- Ishimatsu, A.; Kikkawa, T.; Hayashi, M.; Lee, K.-S.; Kita, J. Effects of CO2 on marine fish: Larvae and adults. J. Oceanogr. 2004, 60, 731–741. [Google Scholar] [CrossRef]
- Brownell, C.L. Water quality requirements for first-feeding in marine fish larvae. II. pH, oxygen, and carbon dioxide. J. Exp. Mar. Biol. Ecol. 1980, 44, 285–298. [Google Scholar] [CrossRef]
- Ross, P.M.; Parker, L.M.; O’Connor, W.A. The impact of ocean acidification on reproduction and early development of marine organisms. Aust. Zool. in press.
- Gattuso, J.P.; Gao, K.; Lee, K.; Rost, B.; Schulz, K.G. Approaches and tools to manipulate the carbonate chemistry. In Guide to Best Practices for Ocean Acidification Research and Data Reporting; Riebesell, U., Fabry, V.J., Hansson, L., Gattuso, J.-P., Eds.; Publications Office of the European Union: Luxembourg, Luxembourg, 2010; pp. 41–51. [Google Scholar]
- Intergovernmental Panel on Climate Change. The Scientific Basis: Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change; Houghton, J.T., Ding, Y., Griggs, D.J., Noguer, M., van der Linden, P.J., Dai, X., Maskell, K., Johnson, C.A., Eds.; Cambridge University Press: Cambridge, UK and New York, NY, USA, 2001; p. 881. [Google Scholar]
- Intergovernmental Panel on Climate Change. Synthesis Report: Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R.K., Reisinger, A., Eds.; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2007; p. 104. [Google Scholar]
- Caldeira, K.; Wickett, M. Ocean model predictions of chemistry changes from carbon dioxide emissions to the atmosphere and ocean. J. Geophys. Res. C 2005, 110, 1–12. [Google Scholar] [CrossRef]
- Orr, J.C.; Fabry, V.J.; Aumont, O.; Bopp, L.; Doney, S.C.; Feely, R.A.; Gnanadesikan, A.; Gruber, N.; Ishida, A.; Joos, F.; et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 2005, 437, 681–686. [Google Scholar] [CrossRef]
- Dupont, S.; Lundve, B.; Thorndyke, M. Near future ocean acidification increases growth rate of the lecithotrophic larvae and juveniles of the sea star Crossaster papposus. J. Exp. Zool. Part B 2010, 314 B, 382–389. [Google Scholar] [CrossRef]
- Kurihara, H.; Kato, S.; Ishimatsu, A. Effects of increased seawater pCO2 on early development of the oyster Crassostrea gigas. Aquat. Biol. 2007, 1, 91–98. [Google Scholar] [CrossRef]
- Byrne, M.; Ho, M.; Selvakumaraswamy, P.; Nguyen, H.D.; Dworjanyn, S.A.; Davis, A.R. Temperature, but not pH, compromises sea urchin fertilization and early development under near-future climate change scenarios. Proc. R. Soc. B Biol. Sci. 2009, 276, 1883–1888. [Google Scholar] [CrossRef]
- Parker, L.M.; Ross, P.M.; O’Connor, W.A. The effect of ocean acidification and temperature on the fertilization and embryonic development of the Sydney rock oyster Saccostrea glomerata (Gould 1850). Glob. Change Biol. 2009, 15, 2123–2136. [Google Scholar] [CrossRef]
- Dupont, S.; Ortega-Martínez, O.; Thorndyke, M. Impact of near-future ocean acidification on echinoderms. Ecotoxicology 2010, 19, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Beiras, R.; His, E. Effects of dissolved mercury on embryogenesis, survival, growth and metamorphosis of Crassostrea gigas oyster larvae. Mar. Ecol. Prog. Ser. 1994, 113, 95–104. [Google Scholar] [CrossRef]
- Green, M.A.; Jones, M.E.; Boudreau, C.L.; Moore, R.L.; Westman, B.A. Dissolution mortality of juvenile bivalves in coastal marine deposits. Limnol. Oceanogr. 2004, 49, 727–734. [Google Scholar] [CrossRef]
- Parker, L.M.; Ross, P.M.; O’Connor, W.A. Comparing the effect of elevated pCO2 and temperature on the fertilization and early development of two species of oysters. Mar. Biol. 2010, 157, 2435–2452. [Google Scholar] [CrossRef]
- Gosselin, L.A.; Qian, P.-Y. Juvenile mortality in benthic marine invertebrates. Mar. Ecol. Prog. Ser. 1997, 146, 265–282. [Google Scholar] [CrossRef]
- Impacts of Ocean Acidification. Science Policy Briefing 37; European Science Foundation: Strasbourg, France, 2009; pp. 1–12. [Google Scholar]
- Moulin, L.; Catarino, A.I.; Claessens, T.; Dubois, P. Effects of seawater acidification on early development of the intertidal sea urchin Paracentrotus lividus (Lamarck 1816). Mar. Pollut. Bull. 2011, 62, 48–54. [Google Scholar] [CrossRef]
- Parker, L.M.; Ross, P.M.; O’Connor, W.A. Populations of the Sydney rock oyster, Saccostrea glomerata, vary in response to ocean acidification. Mar. Biol. 2011, 158, 689–697. [Google Scholar] [CrossRef]
- Waldbusser, G.G.; Bergschneider, H.; Green, M.A. Size-dependent pH effect on calcification in post-larval hard clam Mercenaria spp. Mar. Ecol. Prog. Ser. 2010, 417, 171–182. [Google Scholar] [CrossRef]
- Sheppard Brennand, H.; Soars, N.; Dworjanyn, S.A.; Davis, A.R.; Byrne, M. Impact of ocean warming and ocean acidification on larval development and calcification in the sea urchin Tripneustes gratilla. PLoS One 2010, 5, 1–7. [Google Scholar] [CrossRef]
- Parker, L.M.; Ross, P.M.; Raftos, D.A.; Thompson, E.; O’Connor, W.A. The proteomic response of larvae of the Sydney rock oyster, Saccostrea glomerata to elevated pCO2. Aust. Zool. in press.
