Effects of Freeze–Thaw Cycles on Soil Nitrogen Transformation in Improved Saline Soils from an Irrigated Area in Northeast China
Abstract
1. Introduction
2. Materials and Methods
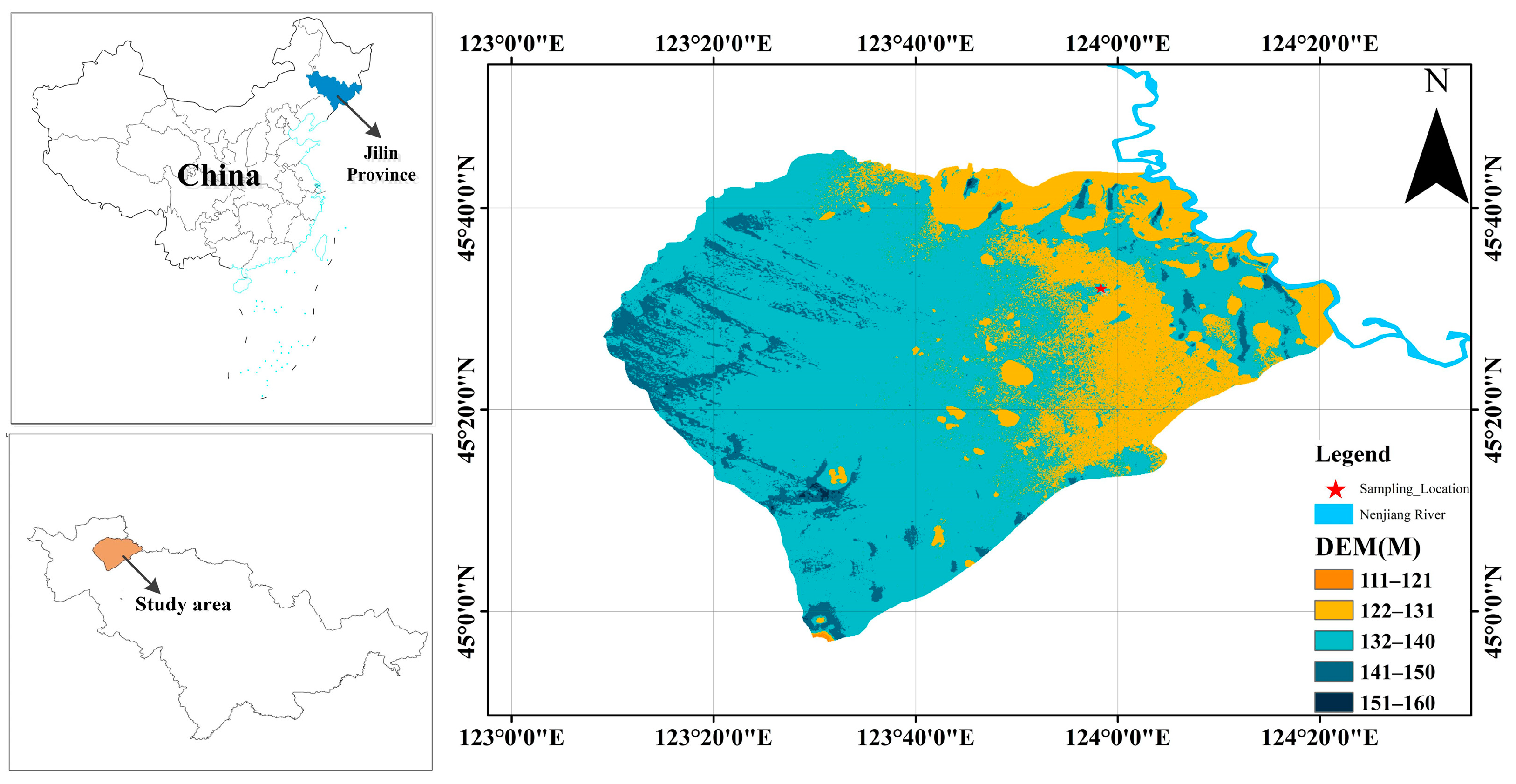
2.1. Site Description and Soil Sampling
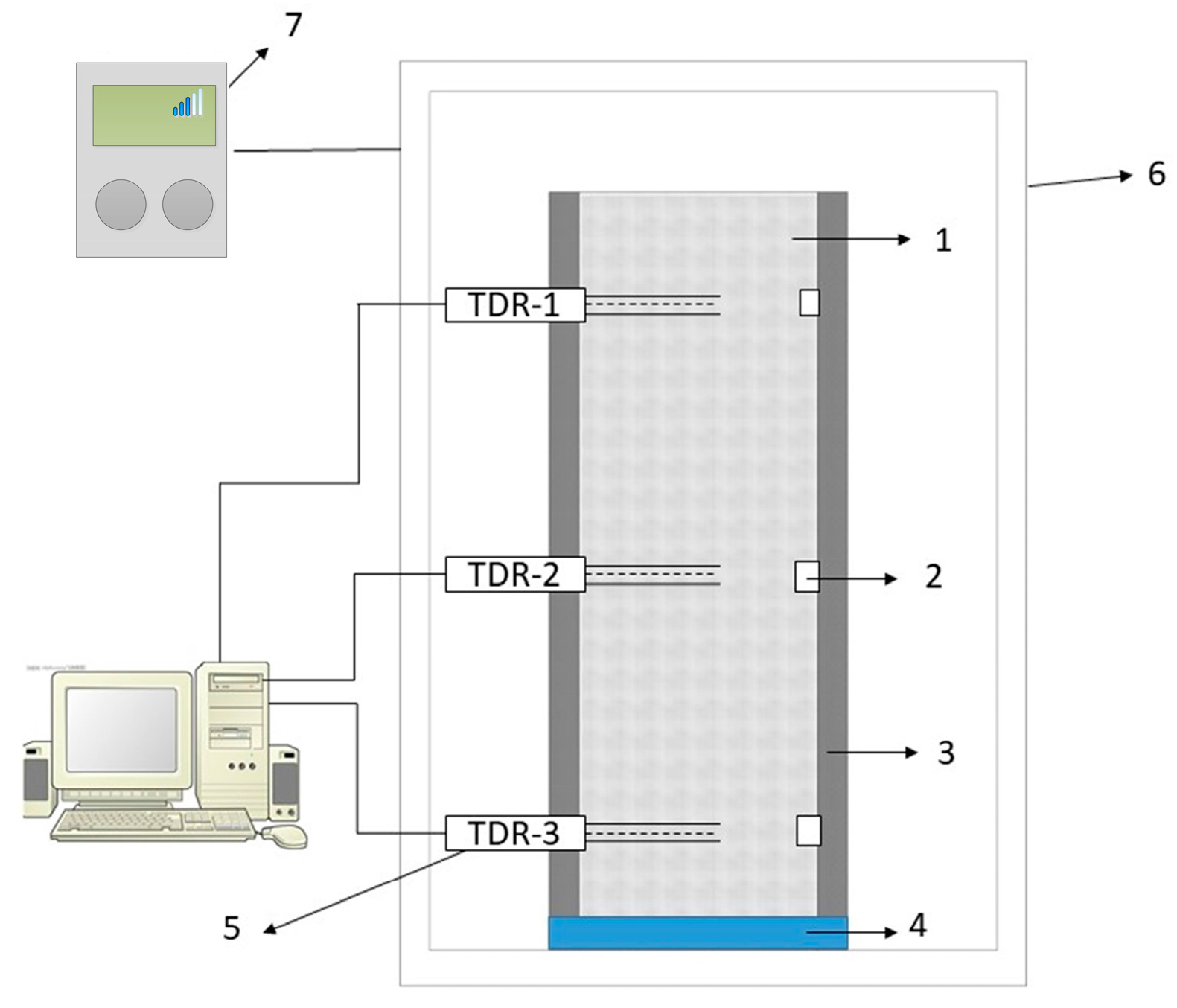
2.2. Test Device
2.3. Experimental Design
2.4. Analytical Methods and Statistical Analyses
3. Results
3.1. Soil Physical Properties
3.1.1. Soil Moisture Characteristics
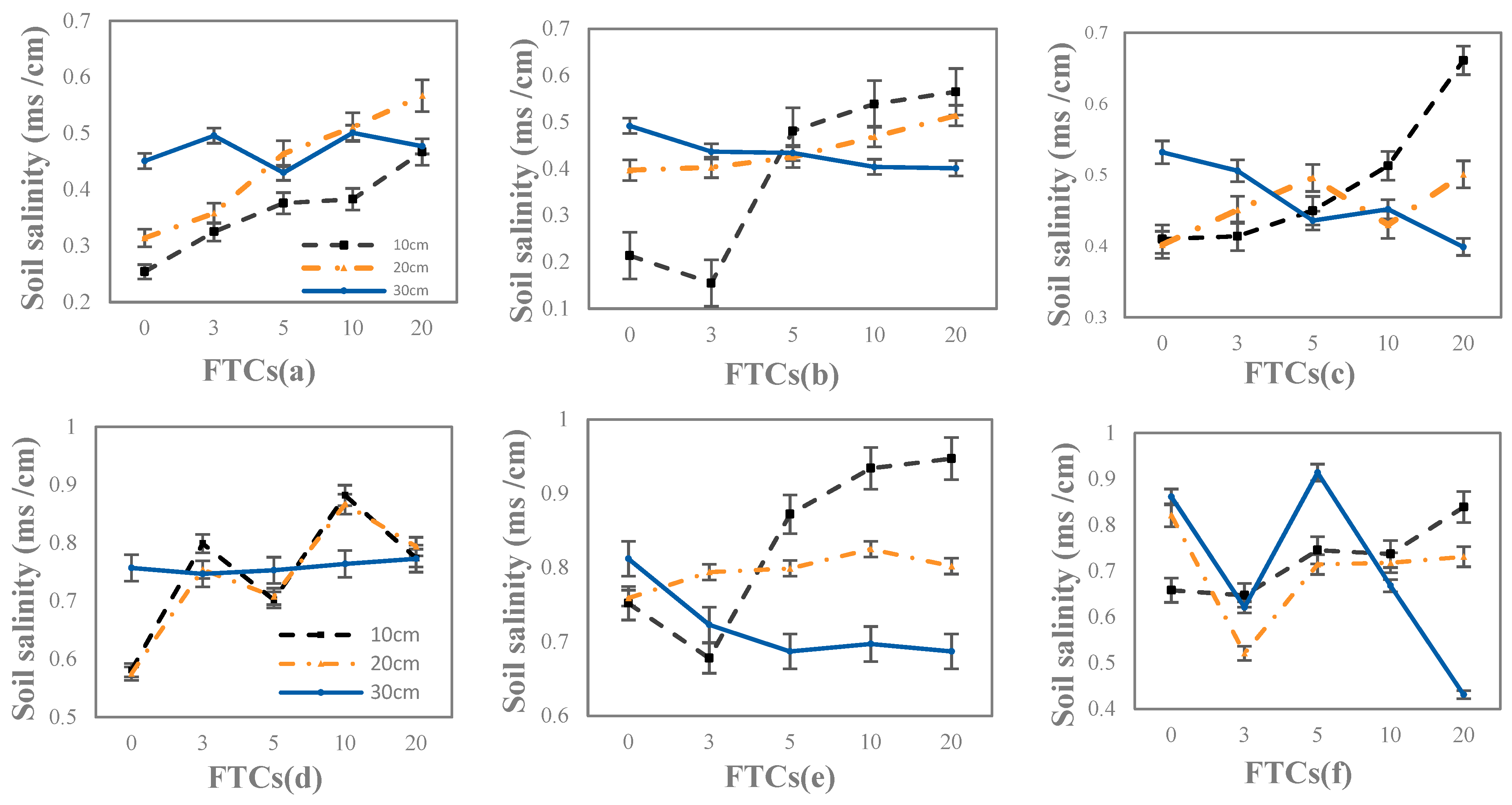
3.1.2. Soil Salinity Characteristics
3.2. Responses of the Nitrogen to FTCs
3.2.1. NH4+-N Content
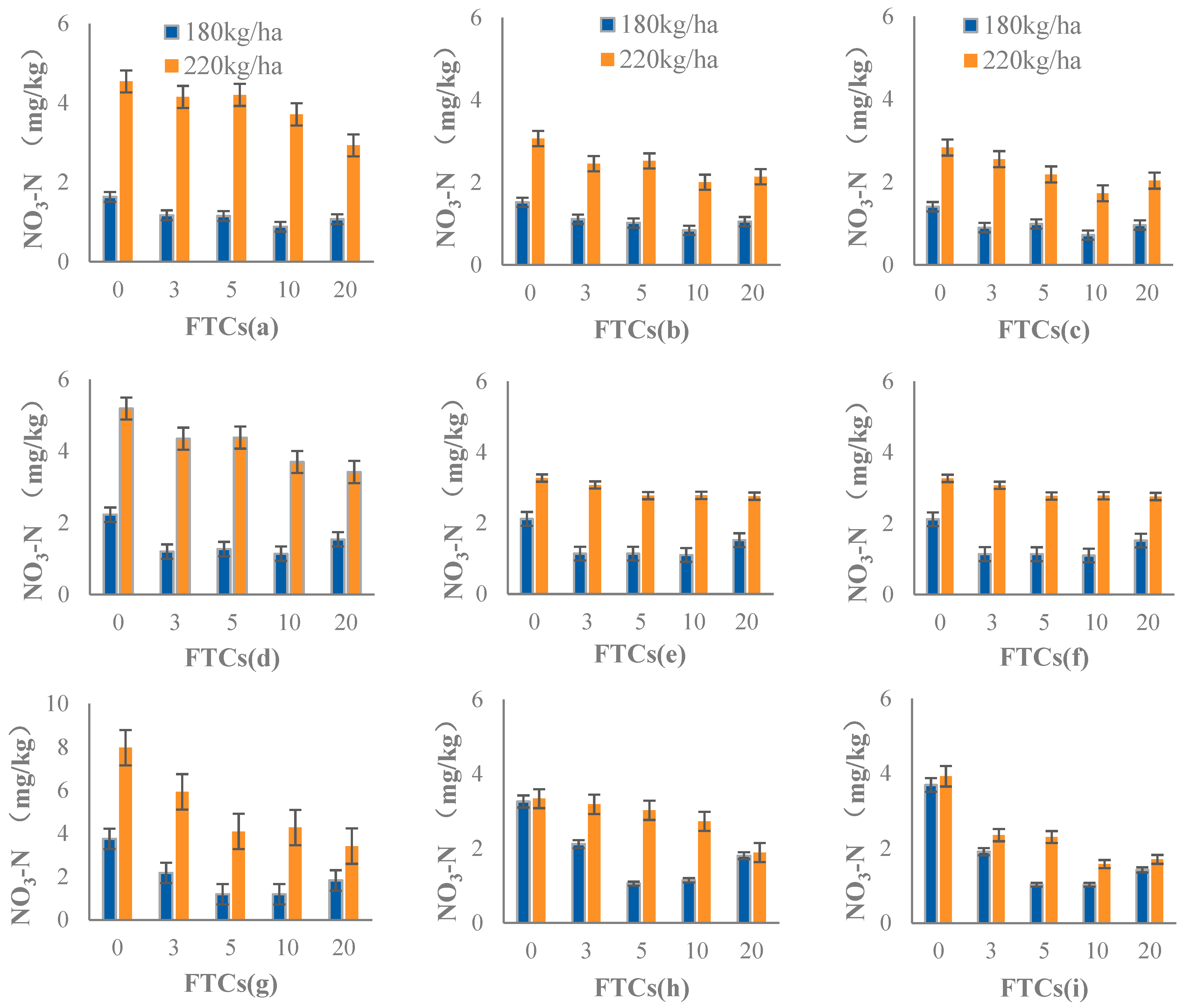
3.2.2. NO3−-N Content
3.3. Variation in the Characteristics of Nitrogen Mineralization under the Influence of FTCs
4. Discussion
4.1. Effect of FTCs on the N Content
4.2. Effects of Soil Salinity and Exogenous Nitrogen on Nitrogen
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, L.; Zhang, A.; Wang, Q.; Han, Y.; Li, K.; Gao, X.; Tang, Z. Corrosion resistance of wollastonite modified magnesium phosphate cement paste exposed to freeze-thaw cycles and acid-base corrosion. Case Stud. Constr. Mat. 2020, 13, e00421. [Google Scholar] [CrossRef]
- Hou, R.; Li, T.; Fu, Q.; Liu, D.; Li, M.; Zhou, Z.; Li, Q.; Zhao, H.; Yu, P.; Yan, J. Effects of biochar and straw on greenhouse gas emission and its response mechanism in seasonally frozen farmland ecosystems. Catena 2020, 194, 104735. [Google Scholar] [CrossRef]
- Aanderud, Z.T.; Jones, S.E.; Schoolmaster, D.R., Jr.; Fierer, N.; Lennon, J.T. Sensitivity of soil respiration and microbial communities to altered snowfall. Soil Biol. Biochem. 2013, 57, 217–227. [Google Scholar] [CrossRef]