- Byrne, M.; Soars, N.; Selvakumaraswamy, P.; Dworjanyn, S.A.; Davis, A.R. Sea urchin fertilization in a warm, acidified and high pCO2 ocean across a range of sperm densities. Mar. Environ. Res. 2010, 69, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Byrne, M.; Soars, N.A.; Ho, M.A.; Wong, E.; McElroy, D.; Selvakumaraswamy, P.; Dworjanyn, S.A.; Davis, A.R. Fertilization in a suite of coastal marine invertebrates from SE Australia is robust to near-future ocean warming and acidification. Mar. Biol. 2010, 157, 2061–2069. [Google Scholar] [CrossRef]
- Havenhand, J.N.; Schlegel, P. Near-future levels of ocean acidification do not affect sperm motility and fertilization kinetics in the oyster Crassostrea gigas. Biogeosciences 2009, 6, 3009–3015. [Google Scholar] [CrossRef]
- Kurihara, H.; Ishimatsu, A. Effects of high CO2 seawater on the copepod (Acartia tsuensis) through all life stages and subsequent generations. Mar. Pollut. Bull. 2008, 56, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Frommel, A.Y.; Stiebens, V.; Clemmesen, C.; Havenhand, J. Effect of ocean acidification on marine fish sperm (Baltic cod: Gadus morhua). Biogeosci. Discuss. 2010, 7, 5859–5872. [Google Scholar]
- Kurihara, H.; Shirayama, Y. Effects of increased atmospheric CO2 on sea urchin early development. Mar. Ecol. Prog. Ser. 2004, 274, 161–169. [Google Scholar] [CrossRef]
- Kurihara, H.; Shimode, S.; Shirayama, Y. Sub-lethal effects of elevated concentration of CO2 on planktonic copepods and sea urchins. J. Oceanogr. 2004, 60, 743–750. [Google Scholar] [CrossRef]
- Ericson, J.A.; Lamare, M.D.; Morley, S.A.; Barker, M.F. The response of two ecologically important Antarctic invertebrates (Sterechinus neumayeri and Parborlasia corrugatus) to reduced seawater pH: Effects on fertilisation and embryonic development. Mar. Biol. 2010, 157, 2689–2702. [Google Scholar] [CrossRef]
- Reuter, K.E.; Lotterhos, K.E.; Crim, R.N.; Thompson, C.A.; Harley, C.D.G. Elevated pCO2 increases sperm limitation and risk of polyspermy in the red sea urchin Strongylocentrotus franciscanus. Global Change Biol. 2011, 17, 163–171. [Google Scholar] [CrossRef]
- Havenhand, J.N.; Buttler, F.-R.; Thorndyke, M.C.; Williamson, J.E. Near-future levels of ocean acidification reduce fertilization success in a sea urchin. Curr. Biol. 2008, 18, 651–652. [Google Scholar] [CrossRef]
- Kurihara, H.; Asai, T.; Kato, S.; Ishimatsu, A. Effects of elevated pCO2 on early development in the mussel Mytilus galloprovincialis. Aquat. Biol. 2008, 4, 225–233. [Google Scholar] [CrossRef]
- Albright, R.; Mason, B.; Miller, M.; Langdon, C. Ocean acidification compromises recruitment success of the threatened Caribbean coral Acropora palmata. Proc. Natl. Acad. Sci. USA 2010, 107, 20400–20404. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.M.; Ross, P.M.; O’Connor, W.A.; Borysko, L.; Raftos, D.A.; Portner, H.O. Adult exposure influences offspring response to ocean acidification in oysters. Glob. Change Biol. in press. [CrossRef]