- He, X.; Chi, Q.; Zhao, C.; Cheng, Y.; Huang, X.; Zhao, J.; Cai, Z.; Zhang, J.; Müller, C. Plants with an ammonium preference affect soil N transformations to optimize their N acquisition. Soil Biol. Biochem. 2021, 155, 108158. [Google Scholar] [CrossRef]
- Wan, H.; Bian, J.; Zhang, H.; Li, Y. Assessment of future climate change impacts on water-heat-salt migration in unsaturated frozen soil using CoupModel.Front. Environ. Sci. Eng. 2020, 15, 1–10. [Google Scholar]
- Harrison, J.L.; Schultz, K.; Blagden, M.; Sanders-DeMott, R.; Templer, P.H. Growing season soil warming may counteract trend of nitrogen oligotrophication in a northern hardwood forest. Biogeochemistry 2020, 151, 139–152. [Google Scholar] [CrossRef]
- Kreyling, J.; Schumann, R.; Weigel, R. Soils from cold and snowy temperate deciduous forests release more nitrogen and phosphorus after soil freeze-thaw cycles than soils from warmer, snow-poor conditions. Biogeosciences 2020, 17, 4103–4117. [Google Scholar] [CrossRef]
- Jiang, N.; Juan, Y.; Tian, L.; Chen, X.; Sun, W.; Chen, L. Modification of the composition of dissolved nitrogen forms, nitrogen transformation processes, and diversity of bacterial communities by freeze–thaw events in temperate soils. Pedobiologia 2018, 71, 41–49. [Google Scholar] [CrossRef]
- Rathore, V.S.; Nathawat, N.S.; Bhardwaj, S.; Yadav, B.M.; Kumar, M.; Santra, P.; Kumar, P.; Reager, M.L.; Yadava, N.D. Optimization of deficit irrigation and nitrogen fertilizer management for peanut production in an arid region. Sci. Rep. 2021, 11, 5456. [Google Scholar] [CrossRef]
- Zheng, J.; Fan, J.; Zhang, F.; Guo, J.; Yan, S.; Zhuang, Q.; Cui, N.; Guo, L. Interactive effects of mulching practice and nitrogen rate on grain yield, water productivity, fertilizer use efficiency and greenhouse gas emissions of rainfed summer maize in northwest China. Agric. Water Manag. 2021, 248, 106778. [Google Scholar] [CrossRef]
- Hao, D.-C.; Su, X.-Y.; Xie, H.-T.; Bao, X.-L.; Zhang, X.-D.; Wang, L.-F. Effects of tillage patterns and stover mulching on N2O production, nitrogen cycling genes and microbial dynamics in black soil. Environ. Manag. 2023, 345, 118458. [Google Scholar] [CrossRef]
- Yang, X.; Hou, R.; Fu, Q.; Li, T.; Wang, J.; Su, Z.; Shen, W.; Zhou, W.; Wang, Y. Effect of freeze-thaw cycles and biochar coupling on the soil water-soil environment, nitrogen adsorption and N2O emissions in seasonally frozen regions. Sci. Total Environ. 2023, 893, 164845. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Tateno, R.; Imada, S.; Fukuzawa, K.; Isobe, K.; Urakawa, R.; Oda, T.; Hosokawa, N.; Sasai, T.; Inagaki, Y.; et al. The effect of a freeze-thaw cycle on dissolved nitrogen dynamics and its relation to dissolved organic matter and soil microbial biomass in the soil of a northern hardwood forest. Biogeochemistry 2019, 142, 319–338. [Google Scholar] [CrossRef]
- Song, Y.; Zou, Y.; Wang, G.; Yu, X. Stimulation of nitrogen turnover due to nutrients release from aggregates affected by freeze-thaw in wetland soils. Phys. Chem. Earth. 2017, 97, 3–11. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, W.; Feng, W.; Xiao, D.; Hou, X. Reconstruction of Soil Particle Composition During Freeze-Thaw Cycling: A. Review. Pedosphere 2016, 26, 167–179. [Google Scholar] [CrossRef]
- Jackson-Blake, L.; Helliwell, R.C.; Britton, A.J.; Gibbs, S.; Coull, M.C.; Dawson, L. Controls on soil solution nitrogen along an altitudinal gradient in the Scottish uplands. Sci. Total Environ. 2012, 431, 100–108. [Google Scholar] [CrossRef]