- Clark, D.; Lamare, M.; Barker, M. Response of sea urchin pluteus larvae (Echinodermata: Echinoidea) to reduced seawater pH: A comparison among a tropical, temperate, and a polar species. Mar. Biol. 2009, 156, 1125–1137. [Google Scholar] [CrossRef]
- Dupont, S.; Havenhand, J.; Thorndyke, W.; Peck, L.; Thorndyke, M. Near-future level of CO2-driven ocean acidification radically affects larval survival and development in the brittlestar Ophiothrix fragilis. Mar. Ecol. Prog. Ser. 2008, 373, 285–294. [Google Scholar] [CrossRef]
- Gutowska, M.A.; Melzner, F. Abiotic conditions in cephalopod (Sepia officinalis) eggs: Embryonic development at low pH and high pCO2. Mar. Biol. 2009, 156, 515–519. [Google Scholar] [CrossRef]
- Ellis, R.P.; Bersey, J.; Rundle, S.D.; Hall-Spencer, J.M.; Spicer, J.I. Subtle but significant effects of CO2 acidified seawater on embryos of the intertidal snail, Littorina obtusata. Aquat. Biol. 2009, 5, 41–48. [Google Scholar] [CrossRef]
- Watson, S.-A.; Southgate, P.C.; Tyler, P.A.; Peck, L.S. Early larval development of the Sydney rock oyster Saccostrea glomerata under near-future predictions of CO2-driven ocean acidification. J. Shellfish Res. 2009, 28, 431–437. [Google Scholar] [CrossRef]
- Gazeau, F.; Gattuso, J.-P.; Dawber, C.; Pronker, A.E.; Peene, F.; Peene, J.; Heip, C.H.R.; Middelburg, J.J. Effect of ocean acidification on the early life stages of the blue mussel (Mytilus edulis). Biogeosci. Discuss. 2010, 7, 2927–2947. [Google Scholar]
- Lischka, S.; Büdenbender, J.; Boxhammer, T.; Riebesell, U. Impact of ocean acidification and elevated temperatures on early juveniles of the polar shelled pteropod Limacina helicina: Mortality, shell degradation, and shell growth. Biogeosci. Discuss. 2010, 7, 8177–8214. [Google Scholar] [CrossRef]
- Talmage, S.C.; Gobler, C.J. Effects of past, present, and future ocean carbon dioxide concentrations on the growth and survival of larval shellfish. Proc. Natl. Acad. Sci. USA 2010, 107, 17246–17251. [Google Scholar] [CrossRef] [PubMed]
- Comeau, S.; Gorsky, G.; Alliouane, S.; Gattuso, J.-P. Larvae of the pteropod Cavolinia inflexa exposed to aragonite undersaturation are viable but shell-less. Mar. Biol. 2010, 157, 2341–2345. [Google Scholar] [CrossRef]
- Range, P.; Chícharo, M.A.; Ben-Hamadou, R.; Piló, D.; Matias, D.; Joaquim, S.; Oliveira, A.P.; Chícharo, L. Calcification, growth and mortality of juvenile clams Ruditapes decussatus under increased pCO2 and reduced pH: Variable responses to ocean acidification at local scales? J. Exp. Mar. Biol. Ecol. 2011, 396, 177–184. [Google Scholar] [CrossRef]
- Gutowska, M.A.; Pörtner, H.O.; Melzner, F. Growth and calcification in the cephalopod Sepia officinalis under elevated seawater pCO2. Mar. Ecol. Prog. Ser. 2008, 373, 303–309. [Google Scholar] [CrossRef]
- Lacoue-Labarthe, T.; Martin, S.; Oberhänsli, F.; Teyssié, J.-L.; Markich, S.; Ross, J.; Bustamante, P. Effects of increased pCO2 and temperature on trace element (Ag, Cd and Zn) bioaccumulation in the eggs of the common cuttlefish, Sepia officinalis. Biogeosciences 2009, 6, 2561–2573. [Google Scholar] [CrossRef]
- Weiss, I.M.; Tuross, N.; Addadi, L.; Weiner, S. Mollusc larval shell formation: Amorphous calcium carbonate is a precursor phase for aragonite. J. Exp. Zool. 2002, 293, 478–491. [Google Scholar] [CrossRef]
- Wilt, F.H. Developmental biology meets materials science: Morphogenesis of biomineralized structures. Dev. Biol. 2005, 280, 15–25. [Google Scholar] [CrossRef]
- Zippay, M.L.; Hofmann, G.E. Effect of pH on gene expression and thermal tolerance of early life history stages of red abalone (Haliotis rufescens). J. Shellfish Res. 2010, 29, 429–439. [Google Scholar] [CrossRef]
- Sunday, J.M.; Crim, R.N.; Harley, C.D.G.; Hart, M.W. Quantifying rates of evolutionary adaptation in response to ocean acidification. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Orr, J.C.; Caldeira, K.; Fabry, V.; Gattuso, J.P.; Haugan, P.; Lehodey, P.; Pantoja, S.; Pörtner, H.O.; Riebesell, U.; Trull, T.; et al. Research priorities for ocean acidification. In Report from the Second Symposium on the Ocean in a High-CO2 World; SCOR, IOC-UNESCO, IAEA, and IGBP: Monaco, Monaco, 9 October 2008. [Google Scholar]
- Pörtner, H.-O. Ecosystem effects of ocean acidification in times of ocean warming: A physiologist’s view. Mar. Ecol. Prog. Ser. 2008, 373, 203–217. [Google Scholar] [CrossRef]
- Doney, S.C.; Fabry, V.J.; Feely, R.A.; Kleypas, J.A. Ocean acidification: The other CO2 problem. Annu. Rev. Mar. Sci. 2009, 1, 169–192. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Hammond, L.M.; Hofmann, G.E. Predicted impact of ocean acidification on a marine invertebrate: Elevated CO2 alters response to thermal stress in sea urchin larvae. Mar. Biol. 2009, 156, 439–446. [Google Scholar] [CrossRef]
- O’Donnell, M.J.; Todgham, A.E.; Sewell, M.A.; Hammond, L.M.; Ruggiero, K.; Fangue, N.A.; Zippay, M.L.; Hofmann, G.E. Ocean acidification alters skeletogenesis and gene expression in larval sea urchins. Mar. Ecol. Prog. Ser. 2009, 398, 157–171. [Google Scholar] [CrossRef]
- Todgham, A.E.; Hofmann, G.E. Transcriptomic response of sea urchin larvae Strongylocentrotus purpuratus to CO2-driven seawater acidification. J. Exp. Biol. 2009, 212, 2579–2594. [Google Scholar] [CrossRef]
- Stumpp, M.; Dupont, S.; Thorndyke, M.C.; Melzner, F. CO2 induced seawater acidification impacts sea urchin larval development II: Gene expression patterns in pluteus larvae. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2011, 160, 320–330. [Google Scholar] [CrossRef]
- Stumpp, M.; Wren, J.; Melzner, F.; Thorndyke, M.C.; Dupont, S.T. CO2 induced seawater acidification impacts sea urchin larval development I: Elevated metabolic rates decrease scope for growth and induce developmental delay. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2011, 160, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Deigweiher, K.; Hirse, T.; Bock, C.; Lucassen, M.; Pörtner, H.O. Hypercapnia induced shifts in gill energy budgets of Antarctic notothenioids. J. Comp. Physiol. B 2010, 180, 347–359. [Google Scholar] [CrossRef]