- Sorensen, P.O.; Templer, P.H.; Christenson, L.; Duran, J.; Fahey, T.; Fisk, M.C.; Groffman, P.M.; Morse, J.L.; Finzi, A.C. Reduced snow cover alters root-microbe interactions and decreases nitrification rates in a northern hardwood forest. Ecology 2016, 97, 3359–3368. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Chen, Y.; Li, T.; Wang, C.; Wang, D.; Fu, B.; Lv, Y.; Wu, X. Grazing Reduces the Soil-Atmosphere Exchange of Greenhouse Gases During Freeze-Thaw Cycles in Meadow Steppes in Inner Mongolia. Front. Ecol. Evol. 2021, 9, 795203. [Google Scholar] [CrossRef]
- Fu, Q.; Yan, J.; Li, H.; Li, T.; Hou, R.; Liu, D.; Ji, Y. Effects of biochar amendment on nitrogen mineralization in black soil with different moisture contents under freeze-thaw cycles. Geoderma 2019, 353, 459–467. [Google Scholar] [CrossRef]
- Zhang, L.; Ren, F.; Li, H.; Cheng, D.; Sun, B. The Influence Mechanism of Freeze-Thaw on Soil Erosion: A Review. Water 2021, 13, 1010. [Google Scholar] [CrossRef]
- Reinmann, A.B.; Templer, P.H. Reduced Winter Snowpack and Greater Soil Frost Reduce Live Root Biomass and Stimulate Radial Growth and Stem Respiration of Red Maple Trees in a Mixed-Hardwood Forest. Ecosystems 2016, 19, 129–141. [Google Scholar] [CrossRef]
- Herrmann, A.; Witter, E. Sources of C and N contributing to the flush in mineralization upon freeze-thaw cycles in soils. Soil Biol. Biochem. 2002, 34, 1495–1505. [Google Scholar] [CrossRef]
- Urakawa, R.; Shibata, H.; Kuroiwa, M.; Inagaki, Y.; Tateno, R.; Hishi, T.; Fukuzawa, K.; Hirai, K.; Toda, H.; Oyanagi, N.; et al. Effects of freeze–thaw cycles resulting from winter climate change on soil nitrogen cycling in ten temperate forest ecosystems throughout the Japanese archipelago. Soil Biol. Biochem. 2014, 74, 82–94. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, W.; Wang, D.; Heenan, M.; Xu, Z. Short-term responses of soil nitrogen mineralization, nitrification and denitrification to prescribed burning in a suburban forest ecosystem of subtropical Australia. Sci. Total Environ. 2018, 642, 879–886. [Google Scholar] [CrossRef]
- Li, S.; Du, Y.; Guo, P.; Guo, L.; Qu, K.; He, J. Effects of different types of N deposition on the fungal decomposition activities of temperate forest soils. Sci. Total Environ. 2014, 497, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Juan, Y.; Tian, L.; Sun, W.; Qiu, W.; Curtin, D.; Gong, L.; Liu, Y. Simulation of soil freezing-thawing cycles under typical winter conditions: Implications for nitrogen mineralization. J. Soil Sediment. 2020, 20, 143–152. [Google Scholar] [CrossRef]
- Sharma, S.; Bhatt, R. Monitoring Water and Soil Salinity across Mentor Marsh to Investigate Their Temporal and Spatial Variability. Water 2023, 15, 4172. [Google Scholar] [CrossRef]
- Nie, E.; Zhang, X.; Zheng, G.D.; Wang, H.M.; Chen, F.S.; Sun, X.M. Effects of nitrogen and phosphorus additions on soil organic carbon and nitrogen mineralization and hydrolase kinelies in Chinese fir plantations. Acta Ecol. Sin. 2018, 38, 615–623. [Google Scholar]
- Hou, R.; Li, T.; Fu, Q.; Liu, D.; Cui, S.; Zhou, Z.; Yan, P.; Yan, J. Effect of snow-straw collocation on the complexity of soil water and heat variation in the Songnen Plain, China. Catena 2019, 172, 190–202. [Google Scholar] [CrossRef]
- Xu, W.; Zhao, D.; Ma, Y.; Yang, G.; Ambus, P.L.; Liu, X.; Luo, J. Effects of long-term organic fertilizer substitutions on soil nitrous oxide emissions and nitrogen cycling gene abundance in a greenhouse vegetable field. Appl. Soil Ecol. 2023, 188, 104877. [Google Scholar] [CrossRef]
- He, X.; Dai, S.; Meng, L.; He, M.; Wang, X.; Cai, Z.; Zhu, B.; Zhang, J.; Nardi, P.; Müller, C. Effects of 18 years repeated N fertilizer applications on gross N transformation rates in a subtropical rain-fed purple soil. Appl. Soil Ecol. 2023, 189, 104952. [Google Scholar]