- Melzner, F.; Gutowska, M.A.; Langenbuch, M.; Dupont, S.; Lucassen, M.; Thorndyke, M.C.; Bleich, M.; Pörtner, H.-O. Physiological basis for high CO2 tolerance in marine ectothermic animals: Pre-adaptation through lifestyle and ontogeny? Biogeosciences 2009, 6, 2313–2331. [Google Scholar] [CrossRef]
- Kurihara, H.; Shimode, S.; Shirayama, Y. Effects of raised CO2 concentration on the egg production rate and early development of two marine copepods (Acartia steueri and Acartia erythraea). Mar. Pollut. Bull. 2004, 49, 721–727. [Google Scholar] [CrossRef]
- Kurihara, H.; Matsui, M.; Furukawa, H.; Hayashi, M.; Ishimatsu, A. Long-term effects of predicted future seawater CO2 conditions on the survival and growth of the marine shrimp Palaemon pacificus. J. Exp. Mar. Biol. Ecol. 2008, 367, 41–46. [Google Scholar] [CrossRef]
- Hauton, C.; Tyrrell, T.; Williams, J. The subtle effects of sea water acidification on the amphipod Gammarus locusta. Biogeosci. Discuss. 2009, 6, 919–946. [Google Scholar]
- Findlay, H.S.; Kendall, M.A.; Spicer, J.I.; Widdicombe, S. Relative influences of ocean acidification and temperature on intertidal barnacle post-larvae at the northern edge of their geographic distribution. Estuar. Coast. Shelf Sci. 2010, 86, 675–682. [Google Scholar] [CrossRef]
- Findlay, H.S.; Kendall, M.A.; Spicer, J.I.; Widdicombe, S. Post-larval development of two intertidal barnacles at elevated CO2 and temperature. Mar. Biol. 2010, 157, 725–735. [Google Scholar] [CrossRef]
- McDonald, M.R.; McClintock, J.B.; Amsler, C.D.; Rittschof, D.; Angus, R.A.; Orihuela, B.; Lutostanski, K. Effects of ocean acidification over the life history of the barnacle Amphibalanus amphitrite. Mar. Ecol. Prog. Ser. 2009, 385, 179–187. [Google Scholar] [CrossRef]
- Arnold, K.E.; Findlay, H.S.; Spicer, J.I.; Daniels, C.L.; Boothroyd, D. Effect of CO2-related acidification on aspects of the larval development of the European lobster, Homarus gammarus (L.). Biogeosciences 2009, 6, 1747–1754. [Google Scholar] [CrossRef]
- Walther, K.; Anger, K.; Pörtner, H.O. Effects of ocean acidification and warming on the larval development of the spider crab Hyas araneus from different latitudes (54° vs. 79°N). Mar. Ecol. Prog. Ser. 2010, 417, 159–170. [Google Scholar] [CrossRef]
- Egilsdottir, H.; Spicer, J.I.; Rundle, S.D. The effect of CO2 acidified sea water and reduced salinity on aspects of the embryonic development of the amphipod Echinogammarus marinus (Leach). Mar. Pollut. Bull. 2009, 58, 1187–1191. [Google Scholar] [CrossRef]
- Mayor, D.J.; Matthews, C.; Cook, K.; Zuur, A.F.; Hay, S. CO2-induced acidification affects hatching success in Calanus finmarchicus. Mar. Ecol. Prog. Ser. 2007, 350, 91–97. [Google Scholar] [CrossRef]
- Findlay, H.S.; Kendall, M.A.; Spicer, J.I.; Widdicombe, S. Future high CO2 in the intertidal may compromise adult barnacle Semibalanus balanoides survival and embryonic development rate. Mar. Ecol. Prog. Ser. 2009, 389, 193–202. [Google Scholar] [CrossRef]