- Payandi-Rolland, D.; Shirokova, L.S.; Labonne, F.; Bénézeth, P.; Pokrovsky, O.S. Impact of freeze-thaw cycles on organic carbon and metals in waters of permafrost peatlands. Chemosphere 2021, 279, 130510. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Chua-Ona, T.; Thompson, M.L. Short-term Nitrogen Mineralization Potential in Soils of Biofuel Cropping Systems. Soil Sci. 2016, 181, 503–512. [Google Scholar] [CrossRef]
- Sulkava, P.; Huhta, V. Effects of hard frost and freeze-thaw cycles on decomposer communities and N mineralisation in boreal forest soil. Appl. Soil Ecol. 2003, 22, 225–239. [Google Scholar] [CrossRef]
- Hu, R.; Wang, X.; Xu, J.; Zhang, Y.; Pan, Y.; Su, X. Themechanism of soil nitrogen transformation under different biocrusts to warming and reduced precipitation: From microbial functional genes to enzyme activity. Sci. Total Environ. 2020, 722, 137849. [Google Scholar] [CrossRef] [PubMed]
- Maenhout, P.; van den Bulcke, J.; van Hoorebeke, L.; Cnudde, V.; De Neve, S.; Sleutel, S. Nitrogen Limitations on Microbial Degradation of Plant Substrates Are Controlled by Soil Structure and Moisture Content. Front. Microblol. 2018, 9, 01433. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Xu, J.; Tang, C.; Zhang, J.; Brookes, P.C. Differences in carbon and nitrogen mineralization in soils of differing initial pH induced by electrokinesis and receiving crop residue amendments. Soil Biol. Biochem. 2013, 67, 70–84. [Google Scholar] [CrossRef]
- Zuo, Y.; Meng, F.; Li, T.; Fu, Q.; Liu, D.; Hou, R.; Li, Q.; Li, M.J.G. Effect of biochar application on freezing-thawing deformation of farmland soil during freeze–thaw cycling. Geoderma 2022, 405, 115510. [Google Scholar] [CrossRef]