- Walther, K.; Sartoris, F.J.; Pörtner, H.O. Impacts of temperature and acidification on larval calcium incorporation of the spider crab Hyas araneus from different latitudes (54° vs. 79°N). Mar. Biol. 2011, 158, 2043–2053. [Google Scholar] [CrossRef]
- Suwa, R.; Nakamura, M.; Morita, M.; Shimada, K.; Iguchi, A.; Sakai, K.; Suzuki, A. Effects of acidified seawater on early life stages of scleractinian corals (Genus Acropora). Fish. Sci. 2010, 76, 93–99. [Google Scholar] [CrossRef]
- Anlauf, H.; D’Croz, L.; O’Dea, A. A corrosive concoction: The combined effects of ocean warming and acidification on the early growth of a stony coral are multiplicative. J. Exp. Mar. Biol. Ecol. 2011, 397, 13–20. [Google Scholar] [CrossRef]
- Albright, R.; Langdon, C. Ocean acidification impacts multiple early life history processes of the Caribbean coral Porites astreoides. Glob. Change Biol. 2011, 17, 2478–2487. [Google Scholar] [CrossRef]
- Kikkawa, T.; Ishimatsu, A.; Kita, J. Acute CO2 tolerance during the early developmental stages of four marine teleosts. Environ. Toxicol. 2003, 18, 375–382. [Google Scholar] [CrossRef]
- Munday, P.L.; Crawley, N.E.; Nilsson, G.E. Interacting effects of elevated temperature and ocean acidification on the aerobic performance of coral reef fishes. Mar. Ecol. Prog. Ser. 2009, 388, 235–242. [Google Scholar] [CrossRef]
- Munday, P.L.; Dixson, D.L.; Donelson, J.M.; Jones, G.P.; Pratchett, M.S.; Devitsina, G.V.; Døving, K.B. Ocean acidification impairs olfactory discrimination and homing ability of a marine fish. Proc. Natl. Acad. Sci. USA 2009, 106, 1848–1852. [Google Scholar] [CrossRef]
- Munday, P.L.; Donelson, J.M.; Dixson, D.L.; Endo, G.G.K. Effects of ocean acidification on the early life history of a tropical marine fish. Proc. R. Soc. B 2009, 276, 3275–3283. [Google Scholar] [CrossRef]
- Munday, P.L.; Gagliano, M.; Donelson, J.M.; Dixson, D.L.; Thorrold, S.R. Ocean acidification does not affect the early life history development of a tropical marine fish. Mar. Ecol. Prog. Ser. 2011, 423, 211–221. [Google Scholar] [CrossRef]
- Munday, P.L.; Hernaman, V.; Dixson, D.L.; Thorrold, S.R. Effect of ocean acidification on otolith development in larvae of a tropical marine fish. Biogeosciences 2011, 8, 1631–1641. [Google Scholar] [CrossRef]
- Anger, K. The D0 threshold: A critical point in the larval development of decapod crustaceans. J. Exp. Mar. Biol. Ecol. 1987, 108, 15–30. [Google Scholar] [CrossRef]
- Hart, M.W.; Strathmann, R.R. Mechanisms and rates of suspension feeding. In Ecology of Marine Invertebrate Larvae; McEdward, L., Ed.; CRC Press: Boca Raton, FL, USA, 1995; pp. 193–222. [Google Scholar]
- Strathmann, M.F. Opisthobranchia. In Reproduction and Development of Marine Invertebrates of the Northern Pacific Coast; Strathmann, M.F., Ed.; University of Washington Press: Seattle, WA, USA, 1987; pp. 268–302. [Google Scholar]
- Cigliano, M.; Gambi, M.C.; Rodolfo-Metalpa, R.; Patti, F.P.; Hall-Spencer, J.M. Effects of ocean acidification on invertebrate settlement at volcanic CO2 vents. Mar. Biol. 2010, 157, 2489–2502. [Google Scholar] [CrossRef]