- Wang, J.; Song, C.; Hou, A.; Wang, L. CO2 emissions from soils of different depths of a permafrost peatland, Northeast China: Response to simulated freezing-thawing cycles. J. Plant Nutr. Soil Sci. 2014, 177, 524–531. [Google Scholar] [CrossRef]
- Clein, J.S.; Schimel, J.P. Microbial activity of tundra and taiga soils at sub-zero temperatures. Soil Biol. Biochem. 1995, 27, 1231–1234. [Google Scholar] [CrossRef]
- Cookson, W.R.; Cornforth, I.S.; Rowarth, J.S. Winter soil temperature (2–15 degrees C) effects on nitrogen transformations in clover green manure amended or unamended soils; a laboratory and field study. Soil Biol. Biochem. 2002, 34, 1401–1415. [Google Scholar] [CrossRef]
- Kang, F.; Meng, Y.; Ge, Y.; Zhang, Y.; Gao, H.; Ren, X.; Wang, J.; Hu, S. Calcium-based polymers for suppression of soil acidification by improving acid-buffering capacity and inhibiting nitrification. J. Environ. Sci. 2024, 139, 138–149. [Google Scholar] [CrossRef] [PubMed]



| Nitrogen Fertilization (kg/ha) | Soil Layer (cm) | Total Nitrogen (%) | pH | Bulk Density (g/cm3) | Soil Salinity (g/kg) | Mechanical Composition (%) | ||
|---|---|---|---|---|---|---|---|---|
| Clay | Silt | Sand | ||||||
| 180 | 0–10 | 0.075 | 8.8 | 1.29 | 1.76 | 38.8 | 30.3 | 30.9 |
| 10–20 | 0.081 | 8.8 | 1.26 | 1.71 | 39.7 | 28.2 | 32.1 | |
| 20–30 | 0.087 | 8.9 | 1.32 | 1.83 | 38.9 | 26.7 | 34.4 | |
| 220 | 0–10 | 0.131 | 8.9 | 1.34 | 1.82 | 38.6 | 23.7 | 37.7 |
| 10–20 | 0.152 | 8.9 | 1.35 | 1.94 | 40.4 | 20.2 | 39.4 | |
| 20–30 | 0.164 | 9.1 | 1.55 | 2.12 | 43.7 | 20.5 | 35.8 | |
| Source | NH4+-N | NO3−-N | SNMR | EC | ||||
|---|---|---|---|---|---|---|---|---|
| F-Value | Sig. | F-Value | Sig. | F-Value | Sig. | F-Value | Sig. | |
| FTCs | 80.15 | 0.000 | 3.876 | 0.006 | 20.30 | 0.000 | 0.814 | 0.520 |