- Hall-Spencer, J.M.; Rodolfo-Metalpa, R.; Martin, S.; Ransome, E.; Fine, M.; Turner, S.M.; Rowley, S.J.; Tedesco, D.; Buia, M.-C. Volcanic carbon dioxide vents show ecosystem effects of ocean acidification. Nature 2008, 454, 96–99. [Google Scholar] [CrossRef]
- Fabricius, K.E.; Langdon, C.; Uthicke, S.; Humphrey, C.; Noonan, S.; De’ath, G.; Okazaki, R.; Muehllehner, N.; Glas, M.S.; Lough, J.M. Losers and winners in coral reefs acclimatized to elevated carbon dioxide concentrations. Nat. Clim. Change 2011, 1, 165–169. [Google Scholar] [CrossRef]
- Reynaud, S.; Leclercq, N.; Romaine-Lioud, S.; Ferrier-Pagès, C.; Jaubert, J.; Gattuso, J.-P. Interacting effects of CO2 partial pressure and temperature on photosynthesis and calcification in a scleractinian coral. Glob. Change Biol. 2003, 9, 1660–1668. [Google Scholar] [CrossRef]
- Metzger, R.; Sartoris, F.J.; Langenbuch, M.; Pörtner, H.O. Influence of elevated CO2 concentrations on thermal tolerance of the edible crab Cancer pagurus. J. Therm. Biol. 2007, 32, 144–151. [Google Scholar] [CrossRef]
- Anthony, K.R.N.; Kline, D.I.; Diaz-Pulido, G.; Dove, S.; Hoegh-Guldberg, O. Ocean acidification causes bleaching and productivity loss in coral reef builders. Proc. Natl. Acad. Sci. USA 2008, 105, 17442–17446. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.; Gattuso, J.-P. Response of Mediterranean coralline algae to ocean acidification and elevated temperature. Glob. Change Biol. 2009, 15, 2089–2100. [Google Scholar] [CrossRef]
- Bibby, R.; Widdicombe, S.; Parry, H.; Spicer, J.; Pipe, R. Effects of ocean acidification on the immune response of the blue mussel Mytilus edulis. Aquat. Biol. 2008, 2, 67–74. [Google Scholar] [CrossRef]
- Connell, J.H. Some mechanisms producing structure in natural communities: A model and evidence from field experiments. In Ecology and Evolution of Communities; Cody, M., Diamond, J., Eds.; Harvard University Press: Cambridge, MA, USA, 1975; pp. 460–490. [Google Scholar]
- Underwood, A.J. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1997; p. 504. [Google Scholar]
- Byrne, M.; Ho, M.; Wong, E.; Soars, N.A.; Selvakumaraswamy, P.; Shepard Brennand, H.; Dworjanyn, S.A.; Davis, A.R. Unshelled abalone and corrupted urchins: Development of marine calcifiers in a changing ocean. Proc. R. Soc. B 2011, 278, 2376–2383. [Google Scholar] [CrossRef]
- Martin, S.; Richier, S.; Pedrotti, M.-L.; Dupont, S.; Castejon, C.; Gerakis, Y.; Kerros, M.-E.; Oberhänsli, F.; Teyssié, J.-L.; Jeffree, R.; et al. Early development and molecular plasticity in the Mediterranean sea urchin Paracentrotus lividus exposed to CO2-driven acidification. J. Exp. Biol. 2011, 214, 1357–1368. [Google Scholar] [CrossRef]
- Yu, P.C.; Matson, P.G.; Martz, T.R.; Hofmann, G.E. The ocean acidification seascape and its relationship to the performance of calcifying marine invertebrates: Laboratory experiments on the development of urchin larvae framed by environmentally-relevant pCO2/pH. J. Exp. Mar. Biol. Ecol. 2011, 400, 288–295. [Google Scholar] [CrossRef]
- Crim, R.N.; Sunday, J.M.; Harley, C.D.G. Elevated seawater CO2 concentrations impair larval development and reduce larval survival in endangered northern abalone (Haliotis kamtschatkana). J. Exp. Mar. Biol. Ecol. 2011, 400, 272–277. [Google Scholar] [CrossRef]
- Miller, A.W.; Reynolds, A.C.; Sobrino, C.; Riedel, G.F. Shellfish face uncertain future in high CO2 world: Influence of acidification on oyster larvae calcification and growth in estuaries. PLoS One 2009, 4. [Google Scholar] [CrossRef]
- Jokiel, P.L.; Rodgers, K.S.; Kuffner, I.B.; Andersson, A.J.; Cox, E.F.; Mackenzie, F.T. Ocean acidification and calcifying reef organisms: A mesocosm investigation. Coral Reefs 2008, 27, 473–483. [Google Scholar] [CrossRef]
- Morita, M.; Suwa, R.; Iguchi, A.; Nakamura, M.; Shimada, K.; Sakai, K.; Suzuki, A. Ocean acidification reduces sperm flagellar motility in broadcast spawning reef invertebrates. Zygote 2010, 18, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Holcomb, M.; McCorkle, D.C.; Cohen, A.L. Long-term effects of nutrient and CO2 enrichment on the temperate coral Astrangia poculata (Ellis and Solander, 1786). 2010, 386, 27–33. [Google Scholar] [CrossRef]
- Krief, S.; Hendy, E.J.; Fine, M.; Yam, R.; Meibom, A.; Foster, G.L.; Shemesh, A. Physiological and isotopic responses of scleractinian corals to ocean acidification. 2010, 74, 4988–5001. [Google Scholar] [CrossRef]
- Rodolfo-Metalpa, R.; Martin, S.; Ferrier-Pagès, C.; Gattuso, J.P. Response of the temperate coral Cladocora caespitosa to mid- and long-term exposure to pCO2 and temperature levels projected for the year 2100 AD. Biogeosciences 2010, 7, 289–300. [Google Scholar] [CrossRef]
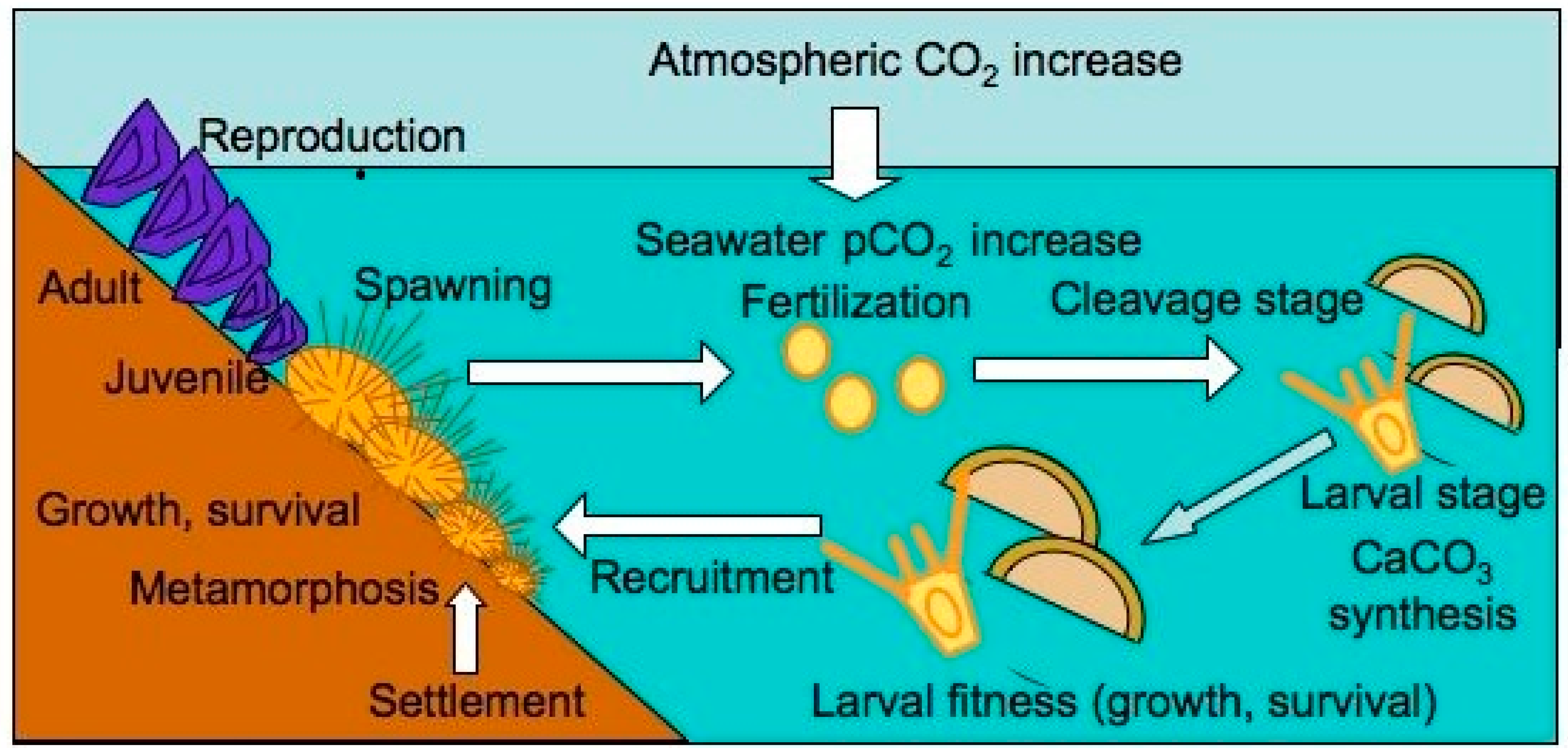
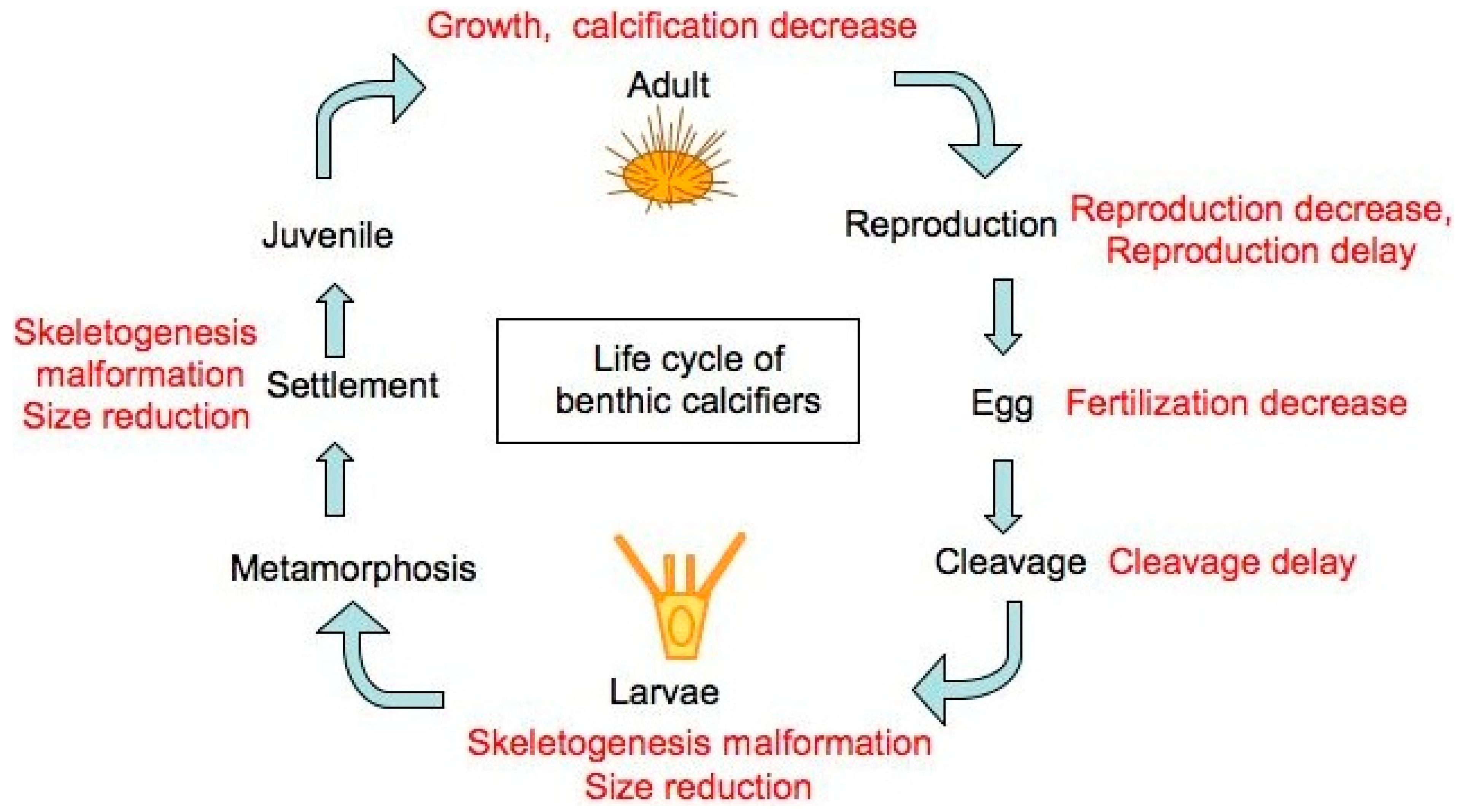
| Organism | Skeleton type * | Mineralogy |
|---|---|---|
| Echinoderms | Endoskeleton | ACC, High-Magnesium Calcite |
| Molluscs | Exoskeleton | ACC, Aragonite, Calcite ^, High- or Low-Magnesium Calcite |
| Crustaceans | Exoskeleton | ACC, High-Magnesium Calcite |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2011 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ross, P.M.; Parker, L.; O’Connor, W.A.; Bailey, E.A. The Impact of Ocean Acidification on Reproduction, Early Development and Settlement of Marine Organisms. Water 2011, 3, 1005-1030. https://doi.org/10.3390/w3041005
Ross PM, Parker L, O’Connor WA, Bailey EA. The Impact of Ocean Acidification on Reproduction, Early Development and Settlement of Marine Organisms. Water. 2011; 3(4):1005-1030. https://doi.org/10.3390/w3041005
Chicago/Turabian StyleRoss, Pauline M., Laura Parker, Wayne A. O’Connor, and Elizabeth A. Bailey. 2011. "The Impact of Ocean Acidification on Reproduction, Early Development and Settlement of Marine Organisms" Water 3, no. 4: 1005-1030. https://doi.org/10.3390/w3041005
APA StyleRoss, P. M., Parker, L., O’Connor, W. A., & Bailey, E. A. (2011). The Impact of Ocean Acidification on Reproduction, Early Development and Settlement of Marine Organisms. Water, 3(4), 1005-1030. https://doi.org/10.3390/w3041005