| Moisture content | 0.036 | 0.964 | 6.718 | 0.002 | 0.013 | 0.987 | 0.069 | 0.933 |
| Nitrogen fertilizer | 1.330 | 0.252 | 69.21 | 0.000 | 7.420 | 0.008 | 225.27 | 0.000 |
| Soil layers | 0.169 | 0.845 | 2.405 | 0.096 | 0.388 | 0.679 | 0.006 | 0.994 |
| FTCs × Moisture content | 0.317 | 0.957 | 0.177 | 0.993 | 0.503 | 0.850 | 0.310 | 0.960 |
| FTCs × Nitrogen fertilizer | 13.25 | 0.000 | 0.793 | 0.533 | 3.288 | 0.015 | 0.705 | 0.591 |
| FTCs × Soil layers | 0.379 | 0.928 | 0.515 | 0.841 | 4.096 | 0.000 | 0.846 | 0.565 |
| Moisture content × Nitrogen fertilizer | 0.051 | 0.950 | 11.97 | 0.000 | 0.176 | 0.839 | 3.314 | 0.041 |
| Moisture content × Soil layers | 0.009 | 1.000 | 0.188 | 0.944 | 0.456 | 0.768 | 0.230 | 0.921 |
| Soil layers × Nitrogen fertilizer | 0.566 | 0.570 | 0.101 | 0.904 | 1.265 | 0.288 | 1.506 | 0.228 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, S.; Jia, X.; Zou, Y.; Bian, J. Effects of Freeze–Thaw Cycles on Soil Nitrogen Transformation in Improved Saline Soils from an Irrigated Area in Northeast China. Water 2024, 16, 653. https://doi.org/10.3390/w16050653
Nie S, Jia X, Zou Y, Bian J. Effects of Freeze–Thaw Cycles on Soil Nitrogen Transformation in Improved Saline Soils from an Irrigated Area in Northeast China. Water. 2024; 16(5):653. https://doi.org/10.3390/w16050653
Chicago/Turabian StyleNie, Siyu, Xian Jia, Yuanchun Zou, and Jianmin Bian. 2024. "Effects of Freeze–Thaw Cycles on Soil Nitrogen Transformation in Improved Saline Soils from an Irrigated Area in Northeast China" Water 16, no. 5: 653. https://doi.org/10.3390/w16050653
APA StyleNie, S., Jia, X., Zou, Y., & Bian, J. (2024). Effects of Freeze–Thaw Cycles on Soil Nitrogen Transformation in Improved Saline Soils from an Irrigated Area in Northeast China. Water, 16(5), 653. https://doi.org/10.3390/w16050653









